Abstract
Cerebral atrial natriuretic peptide (ANP), which is generated in the brain, has functions in the regulation of brain water and electrolyte balance, blood pressure and local cerebral blood flow, as well as in neuroendocrine functions. However, cerebral ANP clearance is still poorly understood. The purpose of this study was to clarify the mechanism of blood–brain (BBB) efflux transport of ANP in mouse. Western blot analysis showed expression of natriuretic peptide receptor (Npr)-A and Npr-C in mouse brain capillaries. The brain efflux index (BEI) method confirmed elimination of [125I]human ANP (hANP) from mouse brain across the BBB. Inhibition studies suggested the involvement of Npr-C
Keywords
Introduction
Natriuretic peptides in the central nervous system have been proposed to be involved in the central control of cardiovascular and neuroendocrine functions (Imura et al, 1992). Atrial natriuretic peptide (ANP), originally identified in the heart and peripheral tissues, has been detected in rodent brain and human brain by radioimmunoassay and immunohistochemistry (McKenzie et al, 1994; Morii et al, 1985). Accumulating evidence indicates that cerebral ANP has important functions in the regulation of brain water and electrolyte balance, blood pressure and local cerebral blood flow (Imura et al, 1992; Levin et al, 1998; Nakao et al, 1992), as well as in neuroendocrine functions (Itoh et al, 1986; Yamada et al, 1986). Therefore, changes in cerebral ANP levels may affect central nervous system homeostasis.
The cerebral level of ANP is thought to be controlled not only by production, but also by clearance, as peripheral clearance regulates the ANP level in circulating blood (Chiu et al, 1991). We have reported that the blood–brain barrier (BBB) possesses a brain-to-blood efflux transport system for neuromodulators and their metabolites as a part of the cerebral clearance system (Terasaki and Ohtsuki, 2005). As regards peptides, it was reported that a somatostatin analogue was bound to and internalized into isolated brain microvessels, and this internalization was proposed to be one mechanism of inactivation of neuropeptides (Pardridge et al, 1985). [125I]Human ANP (hANP) binding studies indicated the existence of an ANP binding receptor in brain capillaries (Chabrier et al, 1987; Steardo and Nathanson, 1987). These reports raised the possibility that the BBB possesses a receptor-mediated brain-to-blood efflux transport system for ANP, although there is as yet no report demonstrating the elimination of ANP across the BBB.
The ANP exerts its biological effects by binding to guanylyl-cyclase (GC)-linked natriuretic peptide receptor (Npr)-A and non-GC-linked Npr-C with similar affinity (Potter et al, 2006). The Npr-A is involved in signal transduction through elevation of intracellular cyclic guanosine monophosphate, whereas Npr-C acts as a clearance receptor to modulate extracellular ANP levels through internalization and degradation. Interestingly, [125I]hANP was cross-linked to a 60- to 70-kDa protein in bovine brain capillary endothelial cells (Whitson et al, 1991), and the size of Npr-C is almost the same as this (Fuller et al, 1988), whereas that of Npr-A is different (~135 kDa) (Oliver et al, 1997). Therefore, if the BBB possesses an efflux transport system for cerebral clearance of ANP, Npr-C seems to be a good candidate. However, it is still not known whether Npr-C protein is expressed in brain capillaries.
The purpose of this study was to identify the mediator of ANP clearance across the BBB by application of the brain efflux index (BEI) method (Kakee et al, 1996; Nishida et al, 2009) and to investigate the protein expression of Npr-A and Npr-C in brain capillaries by Western blot analysis. Further, the transport mechanism of ANP at the BBB was investigated by means of cellular uptake experiments, and the regulatory mechanism of Npr-C at the BBB was examined by highly selective quantification with liquid chromatography–tandem mass spectrometry (LC-MS/MS)-based targeted absolute proteomics (Kamiie et al, 2008) in conditionally immortalized mouse brain capillary endothelial cells, TM-BBB4 (Hosoya et al, 2000).
Materials and methods
Reagents
[125I]Human ANP (2000 Ci/mmol) radioiodinated with sodium [125I]iodide and lactoperoxidase was purchased from GE Healthcare (Buckinghamshire, UK) and Institute of Isotopes Co., Ltd (Budapest, Hungary). [3H]Dextran (100 mCi/mg) was obtained from American Radiolabeled Chemicals, Inc. (St Louis, MO, USA). Unlabeled hANP and human C-type natriuretic peptide (hCNP) were purchased from Peptide Institute (Osaka, Japan). cANP(4–23) was obtained from Phoenix Pharmaceuticals, Inc. (Burlingame, CA, USA). 1α,25-dihydroxyvitamin D3 (1,25(OH)2D3) was purchased from Biomol Research Laboratories (Plymouth Meeting, PA, USA). Xylazine hydrochloride was obtained from Sigma-Aldrich (St Louis, MO, USA). Ketaral 50 (ketamine hydrochloride) was purchased from Sankyo Co. (Tokyo, Japan). All peptides (purity > 95%) listed in Table 1 were synthesized by Thermoelectron Corporation (Sedanstrabe, Germany). All other chemicals were analytical grade commercial products.
Peptide probe sequences and selected ions for quantification of each protein with LC-MS/MS in the MRM mode
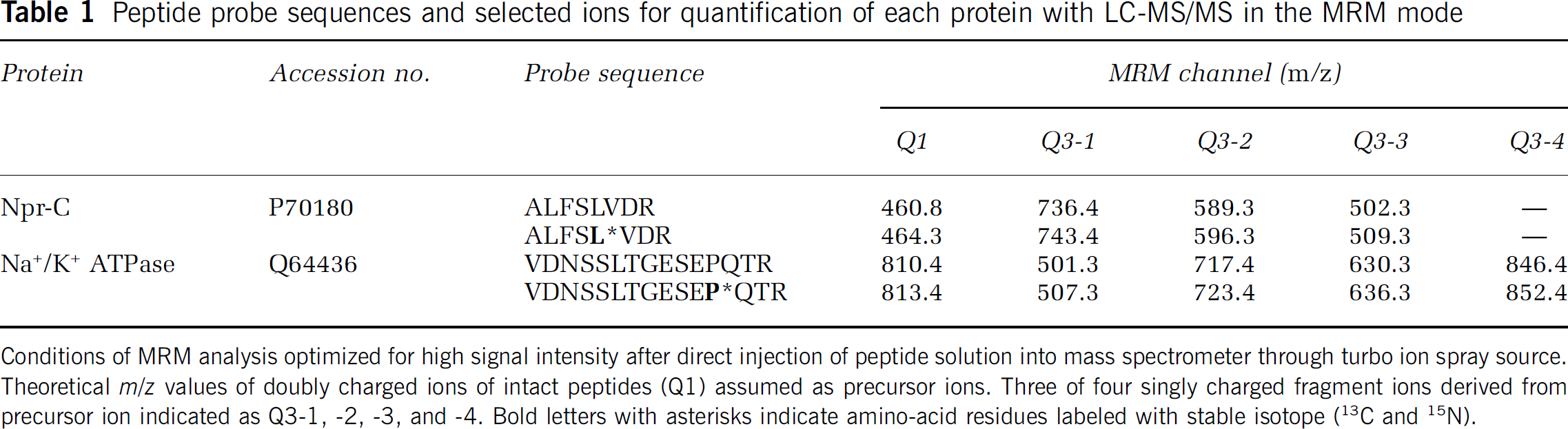
Conditions of MRM analysis optimized for high signal intensity after direct injection of peptide solution into mass spectrometer through turbo ion spray source. Theoretical
Animals
Male C57BL/6 mice (8 to 10 weeks old) were purchased from Japan SLC (Hamamatsu, Japan). All experiments were approved by the Animal Care Committee of the Graduate School of Pharmaceutical Sciences, Tohoku University.
TM-BBB4 Cell Culture
The TM-BBB4 cell line established from transgenic mice harboring the temperature-sensitive SV40 large T-antigen gene was used in this study (Hosoya et al, 2000). TM-BBB4 cells were cultured at 33°C in Dulbecco's modified Eagle's medium (DMEM; Nissui Pharmaceutical Co., Tokyo, Japan), supplemented with 20 mmol/L NaHCO3, 2 mmol/L
Western Blot Analysis
Under deep anesthesia induced with pentobarbital, mice were transcardially perfused with phosphate-buffered saline to remove blood, and then tissues were collected. The brain capillary fraction was separated from mouse cerebrum by the glass bead column method using the reported procedure (Ohtsuki et al, 2007). The crude membrane protein fraction of mouse kidney, brain capillary-rich fraction and TM-BBB4 cells were prepared using nitrogen cavitation. Briefly, the cells were washed with 1 mL phosphate-buffered saline twice and then scraped in 10 mmol/L Tris-HCl (pH 7.4) containing 250 mmol/L sucrose and protease-inhibitor cocktail (Sigma-Aldrich) and collected in a precooled cavitation chamber. The cell suspension was stirred under a pressure of 450 p.s.i. for 15 minutes. The crude membrane protein fraction of TM-BBB4 cells treated with or without 10 or 100 nmol/L 1,25(OH)2D3 was isolated using the reported procedure (Kamiie et al, 2008). TM-BBB4 cells were homogenized in buffer containing 10 mmol/L Tris-HCl (pH 7.4), 10 mmol/L NaCl, 1.5 mmol/L MgCl2, 1 mmol/L and a protease-inhibitor cocktail (Sigma-Aldrich). These homogenized samples were centrifuged at 10,000
In Vivo Brain Efflux Index Study
The


The apparent elimination rate constant (
To characterize the efflux transport system at the BBB, the percentage of [125I]hANP remaining in the brain at 30 minutes was determined in the presence or absence of preadministration of inhibitors. When the inhibitor concentration at the microinjection site could not be maintained high enough by coadministration, or to avoid possible physicochemical interactions, a sufficient volume (30 μL) of the inhibitor solution was preinjected 5 minutes before at the microinjection site of [125I]hANP to minimize the dilution effect. The percentage of [125I]hANP remaining in the brain at 30 minutes when 30 μL of ECF buffer was preinjected was not significantly different from that of the coadministered control (
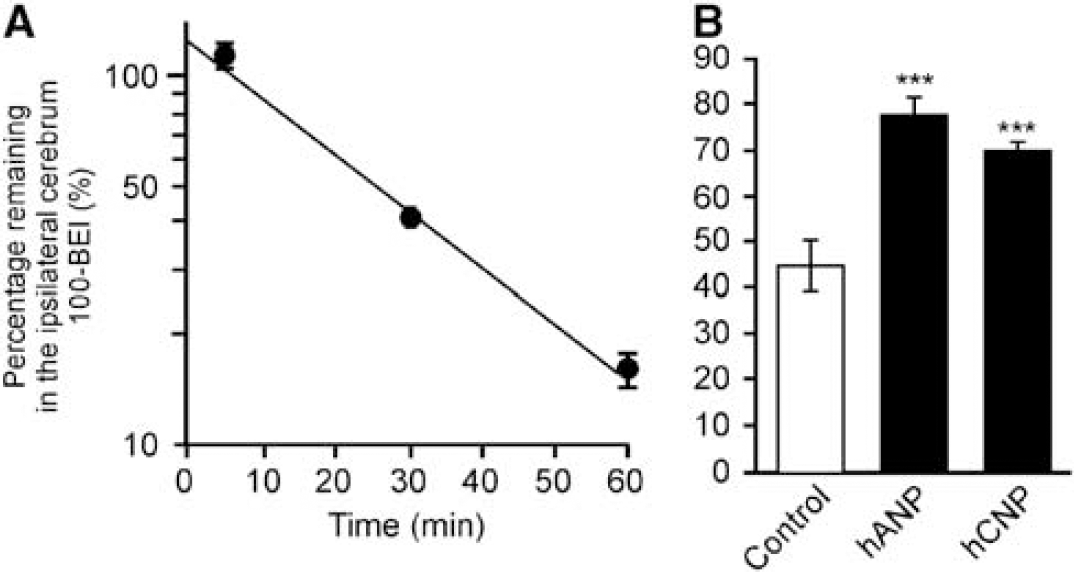
Elimination of [125I]hANP from mouse brain after microinjection. (
In Vitro Internalization Study in TM-BBB4 Cells
The internalization of [125I]hANP into TM-BBB4 cells was examined as described earlier (Ito et al, 2007). Briefly, TM-BBB4 cells were seeded on a collagen-coated 24-well plate at a density of 1.0 × 105 cells/well (Becton Dickinson, Bedford, MA, USA) and cultured for 48 hours. The extent of TM-BBB4 cell-[125I]hANP (0.015 μCi/200 μL in ECF buffer) binding was measured at 37°C. After the indicated times, the ECF buffer was removed, and the cells were washed three times with 1 mL ice-cold ECF buffer. The cells were then incubated with 1 mL ice-cold acetate-barbital buffer (28 mmol/L CH3COONa, 120 mmol/L NaCl, 20 mmol/L barbital sodium (pH 3.0) and 360 mOsm/kg) for 20 minutes at 4°C to remove [125I]hANP bound to the cell surface. After incubation, the buffer was recovered (fraction of acid-soluble binding) and the cells were washed three times with ice-cold acetate-barbital buffer.
Acid-resistant binding represents the amount of internalized [125I]hANP in the TM-BBB4 cells. The cells were solubilized with 200 μL of 5 mol/L NaOH overnight and neutralized with 200 μL of 5 mol/L HCl. [125I]Radioactivity was measured using a γ-counter (ART310, Aloka). The protein content of the cultured cells was measured using a DC protein assay kit (Bio-Rad) with bovine serum albumin as a standard. Total binding was obtained as the sum of acid-resistant binding and acid-soluble binding.
For kinetic studies, the Michaelis–Menten constant (
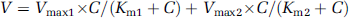
where
The half-saturation constant (
Transfection of Short Interfering RNA into TM-BBB4 Cells
Short interfering RNA (siRNA) knockdown of mNpr-C was accomplished using the following sequences: siRNA #1 sense-5'ACACGACUCUGAAGCUAAA-3′ and antisense-5'UUUAGC UUCAGAGUCGUGU-3′; siRNA #2 sense-5'GGU CAUUGGUGAUUACUUU-3′ and antisense-5'AAAGUAA UCACCAAUGACC-3′ and negative control siRNA (Ambion/Applied Biosystems, Foster City, CA, USA). Because of low transfection efficiency and toxicity of the transfection reagent to TM-BBB4 cells, we examined four different siRNAs and various transfection conditions, including time of incubation, siRNA concentration, medium volume, chemically modified siRNA, and lipofection reagents, to optimize the conditions of siRNA treatment of TM-BBB4 cells. Briefly, TM-BBB4 cells were seeded into collagen-coated 24-well plates at 0.75 × 105 cells/well, and grown for 24 hours in DMEM supplemented with 10% FBS. TM-BBB4 cells were transfected with 5 nmol/L mouse Npr-C siRNA-#1, #2 or negative control siRNA using Lipofectamine RNAiMAX (1 μ/200 μL/well; Invitrogen, Carlsbad, CA, USA) and OPTI-MEM I reduced serum medium (Invitrogen). Five hours after transfection, 200 μL DMEM supplemented with 20% FBS and 600 μL DMEM supplemented with 10% FBS were add to each well, and incubation was continued for 43 hours. The mRNA expression level and the transport activity were examined at 48 hours after the transfection.
Quantitative Real-Time Polymerase Chain Reaction Analysis
Total RNA was extracted from TM-BBB4 cells with an RNeasy kit (QIAGEN, Hilden, Germany) according to the manufacturer's protocol. RNA integrity was checked by electrophoresis on agarose gel. Single-stranded cDNA was prepared from 1 μg total RNA by reverse transcription (ReverTraAce, Toyobo, Osaka, Japan) using oligo (dT) primer. The sequences of the primers were as follows: sense primer 5′-TCT GCC TAC AAT TTC GAC GAG-3′ and antisense primer 5′-CAC AGA GAA GTC CCC ATA CCG-3′ for Npr-C (GenBank accession number; NM_008728); sense primer 5′-TTT GAG ACC TTC AAC ACC CC-3′ and antisense primer 5′-ATA GCT CTT CTC CAG GGA GG-3′ for β-actin (GenBank accession number; NM_031144). Quantitative real-time PCR analysis was performed using an ABI PRISM 7700 sequence detector system (Applied Biosystems) with 2 × SYBR Green PCR Master Mix (Applied Biosystems) according to the manufacturer's protocol. To quantify the amount of specific mRNA in the samples, a standard curve was generated for each run using pGEM-T Easy vector (Promega, Madison, WI, USA) containing Npr-C or β-actin (dilution ranging from 0.1 fg/μL to 1 ng/μL). This enabled standardization of the initial mRNA content of cells relative to the amount of β-actin.
Quantitation of Proteins Using LC-MS/MS System
Quantitation of crude membrane fraction of proteins in TM-BBB4 cells was performed using a nano-LC-MS/MS system as described earlier (Kamiie et al, 2008; Niessen et al, 2009). Tryptic peptides were chosen for synthesis based on in-silico selection criteria as described earlier (Kamiie et al, 2008). The amino-acid sequences and
The proteins of the crude membrane fraction were suspended in 100 mmol/L Tris-HCl (pH 8.5) containing 8 mol/L urea. The proteins were S-carbamoylmethylated as described (Mawuenyega et al, 2003). The alkylated proteins were fourfold diluted with 100 mmol/L Tris-HCl (pH 8.5) and digested with TPCK-treated trypsin (Promega) at an enzyme/substrate ratio of 1:100 at 37°C for 16 hours.
The tryptic digests were acidified with formic acid and 50 fmol of the stable isotope-labeled peptides were spiked into the digest to provide internal references. The mixtures were then analyzed with a nano-LC system (LC-assist, Tokyo, Japan) connected to an ESI-triple quadrupole mass spectrometer (4000Q trap, Applied Biosystems). Nano-LC was performed with a direct nano flow spray tip reversed-phase column (150-μm inner diameter × 50 mm) of Mightysil-C18 (3-μm particles; Kanto Chemicals, Tokyo, Japan) connected in tandem through an automated solvent desalting device and nano ion source (Nanospray, Applied Biosystems). Linear gradients of 1% to 35% acetonitrile in 0.1% formic acid were applied to elute the peptides at a flow rate of 100 nL/min for 35 minutes. Four products derived from single peptides are monitored in specific
Data Analysis
Unless otherwise indicated, all data represent the mean ± s.e.m. Unpaired two-tailed Student's
Results
Expression of Natriuretic Peptide Receptor-A and Natriuretic Peptide Receptor-C in Mouse Isolated Brain Capillaries and TM-BBB4 Cells
Protein expression of Npr-A and Npr-C in mouse isolated brain capillaries and TM-BBB4 cells, which are conditionally immortalized mouse brain capillary endothelial cells, was determined by Western blot analysis. As shown in Figure 1A, bands reacting with anti-Npr-A antibody were observed at ~130 kDa in mouse isolated brain capillaries, but not in TM-BBB4 cells. A band of the same size was detected in the kidney, which has been reported to express Npr-A and Npr-C (Fuller et al, 1988; Oliver et al, 1997). As shown in Figure 1B, bands of ~65 kDa were detected with anti-Npr-C antibody in mouse isolated brain capillaries and TM-BBB4 cells, as well as in the kidney, and the band intensity in TM-BBB4 cells was much greater than that in isolated brain capillaries. These results indicate that Npr-A and Npr-C are expressed in mouse brain capillary endothelial cells, and Npr-C is highly expressed in TM-BBB4 cells.
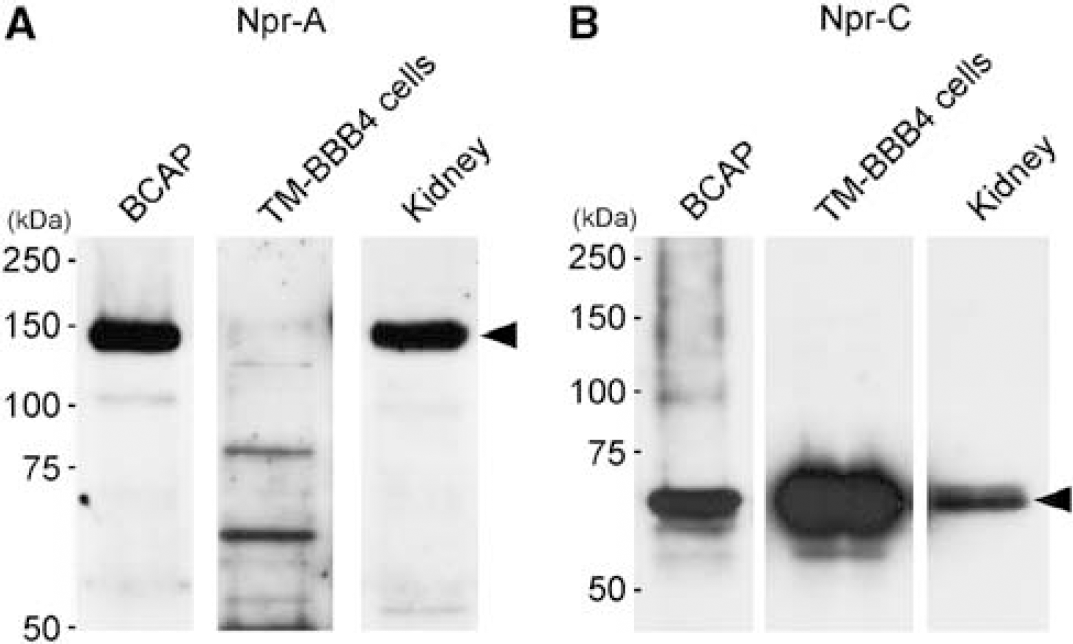
Expression of natriuretic peptide receptor (Npr)-A and Npr-C proteins in mouse brain capillaries, TM-BBB4 cells, and kidney. The crude membrane fractions (1 to 4.7 μg) were subjected to Western blot analysis. The bands at 130 kDa (
[125I]Human Atrial Natriuretic Peptide Elimination from the Brain After Intracerebral Microinjection
The
To characterize the hANP elimination process at the BBB
Characteristics of [125I]Human Atrial Natriuretic Peptide Internalization into TM-BBB4 Cells
To characterize ANP transport at the BBB, TM-BBB4 cells were used as an
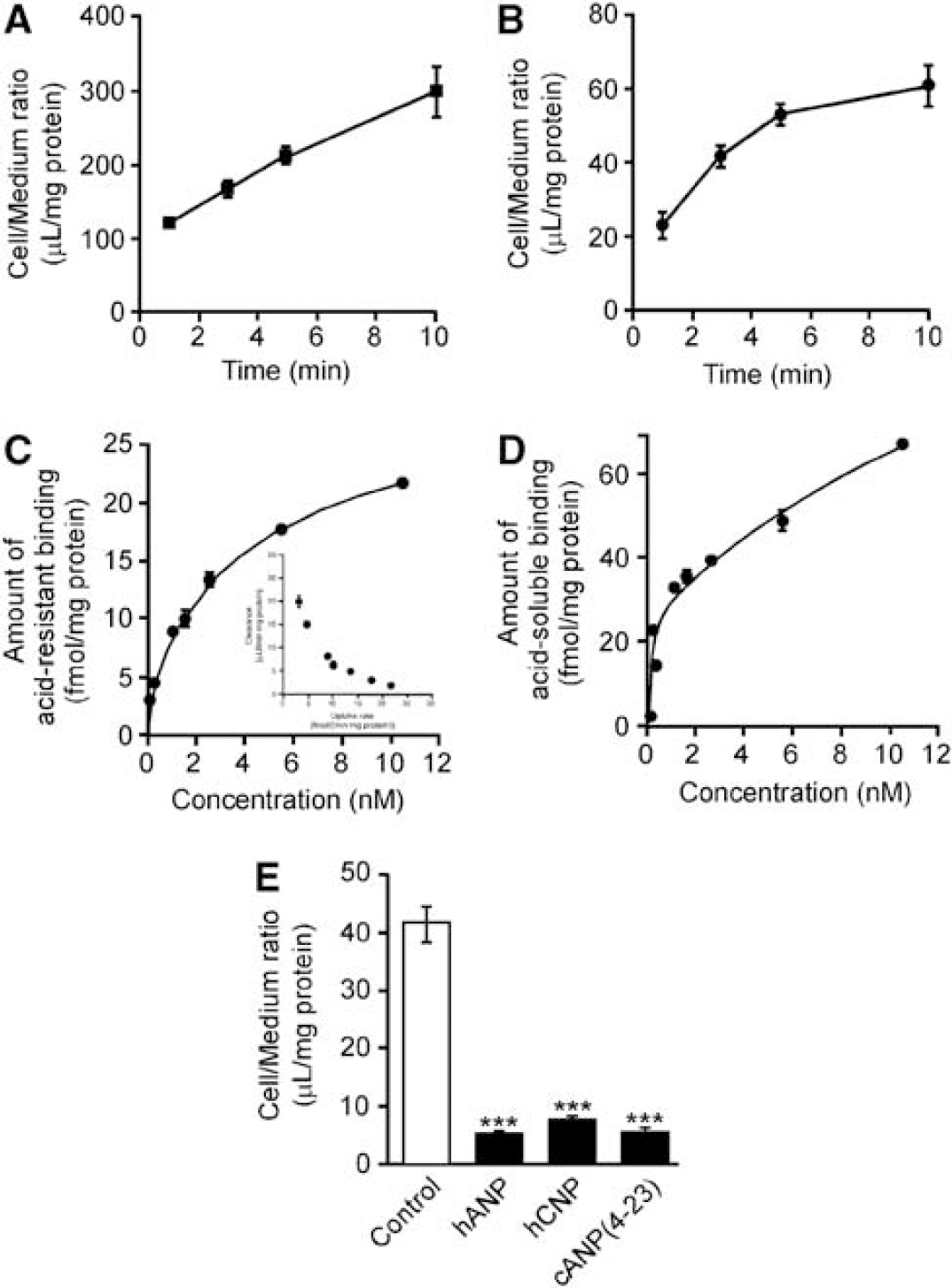
Internalization of [125I]hANP in TM-BBB4 cells. (
The acid-soluble binding reflects the amount of surface binding of [125I]hANP not internalized into the cells. The acid-soluble binding showed similar concentration dependency to the acid-resistant binding (Figures 3C and 3D). Analysis of the acid-soluble binding based on two saturable components indicated an
To investigate the substrate selectivity of the hANP internalization system in TM-BBB4 cells, the inhibitory effects of several natriuretic peptides on [125I]hANP uptake by TM-BBB4 cells were examined (Figure 3D). Unlabeled hANP and hCNP inhibited [125I]hANP internalization by 86.2% and 80.8%, respectively. cANP(4–23), a selective ligand for Npr-C, also reduced [125I]hANP internalization by 85.7%. This result suggests that Npr-C is involved in [125I]hANP internalization into TM-BBB4 cells.
Effect of Natriuretic Peptide Receptor-C Knockdown on [125I]Human Atrial Natriuretic Peptide Internalization into TM-BBB4 Cells
To clarify the involvement of Npr-C in hANP internalization into TM-BBB4 cells, we examined the effect of siRNA knockdown of Npr-C. The Npr-C mRNA expression and [125I]hANP internalization were determined 48 hours after siRNA treatment. The expression of Npr-C mRNA in TM-BBB4 cells was significantly decreased by Npr-C siRNA treatment by 53.2% (siRNA-1) and 28.1% (siRNA-2) compared with that in the case of negative control siRNA treatment (Figure 4A). The [125I]hANP internalization into TM-BBB4 cells was also reduced by Npr-C siRNA treatment, by 22.8% (siRNA-1) and 12.2% (siRNA-2) compared with that in the case of negative control siRNA treatment (Figure 4B).
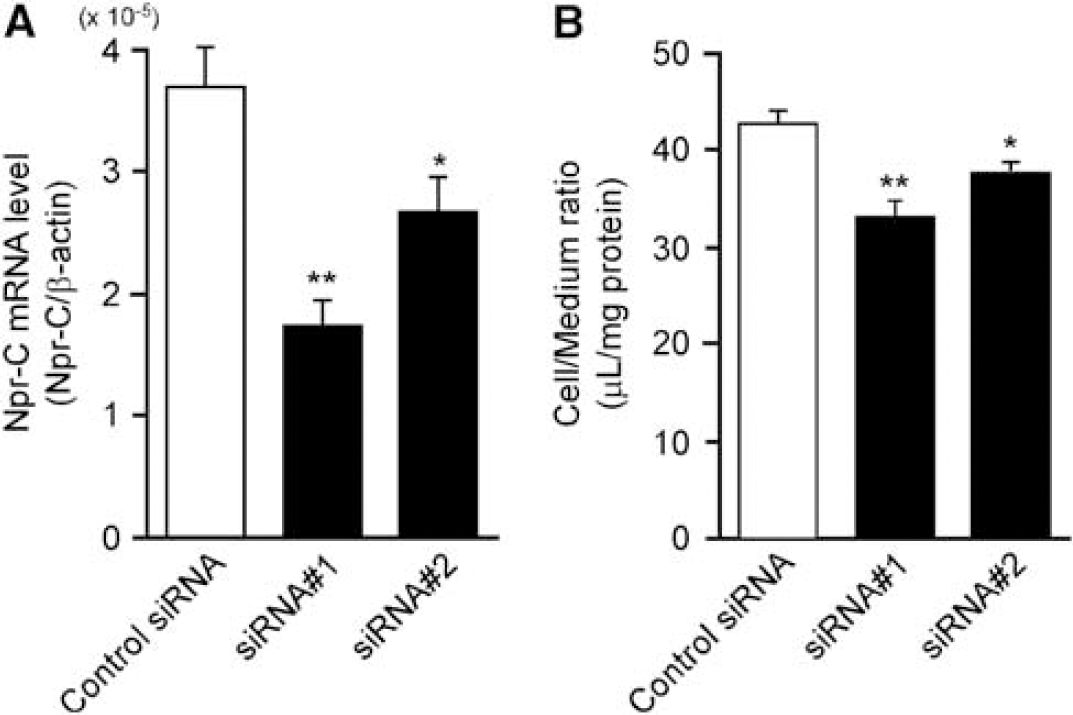
Effect of natriuretic peptide receptor (Npr)-C short interfering RNAs (siRNAs) on Npr-C mRNA expression and the acid-resistant [125I]hANP binding to TM-BBB4 cells. (
Effect of 1α,25-Dihydroxyvitamin D3 on the Expression Level of Natriuretic Peptide Receptor-C in TM-BBB4 Cells
Protein expression of Npr-C is upregulated by 1,25(OH)2D3, a key regulator of mineral metabolism, in osteoblasts (Yanaka et al, 1998). To investigate the regulation mechanism of Npr-C in brain capillary endothelial cells, we examined the effect of 1,25(OH)2D3 treatment on protein expression of Npr-C in TM-BBB4 cells. After treatment of TM-BBB4 cells with 10 and 100 nmol/L 1,25(OH)2D3 for 24 hours, an increase of Npr-C expression was observed by Western blot analysis (Figure 5A). To quantify Npr-C protein expression, multiplexed multiple reaction monitoring (MRM) analysis was performed with nano-LC/MS/MS, which measures the absolute amount of a tryptic peptide specific for the target molecule in trypsin-digested protein samples. The expression level of Npr-C protein in TM-BBB4 cells was increased to 1.23 ± 0.06 and 1.55 ± 0.18 fmol/μg protein after treatment of 10 and 100 nmol/L 1,25(OH)2D3, respectively, compared with that in the control (0.57 ± 0.05 fmol/μg protein) (Figure 5B). The protein expression of Na+/K+ ATPase was not significantly affected by 1,25(OH)2D3.
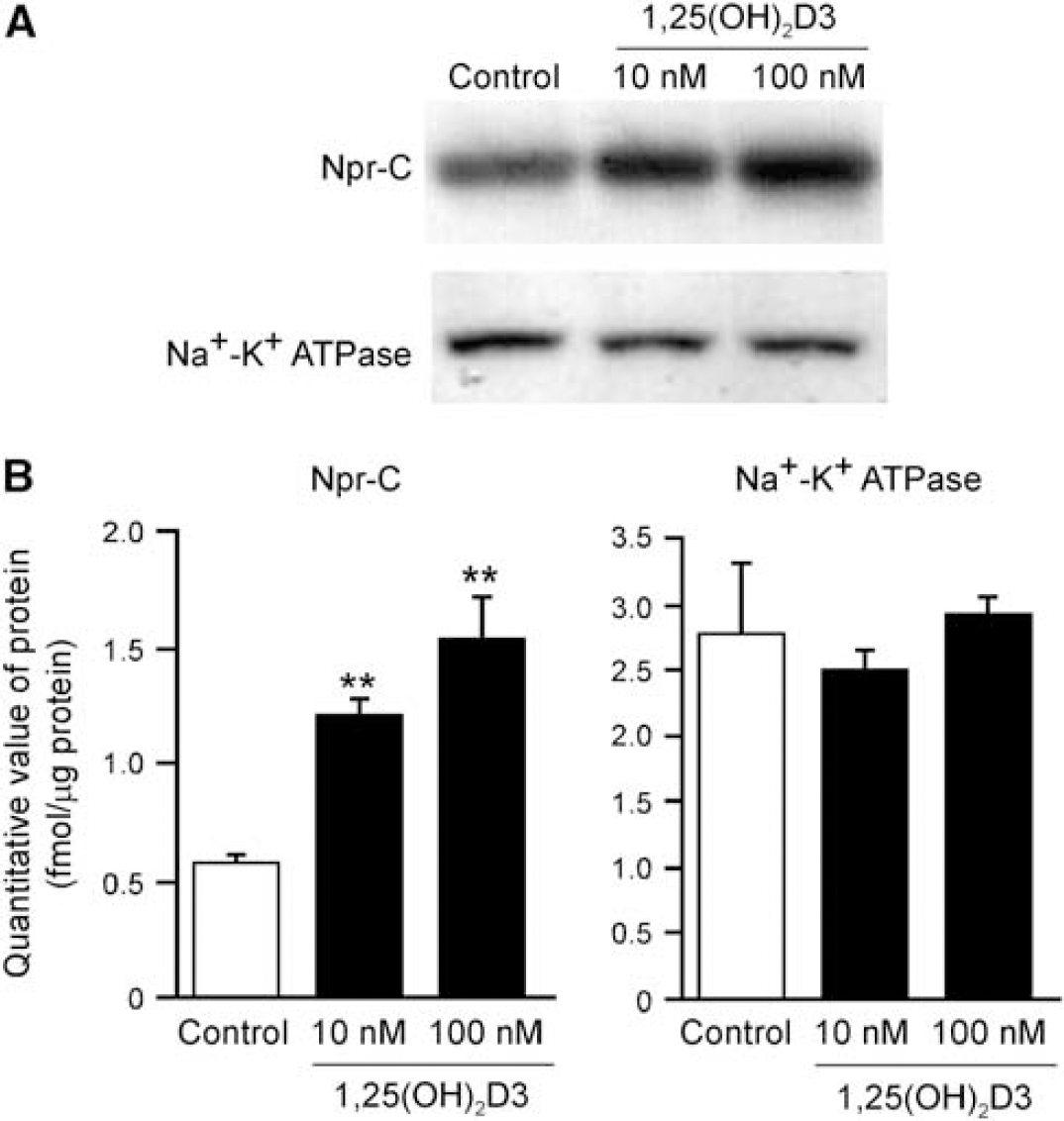
Effect of 1α,25-dihydroxyvitamin D3(1,25(OH)2D3) on the expression of natriuretic peptide receptor (Npr)-C in TM-BBB4 cells. (
Discussion
This study has provided evidence that ANP is eliminated from the brain across the BBB after intracerebral administration of [125I]hANP into mouse cerebral cortex
It has been reported that ANP primarily binds Npr-A and Npr-C, whereas CNP preferentially binds Npr-B and Npr-C (Potter et al, 2006). The Npr-C is known to bind ANP, Npr-B and CNP with similar affinity (He et al, 2001). The [125I]hANP elimination process was significantly inhibited by preadministration of unlabeled hANP and hCNP (Figure 2B). Western blot analysis using anti-Npr-A and anti-Npr-C antibodies showed that Npr-A and Npr-C proteins were expressed in mouse isolated brain capillaries, as well as kidney (Figure 1). These results suggest the involvement of Npr-A and/or Npr-C in the clearance of ANP from mouse brain across the BBB.
Cells of the conditionally immortalized mouse brain capillary endothelial cell line TM-BBB4 expressed Npr-C protein and exhibited internalization of [125I]hANP (Figures 1 and 3). As internalization of [125I]hANP into TM-BBB4 cells was inhibited by hANP and hCNP, in accordance with the
This study in mice was performed with hANP. The amino-acid sequence of hANP differs by only one amino acid from that of mouse; methionine at position 12 in hANP is isoleucine in mouse ANP. We confirmed that the acid-resistant binding of [125I]hANP in HEK293 cells transfected with mouse Npr-C isolated from TM-BBB4 cells was significantly greater than that in mock HEK293 cells; the values were 20.1 ± 0.5 μL/mg protein and 4.77 ± 0.15 μL/mg protein, respectively, at 30 minutes (mean ± s.e.m.,
In contrast to Npr-C, the expression of Npr-A was suppressed in TM-BBB4 cells. Therefore, involvement of not only Npr-C, but also Npr-A, in the efflux transport of [125I]hANP at the BBB
Cerebral ANP has been reported to participate in the regulation of brain volume (Doczi et al, 1987; Vajda et al, 2001). Intracerebral injection of ANP decreased brain sodium levels and attenuated brain edema in rat models of cerebral ischemia (Doczi et al, 1987; Nakao et al, 1990; Naruse et al, 1991). Therefore, the regulation of ANP efflux transport at the BBB through Npr-C is expected to be important to regulate brain water content and brain volume. The protein expression of Npr-C was reported to be upregulated by 1,25(OH)2D3 in osteoblasts (Yanaka et al, 1998), and our results show that protein expression of Npr-C was also induced by 1,25(OH)2D3 in TM-BBB4 cells (Figure 5), suggesting that ANP efflux transport at the BBB through Npr-C is regulated by 1,25(OH)2D3. Spontaneously hypertensive rats, an animal model of essential hypertension, were resistant to stimulation with 1,25(OH)2D3 (Toraason and Wright, 1981) and the number of [125I]ANP binding sites in the brain capillaries was significantly lower than that in the case of Wistar-Kyoto rat brain capillaries (Ibaragi and Niwa, 1989; Okazaki et al, 1990). This could be explained by regulation of Npr-C expression of brain capillaries by 1,25(OH)2D3. The effect of 1,25(OH)2D3 on the efflux transport of ANP and cerebral ANP level should be examined in further studies.
Our results show the involvement of Npr-C in the brain-to-blood efflux transport of cerebral ANP at the BBB. However, it is also suggested that other molecules are involved in the BBB efflux transport of ANP, as Eadie–Hofstee plots indicates that internalization of [125I]hANP into TM-BBB4 cells involved both high- and low-affinity internalization processes with
The present results imply that lower levels of serum 25(OH)D3, a major circulating form of vitamin D, attenuate cerebral clearance of ANP by impairing Npr-C induction at the BBB, leading to elevation of ANP level in the brain. Although the brain level of ANP is unknown, an elevated level of ANP might suppress the insulin-degrading enzyme-mediated Aβ degradation process, as ANP has 200-fold higher affinity for insulin-degrading enzyme than does Aβ (60 nmol/L versus 1.2 μmol/L; Muller et al, 1991; Perez et al, 2000). Epidemiological study indicated that a low level of serum 25(OH)D3 may result in an increased risk of Alzheimer's disease (Oudshoorn et al, 2008). Therefore, it can be hypothesized that reduced levels of serum 25(OH)D3 have a part in increasing Aβ level in the brain as a result of inhibition of Aβ degradation by attenuation of cerebral clearance of ANP.
In conclusion, our findings indicate that Npr-C is expressed at brain capillary endothelial cells and mediates cerebral clearance of hANP from the brain, at least in part. Therefore, Npr-C may be involved in physiological control of the ANP level in the brain, and thus in brain fluid homeostasis.
Footnotes
The authors declare no conflict of interest.
