Abstract
Calcitonin gene-related peptide and adrenomedullin belong to a structurally related neuropeptide family and are potent vasodilators expressed in the trigeminovascular system. The molecular identity of receptors for these proteins has only recently been elucidated. Central to functional binding of these neuropeptides is the G-protein–coupled receptor, the calcitonin receptor–like receptor (CRLR), whose cell surface expression and pharmacology is determined by coexpression of a receptor activity-modifying protein (RAMP). CRLR combined with RAMP1 binds calcitonin gene-related peptide with high affinity, whereas CRLR coexpression with RAMP2 or −3 confers high-affinity binding of adrenomedullin. The authors investigated the expression of these receptor components in human cerebral vasculature to further characterize neuropeptide receptor content and the potential functions of these receptors. Localization has been carried out using specific antisera raised against immunogenic peptide sequences that were subsequently applied using modern immunohistochemical techniques and confocal microscopy. The results are the first to show the presence of these receptor component proteins in human middle meningeal, middle cerebral, pial, and superficial temporal vessels, and confirm that both calcitonin gene-related peptide and adrenomedullin receptors may arise from the coassembly of RAMPs with CRLR in these vessel types. These novel data advance the understanding of the molecular function of the trigeminovascular system, its potential role in vascular headache disorders such as migraine, and may lead to possible ways in which future synthetic ligands may be applied to manage these disorders.
Calcitonin gene-related peptide (CGRP), adrenomedullin, amylin, and calcitonin belong to a family of neuropeptides involved in diverse biologic functions, including vasodilation. Over recent years, several receptors that mediate responsiveness to CGRP and adrenomedullin have been cloned, including RDC1 (Kapas and Clark, 1995), adrenomedullin receptor/L1 (Kapas et al., 1995), and calcitonin receptor–like receptor (CRLR) (Aiyar et al., 1996; Fluhmann et al., 1995; Han et al., 1997; Njuki et al., 1993). CRLR is perhaps the most studied of these receptors, and recent data suggest that the cell surface expression and pharmacology of this G-protein–coupled receptor is governed by coexpression with one of three receptor activity-modifying proteins (RAMPs) (Buhlmann et al., 1999; Husmann et al., 2000; McLatchie et al., 1998; Hilairet et al., 2001; Oliver et al., 2001). Association of CRLR with RAMP1 confers responsiveness to CGRP, whereas CRLR association with RAMP2 or RAMP3 confers responsiveness to adrenomedullin. Furthermore, RAMPs have also been shown to alter the pharmacology of the calcitonin receptor, which when coexpressed with RAMP1 or RAMP3 confers high-affinity binding of amylin to this receptor complex (Christopoulos et al., 1999; Muff et al., 1999). Additionally, a further receptor component involved in CGRP-receptor and adrenomedullin-receptor activation is the CGRP-receptor component protein (Luebke et al., 1996). This small intracellular protein is thought to participate in mediating intracellular signaling in response to agonist binding, rather than acting as a chaperone or as part of the extracellular ligand-binding site (Evans et al., 2000; Prado et al., 2001).
Pharmacologic evidence for the presence of CGRP and adrenomedullin receptors in different cerebral and cranial arteries is convincing (Baskaya et al., 1995; Jansen et al., 1992; Kobayashi et al., 2000; Mori et al., 1997). Furthermore, molecular data support the presence of CGRP receptor family members in the cerebral vasculature, including RDC1, CRLR, and all three known RAMP messenger RNAs (mRNAs) (Edvinsson et al., 1997; Moreno et al., 1999; Oliver et al., 1998; Sams and Jansen-Olesen, 1998). Functionally, CGRP has been purported to play a significant role in diseases involving cerebral vasospasm (McCulloch et al., 1986; Juul et al., 1990), though such a strong interrelation with adrenomedullin has not been made. Furthermore, CGRP levels are increased in the circulation and saliva during migraine attacks (Gallai et al., 1995; Fischer et al., 1998; Goadsby and Edvinsson, 1993) and chronic tension-type headache (Ashina et al., 2000a; Fischer et al., 1998), and in migraine sufferers outside of attacks (Ashina et al., 2000b). Subsequent administration of 5-HT1B/1D agonists to migraine patients normalizes CGRP levels and relieves headache, suggesting that this peptide is intimately involved in the pathogenesis of migraine (Goadsby and Edvinsson, 1993; Goadsby et al., 1990) and cluster headache (Goadsby and Edvinsson, 1994; Fanciullacci et al., 1995). Neuroanatomically, CGRP is contained in many small unmyelinated nociceptive primary afferent neurons (C-fibers) and, unlike substance P, is also found in a proportion of myelinated nociceptive sensory afferents (Aδ-fibers) (Quartu et al., 1992; May and Goadsby, 1999). These perivascular afferent fibers may release CGRP and dilate cerebral blood vessels, resulting in the exacerbation of headache pain.
Immunohistochemical techniques have been widely used to investigate the neurovascular and trigeminovascular systems (Edvinsson et al., 1987a; 1998; Quartu et al., 1992; Sugimoto et al., 1997; Uddman et al., 1989). In the present study, we have used specific antisera to detect the presence of CGRP and adrenomedullin receptor component proteins in cerebral vessels. These findings may facilitate further classification and characterization of receptors in cerebral vasculature and lead to the discovery of small-molecule, receptor-subtype-selective compounds for the management of headache disorders.
MATERIALS AND METHODS
Human tissue samples
Middle meningeal, middle cerebral, and pial vessels were obtained postmortem as histopathologic samples from the University Hospital, Lund, Sweden and immersion-fixed in 2% paraformaldehyde (weight/volume) in 0.1-mol/L phosphate-buffered saline (PBS) and 15 mL saturated picric acid per 100 mL fixative. Specimens were collected in accordance with Swedish legislation (Transplantation, β1) and approved by the Ethics Committee (Lund). Human material collected in the U.K. was approved by the University of Cambridge and Addenbrookes Hospital Ethics Committees. This tissue was excess material remaining after the completion of all pathology or surgical procedures, and patients gave written consent for this material to be used. Briefly, tissues were immersion-fixed in 10% formal saline and embedded in paraffin wax (for immunohistochemistry) or frozen fresh in isopentane at −40°C (for radioligand binding). Tissue samples were taken from persons aged 37 to 79 years.
Antibodies
Peptide epitopes specific for CRLR, RAMP1, RAMP2, and RAMP3 were selected, synthesized, and conjugated to keyhole limpet hemocyanin and immunized into at least three rabbits per peptide. Optimal peptides to which polyclonal antisera were raised were GYSHDCPTEHLNGK (CRLR), GRTIRSYRELADC (RAMP1), CWNHYKDQMDPIEKD (RAMP2), and HRQFFSNCTVDRVHLEDPPDEY (RAMP3). Harvest bleeds were subsequently affinity purified using a Sepharose immunoaffinity column (Affiniti Research Products, Exeter, U.K.). To determine the magnitude of the immune reaction raised to the immunogen, antisera from each of the rabbits were characterized using enzyme-linked immunosorbent assay (Affiniti Research Products) and dot blot. After detailed assessment of each of the replicate rabbit antisera for each of the four peptides, a single optimal antiserum per receptor protein target was chosen and subsequently used for all immunohistochemical studies. Antisera were named OA-910 (anti-CRLR), OA-350 (anti-RAMP1), OA-450 (anti-RAMP2), and OA-550 (anti-RAMP3).
To confirm cellular localization of CRLR and RAMPs, an antiactin antibody was obtained commercially (Sigma, Poole, U.K.) to label vascular smooth muscle cells (tunica media). Furthermore, to determine presence and expression of receptor component immunoreactivity in endothelial cells (tunica intima), biotinylated Ulex europaeus agglutinin I (Vector Laboratories, Peterborough, U.K.) was used as a marker for this subpopulation of cells.
Dot blotting
To determine immune reactivity of the antisera, nitrocellulose membranes with focal spots of serially diluted immunogen were incubated in blocking solution (Western Breeze; Novex, San Diego, CA, U.S.A.) and subsequently in equivalent concentrations of preimmune or harvest bleed sera (OA-910, 1/2000; OA-350, 1/3000; OA-450 and OA-550, 1/5000), affinity-purified primary antisera (OA-910, 3.4 μg/mL; OA-350, 0.75 μg/mL; OA-450, 4.5 μg/mL; OA-550, 0.2 μg/mL) or blocking solution alone overnight at 4°C (Oliver et al., 2000; Wainwright et al., 2000). Detection was carried out using Western Breeze reagents (included secondary antibody-enzyme conjugate and chemiluminescent substrate) according to manufacturer's instructions (Novex) and opposed to Hyperfilm ECL (Amersham, Little Chalfont, U.K.). Further specificity of immune sera for immunizing peptides was ascertained by incubation of blots with affinity-purified primary antisera in the presence of 20-fold excess cognate peptide. To eliminate the possibility of nonspecificity of preabsorption, blots were incubated with affinity-purified antibodies in the presence of 20-fold excess unrelated peptide.
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
Tissue protein extraction for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) was carried out as previously described (Oliver et al., 2000; Wainwright et al., 2000). Briefly, cultures of SK-N-MC and Kelly cell lines previously shown to express CRLR and all RAMP mRNAs, respectively (McLatchie et al., 1998), were washed in PBS, scrape-harvested, pelleted, and homogenized in extraction buffer consisting of 40-mmol/L Tris (pH 8.0), 8-mol/L urea, and 65-mmol/L dithiothreitol using a Teflon pestle (Janke and Kunkel, Staufen, Germany). Homogenates were repeatedly centrifuged at 100,000 g to produce purified membrane protein preparations. Samples in NuPAGE sample buffer (Novex) were heated at 95°C for 5 minutes before being run on a 4% to 12% NuPAGE Bis-Tris gel (Novex) at a constant 125 V for 25 minutes using MOPS (3-[N-morpholino]propane sulfonic acid) running buffer (Novex). Using wet transfer apparatus (Novex), proteins were subsequently electrophoretically transferred onto nitrocellulose membranes. After blocking and overnight incubation at 4°C with affinity-purified primary antisera (OA910, 0.85 μg/mL; OA-350, 0.75 μg/mL; OA-450, 2.27 μg/mL; OA-550, 0.15 μg/mL) proteins were visualized as described previously using Western Breeze reagents (Novex).
Immunohistochemistry
Using previously described procedures (Oliver et al., 2000; Wainwright et al., 2000), immunostaining was performed on human vascular sections. Briefly, 6-μm thick paraffin sections were dewaxed in xylene, incubated in 0.3% hydrogen peroxide in methanol, and hydrated in a graded ethanol series. Before overnight incubation at 4°C in affinity-purified antisera, sections were washed in PBS and blocked in 5% normal goat serum in PBS-Triton X-100. After overnight incubation in primary antisera, sections were washed in PBS and immunoreactivity was detected using biotinylated goat antirabbit secondary antibody (1/200, Vector Laboratories) and ABC (Vector Laboratories), and visualized using diaminobenzidine and hydrogen peroxide. To increase intersection staining consistency, all secondary antibody and enzymatic immunodetection procedures subsequent to the overnight primary antibody step were performed using an Optimax robotic immunostainer (BioGenex, Finchampstead, U.K.).
Confocal microscopy
Human middle meningeal artery sections were immunostained for CRLR and RAMP1 using a fluorescent detection method. Briefly, slides were processed as previously detailed before incubation overnight with affinity-purified antisera (OA-910, 6.8 μg/mL; OA-350, 6.0 μg/mL). Primary antibodies were detected using biotinylated goat antirabbit secondary antibody (1/200, Vector Laboratories), followed by a tertiary layer of Texas Red Streptavidin (5.0 μg/mL, Vector Laboratories), and finally counterstained using TOTO-3 iodide (Molecular Probes, Leiden, Netherlands). Sections were mounted in Immu-Mount (Shandon Corp., Pittsburgh, PA, U.S.A.) and visualized using a Multi Band Confocal Imaging Spectrophotometer (Leica TCS SP, Wetzlar, Germany).
Radioligand autoradiography
Radioligand autoradiography was carried out as previously described (Oliver et al., 2001). Briefly, frozen vessels were sectioned at 10-μm onto Superfrost Plus slides (BDH, Poole, U.K.), and stored at −20°C. Slide-mounted tissue sections were allowed to reach room temperature and subsequently incubated for 30 minutes in preincubation buffer containing 50-mmol/L Tris-hydrochloride (pH 7.4), 5-mmol/L magnesium chloride, and 0.2% bovine serum albumin. Sections were then incubated with 100-pmol/L [125I]-CGRP (specific activity, 2000 Ci/mmol; Amersham) for 90 minutes in the same buffer with 0.5-mg/mL bacitracin. Levels of nonspecific binding were evaluated using incubation of radiolabeled CGRP concomitant with cold CGRP or CGRP8–37 added to final concentrations of 1 μmol/L in the incubation mixture. After incubation, slides were washed for 2 minutes in ice-cold washing buffer containing 50-mmol/L Tris-hydrochloride (pH 7.4), 5-mmol/L magnesium chloride, and 0.2% bovine serum albumin, rinsed four times in ice-cold 50-mmol/L Tris-hydrochloride (pH 7.4) and 5-mmol/L magnesium chloride, and finally in chilled distilled water. Sections were then rapidly dried under a stream of cold air and exposed to Kodak Biomax film (Sigma) for 16 to 36 hours.
RESULTS
Antibody characterization
Preimmune sera did not detect immobilized immunogenic peptides after dot blot analysis, suggesting that anti-CRLR and anti-RAMP antibodies were not innately present at significant levels in nonimmunized rabbits (Fig. 1A). In contrast, unpurified harvest and affinity-purified sera showed intense immunolabeling of their desired target epitopes. This was deemed specific because the signal was preabsorbable using cognate, but not unrelated, synthetic peptides (Fig. 1A).
Characterization of anti-CRLR (OA-910), RAMP1 (OA-350), RAMP2 (OA-450), and RAMP3 (OA-550) antisera using dot blot
Specificity of the antisera was further assured using SDS-PAGE immunoblot analysis. A major CRLR-immunoreactive band of 66 to 70 kd was detected in protein extracts from SK-N-MC cells, which express CGRP1 receptors and CRLR mRNA and protein (McLatchie et al., 1998; Evans et al., 2000; Prado et al., 2001) (Fig. 1B). This band likely represents mature glycosylated CRLR (McLatchie et al., 1998; Hilairet et al., 2001). The SDS-PAGE analysis of Kelly cell line extracts with antisera specific for RAMP1, RAMP2, or RAMP3 resulted in intense bands of approximately 17 kd, which is consistent with their expected monomeric mass and with previously published data (Hilairet et al., 2001).
Receptor localization
Immunohistochemistry and radioligand autoradiography. CRLR-, RAMP1-, RAMP2-, and RAMP3-like–immunoreactive material (ir) were present in human middle meningeal, middle cerebral, pial, and superficial temporal arteries. Specificity of staining was ascertained by comparing the intense immunoreactivity obtained with immune sera with the relative absence of reaction product obtained when using matched preimmune sera (Fig. 1C).
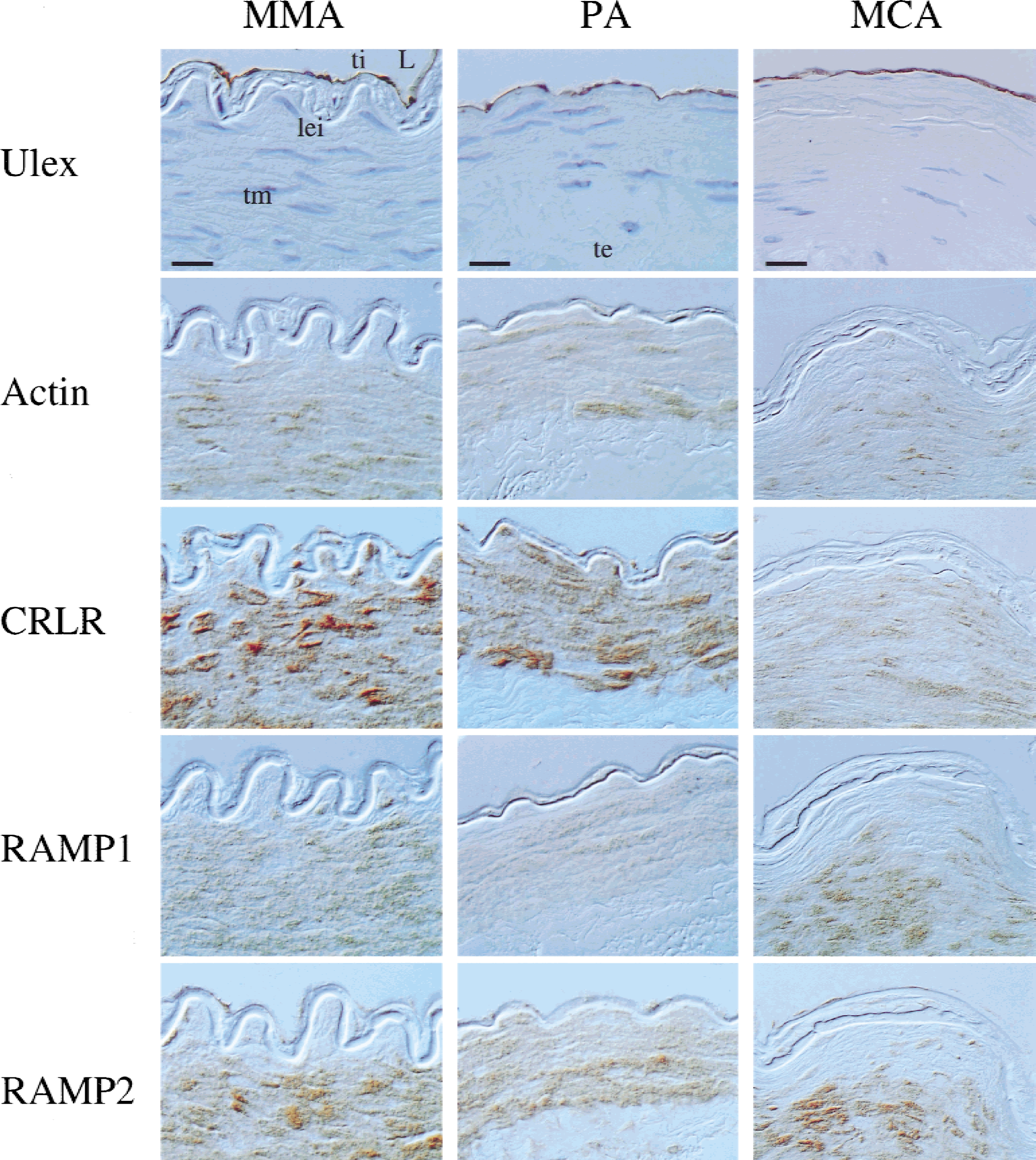
Abundant immunolabeling of the tunica media (smooth muscle layer) was observed using antisera specific for each of the receptor components (Figs. 1C, 2, and 3). This finding was complimentary to the observation that most abundant [125I]-CGRP binding sites were present on the tunica media, as detected by radioligand autoradiography (Fig. 4). Furthermore, the localization of CGRP receptor components to the smooth muscle layer was reaffirmed by staining of adjacent sections with an antibody specific for actin (Fig. 2).
Cellular expression of calcitonin gene-related peptide (CGRP)-receptor component proteins in human middle meningeal (MMA), pial (PA), and middle cerebral artery (MCA) sections. Each vessel possessed intact tunica intima (ti; endothelial cells) as shown using Ulex lectin staining (counterstained blue with hematoxylin). Actin-immunostaining was used as a marker for tunica media (tm; smooth muscle) in these preparations. CRLR- and RAMP2-like–immunoreactive material (ir) were abundant in tm and ti of all vessels shown, whereas RAMP1-ir and RAMP3-ir were particularly expressed by smooth muscle cells. L, lumen; lei, lamina elastica interna (elastin); te, tunica externa, loose fibrous connective tissue; CRLR, calcitonin receptor–like receptor; RAMP, receptor activity–modifying protein. Scale bars = 20 μm (upper left and upper middle panel [MMA and PA]; 30 μm (upper right panel [MCA]).
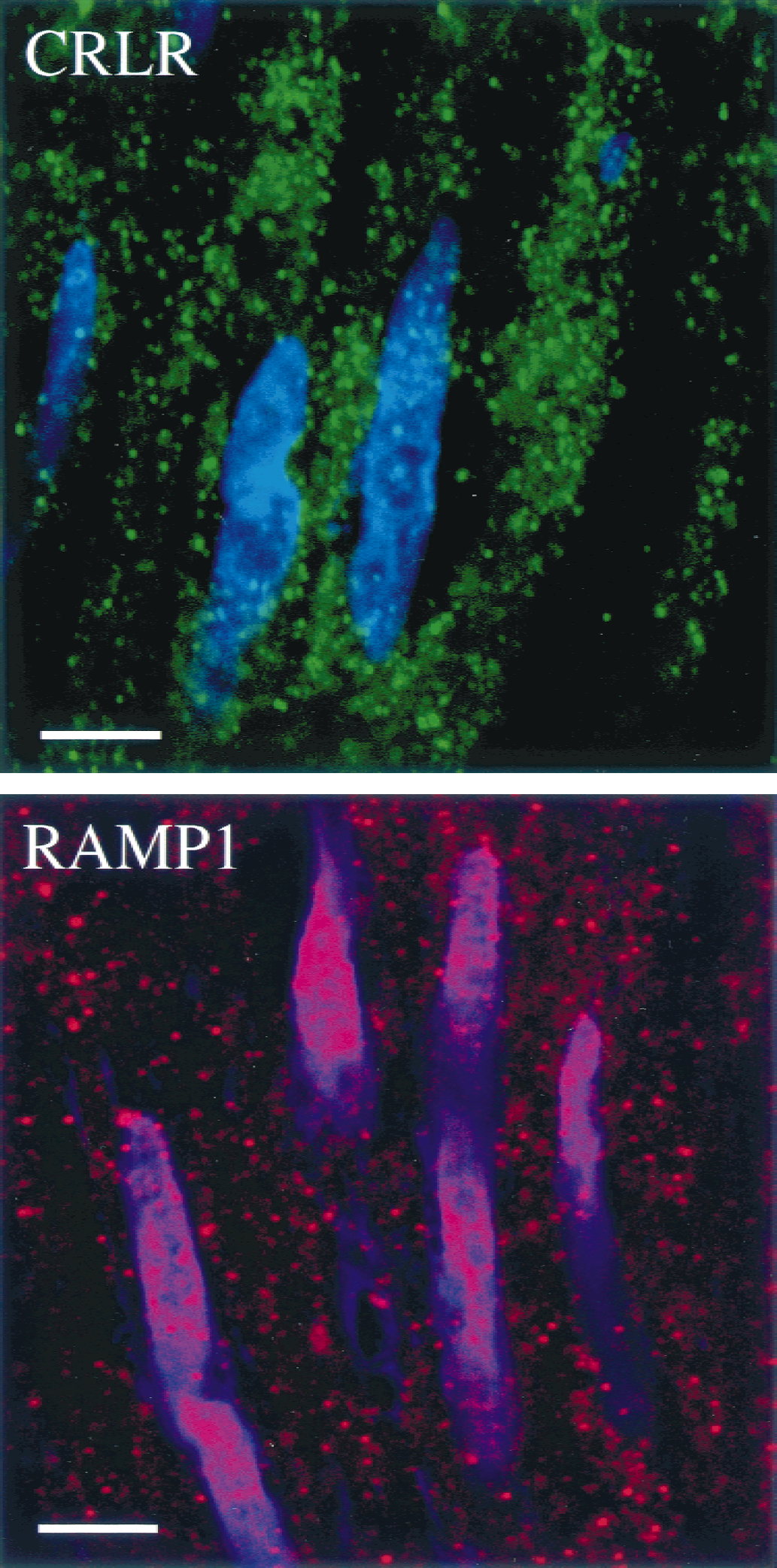
Confocal laser micrographs showing subcellular localization of CRLR-ir and RAMP1-ir in human middle meningeal artery tunica media. Staining of smooth muscle cells in both cases was punctate, typical of synaptic boutons (e.g., at the neuromuscular junction). Intracellular vesicular staining, characteristic of internalized receptor, may also represent a component of such focal staining. Blue nuclear counterstain was produced with TOTO-3 iodide. Scale bar = 5 μm.
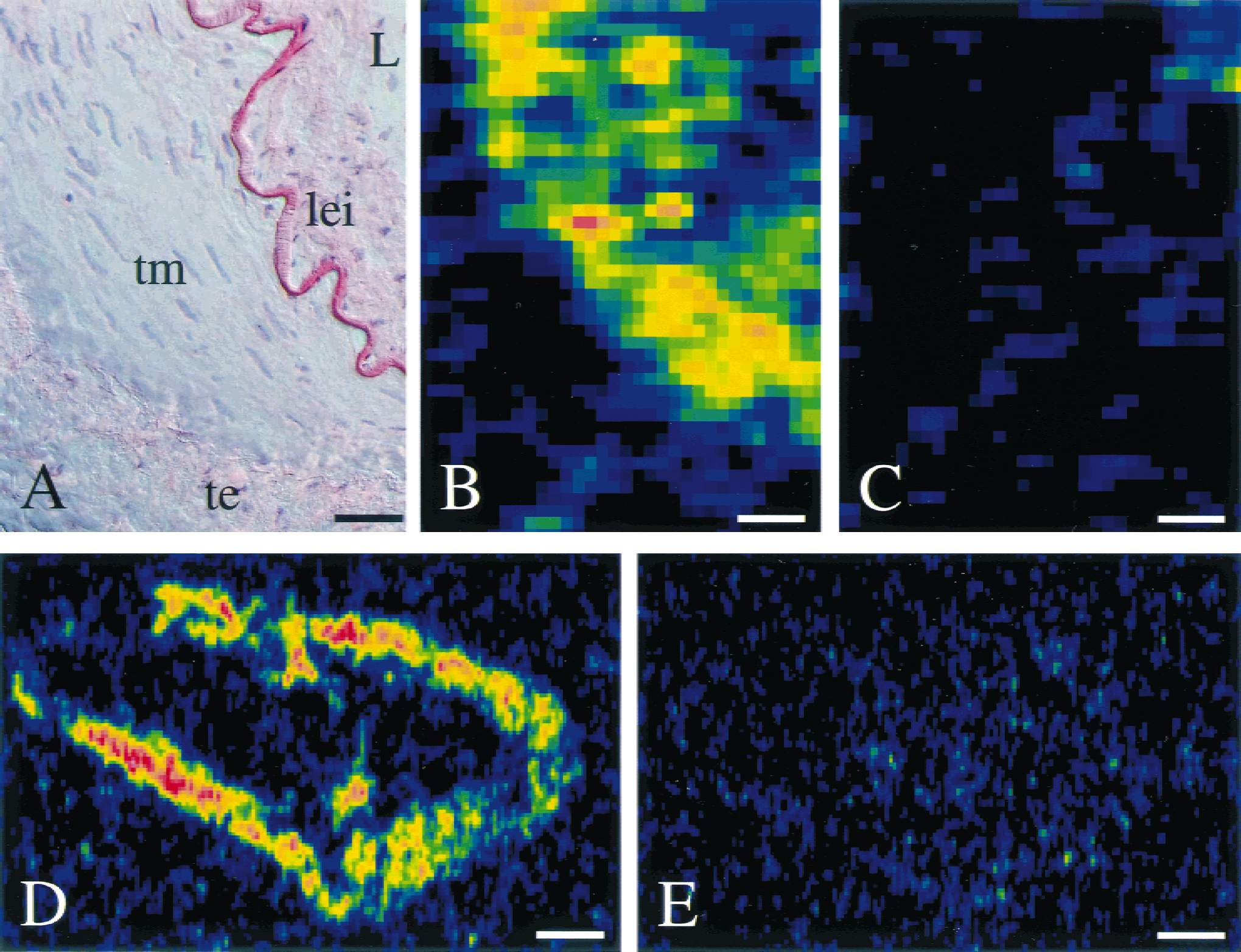
Radioligand autoradiography showing the presence of abundant [125I]-CGRP binding sites on human middle meningeal artery tunica media.
Vascular endothelial cells of the tunica intima also expressed CRLR-ir and RAMP-ir materials (Fig. 2). Widespread and abundant staining of the tunica intima was observed when using antisera specific for CRLR and RAMP2, in particular. Interestingly, immunohistochemistry for RAMP1 showed little, if any, detectable RAMP1-ir in most endothelial cells. The presence of endothelium in vessel samples used was affirmed using Ulex lectin, a marker of these cells (Fig. 2).
The sub-cellular nature of CRLR and RAMP1 immunolabeling was investigated with confocal microscopy. Staining for both components was punctate in character, suggesting focal accumulations of receptor proteins. This aggregation may occur both on the cell surface and in intracellular vesicles. The latter may arise during synthesis, assembly, and transport of CRLR and RAMP1 complexes to the plasma membrane or after agonist-induced sensitization and internalization (Kuwasako et al., 2000). Further studies using electron microscopy will be needed to investigate the precise nature of the punctate foci of CRLR-ir and RAMP1-ir in or on smooth muscle cells.
DISCUSSION
CGRP receptors have been classified pharmacologically using receptor subtype-selective analogs of this neuropeptide, such that CGRP8–37 blocks the type 1 (CGRP1) receptor and [Cys(ACM)2,7]-CGRP is an agonist of the type 2 (CGRP2) receptor, which is also insensitive to CGRP8–37 (Dennis et al., 1990). Until relatively recently, the molecular identities of CGRP1 and CGRP2 receptors were unknown, and this remains the case for the latter subtype. However, it is now accepted that CRLR combined with RAMP1 constitutes a CGRP1 receptor (McLatchie et al., 1998; Oliver et al., 2001). Human cerebral vessels are pharmacologically classified as possessing a CGRP1 pharmacology (Edvinsson et al., 1987b; Faraci and Breese, 1994; Goadsby et al., 1988; Kitazono et al., 1993; Kobari et al., 1995; Wahl et al., 1994). In concordance with this, recent studies have shown that these vessels express CRLR and RAMP1 mRNAs (Edvinsson et al., 1997; Sams and Jansen-Olesen, 1998). Additionally, cerebral vessels express mRNAs encoding RAMP2 and RAMP3 (Edvinsson et al., 2001; Sams and Jansen-Olesen, 1998), which generate adrenomedullin-binding heterooligomers on translation and coassembly (Hilairet et al., 2001; McLatchie et al., 1998; Oliver et al., 2001).
To further understand the molecular basis of CGRP and adrenomedullin receptor-mediated effects on the cerebrovascular system and to determine the cell types expressing receptor proteins for these neuropeptides, we raised and characterized antisera specific for CRLR and all known RAMP proteins. Specificity was confirmed using dot blotting, SDS-PAGE immunoblotting, and immunohistochemistry. Immunohistochemistry showed a general relative abundance of CRLR-ir and RAMP-ir in the tunica media (smooth muscle). CRLR-ir was also present on endothelial cells of the tunica intima. This latter finding is in agreement with previous data showing CRLR mRNA in primary cultures of endothelial cells (Moreno et al., 1999) and with the recent finding of “CGRPR-1′' immunoreactivity in the endothelium of human coronary and bronchial vessels (Hagner et al., 2001). However, a discrepancy between our findings and those of Hagner et al. is the abundant CRLR-ir that we observed in the smooth muscle layer, in which they could not detect receptor protein. The reasons for the discrepancy between these findings are unclear, but may be specifically related to differences in vessel type or histochemical procedures. The findings of the present study would fully corroborate molecular data showing first, that CRLR mRNA is expressed in human smooth muscle cells (Moreno et al., 1999); and second, that the abundance of CRLR reverse transcription polymerase chain reaction product was comparable in endothelium-denuded and endothelium-nondenuded cerebral arteries (Edvinsson et al., 1997). Furthermore, our data concur with functional data showing that CGRP is as potent a vasodilator of endothelium-denuded cerebral arteries as it is of nondenuded arteries (Edvinsson et al., 1985; 1998; Verrecchia et al., 1986). Interestingly, RAMP1 was largely absent or at below-detectable levels in most endothelial cells, further supporting the minor role of endothelial cells in mediating vasomotility in response to CGRP. Finally, our application of autoradiographic binding showed high levels of [125I]-CGRP binding to the tunica media, suggesting that CRLR (and RAMP1) is present on smooth muscle cells.
Therefore, CRLR-ir and RAMP1-ir were detectable on cells of the tunica media, and RAMP2-ir and RAMP3-ir were also expressed in tunica media. These data concur with the presence of adrenomedullin-binding sites on vascular smooth muscle cells (Eguchi et al., 1994; Iwasaki et al., 1998; Moreno et al., 1999), because both RAMP2 and RAMP3 coassemble with CRLR to form adrenomedullin-binding sites (McLatchie et al., 1998; Oliver et al., 2001). These data also corroborate functional data showing that adrenomedullin produces a concentration-dependent vasodilation of cerebral arteries in vivo and in vitro (Baskaya et al., 1995; Sams et al., 2000; Wang et al., 1995). Localization of abundant CRLR-ir and RAMP2-ir to vascular endothelial cells is a finding that is in agreement with the observed functional responses of this cell population to adrenomedullin (Kato et al., 1995).
CGRP-containing fibers are known to innervate cerebral vessels (Edvinsson et al., 1987b), and the stimulation of these fibers causes dilation. It has been hypothesized that the chain of events after stimulation of trigeminal sensory nerves, including neurogenic vasodilation, is perceived as painful. This and other mounting evidence implicate CGRP in the pathophysiology of headache disorders. For example, plasma CGRP levels are increased during chronic tension-type headache (Ashina et al., 2000a) and in migraine patients, both during and outside of attacks (Ashina et al., 2000b; Goadsby and Edvinsson, 1993; Gallai et al., 1995). Subsequent administration of triptan 5-HT1B/1D agonists normalizes CGRP levels and relieves migraine headache (Goadsby and Edvinsson, 1993; 1994; Goadsby et al., 1990). A proposed mechanism of this normalization may be that of 5-HT1D receptor-mediated inhibition of CGRP release from trigeminal neurons, as recently supported experimentally (Durham and Russo, 1999). However, such convincing evidence has not been presented for a pathophysiologic role of adrenomedullin in migraine. These data suggest that blockade of CGRP receptors (i.e., CRLR–RAMP1-containing oligomeric complexes) may facilitate normalization of vessel diameter, possibly resulting in amelioration of headache. This hypothesis will only be fully tested on the clinical evaluation of potent CGRP receptor antagonists, which have only recently become available (Doods et al., 2000).
In conclusion, we present for the first time immunohistochemical localization of RAMPs −1, −2, and −3, and their associated G-protein–coupled receptor, calcitonin receptor–like receptor, in human cerebral vessels. These data corroborate previous findings showing the presence of mRNAs that encode these proteins in the human vasculature. Furthermore, using radioligand autoradiography, we show the presence of [125I]-CGRP binding sites on cranial vessels, suggesting that receptors for CGRP include those formed by CRLR–RAMP1 heterooligomerization. These data allow receptors for the CGRP family to be classified on a molecular basis, rather than an exclusively pharmacologic basis. Furthermore, the proposed involvement of CGRP in migraine, and the presence of its target receptor in vessels implicated as the source of headache pain, may lead to the discovery of receptor subtype-selective compounds directed against CRLR–RAMP1 for the management of headache. Though beyond the scope of the current study, more quantitative future investigations into the expression of these receptor components in healthy and diseased tissues may provide further evidence to support the role of these receptor complexes in headache and other disorders.
Footnotes
Acknowledgments:
The authors would like to express their gratitude to those patients and next of kin who have allowed the present study to be undertaken through their provision of tissues, and hope that this work may eventually lead to the development of improved therapies for the treatment of patients with trigeminovascular pain. The authors also appreciate the expert assistance of L. Maskell (Cambridge).
