Abstract
Recent studies demonstrated that neurogenesis in the adult hippocampus increased after transient global ischemia; however, the molecular mechanism underlying increased neurogenesis after ischemia remains unclear. The finding that proliferation of progenitor cells occurred at least a week after ischemic insult suggests that the stimulus was not an ischemic insult to progenitor cells. To clarify whether focal ischemia increases the rate of neurogenesis in the remote area, the authors examined the contralateral hemisphere in rats subjected to permanent occlusion of the middle cerebral artery. In the subgranular zone of the hippocampal dentate gyrus, the numbers of bromodeoxyuridine (BrdU)-positive cells increased approximately sixfold 7 days after ischemia. In double immunofluorescence staining, more than 80% of newborn cells expressed Musashi1, a marker of neural stem/progenitor cells, but only approximately 10% of BrdU-positive cells expressed glial fibrillary acidic protein (GFAP), a marker of astrocytes. The number of BrdU-positive cells markedly decreased 28 days after BrdU administration after ischemia, but it was still elevated compared with that of sham-operated rats. In double immunofluorescence staining, 80% of newborn cells expressed NeuN, a marker of differentiated neurons, and 10% of BrdU-positive cells expressed GFAP. However, in the other areas of the contralateral hemisphere including the rostral subventricular zone, the number of BrdU-positive cells remained unchanged. These results showed that focal ischemia stimulated the proliferation of neuronal progenitor cells, but did not support survival of newborn cells in the contralateral hippocampus.
Growing evidence indicates that neurogenesis still occurs in the subgranular zone (SGZ) in the hippocampus (Altman and Das, 1967; Gould et al., 1998; Eriksson et al., 1998; Roy et al., 2000) and the rostral migratory stream forms from the subventricular zone (SVZ) to the olfactory bulb (Pincus et al., 1998; Johansson et al., 1999; Doetsch et al., 1999) in the adult mammalian brain. Recent studies demonstrated increased neurogenesis in the dentate gyrus of the hippocampus after transient global ischemia in gerbils, rats, and mice (Liu et al., 1998; Takagi et al., 1999; Kee et al., 2001; Yagita et al., 2001). Although it was shown recently that increased neurogenesis was suppressed by treatment with glutamate receptor antagonists (Bernabeu and Sharp, 2000; Arvidsson et al., 2001) or with acetylsalicylic acid (Kumihashi et al., 2001), the molecular mechanism underlying increased neurogenesis still remains unclear. The proliferating activity in the SGZ reached a peak 7 to 10 days after ischemic insult. Because CA4 neurons in the hilus are the most vulnerable to ischemic insult (Schmidt-Kastner and Freund, 1991; Matsuyama et al., 1993), neuronal progenitor cells in the SGZ resided close to injured CA4 neurons in a transient global ischemia model. Late stimulation and proliferation of neuronal progenitor cells suggest that the stimulus was not an ischemic insult to progenitor cells themselves, but subsequent production of growth factors in brain tissue. Yoshimura et al. (2001) recently demonstrated involvement of basic fibroblast growth factor on increased neurogenesis after kainate seizures and ischemia, using basic fibroblast growth factor knockout mice. In terms of cell proliferation in a remote area, Jin et al. (2001) recently demonstrated increased incorporation of BrdU into cells in bilateral SGZ after focal ischemia. Although increased incorporation of BrdU in the contralateral hippocampus suggests the role of a diffusible or humoral factor, there are still unresolved points such as the characteristics of proliferating cells, cell survival, and neuronal differentiation in the contralateral SGZ. Therefore, we examined the proliferation of neuronal progenitor cells using a neural stem cell marker, Musashi1, and determined cell survival after proliferation and neuronal differentiation of the nonischemic, contralateral hemisphere including the hippocampus after permanent occlusion of the middle cerebral artery.
MATERIALS AND METHODS
Animal model
Adult male Wistar rats (2 months old) weighing 250 to 300 g (Charles River Laboratory, Yokohama, Japan) were used in this study. The experimental protocol was approved by the Institutional Animal Care and Use Committee of Osaka University Graduate School of Medicine. They were fed standard laboratory chow and had free access to water before and after all procedures. Animal care was given according to the guidelines of the Animal Center of Osaka University Graduate School of Medicine.
Each animal was anesthetized with halothane, and occlusion of the middle cerebral artery (MCA) was done according to Koizumi et al. (1986) and Longa et al. (1989). Briefly, the left common carotid artery was exposed through a midline incision, and the internal carotid artery was isolated and carefully separated. A 4–0 nylon monofilament, with its tip rounded by heating, was introduced from a bifurcation of the internal carotid artery and advanced until resistance was felt. Operated rats were used if they showed spastic paralysis of the right forelimb and circling to the right during the ischemic period. Rectal temperatures were monitored routinely during surgery to maintain animals at 37.0 ± 0.5°C. For the sham operation, a nylon filament was introduced from the carotid bifurcation and the tip was kept in the internal carotid artery.
Bromodeoxyuridine labeling and experimental design
We used bromodeoxyuridine (BrdU) (Boehringer Mannheim, Tokyo, Japan), a thymidine analog, to label proliferating cells. Bromodeoxyuridine was incorporated into newly synthesized DNA. In the first experiment (Fig. 1), rats of the control, sham, and ischemia groups (each time point 1, 4, 7, or 14 days after ischemia: n = 4 in each group) received BrdU (50 mg/kg, i.p.) administration three times every 4 hours during a period of 8 hours. The next day (each time point 2, 5, 8, or 15 days after ischemia, respectively), they were decapitated while they were under deep pentobarbital anesthesia, and their brains were removed and divided into coronal sections (approximately 5-mm thickness) in ice-cold methanol, and immersion fixed in methanol at 4°C overnight. After fixation, each tissue block was dehydrated and embedded in paraffin. The 5-μm-thick sections corresponding to the stereotaxic sections 1.5 mm rostral and 3.0 mm caudal to bregma, containing the caudoputamen and the dorsal hippocampus, respectively, were stained with the immunohistochemical reaction for BrdU. In the second experiment (Fig. 1), control and ischemic rats (n = 4 for each) were perfusion-fixed under deep pentobarbital anesthesia 1 day after BrdU administration. After perfusion with 4% paraformaldehyde in a 50-mmol/L phosphate buffer, the brains were removed and cut into coronal blocks containing the hippocampus, and immediately immersion fixed in 4% paraformaldehyde. Coronal sections (30 μm thick) were cut on a vibratome and processed for the double immunofluorescence. In the third experiment (Fig. 1), rats received BrdU (50 mg/kg i.p.) administration three times 7 days after MCA occlusion, and then control and ischemic rats were killed 1 day (n = 4 for each) or 28 days (n = 4 for each) after BrdU injection. The 5-μm-thick paraffin sections containing the dorsal hippocampus were stained with the immunohistochemical reaction for BrdU. Additional control rats and ischemic rats (n = 4 for each) were perfusion fixed 28 days with 4% paraformaldehyde after BrdU administration, and the vibratome sections were processed for the double immunofluorescence.
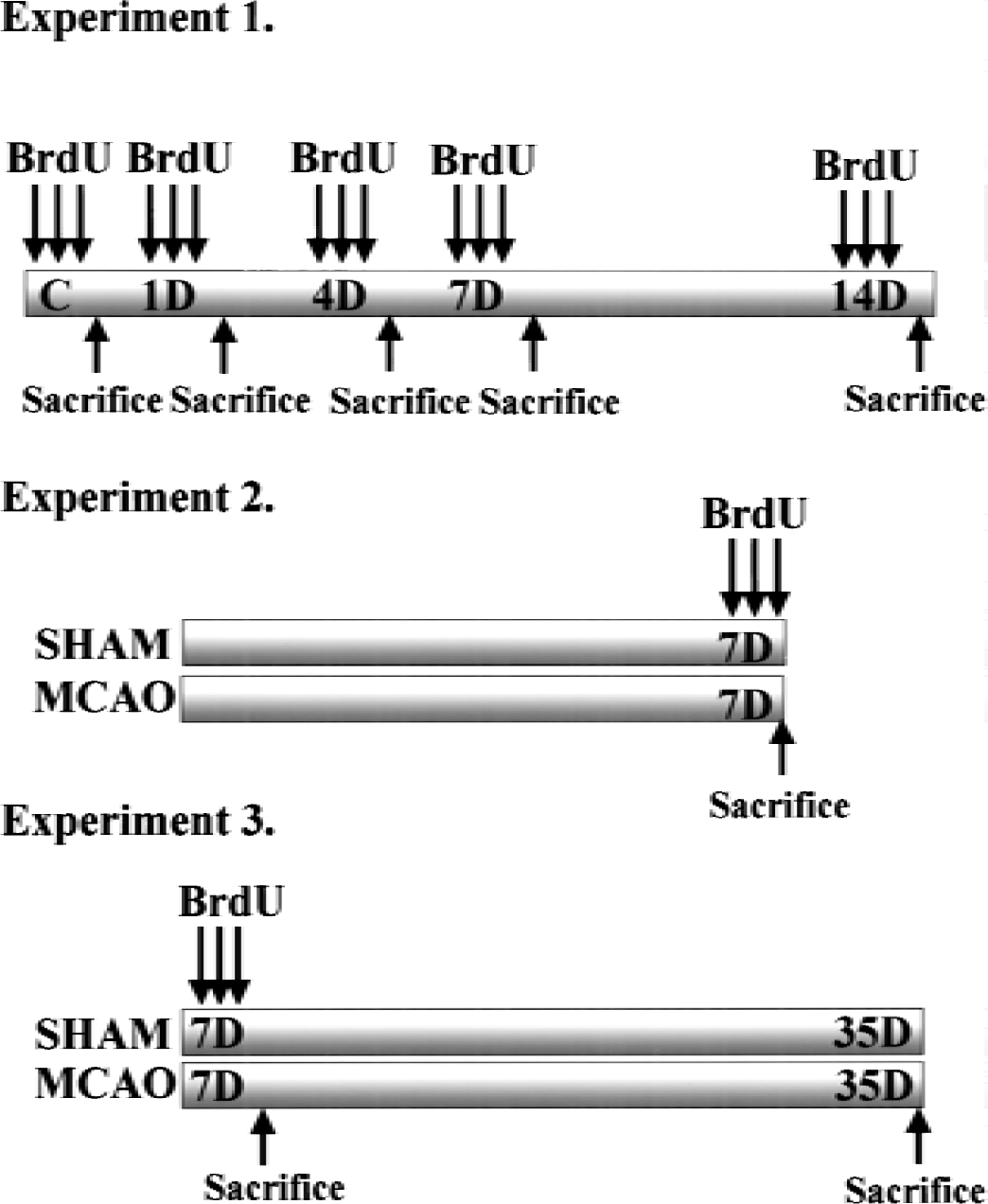
Bromodeoxyuridine (BrdU) labeling of ischemic brain. Bromodeoxyuridine (50 mg/kg, i.p.) was given three times every 4 hours during a period of 8 hours. In the first experiment (experiment 1), rats received BrdU 1, 4, 7, and 14 days after ischemia (n = 4 in each group), and the next day they were killed and the brains were processed for paraffin sections and used for counting BrdU-positive cells. In the second experiment (experiment 2), rats received BrdU on day 7 after ischemia, and the next day they were killed and the vibratome sections were prepared for double immunofluorescence. In the third experiment (experiment 3), rats received BrdU on day 7 after ischemia, and they were killed 1 day or 28 days after BrdU injection. The paraffin and vibratome sections were prepared for counting BrdU-positive cells and for double immunofluorescence, respectively.
Immunohistochemistry
For BrdU immunohistochemistry, DNA denaturing was required. Each deparaffinized section was treated in 50% form-amide and a 2x saline–sodium citrate buffer at 65°C for 2 hours. After washing in the 2x saline–sodium citrate buffer, sections were incubated in 2N HCl at 37°C for 30 minutes. Sections were rinsed in Tris-buffered saline/0.1% Triton X-100 for 20 minutes, and incubated with a monoclonal anti-BrdU antibody (1:100; Amersham, Arlington Heights, IL, U.S.A.) at 4°C overnight. After washing in Tris-buffered saline/0.1% Triton X-100, the sections were incubated with a biotinylated secondary antibody for 1 hour at room temperature. They were washed and further incubated with a streptavidin–biotin–peroxidase complex (Vector, Burlingame, CA, U.S.A.). The sections were finally reacted with 0.05% 3′3-diaminobenzidine in the presence of 0.01% H2O2. The control sections were incubated with the nonimmune serum or ascites.
For the double-immunofluorescence technique, vibratome brain sections were incubated with a primary antibody diluted with Tris-buffered saline/0.1% Triton X-100 at 4°C overnight. We used the following antibodies as primary antibodies; a mouse monoclonal anti-BrdU antibody (Amersham 1:100), a rat monoclonal anti-BrdU antibody (1:200; Harlan Sera-labo, Loughborough, U.K.), a mouse monoclonal anti-NeuN antibody (1:200; Chemicon, Temecyla, CA, U.S.A.), a mouse monoclonal anti-microtubule-associated protein 2 (MAP2) antibody (1:200; Sigma-Aldrich, Tokyo, Japan), a rabbit polyclonal anti-glial fibrillary acidic protein (GFAP) antibody (1:200 Sigma), a goat polyclonal anti-doublecortin (DCX) antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.), and a rat monoclonal anti-Musashi1 antibody. Double immunostaining was performed using immunofluorescence and confocal microscopy (Zeiss). For double labeling of BrdU and cell markers (NeuN and MAP2: neuron, GFAP: astrocytes, DCX: immature neuron, Musashi1: neuronal progenitor cells and astrocytes), sections were incubated with an anti-BrdU antibody and antibodies for each cell marker at 4°C overnight after DNA denaturation. DCX is a protein required for neuronal migration and it is expressed in both migrating neuroblasts and differentiated neurons (Francis et al., 1999; Nacher et al., 2001). Fluorescein isothiocyanate- or rhodamine-labeled goat or donkey anti-immunoglobulin G (IgG) antibodies were used as the secondary antibodies. The combination of antibodies used in each double-immunostaining experiment was as follows: BrdU-NeuN and BrdU-MAP2; the rat anti-BrdU antibody and mouse anti-NeuN or anti-MAP2 antibody as primary antibodies, and the rhodamine-labeled anti-rat IgG antibody and fluorescein isothiocyanate–labeled anti-mouse IgG as secondary antibodies. BrdU-DCX; the rat anti-BrdU antibody and goat anti-DCX antibody as primary antibodies, and the rhodamine-labeled anti-rat IgG antibody and fluorescein isothiocyanate-labeled anti-goat IgG as secondary antibodies. BrdU-GFAP; the rat anti-BrdU antibody and rabbit anti-GFAP antibody as primary antibodies, and the rhodamine-labeled anti-rat IgG antibody and fluorescein isothiocyanate-labeled anti-rabbit immunoglobulin G antibody as secondary antibodies. BrdU-Musashi1; the mouse anti-BrdU antibody and rat anti-Musashi1 antibody as primary antibodies, and the rhodamine-labeled anti-mouse IgG antibody and fluorescein isothiocyanate-labeled anti-rat IgG antibody as secondary antibodies.
Quantification
To count BrdU-positive cells in paraffin sections colored by the peroxidase reaction, five sections from the caudoputamen and the hippocampus were obtained every 150 μm beginning at a section 1.5 mm rostral and 3.0 mm caudal to the bregma, respectively. The area for quantification was as follows (Fig. 2): the frontal, cingulate, and parietal cortex, caudoputamen, and thalamus in the contralateral hemisphere, and the rostral subventricular zone and the dentate gyrus of the hippocampus in both hemispheres. In the hippocampus, the granular cell layer (GCL) (approximately 60 μm) and SGZ, defined as a two-cell-body–wide zone (approximately 10 μm) along the border of the GCL and the hilus, were always combined for quantification. The mean density of BrdU-labeled cells in each rat was calculated as the number of labeled nuclei/area. Statistical analysis was performed using analysis of variance followed by Scheffé post hoc tests.
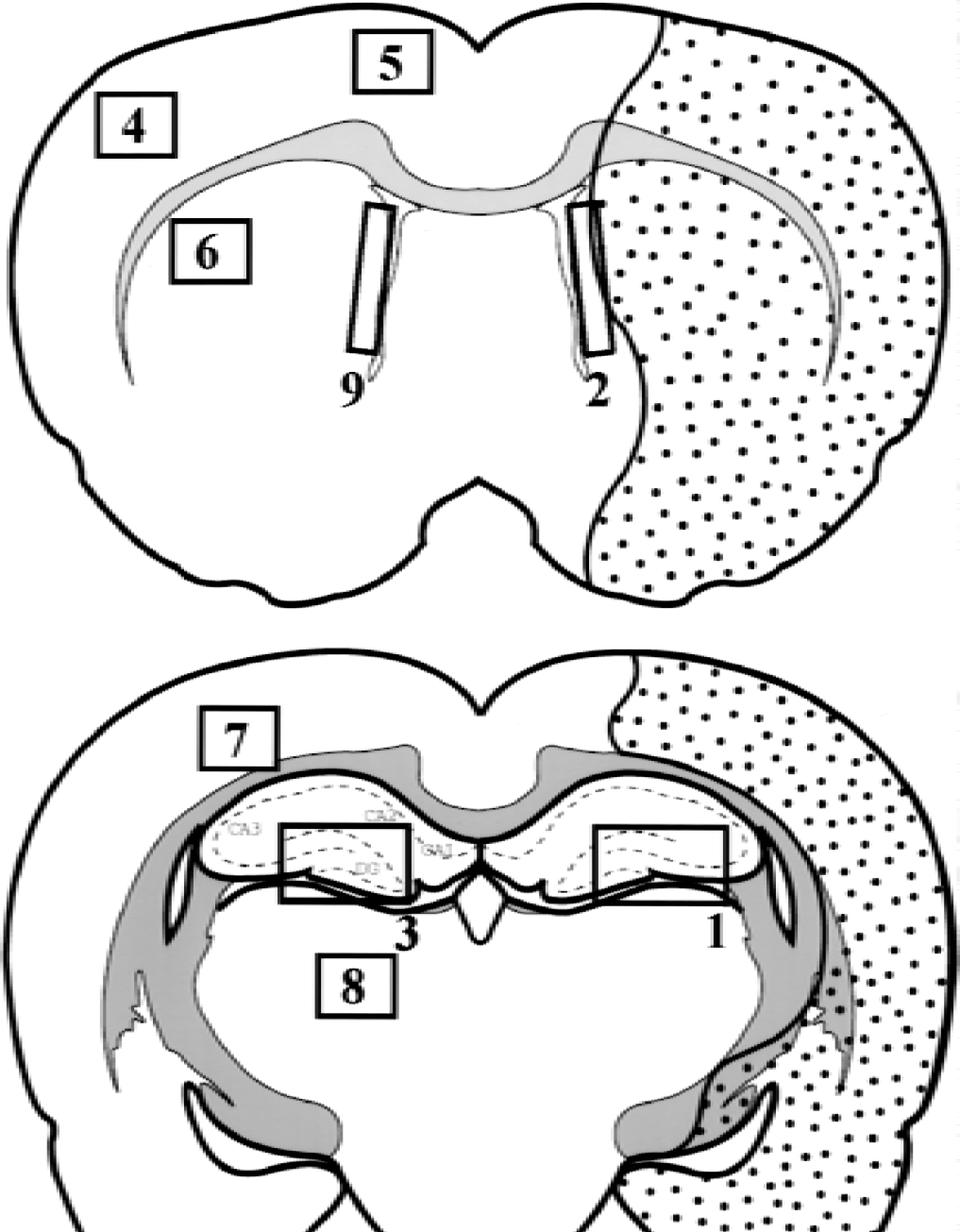
Diagram of the brain sections in the rat brain after middle cerebral artery occlusion. Infarct area was shown as stippled area. Number of BrdU-positive cells was quantified in nine boxed areas as follows: 1,2: ipsilateral hemisphere; 1: hippocampus; 2: rostral subventricular zone; 3–9: contralateral hemisphere; 3: hippocampus; 4: frontal cortex; 5: cingulate cortex; 6: caudoputamen; 7: frontoparietal cortex; 8: thalamus; 9: rostral subventricular zone.
To assess the phenotype of BrdU-positive cells after ischemia, we used a double-immunostaining technique. We detected BrdU-positive cells in the SGZ and GCL, and determined whether they expressed Musashi1, DCX, NeuN, MAP2, or GFAP with confocal microscopy. A double positive percentage was calculated as BrdU+/Musashi1+, BrdU+/NeuN+, or BrdU+/GFAP+ cells for total BrdU-positive cells. The mean value of each data was obtained in eight sections from four rats.
RESULTS
A few BrdU-positive cells were observed in the SGZ of the hippocampal dentate gyrus in the control (Figs. 3 and 4). In the hippocampus that was ipsilateral to ischemia, the number of BrdU-positive cells increased 1 day after MCA occlusion, reached a peak 4 days later, and remained elevated until 7 days after ischemia (Fig. 3). In contrast, in the contralateral hippocampus, the number of BrdU-positive cells in the SGZ remained unchanged until 4 days after MCA occlusion, increased markedly 7 days after ischemia, and then declined to the control level 14 days after ischemia (Fig. 4). Semiquantitative analysis showed that the number of BrdU-positive cells increased approximately sixfold 7 days after ischemia compared with the control (Fig. 4). Only a few BrdU-positive cells were observed in the dentate hilus from 1 to 14 days after ischemia. In other areas of the contralateral hemisphere including the rostral SVZ, the number of BrdU-positive cells remained unchanged from 1 day to 14 days after ischemia (Table 1). In the ipsilateral SVZ, the number of BrdU-positive cells significantly increased 7 days after ischemia.
BrdU-positive cells in the bilateral hemisphere after middle cerebral artery occlusion
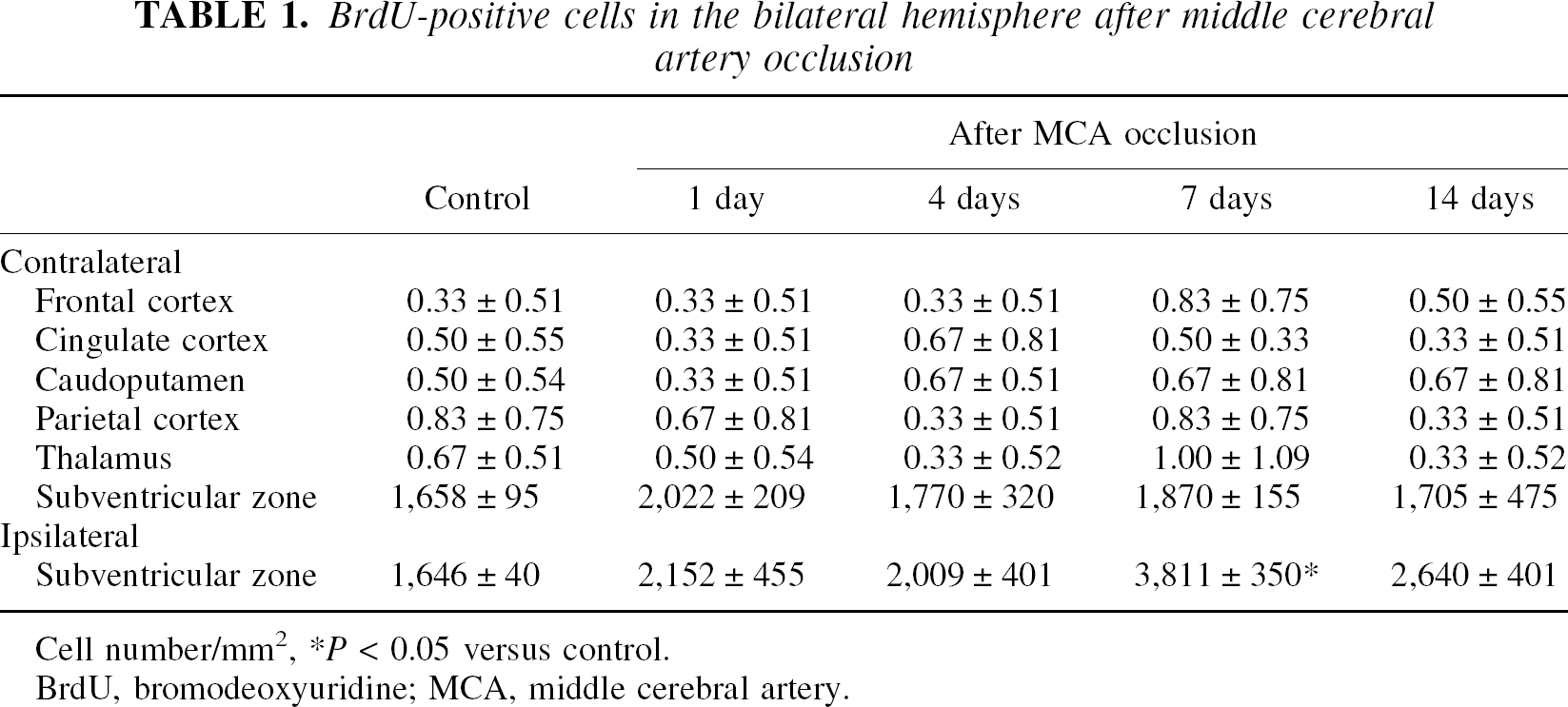
Cell number/mm2
P < 0.05 versus control.
BrdU, bromodeoxyuridine; MCA, middle cerebral artery.
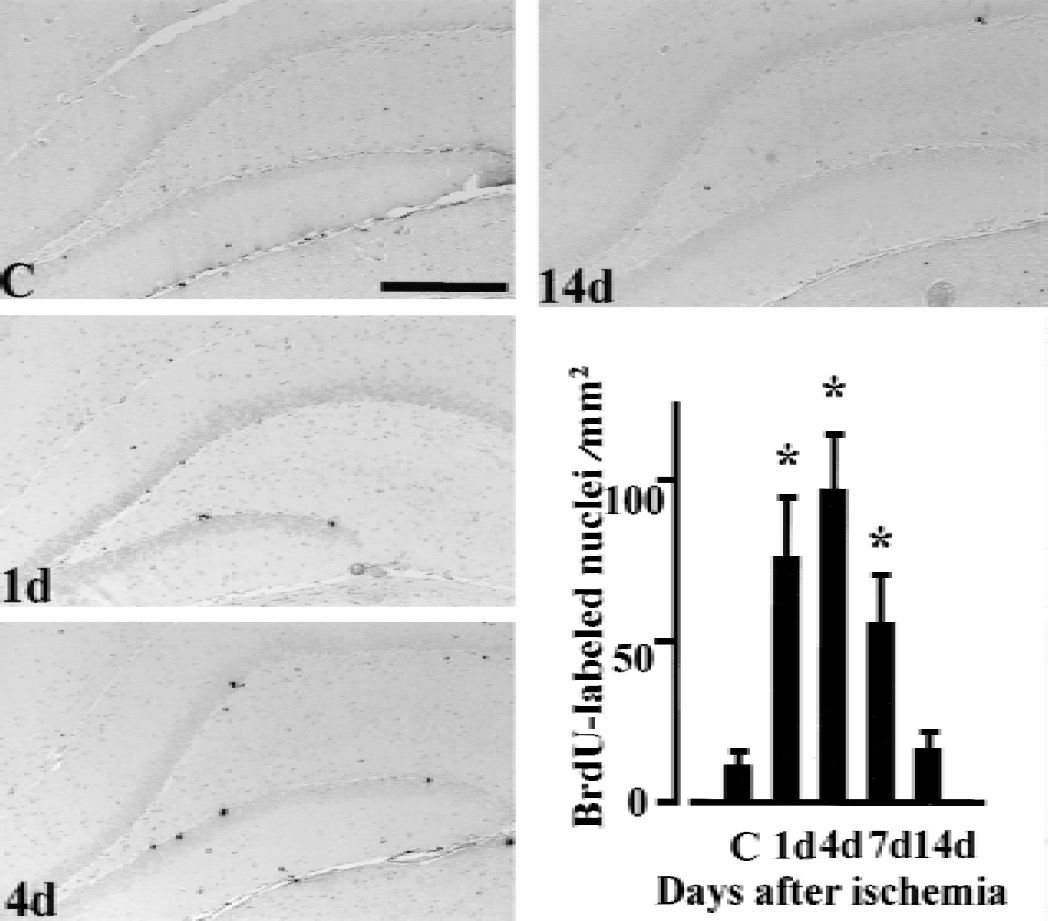
Bromodeoxyuridine (BrdU)-positive cells in the ipsilateral hippocampus after middle cerebral artery occlusion. All rats were administered BrdU (50 mg/kg, i.p.) three times a day before they were killed. Note the increase in BrdU-positive cells in the subgranular zone 1 day (1d) to 7 days (7d) after ischemia. Bar = 0.5 mm. The significance of differences was determined using analysis of variance followed by Scheffé post hoc test. Each column and bar denotes mean ± SD. *P < 0.05 compared with the control group. C: control animal; 1d, 4d, 7d and 14d: 1 day, 4 days, 7 days, and 14 days after middle cerebral artery occlusion.
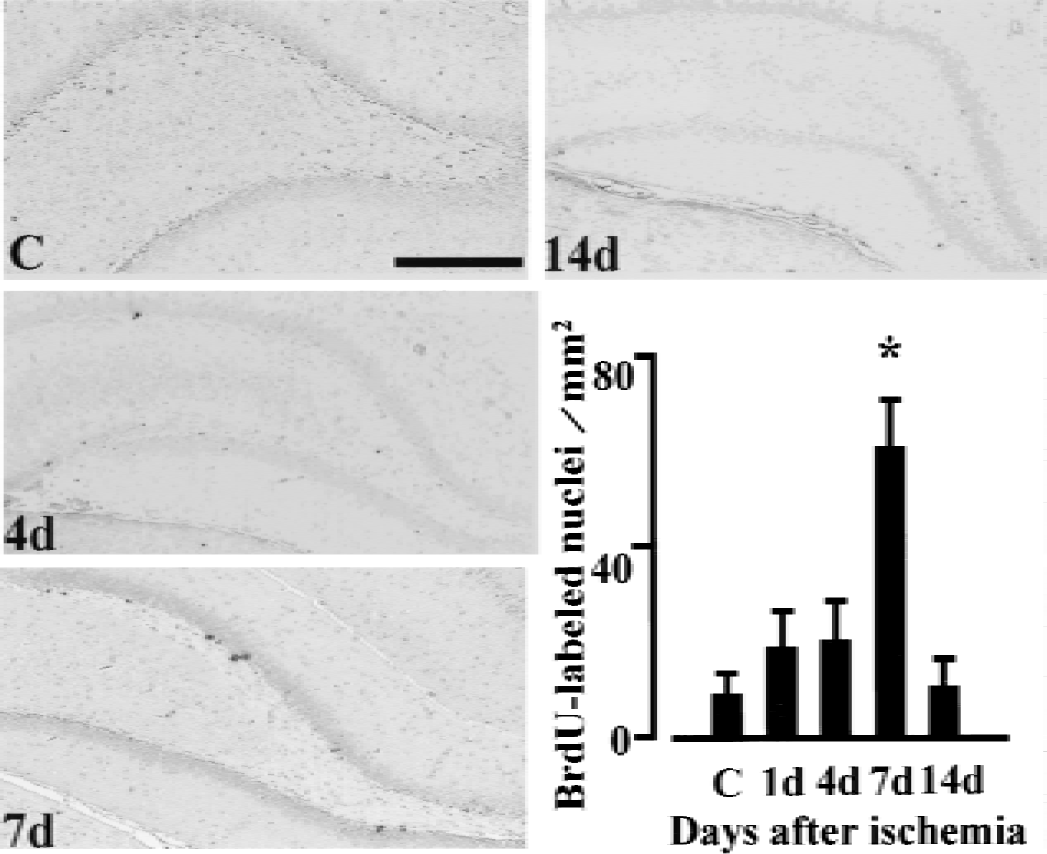
Bromodeoxyuridine (BrdU)-positive cells in the contralateral hippocampus after middle cerebral artery occlusion. All rats were administered BrdU (50 mg/kg, i.p.) three times a day before they were killed. Note the marked increase in BrdU-positive cells in the subgranular zone 7 days after ischemia (7d), compared with the control (C). Bar = 0.5 mm. The significance of differences was determined using analysis of variance followed by Scheffé post hoc test. Each column and bar denotes mean ± SD. *P < 0.05 compared with the control group. C: control animal; 1d, 4d, 7d and 14d: 1 day, 4 days, 7 days, and 14 days after middle cerebral artery occlusion.
To examine the phenotype of BrdU-positive cells in the contralateral hippocampus 1 day after BrdU administration, double immunofluorescence for BrdU/Musashi1, BrdU/DCX, or BrdU/GFAP was examined (Fig. 5). In both sham-operated rats and ischemic rats 7 days after MCA occlusion, the majority of BrdU-positive cells expressed both Musashi1 and DCX. In the semiquantitative analysis, approximately 80% of BrdU-positive cells in the SGZ colocalized with Musashi1 (79.8 ± 9.7% in control rats and 80.8 ± 9.8% in ischemic rats), but only 20% of BrdU-positive cells colocalized with GFAP (18.3 ± 4.5% in control rats and 17.8 ± 5.4% in ischemic rats). However, in the ipsilateral hippocampus of ischemic rats, BrdU-positive but Musashi1-negative cells were often observed (Fig. 6). Only 50.5 ± 10.4% of BrdU-positive cells in the SGZ colocalized with Musashi1, and 42.7 ± 4.7% of BrdU-positive cells colocalized with GFAP (Fig. 6). The percentage of both BrdU- and DCX-positive cells per total BrdU-positive cells in the SGZ was 76.7 ± 7.2% and 31.7 ± 9.7% in the contralateral and in the ispsilateral hippocampus, respectively.
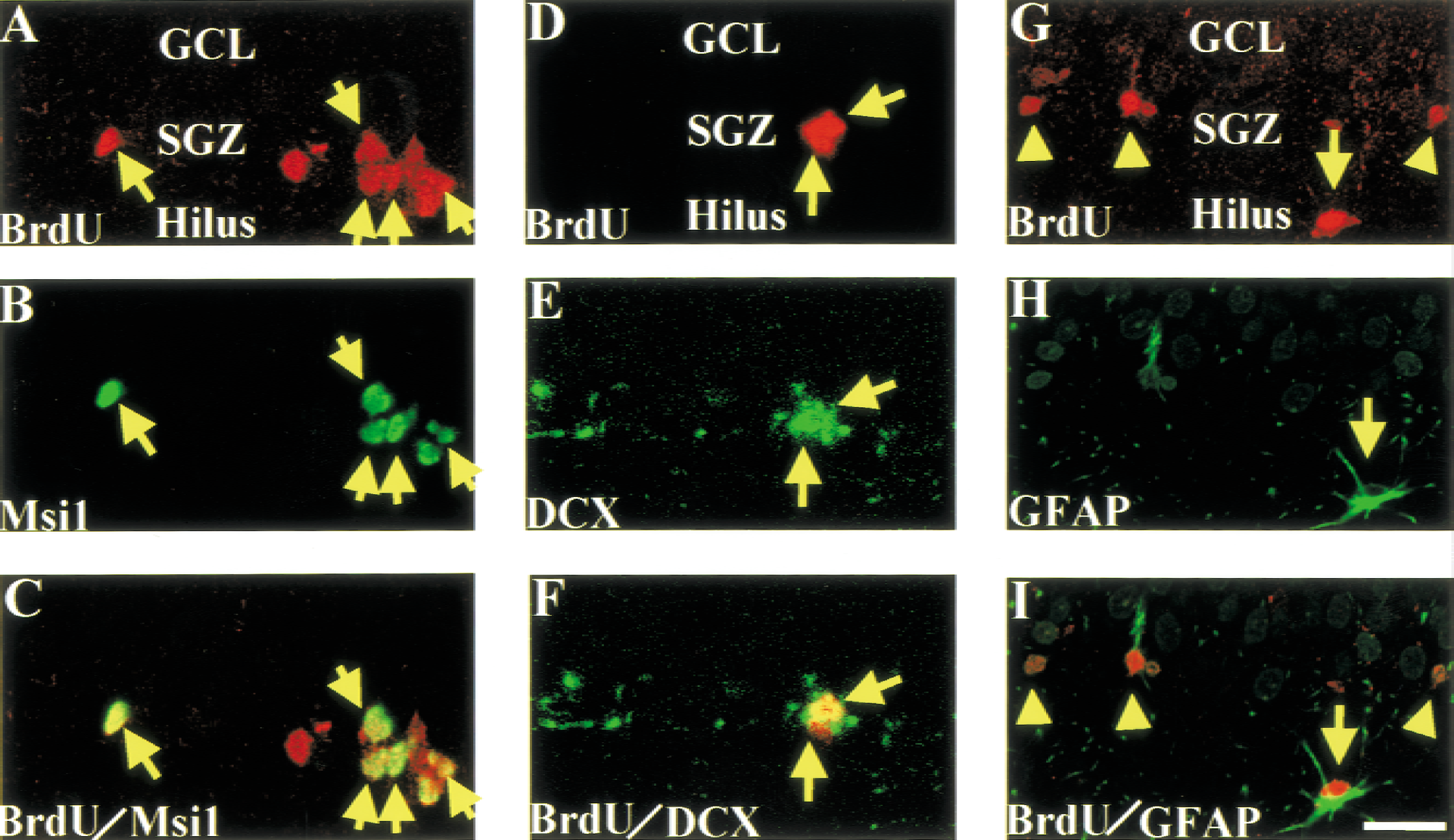
Proliferation of Musashi1 (Msi1) and doublecortin (DCX)-positive and glial fibrillary acidic protein (GFAP)-negative cells in the contralateral subgranular zone (SGZ). To label newborn cells, rats received bromodeoxyuridine (BrdU; 50 mg/kg, i.p.) administration three times, 7 days after ischemia, and were killed a day after BrdU injection. In the SGZ and granular cell layer (GCL) (A–C;
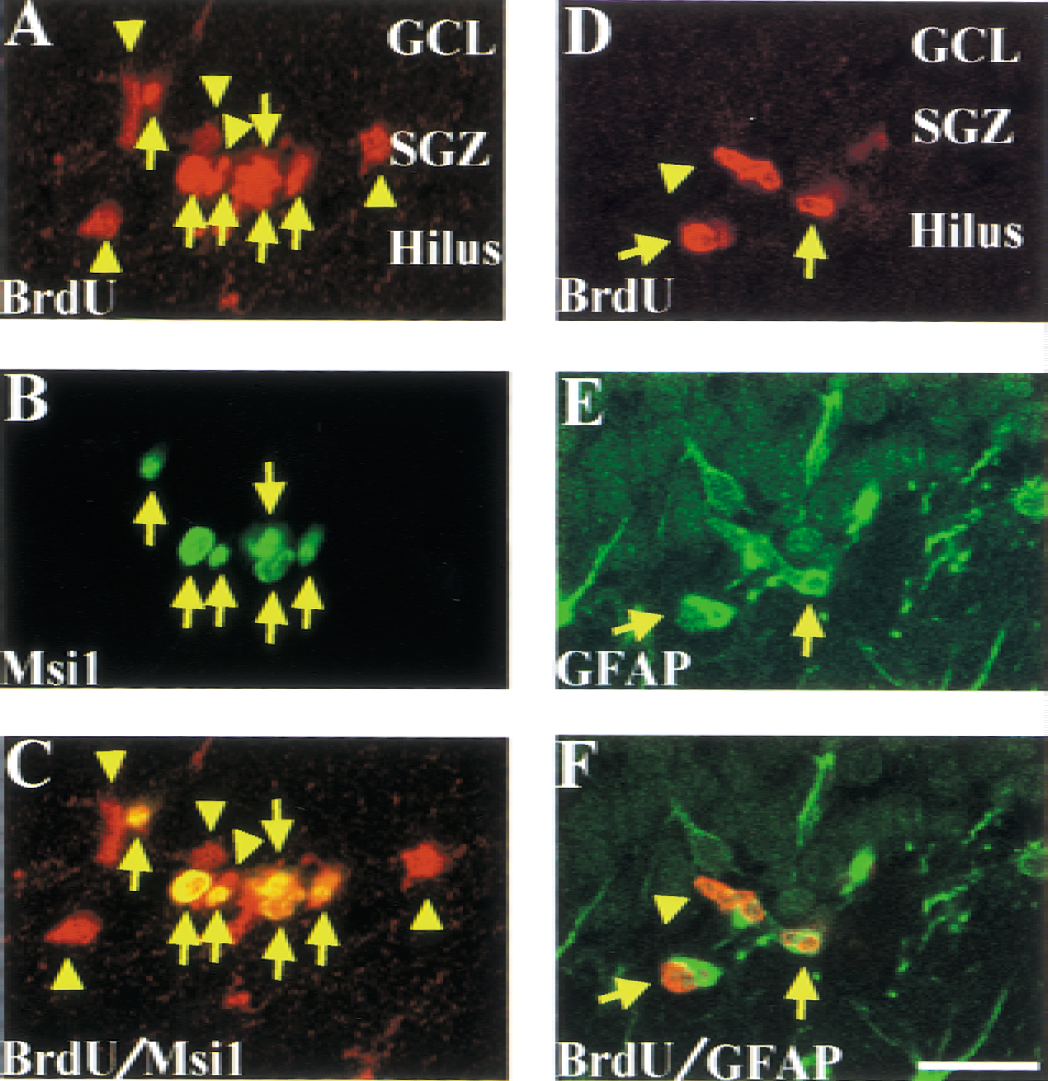
Proliferation of Musashi1 (Msi1)-negative cells in the ipsilateral subgranular zone (SGZ). The same brain sections in Figure 5 were used to examine proliferating cells in the ipsilateral hippocampus after middle cerebral artery occlusion. In the SGZ and granular cell layer (GCL) of the ipsilateral hippocampus (
To examine the survival or reduction of BrdU-positive cells, the number of BrdU-positive cells in the SGZ and GCL of the contralateral hippocampus 28 days after BrdU administration was counted in both control and ischemic rats. The survival rate in control rats was 92%, whereas that in ischemic rats was only approximately 20% (Table 2), although there were still a significantly larger number of BrdU-positive cells in the ischemic rat hippocampus than those of sham-operated rats (Table 2). In the ipsilateral hippocampus, the number of BrdU-positive cells also decreased from 98.0 ± 22.5/mm2 (1 day after BrdU injection) to 20.3 ± 6.6/mm2 (28 days after BrdU injection); however, it was not possible to calculate the residual rate of BrdU-positive cells because many BrdU-positive cells were Musashi1-negative and would likely be inflammatory cells. We further observed that BrdU-positive postmitotic cells expressed the neuronal antigenic phenotypes, NeuN and MAP2, 28 days after BrdU injection with double immunofluorescence (Fig. 7). In the contralateral hippocampus, the majority of BrdU-positive cells expressed both NeuN and MAP2. In the semiquantitative analysis, approximately 80% of all BrdU-positive cells represented NeuN both in the control and ischemia groups (Table 2). In contrast, only 10% of BrdU-positive cells in the SGZ and GCL colocalized with GFAP (Fig. 7; Table 2).
Survival and differentiation into neurons 28 days after BrdU labeling in the contralateral SGZ and GCL

P < 0.05 versus control.
BrdU, bromodeoxyuridine; SGZ, subgranular zone; GCL, granular cell layer; GFAP, glial fibrillary acidic protein.
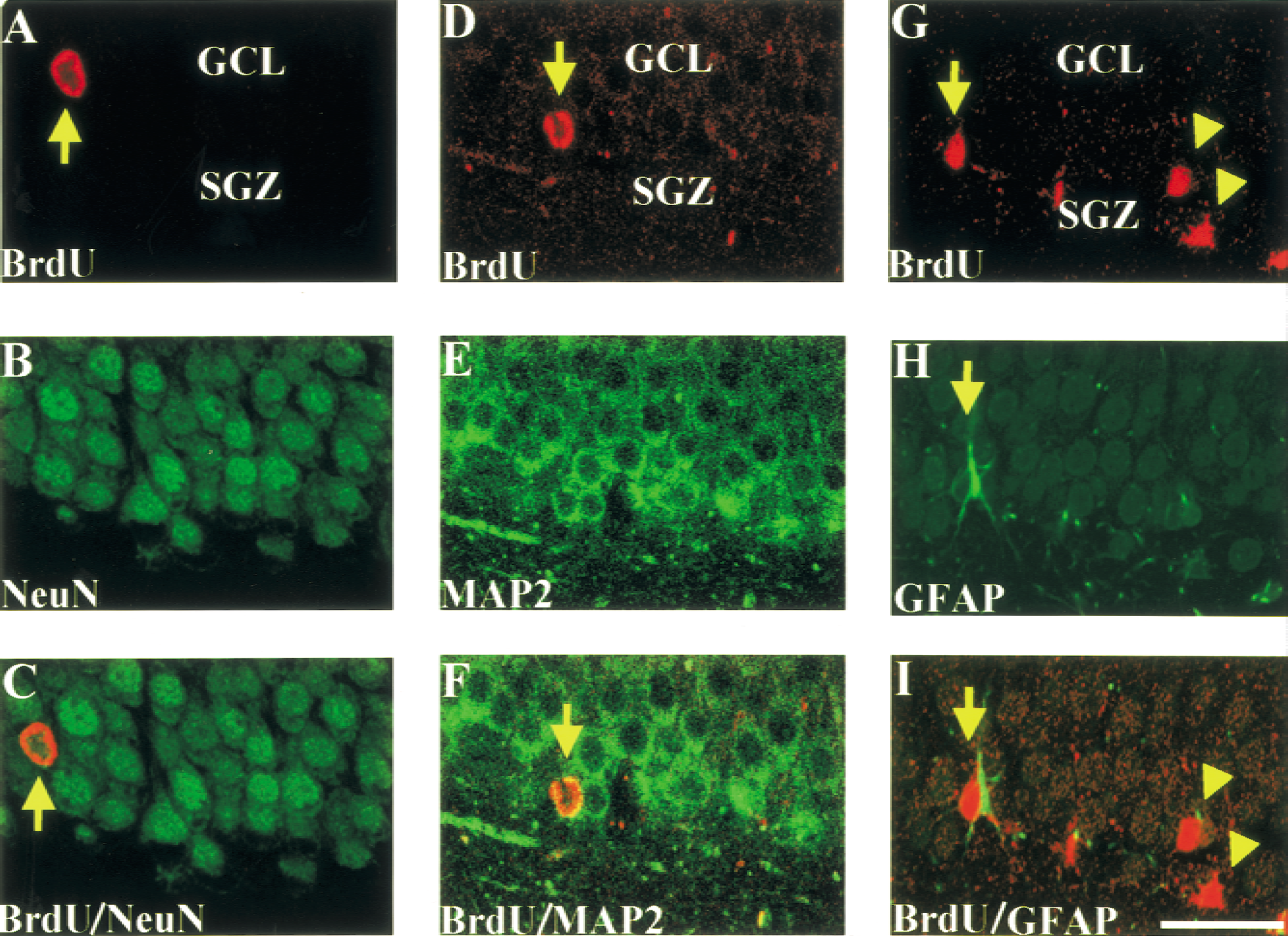
Neuronal differentiation of newborn cells in the contralateral hippocampus after middle cerebral artery occlusion. Rats received BrdU (BrdU; 50 mg/kg, i.p.) administration three times, 7 days after ischemia, and were killed 28 days after BrdU injection. Left and middle panels: In the subgranular zone (SGZ) and granular cell layer (GCL) (
DISCUSSION
In the present study, we demonstrated that proliferation of neuronal progenitor cells in the SGZ of nonischemic hippocampus increased after focal ischemia. However, survival of those proliferating cells markedly reduced, so focal ischemia resulted in only a slight neurogenesis in the contralateral SGZ. As control, we also examined ipsilateral hippocampus and found a rapid increase in the number of BrdU-positive cells (Table 1). However, the majority of BrdU-positive cells could be not progenitor cells but inflammatory cells or astrocytes residing close to injured neurons, because about half of BrdU-positive cells were Musashi1-negative in the ipsilateral hippocampus, which is often injured in the nylon suture model (States et al., 1996). The dissociation of the peaks of cell proliferation in the ipsilateral (4 days) and contralateral (7 days) hippocampi is likely ascribed to the different population of proliferative cells. Liu et al. (1998) first demonstrated increased neurogenesis in the hippocampus after transient global ischemia in gerbils. Later, similar findings were observed after transient global ischemia in the hippocampus of mice (Takagi et al., 1999) and rats (Kee et al., 2001). Recently, we demonstrated proliferation of neural stem/progenitor cells using Musashi1 as a marker of neural stem or progenitor cells after transient global ischemia in rats (Yagita et al., 2001). In a focal-ischemia model, Gu et al. (2000) demonstrated neurogenesis in the peripheral area of cerebral infarction. All previous studies demonstrated that the number of BrdU-positive cells in the SGZ reached a peak 7 to 10 days after ischemia, suggesting the involvement of lesion-induced factors, rather than ischemic insult itself, in the increased neurogenesis after ischemia. If this hypothesis is true, focal ischemia and cerebral infarction result in increased neurogenesis in the contralateral SGZ. There are only two reports (Jin et al., 2001; Zhang et al., 2001) about neurogenesis in the contralateral hippocampus after cerebral infarction. Strictly speaking, Jin et al. demonstrated only an increased incorporation of BrdU in the contralateral SGZ. Zhang et al. found no significant difference in number of BrdU-positive cells between the ipsilateral and the contralateral dentate gyrus.
The first novel finding in the present study was the increased proliferation of neural stem/progenitor cells in the contralateral, nonischemic SGZ. We used Musashi1 as a marker of neural stem/progenitor cells as we did in a transient global ischemia model (Yagita et al., 2001). Musashi1 is a neural RNA-binding protein, and is expressed in neural stem/progenitor cells and astrocytes in the adult brain (Sakakibara et al., 1996; Sakakibara and Okano, 1997; Kaneko et al., 2000). Because the majority of astrocytes express GFAP, neural stem/progenitor cells could be recognized as Musashi1-positive and GFAP-negative cells. However, recently, it has been shown that GFAP-positive astrocytes (designated as “type B” cells) in the SGZ of the adult hippocampal dentate gyrus are able to give rise to new neurons via GFAP-negative intermediate neuronal progenitor cells (designated as “type D” cells) (Seri et al., 2001). In the present study, we observed that more than 80% of BrdU-positive cells expressed Musashi1 after the survival time of 1 day in the control SGZ and the contralateral SGZ after MCA occlusion. However, only 20% of BrdU-positive cells expressed GFAP in the same condition (Table 1). This finding could be explained as follows. Although the primary neuronal progenitor cells are GFAP-positive astrocytes (type B cells), GFAP, an intermediate filament, may be degraded soon after the cell division. It would be interesting if cells at this stage become Musashi1 positive. It should be also noted that BrdU may label neurons undergoing cell death as well as newly divided cells (Bjorklund and Lindvall, 2000). However, there were no cells in the contralateral SGZ positive for fragmented DNA with in situ nick-end labeling (data not shown); thus, most BrdU-positive cells in that area are likely to be newly divided cells. Proliferation of neural stem/progenitor cells reached a peak 7 days after ischemia, similar to a temporal profile observed in the SGZ after transient global ischemia (Liu et al., 1998; Yagita et al., 2001). Increased proliferation of neural stem/progenitor cells in the contralateral SGZ supported the involvement of lesion- or infarction-induced diffusible or humoral factors such as neurotrophic factors. It was already demonstrated that several growth factors such as epidermal growth factors (Reynolds and Weiss, 1992), basic fibroblast growth factor (Kuhn et al., 1997), brain-derived neurotrophic factor (Ahmed et al., 1995), and insulin-like growth factor-1 (Arsenijevic and Weiss, 1998) are important in the proliferation and differentiation of neural stem cells. However, the loss of a synaptic input from the infarcted hemisphere may be also involved in the proliferation of neural stem/progenitor cells in the contralateral SGZ. In contrast to recently reported findings (Jin et al., 2001), we could not observe an increase in the number of BrdU-positive cells in the contralateral SVZ after ischemia. Reperfusion or reoxygenation may accelerate proliferation of neural progenitor cells in the SVZ, because Jin et al. used transient MCA occlusion followed by recirculation, but we used a permanent MCA occlusion model. Future study will be required to resolve this disagreement. No increase in BrdU-positive cells in other areas of the contralateral hemisphere would reflect no or very little neurogenesis in the corresponding area of the contralateral side after cerebral infarction.
The second novel finding was the reduced survival of proliferating cells in the SGZ and dentate gyrus after focal ischemia. Although the percentage of neuronal differentiation of BrdU-positive cells was similar between control (73%) and ischemic rats (85%) (Table 2), survival of BrdU-positive cells 28 days after BrdU administration was only approximately 20% of that 1 day after labeling in ischemic rats. However, survival of BrdU-positive cells in the control rats (92%) was in fair agreement with the previous findings (Yagita et al., 2001). Although we could not exclude the possibility that disappearance of BrdU-positive cells may indicate fast DNA replication or repair, it is unlikely that BrdU-positive cells divide several times after the next day of labeling, because approximately 80% of BrdU-positive cells expressed immature neuronal marker, DCX, a day after labeling (Fig. 5). Jin et al. (2001) demonstrated contralateral hippocampal neurogenesis in the same model of rat focal ischemia, although they did not examine survival or neuronal differentiation of BrdU-positive cells. Survival and neuronal differentiation of proliferating cells after ischemia may require local factors or a high concentration of growth factors secreted by reactive astrocytes or microglia that reside close to the proliferating cells. Recent evidence demonstrated that proliferation and survival/neuronal differentiation of neural stem cells was determined by distinct factors. For example, exposure to an enriched environment or hippocampus-dependent learning task increased the number of surviving newborn cells (van Praag et al., 1999; Gould et al., 1999) but did not affect proliferation (van Praag et al., 1999). In contrast, adrenalectomy, by reducing corticosteroid levels, increases not only cell proliferation, but also granule cell death in the dentate gyrus through apoptosis (Cameron and McKay, 1999). Recently, we also demonstrated that cell proliferation was preserved, but cell survival was hampered, in the ischemic aged rat hippocampus (Yagita et al., 2001).
Although it remains unclear whether and how increased neurogenesis in the remote area after cerebral infarction may be involved in neuronal plasticity, the molecular mechanisms underlying increased proliferation of neural stem cells, and survival and neuronal differentiation of proliferating cells, are distinct and need to be elucidated to aid development of a novel strategy for enhancing neurogenesis in patients who have had stroke.
Footnotes
Acknowledgments:
The authors thank Y. Nishizawa, R. Morimoto, and S. Imoto for their secretarial assistance, and Drs. Arturo Alvarez-Buylla and Kazunobu Sawamoto for their valuable comments on this article.
