Abstract
Ischemic preconditioning models have been characterized in brain, heart, and other tissues, and previous pharmacologic studies have suggested an involvement of adenosine and ATP dependent potassium (KATP) channels in such tolerance phenomena. This question was reexamined in a reproducible gerbil model in which the duration of ischemic depolarization defined the severity of preconditioning and test insults. Agents studied were glibenclamide, a blocker of KATP channels; 1,3-dipropyl-8-cyclopentylxanthine (DPCPX), an adenosine A1 receptor antagonist; and N6-cyclopentyladenosine (CPA), an A1 agonist. Intraventricular glibenclamide injections aggravated neuron damage after brief priming insults, in parallel with a dose-dependent prolongation of ischemic depolarization. However, the depolarization thresholds for ischemic neuronal injury were identical in vehicle- and glibenclamide-treated animals, and glibenclamide did not affect preconditioning when equivalent insult severity was maintained during priming insults. Neither DPCPX nor CPA had any effect on the onset or duration of depolarization after intraperitoneal injection in this model, and neither drug affected neuron damage. In the case of CPA, it was necessary to maintain temperature for 4 to 6 hours of recirculation to avoid significant confounding hypothermia. These results fail to support a direct involvement of A1 receptors or KATP channels during early stages in the development of ischemic tolerance in vivo, and emphasize the need for robust, well-controlled, and quantitative models in such studies.
Keywords
Preconditioning by brief global ischemia activates unknown endogenous protective mechanisms that render neurons resistant to a subsequent, more sustained ischemic insult, as recently reviewed (Dirnagl et al., 2003; Kirino, 2002). Early studies documented the general features of induced tolerance after global ischemia in gerbils (Kirino et al., 1991; Kitagawa et al., 1990) and rats (Liu et al., 1992; Shamloo and Wieloch, 1999). Recent reports have shown the improved predictability of preconditioning by recording ischemic depolarization as a quantitative index of insult severity (Abe and Nowak, 1996, 2004; Nishino and Nowak, 2004).
Among the diverse candidate mechanisms considered as mediators of preconditioning in brain, considerable attention has focused on activation of adenosine A1 receptors and ATP-dependent potassium (KATP) channels. This focus was based on initial pharmacologic results in cardiac preconditioning suggesting the plausible scenario that priming ischemia activated A1 receptors, which then via a protein kinase C–mediated mechanism opened KATP channels, in turn decreasing myocardial damage after a subsequent ischemic insult (Parratt and Kane, 1994). Somewhat analogous studies in brain suggested that both the KATP channel blocker glibenclamide and the A1 receptor antagonist DPCPX (1,3-dipropyl-8-cyclopentylxanthine) could abolish the protective effect of preconditioning in a rat global ischemia model when administered during the preconditioning insult (Heurteaux et al., 1995). Additionally, the adenosine receptor agonist CPA (N6-cyclopentyladenosine) was reported to protect CA1 neurons against transient ischemia (Heurteaux et al., 1995; Von Lubitz et al., 1994), while adenosine antagonists conversely worsened damage (Von Lubitz et al., 1994). Corresponding results have been reported in a model of acute preconditioning in brain slices (Pérez-Pinzón and Born 1999; Pérez-Pinzón et al., 1996).
There is no doubt that modulation of KATP channels can directly impact insult severity after ischemia. Channel openers limit depolarization during metabolic insults (Ben-Ari et al., 1990; Riepe et al., 1992), reduce glutamate release (Zini et al., 1993), and are cytoprotective (Abele and Miller, 1990; Heurteaux et al., 1993; Lauritzen et al., 1997; Reshef et al., 1998; Wind et al., 1997). Conversely, channel blockers facilitate depolarization (Mourre et al., 1989; Murphy and Greenfield, 1991; Riepe et al., 1992), increase glutamate release (Zini et al., 1993), and antagonize protective effects of channel openers (Abele and Miller, 1990; Lauritzen et al., 1997; Wind et al., 1997). However, interpretation of in vivo pharmacologic results is not always straightforward, and this presents a particular problem when dealing with the very short ischemic insults used in preconditioning paradigms. For example, controls for possible injurious effects of agents administered during a priming insult were not included in previous in vivo preconditioning studies in brain (Heurteaux et al., 1995). The aim of these experiments, therefore, was to reexamine the impact of A1 receptor and KATP channel pharmacology on ischemic injury and preconditioning in a quantitative depolarization-monitored gerbil model.
MATERIALS AND METHODS
Ischemia model
Adult female Mongolian gerbils (50–70 g, Charles River Laboratories, West Brookfield, MA, U.S.A.) were subjected to single or repeated intervals of transient global ischemia according to a protocol approved by the Animal Care and Use Committee, University of Tennessee, as previously described (Abe and Nowak, 2004). Animals were anesthetized with 2% halothane in 30% O2 and 70% N2. Common carotid arteries were isolated and looped with 4–0 silk suture and the animal was placed in a stereotaxic frame. Rectal and epidural temperatures were thereafter maintained at 37°C using independent heating lamps. Burr holes were drilled and glass electrodes filled with 2-mol/L NaCl (5–20 MΩ) were positioned bilaterally in the hippocampal CA1 subfields, 2 mm posterior to bregma, 2 mm lateral to the midline, and 1.9 mm ventral to the cortical surface (Loskota et al., 1974). Ground-referenced extracellular voltage was continuously recorded using a DC-coupled amplifier (S-7100A, World Precision Instruments, Inc., New Haven, CT, U.S.A.) and a data-acquisition package (SuperScope 2; GW Instruments, Inc., Somerville, MA, U.S.A.). Ischemia was produced by applying tension to the suture loops, which were then cut and removed to restore blood flow. Electrodes were withdrawn, the animal was released from the stereotaxic frame, and scalp and neck wounds were sutured. Anesthesia was maintained for 1 hour of recirculation, during which time the rectal temperature was continuously controlled at 37°C to avoid the spontaneous hyperthermia that can occur during recirculation in this model (Abe and Nowak, 2000; Kuroiwa et al., 1990). To produce a second ischemic insult, animals were reanesthetized 2 days later and these procedures were repeated.
The duration of ischemic depolarization was determined from the recorded tracing, defined as the interval between the midpoints of the depolarization and repolarization transients (Fig. 1A). Preliminary studies identified optimal preconditioning after insults producing 2.5- to 3.5-minute depolarizations, entirely consistent with previous results (Abe and Nowak, 2004; Nishino and Nowak, 2004). Previous studies also established that sham electrode insertions fail to induce either gene expression changes or preconditioning effects (Abe and Nowak, 2004), confirming that trauma-induced spreading depression is successfully avoided during these experimental procedures.
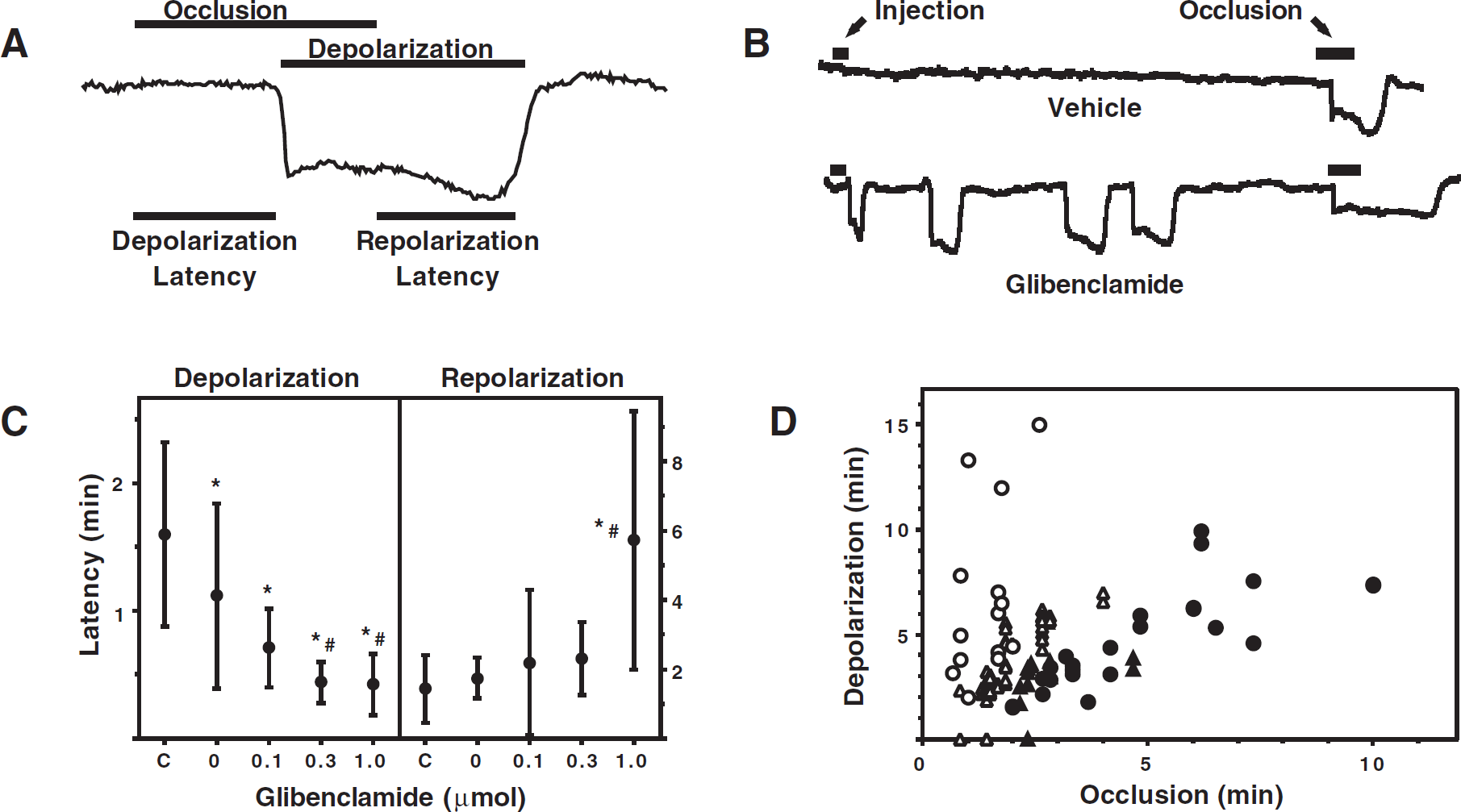
Characteristics of ischemic depolarization and effects of glibenclamide. (
Histologic evaluation of neuronal damage
Animals were perfused transcardially with formalin, glacial acetic acid, and methanol (1:1:8, FAM) at an interval of 5 days after a test ischemic insult. The skull was kept in FAM overnight and the brain was then removed and stored in the same fixative. Blocks containing dorsal hippocampus were dehydrated and embedded in paraffin. Sections were cut at 8 μm and stained with hematoxylin and eosin. Neuron density in CA1 was determined by counting of a calibrated 400-μm field under a 25x objective and the results expressed as the number of intact pyramidal cells per 1 mm length of CA1. Injury thresholds in the various treatment groups were compared as plots of surviving cell number versus occlusion or depolarization time, as indicated for individual experiments.
Drug administration
Gibenclamide, DPCPX, and CPA were obtained from Research Biochemicals International (Natick, MA, U.S.A.). Glibenclamide was dissolved in dimethylsulfoxide (DMSO) at concentrations of 20, 60, or 200 mmol/L, yielding doses of 0.1, 0.3, or 1.0 μmol per 5-μL injection. Intraventricular injections were administered 20 to 30 minutes before occlusion using a glass pipette with a 50-μm tip diameter inserted into the left lateral ventricle (1 mm anterior to bregma, 1.25 mm lateral to the midline, and 2.4 mm ventral to the cortical surface). DPCPX and CPA were dissolved at a concentration of 0.6 mg/mL in DMSO and injected intraperitoneally at a dose of 1 mg/kg 15 minutes before occlusion.
Statistical analyses
Group values are given as mean ± SD. Statistical comparisons were made by using analysis of variance and Scheffé F test, with P>0.05 considered significant.
RESULTS
Effects of glibenclamide on ischemic depolarization
The characteristics of ischemic depolarization in gerbil hippocampus and the relevant consequences of glibenclamide administration are illustrated in Fig. 1. In the absence of drug treatment, transient carotid artery occlusion resulted in a characteristically delayed depolarization, followed by a comparably delayed repolarization upon restoration of blood flow (Fig. 1A). In the absence of ischemia, glibenclamide induced occasional spontaneous depolarizations (Fig. 1B, Table 1), which were never observed after vehicle injection. These occurred in approximately 25% of hippocampi, increasing to 75% ipsilateral to the highest dose, after which multiple depolarizations also became more frequent (Table 1). More striking and consistent were the effects of glibenclamide on the kinetics of ischemic depolarization during brief intervals of occlusion. Intraventricular injection of DMSO alone slightly reduced the latency to depolarization after occlusion from 96 ± 43 seconds to 67 ± 44 seconds, with further dose-dependent reductions to 43 ± 18 seconds, 26 ± 10 seconds, and 25 ± 14 seconds after administration of 0.1, 0.3, and 1 μmol glibenclamide, respectively (Fig. 1C). There was also a marked delay in repolarization after glibenclamide treatment at the highest dose tested. As a consequence of these effects, much longer intervals of depolarization occurred in glibenclamide-treated hippocampi after occlusions of a given duration (Fig. 1D). The increased duration of ischemic depolarization after short periods of occlusion was paralleled by a decrease in the threshold for neuron damage determined as a function of occlusion duration, while the depolarization threshold for CA1 injury was identical in control and glibenclamide-treated groups (Fig. 2A).
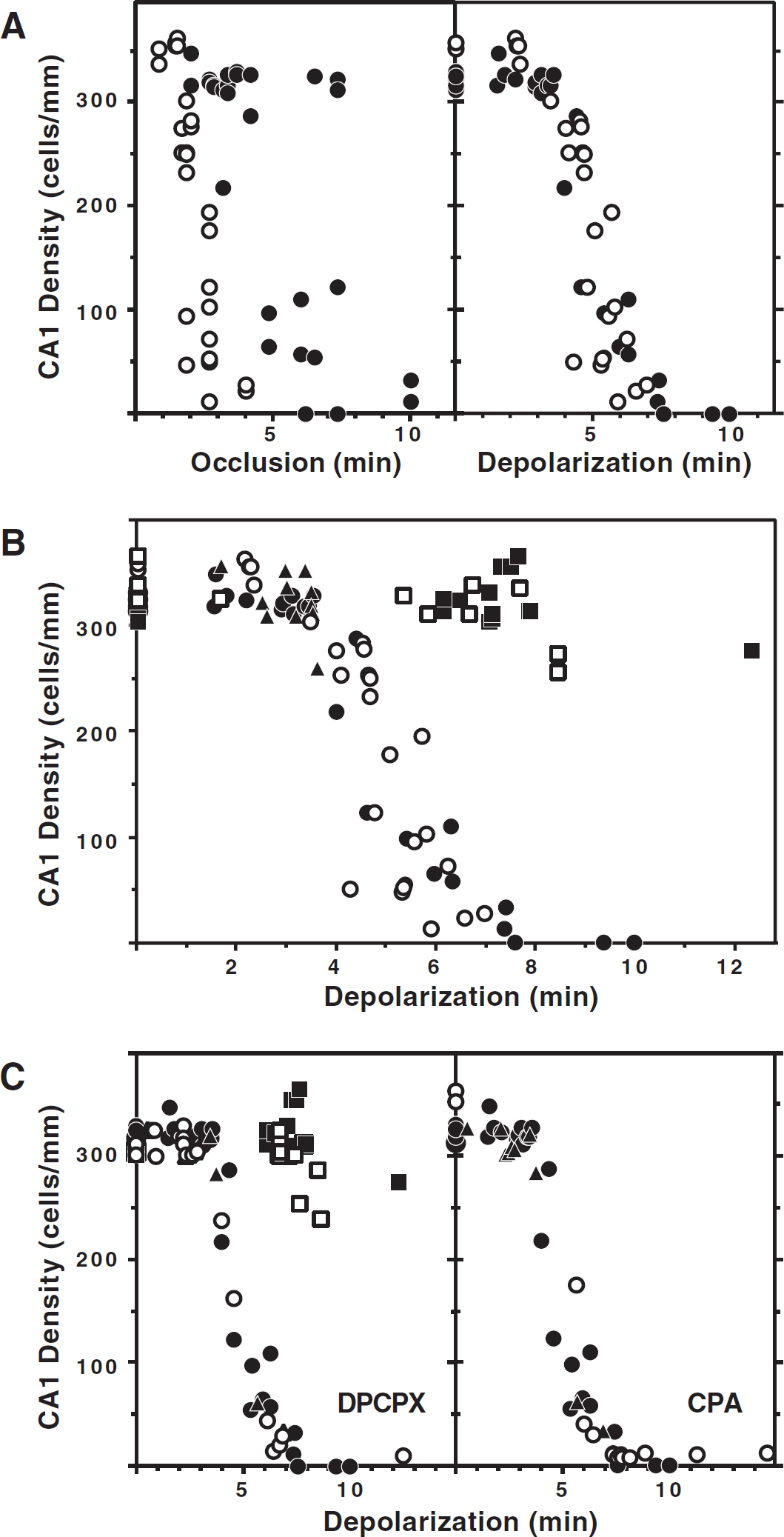
Effects of pharmacologic interventions on ischemic injury and preconditioning. (
Hippocampal depolarizations after glibenclamide administration
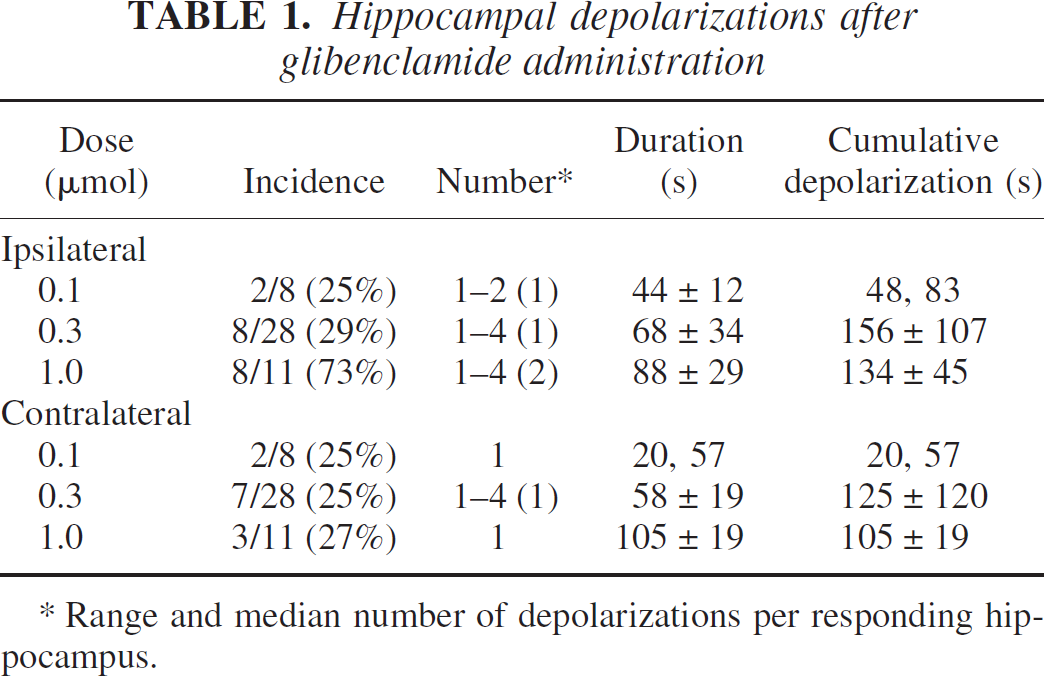
* Range and median number of depolarizations per responding hippocampus.
Based on criteria established in previous studies, untreated and glibenclamide-injected animals were subjected to ischemia yielding hippocampal depolarization within the limits of 2.5 to 3.5 minutes that result in optimal preconditioning in this model. Gerbils were then subjected 2 days later to test insults with depolarization durations sufficient to produce CA1 loss in naive animals. Hippocampi of animals receiving 0.3 μmol glibenclamide before the priming insult showed the same preconditioning effect observed in gerbils that did not receive the drug (Fig. 2B).
Effects of A1 receptor agonists and antagonists
Mean latency and duration of ischemic depolarization did not differ in animals treated with the A1 receptor antagonist, DPCPX, relative to either control or vehicle-injected animals (data not shown). Slight neuron damage was consistently observed in hippocampi after 4-minute depolarizations, and insults of 7 minutes or longer produced maximal CA1 loss 5 days after ischemia, independent of DPCPX injection (Fig. 2C). Furthermore, animals injected with DPCPX before priming insults showed unaltered protection of CA1 neurons after test insults of up to 7 minutes in duration, although there was perhaps some trend toward attenuated preconditioning after more severe insults. Therefore, DPCPX injection had little impact on either the depolarization dependence of neuron damage or the protective effects of priming ischemia.
Mean depolarization latency and duration in animals treated with CPA did not differ from those of untreated or vehicle-injected animals (data not shown). After release from anesthesia, hypothermia and hypokinesia were noted in all animals with CPA injection, requiring 4 to 6 hours of temperature control before homeostasis was regained. Under such conditions of temperature control, histologic examination 5 days after ischemia revealed severe CA1 damage after depolarizations of 5 minutes or longer in all treatment groups (Fig. 2C), failing to identify a neuroprotective effect of CPA.
DISCUSSION
These results show that glibenclamide increases the duration of ischemic depolarization in gerbil hippocampus in a dose-dependent manner and thereby worsens damage after brief occlusions (Figs. 1 and 2A). This finding implies that a previously inferred blockade of preconditioning by glibenclamide in rats (Heurteaux et al., 1995) was derived instead from overt damage that occurred as a result of the intended preconditioning treatment. Ischemic preconditioning was not significantly altered by glibenclamide when consistent insult severity was maintained, as defined by the duration of the priming depolarization (Figs. 2A and 2B). Explanations for the absence of treatment effects for DPCPX and CPA (Fig. 2C) are less definitive, but will be considered in turn.
Effects of glibenclamide on ischemic depolarization and the response to preconditioning
In vivo physiologic consequences of glibenclamide treatment in ischemia have not been extensively investigated. In one other study glibenclamide shortened latency to ischemic depolarization in rat cortex after cardiac arrest, although this was attributed to systemic hypoglycemic effects because it was not observed when the drug was administered by superfusion (Xie et al., 1995). However, the drug load presented to the brain surface for 60 minutes in that study was 100-fold lower than the intermediate intraventricular dose (0.3 μmol) administered acutely in the present experiments. DMSO slightly but significantly reduced average depolarization latency in animals receiving the vehicle alone, which also may have contributed to the effect seen in the present study. An impact on in vivo preconditioning had been previously described after infusion of 1.0 μmol glibenclamide in DMSO (Heurteaux et al., 1995), a dose that dramatically prolonged depolarization in the current experiments (Fig. 1D). This marked amplification of the effect occurred as a result of delayed repolarization, which was only observed at the higher glibenclamide dose (Fig. 1C). Subsequent injury threshold and preconditioning studies reported here (Figs. 2A and 2B) were performed with only 0.3 μmol glibenclamide because of the difficulty in producing controlled depolarizations of short duration at a higher dose.
Most previous studies of glibenclamide's effects in brain have used in vitro preparations. Although producing no effects in control rat hippocampal slices, glibenclamide increased synaptic activity and blunted early hyperpolarizing responses to anoxia of CA3 neurons, which are particularly rich in sulfonylurea binding sites (Mourre et al., 1989). Comparable glibenclamide effects were reported for CA1 neurons in slices challenged with the metabolic inhibitor 3-NP (Riepe et al., 1992). Glibenclamide reduced K+ efflux during hypoxic exposure of hypoglossal nucleus in rat brain slices, increased the magnitude of depolarization, and prevented repolarization after sufficiently prolonged anoxia (Jiang et al., 1992). The present in vivo results in gerbil are entirely consistent with these in vitro observations. Although it is possible that rat and gerbil could differ somewhat in the precise dose dependence of glibenclamide's effects, the available data overwhelmingly support the conclusion that potentiation of ischemic injury by this agent similarly accounts for the cell loss observed after “preconditioning” insults to pretreated rats (Heurteaux et al., 1995). Parallel effects of glibenclamide have sometimes been noted in the cardiac literature, as evidenced by accelerated ATP depletion (Fralix et al., 1993) and even overt infarct expansion (Thornton et al., 1993). Experiments to rigorously exclude glibenclamide exacerbation of injury have been notably lacking in some other preconditioning studies involving in vivo focal brain ischemia (Xiong et al., 2003) and neuron cultures (Reshef et al., 1998).
Although not affecting the interpretation of the results presented here, preconditioning studies in heart have more recently focused on the role of mitochondrial rather than plasma membrane KATP channels (Cohen et al., 2000; Garlid et al., 1997), with a parallel literature evolving in brain (Horiguchi et al., 2003; Liu et al., 2002). Interestingly, recent results in a mouse bilateral carotid artery occlusion model have also demonstrated a worsening of damage after otherwise protective priming insults in animals pretreated with 5-hydroxydecanoate (Muñoz et al., 2003). This agent reportedly blocks preconditioning in a range of models (Fryer et al., 2000), and shows considerable specificity as an antagonist of mitochondrial KATP channels (Garlid et al., 1997). Comparable effects were obtained in control mice and those lacking SUR1 regulatory sulfonylurea receptor channel components, further suggesting a site of action independent of cell surface KATP channels (Muñoz et al., 2003). Therefore, the potentiation of injury after priming insults must be considered as a potential confound for this class of agents as well.
Recent results indicate the involvement of hypothalamic KATP channels in glucose homeostasis, separate from the role of pancreatic KATP channels in regulation of circulating glucose levels, as shown by an absence of functional glucose-responsive neurons in mice lacking the major pore-forming subunit Kir6.2 (Miki et al., 2001). These animals exhibited impaired glucagon release and slower recovery of glucose levels after hypoglycemic challenge, although the resting glucose level was not altered. Glucose levels were not measured in the present study. However, the intermediate decrease in mean depolarization latency after low dose glibenclamide (Fig. 1C) primarily reflected changes restricted to ipsilateral hippocampus (data not shown). The rapid onset of spontaneous depolarizations after intraventricular glibenclamide, and the predominantly ipsilateral response seen at the highest dose (Table 1), likewise argue against a contribution of systemic changes in glucose homeostasis to the observed effects on depolarization kinetics.
Absence of adenosine A1 agonist or antagonist effects
The present results appear to be at odds with the findings of a considerable body of early as well as recent studies noting an impact of adenosine pharmacology on ischemic injury (Ghiardi et al., 1999; Rudolphi et al., 1992; Von Lubitz 1999). For example, in contrast to previous data for acutely injected rats (Heurteaux et al., 1995) and gerbils (Von Lubitz et al., 1994), there was no evidence for any protective effect of CPA in the present study (Fig. 2C). The most straightforward explanation for this discrepancy would be inadequate temperature monitoring and control in the previous experiments. Adenosine agonists have long been known to produce dose-dependent hypothermic effects (Jonzon et al., 1986). Under the conditions of this study, temperature was routinely maintained under halothane anesthesia for 90 minutes of recirculation, but it was necessary to thereafter sustain temperature control for as long as 6 hours in CPA-treated animals to avoid profound cooling. Global ischemia models display striking temperature sensitivity during a substantial recirculation period (Colbourne and Corbett, 1995; Colbourne et al., 1997), and the confounding effects of hypothermia are well recognized for many proposed neuroprotective agents (Buchan and Pulsinelli, 1990; Nurse and Corbett, 1996). Furthermore, the occurrence of transient hyperthermia during recirculation is an established feature of the gerbil model under conditions of early release from anesthesia (Kuroiwa et al., 1990; Suga and Nowak, 1998), and this was intentionally eliminated by the design of the present studies. If such hyperthermia were present in untreated animals, substantial protection could be produced by agents capable of lowering temperature even if overt cooling were avoided by temperature control at 37°C. For example, the adenosine reuptake inhibitor propentofylline shortened the interval of severe hyperthermia after an ischemic insult 24 hours after drug administration in the gerbil, under which condition a potentiation of ischemic preconditioning was also reported (Kawahara et al., 1998).
Negative results with the selective A1 antagonist DPCPX (Fig. 2C) also disagree with several other studies. This and other adenosine antagonists were reported to worsen damage in gerbil hippocampus at high oral doses (Rudolphi et al., 1990). Subsequently it was reported that DPCPX at 1 mg/kg (intraperitoneally) effectively blocked protection when administered before a preconditioning insult in rats (Heurteaux et al., 1995), and equivalent results have been obtained in hippocampal slices (Pérez-Pinzón et al., 1996; Pugliese et al., 2003). The same treatment was also shown to attenuate preconditioning in the gerbil when administered 3 hours after the priming insult, and injury potentiation comparable to that noted above for glibenclamide was explicitly eliminated as a possible contributing effect (Hiraide et al., 2001). Moreover, DPCPX was recently found to exacerbate injury in association with accelerated depolarization in a fetal sheep asphyxia model (Hunter et al., 2003). It was therefore surprising that, in the present study, DPCPX pretreatment had no evident impact on ischemic depolarization, neuron damage, or preconditioning in the gerbil (Fig. 2C). More comprehensive dose–response and time course studies are required to determine whether there may be significant species differences in the pharmacology of adenosine receptors, or in the precise timing of their relevance to ischemic injury and preconditioning. It should be noted that in vivo studies with this agent have not included comprehensive monitoring of long-term temperature changes that could potentially impact outcome after subsequent test insults.
Final comments
Although these results provide direct evidence against an effect of glibenclamide pretreatment to block preconditioning, they do not formally rule out the participation of altered KATP channel function in neuroprotection. For example, preconditioned gerbil hippocampi show only slightly delayed depolarization during subsequent test insults (Abe and Nowak, 2004), whereas recent results indicate that this effect is much more prominent in the rat (M. Ueda and T. S. Nowak, Jr., unpublished observation, 2002). Such delays cannot account for the protection seen under conditions of controlled depolarization duration, but they clearly attenuate insult severity based on occlusion time, and persistent KATP channel potentiation subsequent to preconditioning would be one mechanism to explain such an effect. KATP channel openers confer protection in diverse models, that can be blocked by channel antagonists (Abele and Miller, 1990; Heurteaux et al., 1995; Reshef et al., 1998; Wind et al., 1997), and the present results further indicate that channel activity at the time of an insult is an important determinant of neuron vulnerability to depolarization and subsequent injury. The possible contributions of mitochondrial KATP channels to such effects in brain remain to be fully elucidated.
It should be emphasized that these results do not necessarily conflict with in vitro studies indicating involvement of adenosine receptors and KATP channels in acute preconditioning effects in tissue slices (Pérez-Pinzón and Born, 1999; Pérez-Pinzón et al., 1996; Pugliese et al., 2003). Such studies have typically evaluated short-term physiologic endpoints (Schurr et al., 1986), and lasting cytoprotection has not been obtained in brain after rapid preconditioning protocols (Pérez-Pinzón et al., 1997). Histopathologic protection is evident in organotypic hippocampal cultures at long intervals between priming and test insults comparable to those proven effective in vivo (Raval et al., 2003; Xu et al., 2002), although cumulative damage has also been noted under some conditions (Pringle et al., 1999), and effects of adenosine and KATP channel pharmacology have not yet been reported in such models. The distinction between physiologic and pathologic endpoints has also been noted as a variable in cardiac protection studies in vivo (Parratt and Kane, 1994), although histologically demonstrable myocardial preconditioning clearly develops and declines with a rapid time course (Murry et al., 1986). It would seem that heart is less subject to the injury-exacerbating effects noted for ischemic brain insults repeated at short intervals (Abe and Nowak, 2004; Kato et al., 1989; Tomida et al., 1987). The present findings provide further support for the mechanistic distinction of early and delayed preconditioning phenomena.
