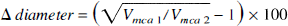Abstract
Dipyridamole is used for secondary prophylaxis in ischemic stroke and as a vasodilator agent in myocardial scintigraphy. An important side effect to administering dipyridamole is headache. The aim of the current study was to investigate the effects of dipyridamole on cerebral blood flow, large artery diameter, and headache induction. Twelve healthy subjects were included in this single-blind placebo-controlled study in which placebo (0.9% NaCl) and dipyridamole 0.142 mg/kg·min were administered intravenously over 4 minutes 1 hour apart. Blood flow velocity in the middle cerebral artery (Vmca) was recorded by transcranial Doppler and regional cerebral blood flow in the middle cerebral artery (rCBFmca) was measured using single photon emission computed tomography and 133Xenon-inhalation. Blood pressure, heart rate, and pCO2 were measured repeatedly. Headache response was scored every 10 minutes on a verbal scale from 0 to 10 (10 = worst). Dipyridamole caused a decrease in pCO2 (P < 0.001). pCO2 corrected rCBFmca was 41.7 ± 6.9 mL/100 g ·min after placebo versus 41.2 ± 6.9 after dipyridamole (P ≥ 0.05). pCO2 corrected Vmca decreased 8.4% ± 11.7 (P < 0.001) after dipyridamole, indicating a mean 5.6% ± 6.7 (P = 0.005) relative increase of the arterial diameter. After dipyridamole the median peak headache score was 2 (range 0 to 7) compared with 0 (range 0 to 3) after placebo (P = 0.02). Dilatation of the middle cerebral artery outlasted the headache response. In conclusion, dipyridamole causes a modest pCO2 independent dilatation of the MCA, which is time-linked to the onset, but not to the cessation, of headache.
Dipyridamole is used clinically for secondary prevention in ischemic cerebrovascular disease (Diener, 1998). Intravenous administration of dipyridamole combined with thallium-201 myocardial perfusion imaging is also used as a nonexercise cardiac stress test in patients unable to perform a physical stress test (Laarman et al., 1988).
Dipyridamole has two main mechanisms of action: inhibition of adenosine reuptake increasing the circulating level of adenosine and inhibition of phosphodiesterases (PDEs) leading to an increase in cyclic guanosine monophosphate (cGMP) and cyclic adenosine monophosphate (cAMP) (FitzGerald, 1987; Sakuma et al., 1990). Dipyridamole has been reported to have effects on the cerebral circulation in animals, but its effect on the cerebral circulation in humans has not been fully elucidated.
The most common side effect of dipyridamole both after oral and intravenous administration is headache, which has been reported in 37% of subjects after oral administration and 25% of subjects after intravenous infusion (Diener, 1998; Laarman et al., 1988). This headache inducing ability of dipyridamole has not been described systematically in a prospective study, and the exact characteristics of the headache and its possible mechanisms have not been evaluated. Previous experimental studies have shown that nitric oxide (NO) releasing compounds, or NO donors, capable of inducing headache, also dilate large cerebral arteries without a concomitant change in cerebral blood flow (Dahl et al., 1989; Lassen et al., 1995; Thomsen et al., 1993). Nitric oxide causes a rise in cGMP in the vascular smooth muscle cell. An increase in cGMP can also be achieved by inhibition of cGMP hydrolyzing PDEs through compounds like dipyridamole. The aim of the current study was to examine the effects of dipyridamole on regional cerebral blood flow (rCBF) in the middle cerebral artery territory (rCBFmca) by 133-Xenon single photon emission computed tomography (SPECT) and the effects on mean blood flow velocity in the middle cerebral artery (Vmca), and furthermore, to quantify, characterize, and time any headache caused by dipyridamole, and relate it to the observed hemodynamic changes. Such a study may not only elucidate the mechanisms of dipyridamole-induced headache, but it may also contribute toward understanding the mechanisms of vascular headache in general.
MATERIALS AND METHODS
Design and material
The current study was a single-blind placebo-controlled study in which placebo (0.9% NaCl) was administered intravenously over 4 minutes. On the same day, 1 hour after placebo infusion, dipyridamole 0.142 mg/kg·min was administered intravenously for 4 minutes.
The subjects were blinded to the order of treatments. In addition, the investigators analyzing the CBF data were also blinded to the design of the study and were unaware of the order of infusions. Preliminary studies had shown that the effect of dipyridamole on blood flow velocity lasted more than 1 hour, therefore the order of infusions was the same for each subject to avoid a carry-over effect of dipyridamole. The study was performed using intravenous administration of dipyridamole to avoid possible variation in bioavailability (FitzGerald, 1987). A dose similar to the dose used for routine cardiovascular performance tests was chosen for the study.
Twelve healthy subjects, seven women and five men, were included (age range 18 to 50 years, mean = 30.1 years). The subjects had no medical history of cerebrovascular or cardiovascular disorders, migraine, hypo-or hypertension, and had a headache frequency of less than 2 days a month. All subjects gave written informed consent before inclusion and a medical examination, including an ECG, was performed before the study. The study was approved by the ethical committee of Copenhagen County and the Danish health authorities and was conducted according to the Helsinki II declaration.
Headache recordings
During the study, headache intensity was scored every 10 minutes on a verbal scale from 0 to 10, where 1 represents a very mild headache (including a feeling of pressing or throbbing), 5 a headache of moderate intensity, and 10 the worst headache imaginable (Iversen et al., 1989). Also, headache characteristics, headache location, and other side effects were recorded. All subjects were asked to record their headache, headache characteristics, and any intake of medication every hour at home on a headache diary card from 2 to 10 hours after the dipyridamole infusion. The subjects were allowed to treat their headache at home with plain analgesics. Time for intake and drugs used were recorded in the headache diary card.
Blood flow velocity
A time-averaged mean of the maximal blood velocities in the middle cerebral artery (Vmca) was recorded bilaterally by transcranial Doppler (2 MHz, Multidop X Doppler, DWL; Lübech & Sipplingen, Sipplingen, Germany) using handheld probes (Thomsen et al., 1993). The average of 4 measurements comprising approximately 4 cardiac cycles each over a time interval of 30 seconds was used. A fixed point for measurements of Vmca was chosen along the MCA as the point that was as close as possible to the bifurcation between the MCA and the anterior cerebral artery. This fixed point was then used throughout the study in each subject, and every measurement was done after carefully optimizing the signal from this point. Measurements of pCO2 were performed simultaneously with the transcranial Doppler measurements (POET; Criticare Systems, Waukesha, WI, U.S.A.) using a mask placed lightly over the nose and mouth.
Cerebral blood flow measurements
Measurements of CBF were performed with 133-Xenon inhalation and SPECT using a brain-dedicated camera, Ceraspect (DSI, Waltham, MA, U.S.A.), with a stationary annular NaI crystal and a fast rotating collimator system. Each rotation took 10 seconds, acquiring 1 frame in a 30-frame dynamic protocol of 133Xenon-inhalation (3 background, 9 wash-in, 18 wash-out) using the Kanno-Lassen algorithm (Kanno and Lassen, 1979). Thirty-two slices were reconstructed in a 64 × 64 matrix with each pixel measuring 0.33 × 0.33 cm. A set of 8 transaxial slices was generated by adding 4 slices together to a total slice thickness of 1.33 cm. Attenuation correction using the Chang algorithm (μ = 0.05 cm) and nose-artefact correction was performed. The output in each pixel was the k1 value. The k1 was corrected with a partition coefficient (λ) = 0.85 mL/g to get CBF values (mL/100 g·min).
A Datex Normocap 200 (Dameca, Rídovre, Denmark) was used for end-tidal CO2 measurements during CBF acquisition. A Xenamatic 4000 (Diversified Diagnostic Products, Houston, Texas, U.S.A.) was used for 133Xenon-administration. The measurement lasted 5 minutes. A standard vascular region of interest set of 5 slices positioned 3.6, 5.0, 6.3, 7.6, and 9.0 cm above the cantho-meatal plane was used.
Because transcranial Doppler was performed on the MCA, the distribution area for the MCA was chosen as a region of interest for further analysis. Mean blood flow in the perfusion area of MCA for each side was calculated, and a mean of left and right rCBFmca values was analyzed. Global CBF was calculated as an average of all regions of interest.
Procedures
The subjects arrived without headaches at 8:30 in the morning on the day of the study. No medication, coffee, tea, alcohol, tobacco, or chocolate were allowed for at least 8 hours before start of the examination. After a short medical examination, including an ECG, a cannula was inserted in the cubital vein for infusion of placebo and dipyridamole. The subjects were kept in supine position during the study. After 30 minutes rest, baseline measurements of blood pressure, heart rate, pCO2, Vmca, and CBF were performed. To avoid accumulation of radioactivity, causing high background readings during CBF measurements, a 40-minute interval was included between baseline CBF measurement and the next CBF measurement, which was performed immediately after a 4-minute placebo infusion. Sixty minutes after start of the placebo infusion, dipyridamole was infused and a third CBF measurement was performed at the end of infusion. After another interval of 1 hour, the final CBF measurement was performed. Vmca was recorded every 20 minutes from 50 minutes before infusion of placebo to end of the final CBF measurement. In connection with the CBF measurements, the Vmca measurements were performed immediately at the end of each CBF measurement. Headache and headache characteristics were scored every 20 minutes from start of placebo infusion.
Blood pressure and heart rate were measured repeatedly every 5 minutes using an automatically inflated arm-cuff. Electrocardiogram was recorded every 10 minutes during the study.
The estimated perfusion area (rCBFx) of a given artery (x) in the brain depends on the mean blood flow velocity (Vx) and the cross-sectional area π(r2) of the artery (Dahl et al., 1989; Sorteberg, 1992). Based on this assumption an estimate of the relative change in the cross-sectional area of the large artery can be made when rCBFx and VX is known. When the cerebral blood flow is unchanged the following equation is valid (Dahl et al., 1989):
Δ diameter is the relative percentage change in diameter, Vmca1 is the blood flow velocity in the MCA before infusion of drugs, and Vmca2 is the velocity at a relevant time point after infusion of the drug.
To circumvent the influence of the difference in pCO2 over time, and because the measurements were not done simultaneously, rCBFmca and Vmca were corrected for pCO2 changes from baseline. Cerebral blood flow and rCBFmca were corrected by 2% per mm Hg change in pCO2 (Shirahata et al., 1985) and Vmca with an exponential factor of 0.034 mm Hg (Markwalder et al., 1984).
Statistics
Statistical analyses were performed on absolute values. Values are presented as means ± SD, with headache values as median and (range). P < 0.05 was considered significant.
Changes over time for each variable—Vmca, CBF, rCBFmca, calculated relative artery diameter change, blood pressure, heart rate, and pCO2—were analyzed with two-way analysis of variance (Statgraphics 3.3; Manugistics, Rockville, MD, U.S.A.) with factors time and subjects. The two-way analysis of variance was repeated for each single variable. If an overall effect of time was found, further analysis was performed using a multiple range test for location of significant changes (Statgraphics 3.3). Mean values were evaluated according to three time periods: baseline, after placebo, and after dipyridamole. These mean values, together with peak values and the calculated area under the curve for the two latter periods, were chosen as summary measures when analyzing difference in response between treatments. The calculated values were compared using a paired t-test (Statgraphics 3.3). Difference in headache response after placebo and dipyridamole (area under the curve and peak response) were analyzed using Wilcoxon Rank Sum Test (SPSS 9.0).
RESULTS
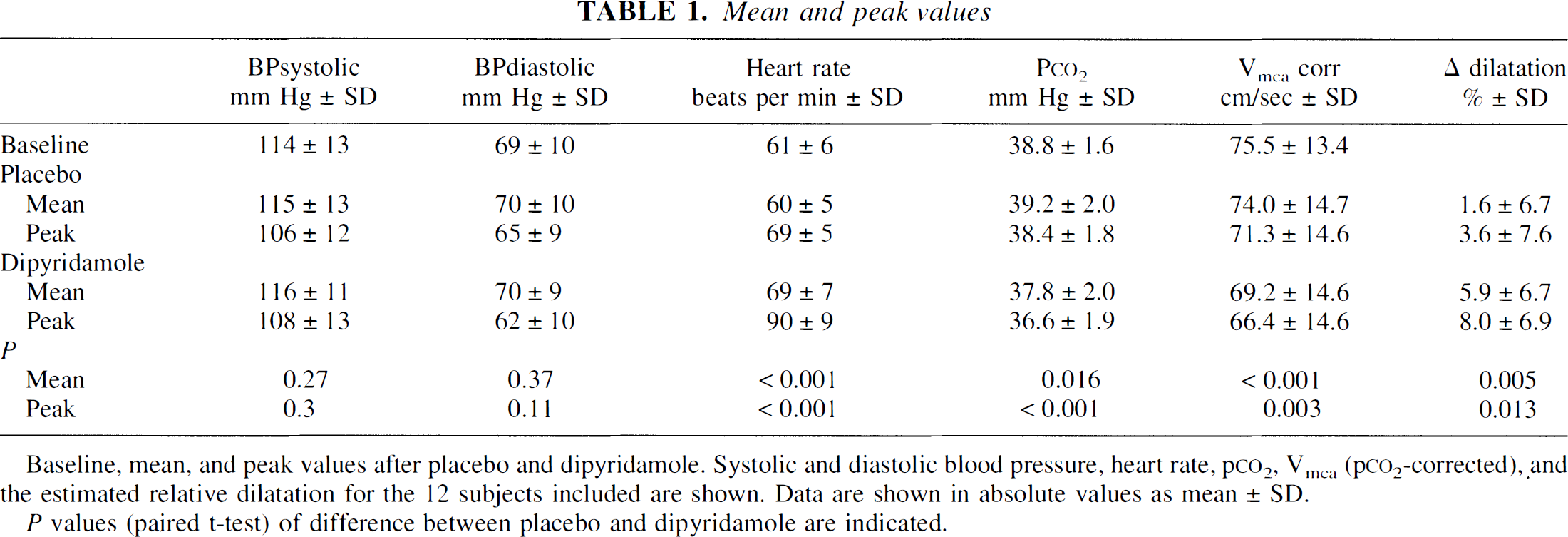
End-expiratory pCO2 decreased significantly (P < 0.001) over time after dipyridamole infusion compared with baseline, returning to baseline 40 minutes after infusion (Fig. 1). The mean and peak end expiratory pCO2 after dipyridamole was significantly different from that seen after placebo (P < 0.003;Table 1). Peak decrease in pCO2 from baseline was 0.4% ± 3.5 after placebo and 5.6% ± 3.3 after dipyridamole.
Mean and peak values
Baseline, mean, and peak values after placebo and dipyridamole. Systolic and diastolic blood pressure, heart rate, p
P values (paired t-test) of difference between placebo and dipyridamole are indicated.
Mean blood flow velocity in the middle cerebral artery (Vmca). Mean values for Vmca ± SD in percent of baseline (n = 12) are shown.
Mean rCBFmca was 41.2 ± 7.0 mL/100 g·min at baseline and global CBF was 42.0 ± 7.1. There was no significant change over time compared with baseline in rCBFmca (P = 0.26) or global CBF (P = 0.24) when values were corrected for the change in pCO2 induced by dipyridamole. Mean rCBFmca was 42.7 ± 7.4 mL/100 g·min after placebo versus 42.3 ± 7.6 after dipyridamole (P = 0.67). Corresponding values for global CBF was 43.3 ± 7.5 mL/100 g·min and 42.4 ± 7.5 mL/100 g·min (P = 0.40) for placebo and dipyridamole, respectively.
When no pCO2 correction was performed, a significant decrease from baseline in rCBFmca (P = 0.02) and global CBF (P = 0.01) was seen immediately after dipyridamole infusion, with values returning to baseline 1 hour after dipyridamole infusion. Without pCO2 correction, rCBFmca values after placebo were 42.7 ± 6.6 mL/100 g·min and after dipyridamole were 39.6 ± 6.7 mL/100 g /min (P = 0.009), and global CBF was 43.3 ± 6.7 after placebo and 39.8 ± 6.6 after dipyridamole (P = 0.005).
At baseline, mean Vmca was 75.5 ± 13.4 cm/second. Vmca decreased significantly after start of dipyridamole infusion and remained decreased until at least 1 hour after end of infusion (P < 0.01). This was seen both with and without correction for change in pCO2 (Fig. 1). Without correction, mean Vmca was 75.2 ± 14.6 cm/second after placebo and 67.8 ± 14.4 cm/second after dipyridamole. When correcting for change in pCO2, mean Vmca in the period after placebo infusion was 74.0 ± 14.7 cm/second and in the period after dipyridamole was 69.2 ± 14.6 cm/second (P < 0.001). This was equal to a mean decrease in velocity from baseline of 8.4% ± 11.7% after dipyridamole (P < 0.001) and 1.6% ± 13.2% after placebo (P = 0.3). Thus, compared with placebo mean and peak, values of Vmca were significantly decreased after dipyridamole (absolute values are shown in Table 1). Also, the calculated area under the curves was significantly decreased after dipyridamole (P = 0.001).
Using the pCO2 corrected values of Vmca (Dahl et al., 1989), the estimated mean relative dilatation from baseline was 1.6% ± 6.7% after placebo and 5.6% ± 6.7% after dipyridamole (P = 0.005); both mean and peak relative dilatation (P = 0.01) was significantly increased after dipyridamole compared with placebo (Table 1).
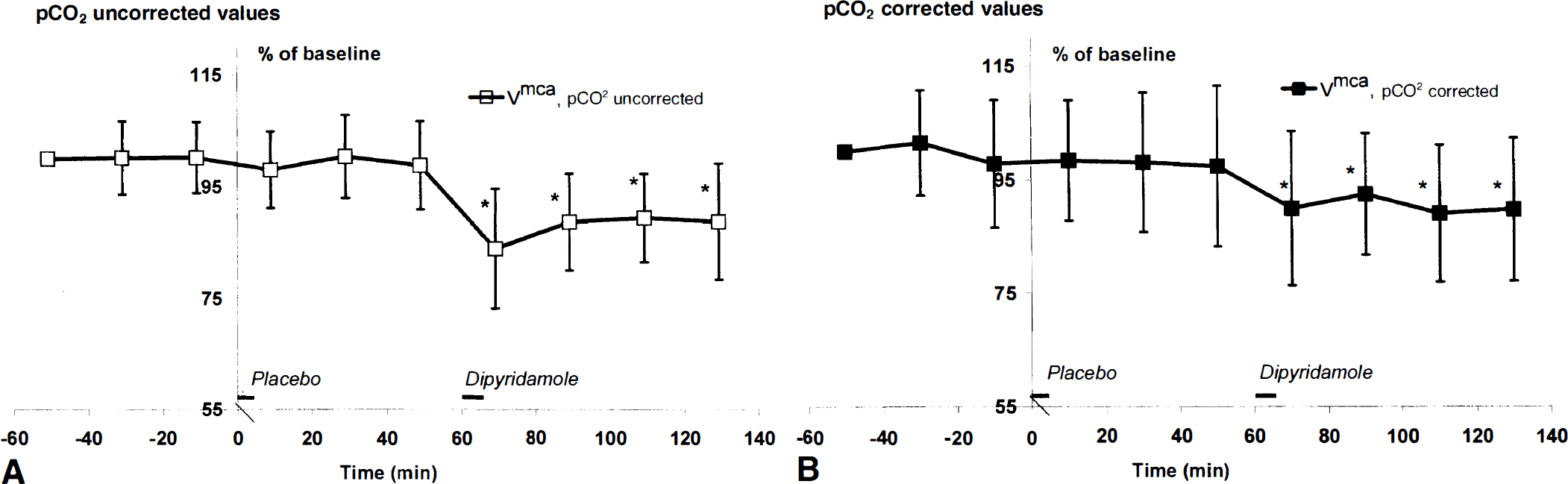
Diastolic and systolic blood pressures were unchanged over time (P = 0.11 and P = 0.1, respectively). In contrast, heart rate increased 43.6% ± 24.7% immediately after infusion of dipyridamole (P < 0.001) compared with baseline (Fig. 2). Mean and peak heart rate was significantly increased (P < 0.001) after dipyridamole compared with placebo, whereas systolic and diastolic blood pressure showed no significant difference (P ≥ 0.05;Table 1).
Mean values of blood pressure and heart rate during the study. Systolic, diastolic blood pressure, and heart rate shown as mean values over time in percent of baseline (n = 12). Values of 10-minute increments and during infusions are shown. Diamonds (♦) represent systolic blood pressure, squares (□) represent diastolic blood pressure, and triangles (▴) represent heart rate. Only heart rate changed significantly after dipyridamole infusion (* P < 0.01).
The area under the headache curve during the first 60 minutes postinfusion was significantly increased after dipyridamole compared with placebo (P = 0.01). Of 12 subjects, 4 reported a mild headache after placebo and 8 reported mild to severe headache after dipyridamole with peak intensity 10 to 20 minutes after infusion (Fig. 3). The median peak headache score after placebo was 0 (range 0 to 3), and the score after dipyridamole was 2 (range 0 to 7, P = 0.02). A second delayed peak in headache scores, exceeding the headache scores immediately after dipyridamole infusion, was reported in 3 subjects with peak intensity at a mean of 3 hours (range 2 to 5 hours) after dipyridamole infusion. A further 2 subjects reported a similar second headache peak, however with a lower headache score than reported after dipyridamole infusion, also occurring 3 hours (range 2 to 4 hours) after dipyridamole infusion.
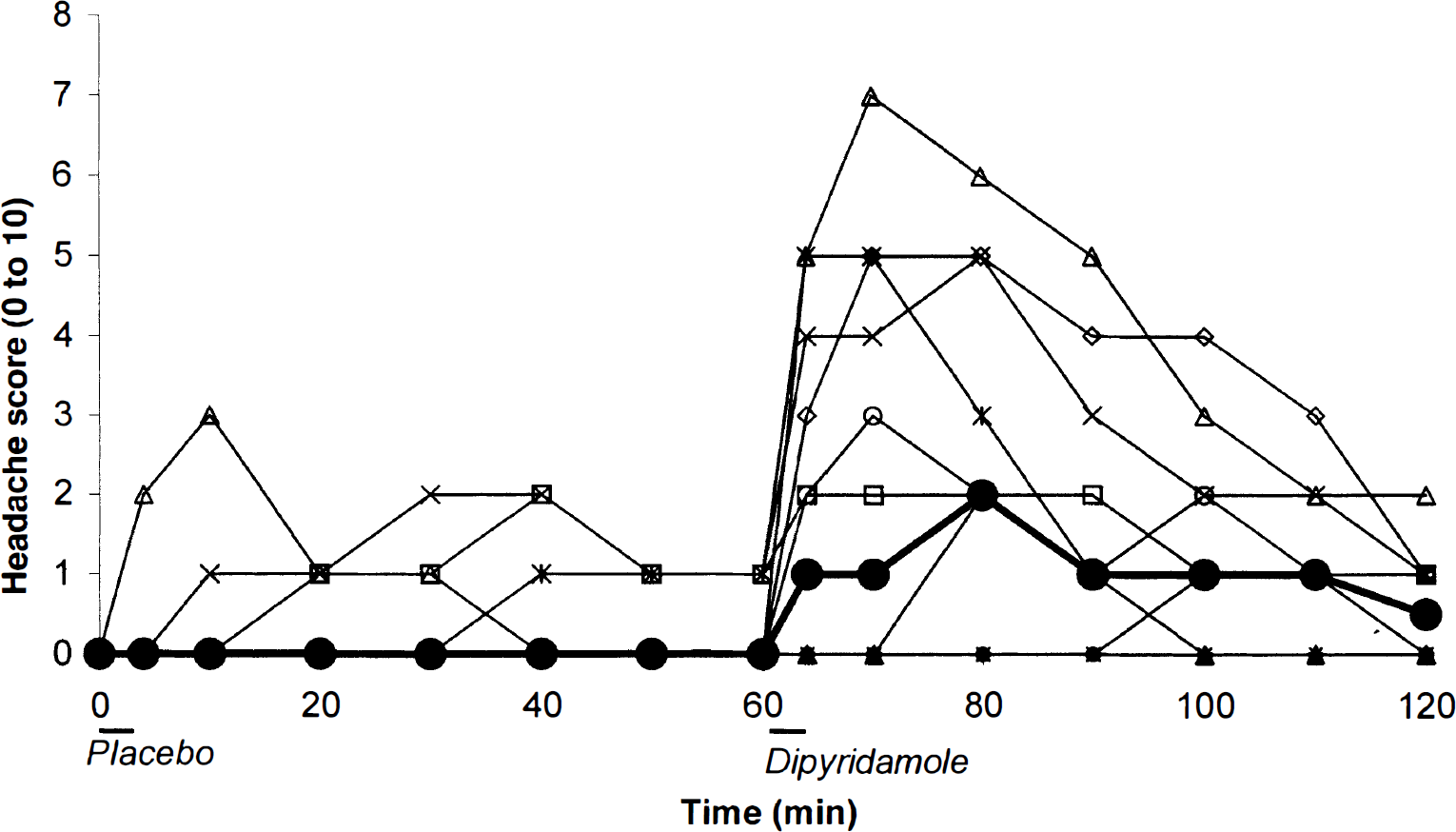
Median headache score over time. Individual headache scores are shown in thin lines. A thick line with filled circles (•) indicates the median headache score over time (n = 12). Significant difference in area under the curves was seen between placebo and dipyridamole treatment (P = 0.02).
The headache was bilateral and of pressing quality in 7 subjects and unilateral in 1 subject who also experienced throbbing pain and nausea. This subject fulfilled the criteria for migraine according to the International Headache Society classification (Headache Classification Committee, 1988), but only at one time-point 20 minutes after dipyridamole, migraine symptoms remitted without therapy; the subject had no family history of migraine. The remaining 7 subjects did not fulfil the migraine criteria at any time-point. Two of the subjects experiencing headache took rescue medication (plain analgesics) at home.
Of other side effects recorded, 11 out of 12 subjects showed objective flushing in the face after dipyridamole and only 1 experienced this after placebo. Eight subjects reported a feeling of warmth, 6 subjects experienced palpitations, and 6 subjects reported a feeling of heaviness in the body after dipyridamole. Electrocardiogram showed no signs of ischemia or arrhythmia during dipyridamole infusion. After placebo only 2 subjects reported palpitations, 1 subject reported a feeling of warmth, and 1 subject complained of nausea.
DISCUSSION
Dipyridamole and cerebrovascular hemodynamics
In the current study, global CBF and regional CBF in the middle cerebral artery territory (rCBFmca) decreased during dipyridamole infusion simultaneous to a decrease in end-expiratory pCO2. Previously, Ito et al. (1999) also observed a decrease in CBF and in pCO2 during dipyridamole infusion. As in the current study, the CBF decrease was thought to be because of hyperventilation attributed to adenosine reuptake inhibition, because adenosine stimulates pulmonary chemoreceptors. Therefore, the authors chose to correct Vmca, CBF, and rCBFmca for pCO2 changes. After correction dipyridamole had no effect on CBF or rCBFmca, whereas Vmca was still significantly decreased. This suggests dilatation of large conductance arteries.
Previous studies have revealed species differences of dipyridamole in the induced CBF response (Heistad et al., 1981). Thus, dipyridamole caused a 2.7-fold increase in CBF in rabbits, whereas no increase in CBF was found in cats and dogs (Heistad et al., 1981). Another study on dogs reported no change in CBF, but did report a decreased cerebral vascular resistance after dipyridamole. It was unclear whether this was a direct effect of dipyridamole on the cerebral resistance vessels or was because of autoregulatory dilatation secondary to a 20% decrease in blood pressure (Boarini et al., 1982). An increase in CBF was correlated to the plasma levels of cAMP but not to adenosine levels (Hegedus et al., 1997).
Topical application of very high concentrations (10 to 100 μmol/L) of dipyridamole dilates pial arterioles in rabbits (Heistad et al., 1981). Likewise, when administered intravenously, only concentrations greater than or equal to 0.7 mg/kg induced a significant increase in CBF (Hegedus et al., 1997). This dose far exceeds the doses that can be administered to humans. Thus, there are quite marked species differences in the cerebrovascular response to dipyridamole, which make human studies essential.
Based on the pCO2 corrected values showing a decrease in Vmca and an unchanged rCBFmca, a significant relative increase in the diameter of the MCA of 5.6% ± 6.7% was calculated. End-expiratory pCO2 returned to normal 40 minutes after dipyridamole, whereas Vmca remained significantly decreased until the end of the study and also during the last CBF measurement, which had returned to baseline. This indicates that the change in Vmca was caused by the drug and not by hyperventilation. Allowing for some species difference, the current findings of large artery dilatation and of no direct effect on CBF are largely compatible with previous findings in vitro and in vivo.
The effect of drugs on CBF are not always similar to their effects on large cerebral arteries (Dahl et al., 1989; Friberg et al., 1991). As demonstrated in the current study and in previous studies, these effects can be distinguished by combining quantitative CBF measurement and blood flow velocity measurement (Sorteberg, 1992). Using 133−Xenon inhalation rCBF measurement and a brain dedicated SPECT scanner provides reliable quantified rCBF measurements, although the spatial resolution is rather poor (Lassen, 1985). The latter is not a problem when evaluating drugs for their vasoreactivity, which in all likelihood is similar within each brain region. 133−Xenon SPECT involves only minor radiation exposure and therefore allows several repeated measurements, a requirement in pharmacologic studies. Transcranial Doppler is easy to handle, has a good reproducibility, and is without known risks for the patients (Thomsen and Iversen, 1993).
Mechanism of action
Pharmacokinetic studies of dipyridamole have revealed a tri-exponential decrease in plasma concentration over time, the first two steep decreases occurring within 4 to 6 hours after intravenous dipyridamole and the terminal half-life being approximately 12 hours (Bjornsson and Mahony, 1983; Sollevi, 1986). The half-life of the effects described below is not known in detail.
Two main mechanisms of action of dipyridamole on the vascular system have been proposed. First, it acts as an inhibitor of adenosine reuptake, increasing the extracellular level of adenosine. Adenosine in turn activates adenylate cyclase and causes a rise in cAMP. Secondly, dipyridamole inhibits phosphodiesterases (PDEs), primarily the cGMP-specific PDE5, thereby increasing intracellular cGMP (Beavo, 1995; FitzGerald, 1987). PDE5 is present in vascular smooth muscle and platelets (Beavo, 1995). Both oral administration and intravenous infusion result in levels of dipyridamole in humans that affect both mechanisms (Bjornsson and Mahony, 1983; FitzGerald, 1987).
Adenosine and cGMP elevating compounds have been shown to induce dilatation of human cerebral arteries (Dahl et al., 1989; Sollevi, 1986). Adenosine increases CBF in rabbits but causes no change in the CBF of dogs and cats (Heistad et al., 1981). Zaprinast, a selective PDE5 inhibitor increasing cGMP levels, did not change CBF or cerebral oxygen consumption in rats when it was applied topically (Wei et al., 1996), but did dilate guinea pig and human cerebral arteries in vitro (Kruuse et al., unpublished results).
A consensus has not been reached as to which effect of dipyridamole is the most important, and diverging results and theories have been put forward, especially concerning the antiaggregatory effect (FitzGerald, 1987; Muller et al., 1990; Sakuma et al., 1990). Adenosine and cAMP have been reported to be increased in plasma after dipyridamole (Hegedus et al., 1997; Sollevi, 1986). A rise in tissue levels of cGMP has also been shown in vitro after dipyridamole administration (Sakuma et al., 1990). Furthermore, dipyridamole enhances the effects of endogenous NO on platelet aggregation (Bult et al., 1991; Sakuma et al., 1990) and enhances the effect of inhaled NO on airway smooth muscle (Ziegler et al., 1998). Unfortunately, neither in CBF studies nor platelet studies have cAMP and cGMP after dipyridamole been consistently and simultaneously measured. At present, it can not be determined whether the dilatory effect of dipyridamole on the large cerebral arteries was because of inhibition of adenosine reuptake, PDE inhibition and a rise in cGMP, or the combined effect of both mechanisms.
Dipyridamole-induced headache
Migraine and vascular headache may be related to dilatation of cerebral and extracranial arteries (Friberg et al., 1991; Thomsen et al., 1995). However, vasodilatation alone seems to be too simplistic of an explanation and no universal agreement exists on this (Thomsen et al., 1995; Zwetsloot et al., 1993). The NO donor Glyceryl-trinitrate (GTN) and 5-isosorbid mononitrate (5-ISMN) can induce headaches in healthy volunteers and migraines in migraine patients concomitant to dilatation of cerebral and extracranial arteries while CBF remains unaltered (Dahl et al., 1989; Olesen et al., 1993). Similar findings have been made with histamine that stimulates endogenous production of NO through H1 receptors on the endothelial cells (Lassen et al., 1995). Nitric oxide induces an increase in cGMP through activation of the enzyme soluble guanylate cyclase. This eventually leads to smooth muscle cell relaxation and arterial dilatation. Some doubt has been raised whether headache is induced by stimulation of perivascular pain-sensitive nerve-fibers because of the arterial dilatation, or if NO diffuses through the smooth muscle cells and stimulates the pain-sensitive nerve-fibers directly (Thomsen et al., 1995).
Dipyridamole induces headache after oral and intravenous administration, but only the frequency and not the headache intensity, characteristics, or timing have been reported (Diener, 1998; Laarman et al., 1988). In the current study, 8 out of 12 subjects (67%) reported headache. Previously, in studies that did not focus on headache, the frequency of headache after intravenous administration was reported to be between 8% and 25% (Laarman et al., 1988). Headache occurred in 37.2% of stroke patients after administration of 2 × 75 mg slow release tablet daily (Diener, 1998), and 11 out of 12 healthy subjects reported headache after 200 mg oral dose twice daily (Bult et al., 1991). The variation in reported headache may be because of difference in doses of dipyridamole, age-related difference in cerebrovascular response, or the lack of a systematic registration of this side effect.
In the current study, the authors selected a single blind design to minimize the radiation exposure. Furthermore, the color of dipyridamole used for intravenous infusion (bright yellow) and the side effects of facial flushing were difficult to disguise for the investigator and made it impossible to double blind the study properly. The nonrandomized order of drug administration chosen reflects the limitations of studies including human subjects. The chosen design may be a confounding factor, which should be kept in mind when interpreting the results of the current study. However, because of the objective measurements used, the single blind design seems unlikely to have biased the hemodynamic results. The reports of delayed headache at the end of the study may have been partially biased by the subjects' knowledge of active treatment. Thus, less emphasis should be placed on these data in the interpretations of the effects of dipyridamole.
The decrease of blood flow velocity in the MCA of 8.4% in the current study is smaller than the 28% seen with GTN (0.5 μg/kg·min) (Thomsen et al., 1993). However, it is in the same range as seen during spontaneous migraine attacks (Thomsen et al., 1995). The onset of dipyridamole-induced arterial dilatation is time-wise correlated to the induced headache. Compared with headache induced by GTN, dipyridamole-induced headache started later, reached its peak more slowly, and lasted longer, which corresponds to the longer half-life of dipyridamole (Iversen et al., 1989; Olesen et al., 1993).
Although headache initiation after dipyridamole corresponded to the decrease in Vmca, headache intensity decreased before vasodilatation had diminished. Thus, arterial dilatation may mechanically activate perivascular pain sensitive nerves, but either the algogenic effect of vasodilatation is transient or endogenous antinociceptive mechanisms become temporarily activated to counteract the nociceptive input.
A delayed headache, defined as a second peak of headache, occurred several hours after dipyridamole infusion. The occurrence of delayed headache has also been reported after GTN in migraine patients and more rarely in healthy subjects (Iversen et al., 1989). In previous studies of GTN induced headache in normal subjects, delayed headache occasionally fulfilled the International Headache Society criteria for migraine without aura (Olesen et al., 1993) and this was seen in one subject in the current study.
A possible mechanism of the delayed headache may be a release of neurotransmitters such as calcitonin gene-related peptide, substance P, and NO during vasodilatation. The release may sensitize the perivascular nerve-fibers. Also, the initial volley of nociceptive impulses may sensitize central pain pathways. If one or both of these mechanisms becomes sufficiently activated, even normal pulsation in the artery may be perceived as painful. At the molecular level these effects may be related to the rise in cGMP caused by PDE5 inhibition because other headache-inducing compounds, such as GTN and histamine, are vasoactive and work through the NO-cGMP pathway. Understanding these mechanisms in further detail will not only shed light on dipyridamole-induced headache and cerebral artery dilatation, thereby offering possibilities of preventing the side effects related to the therapeutic use of this agent, but also will add useful information to the understanding of basic headache mechanisms.
Footnotes
Abbreviations used
Acknowledgements
The authors thank laboratory technicians Kirsten Enghave and Olga Jonassen for their skillful assistance.