Abstract
The present study determined whether gene transfer of human copper/zinc superoxide dismutase-1 (Cu/Zn SOD-1) prevented the autoregulatory impairment of CBF induced by subarachnoid hemorrhage (SAH). After application of recombinant adenovirus (100 μL of 1 × 1010 pfu/mL, intracisternally) encoding human Cu/Zn SOD-1 3 days before experiments, Cu/Zn SOD-1 activity significantly increased in association with increase in Cu/Zn SOD-1 mRNA and protein expression in the cerebral vasculature of both sham-operated and SAH rats as determined by reverse transcriptase-polymerase chain reaction, Western blotting, and immunohistochemistry, and SAH-induced increase in superoxide anion was markedly reduced in accordance with increased nitric oxide production. In line with these findings, rats that received human Cu/Zn SOD-1 therapy showed the prominent restoration of blunted vasodilation of the pial artery in response to calcitonin gene-related peptide and levcromakalim, and the recovery of impaired autoregulatory vasodilation in response to acute hypotension, thereby leading to significant restoration of CBF autoregulation. These results provide a rational basis for application of Cu/Zn SOD-1 gene therapy for protection of the impairment of autoregulatory CBF during the acute stage of SAH.
The alteration of cerebral microcirculation is an important cause of cerebral ischemia (Kassell et al., 1985). The autoregulatory disturbance of CBF has been documented in patients with subarachnoid hemorrhage (SAH) (Voldby et al., 1985; Yundt et al., 1998). The major derangements in cerebral vessels after SAH are reported to be the increased vasoconstriction (vasospasm) and decreased vasorelaxation. Reactive oxygen species including superoxide anion play an important role in regulating vascular tone and in intracellular signaling of the vessels. Superoxide anion inactivates nitric oxide (NO) in vivo, resulting in accentuation of vasoconstriction (Wei et al., 1985).
Marked membrane depolarization of cerebral vascular smooth muscles, however, was demonstrated from 30 minutes to 24 hours after SAH as a result of inhibition of potassium channels (Waters and Harder, 1985; Harder et al., 1987), and its importance was correlated with the production of endothelial superoxide production (Al-Mehdi et al., 1998). Superoxide dismutase has also been demonstrated to improve the outcome after traumatic brain injury in phase II clinical trials (Muizelaar et al., 1993). In the transgenic mice overexpressing copper, zinc-superoxide dismutase (Cu/Zn SOD), the vasospasm induced by SAH was ameliorated, indicative of a deleterious role of the oxidative stress and the importance of SOD in the pathogenesis of vasospasm after SAH (Kamii et al., 1999). Even though SOD has been reported to be effective in preventing the cerebrovascular dysfunction, its effectiveness is limited because the entry of exogenous SOD into the brain is normally limited owing to the blood-brain barrier and because native SOD has a short half-life of approximately 6 minutes in rat plasma (Odlind et al., 1988). Thus, it is not easy to maintain a therapeutic concentration of SOD in the targeted tissues after the bolus injection.
Recently, some reports have documented the perivascular adenoviral transfection of reporter gene encoding β-galactosidase to rabbit, mice and rat cerebral vasculatures (Weihl et al., 1999; Toyoda et al., 2000). Most recently, Kim et al. (2002) have shown that the intracisternal administration of recombinant adenovirus-mediated transfer of human Cu/Zn SOD-1 gene prevents the CBF autoregulatory impairment induced by fluid percussion injury. Furthermore, we observed that SAH rats treated with NG-nitro-L-arginine methyl ester (L-NAME) (10 mg/kg, intraperitoneally) resulted in a significant reduction of autoregulatory vasodilation at 24 hours in association with increased superoxide production, in that increased β-nicotinamide adenine dinucleotide (phosphate) reduced form [NAD(P)H] oxidase was associated with activated tyrosine phosphorylation-coupled increased expression of gp91phox and membrane translocation of Rac protein (Shin et al., 2002).
Based on these reports that acute elevation of intracellular superoxide anion after SAH results in the impairment of CBF autoregulation in rat cerebral vessels, it is hypothesized that effective gene transfer of Cu/Zn SOD-1 may prevent the impairment of CBF autoregulation in the pathologic field of SAH. To address this hypothesis, the present study was to prove the fact that intracisternal application of recombinant adenovirus encoding human Cu/Zn SOD-1 3 days before SAH prevents the impairment of vasodilation to hypotension and vasorelaxants, thereby resulting in restoration of CBF autoregulation.
MATERIALS AND METHODS
Preparation of animals
All animal studies carefully conformed to the guidelines outlined in the Guide for Animal Experiments edited by the Korean Academy of Medical Sciences.
Male Sprague-Dawley rats (250 to 300 g) were anesthetized with anesthesia cocktail composed of 1 mg/kg acetopromazine, 5 mg/kg xylazine, and 50 mg/kg ketamine, and placed in the supine position on the stereotactic frame (Stoelting, Wood Dale, IL, U.S.A.). The arterial catheter was inserted into the femoral artery for withdrawing and sampling of the arterial blood. Through the incised atlanto-occipital membrane, a 27-gauge needle was inserted into the cisterna magna and 0.3 mL CSF was aspirated and then 0.3 mL autologous whole blood was injected. For the sham-operated control group, 0.3 mL of normal saline was injected in the same manner. Free access to food and water was allowed after recovery from anesthesia.
Preparation of adenoviral vector and in vivo gene transfer
Replication-deficient recombinant adenoviral vector (sero-type 5, produced in HEK293 cells) driven by the cytomegalovirus immediate early promoter was used to transfer the genes to the cerebral vasculature. Adenoviral vectors containing human Cu/Zn SOD-1 (AdCMVSOD) and Escherichia coli β-galactosidase (AdCMVLacZ) genes were obtained from Dr. John Engelhardt (Gene Therapy Core Center, University of Iowa, Iowa, U.S.A.). The DNA constructs of replication-deficient adenovirus comprise almost a full-length copy of the adenoviral genome, in which Cu/Zn SOD-1 and LacZ expression cassette is incorporated at the site of E1 region deletions. For each vector, high-titer adenoviral vector stocks were prepared by double cesium gradient purification, and viral titer of plaque forming units (pfu/mL) was determined. Virus preparations were suspended in phosphate-buffered saline containing 3% sucrose and stored at −70°C until use.
To transfer the gene in vivo to the cerebral vasculature, rats were anesthetized with thiopental sodium (50 mg/kg, intraperitoneally) and a 27-gauge needle was aseptically inserted into the cisterna magna. An equal volume (100 μL) of CSF was removed before injection of viral suspension (AdCMVSOD and AdCMVLacZ, 100 μL of 1 × 1010 pfu/mL, each) to avoid an increase in intracranial pressure.
Expression of human copper, zinc-superoxide dismutase messenger RNAs
The expression of human Cu/Zn SOD-1 was determined by reverse transcription-polymerase chain reaction (RT-PCR). The cerebral vessels were homogenized in Trizol reagent (Gibco BRL; Life Technologies, Rockville, MD, U.S.A.), and chloroform was added to the lysates. After centrifugation, the upper layer was taken and subjected to isopropanol precipitation. Final total RNA concentration and purity were assessed by measurement of absorbance at 260 and 280 nm. The human Cu/Zn SOD-1 primers were composed of a 5‘ primer of TGT-GATCTCACTCTCAGGAGA with the 3‘ primers of TTA-CAGTGTTTAATGTTTATCAGGA. The expected size of the Cu/Zn SOD-1 PCR reaction was 248 bp. The conditions were 35 cycles of denaturation at 94°C (1 minute), annealing at 45°C (1 minute), and extension at 72°C (1 minute), followed by a 10-minute extension reaction at 72°C. Polymerase chain reaction products were resolved in a 1.5% agarose gel.
Western blotting for human copper, zinc-superoxide dismutase
The vessels isolated were homogenized at 4°C in 50 mmol/L Tris-HCl (pH 7.4), 1 mmol/L EDTA, and 1 mmol/L phenylmethylsulfonyl fluoride, and then centrifuged at 12,000 g for 10 minutes to remove insoluble material. Each sample (50 μg of total protein) was loaded into a sodium dodecyl sulfate—polyacrylamide gel electrophoresis gel, and transferred to nitrocellulose membrane (Amersham Biosciences, Piscataway, NJ, U.S.A.). The expression of human Cu/Zn SOD-1 protein was determined using specific monoclonal antibody against human CuZnSOD-1 (BD Biosciences, San Diego, CA, U.S.A.). The immunoreactive bands were visualized using chemiluminescent reagent of the Supersignal West Dura Extended Duration Substrate Kit (Pierce, Rockford, IL, U.S.A.). The signals of the bands were quantified using the Calibrated Imaging Densitometer (GS-710; Bio-Rad Laboratories, Hercules, CA, U.S.A.). The protein concentration of the lysate was determined using the Bio-Rad DC assay kit (Bio-Rad Laboratories).
Immunohistochemistry for copper, zinc-superoxide dismutase
Serial 5-μm-thick frozen sections of the pial artery were adhered to poly-L-lysine-coated slides, allowed to dry in room air, and fixed in acetone. After treatment with hydrogen peroxide (0.6%) and bovine serum albumin (2%), the preparations were incubated in the goat antirat Cu/Zn SOD-1 polyclonal antibody (Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.). After primary incubation, the slides were incubated with the biotinylated antigoat secondary antibody (Santa Cruz Biotechnology), and then they were incubated with avidin and biotinylated horseradish peroxidase complex (Vector Laboratories, Burlingame, CA, U.S.A.) for 1 hour. Vessel sections were counterstained with hematoxylin (purple) and examined for positive staining of Cu/Zn SOD-1 (gray—black) by light microscopy. Immunoreactivity for Cu/Zn SOD-1 was quantified by the Image-Pro Plus Imaging software (Media Cybernetics, Silver Spring, MD, U.S.A.) and the stain density was expressed as pixels per square micron tissue.
Measurement of vascular superoxide dismutase activity
After removal of brain samples, cerebral vessels—anterior, middle, and posterior cerebral arteries including basilar artery and pial microvessels—were immediately isolated and immersed in ice-cold physiologic salt solution (pH 7.4). Then, the samples were homogenized at 4°C in 50 mmol/L Tris-HCl (pH 7.4), 1 mmol/L EDTA and 1 mmol/L phenylmethylsulfonyl fluoride, and then centrifuged at 12,000 g for 10 minutes to remove insoluble material. Superoxide dismutase activity was assessed by the ability of the homogenate (10 or 20 μg total protein) to inhibit the lucigenin chemiluminescence induced by addition of 5 μmol/L lucigenin, 100 μmol/L hypoxanthine, and 0.025 U xanthine oxidase. In each experiment, a parallel determination was performed in the presence of 1 mmol/L KCN. The Cu/Zn SOD-1 was calculated as the activities when inhibited by KCN, whereas Mn SOD-2 was calculated as the activities not inhibited by KCN. Values obtained were expressed as units of SOD per milligram protein.
Measurement of superoxide anion
Superoxide production in cerebral vessels after SAH was measured as an index of NAD(P)H oxidase activity. Briefly, cerebral vessels isolated were placed in 200 μL of HEPES buffer containing lucigenin (bis-N-methylacridinium nitrate, 5 μmol/L) and placed into the luminometer (Microlumat LB96P; EG & G Berthold, Germany). After dark adaptation, NADH and NADPH (100 μmol/L, each) were added to the vial. Counts were then recorded every 30 seconds for 10 minutes, and the count of respective background was subtracted. Chemiluminescence was expressed as counts per second per milligram dry weight.
Nitrite/nitrate determination
Nitric oxide metabolites (nitrite/nitrate) in the CSF were determined by using Griess reagent (Promega, Madison, WI, U.S.A.). Briefly, CSF samples (100 μL) were drained from the cisterna magna. Fifty microliters of each sample was added to the wells with 50 μL of the sulfanilamide solution and allowed to react for 5 to 10 minutes at room temperature while protected from light. After adding 50 μL N−1-naphthylethylenediamine dihydrochloride solution 0.1% to all wells, total amounts of NO2/NO3 were determined by measuring absorbance at 540 nm by the Power Wave x340 (Bio-Tek Instrument).
Quantification of nitrotyrosine immunoreactivity
Cerebral vessels were quickly removed at 24 hours after SAH. Then, the samples were homogenized at 4°C in 50 mmol/L Tris-HCl (pH 7.4), 1 mmol/L EDTA, and 1 mmol/L phenylmethylsulfonyl fluoride, and then centrifuged at 12,000 g for 10 minutes to remove insoluble material. The supernatant was retained. Samples (10 μg protein each) were analyzed by dot blots on nitrocellulose membranes. The blocked membranes were then incubated with the antibody to nitrotyrosine (Upstate Biotechnology Lake Placid, NY, U.S.A.). The membranes were incubated overnight (4°C) with the first antibody at 1/1,000, thereafter washed, and then incubated with a secondary peroxidase-conjugated goat antirabbit antibody for 1 hour. Specific proteins were detected on X-ray films using enhanced chemiluminescence (Supersignal West Dura Extended Duration Substrate kit; Pierce). Quantification was performed with densitometric measurement using the Calibrated Imaging Densitometer (GS-710; Bio-Rad Laboratories).
Measurement of vessel diameter
Rats were anesthetized with urethane (1 g/kg intraperitoneally) and placed on a heating pad (Homeothermic Blanket System; Harvard Apparatus, South Natick, MA, U.S.A.) to maintain a constant rectal temperature (37 ± 0.5°C). After a tracheostomy, the animal was mechanically ventilated with room air by a respirator (model 683; Harvard Apparatus) after immobilization with 5-mg/kg gallamine triethiodide. The mean Paco2 was monitored with end-tidal CO2 analyzer (CapStar-100; IITC Life Science, Woodland Hills, CA, U.S.A.). Catheters were placed in a carotid artery for measurement of systemic arterial blood pressure (Statham P23D pressure transducer; Gould, Cleveland, OH, U.S.A.). The blood was collected before and after installation of a cranial window for blood gas and pH determination (i-STAT Portable Clinical Analyzer; Abbott Laboratories, East Windsor, NJ, U.S.A.).
The animal head was fixed in a prone position with a stereotaxic apparatus (Stoelting) and a square shape (5-mm x 5-mm) burr hole was made over the right parietal cortex. After resection of the dura with caution, pial microvessels were visualized through the cranial window, where prewarmed artificial CSF saturated with a mixed gas of 95% O2 and 5% CO2 was constantly suffused over the cortical surface at 0.3 mL/min. The image of pial microvessels was captured with a CCD videocamera (VDC 3900; Sanyo, Japan) through a stereomicroscope (model SMZ-2T; Nikon, Japan). It was fed to a television monitor for direct observation, and the caliber was measured using a Width analyzer (C3161; Hamamatsu Photonics, Hamamatsu, Japan). The intracranial pressure was maintained constant at 5 to 6 mm Hg throughout the experiment by adjusting the height of the free end of the plastic tubing, which was connected to the outlet of the window.
Protocol for in vivo experiment
In order to determine the vasodilating responses of the resting pial arteries to calcitonin gene-related peptide (CGRP, 0.01 and 0.1 μmol/L) and levcromakalim (1 and 10 μmol/L), the cortical surface was suffused with artificial CSF containing increasing concentrations of each agent. For measurement of the autoregulatory vasodilation to hypotension in rat 24 hours after SAH, the vasodilatory function of the pial artery in response to stepwise hypotension was determined under suffusion of the cortical surface with artificial CSF containing vehicle. The decrease in blood pressure at each step was maintained for 2 minutes, and changes in vessel diameter during the last minute were measured. Thereafter, we reexamined the vasodilatory and autoregulatory responses in rats that were intracisternally treated with adenovirus encoding AdCMVLacZ and AdCMVSOD 3 days before SAH operation. Alterations in pial arterial diameter and blood pressure were expressed as percent changes in the baseline diameter and mean arterial blood pressure. When necessary, L-NAME (10 mg/kg) was administered intraperitoneally before SAH operation to exclude the involvement of endothelial NO synthesized endogenously.
Measurement of lower limit of cerebral blood flow autoregulation
The animal's head was fixed in a stereotaxic instrument, and mechanically ventilated with room air by a respirator (model 683; Harvard Apparatus). After craniotomy, CBF to the pial artery over the parietal cortical surface was measured by using laser-Doppler flowmetry (Laserflo BPM; Vasamedics, St. Paul, MN, U.S.A.) equipped with a 1-mm-diameter needle probe (Model P-433–5 Needle probe; Vasamedics). The probe was placed lateral to, but near the pial artery in the open window and advanced into the CSF approximately 0.2 mm above the surface of the cortex. The laser-Doppler flowmetry outputs were adjusted to arbitrary units and the changes in CBF were expressed as a percentage of the baseline CBF. The lower limit of autoregulation was defined as the blood pressure at which CBF decreased by 10% of its value at resting mean arterial blood pressure.
Drugs
Calcitonin gene-related peptide was purchased from the Sigma-Aldrich Chemical Co. (St. Louis, MO, U.S.A.) and dissolved in 0.1% bovine serum albumin to make a stock solution of 0.01 mmol/L. Levcromakalim was obtained from Korea Research Institute of Chemical Technology (Daejon, Korea) and dissolved in ethanol-polyethyleneglycol (50:50% vol/vol) to make a stock solution of 1 mmol/L. NG-nitro-L-arginine methyl ester, reduced form of NAD(P)H, lucigenin, hypoxanthine, and xanthine oxidase were purchased from the Sigma-Aldrich Chemical Co.
Statistical analysis
Results are expressed as means ± SD. Student's t-test was used for analyzing values between the data of vehicle and inhibitor-treated groups. The comparisons of mean arterial blood pressure-dependent arterial diameter changes between SAH and sham-operated control groups was analyzed by repeated measures analysis of variance, and followed by Tukey's multiple comparison tests as a post hoc comparison. A value of P < 0.05 was accepted as statistically significant.
RESULTS
Mean levels of blood gas, pH, and baseline pial arterial diameter of SAH, SAH + L-NAME, SAH + L-NAME + AdCMVLacZ, SAH + L-NAME + AdCMVSOD groups were not significantly different from sham-operated control rats. Mean arterial blood pressure of the L-NAME-treated SAH groups was significantly elevated by approximately 30 mm Hg in comparison with control and SAH alone groups (Table 1).
Physiologic variables: mean arterial blood pressure and blood gas analyses
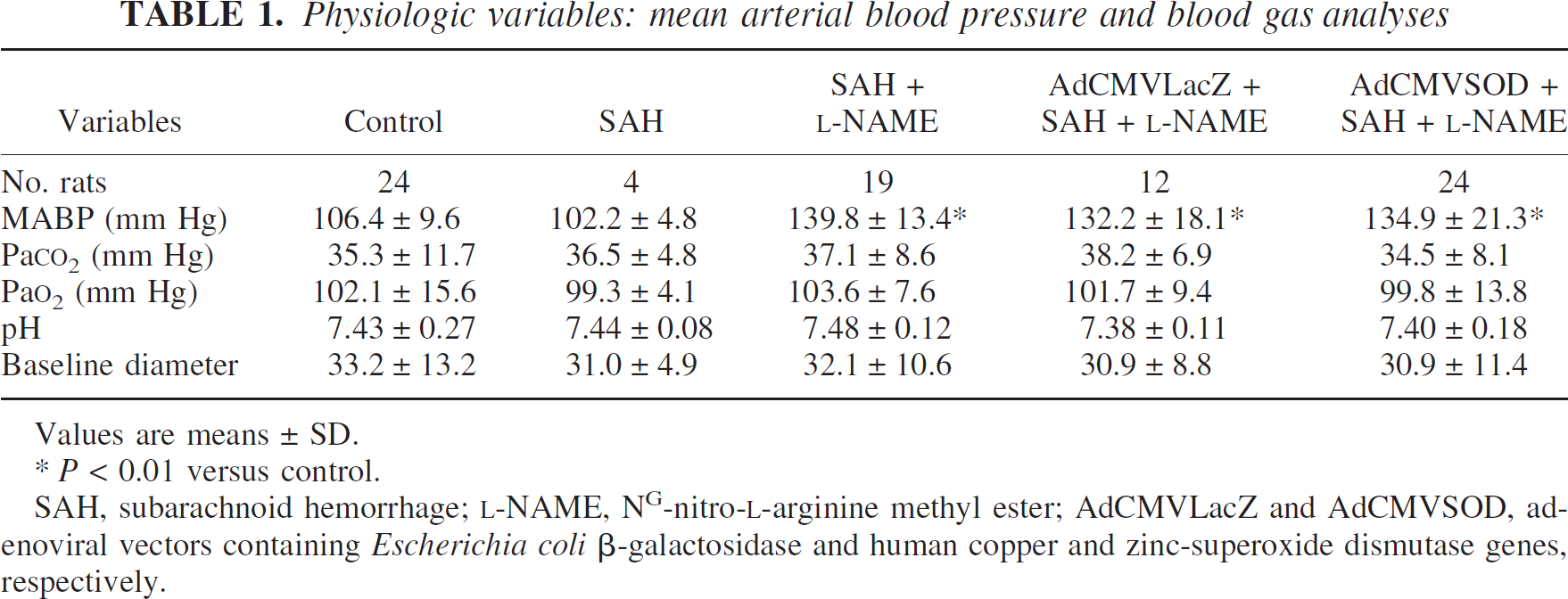
Values are means ± SD.
P < 0.01 versus control.
SAH, subarachnoid hemorrhage; L-NAME, NG-nitro-L-arginine methyl ester; AdCMVLacZ and AdCMVSOD, adenoviral vectors containing Escherichia coli β-galactosidase and human copper and zinc-superoxide dismutase genes, respectively.
AdCMVSOD-mediated transgene expression on the cerebral vessels
After application of recombinant adenovirus (100 μL of 1 × 1010 pfu/mL, intracisternally) encoding human Cu/Zn SOD-1, transgene expression (Cu/Zn SOD-1 mRNA) was evident 1 day and maximized 3 days after administration of AdCMVSOD, as determined by RT-PCR (Fig. 1). Consistent with the results of RT-PCR, human Cu/Zn SOD protein expression was maximized 3 days (232.5 ± 26.9% relative density, P < 0.01) and thereafter, slightly declined 7 days after administration of AdCMVSOD (Fig. 1). In the AdCMVLacZ-treated group, the positive staining for Cu/Zn SOD-1 as determined by immunohistochemistry was identified in the endothelium, but not in the media and adventitial layers of the pial artery. The positive staining for Cu/Zn SOD-1 was manifested in both endothelial and adventitial layers of the vessels from AdCMVSOD-treated group (Fig. 2). The amount of Cu/Zn SOD-1 expression in the endothelium differed little between AdCMVLacZ- and AdCMVSOD-treated groups.
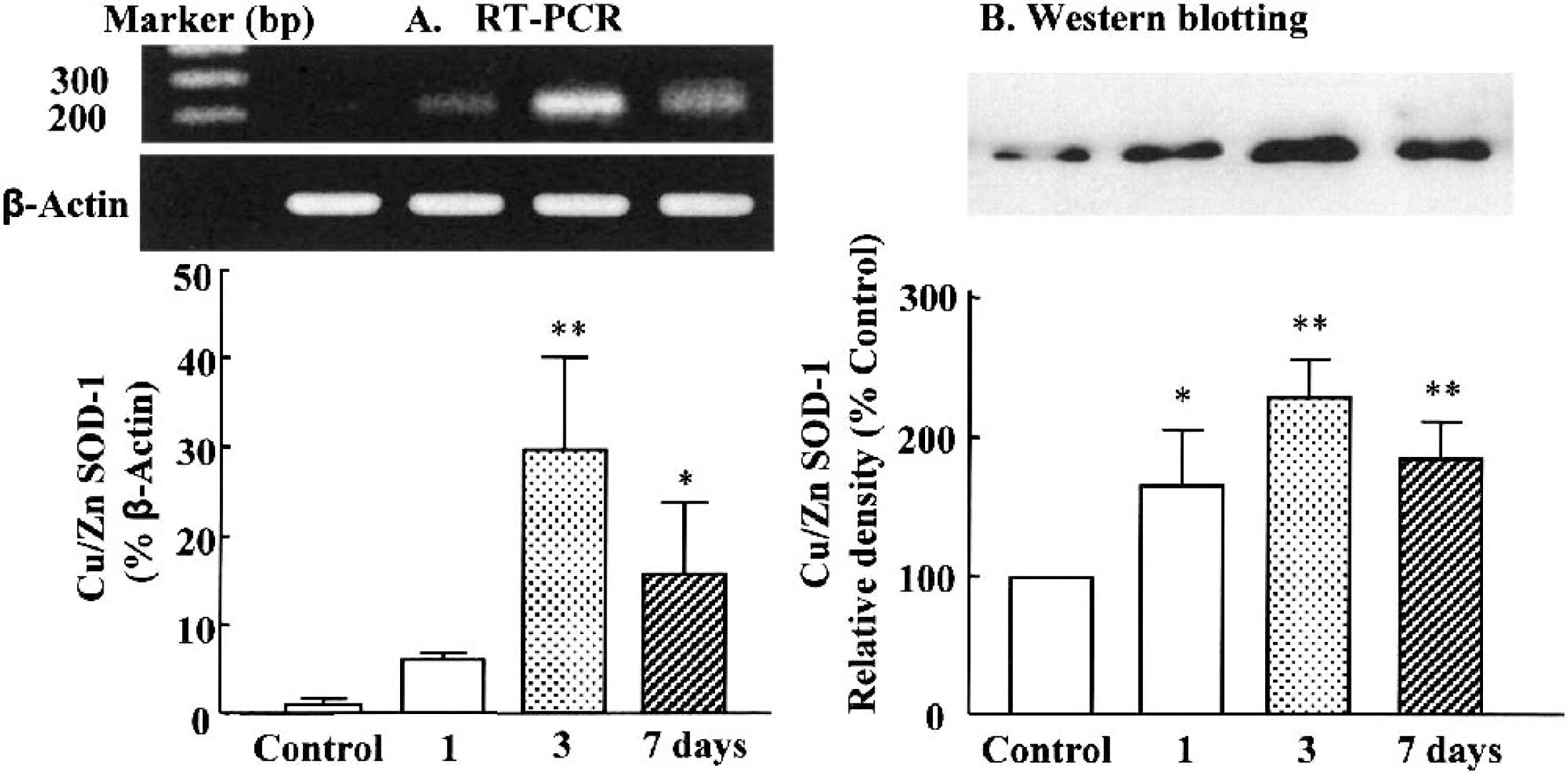
Copper, zinc-superoxide dismutase (Cu/Zn SOD-1) mRNA and protein expression after application of adenoviral vectors containing human Cu/Zn SOD-1 (AdCMVSOD) in the cerebral vessels.
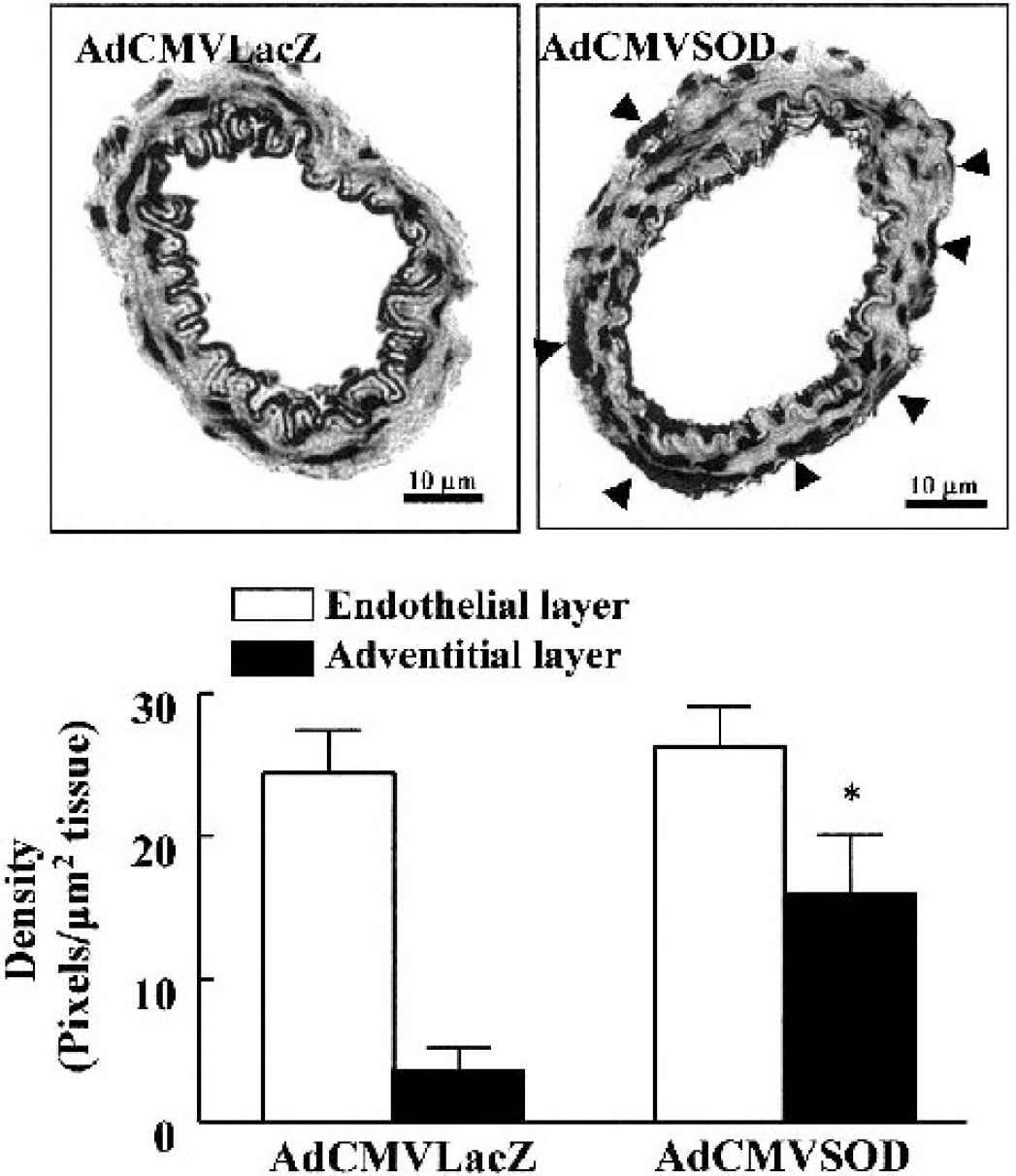
Comparison of densities of immunohistochemical staining for copper, zinc-superoxide dismutase (Cu/Zn SOD-1) in pial arteries in the rats treated with adenoviral vectors containing Escherichia coli β-galactosidase (AdCMVLacZ) and adenoviral vectors containing human Cu/Zn SOD-1 (AdCMVSOD). In the AdCMVLacZ-treated rats, positive staining (gray-black) for Cu/Zn SOD-1 was observed in the endothelium, but not in the media or adventitia. Strong positive staining for Cu/Zn SOD-1 was identified in both endothelial and adventitial cells (arrows) of vessels from the AdCMVSOD-treated group. The density of staining in the pial arterial section was expressed as pixels per square micron tissue. Values are means ± SD from four experiments. *P < 0.05 versus adventitial layer of control.
Effect on superoxide dismutase activities
The SOD activity in the AdCMVLacZ-treated group was not different between control and SAH groups. In control rats treated with AdCMVSOD, KCN-inhibitable Cu/Zn SOD-1 activities significantly elevated from 11.8 ± 1.2 U/mg protein to 16.2 ± 1.9 U/mg protein (P < 0.05), and in the SAH rats, from 11.6 ± 1.1 U/mg protein to 15.7 ± 2.4 U/mg protein, P < 0.05). Mn SOD-2 activity, however, was not altered by treatment with AdCMVSOD (Fig. 3).
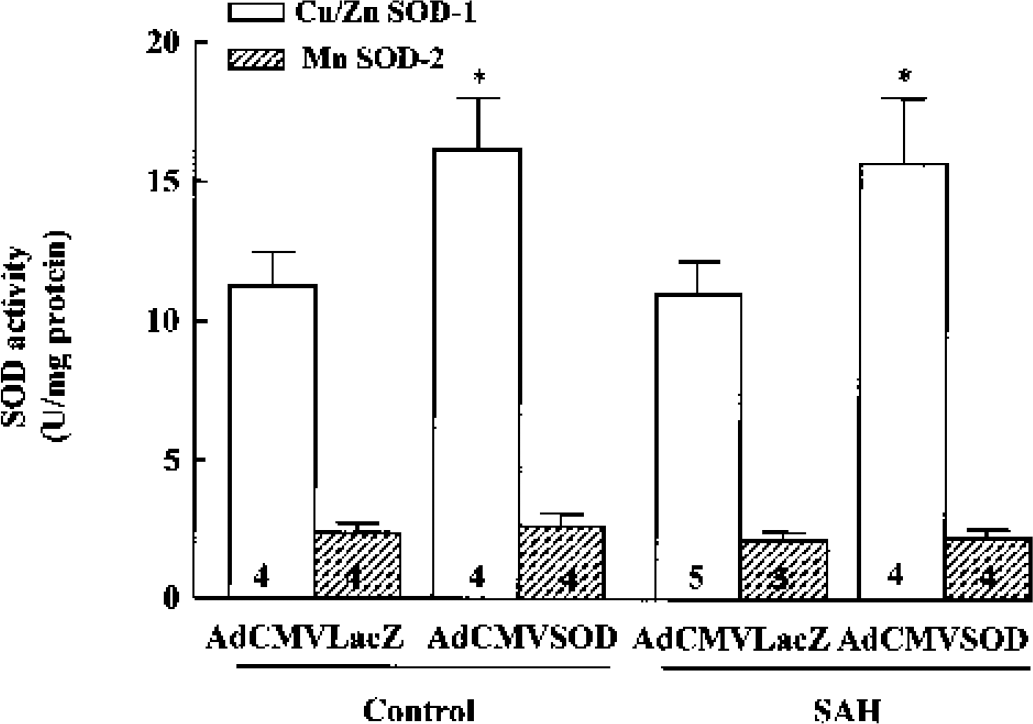
Effects of adenoviral vectors containing Escherichia coli β-galactosidase treatment and adenoviral vectors containing human copper, zinc-superoxide dismutase (Cu/Zn SOD-1) treatment on the SOD activity in cerebral vessels. Cerebral vessels were homogenized and SOD activity was determined by measuring the ability of the homogenate to inhibit hypoxanthine/xanthine oxidase-induced superoxide production. Cu/Zn SOD-1 activity was calculated as the KCN-sensitive SOD activity and Mn SOD-2 activity as KCN-insensitive SOD activity. Each bar represents means ± SD from the numbers indicated in the bars. *P < 0.05 versus vehicle-treated control. SAH, subarachnoid hemorrhage.
NAD(P)H oxidase-dependent superoxide generation
Superoxide production from cerebral vessels in response to NAD(P)H (control, 7.6 ± 6.0 counts sec−1 mg tissue−1) was significantly elevated to 18.3 ± 3.7 counts sec−1 mg tissue−1 (P < 0.01) at 24 hours. The SAH-induced increase in superoxide production was markedly reduced by administration of AdCMVSOD (100 μL of 1 × 1010 pfu/mL) to 12.4 ± 4.2 counts sec−1 mg tissue−1 (P < 0.05), but not by application of AdCMVLacZ (Fig. 4). In the control group, the baseline superoxide production was modestly reduced by administration of AdCMVSOD.
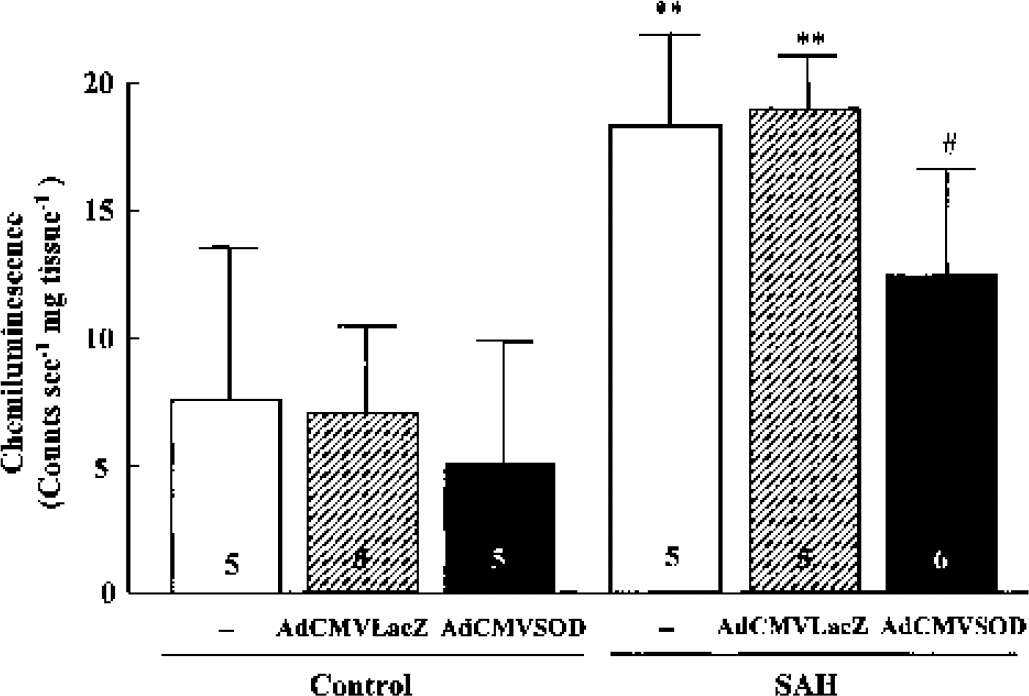
β-Nicotinamide adenine dinucleotide (phosphate) reduced form oxidase-dependent superoxide generation in cerebral vessels after either adenoviral vectors containing Escherichia coli β-galactosidase (AdCMVLacZ) or adenoviral vectors containing human copper, zinc-superoxide dismutase (AdCMVSOD) treatment in sham-operated control and subarachnoid hemorrhage (SAH) group. Superoxide production was measured by lucigenin (5 μmol/L)-enhanced chemiluminescence, and their results are expressed as counts sec−1 mg tissue−1 (dry tissue weight) of cerebral vessels. **P < 0.01 versus corresponding results of control; #P < 0.05 versus SAH alone or AdCMVLacZ treatment. Results represent means ± SD from the numbers indicated in the bars.
Nitrite/nitrate levels and nitrotyrosine products
The levels of NO metabolites nitrite/nitrate (control, 4.0 ± 0.7 μmol/L) in the CSF significantly increased to 11.4 ± 1.9 μmol/L (P < 0.01) at 24 hours after SAH. The elevated levels of nitrite/nitrate were further enhanced under pretreatment with AdCMVSOD (100 μL of 1 × 1010 pfu/mL) to 14.5 ± 1.7 μmol/L (P < 0.05), suggestive of reduced scavenging of NO by superoxide anion (Fig. 5).
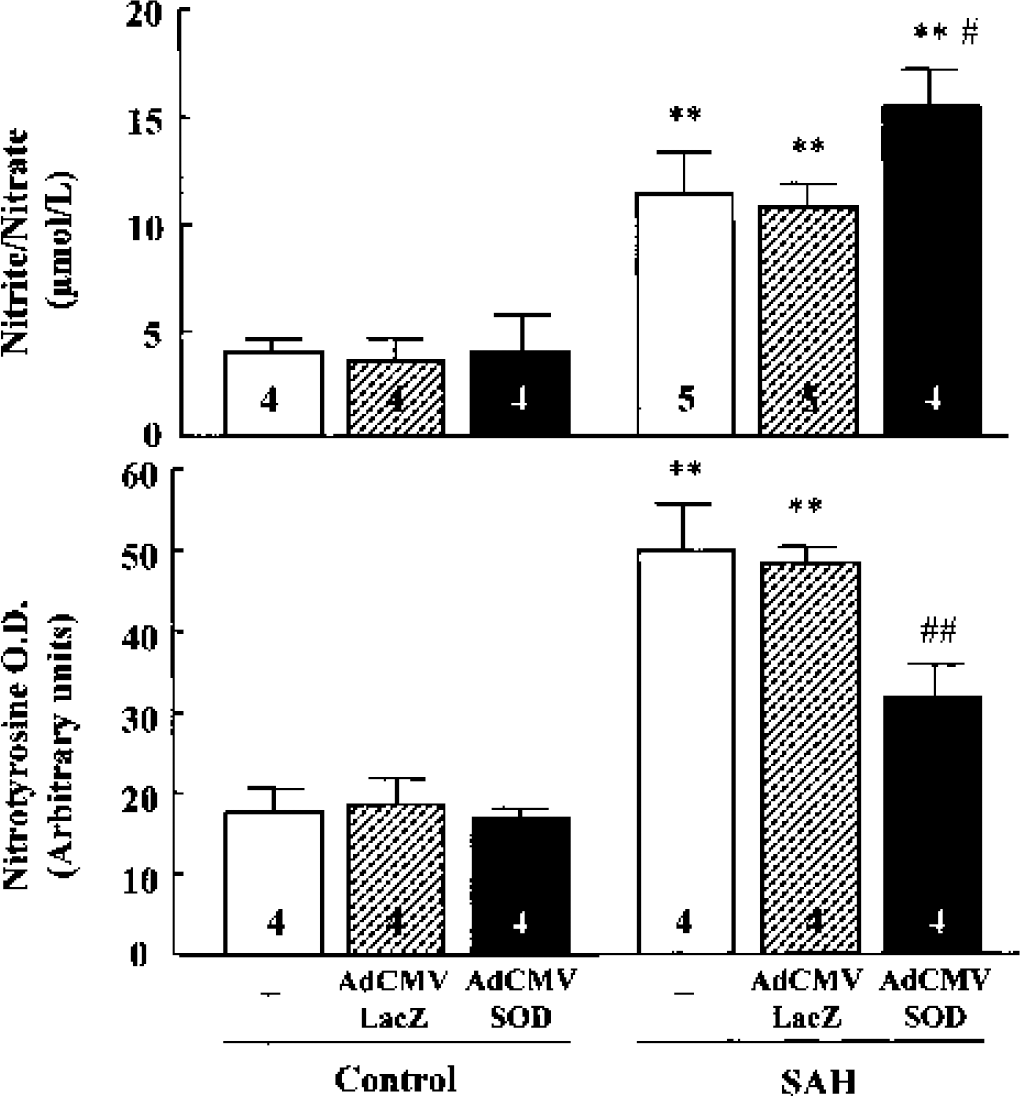
Effects of adenoviral vectors containing human copper, zinc-superoxide dismutase (AdCMVSOD) treatment on nitrite/nitrate and nitrotyrosine levels.
The dual increases in superoxide and NO are the prerequisite conditions for increasing peroxynitrite formation. The nitrotyrosine levels (control, 18.2 ± 3.2 arbitrary unit) significantly increased after SAH (49.3 ± 6.2 arbitrary unit, P < 0.01), which was significantly suppressed by administration of AdCMVSOD (31.8 ± 4.4 arbitrary unit, P < 0.01), indicating that the superoxide anion level was significantly reduced by increased scavenging effect of Cu/Zn SOD-1 (Fig. 5).
Effects on the vasoactivity of the pial artery
Suffusion with artificial CSF containing either CGRP (0.01 and 0.1 μmol/L) or levcromakalim (1 and 10 μmol/L) exerted concentration-dependent vasodilation of the pial arteries, and the maximum diameters of the pial arteries under cranial window reached 102.2 ± 27.2% (n = 5) by 0.1 μmol/L CGRP and 85.3 ± 35.2% (n = 4) by 10 μmol/L levcromakalim. Calcitonin gene-related peptide and levcromakalim caused little change in systemic arterial blood pressure at the concentrations used in this study. At 24 hours after SAH, the vasodilations induced by CGRP and levcromakalim were markedly blunted to 41.6 ± 10.4% (P < 0.01) and 31.3 ± 4.4% (P < 0.05), respectively. In the rats treated with AdCMVSOD (100 μL of 1 × 1010 pfu/mL, intracisternally) 3 days before SAH, the blunted vasodilation to CGRP and levcromakalim induced by SAH was largely restored to 69.0 ± 19.4% (P < 0.05) and 55.1 ± 18.9% (P < 0.05), respectively. The restoration of vasodilation, however, was not manifested at the low dose effects of CGRP (0.01 μmol/L) and levcromakalim (1 μmol/L) (Fig. 6).
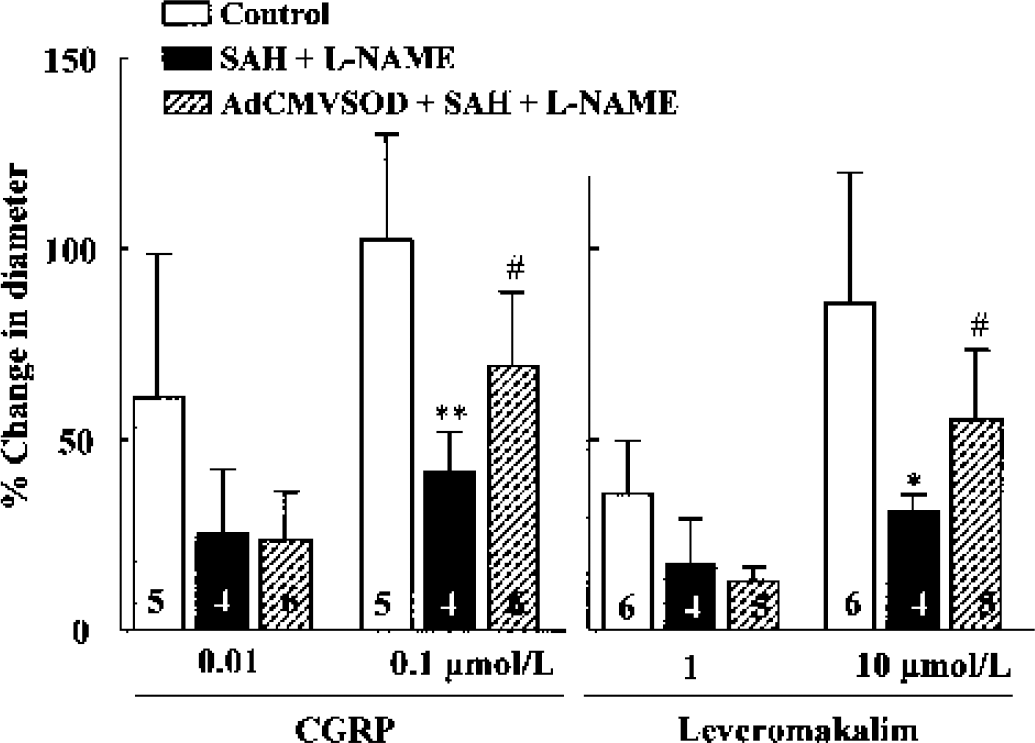
Percent changes in pial arterial diameter in response to calcitonin gene-related peptide (CGRP) and levcromakalim in the sham-operated control and rats subjected to subarachnoid hemorrhage (SAH) + NG-nitro-L-arginine methyl ester (L-NAME) with and without treatment with adenoviral vectors containing human copper, zinc-superoxide dismutase (AdCMVSOD) (100 μL of 1 × 1010 plaque-forming units/mL). The cortical surface was suffused with artificial CSF containing CGRP or levcromakalim. Values are means ± SD from four to six experiments. Graph shows a significant reduction in the vasodilations induced by CGRP and levcromakalim 24 hours after SAH + L-NAME, and its restoration after treatment with AdCMVSOD.
Effect on the impairment of autoregulatory vascular activity
We administered L-NAME (10 mg/kg, intraperitoneally) before SAH operation to exclude any involvement of endogenously synthesized endothelial NO. Under treatment with L-NAME in the SAH rats, the autoregulatory pial arterial vasodilation in response to lowering of blood pressure was markedly blunted [F(1,15) = 97.91, P < 0.0001]. Application of AdCMVSOD (100 μL of 1 × 1010 pfu/mL) 3 days before SAH significantly restored the reduced autoregulatory vasodilation in the SAH + L-NAME group [F(1,15) = 37.29, P < 0.0001] (Fig. 7). After application of AdCMVLacZ, however, the impaired autoregulatory vasodilation was not recovered.
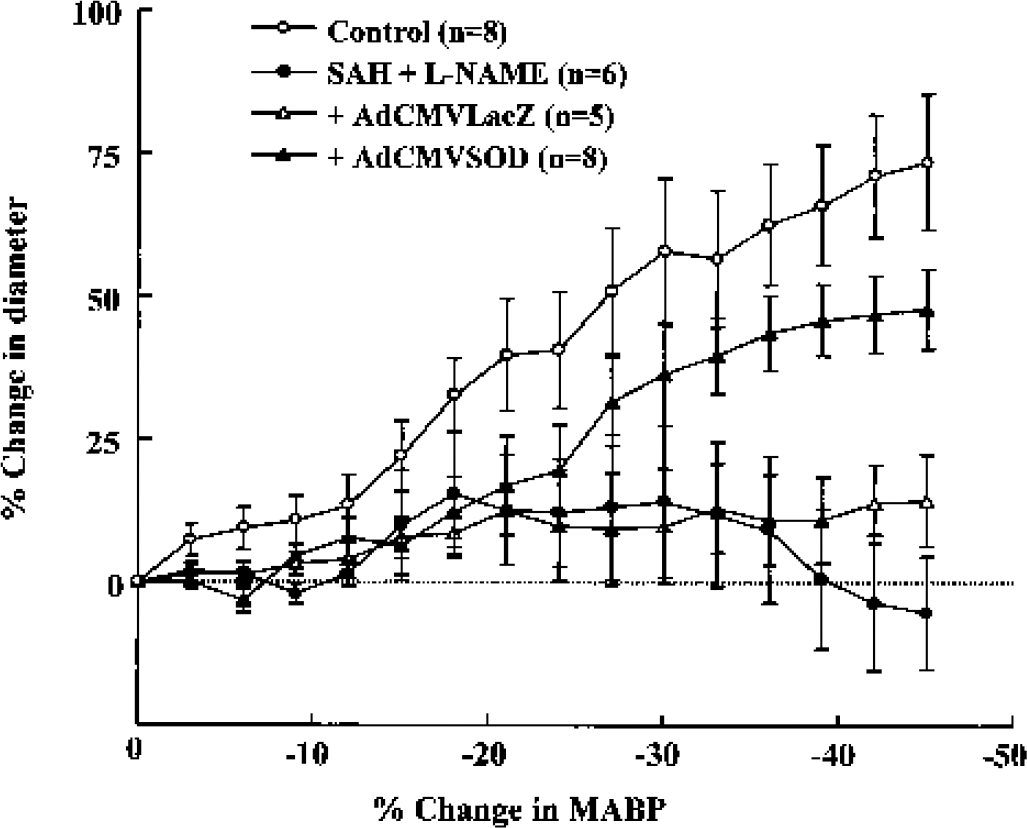
Graph showing a significant reduction in the autoregulatory vasodilator response to change in arterial blood pressure 24 hours after subarachnoid hemorrhage (SAH) + NG-nitro-L-arginine methyl ester (L-NAME) (10 mg/kg, n = 6), and its restoration under pretreatment with adenoviral vectors containing human copper, zinc-superoxide dismutase (AdCMVSOD) (n = 8) (100 μL of 1 × 1010 plaque-forming units/mL, 3 days before SAH), but with adenoviral vectors containing Escherichia coli β-galactosidase treatment. The cortical surface was suffused with artificial CSF during gradual hypotension. Significant differences were shown between the blood pressure-diameter relationship of control and SAH + L-NAME groups, and between SAH + L-NAME and SAH + L-NAME + AdCMVSOD groups by two-way repeated measures analysis of variance.
Effects on the lower limit of cerebral blood flow autoregulation
Cerebral blood flow to the pial artery was preserved in spite of a decrease in mean arterial blood pressure up to approximately 55 to 60 mm Hg in sham-operated control rats. The lower limit of autoregulation was defined as the blood pressure at which CBF decreased by 10% of its value at resting mean arterial blood pressure. The lower limit of CBF autoregulation (control rats; 54.3 ± 12.4 mm Hg, n = 7) was significantly elevated in the SAH group to 73.9 ± 8.4 mm Hg (n = 4, P < 0.05) and in the L-NAME alone group to 83.7 ± 6.4 mm Hg (n = 4, P < 0.01). In the SAH + L-NAME rats, it was further shifted to a higher blood pressure level to 118.8 ± 9.4 mm Hg (n = 5, P < 0.01 versus L-NAME alone). After pretreatment with AdCMVSOD (100 μL of 1 × 1010 pfu/mL) 3 days before SAH, the lower limit of CBF autoregulation in SAH + L-NAME rats was significantly restored to 79.8 ± 6.9 mm Hg (n = 5, P < 0.01) (Fig. 8). Application of AdCMVLacZ was without effect (data not shown).
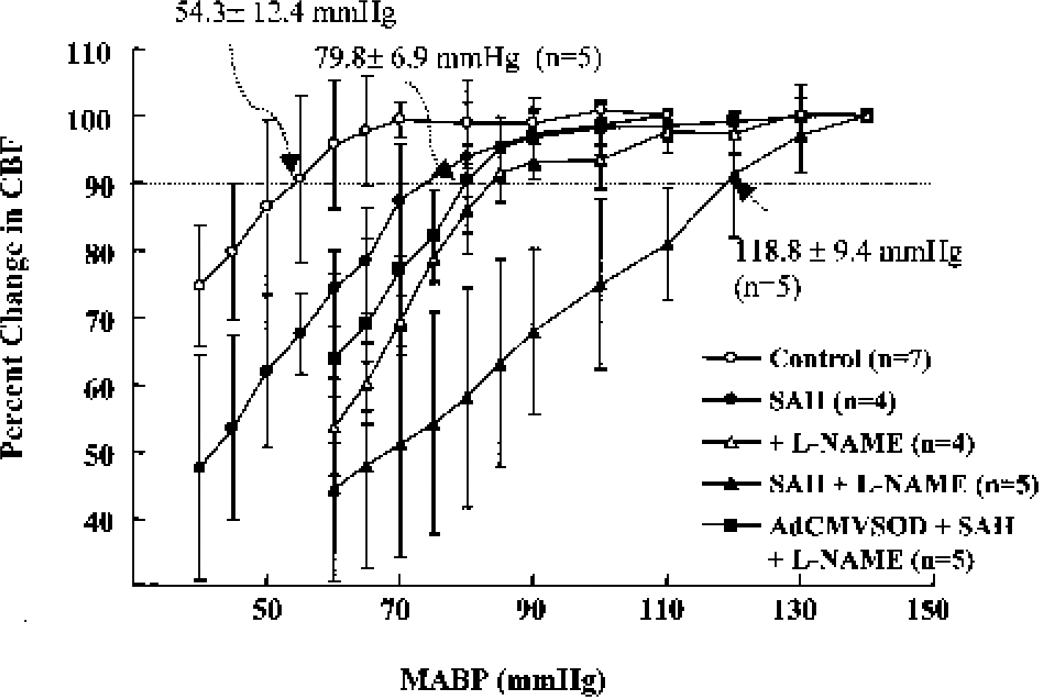
Changes in CBF to changes in mean arterial blood pressure in the pial arteries in the control and rats subjected to subarachnoid hemorrhage (SAH) with and without treatment with NG-nitro-L-arginine methyl ester (L-NAME) (10 mg/kg, intraperitoneally) and L-NAME + adenoviral vectors containing human copper, zinc-superoxide dismutase (AdCMVSOD) (100 μL of 1 × 1010 plaque-forming units/mL, 3 days before SAH). Arrows indicate the lower limits of CBF autoregulation defined as the mean arterial blood pressure at which CBF decreased by 10% of the baseline value. Values are means ± SD from the numbers indicated in parentheses. Significant differences were shown in the lower limit of CBF autoregulation between sham-operated control and SAH alone groups (P < 0.05), and between sham-operated control and SAH + L-NAME (P < 0.01), and between SAH + L-NAME and SAH + L-NAME + AdCMVSOD (P < 0.01).
DISCUSSION
The major findings of this study were that administration of recombinant adenovirus encoding human Cu/Zn SOD-1 3 days before experiments exerted enhanced Cu/Zn SOD-1 activity in association with increase in Cu/Zn SOD-1 mRNA and protein expression in the cerebral vessels of both sham-operated control and SAH rats, which was accompanied by a significant reduction of superoxide anion and increased NO levels, with resultant suppression of nitrotyrosine production. In line with these evidences, the gene therapy markedly restored the blunted vasodilation in response to CGRP and levcromakalim, and significantly improved the autoregulatory vasodilation in response to acute hypotension, leading to significant restoration of CBF autoregulation during the acute stage after SAH.
Cerebral autoregulation may be altered in some disorders of central nervous system including brain trauma (DeWitt et al., 1986; Hong et al., 2001). The severity of neurotic symptoms in the chronic stage of cerebral infarction was reportedly correlated with CBF levels (Heiss et al., 1977). Thus, maintenance of CBF has been considered to be very important from a therapeutic point of view. Although the exact mechanism(s) contributing to the impairment of CBF autoregulation are still unclear, there is convincing evidence that oxygen free radicals including superoxide anion play an important role in the impairment of CBF autoregulation. Recently, Hong et al. (2001) have shown that the altered CBF autoregulation in rat pial artery in response to acute hypotension after fluid percussion injury is associated with reduced vasodilation in response to CGRP and levcromakalim. They postulated that the altered autoregulation of CBF is ascribed to the injury-induced activation of tyrosine kinase-linked inhibition of K+ channels. In line with these considerations, most recently, Shin et al. (2002) have provided further evidence that SAH during acute stage causes an increase in NAD(P)H oxidase-dependent superoxide formation in cerebral vessels, which may contribute to the impaired autoregulatory vasodilation in rat pial artery during acute stage (24 hours) after SAH.
Major sources of superoxide anion in cerebral vessels after SAH include oxyhemoglobin conversion to methemoglobin (Misra and Fridovich, 1972) and NAD(P)H oxidoreductase (Mohazzab et al., 1997). Superoxide dismutase converts superoxide to hydrogen peroxide (H2O2) and represents the first line of defense against oxygen toxicity. Three forms of SOD have been described in humans: one isoform, containing copper and zinc at its active site (Cu/Zn SOD-1), is found in the cytoplasm of cells. The other isoform, containing manganese at its active site, is located in the mitochondria (Mn SOD-2). Another isoform is present in the extracellular fluids such as plasma (Cu/Zn SOD-3) (Halliwell, 1994). According to Spranger et al. (1997), SOD activity was significantly low in patients with ischemic stroke, and the endogenous antioxidants were depleted as a consequence of an excessive production of oxygen free radicals. Inconsistent with their report, Cu/Zn SOD-1 mRNA and their activities in our results little differed between control and SAH rats during acute stage (24 hours). Nevertheless, some controversial results were reported about the effectiveness of the exogenously supplied SOD in amelioration of vasospasm after SAH. Some investigators reported that they did not observe its effectiveness (Macdonald et al., 1992), whereas Shishido et al. (1994) demonstrated that intrathecal injection of human recombinant SOD prevented the occurrence of vasospasm in rabbit SAH model, possibly as a result of preventing endothelial injury initiated by superoxide anions. Kamii et al. (1999) recently demonstrated that, in transgenic mice overexpressing human Cu/Zn SOD, a significant attenuation of vasospasm induced by SAH was ameliorated. Even though SOD has been reported to be effective in eradicating the deleterious role of the oxidative stress and preventing alterations in CBF after SAH, exogenously supplied SOD has a short half-life in plasma by approximately 6 minutes in rats (Odlind et al., 1988), and the entry of exogenous SOD into the brain is normally very limited by the blood-brain barrier (Yoshida et al., 1992). These complicated factors suggest the rational basis for using the gene transfer of recombinant adenovirus encoding human Cu/Zn SOD-1 in the SAH model. Adenoviral vectors are used to achieve efficient transfer and expression of recombinant genes in different vasculatures both in ex vivo and in vivo experiments, thereby raising the possibility of their use to treat vascular disorders (Schneider and French, 1993; Nabel and Nabel, 1994). Recently, it has been demonstrated that intracisternal administration of adenoviral vector encoding β-galactosidase effectively transfers recombinant genes to the cerebral vasculatures of the rat, mice, and rabbit (Ooboshi et al., 1995; Christenson et al., 1998; Toyoda et al., 2000; Kim et al., 2002).
In our present study, increased expression of Cu/Zn SOD-1 mRNA and Cu/Zn SOD-1 protein as well as enhanced immunohistochemical transgene expression in adventitial layer of the pial artery were maximally demonstrated 3 days after intracisternal administration of recombinant adenovirus encoding human Cu/Zn SOD-1, which was accompanied by significantly decreased superoxide anion level. Considering some reports that an NAD(P)H oxidase in adventitial cells was a major source of superoxide in the vascular cells (Pagano et al., 1995; Griendling et al., 2000), periadventitial transfer of the Cu/Zn SOD-1 gene was expected to prevent oxygen radical-mediated alterations in the vascular function. In our results, the findings showing that recombinant adenovirus-mediated transfer to the cerebral vasculature of Cu/ZnSOD-1 cDNA via adventitia has prevented the alterations in CBF autoregulation associated with restoration of vasodilation to vasodilators suggest the adventitial production of an NAD(P)H oxidase-derived superoxide anion.
In the preliminary study, the increased NO formation in association with eNOS mRNA expression was accompanied by increase in superoxide production from cerebral vessels during the acute stage after SAH. The molecular mechanisms underlying increased generation of these radicals are still unclear. It is speculated, however, that these radicals may potently contribute to the exaggerated endothelial cell injury. Given NAD(P)H oxidase in the adventitial cells being the major source of superoxide in the vascular cells (Griendling et al., 2000), periadventitial transfer of the Cu/Zn SOD-1 gene is a good target to prevent oxygen radical-mediated alterations in vascular function. Consequently, recombinant adenovirus-mediated transfer of Cu/Zn SOD-1 gene to the cerebral vasculature has prevented the alterations in CBF autoregulation with restoration of vasodilation to vasodilators (CGRP and levcromakalim) and acute hypotension in the present study. Based on these results, it is postulated that alterations in CBF autoregulation and vascular reactivity to vasodilators may be attributed to the overproduction of superoxide anion produced after SAH.
A growing amount of evidence has addressed the observation that decreased NO availability with decreased endothelial NOS mRNA in the arterial wall was correlated with development of delayed vasospasm 3 to 7 days after SAH (Hino et al., 1996; Pluta et al., 1996; Yamamoto et al., 1997). During acute stage after SAH, however, the NO metabolic products (nitrite/nitrate) significantly increased in CSF at 12 to 24 hours after SAH in association with increase in endothelial NOS mRNA, which was consistent with the result of Suzuki et al. (1997). The results showing that administration of gene SOD 3 days after SAH exhibited a significant elevation of Cu/Zn SOD-1 accompanied by increased available NO provided strong evidence that gene transfer of Cu/Zn SOD-1 could preserve the autoregulatory vasodilation with CBF autoregulation during acute stage after SAH, thereby reducing the morbidity and mortality of patients with SAH. As predicted, the peroxynitrite-induced nitrotyrosine levels, an index of peroxynitrite formation (Beckman, 2001), significantly decreased after administration of gene SOD, indicating diminished levels of superoxide anion, even though the availability of NO was increased. Taken together, superoxide anion rapidly reacts with NO to form peroxynitrite, a strong oxidant, and when protonated, it dissociates to form the hydroxyl radical and nitrogen dioxide, potent oxidizing species. Thus, an endogenous overexpression of SOD stimulated by gene transfer may actually reduce the amount of superoxide and, in contrast, increase the endothelial NO production with prolonged half-life of NO, thereby leading to diminished formation of peroxynitrite and hydroxyl radical (Medele et al., 1996).
It is summarized that SAH-induced overproduction of superoxide anion in cerebral vessels may contribute to the SAH-induced impairment of CBF autoregulation, and administration of recombinant adenovirus-mediated transfer of Cu/Zn SOD-1 gene effectively ameliorates the impairment of CBF autoregulation of the pial artery.
Footnotes
Acknowledgements
The authors thank Dr. John Engelhardt, Gene Therapy Core Center, University of Iowa, Iowa, U.S.A. for his generous donation of adenoviral vectors containing transgenes.
