Abstract
Region-specific cerebral blood flow (CBF) and the apparent diffusion coefficient (ADC) of tissue water in the rat brain were quantified by high-field magnetic resonance imaging at 9.4 T in the rat suture occlusion model. Cerebral blood flow and ADC were compared during the short- (4.5 hours) and long-term (up to 6 days) reperfusion after 80 minutes of transient middle cerebral artery occlusion, and correlated with the histology analysis. On occlusion, average CBF fell from ∼100 to less than 50 mL 100 g−1 min−1 in the cortex, and to less than 20 mL 100 g−1 min−1 in the caudate putamen (CP). Corresponding ADC values decreased from (6.98 ± 0.82) × 10−4 to (5.49 ± 0.54) × 10−4 mm2/s in the cortex, and from (7.16 ± 0.58) × 10−4 to (4.86 ± 0.62) × 10−4 mm2/s in the CP. On average, CBF recovered to ∼50% of baseline in the first 24 hours of reperfusion. After 2 to 4 days, a strong hyperperfusion in the ipsilateral cortex and CP, up to ∼300 mL 100 g−1 min−1, was observed. The ADC ratio in the ipsilateral and contralateral CP was also inverted in the late reperfusion period. Histology revealed more severe tissue damage at the late stage of reperfusion than at 4.5 hours. Significant reversal of CBF and ADC during the late reperfusion period may reflect the impairment of autoregulation in the ischemic regions. Vascular factors may play an important role in the infarct development after 80-minute focal ischemia.
Keywords
Hemodynamic changes during reperfusion after cerebral ischemia are critical for the long-term outcome and have received much attention in the development of treatment strategies. Changes in perfusion usually precede other changes detectable by magnetic resonance imaging (MRI), such as those in apparent diffusion coefficient (ADC) of water and transverse relaxation time, T2. Early transient postischemic hyperperfusion followed by a period of delayed hypoperfusion is believed to result in reperfusion injuries, with microvascular damage as one of the primary contributing factors (Traupe et al., 1982; Todd et al., 1986; Marchal et al., 1999). Recent studies indicated that possible mechanisms of reperfusion injury include secondary hemodynamic disturbances (Carden and Granger, 2000), the enhancement of inflammatory processes (Karibe et al., 1994), free-radical formation (del Zoppo, 1994; Jiang et al., 1995), disturbance of the mitochondrial function, and vasogenic edema (Chan, 1996) secondary to the breakdown of the blood–brain barrier (Yang and Betz, 1994). Very few studies, however, reported observation of increased cerebral hemodynamics in experimental ischemia after more than 24 hours of reperfusion (Dijkhuizen et al., 1998; van Lookeren Campagne et al., 1999).
Diffusion-weighted imaging can detect early tissue damage during focal cerebral ischemia (Moseley et al., 1990) and reperfusion. In the transient model of middle cerebral artery occlusion (MCAO), full recovery of ADC to preocclusion levels usually occurs after 30 to 60 minutes of reperfusion if occlusion time is less than 30 minutes. ADC recovery may be followed by a secondary decline in ADC values, leading eventually to selective neuronal injury (Miyabe et al., 1996; Li et al., 1999; van Lookeren Campagne et al., 1999; Li et al., 2000; Neumann-Haefelin et al., 2000; Olah et al., 2001). When occlusion times exceed 90 minutes, however, no recovery in ADC is expected until 2 to 7 days later (Jiang et al., 1993; Li et al., 1999; Kettunen et al., 2000; Neumann-Haefelin et al., 2000).
The development of secondary microvascular dysfunction and dynamics of ADC changes in the rat brain during and after ischemia of intermediate duration, and their relation to the ischemic lesion development have received relatively little attention in experimental ischemia studies. The aim of the present study was to characterize the cerebral blood flow (CBF) and ADC changes noninvasively in the late reperfusion period after 80 minutes of focal cerebral ischemia using MRI in conjunction with the intraluminal suture withdrawal method.
MATERIALS AND METHODS
The experimental protocol was approved by our Institutional Animal Care and Use Committee. Twenty male Sprague-Dawley rats (190 ± 25 g, Harlan Sprague-Dawley, Indianapolis, IN, U.S.A.) were used. Animals were randomized into sham, short-term, and long-term groups. Except for the sham group, all animals were subjected to 81 ± 5 minutes of occlusion. In the short-term group (five rats), animals were monitored by MRI for 4.5 hours of reperfusion. In the long-term group (seven rats), the reperfusion was extended to up to 6 days. Two of the seven rats in the long-term group received the same MRI examinations during the first 4.5 hours as in the short-term group, so that MRI data can be pooled for analysis. In the sham group (three rats), animals were surgically prepared as described in Surgical Procedures, but were not subjected to ischemia. The remaining five rats showed inadequate occlusion, as judged by the absence of any ADC decrease in the brain, and were excluded from the aforementioned three groups and processed separately.
Surgical procedures
Animals were prepared as described earlier (Liachenko et al., 1998). In brief, rats were intratracheally intubated and mechanically ventilated to maintain normal blood gases during and after MCAO. Anesthesia was induced with 4% isoflurane and maintained with 1.00% to 1.75% isoflurane in a 1:1 mixture of O2 and N2O during surgery and MRI. Paralysis was provided by pancuronium bromide [2 mg kg−1 h−1 (Todd et al., 1986)]. Both femoral arteries were cannulated for monitoring arterial blood pressure and gases. The rat body temperature was maintained at 37.0 ± 0.3°C with a feedback-controlled air-heating blanket. Published procedures for MCAO in rats were followed (Longa et al., 1989). A 3–0 or 4–0 surgical nylon suture (Ethicon, Inc., Somerville, NJ, U.S.A.), with its tip rounded to a diameter of 0.22 to 0.25 mm, was introduced into the external carotid artery lumen through a small puncture. The suture was gently advanced from the external carotid artery into the lumen of the internal carotid artery until it was 16 to 18 mm distal from the carotid bifurcation (depending on the weight of the animals), leading to occlusion of the origin of the middle cerebral artery (MCA). The sham-operated rats underwent the same surgery but did not have the suture insertion or the external carotid artery manipulation. Immediately after occlusion, the incision was closed, and the rats were positioned in a specially designed cradle inside the imaging probe for MRI examination. After a predetermined ischemic period, the animals were quickly removed from the magnet for suture withdrawal and then repositioned in the magnet for continuing MRI evaluation of reperfusion.
Magnetic resonance imaging
MRI experiments were carried out using a Chemagnetics CMX-400SLI spectrometer (Varian Inc., Fort Collins, CO, U.S.A.) equipped with a 9.4 T vertical magnet and a homemade birdcage probe, as described elsewhere (Xu and Tang, 1997). The single imaging plane containing caudate putamen was chosen between brain levels 19 and 26 of the Swanson rat brain atlas (Swanson, 1998). CBF measurements in the selected plane were made using the arterial spin labeling technique (Williams et al., 1992). A two-dimensional Fourier transform spin-echo pulse sequence was preceded by an adiabatic arterial spin-tagging pulse, which was 0.8 seconds in duration and 14 μT in strength and was applied 14 mm away from the imaging plane at the neck level in the presence of a 12-mT/m magnetic field gradient. The relatively low tagging power, which still satisfies the adiabatic condition, was chosen to minimize the power deposition and local tissue heating. A 50-millisecond delay period was inserted between the end of the tagging and the beginning of the image acquisition. The control images were acquired with adiabatic pulses applied on the opposite side relative to the imaging plane, adjusted so as to compensate the magnetization transfer effects. Sine-shaped crusher gradients with amplitude of 180 mT/m and duration of 5 milliseconds were applied in three orthogonal directions at the end of each acquisition. The images were acquired in 64 × 128 complex points and averaged for 2 scans, with a field of view of 50 × 50 mm2, a slice thickness of 2 mm, and a spin-echo time and pulse repetition time of 14 milliseconds and 1 second, respectively. The total acquisition time per image was 2.5 minutes. The control and tagged images were interleaved in a train of six, and perfusion maps were reconstructed pixel-by-pixel from each adjacent pair according to the standard equation (Williams et al., 1992). The values of the apparent spin-lattice relaxation time, T1, for the cortex and caudate putamen, as measured earlier (Liachenko et al., 2001), were 1.95 and 1.97 seconds, respectively, in the normal brain, and 2.13 and 2.16 seconds, respectively, in the ischemic tissue. The difference is relatively minor and in good agreement with published data at comparable magnetic fields (Calamante et al., 1999a; Kettunen et al., 2000). The degree of arterial water spin inversion (α) (Zhang et al., 1993) under our experimental conditions was measured to be 0.46 for rats subjected to brain ischemia, and 0.48 for normal or sham-operated rats. These measured a values are in agreement with the expected values (Zhang et al., 1993) for the radiofrequency power level of 14-μT used in the experiments.
The ADC in the brain was measured using a two-dimensional Fourier transform spin-echo sequence (pulse repetition time = 1.3 seconds, spin-echo time = 25 milliseconds, 2 acquisitions, 64 × 128 complex points) modified to include a pair of 5-millisecond diffusion-weighting gradient pulses, one on each side of the refocusing 180° pulse and applied along the readout direction. The diffusion-weighting gradient was incremented to obtain five images with gradient strengths ranging from 0 to 182 mT/m, corresponding to additional diffusion weighting factor (b) values of 0 to 880 s/mm2. The ADC of water maps were calculated pixel-by-pixel from sets of five diffusion-weighted images by fitting the image intensities as a function of the b value to an exponential function (Le Bihan et al., 1986). The total scan time for each ADC map was 14.5 minutes. The images without additional diffusion weighting (b = 0) were used as T1/T2-weighted anatomic images.
Histologic examination
The conventional hematoxylin and eosin staining was used (Liachenko et al., 1998). The sections corresponding to the MRI planes were evaluated for histologic damages using the grading scales listed in Table 1 (Eke et al., 1990). To verify the location and size of brain infarct, staining with 2,3,5-triphenyltetrazolium chloride was performed in three animals after the MRI scans (Bederson et al., 1986).
Scoring scales of the brain ischemic damages
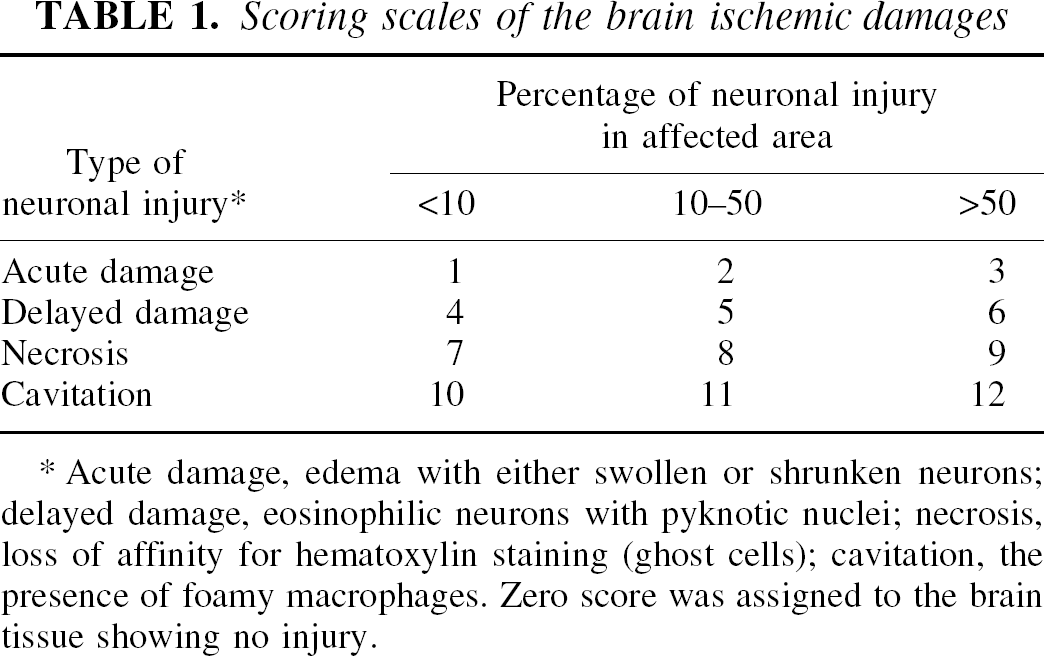
Acute damage, edema with either swollen or shrunken neurons; delayed damage, eosinophilic neurons with pyknotic nuclei; necrosis, loss of affinity for hematoxylin staining (ghost cells); cavitation, the presence of foamy macrophages. Zero score was assigned to the brain tissue showing no injury.
Data analysis
Cerebral perfusion and ADC maps were generated by Fourier transformation after zero-filling the datum matrix to 256 × 256. The average values of CBF and ADC over selected brain regions were calculated using the LabVIEW 5.1 software (National Instruments, Austin, TX, U.S.A.). For quantitative region-specific analysis of MRI data, an anatomic mask was generated using digital images from stereotaxic rat brain atlas (Swanson, 1998). The mask contained four regions of interest: cortical area (including the frontoparietal and piriform cortex) and caudate putamen in the ipsilateral hemisphere, and the homologous regions in the contralateral hemisphere, as shown in Figure 1. The masks were digitally scaled to fit the spin-echo image of each individual brain according to its position and size using the Photoshop program (Adobe Systems, San Jose, CA, U.S.A.). Care was taken to ensure that effects of distortions in brain anatomy (e.g., midline dislocation) were minimal. In most of the animals studied, we found no detectable shift in the midline caused by lesion or brain swelling, consistent with the observation by others (Olah et al., 2000). One-way analysis of variance followed by a post hoc analysis with Bonferroni correction was used for multiple between-group comparisons, and for comparisons of individual parameters in each group over time against the single control value before reperfusion. Non-parametric Mann-Whitney U test was used for histology scores. All data are presented as mean ± SD.
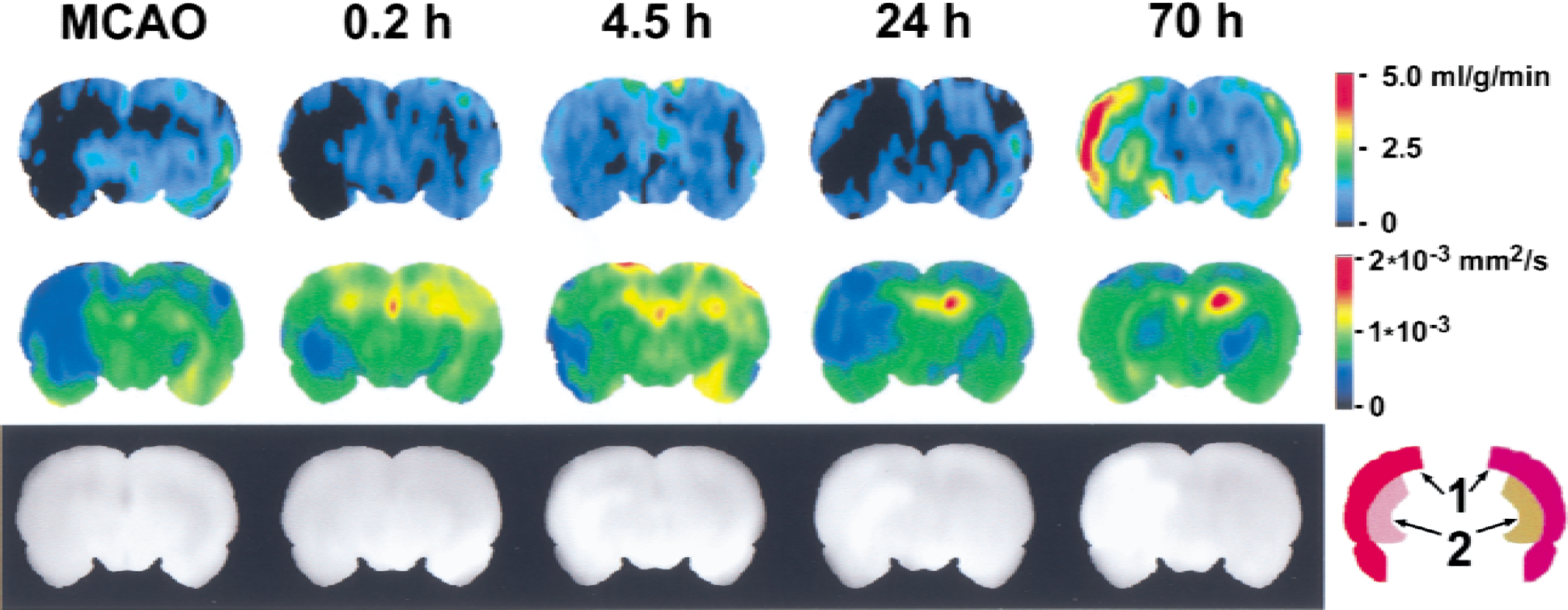
Images of the rat brain taken within 1 hour after middle cerebral artery occlusion (MCAO), and after 0.2, 4.5, 24, and 70 hours of reperfusion (top row: perfusion maps; middle row: corresponding apparent diffusion coefficient maps; and bottom row: corresponding spin-echo images). The colors in the maps of the top two rows encode the values of perfusion and apparent diffusion coefficient, respectively, using the color scales to the right of the corresponding rows. A representative anatomic brain mask used for the image analysis of the cortex (1) and caudate putamen (2) in the ipsilateral and contralateral brain hemispheres is shown in the bottom right corner.
RESULTS
Physiologic variables
Table 2 lists the physiologic parameters for the three groups. Body weight, body temperature, mean arterial blood pressure, and arterial blood pH and gases did not differ significantly either among the three groups or within groups between the occlusion and the reperfusion periods.
Physiologic variables of rats after middle cerebral artery occlusion and in the reperfusion period

Occlusion and reperfusion were not applicable to the sham group.
MABP, mean arterial blood pressure; pHa, arterial blood pH; Pa
Magnetic resonance imaging
The ADC decrease is an early and reliable manifestation of brain ischemia (Moseley et al., 1990; Busza et al., 1992). The presence or the absence of changes in ADC ∼40 minutes after MCAO in the MCA-supplied area of the brain was used to discriminate between animals with successful and unsuccessful (incomplete) occlusion. Five of the 17 MCAO rats showed incomplete occlusion and were analyzed separately. In the rats with successful occlusion, the lateral caudate putamen and the frontoparietal cortex, which are typically involved in the suture MCAO model as the ischemic core (i.e., irreversibly damaged part of the ischemic territory) and penumbra (damaged but still viable part), respectively, appeared most injured in the ADC maps and in the 2,3,5-triphenyltetrazolium chloride staining.
A significant finding in the present study is the reversal of CBF to an abnormally high level in the ipsilateral hemisphere after 2 to 4 days of reperfusion in the successfully occluded animals. Figure 1 shows typical MRI results obtained from the same animal during 3 days of experiment. On the insertion of the suture, successful MCAO is evidenced by the significant perfusion deficit and decrease in ADC value in the caudate putamen and cortex (mainly frontoparietal cortex). Averaged over all rats in the short- and long-term groups, the changes in CBF and ADC in the caudate putamen and cortex are shown in Fig. 2. Clearly, after the suture insertion, the ipsilateral CBF is significantly depressed (P < 0.001 compared to the homologous contralateral areas) to as low as 10 ± 10 and 30 ± 20 mL 100 g−1 min−1 in the caudate putamen and cortex, respectively. The perfusion deficit is more severe in the caudate putamen than in the cortex (P < 0.001). After suture withdrawal, recovery in CBF was not significant until after several hours of reperfusion. In the long-term group, no further recovery was observed during the first day of reperfusion, and in some cases, there was even a secondary decrease in CBF (Fig. 1). As the reperfusion continued into the second to fourth day after occlusion, however, there was an unexpected hyperperfusion to as high as 300 mL 100 g−1 min−1 in the ischemia-damaged areas. This reversal of CBF in the caudate putamen and cortex after prolonged reperfusion is highly significant (P < 0.001) when compared with the homologous contralateral regions before reperfusion. In the anatomic spin-echo images, the signal intensity depends on T2 and, to a lesser extent, on T1 of the tissue water protons in the brain. An increase in image intensity by 10% to 45% was apparent in the areas involved in ischemic damage in all rats and usually appeared on the first day of reperfusion (Fig. 1). On the sixth day of observation, the perfusion difference between brain hemispheres disappeared. Postmortem examination revealed extensive hemorrhage in two rats in the long-term group, presumably resulting from the late hyperperfusion.
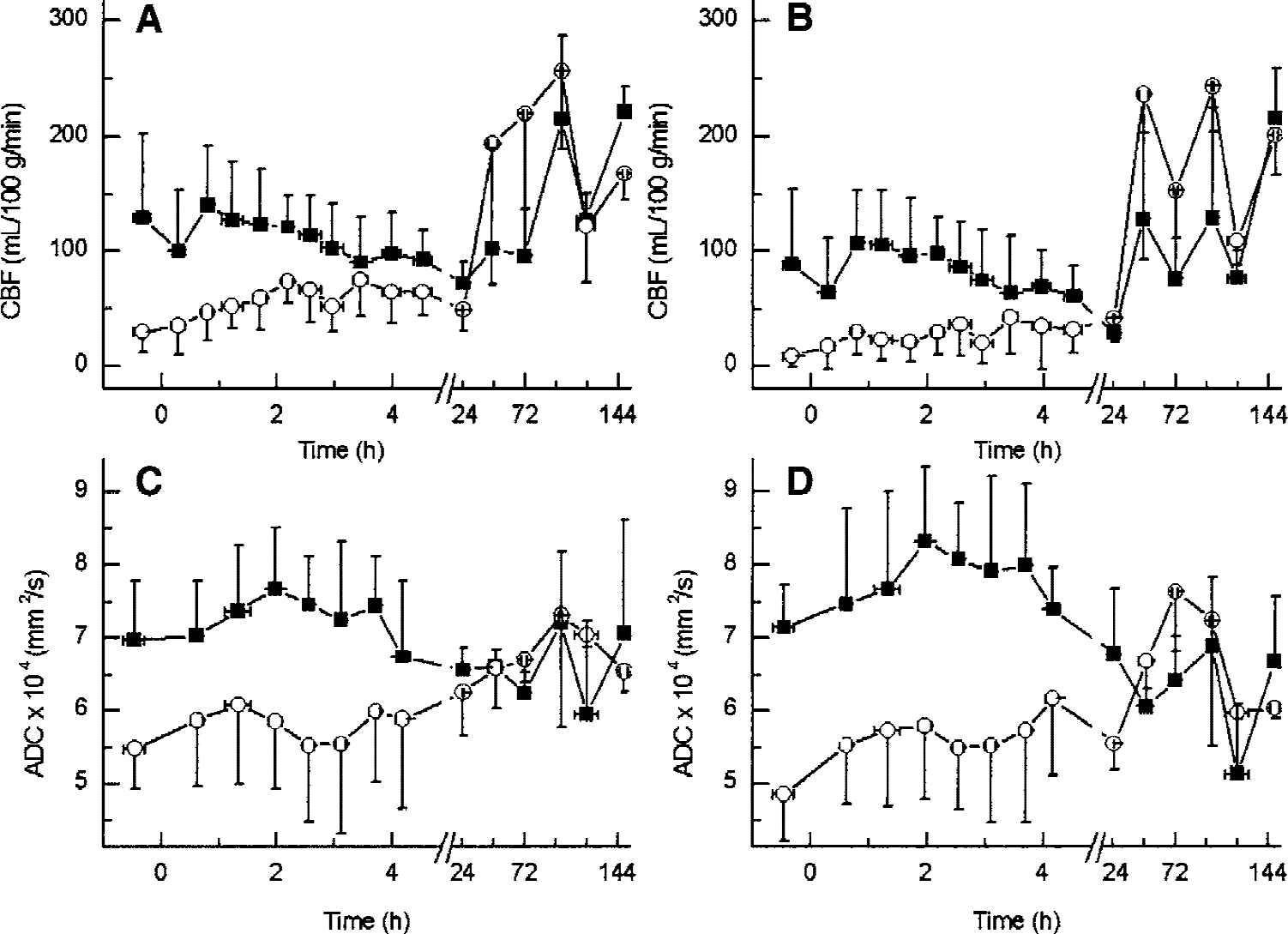
The time course of CBF (charts
The CBF in the contralateral hemisphere was also affected by the ipsilateral MCA occlusion and reperfusion. The CBF in the contralateral hemisphere decreased by approximately one half, reaching the minimum levels 1 day after MCAO (P < 0.05, compared to values before reperfusion, Figs. 2A and 2B), and exhibited some degree of hyperperfusion in the subsequent days.
Changes in ADC that accompany changes in CBF are shown in Figs. 2C and 2D. After occlusion, a significant decline in ADC, from (6.98 ± 0.82) × 10−4 to (5.49 ± 0.54) × 10−4 mm2/s in the cortex and from (7.16 ± 0.58) × 10−4 to (4.86 ± 0.62) × 10−4 mm2/s in the caudate putamen (P < 0.001), was observed in all successfully occluded rats. Partial recovery of ADC, as compared with the ADC in the occlusion period, was seen after 4.5 hours of reperfusion (P < 0.05 for the caudate putamen, Figs. 1 and 2D). In the long-term group, the hyperperfusion was accompanied by the recovery in ADC in the damaged area within 2 to 3 days. In addition, a low ADC area in the contralateral brain hemisphere was always apparent after 2 to 4 days of reperfusion (Fig. 1), albeit the average ADC decline in the contralateral side was not statistically significant.
In the five rats that were not successfully occluded, the perfusion deficit did not result in any decrease in ADC in the brain. Similar to the successfully occluded animals, these rats did not show immediate recovery in CBF on reperfusion. On the long term (1 to 6 days), however, these animals recovered, with no hyperperfusion or other abnormalities in ADC and CBF. In the three sham-operated rats, the CBF and ADC values in all brain regions were normal throughout the experiment.
Histologic outcome
The histologic findings in each group are summarized in Fig. 3, and typical histologic appearances of different grades of ischemic damage are illustrated in Fig. 4. In the sham-operated and unsuccessfully occluded rats, and in the contralateral hemispheres of the successfully occluded rats, no histologic abnormalities were seen. In the short-term group, all rats showed acute ischemic changes (edema with neuronal swelling or shrinkage) and different extent of delayed damage (nuclear pyknosis and cytoplasmic eosinophilia) in the ipsilateral cortex and caudate putamen. As shown in Fig. 3, the ischemic damage in the short-term group is significantly less than in the long-term group. In the long-term group, all rats had cytoplasmic eosinophilia or necrotic ghost cells with the presence of foamy macrophages in the damaged area. In two rats in the long-term group, pannecrosis (total death of all types of cells) was observed.
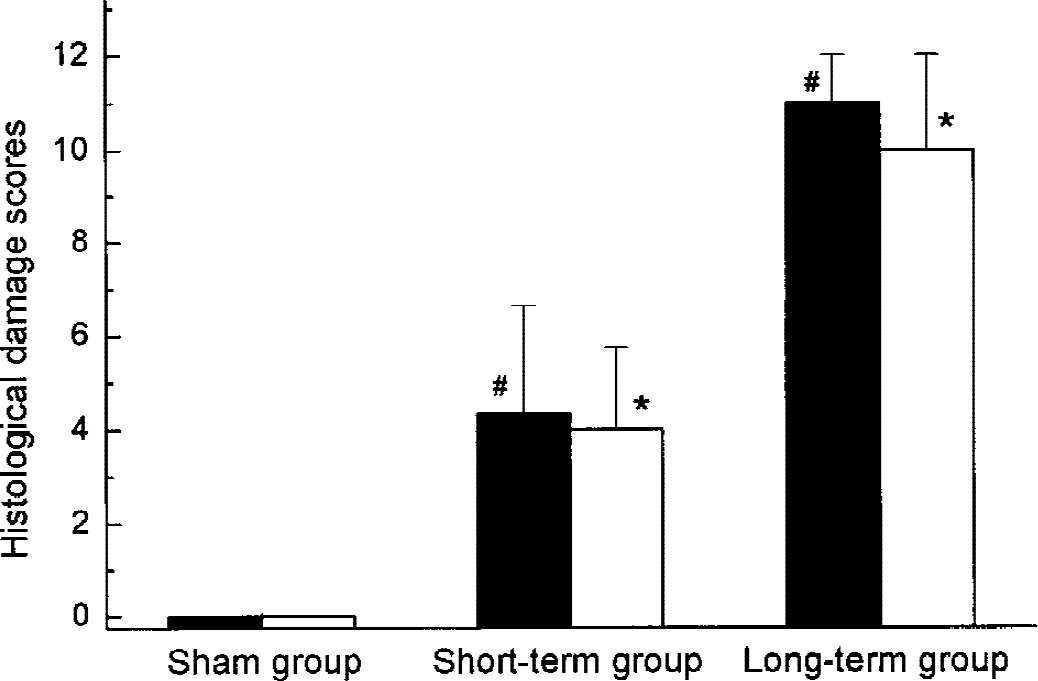
Histology scores in the ipsilateral caudate putamen (black columns) and frontoparietal cortex (white columns) assigned according to Table 1. The difference between the short- and long-term groups is significant (P < 0.05) both in the cortex (*) and caudate putamen (#). n = 3 for all three groups.
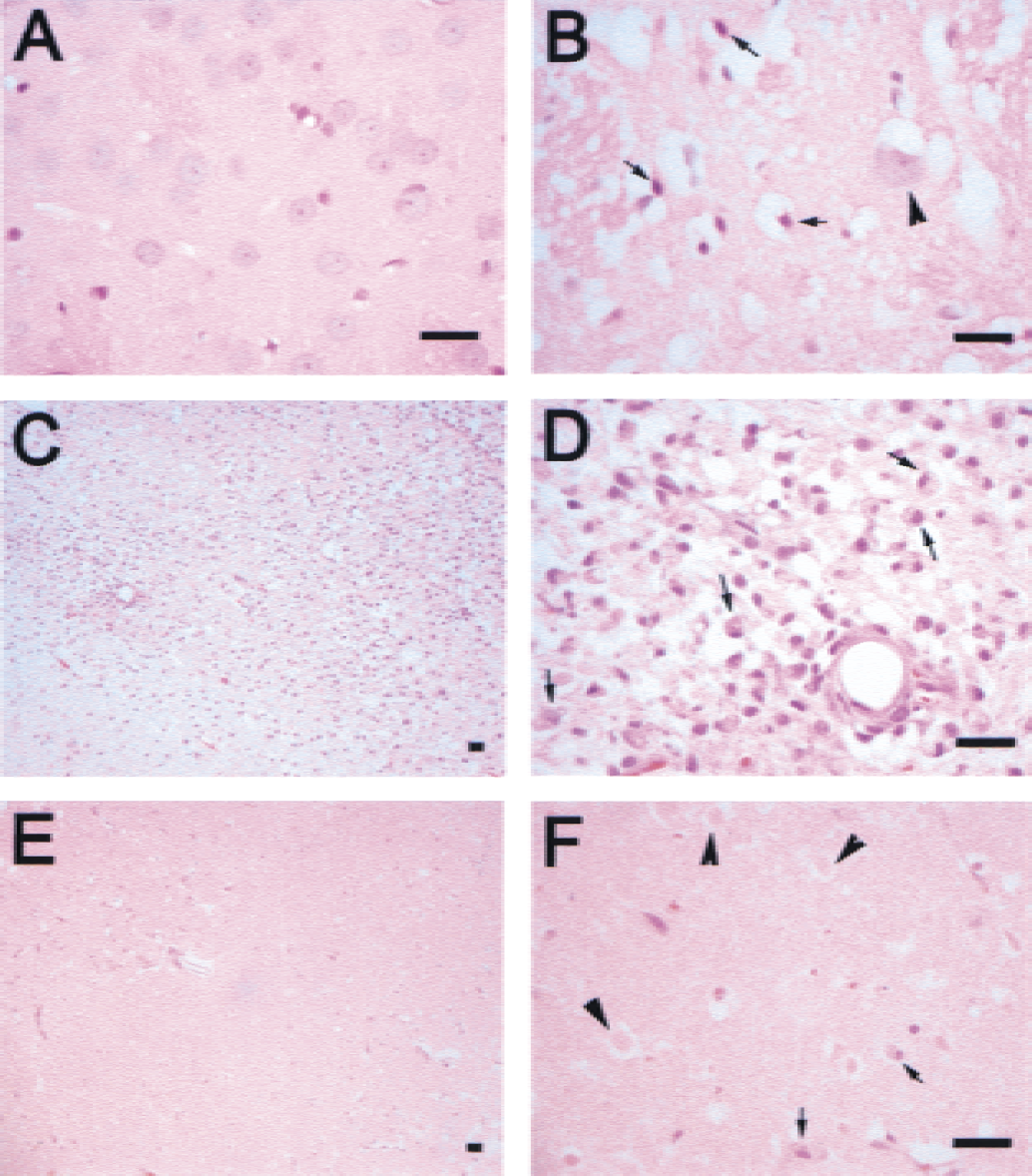
Different grades of histologic changes at 6 hours (
DISCUSSION
A remarkable finding, which has received little attention in the past, is the strong but delayed reversal and overshoot of cerebral perfusion (i.e., apparent hyperperfusion) in the ipsilateral caudate putamen and frontoparietal cortex between 48 and 96 hours of reperfusion after 80 minutes of MCAO. During the same period, ADC recovery in the damaged area and the appearance of a low ADC region in the contralateral hemisphere were noticeable. To date, very few studies reported the observations of increased cerebral hemodynamics in experimental ischemia after reperfusion of 24 hours or longer (Dijkhuizen et al., 1998; van Lookeren Campagne et al., 1999). Relative cerebral blood volume in the ipsilateral parietal cortex and caudate putamen during 24 to 70 hours of reperfusion has been shown to elevate to 130% to 160% of the contralateral values after 20-minute unilateral hypoxia-ischemia (Dijkhuizen et al., 1998) and to 120% to 200% after 30- to 45-minute MCAO (van Lookeren Campagne et al., 1999). An increase in cerebral blood volume, however, is not always associated with an increase in CBF (Kastrup et al., 1999). In our present study, arterial spin tagging technique (Williams et al., 1992) at high magnetic field (9.4 T) provides direct assessment of CBF. Although the absolute values of CBF by the arterial spin labeling method should be taken with caution (Calamante et al., 1999b), the relative changes found in this study are significant, particularly after corrections are made for the minor changes in α and T1 values. To the best of our knowledge, this is the first direct observation of significant increase in CBF in the late phase of reperfusion.
Delayed hyperperfusion should be clearly distinguished from the early postischemic events. In animal stroke models, early postischemic hyperperfusion is regarded as the hallmark of efficient recanalization of the occluded artery with subsequent reperfusion of the tissue (Marchal et al., 1999). It is unclear whether hyperperfusion has a beneficial or, conversely, harmful effect on the reperfused tissue. In rats, numerous studies (see, for example, Traupe et al., 1982; Todd et al., 1986; Marchal et al., 1999) show that restoration of cerebral perfusion pressure after ischemia consistently results in a marked hyperperfusion, usually within the first 10 minutes. This postischemic hyperperfusion is often followed by a phase of secondary hypoperfusion generally associated with a poor tissue outcome. In the rat MCAO model, reports are inconsistent on the duration of transient hyperperfusion, delayed hypoperfusion, and the recovery of CBF. Although some showed quick recovery of CBF (Li et al., 1999; Neumann-Haefelin et al., 2000; Olah et al., 2000), others found a significant delay—up to 2 to 24 hours into the reperfusion period (Jiang et al., 1993; van Lookeren Campagne et al., 1999)—in the renormalization of CBF after MCAO. The delay seems to be unrelated to the duration of occlusion, because it was sometimes observed after 30 to 45 minutes of ischemia (van Lookeren Campagne et al., 1999), whereas in other experiments no delay in perfusion recovery was reported even after much longer (60 to 150 minutes) ischemic periods (Li et al., 1999; Neumann-Haefelin et al., 2000; Olah et al., 2000). In the present study, we found no evidence of immediate CBF recovery or the early hyperperfusion in the successfully occluded animals, although one cannot rule out the possibility that early transient events may have occurred in the first few minutes after the suture withdrawal but before the first postwithdrawal image was acquired (typically, 8 to 10 minutes after suture withdrawal).
The exact mechanisms underlying the delayed hyperperfusion are presently unclear. Possibility of technical artifacts of perfusion measurement can be ruled out. Water is known to be a diffusion-limited tracer (i.e., not a freely diffusible tracer), especially at high flows. Thus, if the barriers to water diffusion are somehow reduced, the apparent perfusion values may be artificially augmented compared with the values under the normal conditions, under which the CBF might be underestimated by the arterial spin tagging method. However, this underestimation of CBF has not been demonstrated (St Lawrence et al., 2000). In fact, it has been shown that CBF measurements by the arterial spin tagging approach are not sensitive to the diffusion limitation in the gray matter (St Lawrence et al., 2000).
Possible physiologic causes for the delayed hyperperfusion include ischemic damages to the vessel wall or the lack of vascular regulation in the damaged tissue. Dysfunction of cerebral autoregulation was reported to occur after stroke (Fieschi et al., 1978). Ischemic damage leads to the decrease of vascular smooth-muscle tone, causing hyperemia. Severity of ischemia before reperfusion may completely abolish the vascular reactivity so that the vessels are not able to respond to potent vasoconstricting stimuli such as hypocapnia and theophylline. At the same time, damaged cells may generate vasodilatory substances, such as basic fibroblast growth factor and NO, in the late phase of reperfusion (Rosenblatt et al., 1994; Takizawa et al., 1999). NO and its derivatives can mediate neuron death by damaging DNA and activating nuclear enzymes to deplete cellular energy stores (Samdani et al., 1997). The generation of reactive oxygen species (Slater et al., 1997) during reperfusion may also at least in part be responsible for the vasodilation. It has been shown that the microvascular abnormalities, including sustained arteriolar dilation, abnormal reactivity to hypocapnia, the reversal of the endothelium-dependent vasodilation response to acetylcholine, and the increased permeability of the blood-brain barrier to proteins, could be inhibited by pretreatment with oxygen radical scavengers (Nelson et al., 1992). Comparison of histologic changes in our study at 6 hours and 4 days after reperfusion shows clear progression of tissue injuries, which developed from the mild acute damage (characterized by edema with either swollen or shrunken neurons) and moderate delayed damage (characterized by eosinophilic neurons with pyknotic nuclei) at 6 hours to necrosis and pannecrosis (appearance of “ghost cells” and foamy macrophages) 4 days later. Irrespective of the underlying causes, the hyperperfusion in the late phase of reperfusion is likely to reflect the microvascular damage that may aggravate the early reperfusion damage and contribute to the final ischemic injury. It has been shown that moderate reactive hyperemia after temporary MCAO in cats was well tolerated when the flow did not exceed approximately 125% of the preischemic values, whereas more severe hyperperfusion tended to be associated with larger infarcts, brain swelling, subarachnoid hemorrhage, and death (Heiss et al., 1997).
In the contralateral brain hemisphere, a certain degree of decrease in CBF during the first several hours after surgery is nonspecific, because this is also found in the sham-operated and permanently occluded animals (data not shown). The decrease in CBF within 24 hours of reperfusion may be attributed to the vasogenic edema, which usually appears as increased T2, as shown in Fig. 1. Microvascular damage leads to an increase in intracranial pressure, which in turn may result in a decrease in cranial perfusion pressure. Alternative explanations include cerebral autoregulation of the perfusion pressure and the vasospasm of the homologous contralateral MCA (Busch et al., 1998).
As mentioned earlier, the ADC changes after MCAO were used as a criterion for discriminating between successful and unsuccessful occlusions. Previous reports have suggested that decrease in ADC occurs only if CBF falls below a critical threshold, which was estimated in the global ischemia models to be 15 to 20 mL 100 g−1 min−1 (i.e., less than 15% to 20% of control) in gerbil (Busza et al., 1992) and rat (van der Toorn et al., 1996). Other studies (Hoehn-Berlage et al., 1995; Mancuso et al., 1995) suggest that perfusion thresholds might depend on the duration of ischemic episode. Our study shows that in most animals, ADC value declined rapidly and significantly when CBF decreased to less than 15% to 20% of control in the MCA territory, especially in the caudate putamen. In five animals, we observed similar deficits in perfusion but without accompanying ADC decrease and histologic changes (incomplete ischemia). This is probably because of the relatively minor CBF fluctuations near the threshold for ADC changes, and variability of the perfusion threshold between animals. Therefore, ADC seems to be a better index of ischemic damage than perfusion deficit alone. Further discussion on perfusion thresholds for ischemic tissue damage, variability between animals and between different brain structures within the same animal, and the use of ADC change as a more stringent criterion for successful occlusion can be found in a recent publication from our group (Yushmanov et al., 2002).
Factors accounting for the ADC changes include tissue water and ionic homeostasis (Mintorovitch et al., 1994), the misbalances of which may result from acute cellular energy failure during ischemia (van der Toorn et al., 1996) and the disruption of the Na+/K+ pump (Benveniste et al., 1992), leading to cytoplasmic membrane dysfunction and accumulation of intracellular water. In our studies, depressed ADC after 4 to 5 hours of reperfusion characterizes the intracellular changes associated with the severe disturbances of the cerebral microcirculation. Reperfusion may give rise to enhanced production of lactate and free radicals, cause intracellular and mitochondrial calcium overload, and hence result in mitochondrial dysfunction, leading to further reduction in ATP and neuronal damage. Although during the late hyperperfusion period ADC appeared to have recovered, corresponding intensity changes in T1/T2-weighted images and the changes in neuron morphology suggest that more severe damage occurs at the cellular level during this period. This observation is in agreement with the transient nature of ADC pseudonormalization after shorter-term MCAO (Li et al., 1999; Li et al., 2000; Miyabe et al., 1996; Neumann-Haefelin et al., 2000; Olah et al., 2001; van Lookeren Campagne et al., 1999). Our MRI and histopathology results and the time course of the tissue damage after delayed hyperperfusion seem to suggest that vascular factors, along with the selective cell death, play an important role in the progression of infarction after ∼80 minutes of focal cerebral ischemia.
Regional differences in ADC behavior may be explained by different vascular anatomy. Cortical regions may be more resistant to microvascular compromise than caudate putamen because of their ability to form perfusion collaterals. The late decrease in ADC in the contralateral hemisphere may result from reduction of CBF and localized vasospasm. The secondary decreases in ADC in the contralateral brain hemisphere have also been found in the acute subarachnoid hemorrhage, where such decreases have been attributed to spreading depolarization of the brain tissue (Busch et al., 1998).
In conclusion, this study presents the direct observation of significant increase in CBF during the long-term reperfusion period with accompanied ADC abnormalities related to infarct progression in the rat suture occlusion model. It is evident that vascular factors play an important role in infarct development after 80 minutes of cerebral ischemia. Our data support the notion that late hyperperfusion may be both the result of reperfusion injury and the cause of further injury, and thus underscore the importance of therapeutic approaches directed at ameliorating reperfusion injury. A better understanding of the mechanisms of CBF and ADC reversal in the late stage of reperfusion may prove useful in characterizing the degree of tissue damage, and in developing and evaluating treatment strategies.
