Abstract
Neurogenesis occurs throughout life in the dentate gyrus of hippocampus and subventricular zone, but this phenomenon has rarely been observed in other brain regions of adult mammals. The aim of the current study was to investigate the cell proliferation process in the ischemically challenged region-at-risk after focal cerebral ischemia in the adult rat brain. A reversible photothrombotic ring stroke model was used, which features sustained hypoperfusion followed by late spontaneous reperfusion and a remarkable morphologic tissue recovery in the anatomically well defined somatosensory cortical region-at-risk. Twelve-week-old male Wistar rats received repeated intraperitoneal injections of the cell proliferation specific marker 5-bromodeoxyuridine (BrdU) after stroke induction. Immunocytochemistry of coronal brain sections revealed that the majority of BrdU-positive cells were of glial, macrophage, and endothelial origin, whereas 3% to 6% of the BrdU-positive cells were double-labeled by BrdU and the neuron-specific marker Map-2 at 7 and 100 days after stroke onset in the region-at-risk. They were distributed randomly in cortical layers II-VI. Three-dimensional confocal analyses of BrdU and the neuronal-specific marker Neu N by double immunofluorescence confirmed their colocalization within the same cells at 72 hours and 30 days after stroke induction. This study suggests that, as a potential pathway for brain repair, new neurons can be generated in the cerebral cortex of adult rats after sublethal focal cerebral ischemia.
Neurogenesis is generally considered to be confined to discrete brain regions in adult mammalian brains under normal physiologic conditions (Kaplan and Hinds, 1977). However, recent data suggest that new neurons are constantly added to the neocortical association area in adult macaques during normal physiologic conditions (Gould et al., 1999). Stem cell manipulation in vitro and in vivo with proliferation and subsequent differentiation into neurons (Reynolds and Weiss, 1992) has been discussed as a potential source for neuronal regeneration. To enable reproducible study of a penumbralike situation in cerebral ischemia, a photothrombotic cortical ring model of rat stroke-in-evolution was developed (Wester et al., 1995). In this model the centrally located cortical region-at-risk is enveloped by a ring-shaped cortical ischemic locus as induced by a type II photochemical reaction leading to microvascular occlusion (Watson, 1998), which results in an anatomically predefined region-at-risk that slowly but inevitably transforms into complete tissue necrosis (Wester et al., 1995). This stroke model was recently modified by using a thinner thickness of the laser irradiation beam to facilitate a sustained hypoperfusion with blood flow levels of 40–23–30% of baseline at 4–24–48 hours followed by a late spontaneous reperfusion starting at 72 hours poststroke in the cortical region-at-risk (Gu et al., 1999c,d). This resulted in a progressively altered neuropil and nerve cell morphology in the region-at-risk that reached their maximum severity at 48 hours after stroke onset with most of the neurons exhibiting eosinophilia and pyknosis (Gu et al., 1999a,b). However, at 72 hours after ischemic onset, a remarkable morphologic restoration of the nerve cells in this region started, which evolved into a chiefly unremarkable cytologic appearance at 7 to 28 days after stroke induction (Gu et al., 1999a,b). The tissue recovery coincided with the onset of spontaneous reperfusion to the cortical region-at-risk (Gu et al., 1999b,c,d). In the cortical region-at-risk some TUNEL-positive stained cells were seen at 24 to 48 hours after ischemic onset, whereas at 72 hours and 7 days after stroke induction such cells were no longer observed (Hu et al., 1999). The current study aimed to investigate the cell proliferation process presumptively related to the morphologic restoration observed in the cortical region-at-risk in this model.
MATERIALS AND METHODS
Animal care and all experimental procedures were carried out in accordance with the European Communities Council Directive (86/609/EEC), and the experimental protocol was approved by the Ethics Committee for Animal Research at Umeå University. Nonfasted 12-week-old male Wistar rats (BK, Sollentuna, Sweden) were subjected to a reversible photothrombotic stroke lesion as described previously (Gu et al., 1999d). To mark many proliferating cells at the same time minimizing any potential cellular toxicity, the cell proliferation-specific marker 5-bromodeoxyuridine (BrdU; Sigma; St. Louis, MO, U.S.A.) was administered repeatedly in small dosages intraperitoneally (10 mg/kg BrdU dissolved in saline at each injection occasion). In the 100 days group (n = 4), BrdU-injections were started at 24 hours after stroke onset and were continued twice daily during the first and second week, once a day during the third and fourth week, and twice a week for the remaining weeks finally ending on day 84. In the 30 days group (n = 4), BrdU was injected as described above, but delivery was stopped on day 10 to observe cell survival after proliferation. To search for the earliest time point when cell proliferation occurred after brain ischemia, three groups of animals that received identical initial BrdU-treatment as described above were sacrificed at day 2, 3, and 7, respectively, after stroke (n = 4 in each group). Sham-operated rats underwent the same surgical procedure as rats subjected to ischemia and were irradiated but did not receive erythrosin B. In these rats BrdU was delivered in the same way as in the ischemic groups and rats were killed at 7 and 100 days (n = 4 in each group). All of the animals were transcardially perfused with 37°C Histochoice tissue fixative (Menezes and Luskin, 1994). Brains were kept in the same fixative at 4°C for at least 24 hours before being processed.
Immunohistochemistry
Paraffin-embedded coronal brain sections (8-μm thick) through the lesioned cortex were immunostained according to the recommended protocol for using the Vector staining kit (Vector Laboratories, Burlingame, CA, U.S.A.). Endogenous peroxidase was deactivated by 30 minutes incubation in freshly prepared 3% H2O2/methanol. Antigen retrieval to facilitate the BrdU-immunostaining was achieved by 21 minutes of boiling in 0.1 mol/L citrate buffer (pH 6.0) in a microwave oven (Tischler, 1995). The boiling procedure also completely deactivated endogenous alkaline phosphatase. Single BrdU-immunostaining was performed with mouse-anti-BrdU (1:50, Becton Dickinson, San Jose, CA, U.S.A.), detected with biotinylated horse-anti-mouse IgG (rat-absorbed, Vector) and Elite Vectastain ABC-peroxidase kit (Vector), and reacted with diaminobenzidine (DAB, Sigma). For double immunolabeling, the BrdU-immunolabeling was performed with the Vectastain ABC-AP- Vector®-red, followed by a second labeling with a neuron (mouse-anti-Map-2a,b, Boehringer Mannheim, Indianapolis, IN, U.S.A.) or glial (rabbit-anti-GFAP; DAKO, A/S, Glostrup, Denmark) cell marker, detected with Elite Vectastain ABC-peroxidase-DAB. To verify the specificity of the immunostaining, primary antibodies were omitted from the staining procedure in a set of control sections. Naive animals without BrdU-injections were used as anti-BrdU negative controls (n = 4).
Immunofluorescence
Ten-μm-thick coronal cryostat brain sections were treated for antigen retrieval by microwaving. The sections were incubated with the neuronal-specific marker mouse-anti-Neu N (Chemicon, Temicula, CA, U.S.A.) in ambient temperature overnight and reacted with cy-3-donkey-anti-mouse F(ab)2 (Jackson ImmunoResearch, West Grove, PA, U.S.A.) for 3 hours. Thirty minutes incubation with goat-anti-mouse Fab was added to block the unsaturated binding sites on the first secondary antibody. The sections were then incubated with 1:50 mouse-anti-BrdU overnight, detected with SP-biotin-donkey anti-mouse F(ab)2 (Jackson) and DTAF-streptavidin (Jackson). The sections were mounted in Vectashield medium and examined with a confocal microscope (Molecular Dynamics, Piscataway, NJ, U.S.A.).
Cell count
To estimate the relative frequency of various types of newborn cortical cells (based on cellular morphology) in the ischemic brain region, two randomly selected coronal brain sections from each brain were single-labeled with anti-BrdU and counted under a CAST-Grid system (Olympus, Albertslund, Denmark). Similarly, the numbers of BrdU and Map-2 double-labeled versus BrdU singly positive cells were counted in the central region-at-risk. Data are expressed as mean ± SD.
RESULTS
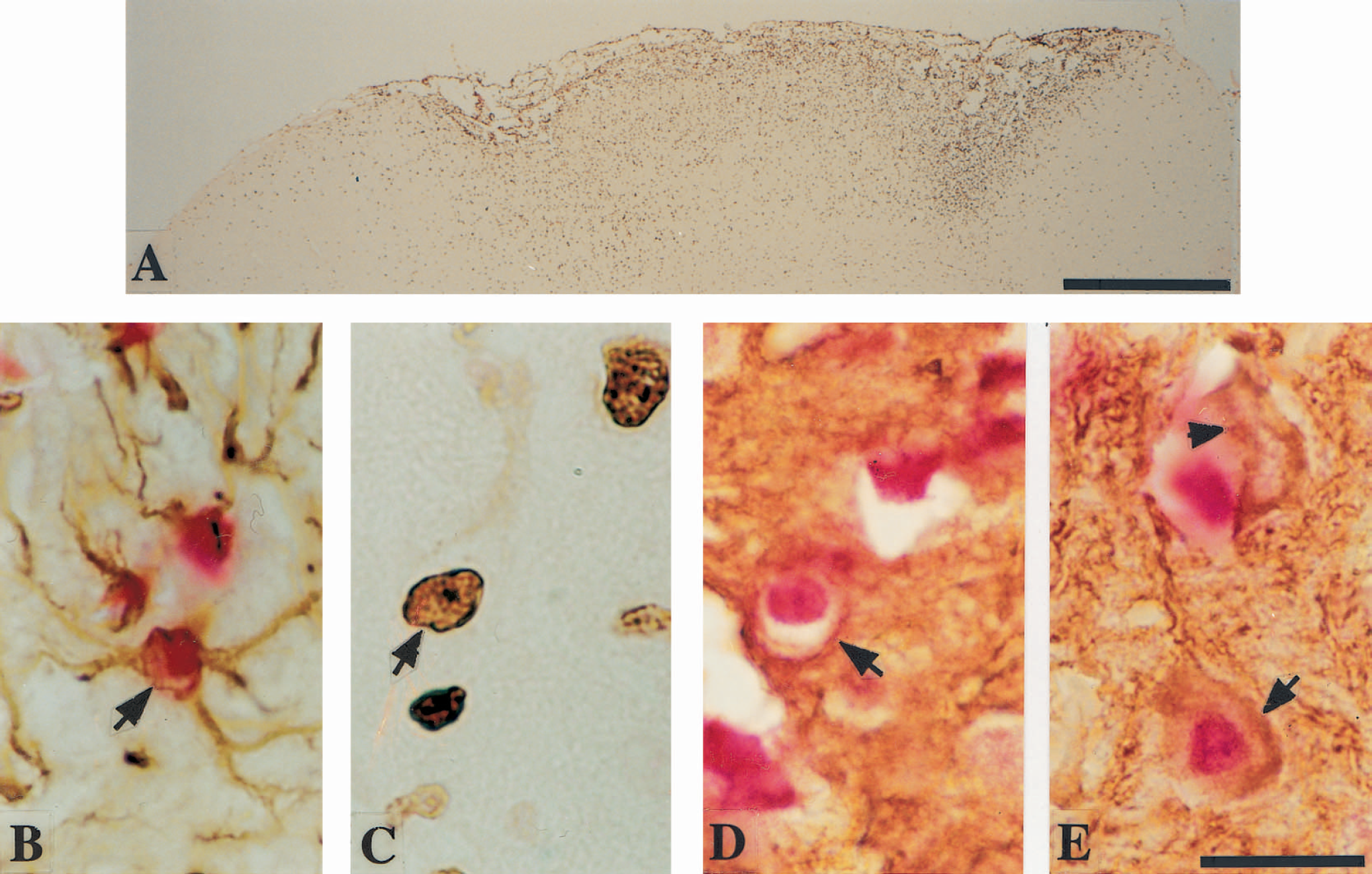
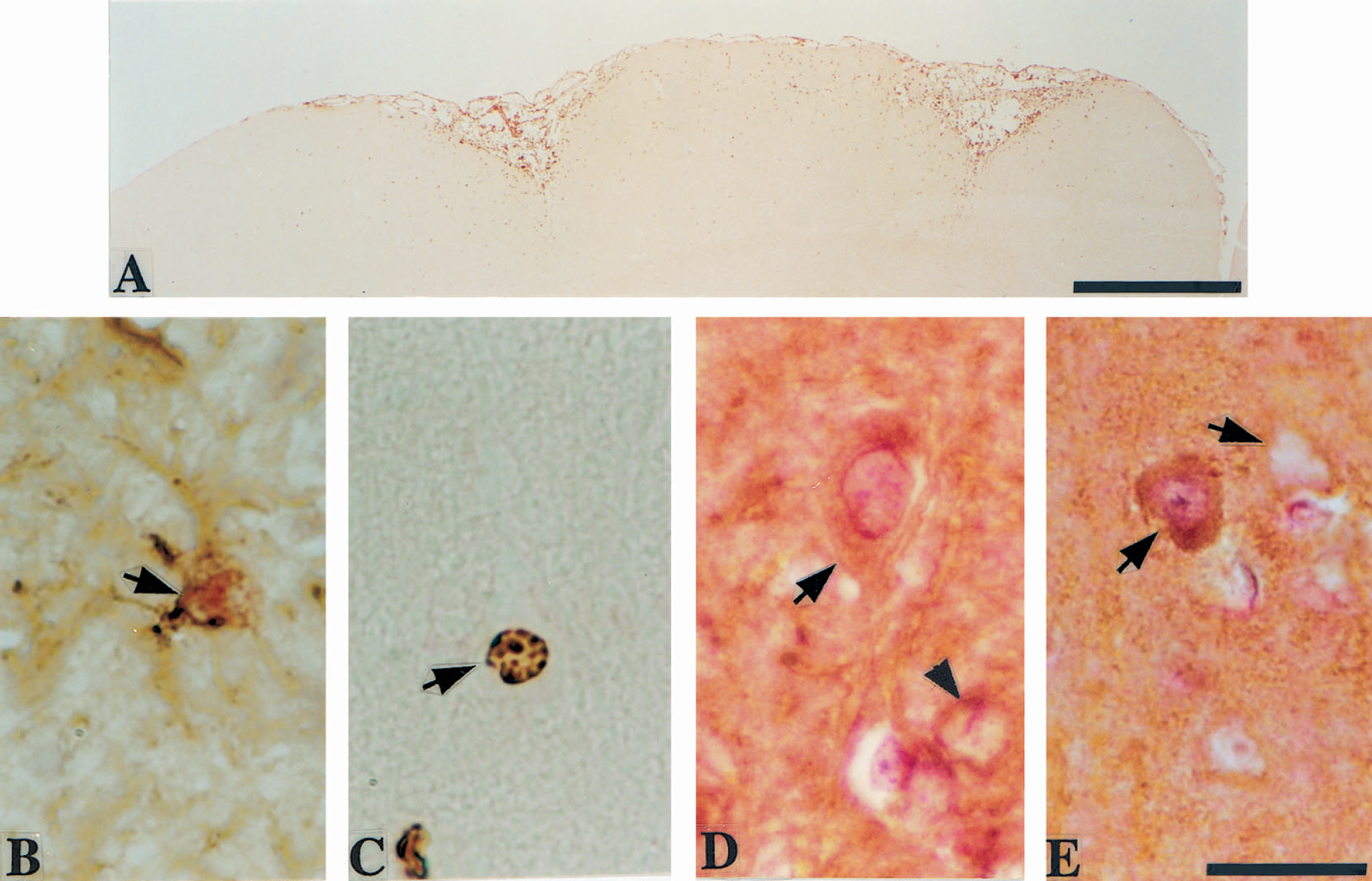
Widespread BrdU-incorporated cells were consistently observed in the cortical region-at-risk and ring lesion region at 7 days after stroke induction (Fig. 1A). At 100 days after ischemic onset, fewer BrdU-positive cells were seen, with a distribution similar to that at 7 days (Fig. 2A), except that few BrdU-labeled cells were observed in the annular ring lesion region. Generally, the color of BrdU nuclear labeling was paler at 100 days than 7 days after stroke induction (Fig. 2A). In the cortical ring lesion region, numerous BrdU-incorporated macrophages, astrocytes and endothelial cells were seen at 7 days after stroke onset. At 100 days poststroke, this region exhibited cystic coagulation necrosis, but contained some BrdU-positive astrocytes and a few endothelial cells. In the cortical region-at-risk, the majority of the BrdU-incorporated cells at 7 days postirradiation were glial cells (62 ± 9.5% of the total 113 ± 34 BrdU single-labeled cells counted per brain), macrophages (20 ± 9.7%), and endothelial cells (12 ± 3.7%). The corresponding proportions at 100 days after stroke onset were 84 ± 2.1% for glial cells (out of a total 82 ± 30 BrdU single-labeled cells counted per brain), 3.2 ± 3.9% for macrophages, and 8.8 ± 5.1% for endothelial cells. In sham-operated rats, no BrdU-positive cells were observed in the cortex corresponding to the region-at-risk under the CAST-grid system. However, in the whole cortex from control animals, 0–5 BrdU-positive glial cells and 0–1 BrdU-positive endothelial cells per brain section were seen at 7 days, whereas at 100 days after operation 3–8 BrdU-positive glial cells, 0–1 BrdU-positive macrophages, and 0–1 BrdU-positive endothelial cells per brain section were observed. In the rats subjected to ischemia, newborn astrocytes, as identified by nuclear BrdU-incorporation in GFAP-positive cells (Figs. 1B and 2B), were mixed with GFAP singly positive astrocytes in the postischemic cortex at 7 (Fig. 1B) and 100 days (Fig. 2B) after stroke onset. To the surprise of the authors, at 7 and 100 days after stroke onset, some of the BrdU single-labeled cells with a similar size to macrophages exhibited a large single nucleus with a distinct centered nucleolus (Figs. 1C and 2C) suggesting a neuronal morphologic appearance. To verify this initial observation, BrdU and Map-2 double immunolabeling was conducted (Figs. 1D, 1E, 2D, and 2E). The cells with nuclear BrdU-incorporation were also Map-2 immunoreactive in their cytoplasm. At 7 days after stroke onset these cells were round without any light microscopically discernible dendritic processes (Fig. 1D), but some may have had neuriticlike processes (Fig. 1E). At 100 days after ischemic onset most of these double-labeled cells in the cortical region-at-risk had pyramidal-shaped cell bodies and clearly discernible dendritic processes (Fig. 2D and 2E). At 7 and 100 days postirradiation these cells were scattered randomly in cortical layers II-VI in the region-at-risk and a few were also found in the exterior counterpart, that is, the boundary zone immediately outside the ring lesion region. The proportion of BrdU and Map-2 double-labeled cells in the region-at-risk was 3.3 ± 0.3% of the total 1405 ± 108 BrdU single-labeled cells per brain counted at 7 days, and 5.8 ± 1.4% of the total 745 ± 95 BrdU-positive cells counted per brain at 100 days after stroke induction. Similar BrdU and Map-2 double-labeled cells were also observed in the dentate gyrus of hippocampus at 7 and 100 days after stroke onset. However, no such cells were seen in the annular ring lesion region. Similarly, no BrdU and Map-2 double-labeled cells were found in the cerebral cortex of sham-operated animals at 7 and 100 days after operation. No specific cell BrdU-labeling was observed in the BrdU-negative controls.
To confirm the finding that nuclear BrdU-incorporation colocalized with a neuron-specific marker within the same cortical cells after stroke, three-dimensional confocal analyses of BrdU and Neu N double immunofluorescence were conducted. At 30 days after stroke induction BrdU and Neu N double-labeled cells were identified (Fig. 3). Neu N labeled the nucleus and part of the cytoplasm of the BrdU-positive cells found in cortical layer IV of the region-at-risk. It thus revealed a cell labeling pattern different from BrdU pure nuclear labeling (Fig. 3). A stronger nuclear BrdU-immunofluorescence signal was detected in the neuron than in a proximate nonneuronal cell (Fig. 3). The spatial distribution of cells double-labeled by BrdU and Neu N was similar to that of BrdU and Map-2 double-labeled cells. The earliest time point when BrdU and Neu N double-positive cells appeared in the region-at-risk was at 72 hours after stroke onset (Fig. 4). In these cells the pure nuclear Neu N labeling merged completely with BrdU nuclear labeling (Fig. 4).
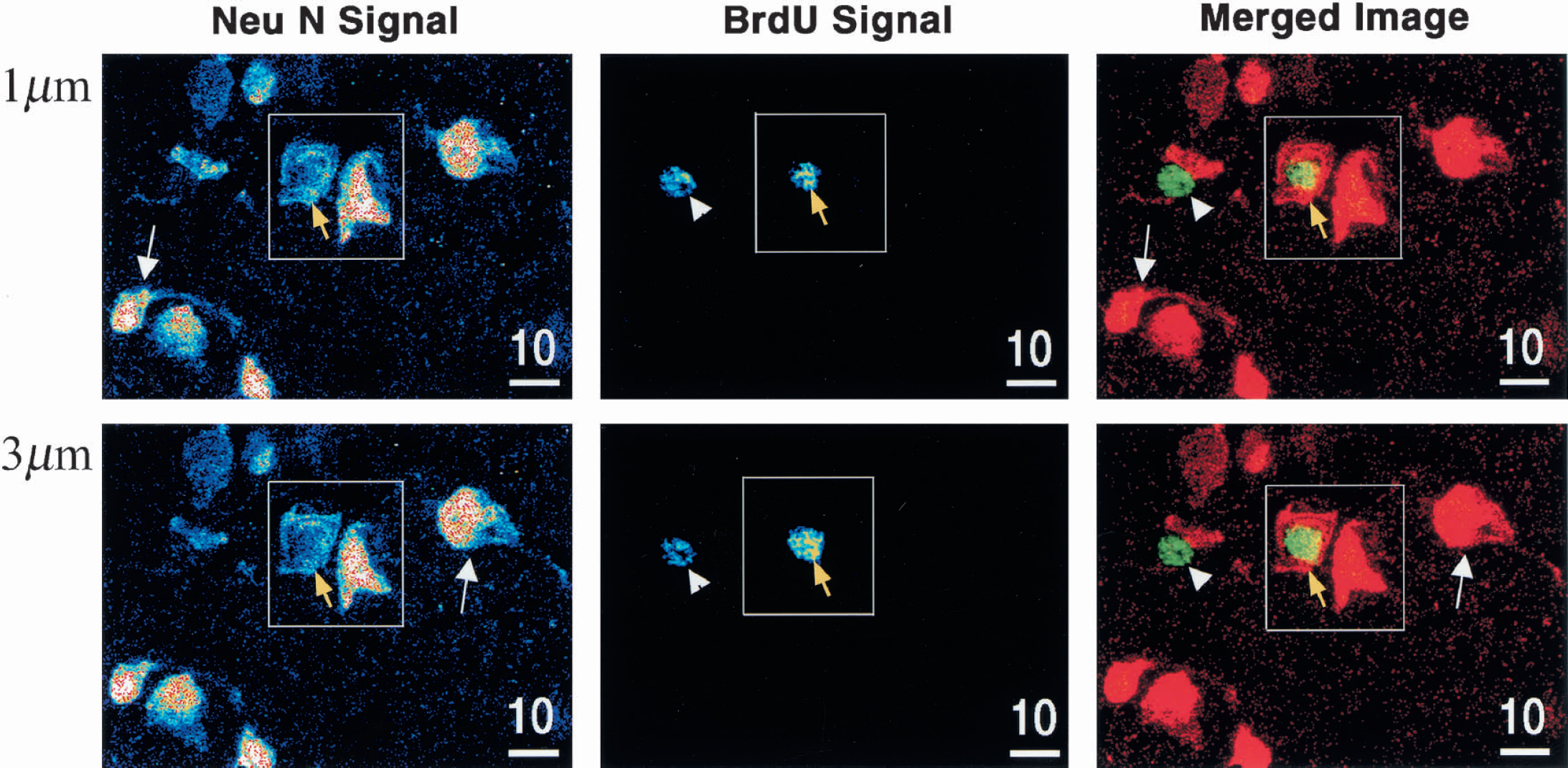
Three-dimensional confocal analyses of Neu N+ BrdU double-labeling of cortical cells at two Z-series planes, at a distance of 1 μm, 30 days after stroke induction. The left column shows the signal intensity for Neu N, the middle column displays the signal intensity for BrdU, and the right column exhibits a merged image of the Neu N and BrdU labeling. In the right column, Neu N is pseudo-colored in red (cy3) and BrdU in green (DTAF) resulting in the double-immunopositive merged image appearing in yellow. The cell designated by the yellow arrow in square was both Neu N positive (left) and BrdU positive (middle), resulting in a yellow Neu N and BrdU double-immunopositive nucleus in the merged image (right). White arrowhead indicates a neighboring Neu N negative (left and right) but BrdU-positive (middle and right) cell, that is, a nonneuronal newborn cell. White arrows outside the square are pointed at two Neu N positive (left and right) but BrdU-negative (middle and right) cells, that is, mature neurons.
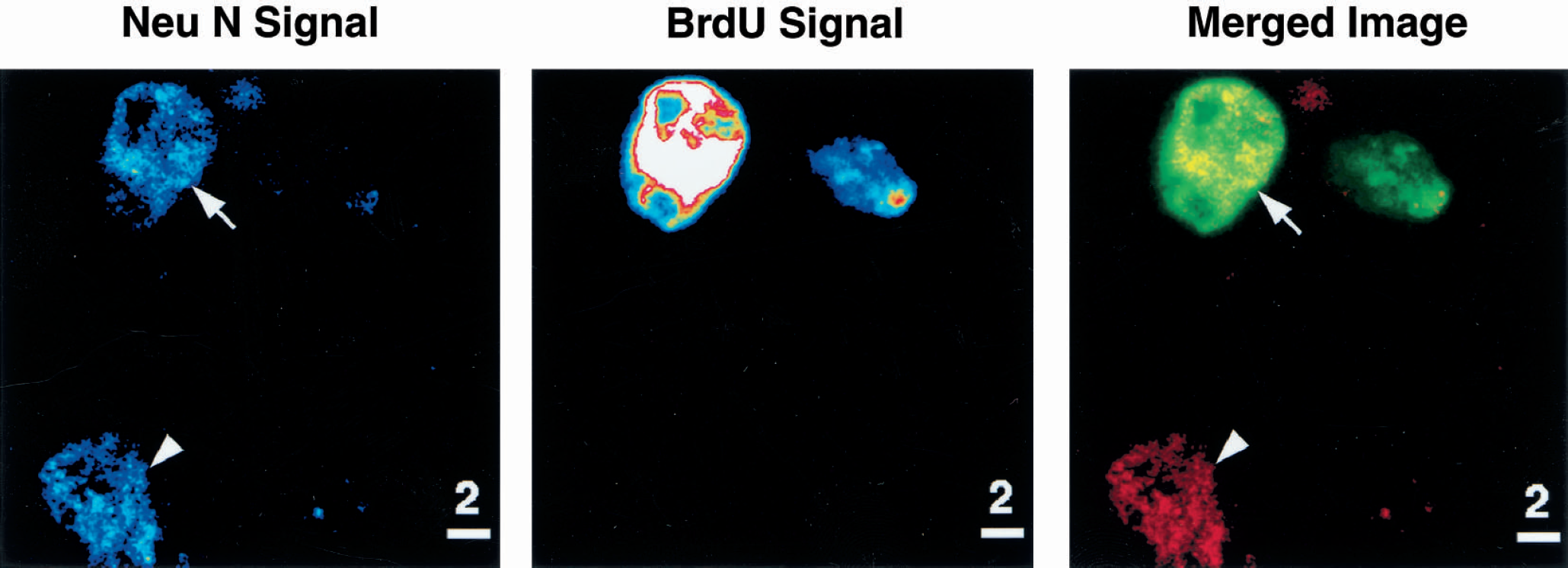
Three-dimensional confocal analysis of Neu N+ BrdU double-labeling of cortical cells at 72 hours after stroke induction. The left column shows the signal intensity for Neu N, the middle column displays signal intensity for BrdU, and the right column exhibits merged image in which Neu N is pseudo-colored in red (cy3) and BrdU in green (DTAF) with the merged color appearing as yellow. White arrow points to a cortical cell observed in layer II of the region-at-risk that was double-labeled by Neu N (left and right) and BrdU (middle and right) with similar nuclear patterns. Arrowhead points to a nearby Neu N positive (left and right), but BrdU-negative (middle and right) cell, that is, a mature neuron.
DISCUSSION
The current study showed that a proportion of cells in the cortical region-at-risk was double-labeled by the cell proliferation marker BrdU and one of the mature neuronal markers Map-2 or Neu N after reversible photothrombotic stroke. BrdU cell labeling has long been a standard method to identify cell proliferation. The thymidine analogue BrdU is incorporated into cell DNA during the S-phase of a cell cycle, which can be detected immunohistochemically (Gratzner, 1982; Miller and Nowakowski, 1988). The combination of anti-BrdU and anti-Map-2 cell labeling has been widely used for the study of neurogenesis (Zigova et al., 1998). Widespread BrdU-incorporation into neurons is observed during the embryonic stage and shortly after birth (Menezes and Luskin, 1994). In discrete brain regions—that is, the subventricular zone and the dentate gyrus of hippocampus—neurogenesis is maintained in adult animals and humans (Eriksson et al., 1998; Kempermann et al., 1997). Neurogenesis in the dentate gyrus of hippocampus is further influenced by aging, enriched environment, epileptic status, and global cerebral ischemia (Kempermann et al., 1997; Kuhn et al., 1996; Liu et al., 1998; Parent et al., 1997). Constant addition of new neurons in the neocortical association area in normal adult macaques has also recently been reported (Gould et al., 1999). Neural stem cells isolated from the adult brain may also proliferate and develop into mature neurons when cultured in epidermal growth factor containing media (Morshead et al., 1994). Transplantation of neural stem cells into the striatum of normal adult rats revealed survival, migration, and integration of thymidine (3H-thy)-labeled cells into the adjacent tissue; 1% to 3% of these cells differentiated into neurons (Lundberg et al., 1997). Survival and integration of fetal transplants into infarcted host brain tissue have also been reported (Borlongan et al., 1997). Thus, the persistence of BrdU-immunopositive mature neurons in the adult rat cortex after reversible photothrombotic stroke, as demonstrated in the current study, suggests cortical neurogenesis. Because Map-2 isotype c may also be induced in glial cells after stimulation by EGF (Rosser et al., 1997), thus confounding clear-cut interpretation of the results, we used Map-2a,b and the neuron-specific marker Neu N to identify neurogenesis (Gould et al., 1999). To rule out the fortuitous occurrence of cortical neurogenesis by possible juxtaposition of BrdU-positive cells of other origin onto Neu N-immunoreactive neurons, we used three-dimensional confocal analyses (Kuhn et al., 1997). With this technique not a single BrdU-incorporated neuron was observed in the cortex in sham-operated BrdU-delivered rats, which agrees with a study performed in adult rat cortex (Kuhn et al., 1997). This is in contrast with the observation in adult macaques in which new neurons are constantly generated in the neocortical association cortex (Gould et al., 1999). Thus, this difference may be species-related. In the current study, three-dimensional confocal micrographs of cells at 30 days after ischemia revealed the coexistence of the cell proliferation-specific marker BrdU with the neuronal-specific marker Neu N within the same cells. Different cellular labeling patterns by BrdU and Neu N signals were demonstrated within the same cell at this time point in which BrdU labeled the newly generated cell nucleus, whereas Neu N marked the nuclear part and the cytoplasmic part. The Neu N cellular labeling pattern observed in the current study agreed with the literature (Wolf et al., 1996). The earliest time point after stroke onset in which BrdU and Neu N double-labeled neurons appeared in the cortical region-at-risk was at 72 hours postirradiation. This time frame was congruent with observations that it takes only 24 hours for stem cells to differentiate in vitro into neurons expressing Map-2 (Svendsen et al., 1995), and that Neu N is expressed immediately after terminal division from progenitors by ventral horn motor neurons and cerebellar granule cells (Mullen et al., 1992). In adult macaques, newborn neurons expressing Neu N were identified 7 days after BrdU delivery (Gould et al., 1999).
The origin of these cortical newborn neurons is unknown. Multipotent stemlike cells from the adult striatum cultured in vitro (Reynolds and Weiss, 1992) and derived from the subventricular zone (SVZ) or adjacent regions (Johansson et al., 1999; Luskin, 1993) have been shown. Similar cells have been demonstrated in the dentate gyrus of hippocampus (Morshead et al., 1994). Under physiologic conditions, newborn cells strictly follow a stereotyped pathway as they migrate from the SVZ or adjacent regions to their final destination in the olfactory bulb (Luskin, 1993). Multipotent stem cells have also been isolated from other regions of the adult central nervous system including the spinal cord (Weiss et al., 1996), other deep brain structures (Gage et al., 1998), and the septum and striatum (Palmer et al., 1995). A recent study provides evidence that ciliated ependymal cells in the central nervous system may be the primary source of adult stem cells in the SVZ (Johansson et al., 1999). In response to spinal cord injury, proliferation of these ependymal cells increased substantially to generate migratory cells that differentiated into astrocytes, but not neurons, (Johansson et al., 1999), which participated in scar formation. In the current study, we did not observe any clear signs of temporal migration of the newborn cells from the ependyma or SVZ to the injured part of the cortex. This may be because of the different type of injury induced presently compared with that of Johansson et al. (1999) and a different general response to injury in the spinal cord versus in the neocortex. Recent data show that neural stem cells isolated from adult rat cerebral cortex can generate neurons after exposure to FGF-2 (Palmer et al., 1999). Therefore, it is conceivable that the newborn cortical neurons observed in the current study originated from quiescent potential neural stem cells that already reside within the neocortex. This is further supported by the finding that FGF expression is induced in the ischemic brain tissue after stroke (Finklestein et al., 1990). A long-distance migration of stem cells from the SVZ in the current experimental setup seems unlikely because neurogenesis was observed in cortical layers II-III already at 72 hours after stroke induction.
The newborn neurons accounted for about 3% of the total newborn cell population in the postischemic cortex at 7 days. It increased to approximately 6% at 100 days, together with a decline of the total of BrdU single-labeled cells in the same regions. This is likely explained by the fact that most macrophages observed at 7 days after stroke onset had disappeared at 100 days. Interestingly, the proportion of newborn cortical neurons in relation to the total number of BrdU-incorporated cells in the current study was in the same range as that reported after stem cell transplantation to adult striatum of nonlesioned rats (Lundberg et al., 1997).
Because BrdU is available for only two hours for cell uptake after each injection (Nowakowski et al., 1989), repeated BrdU intraperitoneal injections were used in the current study to maximize the opportunity to label the cells undergoing proliferation (Gould et al., 1999). To minimize the possibility of any potential neurotoxic effect of BrdU, repeated BrdU injections of 10 mg/kg at each delivery were used as compared with 75 to 120 mg/kg used elsewhere (Craig et al., 1999; Gould et al., 1999). The total amount of BrdU delivered per rat in the current study was 560 mg/kg in 100 day rats and 260 mg/kg in 30 day rats. These doses are well within the range commonly used for neurogenesis studies (Craig et al., 1999; Gould et al., 1999). Agreeing with these studies, no cytotoxic effects were observed in the current study.
Theoretically, BrdU may be incorporated into the cell nucleus as a result of DNA repair. However, if such incorporation occurred, it should be very limited, as discussed elsewhere (Liu et al., 1998), rather than the extensive BrdU-incorporation detected in the current study. BrdU-incorporation, as revealed by semiquantitative signal intensity in the confocal image, was usually equal or stronger in neurons as compared with neighboring newborn cells of nonneuronal lineage. Thus, our findings strongly support the contention that the BrdU-immunopositive cortical neurons represent newborn cells rather than cells repairing their DNA. To our knowledge, no in vivo experiments have ever reported that neurons surviving an injury evince BrdU-incorporation. The traditional dogma that neurons cannot be regenerated in the adult cerebral cortex in mammals after brain injury may be questioned.
Footnotes
Acknowledgments
The authors thank Dr. Anders Björklund, Brant Watson, and Anders Bergh for helpful criticism of this manuscript, Wei Jiang for technical consultation, and Dr. Roland Rosqvist for excellent guidance on the confocal microscopy.
