Abstract
Neurogenesis and angiogenesis in the subventricular zone and peri-infarct region have been confirmed. However, newly formed neuronal cells and blood vessels that appear in the nonischemic ipsilateral ventroposterior nucleus (VPN) of the thalamus with secondary damage after stroke has not been previously studied. Twenty-four stroke-prone renovascular hypertensive rats were subjected to distal right middle cerebral artery occlusion (MCAO) or sham operation. 5′-Bromo-2′-deoxyuridine (BrdU) was used to label cell proliferation. Rats were killed at 7 or 14 days after the operation. Neuronal nuclei (NeuN), OX-42, BrdU, nestin, laminin+, BrdU+/nestin+, BrdU+/NeuN+, nestin+/GFAP+(glial fibrillary acidic protein), and BrdU+/laminin+ immunoreactive cells were detected within the ipsilateral VPN. The primary infarction was confined to the right somatosensory cortex. Within the ipsilateral VPN of the ischemic rats, the number of NeuN+ neurons decreased, the OX-42+ microglia cells were activated, and BrdU+ and nestin+ cells were detected at day 7 after MCAO and increased in number at day 14. Moreover, BrdU+/nestin+ cells and BrdU+/NeuN+ cells were detected at day 14 after MCAO. In addition, the ischemic rats showed a significant increase in vascular density in the ipsilateral VPN compared with the sham-operated rats. These results suggest that secondary damage with neurogenesis and angiogenesis of the ipsilateral VPN of the thalamus occurs after focal cortical infarction.
Introduction
Focal cerebral ischemia causes a series of pathologic changes and results in ischemic neuronal cell deaths and neurologic deficits (Schmidt-Kastner and Hossmann, 1988; Wei et al, 2004). It also leads to secondary neurodegeneration in the nonischemic areas remote from, yet connected synaptically with, the site of primary ischemic lesion, hindering the functional recovery (Iizuka et al, 1990; Liang et al, 2007). One of the best examples is the secondary neurodegeneration in ipsilateral ventroposterior nucleus (VPN) of the thalamus after focal cortical infarction (Dihne et al, 2002; Schroeter et al, 2006). It has been widely accepted that the principal mechanism of secondary thalamic degeneration is retrograde degeneration of thalamocortical projections, which connect the thalamic nuclei, such as VPN, with the ischemic cortex (Kataoka et al, 1989; Carmichael et al, 2001). Until now, most studies of secondary thalamic degeneration have focused only on the neuronal damage and gliosis after cerebral ischemia (Dihne et al, 2002; Schroeter et al, 2006).
Our previous study and other reports have shown that cerebral ischemia enhances neurogenesis and angiogenesis in the subventricular zone and periinfarct region, which may contribute to neurologic improvement (Wang et al, 2004; Sugiura et al, 2005; Ardelt et al, 2007; Ling et al, 2008). To the best of our knowledge, neurogenesis and angiogenesis have not been examined in nonischemic ipsilateral VPN of the thalamus with secondary damage after stroke. Preventing remote damage may be critical to the long-term functional improvement after cerebral ischemia. Moreover, the possibility of preventing the progressive remote damage is greater than that of preventing the primary ischemic damage because the remote damage occurs at a relatively late stage of stroke (Schroeter et al, 2006). As the mechanisms of remote secondary damage, such as neuronal necrosis, apoptosis, and oxidative stress, are similar to those of the peri-infarct region (Schmidt-Kastner and Hossmann, 1988; Iizuka et al, 1990; Natale et al, 2002; Wei et al, 2004; Schroeter et al, 2006; He et al, 2007), we hypothesize that there are newly formed neuronal cells and blood vessels in the ipsilateral VPN of the thalamus after cerebral ischemia. Thus in this study, we aimed at testing whether neurogenesis and angiogenesis would occur in the ipsilateral VPN of the thalamus after focal cortical infarction in hypertensive rats.
Materials and methods
Animal Model
All experimental procedures were approved by the local Institutional Animal Care and Use Committee and were in accordance with the guidelines for animal use. Maximal efforts were applied to minimize the animal's suffering during the experiment.
A total of 30 Sprague—Dawley rats weighting 80 to 100 g were made to become stroke-prone renovascular hypertensive rats (Zeng et al, 1998a,b). Briefly, the rat was anesthetized with 10% chloral hydrate (3 mL/kg, intraperitoneally) and underwent a two-kidney two clips operation to induce renal artery constriction. Systolic blood pressure was measured using a tail-cuff sphygmomanometer after the conscious rats were warmed at a 37°C chamber for 15 mins at baseline and weekly after renal artery constriction. Twelve weeks later, 24 rats weighing 300 to 400 g, whose systolic blood pressure was steadily >180 mmHg and without stroke symptoms, were selected from the 30 stroke-prone renovascular hypertensive rats. They were divided into two groups, namely the middle cerebral artery occlusion (MCAO) group (Bederson et al, 1986) and the sham-operated group (12 rats in each). Briefly, the rat was anesthetized, and the temporal muscle was dissected by a coronal skin incision to expose the underlying skull. A small burr hole was drilled, and the exposed right MCA was occluded above the olfactory tract and distal to the striate branches by bipolar coagulation under a surgical microscope, resulting in a permanent focal infarction in the neocortex. The right MCA of the sham-operated rat was only exposed without occlusion. Body temperature was maintained at 37°C ± 0.5°C during the surgical and recovery periods using a heat lamp.
5′-Bromo-2′-Deoxyuridine Labeling
5′-Bromo-2′-deoxyuridine (BrdU, Sigma-Aldrich, St Louis, MO, USA, 50 mg/kg), a thymidine analog that is incorporated into the DNA of dividing cells during S-phase (Lee et al, 2007), was used to label proliferative cells. It was dissolved in saline and injected intraperitoneally twice daily for 6 consecutive days, starting at 24 h after MCAO or sham operation.
Tissue Preparation
Six rats from each group were killed at 7 and 14 days after the operation, respectively. The rats were anesthetized deeply with 10% chloral hydrate and killed by transcardiac perfusion with heparinized saline initially and thereafter with 4% paraformaldehyde in 0.1 mol/L of PBS (phosphate-buffered saline) (pH = 7.4). After postfixation for 6 h, their brains were immersed in 15, 20, and 30% of sucrose—phosphate buffer solution sequentially, and finally 10-μm-thick coronal sections were obtained between the bregma level of −.2 and −.4 mm using a cryostat vibratome (VT 2800N; Leica, Heidelberg, Germany). For cell counting, four consecutive coronal brain sections per rat containing the thalamic VPN were analyzed at 200-μm intervals from the bregma levels of −.8 to −.6 mm with a field of view of 850 times 640 μm under a magnification of × 200. One of the authors examined the histologic sections under the microscope, and another author who was unaware of the cortical infarct performed the cell counting.
Histology and Immunofluorescent Staining
Hematoxylin and eosin staining was used to detect the infarction. Relative infarct volume was expressed as the percentage of the contralateral hemisphere (Swanson et al, 1990). Briefly, consecutive coronal brain sections at 2.0-mm intervals from the bregma level of +4.7 to −.2 mm were analyzed. Relative infarct volumes were calculated from the areas of the contralateral hemispheric area (V A) and ipsilateral infarcted area (VB) according to the equation VB/VA × 100%0.
For single- or double-labeled immunofluorescent staining, the following primary antibodies were used in this study: mouse monoclonal anti-BrdU (1:1,000, Sigma-Aldrich), sheep polyclonal anti-BrdU (1:500, Abcam, Cambridge, MA, USA), mouse monoclonal anti-nestin (1:1,000, Chemicon International, Temecula, CA, USA), mouse monoclonal anti-neuronal nuclei (NeuN, a marker of neurons, 1:1,000, Chemicon International), mouse monoclonal OX-42 (1:1,000, Chemicon International), rabbit polyclonal anti-glial fibrillary acidic protein (GFAP, a marker of astrocytes, 1:1,000, GeneTex, San Antonio, TX, USA), rabbit polyclonal antilaminin (1:200, Sigma-Aldrich). After being incubated with primary antibodies at 4°C overnight, the fluorescent-labeled secondary antibody Alexa Flour 488 (1:200, Invitrogen, Irvine, CA, USA), Cy3-conjugated antibody (1:400, Chemicon International), and Cy2- or Cy3-conjugated antibody (1:200, 1:400, Jackson Immunoresearch Laboratories, PA, Bar Harbor, ME, USA) were applied for 1 h at room temperature. For BrdU immunofluorescence, brain sections were pretreated with 2 N HCl at 37°C for 30 mins and rinsed in 0.1 mol/L of boric acid (pH = 8.5) at room temperature for 10 mins before being incubated with a blocking solution. Negative control sections were incubated with 0.01 mol/L of PBS as a substitute for the primary antibody. The images were captured under a microscope (Olympus BX51, Olympus, Tokyo, Japan).
Cell Counting
The number of NeuN-labeled neurons or BrdU-labeled cells was counted under a microscope with a × 20 objective (Olympus BX51) plus a magnifier digital camera, and was expressed as the average number of NeuN or BrdU per section. The immunoreactivity of nestin, OX-42, or laminin was measured under a microscope with a × 10 objective, and expressed as the percentage of nestin, OX-42, or laminin-immunopositive area per section. All images were analyzed using the NIH ImageJ 1.38 program (National Institute of Mental Health, Bethesda, Maryland, USA).
Statistical Analysis
All compared data were presented as means ± s.e.m. and analyzed using SPSS (Windows version 13.0; SPSS, Chicago, IL, USA). The comparisons of different groups were analyzed by the two-tailed Student's t-test. A P-value of < 0.05 was considered statistically significant.
Results
Primary Infarction and Secondary Thalamic Damage
In the MCAO group, the primary infarction was confined to the right somatosensory cortex, and the relative infarct volumes were 19.8% ± 1.45% and 17.4% ± 1.11% at 7 and 14 days, respectively. The thalamus was not involved in the primary infarction in all MCAO rats (Figure 1). There was no infarction in the sham-operated rats.
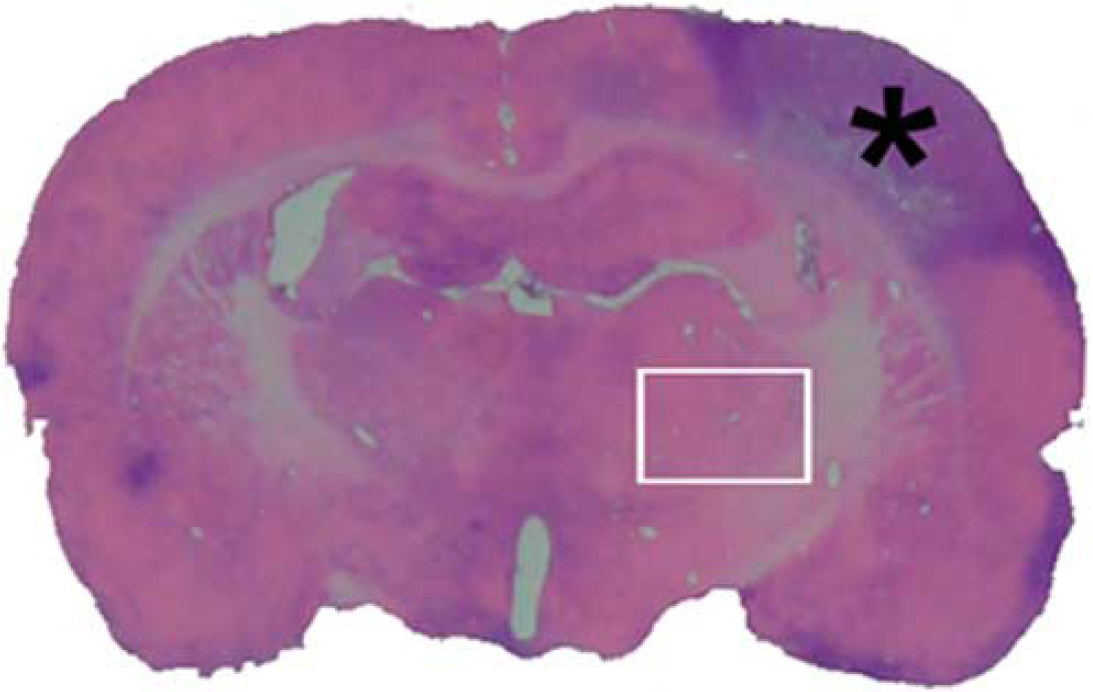
Hematoxylin and eosin (H&E)-stained coronal brain section (at the bregma level of −.3 mm) at day 7 after MCAO. The primary infarction is confined to the right somatosensory cortex and does not involve the ipsilateral thalamus. *Indicates the location of the focal cortical infarct and the rectangle indicates the ipsilateral VPN.
A progressive neuronal damage, characterized by the reduced number of NeuN-postive neurons, was observed in the ipsilateral VPN of the thalamus. The number of neurons within the ipsilateral VPN of the MCAO group decreased after MCAO when compared with the sham-operated group (698 ± 34.2 versus 882 ± 47.0 at day 7, 471 ± 50.0 versus 875 ± 50.7 at day 14, both P<0.05; Figure 2A–2C). Together with the reduced number of neurons, the extent of immunoreactivity for OX-42 increased within the ipsilateral VPN when compared with the sham-operated group (10.9% ± 1.3% versus 1.4% ± 0.4% at day 7, 16.4% ± 1.2% versus 1.5% ± 0.4% at day 14, both P<0.01; Figure 2D–2F). These immunoreactive microglia were characterized by their activated appearance with typical hypertrophic shape and thickened processes. Up to day 14 after MCAO, the OX-42 immunopositive microglia cells were seen over the entire ipsilateral VPN. In contrast, there was no change in the number of NeuN-labeled neurons and only a few OX-42 immunopositive microglia cells without an activated appearance were present within the ipsilateral VPN of the sham-operated rats (Figure 2B and 2E).
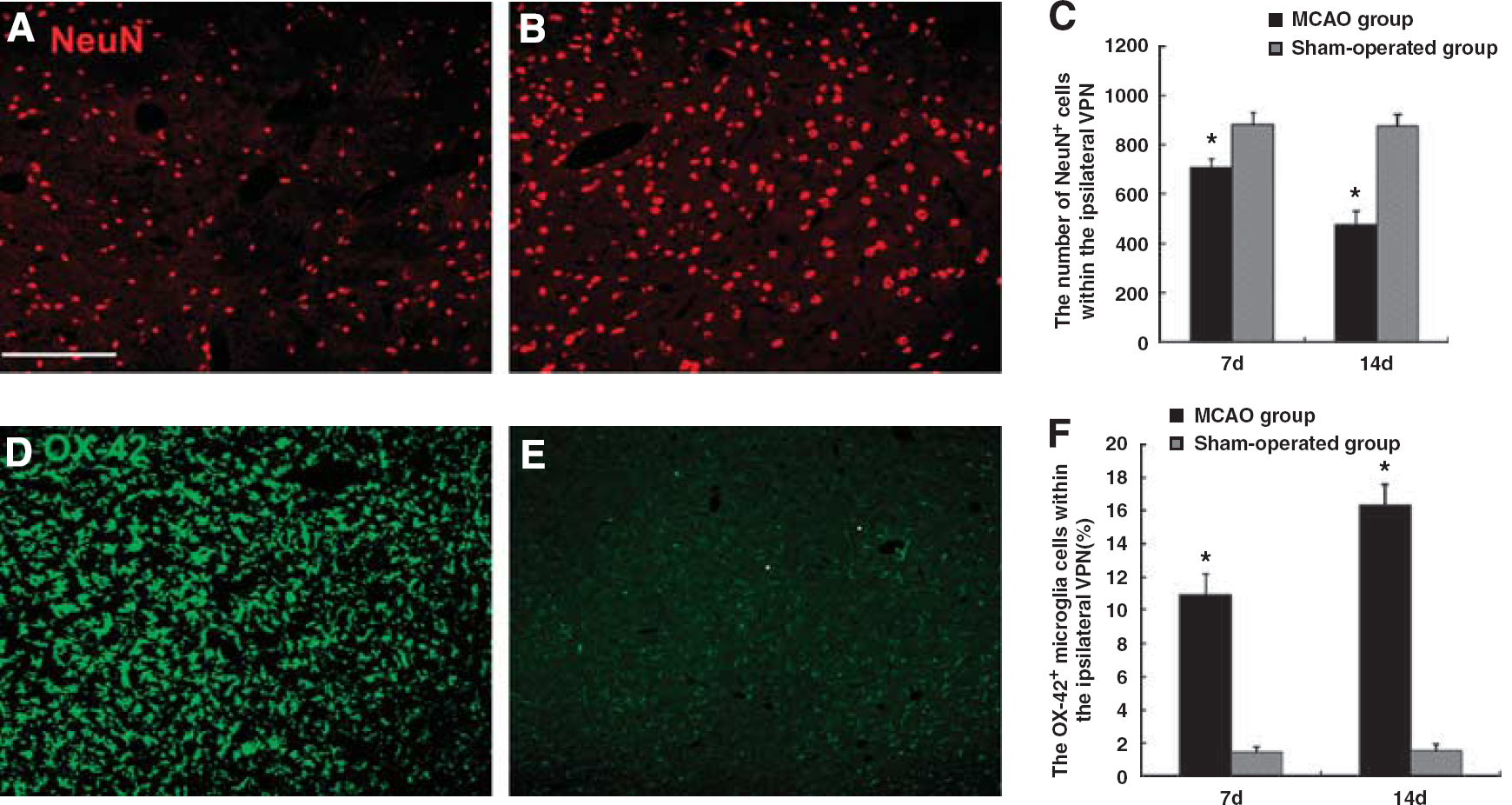
Secondary neuronal loss and microglia activation within the ipsilateral VPN of the thalamus at day 14 after cortical infarction. NeuN-positive neurons (red) within the ipsilateral VPN of the MCAO rats (
Cell Proliferation in the Ipsilateral Ventroposterior Nucleus
In the MCAO group, BrdU-positive cells were detected within the ipsilateral VPN of the thalamus at day 7, and further increased in number at day 14 after MCAO (Figure 3A). Morphologically, some BrdU-positive cells had elongated nuclei or two divided nuclei and a few others were present in pairs. In contrast, BrdU-positive cells were not seen within the ipsilateral VPN of the sham-operated rats (Figure 3B).
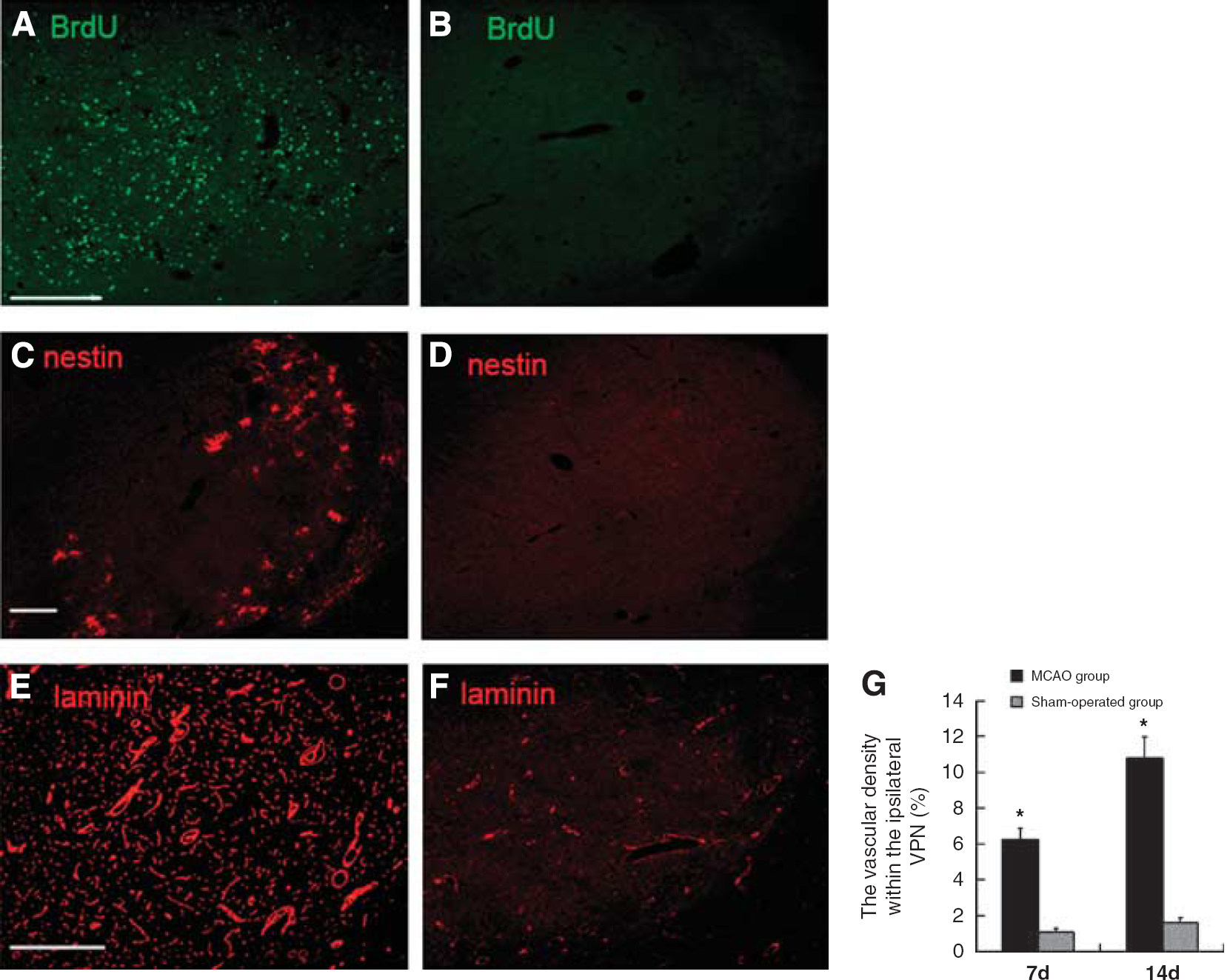
Expression of BrdU, nestin, and laminin immunoreactivities at day 14 after cortical infarction. BrdU-immunopositive cells (green) within the ipsilateral VPN of the MCAO rats (
Neurogenesis in the Ipsilateral Ventroposterior Nucleus
In the MCAO group, the nestin-positive cells were present sporadically within the ipsilateral VPN at day 7 and increased markedly in number at day 14 (Figure 3C). They were distributed initially around the subcortical fiber tracts and at the edge of the ipsilateral VPN, and later seen at the center of the ipsilateral VPN. The nestin-positive cells within the ipsilateral VPN were in clusters and showed a typical morphology of small cell bodies with thin long processes similar to that of GFAP-labeled astrocytes. In addition, nestin-positive and BrdU-positive cells followed a similar time course, and clusters of the nestin-positive cells were frequently present in the places where there were more BrdU-positive cells within the ipsilateral VPN. Moreover, some nestin-positive cells were colocalized with BrdU in the ipsilateral VPN of the ischemic rats at day 14 after MCAO (Figure 4A–4C), suggesting that there are some newly formed neural stem/precursor cells within the ipsilateral VPN of the thalamus, which is remote from the primary infarction in the cortex. In contrast, neither nestin-positive (Figure 3D) nor BrdU-positive cells were detected within the ipsilateral VPN of the sham-operated rats.
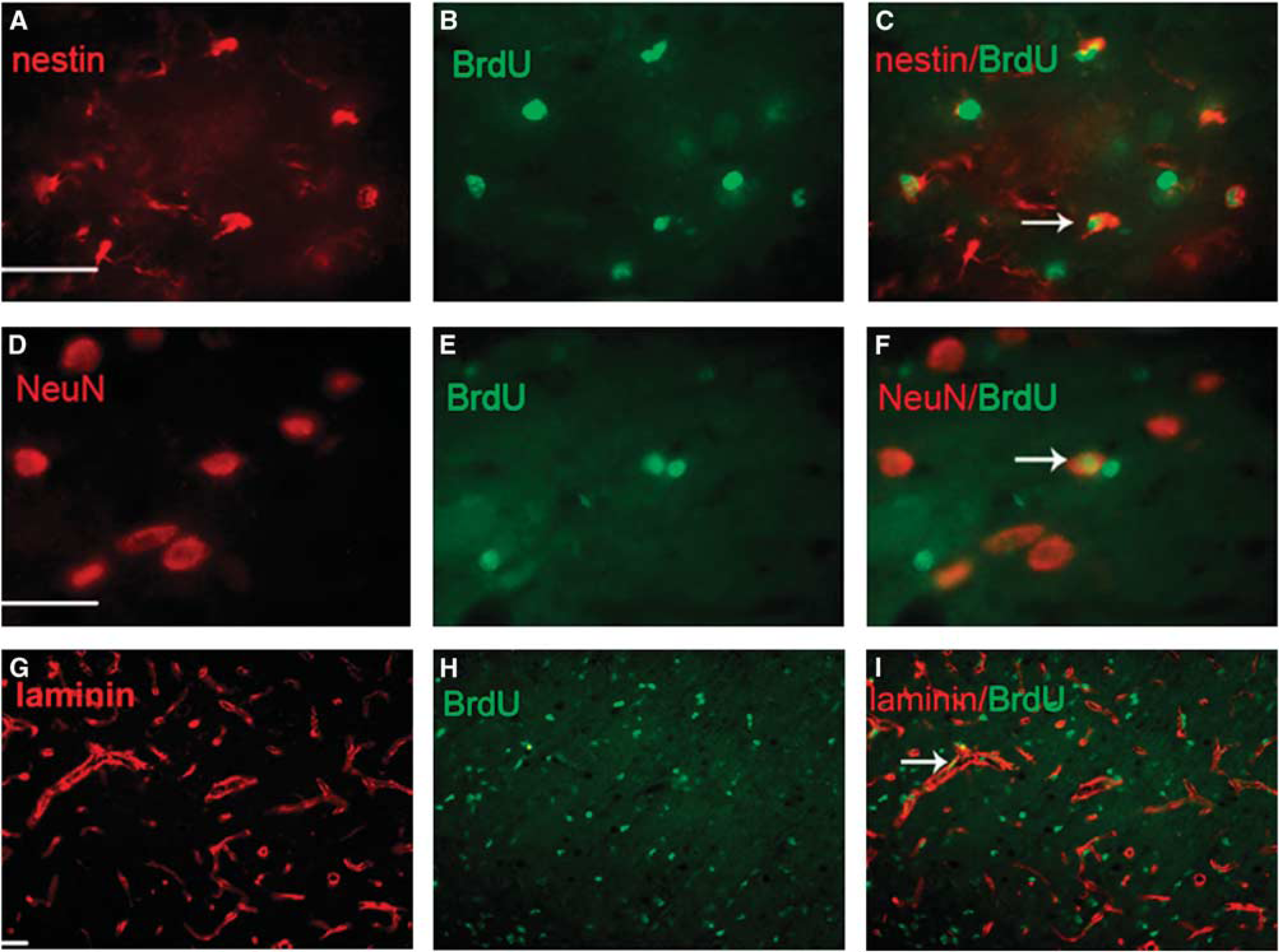
Expression of BrdU+/nestin+ cells, BrdU+/NeuN+ cells at day 14 and BrdU+/laminin+ cells at day 7 after cortical infarction. (
To further investigate the differentiation of newly formed neural stem/precursor cells within the ipsilateral VPN, double-labeled immunofluorescent staining was performed using different cell type-specific markers, such as NeuN, GFAP, and BrdU. Within the ipsilateral VPN of the ischemic rats, some BrdU-positive cells were colabeled with NeuN at day 14 after focal infarction (Figure 4D–4F), but those colabeled with GFAP were not observed. Neither the BrdU/NeuN nor BrdU/GFAP double-positive cell was detected within the ipsilateral VPN of the sham-operated rats.
The majority of nestin-positive cells within the ipsilateral VPN showed a morphology of astrocytes. Double-labeled immunofluorescent staining was performed using antibodies to nestin and GFAP. Some nestin-positive cells were coexpressed with GFAP within the ipsilateral VPN at day 14 after MCAO (Figure 5A–5C).
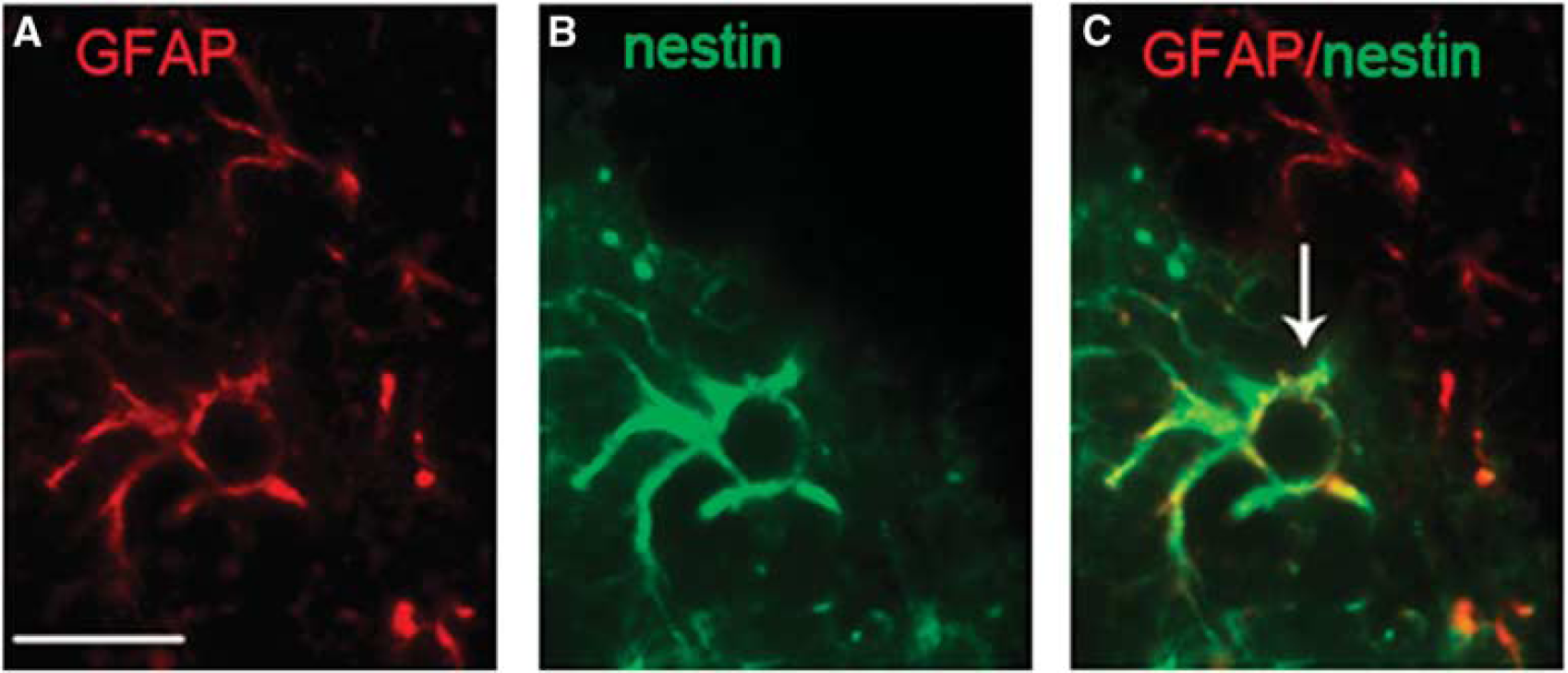
Expression of nestin+/GFAP+ cells at day 14 after cortical infarction. (
Angiogenesis in the Ipsilateral Ventroposterior Nucleus
After MCAO, there was an increase in laminin-immunoreactive blood vessels and staining within the ipsilateral VPN when compared with the sham-operated rats (6.3% ± 0.7% versus 1.4% ± 0.4% at day 7, 10.8% ± 1.2% versus 1.6% ± 0.3% at day 14, both P<0.01; Figure 3E–3G). In addition, some BrdU-positive cell nuclei could be visualized clearly on the verge of laminin-labeled endothelial cells within the ipsilateral VPN of the MCAO rats (Figure 4G–4I), indicating angiogenesis in the ipsilateral thalamus remote from the primary infarction. Angiogenesis was seen at day 7 and up to day 14 after MCAO; the laminin-immunopositive blood vessels almost occupied the entire ipsilateral VPN.
Discussion
In this study, we found that the number of NeuN-labeled neurons was decreased and that OX-42-labeled microglia cells were activated within the ipsilateral VPN of the thalamus after focal infarction localized to the somatosensory cortex. Moreover, within the ipsilateral VPN, BrdU-positive cells were observed at day 7 and further increased in number at day 14 after MCAO. Both BrdU+/nestin+ cells and BrdU+/NeuN+ double-labeled cells were also seen in the ipsilateral VPN. In addition, the MCAO rats showed an increase in vascular density within the ipsilateral VPN when compared with the sham-operated rats. To the best of our knowledge, this is the first study that shows neurogenesis and angiogenesis within the ipsilateral VPN of the thalamus, which is remote from the primary ischemic infarction localized to the cortex.
Focal cortical infarct resulting from occlusion of the distal MCA is a good model for studying the secondary thalamic degeneration. In this model, the infarct is restricted to the ipsilateral somatosensory cortex, and the thalamus is spared because its blood supply through the posterior cerebral artery is unaffected by the MCAO (Scremin, 1995). Although the area of infarction did not involve the thalamus, marked neuron losses and microglia activation occurred within the ipsilateral VPN. Therefore, any thalamic degeneration is secondary to the primary cortical infarction rather than a direct injury due to the MCAO. Hypertensive rats have poor collateral circulation when compared with normal rats, and so an occlusion of the distal MCA would lead to a highly reproducible infarct located in the ipsilateral somatosensory cortex (Zeng et al, 1998a). That is the reason for which we applied the distal MCAO to the stroke-prone renovascular hypertensive rats.
Our findings of progressive neuronal loss plus microglia activation within the ipsilateral VPN after focal cortical infarction are consistent with the results of previous studies (Dihne et al, 2002; Schroeter et al, 2006). Several underlying mechanisms are possible. First, the thalamocortical projections connect the VPN with the ipsilateral somatosensory cortex. As the primary infarct is located in the somatosensory cortex, the cortical inputs to the VPN are interrupted. Second, retrograde degeneration of the thalamocortical projections may complicate focal cortical infarction. In addition, a high extracellular concentration of excitatory amino acids, a series of cytoplasmatic and nuclear events due to intracellular calcium overload, and the production of proinflammatory cytokines, nitrous oxide, glutamate, and oxygen free radicals may be deleterious to the neurons of the ipsilateral VPN and result in activated inflammatory responses (Watanabe et al, 1998; Dihne et al, 2002; Loos et al, 2003; Block et al, 2005).
It is well known that cerebral ischemia can stimulate cell proliferation in the subventricular zone and peri-infarct region (Xia et al, 2006; Ardelt et al, 2007; Ishibashi et al, 2007) in adults animals. Until now, cell proliferation in the thalamus has been described only in the normal or postnatal animals (Pencea et al, 2001; Mooney and Miller, 2007; Simmons et al, 2008). For example, Simmons et al (2008) reported the expression of BrdU-labeled cells in the thalamic nuclei of adult bullfrogs. Pencea et al (2001) described newly generated cells labeled by BrdU in specific regions surrounding the third ventricles, such as the ipsilateral thalamus and hypothalamus, after a combined intracerebroventricular administration of brain-derived neurotrophic factor and BrdU; the authors postulated that brain-derived neurotrophic factor-BrdU flowed caudally from the infused lateral ventricle into the third ventricle. Takasawa et al (2002) reported that the number of BrdU-positive cells was increased in the contralateral nonischemic hippocampus, which was remote from the primary ischemic infarction. To the best of our knowledge, the cell proliferation in the ipsilateral VPN of the thalamus after cerebral ischemia has never been reported. These results suggest that cell proliferation exists within the ipsilateral VPN of the nonischemic thalamus due to secondary damage after focal cortical infarction. Moreover, the delayed detection of BrdU-positive cells within the ipsilateral VPN at day 7 after MCAO would support secondary thalamic damage rather than primary ischemia. However, the exact source of the proliferating cells within the ipsilateral VPN remains elusive. Mooney and Miller (2007) proposed that the cell proliferation would occur in situ in the ventrobasal nucleus of postnatal rats because BrdU-positive cells appeared here as early as 30 mins after BrdU injection. Interestingly, we found that some BrdU-positive cells within the ipsilateral VPN were present in pairs or had two divided nuclei, suggesting cell division in the ipsilateral VPN. Taken together with Mooney's observation, we speculate that the proliferative cells in the VPN may be derived in situ rather than immigrated from outside the thalamus. This study cannot provide sufficient evidence to define the exact source of the proliferative cells within the ipsilateral VPN after cortical infarction.
A previous study has reported that focal ischemia stimulated the proliferation of neuronal progenitor cells in the contralateral hippocampus, a brain area remote from the primary ischemic lesion (Takasawa, et al, 2002). Nevertheless, little is known about the enhanced neurogenesis in areas remote to cerebral ischemia. One of the novel findings of our study was that not only BrdU/nestin double-labeled neural stem/progenitor cells but also BrdU/NeuN double-labeled newly formed neurons were observed within the ipsilateral VPN of the thalamus, indicating that neurogenesis indeed occurs within the ipsilateral VPN after focal cortical infarction. Although many BrdU-positive cells were detected within the ipsilateral VPN, it is important to note that only a few coexpressed with nestin or NeuN. Thus, neurogenesis is limited within this remote area and may be insufficient to compensate for the neuron losses during secondary degeneration. In general, many newly generated neuroblasts would not survive for a long period because of many unfavorable factors in the new environment, such as the limited supply of trophic substances, oxidative stress and excitotoxcity, Further investigations are needed to examine whether enhanced neurogenesis within the ipsilateral thalamus due to focal cerebral infarction would make a significant contribution to brain structural repair and functional recovery. Nevertheless, we confirm that neurogenesis occurs within the ipsilateral VPN of the thalamus after focal cortical infarction.
Another interesting finding was that the morphology of the nestin-positive cells within the ipsilateral VPN was similar to that of astrocytes. Some of the nestin+/GFAP+ cells might be early progenitors that would differentiate into late progenitors and then produce new neurons, but the majority of them would differentiate into astrocytes. Our results are in agreement with a previous study in which nestin is coexpressed with the astrocytic marker GFAP in reactive astrocytes after injuries to the central nervous system (Schwab et al, 2001). Recent studies have shown that astrocytes secrete various neurotrophic factors and evoke and regulate within the neurons the production of neurotrophic factors, provide metabolic support, and regulate glutamate transport to create an environment conducive for viable and functioning neurons (Lim and Alvarez-Buylla, 1999; Nedergaard et al, 2003). Thus, we believe that nestin+/GFAP+ cells within the ipsilateral VPN may promote functional recovery after ischemic stroke. In addition, Fukuda et al (2003) confirmed that nestin+/GFAP+ cells would represent the earliest precursors to neurons in the dentate gyrus. Recent studies have pointed out that the GFAP-positive cells indeed express nestin in the adult dentate gyrus, and that these nestin+/GFAP+ cells were early progenitors, which would differentiate into late progenitors and then produce newly formed neurons. In this study, our results showed that BrdU+ and nestin+/GFAP+ cells appeared within the ipsilateral VPN. Thus, we assume that nestin+/GFAP+ cells may also stand for the neural stem/progenitor cells within the ipsilateral VPN after cerebral ischemia, which are activated in the process of neurogenesis, and contribute to the functional recovery after ischemic stroke.
Numerous studies have shown that focal cerebral ischemia would promote angiogenesis in the periinfarct region (Natale et al, 2002; Wang et al, 2004; Ardelt et al, 2007). However, no report has focused on angiogenesis in the remote areas after cerebral ischemia. These results from laminin immunostaining indicate clearly angiogenesis in the ipsilateral VPN. Similar to glial activation, the blood vessels within the ipsilateral VPN were also activated and increased in number after cerebral ischemia. There were enlarged vessels walls, increased vascular density, and some BrdU-labeled nuclei were seen in the endothelial cells. Angiogenesis after cerebral ischemia is a highly complex process and is believed to involve multiple mechanisms, such as activity and trophic factors. We do not have any evidence to suggest a direct role of the retrograde degeneration of thalamocortical projections in the enhancement of vascular remodeling within the ipsilateral VPN after focal cortical ischemia. Instead, we believe that angiogenesis in the remote areas is associated with proliferating cells. Previously, we and others have shown the cell proliferation in the remote areas after focal cerebral ischemia (Takasawa et al, 2002). Some proliferating cells, such as microglia and astrocytes, can secrete neurotrophic and angiogenic factors, which in turn enhance vascular remodeling. Indeed, we have detected vascular endothelial growth factor-positive cells within the ipsilateral VPN of the ischemic animals but not within that of the sham-operated animals, suggesting that vascular endothelial growth factor secreted from proliferating cells may be at least partially responsible for vascular augmentation (unpublished data). In this study, neurogenesis and angiogenesis followed a similar time course after MCAO.
There are some limitations to our study. This study does not provide information on the source and migration pathway of the newly formed neuronal cells within the ipsilateral VPN, the underlying mechanisms of angiogenesis, and the appropriate microenvironmental signals necessary for neuronal survival in areas remote from the primary ischemic lesion. Further investigations are needed.
In conclusion, this study shows that both neurogenesis and angiogenesis occur within the ipsilateral VPN of the thalamus where secondary damage takes place after focal cortical infarction. Our results indicate that augmenting neurogenesis and angiogenesis may reduce the secondary damage after cerebral ischemia and promote functional recovery after stroke.
Footnotes
Authors declare no conflict of interest.
