Abstract
Stroke increases neurogenesis. The authors investigated whether neural stem cells or progenitor cells in the adult subventricular zone (SVZ) of rats contribute to stroke-induced increase in neurogenesis. After induction of stroke in rats, the numbers of cells immunoreactive to doublecortin, a marker for immature neurons, increased in the ipsilateral SVZ and striatum. Infusion of an antimitotic agent (cytosine-β-D-arabiofuranoside, Ara-C) onto the ipsilateral cortex eliminated more than 98% of actively proliferating cells in the SVZ and doublecortin-positive cells in the ipsilateral striatum. However, doublecortin-positive cells rapidly replenished after antimitotic agent depletion of actively proliferating cells. Depleting the numbers of actively proliferating cells in vivo had no effect on the numbers of neurospheres formed in vitro, yet the numbers of neurospheres derived from stroke rats significantly (P < 0.05) increased. Neurospheres derived from stroke rats self-renewed and differentiated into neurons and glia. In addition, doublecortin-positive cells generated in the SVZ migrated in a chainlike structure toward ischemic striatum. These findings indicate that in the adult stroke brain, increases in recruitment of neural stem cells contribute to stroke-induced neurogenesis, and that newly generated neurons migrate from the SVZ to the ischemic striatum.
Actively proliferating progenitor cells and relatively quiescent neural stem cells are present in the adult rodent subventricular zone (SVZ) (Morshead et al., 1994). Precursor cells isolated from the adult SVZ exhibit neural stem cell characteristics in vitro; they self-renew and are multipotential (Gritti et al., 1996;Morshead et al., 1994;Reynolds and Weiss, 1992a). Stroke enhances neurogenesis in the SVZ (Arvidsson et al., 2002;Iwai et al., 2003;Jin et al., 2001;Parent et al., 2002;Zhang et al., 2001). However, little is known whether neural stem cells or proliferating progenitor cells in the SVZ contribute to stroke-increased neurogenesis.
Neural stem cells in the SVZ give rise to neuroblasts that travel the rostral migratory stream to the olfactory bulb, where they differentiate into granule and periglomerular neurons throughout adult life (Garcia-Verdugo et al., 1998;Luskin, 1998;Morshead et al., 1994). Although the findings of previous studies have suggested that neuroblasts in the SVZ migrate to the striatum after stroke (Arvidsson et al., 2002;Zhang et al., 2002), these studies have not shown that newly generated neurons in the striatum are derived from the SVZ neuroblasts, or how these neuroblasts migrate toward the ischemic striatum.
Here, we show that stroke increases recruitment of neural stem cells in the SVZ. Furthermore, we provide the first direct evidence that neuroblasts in the SVZ migrate toward the ischemic boundary regions via chains.
MATERIALS AND METHODS
The Care of Experimental Animals Committee of Henry Ford Hospital approved all experimental procedures.
Animal model of stroke
Male Wistar rats weighing 320 to 380 g were used for all experiments. The middle cerebral artery was occluded by placement of an embolus at the artery's origin (Zhang et al., 1997).
Bromodeoxyuridine labeling
Bromodeoxyuridine, the thymidine analog that is incorporated into the DNA of dividing cells during S-phase, was used for mitotic labeling (Sigma Chemical, St. Louis, MS, U.S.A.). Stroke rats were intraperitoneally injected with bromodeoxyuridine (50 mg/kg) every 2 hours, three times starting 7 hours before the animals were killed.
Ara-C treatment
Cytosine-β-D-arabiofuranoside (Ara-C, 2%; Sigma-Aldrich, St. Louis, MS, U.S.A.) in vehicle (0.9%, saline) was continuously infused onto the surface of the right hemisphere of the rats for 7 days starting on day of middle cerebral artery occlusion with a mini-osmotic pump (model 2001; Alzet, Palo Alto, CA, U.S.A.) at rate of 1.0 μL/h (Doetsch et al., 1999). Cannulas were implanted on the brain surface at anterior/posterior 0, l, and 1.1 mm relative to bregma (Paxinos and Watson, 1986).
Immunohistochemistry
Coronal or sagittal sections (8 μm) were immunostained as previously described (Zhang et al., 2001) using the following primary antibodies: mouse antibromodeoxyuridine (1:100; Boehringer Mannheim, Indianapolis, IN, U.S.A.) as a marker for proliferating cells, goat antidoublecortin (1:200; Santa Cruz Biotechnology Inc., Santa Cruz, CA, U.S.A.) as a marker for migrating neuroblasts; mouse anti–βIII-tubulin (TuJ1, 1:200; Novus Biologicals, Inc., Littleton, CO, U.S.A.) as a marker for neuroblasts, rabbit anti—glial fibrillary acidic protein (1:200; Dako, Carpinteria, CA, U.S.A.) as a marker for astrocytes. Specimen were analyzed using light (Zeiss Axiophot) or confocal microscopy (1024; Bio-Rad, Cambridge, MA, U.S.A.)
Measurements of the number of bromodeoxyuridine-positive cells were performed on paraffin embedded sections (thickness of 6 μm), because focal cerebral ischemia resulted in cavitation in the ipsilateral hemisphere, which precludes cutting accurate thickness of vibratome sections (Zhang et al., 2001). Bromodeoxyuridine-immunostained sections were digitized under a ×60 objective (Olympus BX40) via the MicroComputer Imaging Device (Imaging Research, St. Catharines, Canada). Bromodeoxyuridine-positive nuclei were counted in the lateral ventricle wall. Every 40th coronal section was selected from each rat for a total seven sections between AP + 10.6 mm -genu corpus callosum and AP + 8.74 mm – anterior commissure crossing. Bromodeoxyuridine-positive nuclei in these areas are presented as the number of the cells per square millimeter (Zhang et al., 2001).
Neurosphere culture and differentiation
Cells from the SVZ were dissociated from the male Wistar rats (3–4 months) treated with Ara-C or saline 7 days after stroke, as previously reported (Chiasson et al., 1999;Morshead et al., 1994). The cells were cultured in the medium containing 20-ng/mL basic fibroblast growth factor and epidermal growth factor (EGF) at cell density of 10,000 cells/mL (Chiasson et al., 1999;Morshead et al., 1994). The total number of primary spheres that formed in each rat was counted after 7 days in vitro. Data are presented as the mean number of primary spheres formed per brain dissection. To generate the secondary spheres, a single primary sphere was mechanically dissociated to a single cell suspension and replated in growth medium. Neurospheres were cultured in differentiating medium for 7 days and then fixed and immunostained, as previously described (Zhang et al., 2001). The number of TuJ1-positive or glial fibrillary acidic protein—positive cells and the total diamidino-2-phenylindoladihydrochloride (DAPI) cell number per sphere were counted and the percentage of each cell type per sphere was determined.
Electron microscopy analysis
Semithin sections (1.5 μm) were stained with 1% toluidine blue. Ultrathin sections (0.05 μm) were analyzed under the EM 201 electron microscope (Philips Electronics Instrument Co., Netherlands). The semithin and ultrathin sections examined included the entire dorsoventral extent of the SVZ of the lateral ventricle. Serial transverse sections were examined to identify cellular types. Criteria used in the present study for identifying type A, B, and C cells were adapted from previous studies (Doetsch et al., 1997;Garcia-Verdugo et al., 1998). Type A cells are elongated with spindle-shaped nuclei, one or two processes, abundant lax chromatin, two to four small nucleoli, and a scant, dark cytoplasm containing many free ribosomes, a few short cisternae of rough endoplasmic reticulum, and many microtubules (Doetsch et al., 1997). Type B cells have irregular nuclei that frequently contain invaginations. Their cytoplasm is light and contains few free ribosomes, abundant intermediate filaments, and dense bodies (Doetsch et al., 1997). Type C cells are spherical and larger than type A cells. Their nuclei contain deep invaginations and mostly lax chromatin. Their cytoplasm contains a large Golgi apparatus and fewer free ribosomes than type A cells (Doetsch et al., 1997).
RESULTS
Stroke enhances recruitment of neural stem cells
To examine temporal profiles of proliferating cells in the SVZ after stroke, stroke rats were given bromodeoxyuridine every 2 hours, three times starting 7 hours before the animals were killed. The number of bromodeoxyuridine-immunoreactive cells in the SVZ was counted, as previously described (Zhang et al., 2001). The number of bromodeoxyuridine-positive cells in the SVZ gradually increased and peaked at 7 days after stroke, and significantly increased numbers of bromodeoxyuridine-positive cells persisted for at least 21 days after stroke (Fig. 1). These data show that stroke induces proliferation of SVZ cells, as has been reported previously (Jin et al., 2001;Zhang et al., 2001).
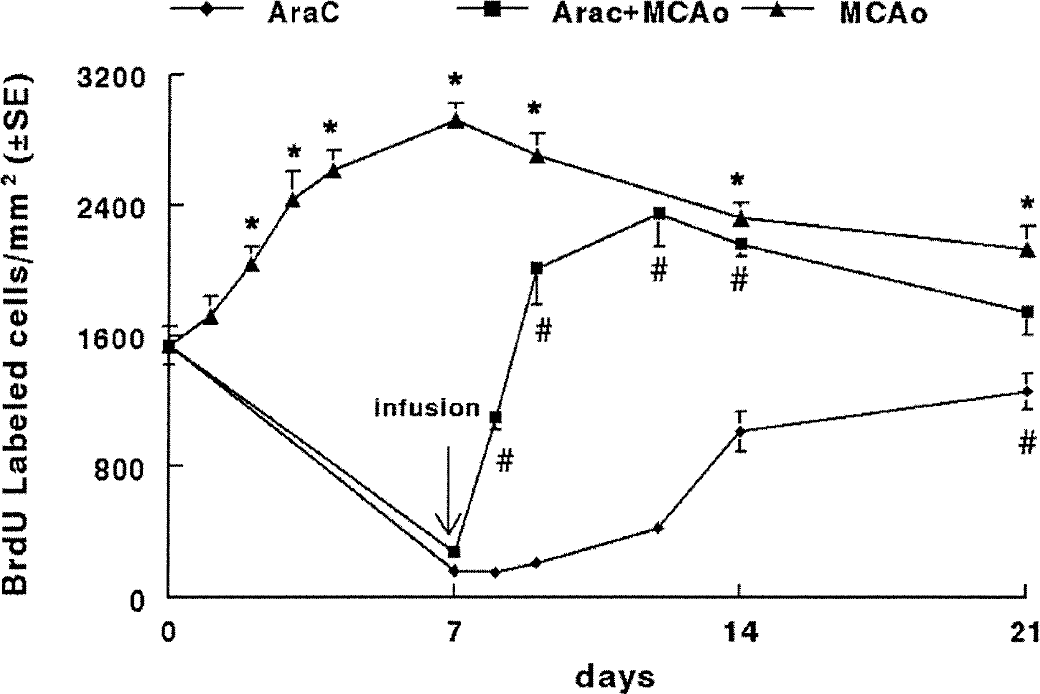
Repopulation of bromodeoxyuridine-positive cells after depletion of the proliferating cells in the SVZ after stroke. The number of bromodeoxyuridine-positive cells significantly increased after stroke (triangles). Infusion of Ara-C for 7 days almost completely eliminated bromodeoxyuridine-positive cells both in stroke rats (squares) and nonstroke rats (diamonds). However, bromodeoxyuridine-positive cells reappeared 1 day after termination of the infusion (squares), and by 5 days after the infusion the number of bromodeoxyuridine-positive cells (squares) approached levels found in ischemic rats without the Ara-C infusion (triangles). In nonstroke rats, the bromodeoxyuridine-positive cells reached preinfusion levels at 14 days after termination of the infusion (diamond, day 21). Data are presented as mean and standard errors of at least three rats for each time point. *P < 0.05 versus the zero (preischemia) time point and #P < 0.05 versus the 7-day time point. The arrow (infusion) indicates end of 7-day infusion. BrdU, bromodeoxyuridine; MCAo, middle cerebral artery occlusion.
The adult SVZ contains actively proliferating progenitor cells and relatively quiescent neural stem cells (Morshead et al., 1994). The increase in the number of bromodeoxyuridine-positive cells after stroke could be due to an increase in progenitor or neural stem cell populations in the SVZ. We tested the hypothesis that stroke increases the number of neural stem cells that give rise to progenitor cells by continuously infusing for 7 days an antimitotic drug Ara-C onto the right hemisphere to kill mitotically active cells in rats subjected to the embolic right middle cerebral artery occlusion (Zhang et al., 1997). Infusion of Ara-C started on the day of stroke and bromodeoxyuridine was administered to these rats in three injections, one every 2 hours starting 7 hours before the animals were killed. In nonstroke rats, 7 hours after termination of the infusion of Ara-C, only a few bromodeoxyuridine-positive cells were detected in the SVZ (Fig. 1). Thereafter, there was a gradual increase in the number of bromodeoxyuridine-positive cells in the SVZ, and at 14 days after infusion, the number of bromodeoxyuridine-positive cells returned to 90% of preinfusion levels (Fig. 1). Thus, treatment with Ara-C eliminates actively proliferating cells, and neural stem cells are able to replenish this population over 14 days. These data are comparable to previous results for repopulation of adult SVZ cells in the mouse after depletion of actively proliferating progenitor cells (Doetsch et al., 1999;Morshead et al., 1994, 1998). In stroke rats receiving infusion of Ara-C, there was a rapid increase in the number of bromodeoxyuridine-positive cells such that by 5 days after infusion, the number of bromodeoxyuridine-positive cells reached 90% of stroke levels compared with saline-infused rats (Fig. 1).
The SVZ contains a heterogeneous population of cells (Doetsch et al., 1997;Garcia-Verdugo et al., 1998). Type A cells divide relatively frequently and type C cells are the most actively proliferating cells in the SVZ (Doetsch et al., 1997). Type B cells form a glial boundary between migrating neuroblasts and the underlying striatum as well as between migrating neuroblasts and the ependymal cells (Doetsch et al., 1997). Using the light and the electron microscope, we analyzed the composition of the SVZ to determine which population of cells increases after stroke according to the criteria defined by Doetsch et al. (1997). The SVZ of nonstroke rats contained clusters of dark (Fig. 2A, arrows) and light cells (Fig. 2A, arrowhead) visible at the light microscope. Under electron microscope, the dark and light cells correspond to type A and type B cells in the SVZ of nonstroke rats, as previously characterized (Doetsch et al., 1997) (Figs. 2A and 2B). However, stroke rats exhibited increases of numbers of type A cells in the ipsilateral SVZ, and type A cells were present in the striatal parenchyma at 7 days after stroke (Figs. 2G and 2H). Few type B cells were detected at the interface of the striatal parenchyma and type A cells (Figs. 2G and 2H). Type A cells are neuroblasts (Doetsch et al., 1997). These observations together with the bromodeoxyuridine data suggest that the number of proliferating neuroblasts is increased. Indeed, when Ara-C was infused for 7 days onto the right cerebral cortex to eliminate actively proliferating cells, the majority of type A and type C cells disappeared, whereas type B and ependymal cells were still detected at 7 hours after infusion (Figs. 2C and 2D). Consistent with previous findings (Doetsch et al., 1999;Morshead et al., 1994, 1998) in the mouse SVZ, type A cells reappeared in the SVZ of nonstroke rats 14 days after infusion (Figs. 2E and 2F). The 7 days of Ara-C infusion onto the ipsilateral cortical surface to stroke rats revealed an identical depletion of type A cells, and a smaller effect on type B and ependymal cells at 7 hours after infusion (Figs. 2I and 2J). However, reappearance of type A cells in the SVZ was observed at 7 days after infusion (Figs. 2K and 2L), which is more rapid compared with nonstroke rats. The organization of SVZ cells was similar to that of 7-day stroke rats without treatment with Ara-C (Figs. 2G and 2H). These morphologic data are consistent with bromodeoxyuridine-immunostaining data, confirming that treatment with antimitotic agents actively kills proliferating cells (Doetsch et al., 1999;Morshead et al., 1994, 1998), and further that neuroblasts in stroke rats after Ara-C treatment are rapidly regenerated. This rapid repopulation of the SVZ cells after the Ara-C infusion in stroke animals may result from increases in neural stem cells or increases in proliferation of remaining proliferating cells.
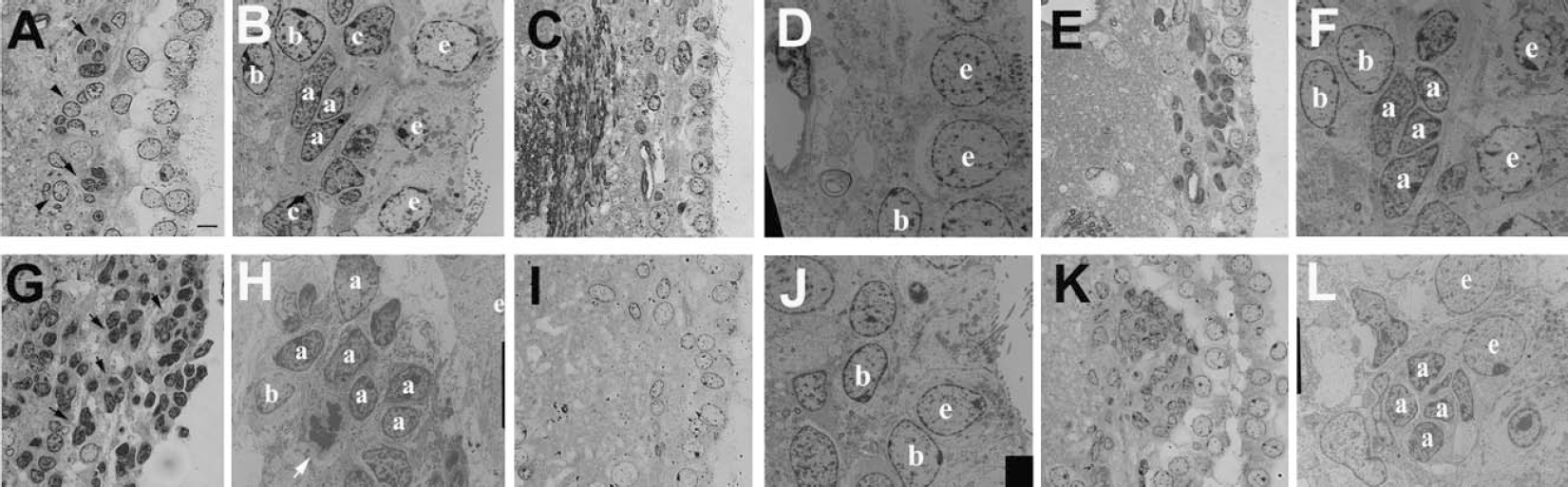
Light and electron microscopic analysis of regenerating SVZ cells in nonstroke and stroke rats after treatment with Ara-C. The coronal semithin (1.5 μm,
Given that neural stem cells isolated from the SVZ can form neurospheres in the presence of growth factors (Gritti et al., 1996;Morshead et al., 1994, 1998) in vitro, we isolated SVZ cells to investigate whether there was a concomitant increase in the numbers of neural stem cells after stroke that could contribute to the increased numbers of proliferating cells observed in vivo after stroke. SVZ cells were isolated from nonstroke rats and from rats 7 days after stroke and cultured at a density of 10,000 cells/mL in the presence of basic fibroblast growth factor and EGF. At 7 days in vitro, the total numbers of spheres and the size of individual spheres were measured. The average sphere diameter from stroke rats was significantly (P < 0.05) larger (108.8 ± 3.98 μm, Fig. 3A) than those derived from nonstroke rats (85.5 ± 9.24 μm, Fig. 3B). Moreover, the number of spheres derived from stroke rats was significantly increased (P < 0.05, Fig. 3C) compared with the number derived from nonstroke rats (Fig. 3C). When SVZ cells were isolated from nonstroke and stroke rats immediately after termination of infusion of Ara-C for 7 days, the numbers and size of the spheres did not differ compared to nonstroke and stroke rats that did not receive Ara-C infusion, respectively (Fig. 3C). Given that Ara-C infused rats have a greater than 98% loss of proliferating cells in vivo at the time when the SVZ cells were isolated, suggests that proliferating progenitor cells are not responsible for the increased number of primary spheres after stroke.
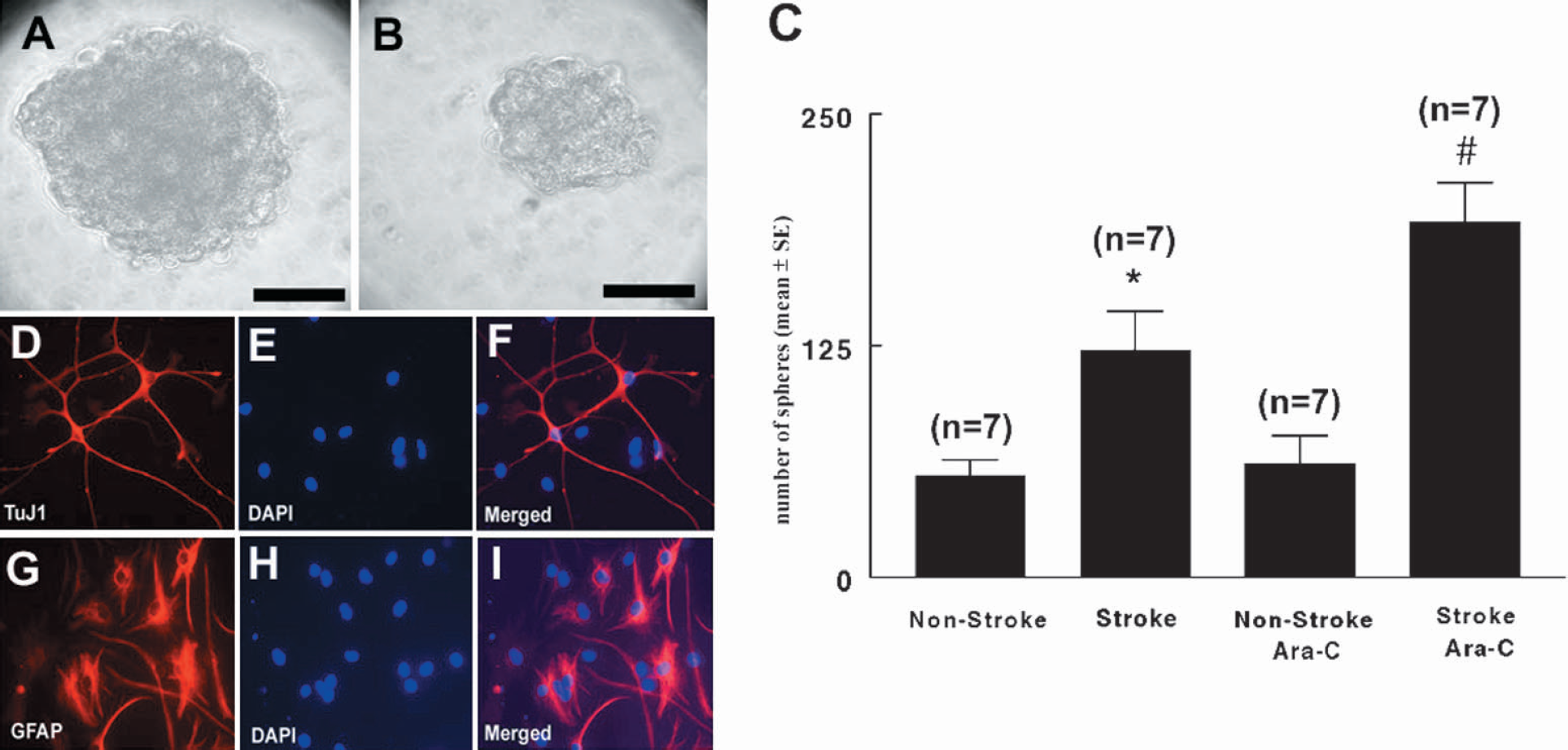
In vitro analysis of spheres. The size of primary spheres derived from the stroke rats (
To confirm that the spheres isolated in culture were derived from neural stem cells, we examined the self-renewal capacity and multipotentiality of the spheres. Individual primary spheres were dissociated into single cells and replated in the medium containing basic fibro-blast growth factor and EGF. Secondary spheres from both nonstroke and stroke derived primary spheres were generated in 100% of the wells, suggesting that these spheres have ability to self-renew, one of the cardinal characteristics of stem cells (Reynolds and Weiss, 1992b). Individual spheres were plated in differentiating media containing 2% fetal bovine serum (Gritti et al., 1996;Reynolds and Weiss, 1992b) and their differentiation profile was examined. Immunostaining revealed 35% TuJ1 (a marker for neuroblasts, Figs. 3D, 3E and 3F) and 45% glial fibrillary acidic protein (a marker for astrocytes, Figs. 3G, 3H and 3I) 7 days after in the differentiation media, indicating that cells in spheres differentiate into neurons and glia. These in vitro results show that spheres derived from stroke rats have characteristics of adult SVZ neural stem cells; i.e., they self-renew and make neurons and glia (Reynolds and Weiss, 1992b). Therefore, our data suggest that stroke increases the numbers of neural stem cells that give rise to neuroblasts.
Neuroblasts migrate from the SVZ to ischemic striatum via chain migration
Migrating neuroblasts generated in the SVZ travel as chains through tunnels formed by astrocytes to the olfactory bulb via the rostral migratory stream (Figs. 4A and 4B) (Doetsch et al., 1997;Garcia-Verdugo et al., 1998). Using immunohistochemistry for the migrating neuroblast marker doublecortin (Francis et al., 1999), we found that stroke significantly increased the number of doublecortin-positive cells in the ipsilateral striatum (Figs. 4C and 4E) compared with the number in the contralateral striatum (Figs. 4D and 4E). The number of doublecortin-positive cells peaked at 14 days after stroke and doublecortin-positive cells persisted in the ipsilateral striatum for at least 42 days after stroke (Fig. 4E). Doublecortin-positive cells were not detected in the ipsilateral cortex. In addition to the similar morphology of doublecortin-positive cells and migrating neuroblasts, we observed three patterns of doublecortin-positive cells in the ipsilateral striatum. Doublecortin-positive cells formed chainlike structures extending from the SVZ toward the ischemic striatum (Figs. 5A and 5B). When they reached the ischemic boundary, these cells aggregated into clusters (Figs. 5D and 5E). To analyze how these patterns change with time, we quantified the numbers of chainlike and clustered doublecortin-positive cells (Fig. 5G). During the first week of stroke, the number of chainlike doublecortin-positive cells significantly increased, whereas the number of clusters of doublecortin-positive cells significantly increased at 7 days after stroke (Fig. 5G). The number of chainlike and clustered doublecortin-positive cells peaked 14 days after stroke (Fig. 5G). Thereafter, doublecortin-positive cells in the clusters dispersed and these cells exhibited multiple processes (Fig. 5H). Furthermore, infusion of Ara-C for 7 days significantly (P < 0.01) reduced number of doublecortin-positive cells in the ipsilateral striatum from 185 ± 45 cells per section in 7-day stroke rats (n = 7) to 11 ± 3 cells per section in 7-day stroke rats treated with Ara-C (n = 7). Together, these data suggest that doublecortin-positive cells in the ipsilateral striatum may migrate from the SVZ via chains.
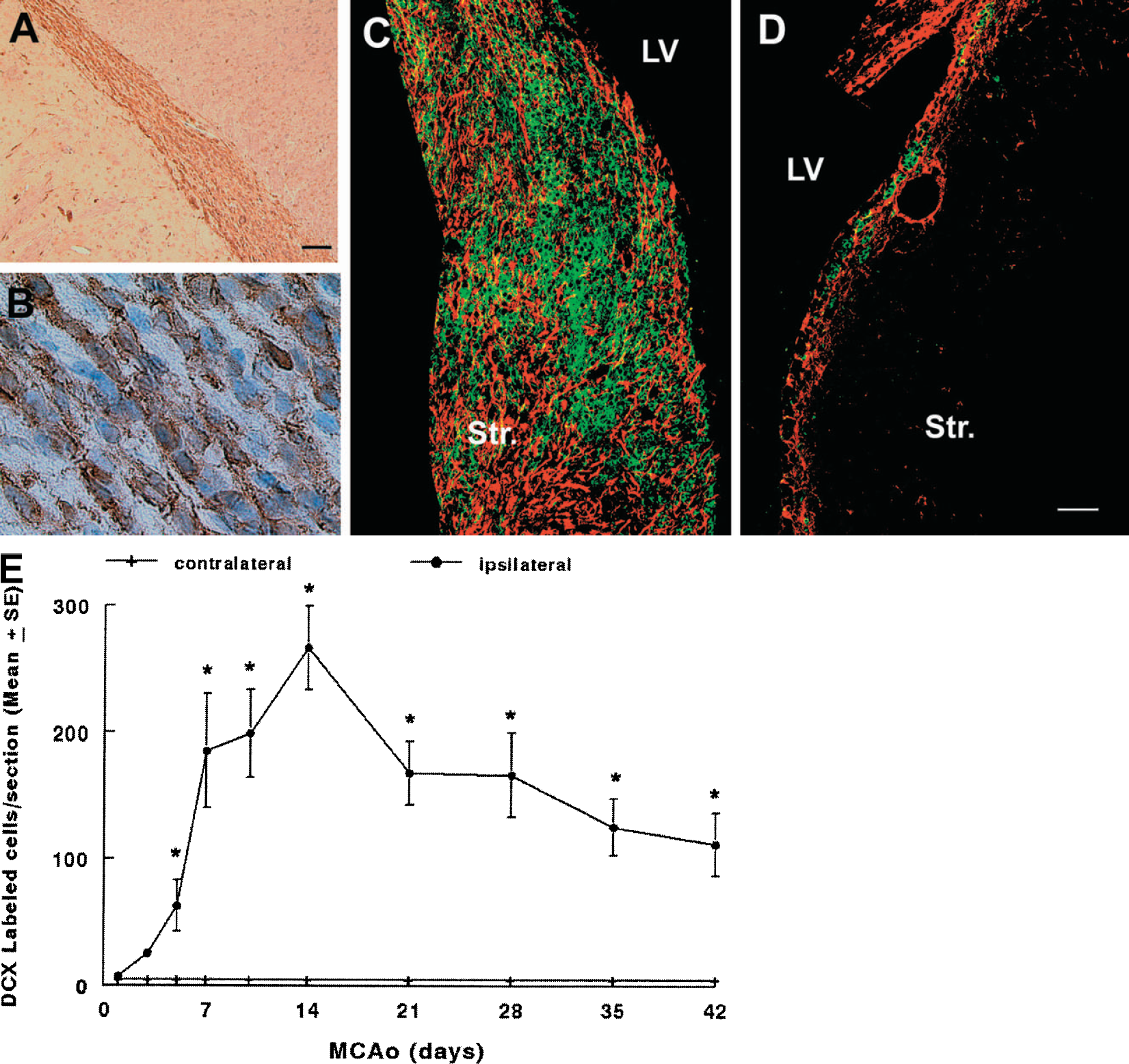
Stroke increases the number of doublecortin-positive cells. Microphotographs of a representative nonstroke rat show that doublecortin-immunoreactive cells are present in the rostral migratory stream (
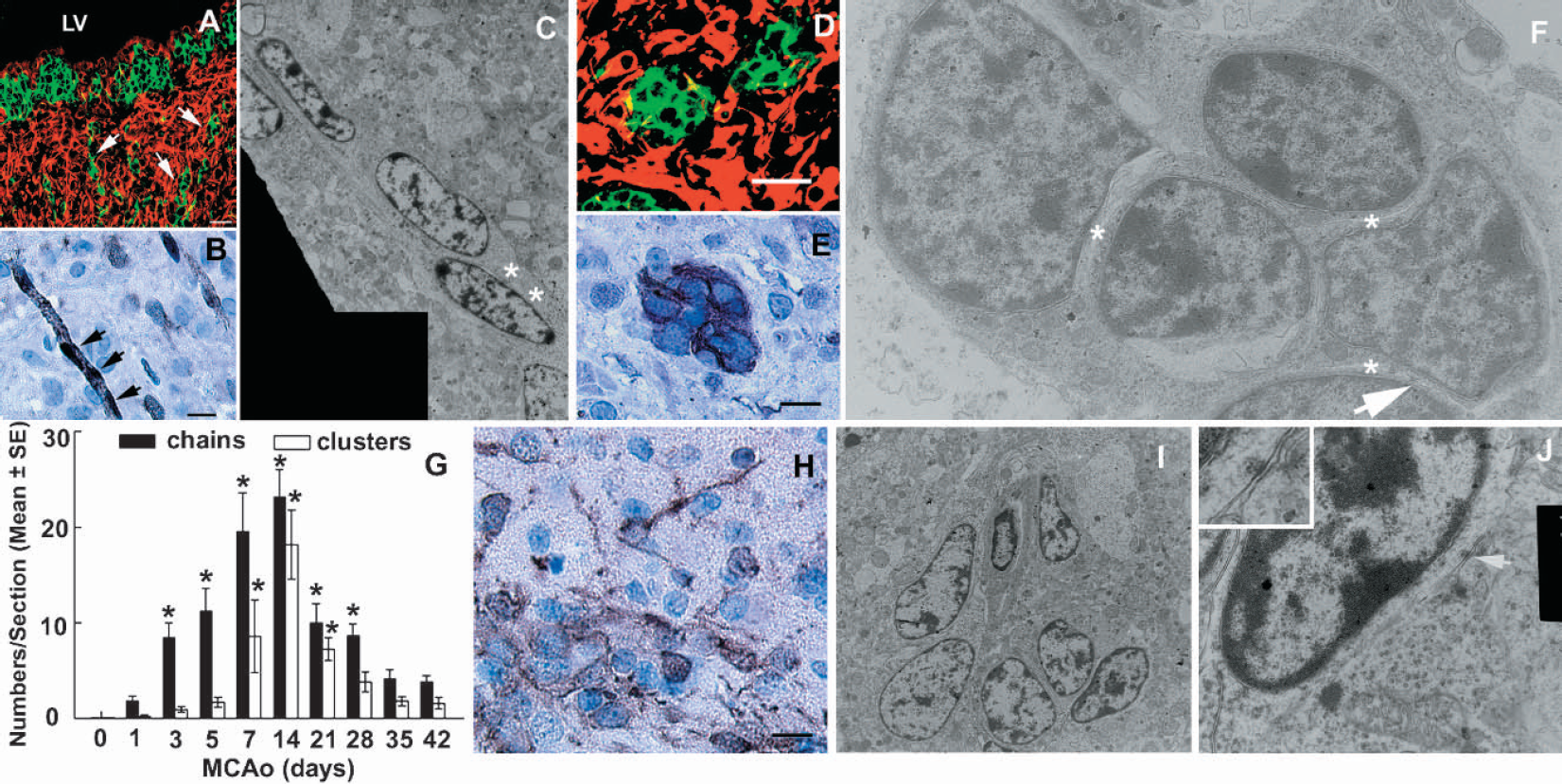
Neuroblasts migrate into the ipsilateral striatum. Panels
To further confirm that doublecortin-positive cells in the striatum migrate from the SVZ, we performed electron microscopic analysis. Electron microscopy revealed that cells in chainlike organization had a spindle-shaped dark nucleus and many free polyribosomes in a leading process (Fig. 5C, asterisks), which is consistent with morphologic characteristics of type A cells in the SVZ (Doetsch et al., 1997). Cells in the clusters of the ipsilateral striatum exhibit abundant free ribosomes in the cytoplasm (Fig. 5F). Open extracellular spaces (Fig. 5F, asterisks) and tight junctions (Fig. 5F, arrow) were frequently observed between cells. Morphology of these cells resembles the ultrastructure of type A cells in the SVZ (Doetsch et al., 1997). Cells dispersed from the cluster had somatodendritic junctions (Figs. 5I and 5J, arrow). Together, ultrastructural data demonstrate that cells in the ipsilateral striatum have morphologic characteristics of type A cells in the SVZ, indicating that these cells migrate from the SVZ to the ischemic boundary.
DISCUSSION
Stroke induces neurogenesis in the SVZ and dentate gyrus (Jin et al., 2001;Yoshimura et al., 2001;Zhang et al., 2001). However, whether increases in neurogenesis after stroke are attributed to increases in the proliferating neuroblasts or to increases in recruitment of neural stem cells has not been investigated. In the present study, we found an increase in recruitment of neural stem cells in the SVZ that correlates with the increased neurogenesis observed after stroke. Support for this conclusion comes from complementary in vivo and in vitro experiments. After stroke, the number of neuroblasts significantly increased in the SVZ, and treatment with the antimitotic agent Ara-C eliminated the actively proliferating cell population in the SVZ. Although neuroblasts were entirely repopulated at 7 days after termination of Ara-C infusion in stroke rats, repopulation of neuroblasts took 14 days in nonstroke rats. Previous studies demonstrate that neural stem cells in the SVZ can repopulate the constitutively proliferating population after its ablation (Doetsch et al., 1999;Morshead et al., 1994, 1998). Thus, rapid repopulation of neuroblasts after its depletion in stroke rats may result from increases in recruitment of neural stem cells. Alternatively, it is possible that repopulation of neuroblasts results from increases in proliferation of the 2% of the remaining proliferating cells after Ara-C treatment. However, based on our in vitro data, this is unlikely the case. Our in vitro experiments reveal that SVZ cells isolated from nonstroke rats treated with Ara-C for 7 days when a greater than 98% loss of proliferating cells in vivo can generate the same number of spheres as the cells isolated from nonstroke rats without Ara-C treatment. These data are consistent with a previous study in mice (Doetsch et al., 1999), suggesting that actively proliferating SVZ cells are not source of stem cells that form spheres in vitro. Moreover, SVZ cells isolated from stroke rats treated with Ara-C exhibit the same capacity to generate spheres as the cells isolated from stroke rats without depleting actively proliferating cells after 7 days in vitro, a time course that coincides with the time course of the recruitment of stem cells to replenish proliferating cells in vivo. Furthermore, primary spheres generate secondary spheres, which differentiate into neurons and glia, indicating that these cells exhibit the cardinal properties of neural stem cells (Reynolds and Weiss, 1992b). Taken together, our data suggest that stroke increases recruitment of quiescent stem cells into proliferation in a symmetric mode of division to give rise to stroke-induced neurogenesis.
Neural stem cells are a relatively quiescent cell population, and proliferation of stem cells in the SVZ is tightly controlled under physiologic conditions (Morshead et al., 1994, 1998). However, infusion of EGF into the lateral ventricles results in 3.7-fold increases in the total number of stem cells isolated in vitro (Craig et al., 1996). Stroke upregulates mitogen expression, including EGF and basic fibroblast growth factor expression (Planas et al., 1998;Speliotes et al., 1996;Tanaka et al., 1999), which may contribute to the increase in neural stem cells. Infusion of basic epidermal growth factor (bEGF) and EGF into the lateral ventricle of forebrain ischemic mice further increases neurogenesis (Nakatomi et al., 2002). In stroke rats, the remarkable fidelity with which increased migrating neuroblasts are reestablished in the SVZ after Ara-C treatment suggests that directional cues for the organization of the SVZ cells remain even after depletion of actively proliferating cells.
Another major finding in the present study is that neuroblasts in the SVZ migrate toward the ischemic boundary regions in the striatum via a chain migration. In the adult brain, doublecortin is primarily expressed in migrating and differentiating neurons in the SVZ and rostral migratory stream (Arvidsson et al., 2002;Nacher et al., 2001). Observations of significant increases in doublecortin-positive cells in the ipsilateral striatum suggest that these cells may migrate from the SVZ. A similar pattern of doublecortin-positive cells has been recently reported after stroke (Arvidsson et al., 2002;Jin et al., 2003). Although doublecortin-immunoreactive cells have been used as a marker for migrating neuroblasts, the presence of doublecortin-immunoreactive neurons in the piriform cortex of the nonstroke adult rat (Nacher et al., 2001) raises the possibility that stroke may upregulate doublecortin expression in neurons. Therefore, increased doublecortin-immunoreactive cells in the ipsilateral striatum could be due to increases in striatal neurons with overexpression of doublecortin rather than to migration of neuroblasts in the SVZ to striatum. To confirm that neuroblasts in the SVZ migrate to the stroke boundary, we performed electron microscope analysis. Ultrastructural analysis in the present study indicates that cluster cells in the ipsilateral striatum have spindle-shaped dark nuclei, many free ribosomes in the cytoplasm, open extracellular spaces, and tight junctions, and are morphologically similar to migrating neuroblasts observed in the nonstroke SVZ (Doetsch et al., 1997). Furthermore, treatment of stroke rats with Ara-C inhibits actively proliferating cells in the SVZ and abolishes doublecortin-immunoreactive cells in the ipsilateral striatum, indicating that actively proliferating progenitor cells in the SVZ differentiate into migrating neuroblasts that travel to the ischemic boundary because doublecortin is expressed exclusively in postmitotic neurons (Feng and Walsh, 2001). Under nonstroke conditions, neuroblasts migrate tangentially from the SVZ to the olfactory bulb along the rostral migratory stream via chains and move radially into the granular and periglomerular layers when they reach the core of the olfactory bulb (Doetsch et al., 1997;Garcia-Verdugo et al., 1998). In the present study, we found that doublecortin-positive cells exhibited chains oriented toward the ischemic boundary regions, formed clusters and dispersed in the ipsilateral striatum. Taken together, our data indicate that neuroblasts in the SVZ migrate toward the ischemic boundary.
Progenitors and stem cells are differentially regulated with age (Seaberg and van der Kooy, 2002;Tropepe et al., 1997). In the SVZ, the number of progenitors decreases with age, whereas population of neural stem cells is relatively constant even into senescence (Tropepe et al., 1997). The majority of stroke patients are older than 60 years. Therefore, our finding that after stroke increased neuroblasts derived from neural stem cells migrate into the ischemic striatum might have important implications for targeting endogenous neural stem cells and progenitor cells toward the ultimate goal of repairing damaged brain tissue, even in the aged patient.
