Abstract
The study aimed to elucidate the effects of cortical ischemia and postischemic environmental enrichment on hippocampal cell genesis. A cortical infarct was induced by a permanent ligation of the middle cerebral artery distal to the striatal branches in 6-month-old spontaneously hypertensive rats. Bromodeoxyuridine (BrdU) was administered as 7 consecutive daily injections starting 24 hours after surgery and animals were housed in standard or enriched environment. Four weeks after completed BrdU administration, BrdU incorporation and its co-localization with the neuronal markers NeuN and calbindin D28k, and the astrocytic marker glial fibrillary acidic protein in the granular cell layer and subgranular zone of the hippocampal dentate gyrus were determined with immunohistochemistry and were quantified stereologically. Compared with sham-operated rats, rats with cortical infarcts had a five-to sixfold ipsilateral increase in BrdU-labeled cells. About 80% of the new cells were neurons. Differential postischemic housing did not influence significantly the total number of surviving BrdU-labeled cells or newborn neurons. However, postischemic environmental enrichment increased the ipsilateral generation of astrocytes normalizing the astrocyte-to-neuron ratio, which was significantly reduced in rats housed in standard environment postischemically.
Neurogenesis, or the generation of new nerve cells, is an inherent feature of the development of the central nervous system. However, in the adult mammalian brain, neurogenesis persists only in certain distinct regions such as the hippocampus (Kuhn et al., 1996) and the rostral subventricular zone from which newly generated neurons migrate via the rostral migratory stream to the olfactory bulb (Lois and Alvarez-Buylla, 1993; Luskin, 1993). Adult hippocampal neurogenesis has been demonstrated in many animal species including primates and humans (Eriksson et al., 1998; Gould et al., 1999; Kornack and Rakic, 1999). Cell proliferation takes place in the subgranular zone (SGZ) of the dentate gyrus, at the border between the granular cell layer (GCL) and hilus, where a pool of multipotent progenitor cells is located. They divide continuously and give rise to mostly neurons as well as astrocytes and oligodendrocytes (Palmer et al., 2000). Newborn neurons migrate into the GCL and establish synaptic contacts with the CA3 region of the hippocampus while newborn astrocytes show morphologic characteristics of radial glia (Stanfield and Trice, 1988; Cameron et al., 1993; Hastings and Gould, 1999; Markakis and Gage, 1999). The physiologic importance of adult neurogenesis has not yet been fully elucidated. It has, however, been shown that hippocampal neurogenesis is important for spatial learning and memory (Shors et al., 2001).
Adult hippocampal neurogenesis is a dynamic process that can be influenced by many factors, physiologic as well as pathologic. Housing in enriched environment has been shown to influence hippocampal neurogenesis. Studies have indicated that enriched environment increases neurogenesis in the dentate gyrus and improves spatial memory in intact animals (Kempermann et al., 1997, 1998a, b; Nilsson et al., 1999; Williams et al., 2001). Moreover, genetic factors can interact with environmental stimuli, and strain and gender differences have been observed in hippocampal cell proliferation and survival in mice (Kempermann et al., 1998b) and in rats (Perfilieva et al., 2001). Transient global cerebral ischemia, which primarily induces ischemic cell damage in the hippocampus, significantly increases cell proliferation in the SGZ (Takagi et al., 1999). These cells migrate into the GCL, where they become mature neurons (Liu et al., 1998; Kee et al., 2001; Yagita et al., 2001). Studies on the proliferation of progenitor cells in the dentate gyrus after transient occlusion of the middle cerebral artery (MCA), which induces striatal and cortical lesions but does not primarily involve the hippocampus, have also shown induced proliferation of progenitor cells in the dentate gyrus (Jin et al., 2001) and increased neurogenesis (Arvidsson et al., 2001).
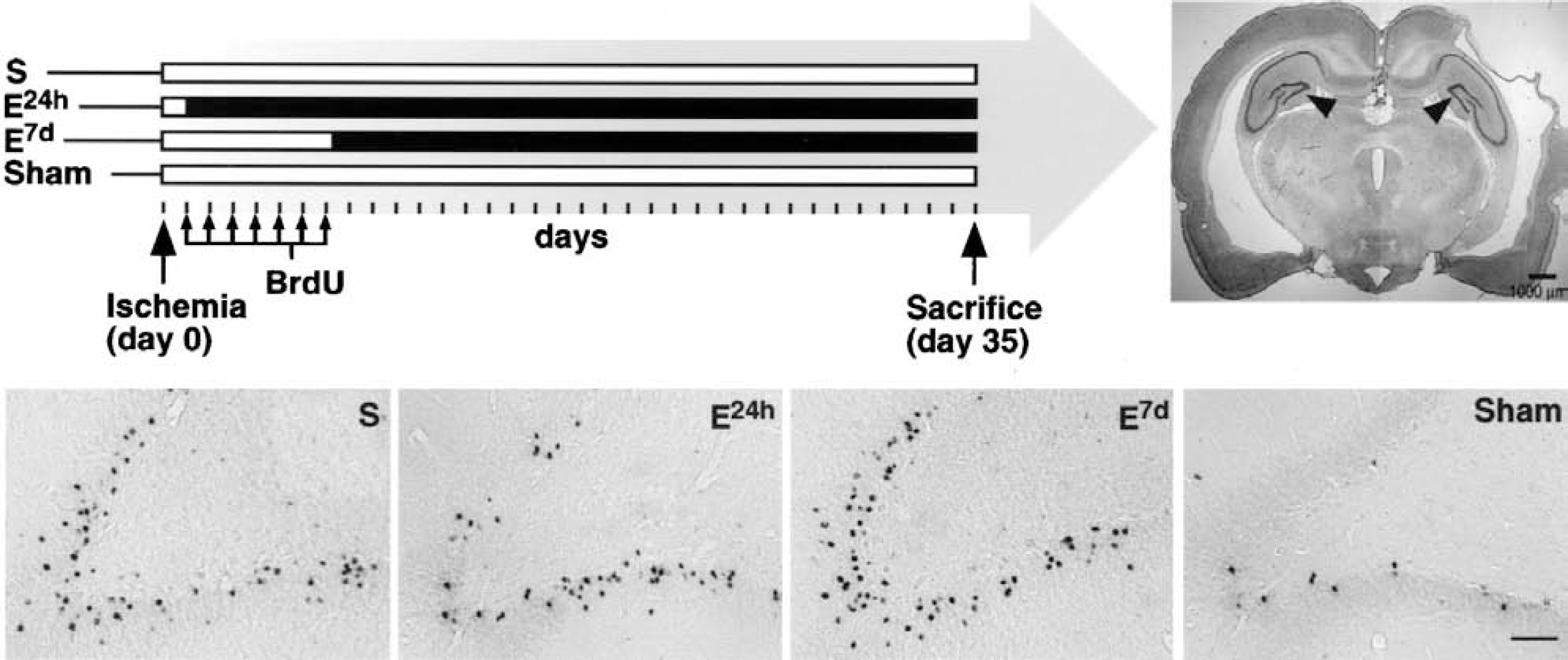
Our study aimed to elucidate the interactions between two important factors known to significantly influence hippocampal cell genesis, namely, ischemia and enriched environment. This has not been studied so far and could be potentially interesting for the interpretation of plasticity and repair processes in the central nervous system. Our hypothesis was that postischemic environmental enrichment, earlier shown to improve functional outcome after focal cerebral ischemia (Olsson and Johansson, 1995; Johansson, 2000), modulates cell genesis in the hippocampus. To obtain selective cortical infarcts, the MCA was ligated distal to the striatal branches in spontaneously hypertensive rats (SHR), which do not develop cerebral lesions spontaneously. However, chronic hypertension leads to a functional and structural adaptation of the vascular bed, resulting in a reduced collateral circulation and more consistent ischemic lesions distal to an arterial ligation (Duverger and MacKenzie, 1888; Grabowski et al., 1988, 1993). Thus, in contrast to other rat strains, SHR develop cortical infarcts after a selective ligation of the MCA distal to the striatal branches (Coyle, 1982). BrdU administration, starting 24 hours after surgery and given as seven daily injections, was used to label proliferating cells. Enriched environment was introduced at 2 points of time after ischemia: at 24 hours (i.e., in connection with the first BrdU injection) and at 7 days (i.e., after the last BrdU injection). The total number of BrdU-labeled cells in the hippocampus and their phenotype (neuronal or astrocytic) were analyzed 4 weeks after completed BrdU administration, when the newborn cells are expected to have matured (Cameron et al., 1993; Hastings and Gould, 1999; Markakis and Gage, 1999). This design enabled us to determine whether survival and differentiation of newly formed cells could be modified by an early exposure to environmental enrichment, which might also influence proliferation, or by a delayed enrichment that could influence survival and differentiation but not proliferation of the BrdU-labeled cells. Our results indicate that the main effect of postischemic housing in an enriched environment is on genesis or survival of astrocytes.
MATERIALS AND METHODS
Animals, housing conditions and surgery
The Ethics Committee for Animal Research at Lund University approved the experimental protocol. Six-month-old male SHR, obtained from Mollegard Breeding Center, Ejby, Denmark, 2 months earlier and preoperatively housed in standard cages (550 × 350 × 200 mm, 3 to 4 rats in each cage), were anesthetized with methohexital sodium (Brietal, 37°C) 50 mg/kg intraperitoneally. The right MCA was accessed via a small craniotomy, and the artery was ligated distal to the striatal branches (Coyle, 1982; Grabowski et al, 1995), causing a neocortical infarct (Fig. 1, top right). The mean surgery time was about 20 minutes and body temperature was maintained close to 37°C. Postoperatively, the rats were kept in individual cages for 24 hours before all, except one group of rats, were returned to standard environment. One group was placed in a large, vertical, enriched-environment cage (815 × 610 × 1,280 mm), equipped with horizontal and vertical boards, chains, swings, wooden blocks, and objects of different sizes and materials. The distance between the boards and the movable objects was changed twice a week. Seven days after the arterial ligation, and following the last BrdU injection, another group of rats was transferred to a similar enriched-environment cage. Thus, the study consisted of four groups: ischemic rats housed in standard environment (standard group, n = 10); rats transferred to enriched environment 24 hours after surgery (early enriched group, n = 12); rats transferred to enriched environment 7 days after surgery (delayed enriched group, n = 12), and sham-operated rats in standard environment (sham group, n = 4). The design is illustrated in Figure 1. All animals received food and water ad libitum. Sensorimotor function tests were performed before the surgery and 4 weeks after surgery. The tests and results will be described in a second study in which we analyze possible changes in cortical regions in the same animals.
Bromodeoxyuridine injections
BrdU was dissolved in 0.9% NaCl and filtered at 22 μm. BrdU is a thymidine analogue that is incorporated into the DNA during the S phase of the cell cycle and can thus be detected immunohistochemically in proliferating cells. BrdU is available for incorporation 2 hours after injection and does not contribute to detectable labeling because of DNA repair (del Rio and Soriano, 1989; Nowakowski et al., 1989; Kuhn et al., 1996; Palmer et al., 2000). BrdU-labeled cells also include the progeny of labeled cells that have divided after BrdU has been discontinued and is no longer available. However, administering BrdU repeatedly should reduce the proportion of BrdU-positive cells that divide again compared to a single dose of BrdU. The animals in this study were given daily intraperitoneal injections (50 mg/kg body weight) of BrdU (Boehringer Mannheim, Scandinavia AB, Bromma, Sweden) for 7 days after surgery.
Tissue preparation
Five weeks after the surgery, the animals were deeply anesthetized with an overdose of methohexital sodium and then were perfused transcardially with saline solution followed by 4% paraformaldehyde in 0.1 mol/L phosphate buffer. The brains were removed, postfixed overnight in 4% paraformaldehyde in 0.1 mol/L phosphate buffer, and thereafter transferred to 30% sucrose. Coronal sectioning (40 μm) was performed on a freezing microtome and sections were stored in tissue cryoprotectant solution (25% ethylene glycol, 25% glycerol, and 0.1 mol/L phosphate buffer) at −20°C. Three, three, and two animals from the standard, early enriched, and delayed enriched groups, respectively, were excluded because of lack of or very small infarcts. One animal was excluded from the standard group because of tissue degradation. Thus, the final number of animals was 6, 9, 10, and 4 in the standard, early enriched, delayed enriched, and sham groups, respectively.
Immunohistochemistry
Pretreatment for bromodeoxyuridine detection.
Immunolabeling was performed on free-floating sections. To detect BrdU, the halogenated pyrimidine was exposed through DNA denaturation. This was achieved by treating the sections for 2 hours at 65°C in sodium citrate buffer (0.03 mol/L NaCl, 0.3 mol/L sodium citrate, pH 7) containing 50% formamide. After that, the sections were rinsed in sodium citrate buffer for 15 minutes, incubated in 2 mol/L HCl for 30 minutes at 37°C, rinsed in 0.1 mol/L borate buffer (pH 8.5) for 10 minutes, followed by several rinses in Tris buffered saline (TBS: 0.08 mol/L Trizma-HCl, 0.016 mol/L Trizma-Base, 0.15 mol/L NaCl, pH 7.5).
Immunoperoxidase staining.
After blocking endogenous peroxidase with 0.6% H2O2 in TBS for 30 minutes and following BrdU pretreatment, sections were incubated in blocking solution for 30 minutes (3% donkey serum and 0.1% Triton X-100 in TBS) and then incubated with primary antibody (monoclonal mouse anti-BrdU IgG, 1:400, Boehringer Mannheim) in blocking solution for 16 hours at 4°C. After rinsing with TBS and incubating in blocking solution for 15 minutes, sections were incubated for 2 hours at room temperature with secondary antibody (biotinylated rat-adsorbed horse anti-mouse IgG, 1:125, Vector Laboratories, Kemila, Stockholm). After rinsing in TBS, the sections were reacted with avidin—peroxidase for 1 hour (ABC-kit, Vectastatin Elite, Vector Laboratories) and then reacted with detection solution (26.5 mg/mL diaminobenzidine, 0.01% H2O2, 0.04% NiCl). Differential interference contrast images were obtained with a Nikon Diaphot microscope equipped with a cooled CCD camera (Hamamatsu, Hamamatsu, Japan).
Immunofluorescence staining.
Triple immunofluorescence staining for BrdU, a neuronal marker (NeuN or calbindin D28k), and the astrocytic marker GFAP were used to detect co-labeling of cell specific markers with BrdU. NeuN stands for neuronal nuclei and is a transcription factor that is expressed in the nucleus and cytoplasm of mature neurons (Mullen et al., 1992; Magavi et al., 2000), whereas calbindin D28k is a neuronal calcium binding protein and marker for granule cells in the dentate gyrus (Sloviter, 1989). After pretreating sections as for immunoperoxidase detection, omitting the step for endogenous peroxidase inactivation, incubation with primary antibodies in blocking solution was performed for 16 hours at 4°C. After rinsing with TBS and incubating in blocking solution for 15 minutes, sections were incubated for 2 hours at 37°C with secondary antibodies. The antibody combinations used were as follows: rat anti-BrdU antiserum (1:300, Harlan, Loughborough, England) detected with fluorescein isothiocyanate conjugated donkey anti-rat IgG (1:150); mouse anti-NeuN IgG (1:50, Chemicon, Temecula, CA, U.SA.) detected with Texas Red conjugated donkey anti-mouse IgG (1:150); rabbit anti-calbindin D28k antiserum (1:1,000, Swant, Bellizona, Switzerland) detected with Texas Red conjugated donkey anti-rabbit IgG (1:150); rabbit anti-GFAP antiserum (1:500, Dako, Glostrup, Denmark) detected with Cy5 conjugated donkey anti-rabbit IgG (1:200); and mouse anti-GFAP antiserum (1:300, Advanced Immunochemicals, Long Beach, CA, U.S.A.) detected with Cy5 conjugated donkey anti-mouse IgG (1:150). All secondary antibodies were from Jackson Immunoresearch, West Grove, PA, U.S.A. Fluorescent signals were analyzed using a confocal scanning microscope (MRC 1024, BioRad, Richmond, CA, U.S.A.).
Cell sampling, counting, and phenotyping.
BrdU-labeled cells in the GCL and the SGZ were counted and phenotyped. The SGZ was defined as a 20-μm band immediately adjacent to the hilar surface of the GCL. The GCL and the SGZ are referred to as the GCL throughout the text, except with respect to volume measurements, where only the GCL proper was considered. All cell counts were performed using unbiased stereologic counting techniques (Gundersen et al., 1988; Coggeshall and Lekan, 1996). Images were taken with a Nikon Optiphot microscope connected to a video camera, and area measurements of the GCL were performed using an Intuos Graphics tablet (Wacom, Japan) and digital image processing software (Nikon, Göteborg, Sweden). BrdU immunopositive cells were counted on 10 anatomically matched immunoperoxidase-stained sections per animal, 240 μm apart. The GCL sample volume was calculated by multiplying the cross-sectional GCL area with the thickness of the section (40 μm). The number of counted cells was then divided by the GCL sample volume. The total number of BrdU-labeled cells was achieved by multiplying mean cell density with the total GCL volume. Determination of co-labeling of BrdU with NeuN, calbindin D28k, and GFAP, respectively, was performed on five to six anatomically matched sections 240 μm apart and at least 40 BrdU-labeled cells per animal were analyzed concerning phenotype. Optical Z sectioning of the samples at 0.5-μm intervals was used to verify co-localization of BrdU with cell-specific markers. A percentage of co-labeling was calculated, and the total numbers of newborn neurons and astrocytes were calculated by multiplying that percentage with the total number of BrdU-labeled cells per GCL.
Measurement of total tissue loss and granular cell layer volume
Measurements of ipsi- and contralateral hemisphere cross-sectional area were performed for each animal on 10 Nissl-stained sections 960 μm apart. The hemispheric volume was calculated from the cross-sectional areas and the distance between the sections according to the Cavallieri principle (Gundersen et al., 1988). Measurement of the GCL volume was performed in a similar fashion on 10 Nissl-stained sections, 480 μm apart, encompassing the entire dentate gyrus. Total tissue volume loss was expressed as percentage of the contralateral hemisphere volume.
Statistical analysis
The presented values are mean values ± SD. Comparisons between groups were made with one-way analysis of variance followed by the Fisher PLSD post hoc test (Statview 4.01 for Macintosh). Student's t-test was used to compare ipsilateral and contralateral astrocyte-to-neuron ratios within a group. P values <0.05 were considered statistically significant.
RESULTS
Bromodeoxyuridine labeling
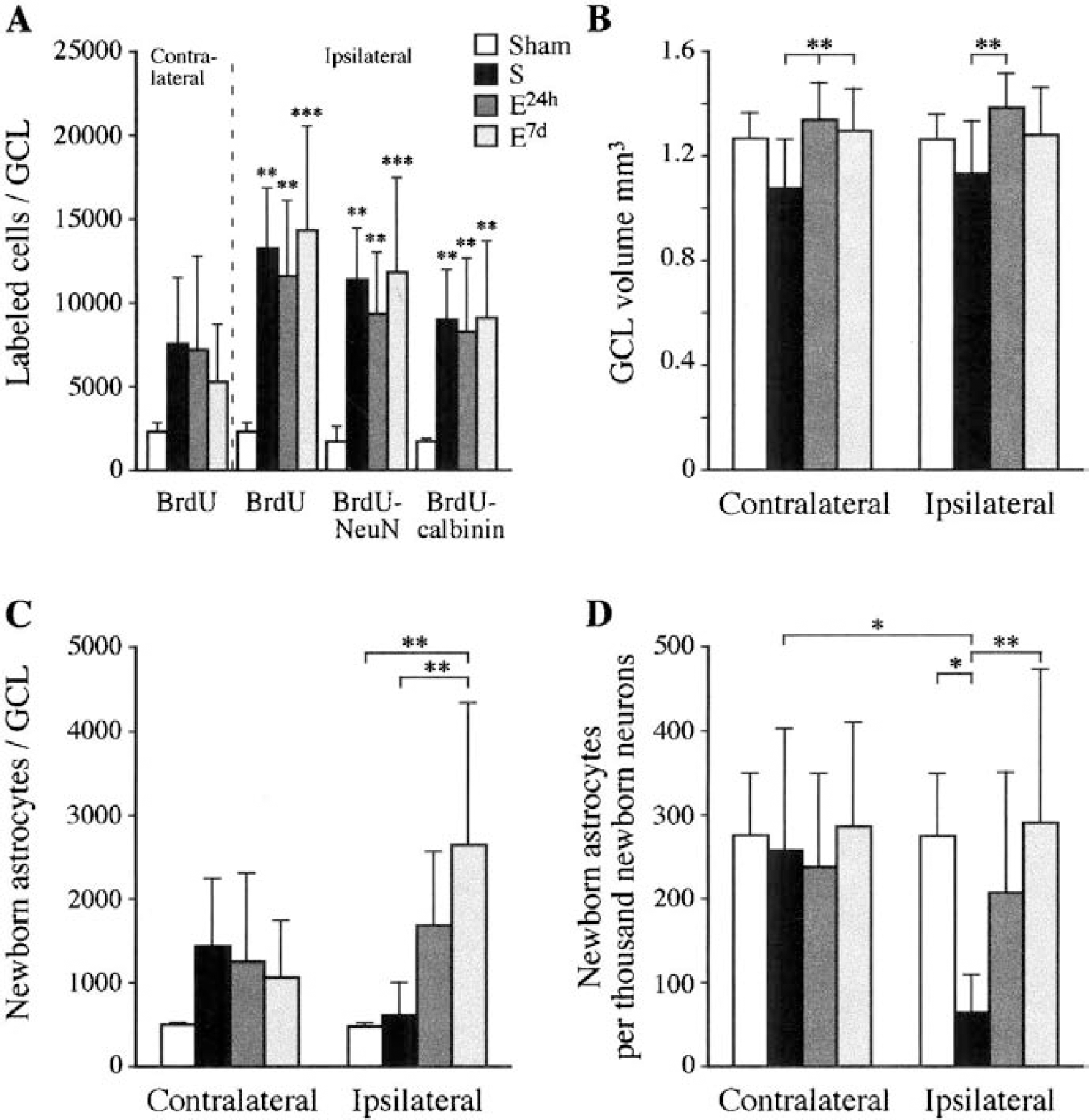
The number of BrdU-positive cells, labeled during the first week after induction of permanent cortical ischemia, was increased five-to sixfold compared to sham-operated rats 5 weeks after the ischemic insult in the GCL ipsilateral to the lesion (Figs. 1 and 2A). The total number of BrdU-labeled cells in the ipsilateral GCL of the standard, early-enriched, delayed-enriched, and sham groups was 13,062 ± 3,524; 11,546 ± 4,380; 13,927 ± 6,479; and 2,289 ± 391, respectively. In the contralateral GCL, the lesioned groups had more new cells than sham-operated rats (7,580 ± 3,832; 7,038 ± 5,364; 5,152 ± 3,434 for standard, early, and delayed enriched, respectively), although the differences were not significant. Thus, postischemic environmental enrichment had no significant influence on the number of surviving cells.
Phenotype determination
Neurons.
Triple labeling with BrdU, the glial marker GFAP, and two different neuronal markers was used to study neurogenesis (Fig. 3A—L). Most of the BrdU-positive cells were verified to be neurons. About 80% of the BrdU-positive cells co-labeled with NeuN and about 70% co-labeled with calbindin D28k. Ischemia induced an ipsilateral increase in total numbers of newborn neurons in all lesioned groups compared to control (Fig. 2A). In the ipsilateral GCL, the total number of BrdU-NeuN co-labeled cells was increased about fivefold in the standard (11,450 ± 2,760), early-enriched (9,308 ± 3,565), and delayed-enriched (11,587 ± 5,936) groups compared to the sham group (1,876 ± 408). The corresponding values for BrdU—calbindin D28k co-labeled cells in the standard, early-enriched, and delayed-enriched groups were 8,814 ± 3,033; 8,353 ± 4,256; and 8,943 ± 4,700; a more than sixfold increase compared to the sham group (1,368 ± 421). Thus, postischemic environment did not influence the neurogenetic response.
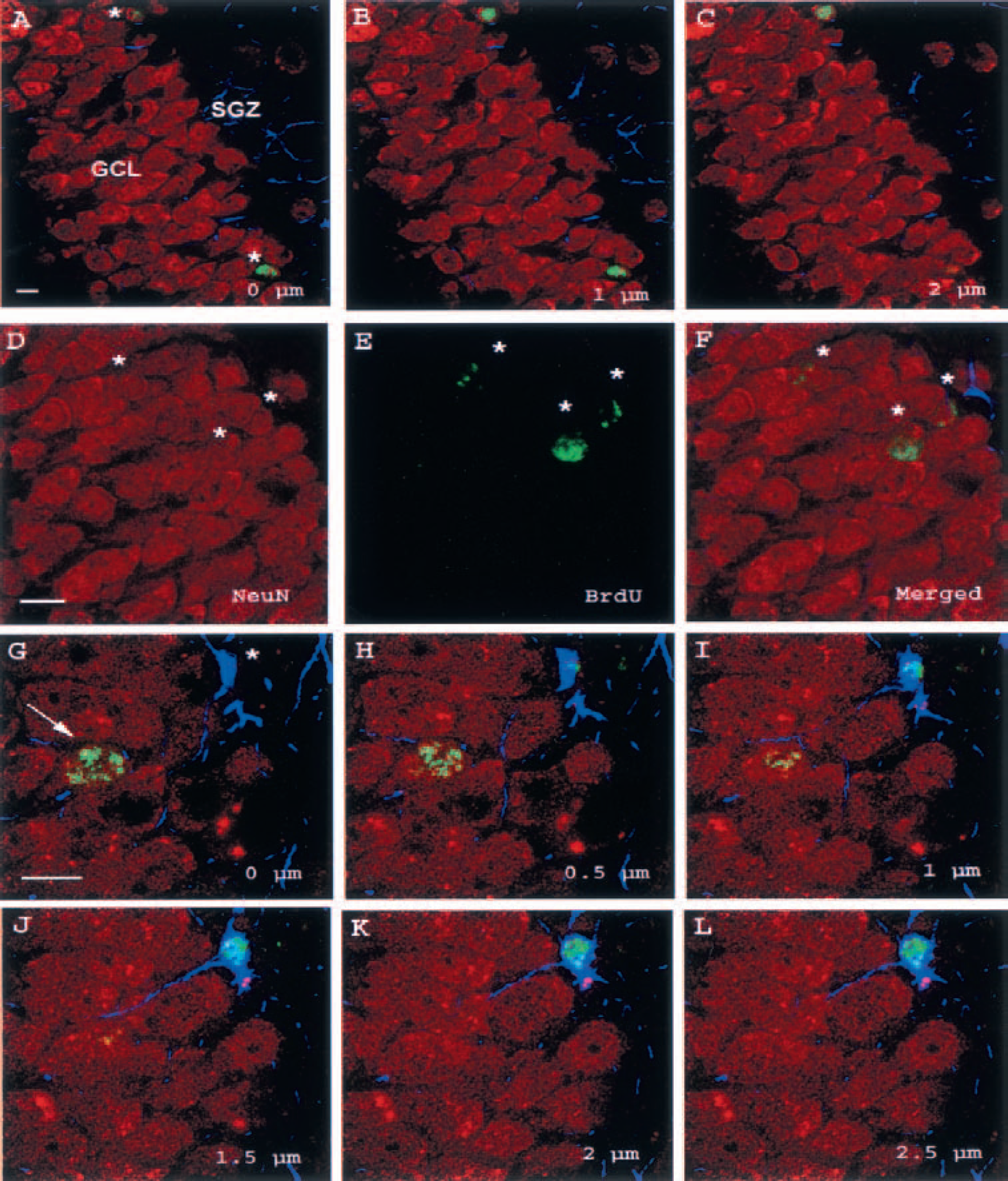
Triple immunofluorescence staining of the ipsilateral dentate gyrus of the standard group with cell-specific markers.
Astrocytes.
Newborn astrocytes represented 6% to 22% of the newborn cells in the GCL. They were mostly located in the SGZ and extended their processes into the GCL proper (Fig. 3G—L). The number of BrdU-GFAP co-localizing cells in the standard group did not differ from the sham group, whereas the early-enriched group showed a threefold increase and the delayed enriched group a fivefold increase in the ipsilateral GCL compared to sham. The difference to the sham and standard groups was significant for the late-enriched group. Because of a large interindividual variation, the difference was not significant for the early-enriched group (P = 0.09). The total numbers of BrdU-GFAP co-localizing cells in the ipsilateral GCL for the sham, standard, early-enriched, and delayed-enriched groups were 495 ± 29; 643 ± 371; 1,699 ± 881; and 2,658 ± 1,696, respectively.
Astrocyte-to-neuron ratio.
To correlate the generation of astrocytes to neurogenesis in the GCL, the ratios between the total numbers of BrdU-GFAP and BrdU-NeuN co-labeled cells were calculated. All lesioned groups displayed contralateral ratios that did not differ from control. Ipsilaterally, however, the ratio of the standard group was significantly lower than that of the delayed enriched and sham groups (Fig. 2D). Furthermore, the ratio was significantly lower in the ipsilateral than in the contralateral GCL in the standard group. The standard group had also a lower ipsilateral ratio than the early enriched group, although the difference was not significant (P = 0.068). Thus, postischemic environmental enrichment, initiated after completed BrdU administration, had a significant effect in normalizing the ipsilateral ratio between newborn astrocytes and neurons that was decreased by ischemia.
Total tissue volume loss and granular cell layer volume
Total tissue volume loss was measured to ensure homogeneity of the lesion size between the groups and was found not to differ significantly, confirming previous results in the same experimental model (Johansson, 1996). The ipsilateral GCL volume was significantly smaller in the standard group (1.13 ± 0.20 mm3) than in the early-enriched group (1.39 ± 0.13 mm3), and the volume of the contralateral GCL was smaller in the standard group (1.08 ± 0.17 mm3) than in the early (1.34 ± 0.14 mm3) and delayed-enriched groups (1.3 ± 0.16 mm3) (Fig. 2B). Thus, postischemic housing conditions significantly influenced the ipsilateral and contralateral GCL volumes.
DISCUSSION
Five weeks after permanent cortical ischemia, the number of surviving BrdU-positive cells, labeled during the first week after the lesion, was five-to sixfold increased in the GCL ipsilateral to the lesion. Phenotypic analysis with cell specific markers showed that for the most part, the newborn cells had differentiated into neurons. More BrdU-positive cells co-labeled with the neuronal marker NeuN than with calbindin D28K, possibly reflecting that calbindin D-28k, a marker for mature granule cells (Sloviter, 1989), does not label certain interneurons in the GCL and the SGZ, whereas NeuN labels most types of postmitotic neurons (Mullen et al., 1992; Magavi et al., 2000).
In the standard group, cortical ischemia did not lead to a corresponding ipsilateral increase of newborn astrocytes, resulting in a reduced ratio between newborn astrocytes and neurons. Postischemic environmental enrichment had no significant influence on the number of surviving cells or neurogenesis, whereas it increased the number of new astrocytes in the ipsilateral GCL. The delayed enrichment induced a fivefold and significant increase in astrocytes compared to sham and lesioned rats in standard environment and normalized the lesion-induced decrease of the ratio between newborn astrocytes and neurons. Although a threefold increase in newly formed astrocytes was observed in the early-enriched group, the difference to sham and standard rats did not reach significance because of a large interindividual variation.
A population of differentiated astrocytes divides at a slow rate in the adult dentate gyrus (Cameron et al., 1993). Because no animals were killed directly after ending BrdU administration, it is impossible to know how many newly formed astrocytes were present at that time. However, the majority of the newly labeled BrdU-positive cells in the SGZ have been shown to express the neural progenitor cell marker nestin and NG2, a marker for glial progenitors, whereas only a small population expressed astrocyte-specific markers (Palmer et al., 2000), suggesting that the majority of differentiated astrocytes in the SGZ originated from progenitor cells. Whether that is the case in the postischemic phase is not known. However, because the largest difference was observed between rats in the standard and delayed-enriched groups, which shared the same environment during the time of BrdU injection, the difference cannot be explained by events happening during the early postischemic phase. Because the total numbers of BrdU-labeled cells and new neurons did not differ between the lesioned groups, it is possible that postischemic environmental enrichment selectively enhanced the survival of progenitor cells, committed to the astrocytic lineage. Another possibility is that postischemic environmental enrichment influenced the differentiation of the BrdU-labeled hippocampal progenitor cells, steering them toward the astrocytic lineage.
Astrocytes are essential for optimal neuronal function. The ischemia-induced reduction in gliogenesis and altered ratio between newborn astrocytes and neurons are likely to result in a poor environment for the new neurons.
Astrocyte-derived soluble factors of the FGF, TGF, and EGF families play an important role in several steps of neuronal morphogenesis from the early events of neuronal precursor proliferation during brain development to later periods of neuronal differentiation and formation of neural circuits where astrocytes provide a scaffold for the correct migration of neurons and growth cones (Gomes et al., 2001). In the adult brain, astrocytes maintain neuronal and synaptic homeostasis (Pfrieger and Barres, 1996; Vernadakis, 1996; Chvatal and Sykova, 2000), and they take an active part in synaptic plasticity and generation (Müller, 1992, 1993; Araque et al., 2001, Ullian et al., 2001). Astrocytes are important mediators of environmentally induced plasticity. They increase in number and contact with synaptic elements in rats reared in a complex environment (Diamond et al., 1966; Walsh, 1969; Jones and Greenough, 1996; Sirevaag and Greenough, 1991), and seasonal changes in astrocytes parallel neuronal plasticity in the song control area of the canary (Kafitz et al., 1999). The environmental promotion of gliogenesis observed in our study is in line with these data.
Compared to standard environment, early environmental enrichment significantly increased the GCL volume on both sides, whereas delayed enrichment had a significant effect only on the contralateral side. There were, however, no corresponding increases in total number of BrdU-labeled cells or in cell density in the GCL (data not shown) that could account for the volume change. The increase in GCL volume may very well be explained by changes in cell morphology induced by enriched environment. Environmental stimulation and complexity have been found to increase brain weight, cortical and hippocampal thickness, neuronal cell body and nucleus size, and to increase dendritic branching, dendritic spine density, and the number of discontinuous synapses (Schrott, 1997; Johansson and Belichenko, 2002). Among the mechanisms proposed to be responsible for the neuroanatomical changes induced by an enriched environment are transcriptional activation and altered levels of a variety of growth factors. Expression of glial-derived neurotrophic factor, brain-derived neurotrophic factor, nerve growth factor, and phosphorylation of the transcription factor cyclic-AMP response element binding protein are all induced in the hippocampus by enriched environment (Young et al., 1999; Ickes et al., 2000).
MCA occlusion has been shown to induce expression of the immediate early genes c-fos and junB, and the heat shock protein gene hsp 70 in the hippocampus (Kinouchi et al., 1994a). Systemic administration of an N-methyl-d-aspartate receptor blocker inhibited the induction of c-fos and junB (Kinouchi et al., 1994b) and completely suppressed the elevated hippocampal neurogenesis following 2 hours of MCA occlusion (Arvidsson et al., 2001). Thus, the hippocampal cell response after ischemia appears to be mediated by glutamatergic mechanisms. However, the mechanisms by which postischemic environmental enrichment might act to influence this response are largely unknown. In the same experimental model as in the present study, enriched environment has been shown to influence postischemic levels of brain-derived neurotrophic factor mRNA and brain-derived neurotrophic factor protein (Zhao et al., 2000, 2001) and expression of the immediate early gene transcription factor nerve growth factor—induced gene A and the glucocorticoid receptor (Dahlqvist et al., 1999).
A permanent embolic occlusion of the MCA was reported not to elicit any changes in BrdU labeling in the dentate gyrus (Zhang et al., 2001). Jin et al. (2001), who studied hippocampal cell proliferation after transient MCA occlusion with the intraluminal suture model, observed a maximal eightfold increase in BrdU labeling ipsilaterally and a fourfold increase contralaterally 1 week after ischemia. Neither cell survival nor differentiation, however, was determined. Following transient global ischemia, about one fourth of the newborn cells disappeared within a month after the maximum rate of cell birth was reached (Liu et al., 2000), and fewer than 50% of the BrdU-labeled cells in the hippocampus could be detected 4 weeks after BrdU administration in intact rats (Nilsson et al., 1999). It is possible that the contralateral differences between sham and lesioned rats in this study would have been more prominent at an earlier stage. In a study on cell survival in the same experimental model as used by Jin et al. (2001), a slight but not significant increase of BrdU-labeled cells was observed on the contralateral side 4 weeks after BrdU administration (Arvidsson et al., 2001), a finding that is comparable to our data.
A neocortical infarct, which does not primarily involve the hippocampus or the entorhinal cortex targeting it, results in loss of cortical connectivity that might be mediated to the hippocampus through subcortical structures. In this study, we explored the influence of cortical ischemia and postischemic environmental enrichment on the generation of new neurons and astrocytes in the GCL. The combination of these two factors, known to independently influence hippocampal cell genesis, has not been studied before. We show that selective cortical ischemia increased ipsilateral neurogenesis and that the response was not influenced by postischemic housing. However, postischemic environmental enrichment increased the generation of astrocytes ipsilaterally and normalized the ratio between newborn astrocytes and neurons that was perturbed by ischemia.
Previous studies of the effect of enriched environment on hippocampal cell genesis in a BrdU labeling paradigm have shown specific neurogenesis-promoting effects and no influence on the generation of astrocytes (Kempermann et al., 1997; Nilsson et al., 1999). We suggest that enriched environment might act in a different way in the postischemic phase and that more attention has to be given to the astrocytic response and influences on the ratio between newborn astrocytes and neurons.
The dentate gyrus can be used as a model area of cell proliferation and neurogenesis with its well-known cytoarchitectonic structure and circuitry and its important function in learning and memory. Knowing more about the cellular responses that ischemia and enriched environment elicit in the hippocampus is potentially important for future treatment strategies against stroke since multipotent precursor cells also exist in several extrahippocampal regions (Palmer et al., 1999) and can possibly be recruited by an injury (Magavi et al., 2000).
Footnotes
Acknowledgments
The authors acknowledge the technical assistance of Dr. Michelle Anderson and Anette Risedal. The authors are also grateful to professors Annica Dahlström, Pam Fredman, and Keiko Funa for generously providing access to their confocal microscope.
