Abstract
An impaired CBF autoregulation can be restored by hyperventilation at a PaCO2 level of about 2.9 to 4.1 kPa (22 to 31 mm Hg). However, it is uncertain whether the restoring effect can take place at lesser degrees of hypocapnia. In the current study, CBF autoregulation was studied at four PaCO2 levels: 5.33 kPa (40 mm Hg, normoventilation), 4.67 kPa (35 mm Hg, slight hyperventilation), 4.00 kPa (30 mm Hg, moderate hyperventilation), and 3.33 kPa (25 mm Hg, profound hyperventilation). At each PaCO2 level, eight rats 2 days after experimental subarachnoid hemorrhage (SAH) and eight shamoperated controls were studied. The CBF was measured by the intracarotid 133Xe method. The CBF autoregulation was found to be intact in all controls but completely disturbed in the normoventilated SAH rats. However, by slight hyperventilation, CBF autoregulation was restored in seven of eight SAH rats with a decline in CBF of 10%. The CBF autoregulation was found intact in all of the moderately or profoundly hyperventilated SAH rats, whereas the decline in CBF was 21% and 28%, respectively. In conclusion, hyperventilation to a PaCO2 level between 4.00 and 4.67 kPa (30 to 35 mm Hg) appears to be sufficient for reestablishing an impaired autoregulation after SAH.
Vasomotoric instability, seen as defective CBF autoregulation, arterial vasospasm, and disturbed relationship between CBF and CMR, is frequently observed after subarachnoid hemorrhage (SAH) (Voldby, 1988). These disturbances contribute to development of delayed cerebral ischemic dysfunction, which is a major cause of the poor outcome in patients surviving the initial SAH (Mendelow, 1988).
Hyperventilation has been used for decades to treat intracranial hypertension in patients with acute intracranial disease. It also has been reported both in clinical patients and experimental models that marked hyperventilation to a PaCO2 level of about 2.9 to 4.1 kPa (22 to 31 mm Hg) can restore an impaired CBF autoregulation (Paulson et al., 1972; Hauerberg et al., 1993). However, hyperventilation carries potential risks as well as considerable benefits. Hyperventilation produces vasoconstriction and thereby a decline in CBF. Because the CBF-reducing effect of hyperventilation is suggested to be independent of CMR, CBF passively follows the variations in PaCO2 in a wide range without apparent physiologic purpose; marked hyperventilation also implies a risk of inducing or exacerbating cerebral ischemia, especially in conditions with disturbed vasoreactivity (Darby et al., 1988; Stringer et al., 1993; Muizelaar et al., 1991). Therefore, it would be favorable if the restoring effect can take place at lesser degrees of hypocapnia, which is uncertain, however, as indicated by the previous studies.
The current study investigates the effects of graded hyperventilation on CBF autoregulation after SAH.
MATERIALS AND METHODS
Investigations of CBF autoregulation require extensive manipulation of blood pressure that entails the risk of cerebral edema at high or ischemia at low blood pressure. Therefore, the study was performed experimentally in rats. Sprague-Dawley male rats (Møllegärden, Lille Skensved, Denmark) weighing about 300 g were used. The rats were divided into four groups according to predetermined levels of PaCO2:
Eucapnia: PaCO2 = 5.33 kPa (5.07 to 5.60 kPa (38.0 to 42.0 mm Hg) Slight hyperventilation: PaCO2 = 4.67 kPa (4.33 to 5.00 kPa (32.5 to 37.5 mm Hg) Moderate hyperventilation: PaCO2 = 4.00 kPa (3.67 to 4.32 kPa (27.5 to 32.4 mm Hg) Profound hyperventilation: PaCO2 = 3.33 kPa (3.00 to 3.66 kPa (22.5 to 27.4 mm Hg)
In each group, a subgroup of eight rats with experimental SAH and a subgroup of eight sham-operated controls were studied. All surgical and experimental procedures were approved by the Committee on Animal Experimentation under the Ministry of Justice, Denmark.
Induction of subarachnoid hemorrhage and sham operation
The animals were prepared as described by Delgado et al. (1985). In each animal, a thin indwelling catheter was implanted into the cisterna magna during anesthesia with 2% halothane in 70% N2O and 30% O2. Experimental SAH then was induced by injecting 0.07 mL of autologous blood through the catheter into the cisterna magna. The skin was closed over the catheter, and the animal was allowed to emerge from anesthesia. In the controls, an indwelling catheter was placed as just described without injecting any substances through it.
General surgical preparation
The investigations were performed 2 days after induction of SAH or sham operation. At this time, after experimental SAH, previous studies have demonstrated severe cerebrovascular instability (Delgado et al., 1985; Delgado et al., 1986; Rasmussen et al., 1992). On the day of the experiment, the animal was anesthetized again with a mixture of 2% halothane in 70% N2O and 30% O2. After tracheotomy, the concentration of halothane was decreased to 0.5% for the rest of the experiment, and the animal was connected to a respirator, through which the predetermined PaCO2 level could be obtained by adjusting the inspiratory volume. Catheters were inserted into both femoral arteries for blood sampling and monitoring of MABP, and in both femoral veins for drug administration and blood substitution.
Measurement of CBF
Cerebral blood flow was measured using the method described by Hertz et al. (1977). The 133Xe dissolved in 30 μL of saline (10 mCi/mL) was injected into the internal carotid artery through a catheter placed in the external carotid artery with the tip just cranial to the carotid bifurcation. The branches of the external carotid artery and the pterygopalatine artery were ligated to minimize extracerebral contribution to the measurement. A single collimated NaI (Th) crystal placed ipsilaterally over the exposed cranium recorded the 133Xe activity. The CBF was calculated from the initial slope of a semilogarithmic plot of the washout curve with correction for background activity (Olesen et al., 1971). This method allows rapid and repetitive measurements of CBF in mainly cortical gray matter.
Measurement of physiologic variables
After every CBF measurement, PaCO2, pH, and PaO2 were analyzed in an arterial blood sample of 100 μL using an ABL-500 Acid-Base Analyser (Radiometer, Copenhagen). The blood withdrawn for these analyses was replaced by blood from a donor rat of the same strain. Body temperature was measured through a rectal thermometer and maintained at about 37.5°C with a heating pad. Intracranial pressure (ICP) was measured through the catheter implanted in the cisterna magna and was monitored continuously with MABP by a three-channel polygraph. Cerebral perfusion pressure (CPP) was calculated as the difference between MABP and ICP.
After surgery, the animal was allowed to rest for 30 minutes. In all groups, baseline values of CBF, MABP, ICP, pH, PaCO2, PaO2, and body temperature were measured three times during normoventilation to ensure stable baseline conditions before commencement of autoregulation studies.
Study of CBF autoregulation
After recording baseline values, MABP was increased to about 120 mm Hg by continuous intravenous administration of norepinephrine (0.4%, 0.15 to 0.25 mL/h), and CBF was measured 5 minutes after achieving the new steady MABP level. Thereafter, MABP was decreased stepwise, initially by reducing the infusion rate of norepinephrine and later by controlled bleeding. In this way, CBF was measured repetitively at every 10- to 20-mm Hg decrement of MABP. Each measurement of CBF was carried out 5 minutes after stabilization of the new MABP level.
After the experiment, the rat was killed during the anesthesia by a lethal intravenous bolus of KCl.
Data analysis
The baseline values at different levels of PaCO2, both in controls and in SAH rats, were compared by one-way analysis of variance, followed by two-tailed, unpaired Student's t-test. Baseline values between the controls and the SAH rats were compared by two-tailed unpaired Student's t test. One-way analysis of variance also was used to compare the physiologic state values at each 20-mm Hg change of MABP during the study of autoregulation. Differences were found to be significant at the 0.05 level.
Because of differences in baseline CBF between the individual animals in each group, the CBF values in each animal were normalized to a percentage of baseline CBF values (CBF%), which were used to plot CBF/CPP curves in every single rat. A computer program was used to evaluate CBF autoregulation. This program repetitively fits two regression lines to the pooled CBF%/CPP values in each group: one sloped regression line through the CBF%/CPP points below a given CPP value, and one horizontal regression line through the CBF%/CPP above the given CPP value (Schmidt et al., 1990). One set of these regression lines was calculated for every 1-mm Hg increment of CPP, and the corresponding sum of squares was calculated. The pair of regression lines yielding the minimum sum of squares was chosen as the autoregulation curve for the group. In addition, a single linear regression line was fitted through the pooled CBF%/CPP values in each group. The sum of squares of this line was calculated and compared with the aforementioned sum of squares obtained by the two regression lines. The CBF autoregulation was defined intact only if all of the following criteria were fulfilled:
The minimum sum of squares obtained by the two regression lines is less than the sum of squares obtained by the single regression line. The lower limit of autoregulation (LL) identified by the program is physiologically acceptable, with a SE of less than 25% of the CPP value of LL. The lowest CPP used in the study exceeds the identified LL by at least 10 mm Hg.
RESULTS
Baseline values
The baseline physiologic state in all eight subgroups is shown in Table 1. As seen, ICP was significantly higher in the subgroups of SAH rats (P < 0.05) but was not different within either the subgroups of SAH rats or the controls. There were no differences concerning CBF between the subgroups of SAH rats and controls, and only small differences were seen between the groups concerning MABP, PaCO2, PaO2, and arterial pH.
Mean baseline CBF and physiologic state in the 4 subgroups of controls and the 4 subgroups of SAH rats before the manipulation of PaCO2
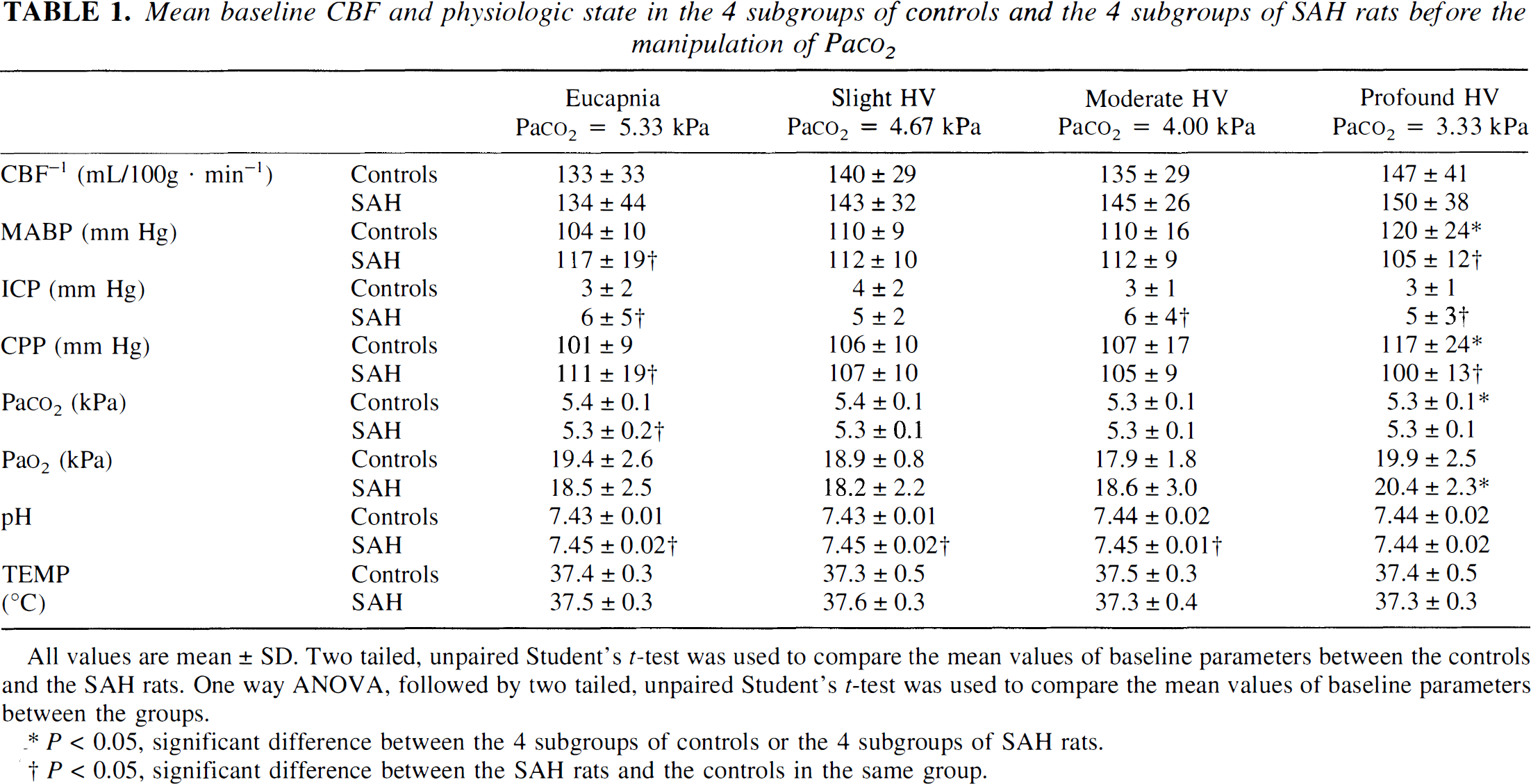
All values are mean ± SD. Two tailed, unpaired Student's t-test was used to compare the mean values of baseline parameters between the controls and the SAH rats. One way ANOVA, followed by two tailed, unpaired Student's t-test was used to compare the mean values of baseline parameters between the groups.
P < 0.05, significant difference between the 4 subgroups of controls or the 4 subgroups of SAH rats.
P < 0.05, significant difference between the SAH rats and the controls in the same group.
However, none of the observed differences in baseline values is assumed to influence the autoregulatory function either in the controls or in the SAH rats. Acute elevation of ICP to at least 50 mm Hg does not abolish autoregulatory function; therefore, an elevation of 2 to 3 mm Hg in ICP per se is assumed not to influence the autoregulatory function in the SAH rats (Hauerberg et al., 1998). The observed variation in MABP, which was within 15% in the controls and within 10% in the SAH rats, may influence the LL but not the function of CBF autoregulation (Paulson et al., 1990). The PaCO2 was maintained within the predetermined eucapnic range, and PaO2 was kept above the ischemic level in both controls and SAH rats, although minor differences were observed. Arterial pH changes appear not to influence CBF at a constant PaCO2 level (Lassen, 1968); therefore, the minor differences in arterial pH between the controls and the SAH rats also are assumed not to influence CBF autoregulatory function.
Study of autoregulation
The physiologic condition in the control and SAH subgroups during the study of autoregulation is shown in Tables 2 and 3, respectively. Throughout the studies, PaCO2 was maintained constant within the predetermined range, and PaO2 was maintained above the ischemic level. Arterial pH also was kept constant during the study except at low CPP values, where a decline in arterial pH was observed in the controls (P < 0.001) and in the SAH rats (P < 0.05). However, since the decline in arterial pH was observed only at the CPP values below the LL and since arterial pH changes appear not to influence CBF at a constant PaCO2 level (Lassen, 1968), the decline in arterial pH probably does not influence the CBF autoregulation curves. Thus, it is possible to evaluate the CBF autoregulation in the controls and SAH rats.
Physiologic state during the study of autoregulation in each 20 mm Hg change of CPP in the 4 subgroups of controls
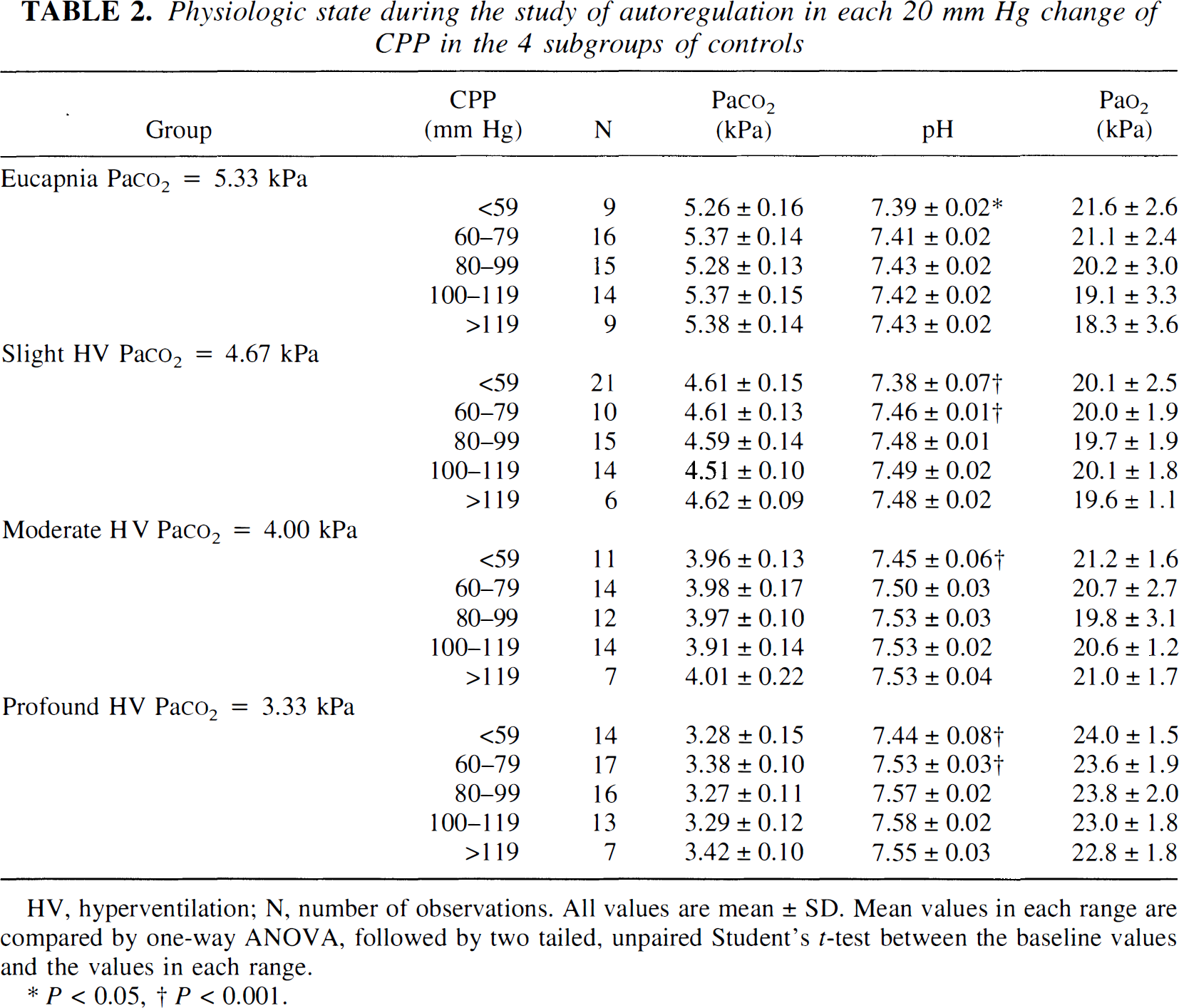
HV, hyperventilation; N, number of observations. All values are mean ± SD. Mean values in each range are compared by one-way ANOVA, followed by two tailed, unpaired Student's t-test between the baseline values and the values in each range.
P < 0.05.
P < 0.001.
Physiologic state during the study of autoregulation in each 20 mm Hg change of CPP in the 4 subgroups of SAH rats
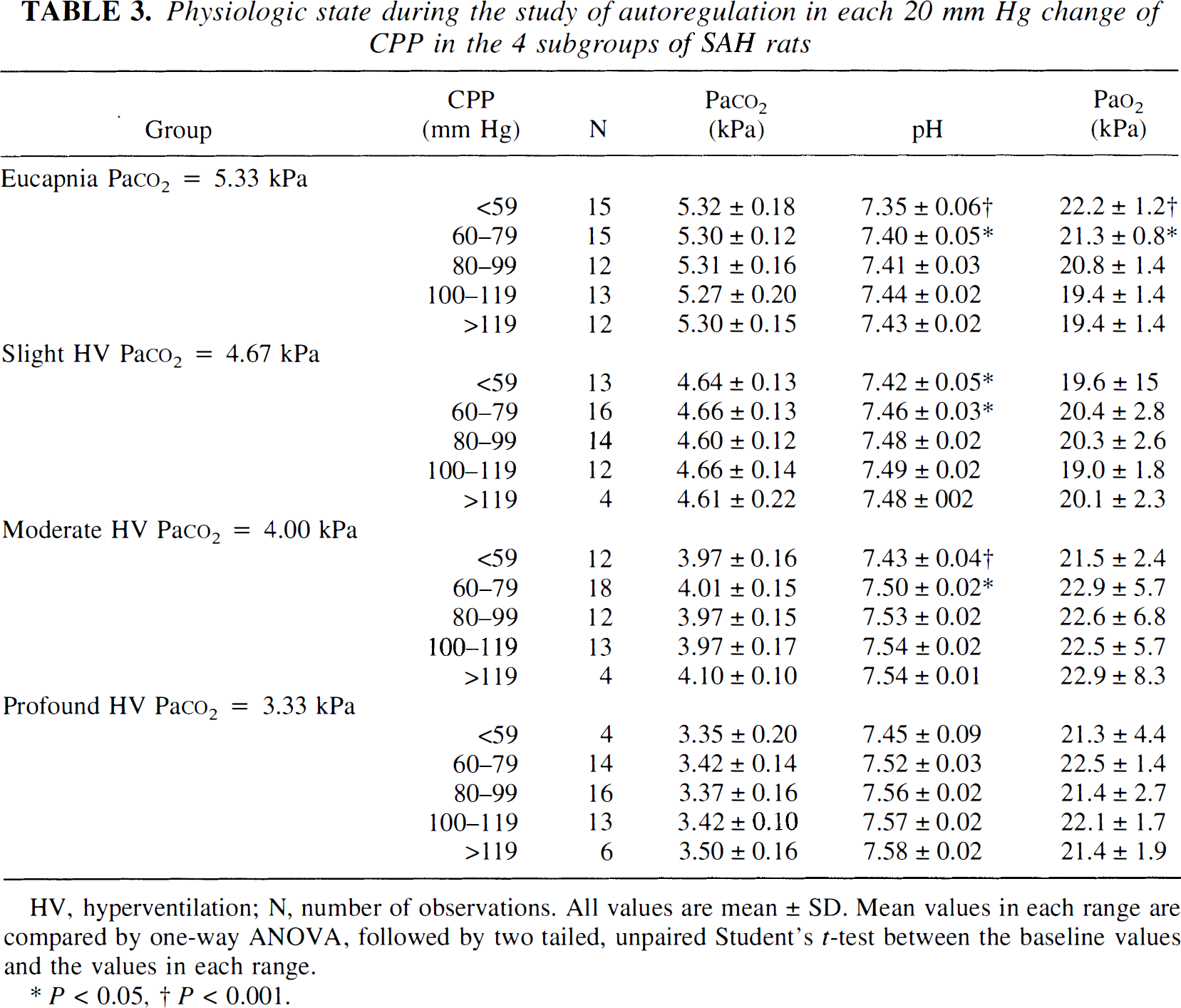
HV, hyperventilation; N, number of observations. All values are mean ± SD. Mean values in each range are compared by one-way ANOVA, followed by two tailed, unpaired Student's t-test between the baseline values and the values in each range.
P < 0.05.
P < 0.001.
All 64 individual autoregulation curves (CBF versus CPP in percentage of baseline value) are presented in Fig. 1. For each group, a mean autoregulation curve was calculated from one or two regression lines, as described in the section of data analysis. The results are shown in Fig. 2.
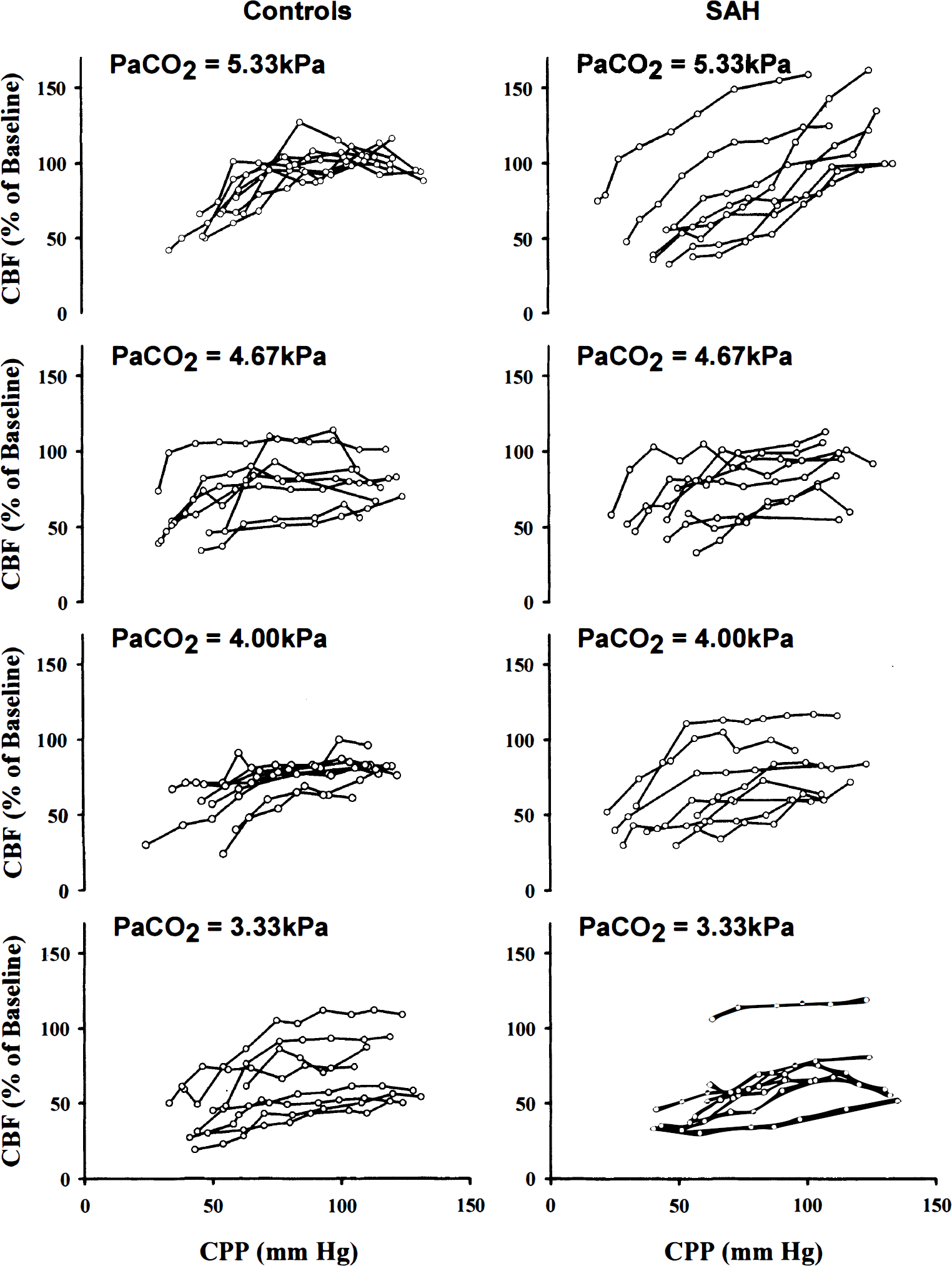
CBF autoregulation curves from each rat in each subgroup of the controls and the SAH rats.
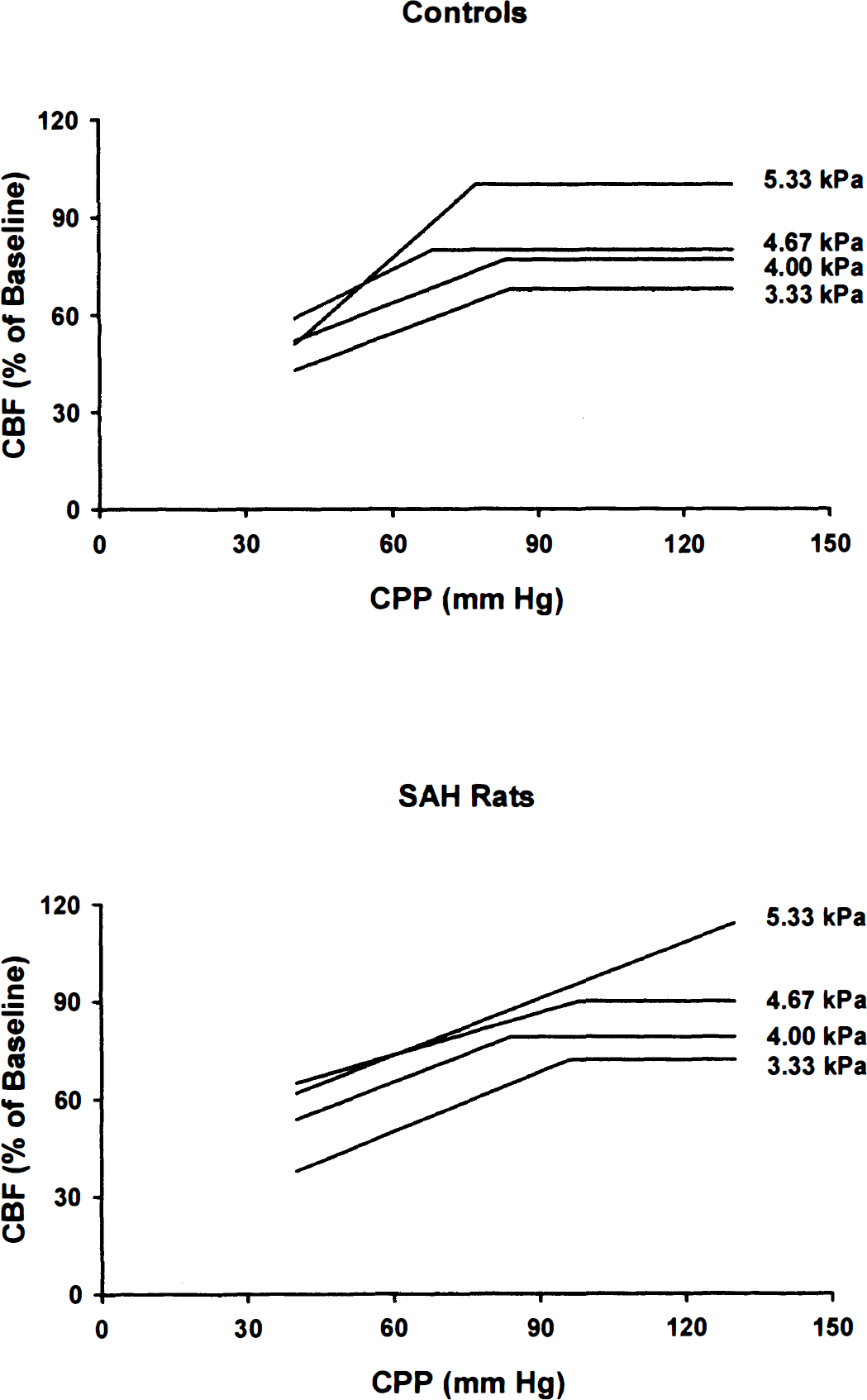
Mean CBF autoregulation curves from each subgroup of the controls and the SAH rats.
In agreement with previous studies, each autoregulation curve in the normoventilated controls consists of two parts (Hauerberg et al., 1993; Rasmussen et al., 1992; Paulson et al., 1990; Hauerberg et al., 1998): (1) a plateau part over a threshold CPP (equal to LL), where CBF is independent of variation in CPP; and (2) a sloped part below the threshold CPP, where CBF declines with decreased CPP. The LL was found at CPP = 77 mm Hg (Table 4).
Lower limits and CBF plateaus in controls and SAH rats during eucapnia and different levels of hyperventilation
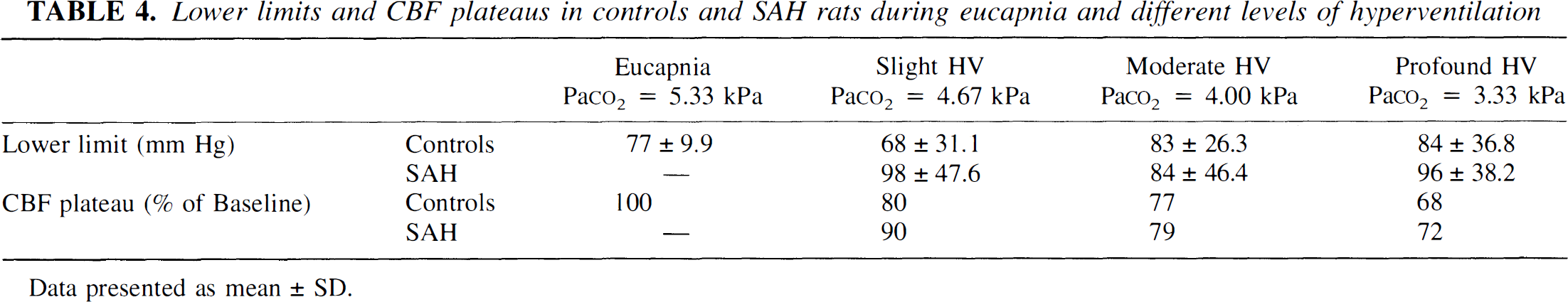
Data presented as mean ± SD.
Similar autoregulation curves were found both in the slightly, moderately, and profoundly hyperventilated subgroups of controls. However, absolute values of CBF decreased in these groups, resulting in CBF plateaus at 80%, 77%, and 68%, respectively, of the eucapnic baseline CBF, which also indicates a preserved CO2 reactivity in the controls. The LL was found when CPP equaled 68, 83, and 84 mm Hg, respectively. The difference between the LL was not significant (Table 4).
In the normoventilated SAH rats, a single linear regression line best described the relationship between CBF and CPP. The CBF in these rats passively follows the changes in CPP. A plateau of CBF could not be found, indicating severely disturbed CBF autoregulation.
However, in the slightly hyperventilated subgroup, seven of the eight autoregulation curves were normalized with a CBF plateau, suggesting a restoration of the autoregulatory function. The one rat in this subgroup that showed a single linear relationship between CBF and CPP was excluded in the calculation of the mean autoregulation curve shown in Fig. 2. The CBF plateau value in this subgroup then was found at 90% of the eucapnic baseline CBF, and the LL was located at CPP = 98 mm Hg (Table 4).
In the moderately or profoundly hyperventilated SAH subgroups, a normal shape of the autoregulation curve was found in every rat. All autoregulation curves in these subgroups included a CBF plateau. The calculated CBF plateau values were at 79% and 72% of the eucapnic baseline CBF, respectively. The LL was found at CPP = 84 mm Hg and CPP = 96 mm Hg, respectively. The difference between the LL of the three hyperventilated SAH subgroups was not significant (Table 4).
DISCUSSION
Dysfunction of CBF autoregulation after SAH is a well-documented phenomenon, both in patients in bad clinical condition as well as in animal models (Voldby, 1988; Mendelow, 1988; Paulson et al., 1972; Delgado et al., 1985; Delgado et al., 1986; Rasmussen et al., 1992; Jakubowski et al., 1982; Kamiya et al., 1983; Dernbach et al., 1988; Voldby et al., 1985; Boisvert et al., 1979; Mendelow et al., 1981). A disturbed CBF autoregulation can be restored by marked hyperventilation (Paulson et al., 1972; Hauerberg et al., 1993). Paulson et al. first reported in six patients with ischemic stroke or intracranial tumor that a disturbed autoregulation can be reestablished by hyperventilation to a PaCO2 between 2.9 and 4.1 kPa (22 to 31 mm Hg) (Paulson et al., 1972). This finding was verified experimentally in SAH rats, where the disturbed autoregulatory function was restored by hyperventilation to a PaCO2 between 3.3 and 4.0 kPa (25 to 30 mm Hg) (Hauerberg et al., 1993). In accordance with those findings, the current study shows that the CBF autoregulation was severely disturbed in the normoventilated SAH rats but was intact in the profoundly hyperventilated SAH rats.
Because CO2 reactivity normally is preserved after SAH (Kamiya et al., 1983; Dernbach et al., 1988; Voldby et al., 1985; Boisvert et al., 1979, profound hyperventilation results in marked decrease in CBF, which amounted to 30% in the current study and 42% in a previous study (Hauerberg et al., 1993)). This pronounced decline in CBF might exacerbate the existing or borderline cerebral ischemia after SAH, and thus may enhance rather than reduce secondary ischemic injury after SAH (Darby et al., 1988; Stringer et al., 1993; Muizelaar et al. 1991).
The important observation in the current study is, therefore, the intact CBF autoregulation found in seven of eight slightly and in all eight moderately hyperventilated SAH rats. This observation shows that the disturbed CBF autoregulation after SAH can be restored without profound reduction of PaCO2. At these PaCO2 levels, CBF is reduced by only 10% to 20%, which may have significance in reducing the risk of hyperventilation-related cerebral ischemia.
Although CBF autoregulation is a much studied phenomenon, the nature of this physiologic regulation remains unclear. It has been shown that cerebral extracellular pH is of major importance for the regulation of the tone of the cerebral resistance vessels and thus for CBF and CBF autoregulation (Paulson et al., 1972, 1990; Jakubowski et al., 1982; Wahl et al., 1970; Reivich, 1964). In patients with aneurysmal SAH, Voldby et al. (1985) have found a correlation between defective autoregulation and increased value of CSF lactate. This suggests that autoregulation is dependent on pH in the arteriolar wall or the perivascular space in the brain. By hyperventilation arterial pH is increased, which counteracts a possible, slight cerebral acidosis. This could be part of the mechanism responsible for the reestablishment of autoregulation (Paulson et al., 1972, 1990; Hauerberg et al., 1993). However, this theory needs to be further clarified by studying cerebral vasoreactivity during pH variations in cerebral perivascular space or brain parenchyma.
Compared with CBF autoregulation, CO2 reactivity seems more robust after SAH (Kamiya et al., 1983; Dernbach et al., 1988; Voldby et al., 1985; Boisvert et al., 1979. In the current study, mean CO2 reactivity in the SAH rats appears to be similar to the controls, since the mean CBF plateau value is comparable between the SAH subgroups and the controls at each PaCO2 level. The CBF-reducing effect of hyperventilation depends on an intact CO2 reactivity, which thereby determines the CBF plateau (Paulson et al., 1990). However, the restoration of the autoregulation after SAH seems not to be dependent on an intact CO2 reactivity, since CBF autoregulation also is restored in the rats with a sparse CO2 reactivity (Fig. 1). Furthermore, in the one slightly hyperventilated SAH rat where CBF autoregulation was not restored, CO2 reactivity appeared to be normal. Thus, although CBF autoregulation can be reestablished by hyperventilation, CBF autoregulation and CO2 reactivity appear to be two independent reactive functions of the cerebral vascular system.
On the other hand, interactions between CBF autoregulation and PaCO2 have been demonstrated previously (Paulson et al., 1990). Not only the level of the CBF plateau, but also the length of the CBF plateau, is dependent on the actual PaCO2 level (Paulson et al., 1990). Hypocapnia induces constriction in the cerebral resistance vessels, which improves the dilatory capacity at the LL and increases resistance at the upper limit. Therefore, hypocapnia with both a downward shift of LL and an upward shift of upper limit widens the plateau of the autoregulation curve (Paulson et al., 1990). In this study, however, the LL are not different between the PaCO2 levels in the controls or in the SAH rats. These results can be partly explained by the large interindividual variations in CO2 reactivity, which produce a considerable SD in the calculations of the mean LL. However, the mathematic method used to determine the LL also may have an impact on the results. As seen especially in the SAH rats, there is a significant discrepancy between the calculated and the visually detected LL (Fig. 1). In the slightly hyperventilated SAH group, for instance, a LL between 50 and 70 mm Hg would be expected from a visual point of view instead of the calculated 98 mm Hg. These differences, however, do not influence the main conclusion of the study.
CONCLUSION
It has been generally recommended but so far not substantially documented that hyperventilation to a PaCO2 of approximately 4.00 kPa (30 mm Hg) is appropriate both in optimizing the beneficial and in minimizing the detrimental effects of hyperventilation. The current results are the first supportive experimental evidence indicating that this PaCO2 level is sufficient for the restoration of impaired CBF autoregulation after SAH. However, there is still a need for clinical investigations.
Footnotes
Abbreviations used
Acknowledgements
The authors thank professor Gitte Moos Knudsen for her critical review and Mrs. Yan Cai for her assistance in data processing and manuscript preparation.
