Abstract
Impaired CBF autoregulation during vasospasm after aneurysmal subarachnoid hemorrhage (SAH) could reflect impaired capacity of distal vessels to dilate in response to reduced local perfusion pressure or simply indicate that the perfusion pressure distal to large arteries in spasm is so low that vessels are already maximally dilated. Autoregulatory vasodilation can be detected
Keywords
Autoregulation of cerebral blood flow (CBF) to changes in systemic arterial pressure has been extensively studied in patients with aneurysmal subarachnoid hemorrhage (SAH). During the early period before the development of vasospasm, autoregulation is often intact unless complicating factors such as hydrocephalus or intracerebral hemorrhage exist. With the development of large artery vasospasm, autoregulation is typically impaired (Darby et al., 1994; Voldby et al., 1985; Ishii, 1979; Nornes et al., 1977; Heilbrun et al., 1972). Normally, as regional cerebral perfusion pressure (rCPP) decreases, distal arterioles dilate. Vascular resistance is reduced, thus maintaining CBF and brain oxygenation. When vasodilation can no longer compensate for reduction in rCPP, this autoregulatory mechanism fails with subsequent reduction in CBF if CPP decreases further (Grubb et al., 1975; Kontos et al., 1978; MacKenzie et al., 1979; Paulson et al., 1990; Powers, 1992). Thus, impaired autoregulation of CBF in response to changes in systemic arterial pressure in patients with arteriographic spasm of large arteries at the base of the brain could be due either to impaired capacity of the distal vessels to dilate appropriately in response to reductions in local perfusion pressure or simply reflect the fact that the perfusion pressure distal to a large artery in spasm is already so low that the autoregulatory vessels are maximally dilated.
Autoregulatory vasodilation of distal blood vessels can be detected
METHODS
Subjects
Aneurysmal SAH
Twenty-nine PET studies of regional CBV (rCBV), regional CBF (rCBF), regional OEF (rOEF), and regional CMRO2 (rCMRO2) performed on 18 patients after aneurysmal SAH were identified from our laboratory records. The ages of the patients ranged from 24 to 70 years (mean age, 44 years). Clinical characteristics for this group are shown in Table 1. Data from 16 of 29 PET studies were presented in an earlier publication (Carpenter et al., 1991). This previous study was intentionally limited to preoperative patients without confounding factors such as hydrocephalus, intracerebral hemorrhage, or later evidence of ischemic stroke. The present study includes all patients studied as part of a research project investigating cerebral hemodynamics and metabolism after SAH performed from 1981–1988. None of the patients with SAH received calcium antagonists or vasopressor. Several received phenobarbital or epsilon amino caproic acid (Amicar®).
Clinical characteristics of the patients with subarachnoid hemorrhage
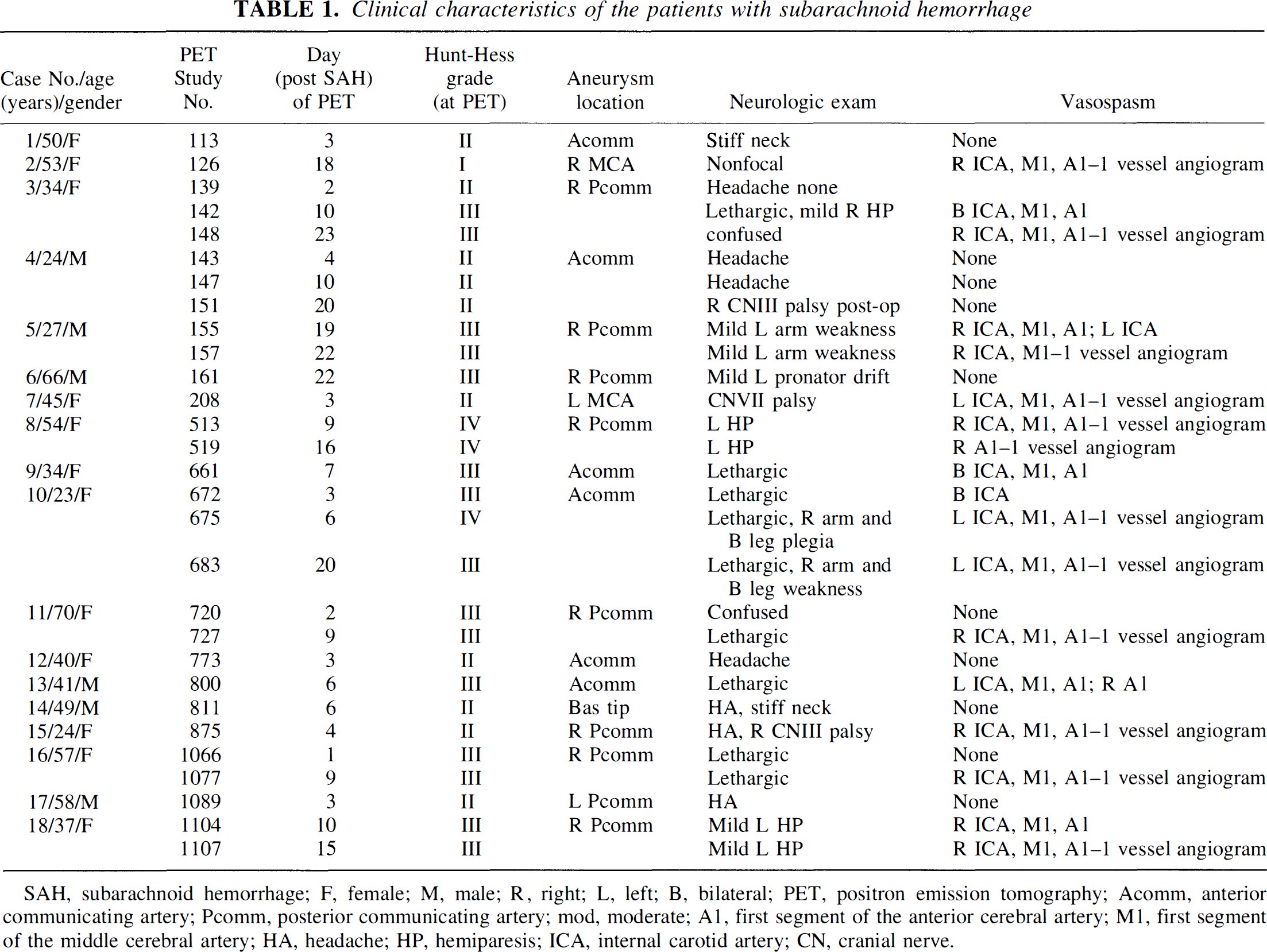
SAH, subarachnoid hemorrhage; F, female; M, male; R, right; L, left; B, bilateral; PET, positron emission tomography; Acomm, anterior communicating artery; Pcomm, posterior communicating artery; mod, moderate; Al, first segment of the anterior cerebral artery; Ml, first segment of the middle cerebral artery; HA, headache; HP, hemiparesis; ICA, internal carotid artery; CN, cranial nerve.
Cerebral arteriography was performed within 1 day of PET for all but three studies. One patient underwent PET on day 3 with arteriography performed on day 1 and day 6 post-SAH. (Day of PET refers to day post-SAH, with hemorrhage occurring on day 0.) Both arteriograms revealed vasospasm. The second patient had a PET study performed on day 15 with arteriograms on day 9 and day 18 post-SAH that both showed vasospasm. The third patient underwent arteriography on day 1 post-SAH which showed no evidence of vasospasm and had a PET study performed 2 days later. None of these patients had clinical changes between the time of angiogram and PET study. For the purposes of this report, the first two patients were categorized as having vasospasm at the time of PET and the third was categorized as having no spasm.
All angiograms were carotid or combined carotid-vertebral selective arteriograms, filmed in two or more planes of view. Vasospasm affecting the internal carotid artery territory was determined by measuring the arterial caliber of the supraclinoid internal carotid artery, M1 branch of the middle cerebral artery (MCA) and A1 branch of the anterior cerebral artery (ACA) and comparing this to either an arteriogram completed before the onset of angiographic vasospasm, or by measuring the arterial caliber in the unaffected contralateral hemisphere. Spasm was graded as severe if the caliber of the artery was reduced by greater than 50% and mild if the caliber reduction was less than 50% (Grubb et al., 1977; Carpenter et al., 1991). For this report, arterial territories with mild and severe spasm were combined into one group.
Normal volunteers
PET studies were also performed on 19 age-matched normal volunteers. Their ages ranged from 24 to 75 years (mean age, 45 years). Six of volunteers, aged 68 to 75 years, were volunteer participants in the Washington University Memory and Aging Project. Each had a clinical interview, general physical examination, standardized 2-hour battery of psychological tests, blood and urine tests, and a CT scan of the brain. Only those classified as normal and healthy according to previously published criteria were included in our study (Berg et al., 1982). The remaining 13, aged 24 to 50 years, were volunteers who had no history or evidence of neurologic disease and were recruited by public advertisement.
Carotid occlusion
We also identified from our laboratory records all PET studies of rCBF, rCBV, rOEF, and rCMRO2 performed on patients with unilateral carotid artery occlusion who had normal head CT scans. We chose only patients with normal head CT scans to eliminate the complex changes in cerebral hemodynamics and metabolism that follow ischemic stroke (Powers, 1992). From this group we selected all patients with increased OEF distal to the occluded vessel. Five patients met these criteria. Although not a criterion for inclusion, all five patients had a history of transient ischemic attacks. Their ages ranged from 47 to 70 years (mean age, 61 years). We and others have described increases in CBV in similar patients (Gibbs et al., 1984; Powers et al., 1984).
Patient and normal volunteer PET studies were intermixed throughout the same period. All were all studied on the same scanner with the same methods.
Informed written consent was obtained from each subject or next of kin before all PET studies. This protocol was approved by the Human Studies Committee (Institutional Review Board) and the Radioactive Drug Research Committee of the Washington University School of Medicine.
PET
Measurements of rCBF, rCBV, and rOEF were performed on the PETT VI tomograph in the low-resolution mode with a reconstructed transverse resolution of 18-mm full-width at half-maximum (Ter-Pogossian et al., 1982). This tomograph collects seven parallel slices, 1.4 cm apart. Venous and arterial catheters were placed to permit radiotracer injection and collection of arterial blood samples. Subjects were positioned so that the lowest tomographic slice was near the canthomeatal line. The head was then immobilized by an individual face mask made of heat-molded plastic and attached to a headrest. A plastic plate with seven radio-opaque wires was used to record the position of each of the seven PET slices on a lateral skull radiograph. A transmission scan with a 68Ge/68Ga-ring source was performed for each subject to provide data on photon attenuation necessary for quantitative reconstruction of subsequent scans.
rCBF was measured after bolus intravenous injection of 15O-labeled water (H215O)(Videen et al., 1987; Raichle et al., 1983). rCBV was measured after brief inhalation of 15O-labeled carbon monoxide (C15O) (Videen et al., 1987; Martin et al., 1987). rOEF was measured after a brief inhalation of 15O-labeled oxygen (O15O) (Videen et al., 1987; Mintun et al., 1984). rCMRO2 was calculated as the product of rCBF, rOEF, and arterial oxygen content (CaO2). The regional cerebral venous oxygen content (rCvO2) was calculated from the formula CaO2 × (1 – rOEF). Subjects' eyes and ears were not masked. The short half-life of 15O (122.1s) allowed these studies to be performed in rapid succession. Total study duration ranged from 1 to 2 hours. All radiotracers were produced by the Washington University medical cyclotrons (Welch and Kilbourn., 1985).
During each subject's PET study a minimum of three arterial blood samples were taken and analyzed for the Pa
Analysis
For each subject's PET study, a stereotactic system was used to define 18, 1.43 × 1.43 cm regions of interest (ROI), 7 in the cortical territory of each middle cerebral artery, and 2 in the cortical territory of each ACA (Powers et al., 1985). Each ROI was placed deep to the cortical surface to avoid the effect of partial volume averaging from the pial vessels, skull, and scalp. Any region that fell into an area of reduced CMRO2 that corresponded to ischemic infarction or hematoma on CT was removed from the analysis. From the remaining regions, a mean value for the middle cerebral artery and ACA ROI in each hemisphere was calculated for CBV, CBF, OEF, and CMRO2.
For the subjects with SAH, MCA, and ACA regions were divided into two groups based on the presence or absence of angiographic cerebral vasospasm. Some arteriograms were only unilateral studies; data from the hemisphere for which there was no accompanying arteriographic data were not used. This yielded a total of 23 regions without vasospasm and 23 regions with vasospasm. Data were also obtained from both hemisphere in 19 control subjects yielding 38 regions. From the five patients with ipsilateral carotid occlusion, data was used only from the hemisphere ipsilateral to the occluded vessel.
Statistical analysis
One-way analysis of variance (ANOVA) was used to compare the four groups (SAH without vasospasm, SAH with vasospasm, carotid occlusion and the normal controls) with respect to PET measurements of rCBV, rCBF, rOEF, rCMRO2, and rCvO2, as well as systemic PaCO2, hematocrit, and CaO2. An alpha probability of
All data are presented as mean ± SD. Statistical computations were performed with SPSS (SPSS, 1993).
RESULTS
Data are summarized in Table 2. Patients with SAH without vasospasm showed significant reductions in CBF, CMRO2, and CBV compared to normal controls whereas OEF was normal. Patients with vasospasm also showed significant reductions in CBF and CBV. CMRO2, although lower than normal, was not significantly different whereas OEF was significantly increased. Patients with carotid occlusion also showed significant reductions in CBF with no change in CMRO2. Both OEF and CBV were significantly increased. Findings similar to OEF but in the opposite direction were found for rCvO2. No differences in PaCO2, hematocrit, and CaO2 were found.
Physiologic measurements in normal volunteers, patients with subarachnoid hemorrhage, and patients with carotid occlussion
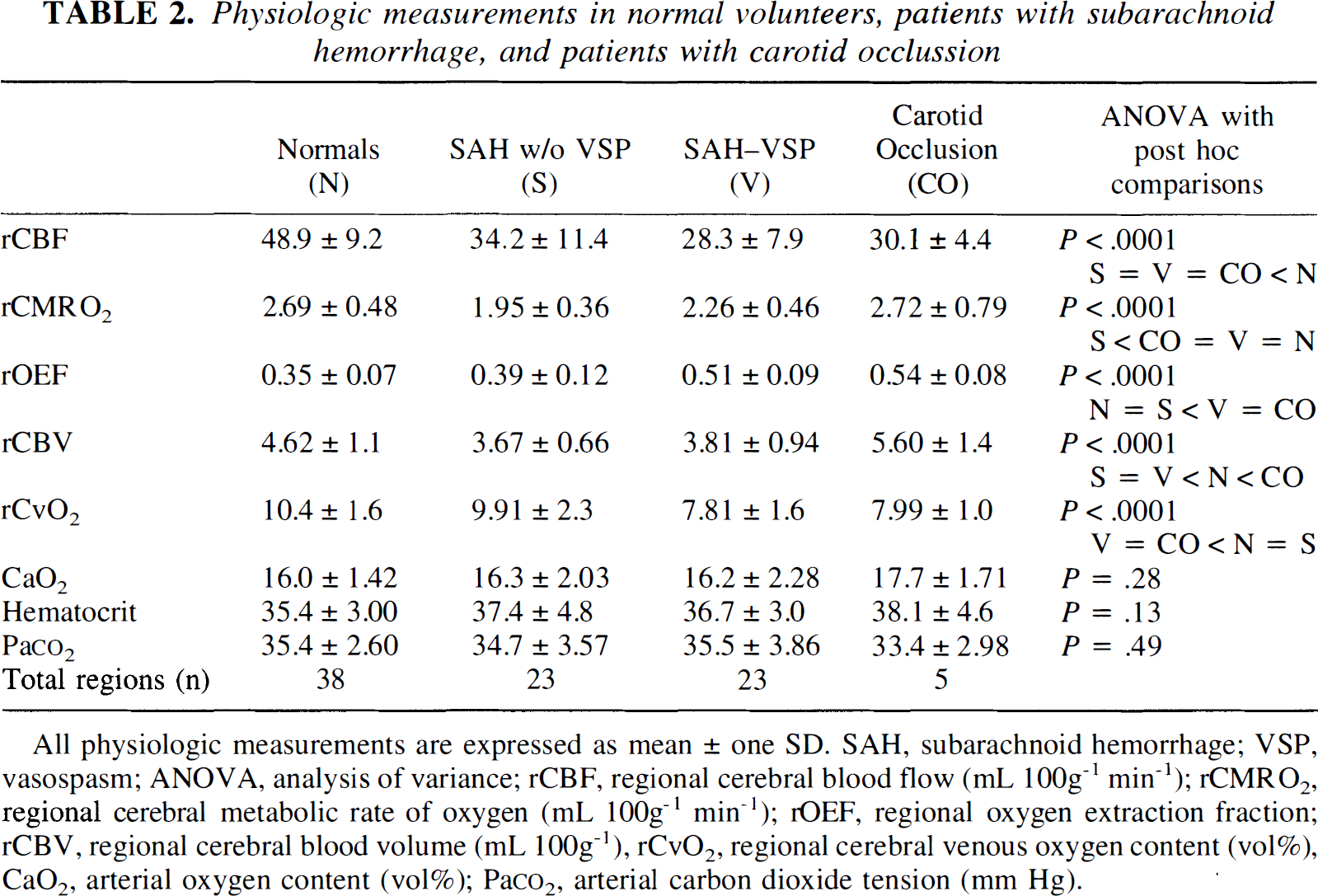
All physiologic measurements are expressed as mean ± one SD. SAH, subarachnoid hemorrhage; VSP, vasospasm; ANOVA, analysis of variance; rCBF, regional cerebral blood flow (mL 100g−1 min−1); rCMRO2, regional cerebral metabolic rate of oxygen (mL 100g−1 min−1); rOEF, regional oxygen extraction fraction; rCBV, regional cerebral blood volume (mL 100g−1), rCvO2, regional cerebral venous oxygen content (vol%), CaO2, arterial oxygen content (vol%); Pa
A closer comparison of the patients with vasospasm and those with carotid occlusion shows that these two groups were closely matched for rCBF (28.3 ± 7.9 versus 30.1 ± 4.4 mL 100 g−1 min−1), rOEF (.51 ± .09 versus .54 ± .08) and rCvO2 (7.81 ± 1.6 versus 7.99 ± 1.0 vol%). The rCBV findings were very different, however. In patients with vasospasm, rCBV (3.81 ± 0.94 mL 100 mg−1) was significantly less than normal (4.62 ± 1.1) whereas in patients with carotid occlusion rCBV (5.60 ± 1.4) was significantly higher than normal. Because the five patients with carotid occlusion had been chosen from a larger group based on the finding of increased OEF, we also separately considered those patients with SAH and vasospasm who had increased OEF. Fifteen of 23 patients with SAH and vasospasm had OEF values above the upper limit of 0.50 observed in the control group. Mean CBV in this subset of 15 patients was 3.50 ± 0.86 mL 100 g−1, even less than in the total group of 23.
DISCUSSION
Autoregulation can be defined on a regional basis as the ability of individual resistance vessels to dilate in response to reductions in perfusion pressure and constrict in response to increases in perfusion pressure (Paulson et al., 1990). The vasodilatory responses to reduced perfusion pressure have been studied primarily in visible pial arterioles but small parenchymal arterioles are believed to participate as well (Kontos et al., 1978; MacKenzie et al., 1979). Vasodilation in response to global or regional reductions in CPP produces an increase in CBV, which can be measured using a variety of techniques including PET (Ferrari et al., 1992; Grubb et al., 1973; Grubb. et al., 1975; Toyama et al., 1990; Gibbs et al., 1984; Powers et al., 1984; Powers et al., 1987).
Autoregulation of CBF to changes in systemic arterial pressure is defective in the majority of patients with angiographic vasospasm of large arteries after aneurysmal SAH (Darby et al., 1994; Voldby et al., 1985; Ishii, 1979; Nornes et al., 1977; Heilbrun et al., 1972). Such abnormalities could be due to impaired capacity of the distal vessels to dilate appropriately in response to reductions in local perfusion pressure or simply reflect the fact that the perfusion pressure distal to the large arteries in spasm is already so low that the vessels are maximally dilated. Under the latter circumstances, rCBV will be increased.
In this study, we found that patients with aneurysmal SAH without vasospasm showed reductions in CBF, CMRO2, and CBV with normal OEF. This is the pattern of primary metabolic depression as defined by Sette et al. (1989) in which reduction in metabolic demand cause secondary decrease in CBF with attendant constriction of parenchymal vessels. Similar findings in a subset of these patients were reported by Carpenter et al. (1991) and attributed to the effects of the subarachnoid blood. In the patients with SAH and arteriographic vasospasm, CBF and CBV were decreased with minimal change in CMRO2 and with increased OEF. The pattern of reduced CBF with increased OEF has been termed misery perfusion (Baron et al., 1981). When this pattern of misery perfusion occurs in patients with chronic atherosclerotic obstruction of the carotid arteries, it is associated with increased CBV reflecting autoregulatory vasodilation of parenchymal vessels as it was in the patients with carotid artery occlusion in this series (Gibbs et al., 1984; Powers et al., 1991). Even in the setting of coincident metabolic depression with misery perfusion, CBV is still increased (Sette et al., 1989). However, the patients with SAH and vasospasm in this series did not show increased CBV in the setting of misery perfusion. In fact, CBV was significantly less than normal controls. While there are several possible explanations for this finding, we believe that it shows a primary impairment in the capacity of distal parenchymal vessels to undergo normal autoregulatory vasodilation during the period of large artery vasospasm after SAH.
Experimental research has shown that the main stimulus to autoregulatory vasodilation
We cannot ascribe our findings to other factors affecting vascular reactivity. Differences in PaCO2 and CaO2 can impair normal autoregulatory vasodilation (Paulson et al., 1990; Maruyama et al., 1985). There was no difference in PaCO2, CaO2, or hematocrit among any of the patient groups or normal controls. We also do not believe that the observed differences were related to medications. All of the PET studies were completed in the 1980s. None of the patients with SAH received calcium antagonists or vasopressors. Many of the SAH patients received epsilon amino caproic acid (Amicar®). There is little information available on the cerebrovascular effects of this drug, but what data there are do not indicate that it has vasoconstrictive properties
Previous studies of CBV in patients with SAH have yielded conflicting results. Grubb et al. (1977) reported a statistically significant increase in CBV in Hunt/Hess grade III—IV patients with angiographic vasospasm when compared to normals. Part of the explanation for the difference between these results and the present series may be methodologic. Grubb et al. used a multiprobe radiation detection system that was targeted primarily on the pial vessels. In this study, we intentionally set our regions of interest deep to the cortical surface to minimize any partial volume averaging effect from the vessels of the scalp, skull, and pia. In an arteriographic study, Bergvall et al. (1973) frequently observed dilation of the distal pial arteries co-existing with proximal arterial vasospasm. However, since we showed an increase in CBV in patients with carotid occlusion using the same region location, we know that these parenchymal vessels participate in the autoregulatory response and that the response can be detected by PET. In a PET study of SAH, Hino et al. (1989) reported a significant increase in CBV in regions of symptomatic angiographic vasospasm. They did not, however, observe an increase in rOEF as we did. Thus, vasospasm was less severe than in the present study. Grubb et al. (1977) did not report OEF values, but examination of the CBF and CMRO2 data indicates that OEF was probably not increased in the patients with vasospasm as compared to those without. It may be that only with severe vasospasm do the parenchymal vessels lose the capacity for autoregulatory vasodilation.
In conclusion, we have shown that parenchymal CBV is reduced in patients with vasospasm secondary to aneurysmal SAH under conditions of tissue hypoxia that produce increased CBV in patients with carotid occlusion. We have not been able to identify any factors to account for this difference other than a direct effect of SAH. These observations provide evidence that distal parenchymal vessels do not show normal autoregulatory vasodilation in patients with angiographic large-vessel vasospasm after SAH.
