Abstract
Cyclic nucleotides are important hemodynamic regulators in many tissues. Glyceryl trinitrate markedly dilates large cerebral arteries and increases cGMP. Here, the authors study the effect of sildenafil, a selective inhibitor of cGMP-hydrolyzing phosphodiesterase 5 on cerebral hemodynamics and headache induction. Ten healthy subjects were included in a double-blind, placebo-controlled crossover study where placebo or sildenafil 100 mg (highest therapeutic dose) were administered on two separate days. Blood velocity in the middle cerebral artery (Vmca) was recorded by transcranial Doppler, and regional cerebral blood flow in the perfusion area of the middle cerebral artery (rCBFmca) was measured using single photon emission computed tomography and 133xenon inhalation. Radial and temporal artery diameters were studied using high-frequency ultrasound. Blood pressure and heart rate were recorded repeatedly. Headache responses and tenderness of pericranial muscles were scored verbally. Sildenafil caused no significant changes in rCBFmca, Vmca, or in temporal or radial artery diameter, but heart rate increased and diastolic blood pressure decreased significantly compared to placebo. Despite the lack of cerebral arterial dilatation, sildenafil caused significantly more headache than placebo. The present results show that sildenafil 100 mg does not dilate cerebral or extracerebral arteries but nevertheless causes headache, which may be attributed to nonvascular mechanisms.
Keywords
Sildenafil is a new highly selective inhibitor of the cGMP-degrading intracellular enzyme phosphodiesterase 5 (PDE5) widely used as treatment for erectile dysfunction. Sildenafil potentiates the actions of endogenous nitric oxide released during sexual stimulation from the endothelial cells or parasympathetic nerves (Ballard et al., 1998). Through inhibition of PDE5, the major enzyme responsible for cGMP degradation in that tissue, the accumulation of cGMP leads to relaxation of the smooth muscle cells, which in the penile tissue causes erection.
In cerebral arteries from different animals, the presence and activity of the Ca2+/calmodulin-stimulated PDE1, the cGMP-inhibited PDE3, the AMP-degrading PDE4, and the cGMP-degrading PDE5 have been described (Kruuse et al., 2001; Willette et al., 1997). Several studies have investigated the role of PDEs in the regulation of cerebral blood flow and cerebral artery diameter in animals using selective inhibitors of PDE (Kawamura et al., 1985; Parfenova et al., 1993). Of the selective PDE inhibitors, only PDE3 inhibitors have been studied using cerebral blood flow measurements, and no in vivo studies have investigated the ability of selective PDE inhibitors to dilate cerebral arteries.
Since species variability has been shown for the distribution and function of PDEs and since PDEs and their regulatory effects on cyclic nucleotide levels may be potentially important in cerebral hemodynamics, we studied the effects of sildenafil on cerebral hemodynamics. Furthermore, the headache-generating potential of sildenafil was evaluated since headache is one of the major side effects of this compound (Morales et al., 1998). Cerebral vasodilatation due to the accumulation of cGMP in the smooth muscle cells of the arteries has previously been proposed as the mechanism of headache induction by sildenafil (Morales et al., 1998).
MATERIALS AND METHODS
The study design was a double-blind, placebo-controlled crossover study with balanced, random oral administration of sildenafil 100 mg or placebo on 2 separate days, at least 1 week apart. Ten healthy subjects were included, 4 women and 6 men, mean age 24 years (range 20 to 31 years), with mean body weight of 77.3 kg (range 73.5 to 87 kg), and with no medical history of cerebrovascular or cardiovascular disorders, migraine, hypotension, or hypertension. When asked, no subjects reported having first-degree relatives (parents, siblings, children) suffering from migraine. All subjects gave informed consent before inclusion, and a medical examination, including an electrocardiogram, was performed before the study. The study was approved by the ethical committee of Copenhagen County and the Danish health authorities and was conducted according to the Helsinki II declaration.
Procedure on the day of the study
No food, medication, coffee, tea, alcohol, chocolate, or tobacco was allowed for the last 8 hours before the examination that began at 8:30 a.m. A brief medical examination was performed. After 30 minutes of rest, baseline values of blood velocities, blood pressure, muscle tenderness, heart rate, and regional cerebral blood flow in the perfusion area of the middle cerebral artery (rCBFmca) were measured. A nontransparent gelatin capsule containing either sildenafil or placebo was ingested in a double-blinded fashion. The subject was kept for observation and measurements for a period of 120 minutes. The subjects remained in the supine position during the entire examination in quiet surroundings.
Single photon emission computed tomography (SPECT) acquisitions were performed at baseline, at 60 minutes, and at 120 minutes. Simultaneous transcranial Doppler (TCD) and radial artery diameter measurements were performed at baseline and every 15 minutes during the observation period. Each TCD measurement was followed by measurements of temporal artery diameter. TCD measurements were carried out immediately after SPECT acquisitions. Blood pressure and heart rate were measured repeatedly. Headache including side effects were recorded every 15 minutes, and the tenderness of the pericranial muscles was scored every 60 minutes during the observation period (120 minutes).
Transcranial Doppler
A time-averaged mean of the maximal blood velocities in the middle cerebral artery (Vmca) was recorded bilaterally by transcranial Doppler (TCD) (2 MHz, Multidop X Doppler, DWL; Lübech & Sipplingen, Germany; Transscan, EME, Uberlingen, Germany) using handheld probes (Thomsen and Iversen, 1993). The average of 4 measurements comprising approximately 4 cardiac cycles each over a time interval of 30 seconds was used. A fixed point for measurements of Vmca was chosen along the middle cerebral artery as the point that was as close as possible to the bifurcation between the middle cerebral and the anterior cerebral artery. This fixed point was then used throughout the study in each individual, and every measurement was done after carefully optimizing the signal from this point.
Measurements of end-tidal P
Single photon emission computed tomography
Measurements of CBF were performed with 133Xe inhalation and SPECT using a brain-dedicated camera—Ceraspect (DSI, Inc., Waltham, MA, U.S.A.) with a stationary annular NaI crystal and a fast rotating collimator system. Each rotation took 10 seconds, acquiring 1 frame in a 30-frame dynamic protocol of 133Xe inhalation (3 background, 9 wash-in, 18 washout) using the Kanno-Lassen algorithm (Kanno and Lassen, 1979). Thirty-two slices were reconstructed in a 64 × 64 matrix with each pixel measuring 0.33 × 0.33 cm. A set of eight transaxial slices was generated by adding four slices together to a total slice thickness of 1.33 cm. Attenuation correction using the Chang algorithm (μ = 0.05 cm) and nose-artifact correction were performed. The output in each pixel was the k1 value. The k1 was corrected with a partition coefficient (Λ) = 0.85 mL/g to get CBF values (mL·100 g−1·min−1).
A Datex Normocap 200 (Dameca, Rødovre, Denmark) was used for end-tidal P
Since transcranial Doppler was performed on the middle cerebral artery, the distribution area for the middle cerebral artery was chosen as ROI for further analysis. Mean blood flow in the rCBFmca for each side was calculated, and a mean of left and right rCBFmca values was analyzed. Global CBF (gCBF) was calculated as an average of all regions of interests.
Diameter of temporal and radial artery
The temporal and radial artery diameters were measured by a high-resolution ultrasound unit (20 MHz, bandwidth 15 MHz; Dermascan C, Cortex Technology, Hadsund, Denmark) (Nielsen et al., 1993). Marks were drawn on the skin to ensure that the repeated measurements were performed in the same place and to ensure reproducibility in measurements from day to day. The diameter of the temporal artery was measured at the frontal branch of the superficial temporal artery on the side of the dominant brain hemisphere assessed by hand dominance (on left side when right-handed).
Measurements of temporal artery diameter were not performed in 3 of the 10 subjects (hence, n = 7 in the results) because of technical difficulties.
Measurements of the radial artery were made on the left arm proximal to the distal volar wrist crease where maximal pulsation of the artery was felt by palpation.
Headache recordings
Headache intensity was scored every 15 minutes on a verbal scale from 0 to 10, where “1” represents a very mild headache (including a feeling of pressing or throbbing), “5” a headache of moderate intensity, and “10” the worst headache imaginable (Iversen et al., 1989). Headache characteristics, location, and other side effects were recorded. All subjects were asked to record their headache, headache characteristics, and any intake of medication as well as other side effects every hour at home on a headache diary card from 3 to 12 hours after drug administration.
Tenderness score
Bilateral palpation of 8 pairs of muscle and tendon insertion (masseter, temporal, frontal, sternocleidomastoid and trapezius muscles, coronoid and mastoid processes, and neck muscle insertions) was performed at baseline, 60 minutes, and 120 minutes. Tenderness of pericranial muscles was recorded according to the Total Tenderness Scoring system on a 4-point scale (0–3) (Langemark and Olesen, 1987). Summed values of left and right side were summed to a total tenderness score for analysis.
Statistics
All values are presented as means ± SDs. P < 0.05 was considered significant.
The area under the curve for each variable, Vmca, CBF, rCBFmca, blood pressure, heart rate, and P
RESULTS
Hemodynamics
There was no significant difference between the area under the curve for rCBFmca (P = 0.43) or gCBF (P = 0.58) between the two treatments. Baseline values for gCBF were 46.5 ± 8.7 mL·100 g brain tissue−1 · min−1 on placebo and 47.3 ± 9.5 mL·100 g brain tissue−1 ·min−1 on sildenafil and showed no change over time compared to baseline on either day (P = 0.67 and P = 0.94, sildenafil and placebo, respectively). Baseline value for rCBFmca was 45.8 ± 10.1 mL·100 g brain tissue−1·min−1 on placebo and 45.7 ± 7.5 mL·100 g brain tissue−1·min−1 on sildenafil, also without changes from baseline after the two treatments (P = 0.70 after sildenafil and P = 0.66 after placebo).
End-expiratory P
Blood velocity in the middle cerebral artery (Vmca) did not change significantly over time on the placebo day (P = 0.33) or the day of sildenafil (P = 0.30), and the area under the curves did not differ between treatments (P = 0.91) (Fig. 1).
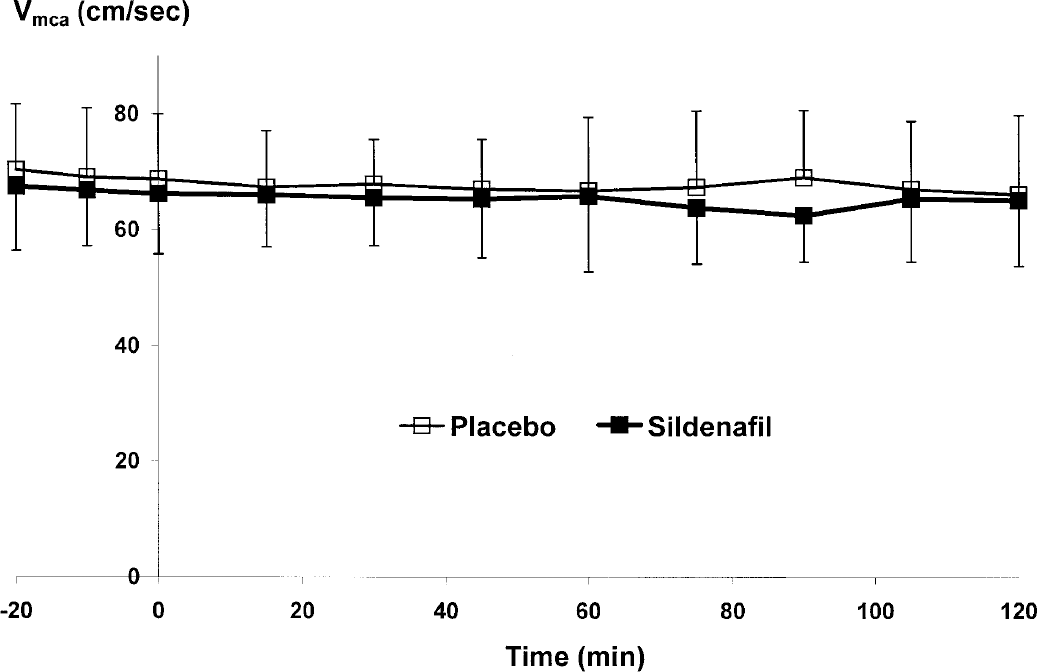
Mean (n = 10) blood velocity in the middle cerebral artery (Vmca) over time. Absolute values (± SD) of the mean blood velocity are presented after sildenafil (represented by filled squares, ■) and after placebo (represented by open squares, □). No significant difference in responses between the two treatments was seen. Since regional cerebral blood flow remained unchanged, sildenafil did not dilate the middle cerebral artery.
According to the following equation: rCBF = velocity x lumen area, Vmca may be taken as an indirect measure of change in diameter of the middle cerebral artery when rCBF remains unchanged (Sorteberg, 1992). Sildenafil 100 mg induced a mean 0.8 ± 3.3% increase in diameter after 1 hour and mean 1.9 ± 3.6% increase after 2 hours. The mean dilatory response after placebo was 1.9 ± 4.5% after 1 hour and 2.3 ± 4.2% after 2 hours (P = 0.58 and P = 0.82, 1 and 2 hours, respectively, in comparisons between treatments).
Radial artery diameter was unchanged by sildenafil compared to placebo (P = 0.42). The temporal artery diameter increased compared to baseline at 60 minutes (P = 0.03) after sildenafil, but not at other time points, and the difference between the area under the temporal diameter curve for sildenafil and placebo was not significant (P = 0.39)(Fig. 2).
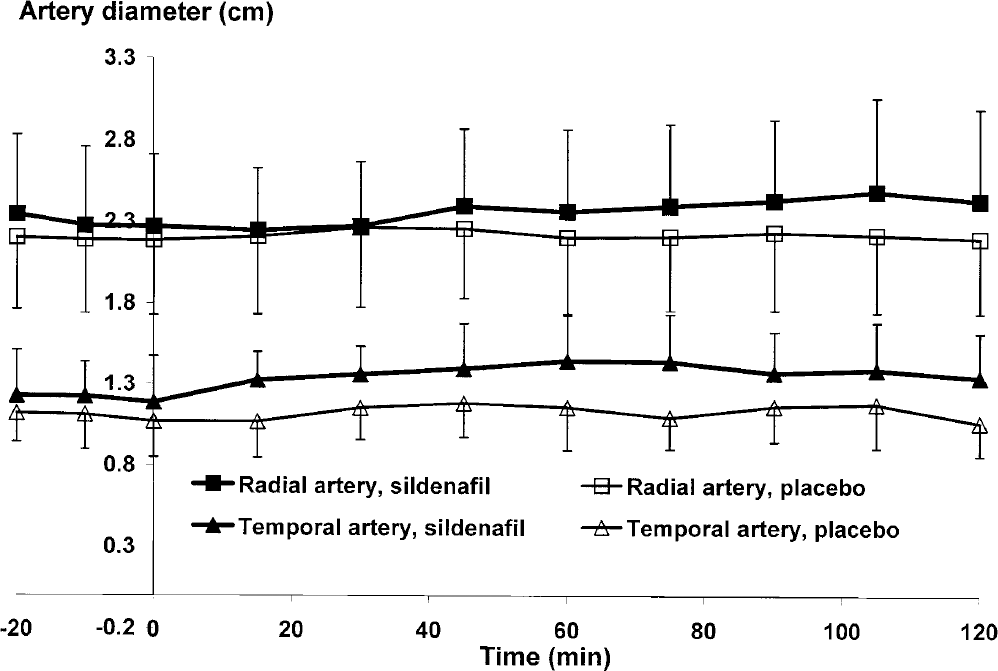
Temporal (n = 7) and radial (n = 10) arterial diameter over time. Mean absolute values (± SD) of temporal (represented by triangles, ▲) and radial artery diameter (represented by squares, ■) are shown after intake of placebo (by open symbols, Δ and □) and after sildenafil (by filled symbols, ▲ and ■). The temporal artery increases significantly (P = 0.03) at time 60 minutes after sildenafil; however, the change was very minor and was not significantly different from placebo.
Diastolic blood pressure decreased from mean values of 60 ± 5 mm Hg at baseline to 56 ± 6 at 90 minutes (P = 0.01) and heart rate increased from a mean value of 55 ± 9 beats per minute at baseline to a maximum of 64 ± 10 (P = 0.02) beats per minute 60 minutes after sildenafil. Although the change was small, it was significantly different from that seen on the day of placebo (P = 0.01 for diastolic blood pressure and P = 0.02 for heart rate). Systolic blood pressure was not significantly affected compared to placebo (P = 0.16) (Fig. 3).
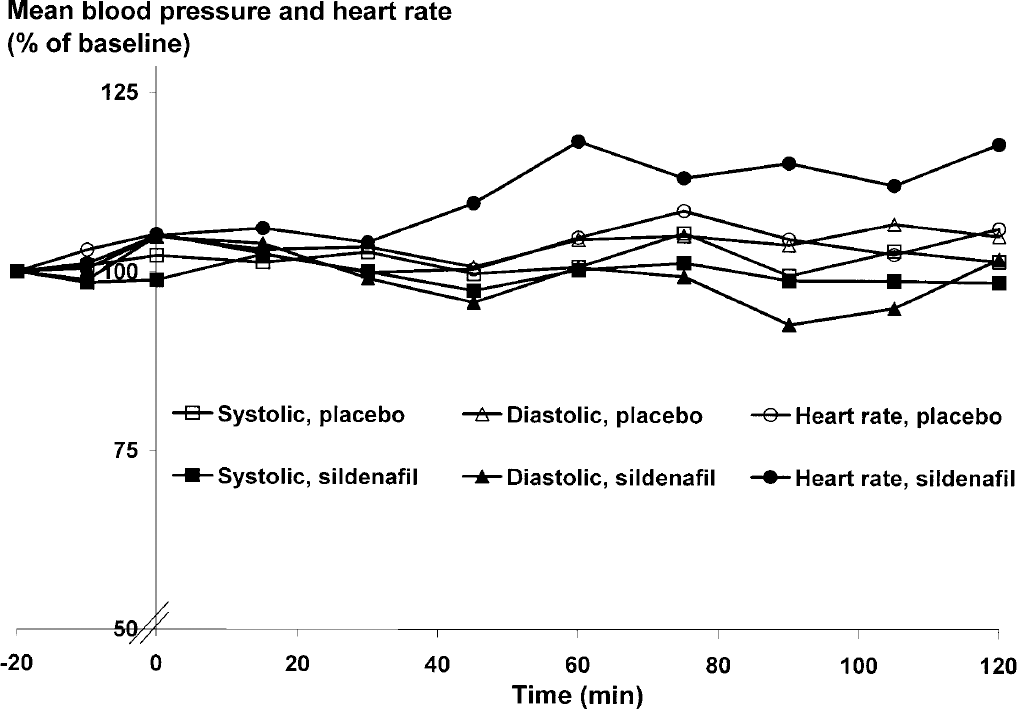
Mean values (n = 10) of heart rate (●), systolic (■), and diastolic (▲) blood pressure are shown in percent of baseline over time after placebo (open symbols) and sildenafil (filled symbols). Systolic blood pressure remained unchanged (P = 0.16), diastolic blood pressure decreased slightly (P = 0.01), and heart rate increased (P = 0.02) after ingestion of sildenafil compared to placebo.
Headache and other side effects
Sildenafil induced headache in nine subjects compared to three after placebo (P = 0.031). Median peak headache score after placebo was 0 (range 0 to 3) and after sildenafil 2.5 (range 0 to 6) (P = 0.007)(Figs. 4A and B). Area under the headache curve was significantly different between sildenafil and placebo (P = 0.008). Median time-to-peak headache score was 2 hours from administration of sildenafil, but a second peak was seen also at 6 hours (Fig. 4B).
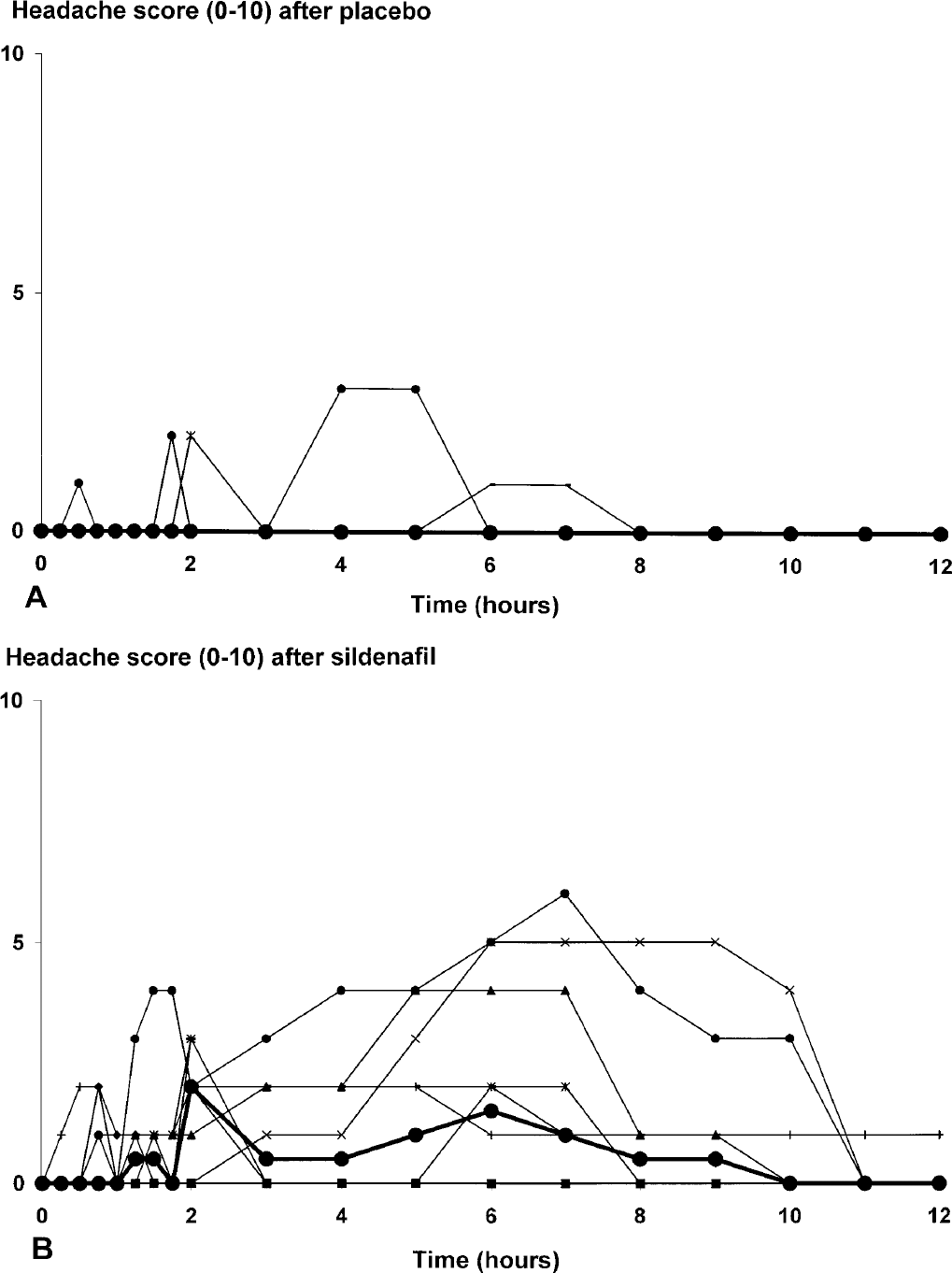
Individual (thin lines) and median headache scores (thick line with filled circle, ●) after placebo
Three subjects fulfilled the IHS criteria for a single attack of migraine without aura including accompanying symptoms such as nausea 5 to 7 hours after sildenafil administration but none after placebo. Headache characteristics and accompanying symptoms for each subject on the day of sildenafil are shown in Table 1.
Headache characteristics in healthy subjects after the intake of sildenafil
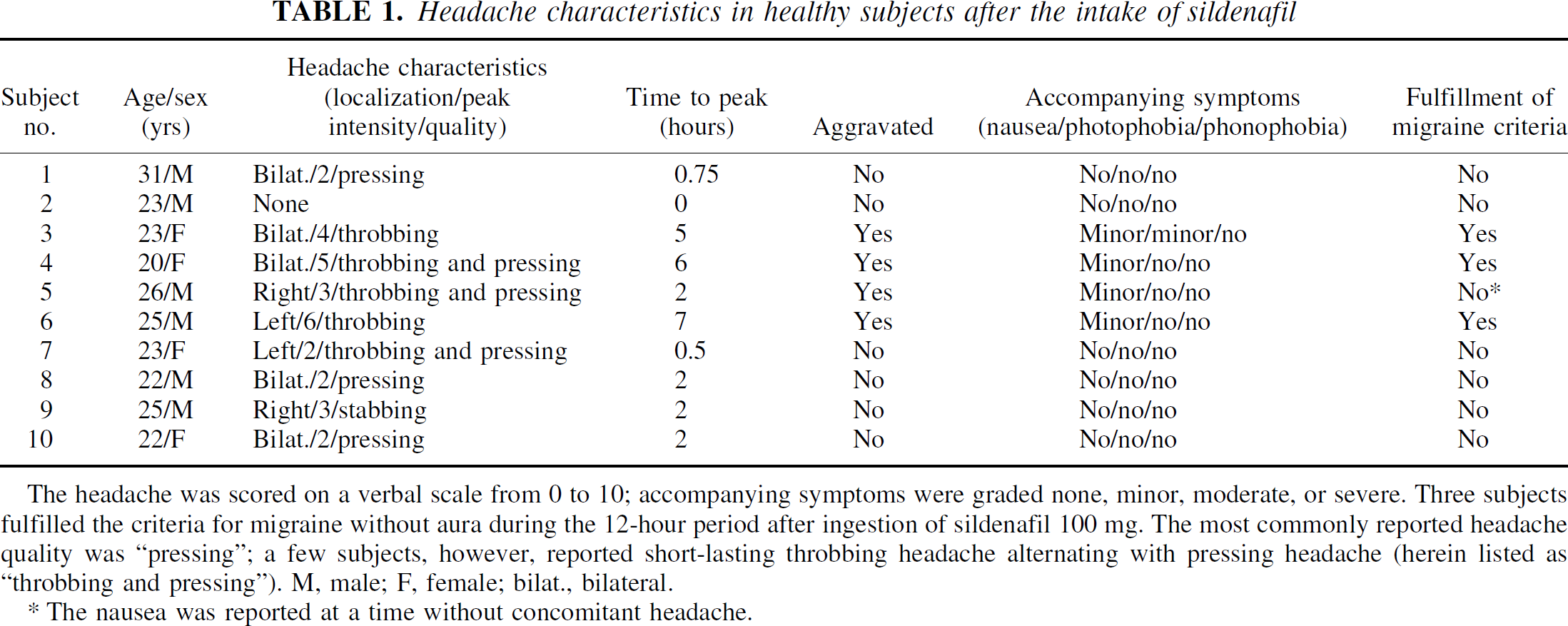
The headache was scored on a verbal scale from 0 to 10; accompanying symptoms were graded none, minor, moderate, or severe. Three subjects fulfilled the criteria for migraine without aura during the 12-hour period after ingestion of sildenafil 100 mg. The most commonly reported headache quality was “pressing”; a few subjects, however, reported short-lasting throbbing headache alternating with pressing headache (herein listed as “throbbing and pressing”). M, male; F, female; bilat., bilateral.
The nausea was reported at a time without concomitant headache.
Flushing was present in all subjects after sildenafil and in three after placebo. Nine reported a feeling of warmth after sildenafil and three after placebo, while two had palpitations with sildenafil and one had palpitations with placebo.
The subjects were asked to report any effects on sexual functions. Only two of six male subjects reported increased frequency of erection on the day of sildenafil, and none reported any effects on the placebo day. The female subjects reported no sexually related effects of sildenafil.
Tenderness of pericranial muscles
No difference in total tenderness score was seen between placebo and sildenafil (P = 0.14). The median baseline values were 4.5 (range 2 to 29) on the day of placebo and 4 (range 1 to 31) on the day of sildenafil.
DISCUSSION
The effects of inhibitors of phosphodiesterases on cerebral blood flow have been studied during the last decades in relation to treatment of stroke, dementia, and cerebral vasospasm, using nonselective and nonspecific inhibitors such as pentoxifylline and papaverine (Kruuse et al., 2000a). More recently, selective PDE3 inhibitors such as cilostazol, olprinone, and milrinone (Arakawa et al., 2001; Kobayashi et al., 1985; Yu et al., 2000) and to smaller degree PDE5 inhibitors, such as dipyridamole (Kruuse et al., 2000b) have been investigated in cerebral vascular disease.
In the present study, we demonstrate that inhibition of cGMP degradation by the selective PDE5 inhibitor sildenafil had no significant effects on cerebral blood flow and diameters of cerebral and extracerebral arteries. However, sildenafil was able to induce headache in healthy subjects without concomitant cerebral large artery dilatation.
Phosphodiesterase 5 distribution and sites of action of sildenafil
Phosphodiesterase 5 is present in penile vascular smooth muscle cells and has recently been reported to be present in brain tissue, mostly cerebellum and hippocampus and in the superior cervical ganglion (Giordano et al., 2001; Giorgi et al., 1994; Loughney et al., 1998). PDE5 has also been shown to be present and active in the guinea pig basilar artery (Kruuse et al., 2001) and in human cerebral arteries (unpublished data).
Oral administration of sildenafil 100 mg results in a peak plasma concentration of 1 μg/mL after approximately 1 hour (Jackson et al., 1999). The previously described intracellular increase of cGMP in smooth muscle cells and enhancement of relaxation by nitric oxide (NO) occurs well within this plasma concentration of sildenafil (Ballard et al., 1998; Jeremy et al., 1997). Sildenafil is the most selective PDE5 inhibitor available for human use, the mean IC50 being 3.5 nmol/L for the isolated PDE5 enzyme. For the retinal PDE6, the mean IC50 was 33 nmol/L and 281 nmol/L for PDE1, but the potency against PDE2, PDE3, and PDE4 is very low (Ballard et al., 1998).
Apart from the effect on smooth muscle cells, sildenafil may also have a central effect in the brain. Inhibitors of PDE5 increase cGMP concentrations in certain brain areas (Van Staveren et al., 2001) and it was found that sildenafil enhances auditory attention in healthy subjects suggesting a central effect of this lipophilic drug (Schultheiss et al., 2001).
This is consistent with previous studies, showing that sildenafil also acts on sensory nerve fibers in stimulating penile erection (Ballard et al., 1998; Medina et al., 2000) and modulates central NO-cGMP pathways in the rat brain involved in erection (Sato et al., 2001). It remains to be investigated whether activation of sensory nerve fibers is also taking place in humans. However, in light of the aforementioned data this seems very likely.
Cyclic nucleotides, phosphodiesterases, and cerebral vasodilatation
The endothelium of cerebral arteries has a basic production of NO, which through activation of soluble guanylate cyclase produces cGMP. Modulation of both cGMP and cAMP elicits essential regulation of cell signaling and probably cerebral vasodilatation through protein kinases, ion channels, and intracellular Ca2+ (Faraci and Sobey, 1999; Parfenova et al., 1993). The intracellular concentration of the cyclic nucleotides is regulated through their rate of production by adenylate cyclases or guanylate cyclases, through degradation by phosphodiesterases, or through efflux from the cell (Beavo, 1995).
Nitric oxide may cause an increase not only in cGMP but also in cAMP in some tissue, because the increase in cGMP inhibits PDE3 and thus increases intracellular cAMP in vitro (Sandner et al., 1999). This effect of NO has not been verified in cerebral arteries so far, and, at least in guinea pig cerebral arteries, the NO donor, sodium nitroprusside, did not increase cAMP levels (Kruuse et al., 2001). Upregulation of phosphodiesterases may also modulate the effects of NO. Thus, continuous infusion of NO donors in rats induced tolerance to the vasodilating properties of NO on isolated rat aortas by upregulation of PDE1A1, thus increasing the Ca2+/calmodulin-stimulated cGMP degradation (Kim et al., 2001).
As mentioned previously, the presence of several PDE families, PDE1, PDE3, PDE4, and PDE5 have been found to be active in cerebral arteries of animals (Kruuse et al., 2001; Willette et al., 1997). The presence and importance of the different PDE families has, however, not yet been established in human cerebral arteries. In studies on animals, most nonselective and selective PDE inhibitors induce cerebral artery dilatation, although with different potency depending on species and vessels tested. Selective PDE3 inhibitors show effective endothelial independent relaxation, whereas PDE1, PDE4, and PDE5 inhibitors elicit endothelial-dependent relaxation (Kruuse et al., 2001; Parfenova et al., 1993; Rosenblum et al., 1993).
Table 2 (Kruuse et al., 2000a, b ; Bowton et al., 1988; Nishimura et al., 1993; McHenry et al., 1983; Wang and Obrist, 1976; Schmidt and Waldemar, 1990; Yu et al., 2000; Ito et al., 1999; Beavo, 1995; Stoclet et al., 1995; Fujishige et al., 1999; Fawcett et al., 2000) gives an overview of the previously reported effects of PDE inhibitors on CBF, blood velocity, and general hemodynamics as well as the occurrence of headache in healthy subjects. Recently, milrinone, which is mainly a PDE3 inhibitor, was shown to reverse cerebral vasospasms and possibly improved clinical outcome in patients with sub-arachnoidal hemorrhage (Arakawa et al., 2001).
Effects of phosphodiesterase inhibitors on cerebral hemodynamics in healthy subjects

The above compounds are considered inhibitors of phosphodiesterases (PDE) (Beavo, 1995; Stoclet et al., 1995), although some are not only nonselective and inhibit more than one PDE family but also nonspecific and thus have other effects than PDE inhibition. Table lists reported significant changes in different hemodynamic parameters and headache after administration of the PDE inhibitors compared with placebo in healthy subjects. Some studies have not investigated (ND, not done), or do not report, all parameters. PDE1 is the Ca2+/calmodulin-stimulated PDE; PDE3 is inhibited by cGMP and hydrolyzes cAMP at a higher rate than cGMP. PDE5 is specific for cGMP hydrolysis. Dipyridamole is considered an inhibitor of PDE5, but it has recently also been described as inhibiting the isolated enzymes of PDE10 (present in brain; Fujishige et al., 1999) and PDE11 (present in skeletal muscle; Fawcett et al., 2000), which hydrolyze both cAMP and cGMP.
cAMP, cyclic adenosine monophosphate; cGMP, cyclic guanine monophosphate; CBF, cerebral blood flow; Vmca, middle cerebral artery blood velocity; P
Theoretically, sildenafil should be an equally effective vasodilator but, unexpectedly, it had no effect in the present study. The lack of a dilatory response to sildenafil in cerebral arteries and arterioles in healthy subjects may indicate that cGMP is not accumulated in cerebral smooth muscle or that a compensatory regulatory mechanism very effectively decreases cGMP. This could be through a negative feedback mechanism, where an increased activity of PDE5 overruled the effect of PDE5 inhibition or where an upregulation of PDE1 is initiated. Another PDE may also be responsible for lack of dilatation, most likely PDE10A that degrades both cAMP and cGMP, with cGMP degradation being inhibited by cAMP. It is also inhibited by dipyridamole and is present in brain tissue (Fujishige et al., 1999). Another explanation may be that the PDE5 in smooth muscle cells is a splice variant, which only to a minor degree is inhibited by sildenafil, but not enough to elicit vasodilatation. The expression of different splice variants of PDE5 is known in the human penile cavernosum (Lin et al., 2000), but the functional importance of this remains to be investigated.
Mechanisms of nitric oxide- and sildenafil-induced headache
Nitric oxide is proposed to induce headache either by stimulating pain-sensitive nerve fibers around cerebral arteries or by arterial dilatation (Olesen et al., 1993). Similar to NO donors, dipyridamole, an adenosine reuptake inhibitor and a much less selective inhibitor of PDE5 than sildenafil, induces headache concomitant to a dilatation of large cerebral arteries in healthy subjects, but there was a temporal dissociation between headache and arterial dilatation. It was not possible to conclude which effect of dipyridamole was responsible for the headache, except that it was due to mechanisms related to cyclic nucleotides, either directly by increased cGMP or indirectly by adenosine-stimulated increase in cAMP (Kruuse et al., 2000b).
Sildenafil induced headache in 9 of 10 healthy subjects compared with 3 of 10 after placebo. The major difference in the sildenafil-induced headache compared with headache induced by NO donors was the apparent lack of effects on cerebral hemodynamics in the number of subjects studied. Thus, no clear dilatation of the cerebral arteries was seen after sildenafil on the same number of subjects used previously to show dilation by NO donors and histamine.
Sildenafil may theoretically have an effect on veins; however, in previous studies it seems to be equally modest to the effect on arteries (Jackson et al., 1999; Wallis et al., 1999). Also, in a previous study on migraine patients during attack of migraine, Queckenstedt's maneuver was performed to increase the pressure on the venous sinuses, without causing an increase in the headache pain. This speaks against a role of dilating venous sinuses in the migraine pain (Daugaard et al., 1998).
Three subjects fulfilled the IHS criteria for one attack of migraine without aura, although they had never experienced migraine before, and reported having no first-degree relatives suffering from migraine. The only major difference compared to migraine was in the quality of headache. In migraine a throbbing headache is dominant, while sildenafil predominantly induced headache of pressing quality.
The frequency of induced headache by sildenafil was considerably higher in the present study than reported in previous safety data (Morales et al., 1998). This is most likely due to a lack of focus on headache in the previous studies.
In parallel with the present study, we have studied the effects of sildenafil in migraine sufferers. It caused no change in cerebral blood flow or cerebral artery diameter, but nevertheless induced migraine attacks.
The present results, where headache without significant dilatation of cerebral arteries was induced by an inhibitor of a cGMP degrading enzyme, make it likely that NO induces headache through cGMP-related mechanisms and not by a direct action of NO on the pain-sensitive perivascular nerves. The marked NO-induced dilatation of large cerebral arteries may be an epiphenomenon rather than a pain-activating mechanism, because the headache-generating effects of sildenafil do not seem to be via intracerebral or extracranial arterial dilatation.
Footnotes
Acknowledgements
We thank the subjects for participating and enduring the provoked pain. We also thank L. Elkaer, K. Enghave, and O. Jonassen for technical assistance, and H. Dige-Petersen for generous use of equipment. Pfizer A/S, Denmark, generously supplied the sildenafil tablets.
