Abstract
Tissue hypoperfusion during cerebral ischemia results from occlusion of large and small vessels. Combination treatment strategies using fibrinolytics to thrombolyse an embolic clot and antiplatelet agents to prevent reocclusion and the formation of new platelet thrombi in the microcirculation may offer advantages over single-agent therapy. The authors report on the effects of tissue plasminogen activator (rt-PA), a glycoprotein (GP) IIb/IIIa receptor antagonist, 7E3 F(ab′)2, or a combination of the two agents in a focal embolic model of cerebral ischemia in Wistar rats. Focal ischemia was produced by introducing an autologous thrombus into the right side middle cerebral artery. Forty-six male Wistar rats were randomly divided into 6 groups: control (n = 8), 7E3 F(ab′)2 (n = 9, 6 mg/kg), rt-PA (n = 9, 10 mg/kg), rt-PA (n = 6, 20 mg/kg), and 7E3 F(ab′)2 with either 10 mg/kg (n = 10) (low-dose combination) or 20 mg/kg (n = 6) (high-dose combination) rt-PA. Evaluation of neurobehavioral scores, cerebral angiography, bleeding time, and measurement of brain infarction volume were used to determine efficacy. All actively treated groups showed a significant reduction in the infarct volume. Animals treated with 7E3 F(ab′)2 showed reduced infarction volumes (24.0 ± 5.1%) compared with controls (42.43 ± 5.6%, P < 0.02). Treatment with rt-PA significantly reduced infarction volume (20.7 ± 3.3, = 0.01) at 10 mg/kg and at 20 mg/kg (19.5 ± 8.2%, P < 0.05). Compared with vehicle-treated animals, the low-dose combination (16.4 ± 5.5, P < 0.003) and high-dose combination (23.7 ± 6.2%, P < 0.05) showed significant reduction in infarction volume. Cerebral angiography revealed significantly better recanalization in the combination group (5/6 animals in the high dose and 4/6 in low dose) compared with animals treated with 7E3 F(ab′)2 (3/10) or rt-PA alone (2/6). Bleeding time significantly increased from 11.25 ± 1.9 minutes in the control group to 17 ± 3.1 minutes in the rt-PA group, 24.5 ± 2.6 minutes in the 7E3 F(ab′)2 group, 25.7 ± 3.1 minutes in the low-dose combination group, and 32.5 ± 4.7 minutes in the high-dose combination group. The incidence of intercerebral hemorrhage was highest in the high-dose combination group (6 of 6 animals) and lowest in the single treatment with 7E3 F(ab′)2 alone (1 of 10 animals) (P < 0.05). Our data show that murine 7E3 F(ab′)2 alone has therapeutic effects when used after cerebral ischemia. Although this study suggests that higher doses of thrombolytic combined with anti-GPIIb/IIIa therapy may increases the risk of intracranial hemorrhage, the data also support the notion that anti-GPIIb/IIIa agents can safely be combined with low doses of thrombolytic agent to produce significant attenuation of neuronal damage with no increase in the incidence of cerebral hemorrhage.
Keywords
Proximal middle cerebral artery (MCA) occlusion results in a large cerebral infarction. Initially after occlusion of a large intracranial vessel, there is a decrease in cerebral blood flow in the distribution of the vessel. Over time as the vessel recanalizes, smaller distal occlusions are evident in the microcirculation (Wang et al., 2001). These occlusions may be a result of platelet activation and microthrombus formation in regions of poor blood flow. The time from onset of arterial obstruction to recanalization is one of the most important determinants of the extent of neuronal injury. With short-interval reversible ischemia, damage may be restricted to neurons. Prolonged severe ischemia produced pannecrosis with injury to neurons, support structures, and blood vessels. In experimental models of focal cerebral ischemia (Umemura et al., 1994; Busch et al., 1998; Shuaib et al., 1998; Li et al., 1998), neuronal death can be reduced if cerebral blood flow is rapidly restored. Unfortunately, a number of factors, including the short time window for the treatment, and a serious increase in the risk of cerebral and systemic hemorrhage, limit the use of tissue plasminogen activator (rt-PA) therapy to only a small number of patients (Koller and Anderson, 1998; Larrue et al., 1997). Any strategy that can increase the time window for efficacy and decrease the risk of hemorrhage may have clinical usefulness in the management of acute ischemic stroke.
The platelet glycoprotein IIb/IIIa receptor (GP IIb/IIIa) mediates platelet aggregation. Expressed at high levels on circulating platelets, this receptor cannot engage its ligand, fibrinogen, unless platelets are activated by prothrombotic stimuli such as ADP, thrombin, or shear. On activation, GPIIb/IIIa assumes a conformation that causes high affinity and rapid binding to fibrinogen. Inhibition of GP IIb/IIIa, using monoclonal antibodies or small molecule antagonists, reduces patient morbidity and mortality in the treatment of unstable angina (Ferrario et al., 1999) and acute myocardial infarction (Ellis et al., 1998; Shetler et al., 1998; Narins et al., 1999; Mickelson et al., 1999). Abciximab is a chimeric monoclonal antibody Fab fragment that blocks GPIIb/IIIa and has been shown not only to prevent platelet-mediated thrombosis, but also in animal models and clinical experience to actually recanalize occluded coronary vessels without the need for concomitant fibrinolytic therapy in patients (Gold et al., 1997; Domanovits et al., 1998; Rechavia and Wurzel, 1998; Rerkpattanapipat et al., 1999). This apparent thrombolytic effect may to be caused by the displacement of fibrinogen from the GPIIb/IIIa receptor, or could be related to the ability of abciximab to prevent plasminogen activator inhibitor-1 secretion from platelet granules (Chronos et al., 1999). Alternatively, addition of abciximab may prevent the accrual of new platelets to the thrombus, thus tipping the balance in favor of endogenous lysis. Recently, the utility of 7E3 F(ab′)2 in rats has been reported (Nakada et al., 1999). This agent appears to have pharmacodynamic properties that are similar to abciximab in humans (Nakada et al., 1999) and was therefore used in this rat model. The effects of blockade of GPIIb/IIIa have been studied in several animal models of cerebral ischemia studies (Choudhri et al., 1998; Huang et al., 1999). In a murine model of reversible focal cerebral ischemia, administration of the GPIIb/IIIa antagonist, GPI 562, reduced cerebral infarct volume (Choudhri et al., 1998). Additional studies have shown that inhibition of GPIIb/IIIa prevents anodal electrolytic-stimulated thrombosis in the carotid artery in cynomolgus monkeys (Rote et al., 1994) and dogs (Mousa et al., 1997).
Based on the pharmacological actions of GPIIb/IIIa receptor antagonists in inhibiting platelet aggregation, platelet procoagulant activity (thrombin generation and hence fibrin formation), and facilitating spontaneous thrombolysis, we hypothesized that such inhibitors may have therapeutic benefits in focal embolic cerebral ischemia in which a thrombus is already formed and lodged in an affected cerebral artery. We tested 7E3 F(ab′)2, in a focal embolic model of cerebral infarction and found that it reduced the size of cerebral infarction without producing an increase risk of cerebral or systemic hemorrhage (Yang et al., 2001). Therapeutic effects were observed when the antibody was administered 1 or 3 hours after onset of arterial occlusion. Because 7E3 F(ab′)2 and rt-PA work through distinct separate mechanisms, combining the two may offer superior results compared with those obtained with either agent alone. We also hypothesized that addition of anti-GPIIb/IIIa therapy may reduce the dose requirement for rt-PA efficacy, thus lowering the risk of cerebral hemorrhage. In this study, we report our results with the use of 7E3 F(ab′)2 alone or in combination with rt-PA in an embolic cerebral ischemia model in Wistar rats.
MATERIALS AND METHODS
All experiments in this study were approved by the Health Sciences Animal Welfare Committee of University of Alberta and performed in strict accordance with the related guidelines. All animals were fasted 12 hours before surgery to normalize the blood glucose level. Any animals that did not exhibit neurologic deficits on awakening from surgery were excluded from the study. A total of eight animals that did not show neurologic deficits were not analyzed further.
Cerebral focal ischemia model
Embolic focal cerebral ischemia was induced in anesthetized rats with the technique detailed in previous reports (Shuaib et al., 1998). Briefly, male Wistar rats (350–400) were anesthetized with 3.0% halothane and anesthesia maintained with 1.5% halothane in 70% N2O and 30% O2 (vol/vol) mixture with a face mask for the duration of surgery. The rectal temperature of animals was monitored and kept at approximately 37°C using a heating pad and an overhead lamp. Other physiological parameters including MABP, O2 saturation, and pulse were also monitored throughout the experiment using a Biopac “MP100” system (Biopac System, Santa Barbara, CA, U.S.A.). The right MCA was occluded by placement of an autologous embolus at the origin of the MCA via a 15-mm length of modified polyethylene catheter.
Neurologic deficit
Neurologic deficit evaluation was conducted at 2 and 24 hours, respectively, after injection of autogenous arterial thrombus into the MCA. The neurologic findings were scored on a four-point grade scale: no observable deficit, 0; forelimb flexion, 1; forelimb flexion and decreased resistance to lateral push, 2; forelimb flexion, decreased resistance to lateral push, 3; and unilateral circling in three successive trials and/or decreased consciousness, 4. This scale was originally reported elsewhere (Bederson et al., 1986). Any other neurologic behavior such as seizure was also recorded.
Evaluation of recanalization by the carotid angiogram
Within 30 minutes after injection of the autologous thrombus into the right side MCA, contrast angiography was performed to confirm successful clot embolization in the MCA. This was done by reinserting a catheter through the previous puncture in the right external carotid artery and injecting 0.2 mL heparinized (5 IU/ml) iohexol (Omnipaque, 300 mg I/mL, Nycomed, Denmark) into the ipsilateral internal carotid artery and its distal territory. The patency of the MCA stem or branches was blindly evaluated. For evaluating the efficacy of recanalization by postischemic administration of 7E3 F(ab′)2, rt-PA, or combination therapy, a second angiogram was performed using the same procedure at 3 hours after initial therapy. The X-ray exposure settings for angiogram in this study (62 kV, 100 mA, 1/200 seconds) were kept constant throughout the experiment. Cerebral angiography was done in 6 of 8 animals of control animals, 6 of 9 of animals treated with 7E3 F(ab′)2, 6 of 13 animals treated with high-dose rt-PA, 6 of 6 animals treated with high-dose combination, and 6 of 10 animals treated with lose-dose combination therapy.
Quantification of brain infarct volume
The details of the evaluation of following methods related to quantification of infarct volume were reported elsewhere (Yang et al., 1998). Briefly, 72 hours after MCA embolism, the rats received intracardiac perfusion of 100 mL normal saline under deep anesthesia by injection of overdose thiopental (Pentothal) (100 mg/kg). The brain was removed from the skull and cooled in ice-cold saline approximately 5 minutes. For morphometric study, 2-mm-thick coronal sections were cut using a rat brain matrix. Total eight coronal sections were prepared for estimation of the degree of infarct damage. Then the coronal sections were stained using 2,3,5-triphenyltetrazolium chloride (TTC). The stained brain sections were placed directly on the scanning screen of a color flatbed scanner (Scanjet 4p, Hewlett-Packard) within 7 days. A colorless transparent glass cover and a piece of black cloth put on the glass cover were used to provide a black background. After image acquisition, the images were analyzed blindly using a commercial image-processing software program, PhotoShop, version 4.0 (Adobe system) installed on the same computer. Measurements were made by manually outlining the margins of infarct areas. The total volume of infarction was determined by integration of the distance of the eight chosen sections.
Because brain edema may affect the accuracy of infarct estimation, to compensate for brain swelling in the ischemic hemisphere, we measured the corrected infarct size instead of using the direct measurement. This method corrects infarct size and involves measuring both hemispheres and applying the following formula: Corrected infarct size = the size of the left hemisphere – (the right hemisphere–measured infarct size). In this study, the infarct size was expressed as a percentage of the volume in the region examined. The percentage of infarct volume was obtained by calculating the portion of the corrected infarct size in the total size of the normal hemisphere.
Therapeutic regimen
A total of 54 rats were used for the experiments. However, eight animals were excluded from analysis because they lacked any neurobehavioral deficits at 2 hours after surgery. The lack of such deficits suggests that embolization was not successful. The remainder of the 46 male Wistar rats were randomly divided into six groups: (1) control group receiving vehicle (normal saline) infusion at 3 hours after cerebral embolism (n = 8); (2) 7E3 F(ab′)2 (Centocor, Malvern, U.S.A.) treatment group, 6 mg/kg (n = 9); (3) rt-PA (Activase; Genentech, Inc., Mississauga, Canada) at low dose (10 mg/kg, IV; n = 9) and (4) rt-PA at high dose (20 mg/kg, IV; n = 6); (5) 7E3 F(ab′)2 (6 mg/kg) and rt-PA (10 mg/kg) (low-dose combination) (n = 8); (6) 7E3 F(ab′)2 (6 mg/kg) and rt-PA (20 mg/kg) (high-dose combination) (n = 6). Animals treated with rt-PA were started 3 hours after onset of ischemic stroke and 7E3 F(ab′)2 received an initial bolus injection of one third of the dose, followed by intravenous infusion of the remainder of 7E3 F(ab′)2 for 30 minutes beginning at 3 hours after cerebral embolization through the tail vein. The 6 mg/kg dose of 7E3 F(ab′)2 was selected to provide >80% inhibition of platelet aggregation at 6 hours after dosing. Previous studies demonstrated that 4 mg/kg provided >80% inhibition of platelet aggregation at 6 hours after dose and ∼40% inhibition 24 hours after dose in rats (Sassoli et al., 2001). The amount of inhibition observed 6 hours after dose with 7E3 F(ab′)2 is similar to what is observed in patients treated with the currently approved drug regimen. We used computer-generated (Minitab V3.0) number table to perform random allocation of animals to different groups. A total of 54 animals received the embolic occlusion in the distal internal carotid artery, but only 46 were analyzed for this study. The remaining eight animals did not show behavioral change at 2 hours after embolization, suggesting a failure of arterial occlusion.
Bleeding time and intercerebral hemorrhage observation
Rat tail bleeding was induced by sectioning the extremity of the tail 3 mm from the tip. The rat's tail was gently blotted with tissue paper every 2 minutes, and the time in minutes to cessation of bleeding was recorded. No pressure was exerted on the tail tip, because this may affect hemostasis. Intercerebral hemorrhagic conversion was evaluated at the time of brain cutting. The location and extent of hemorrhage was recorded. Direct visualization of the brain surface (for subarachnoid hemorrhage) and careful study of sliced section (for intracerebral hemorrhage) were used to evaluate the extent of hemorrhage. The exact extent of the hemorrhage, however, was not quantified.
Data expression and statistical analysis
All data in this study were expressed as mean ± SD. Statistical analysis of more than two groups of animals was performed with one-way analysis of variance, with subsequent individual comparisons using Scheffe's test. The x2 test was used to compare the difference of recannulization rate or intracranial hemorrhagic complications among the groups. The difference was considered significant when P < 0.05. To avoid possible bias in interpretation of data, the investigator who carried out the assessment of brain infraction and neurobehavioral scoring was blinded to the embolization procedure or the treatment regimen.
RESULTS
There were no significant differences in physiologic variables recorded before and after introduction of thrombus into the MCA and among different groups.
Evaluation of recanalization by cerebral angiography
No spontaneous recanalization was observed in the control group treated with normal saline. Postischemia treatment with 7E3 F(ab′)2 fully reopened the occluded MCA in three of nine animals (33%, P < 0.05 when compared with that in control animals). In the group treated with rt-PA, recanalization was evident in 3 of 9 animals (33%) that received 10 mg/kg rt-PA and 2 of 6 (33%) animals that received 20 mg/kg rt-PA that underwent cerebral angiography (both P < 0.05). Low-dose combination therapy resulted in 5 of 8 animals recanalizing (62%, P < 0.05) and the high-dose combination resulted in 4 of 6 animals recanalizing (67%, P < 0.05). Figure 1 shows one representative set of MCA angiographs in the high-dose combination treatment group.
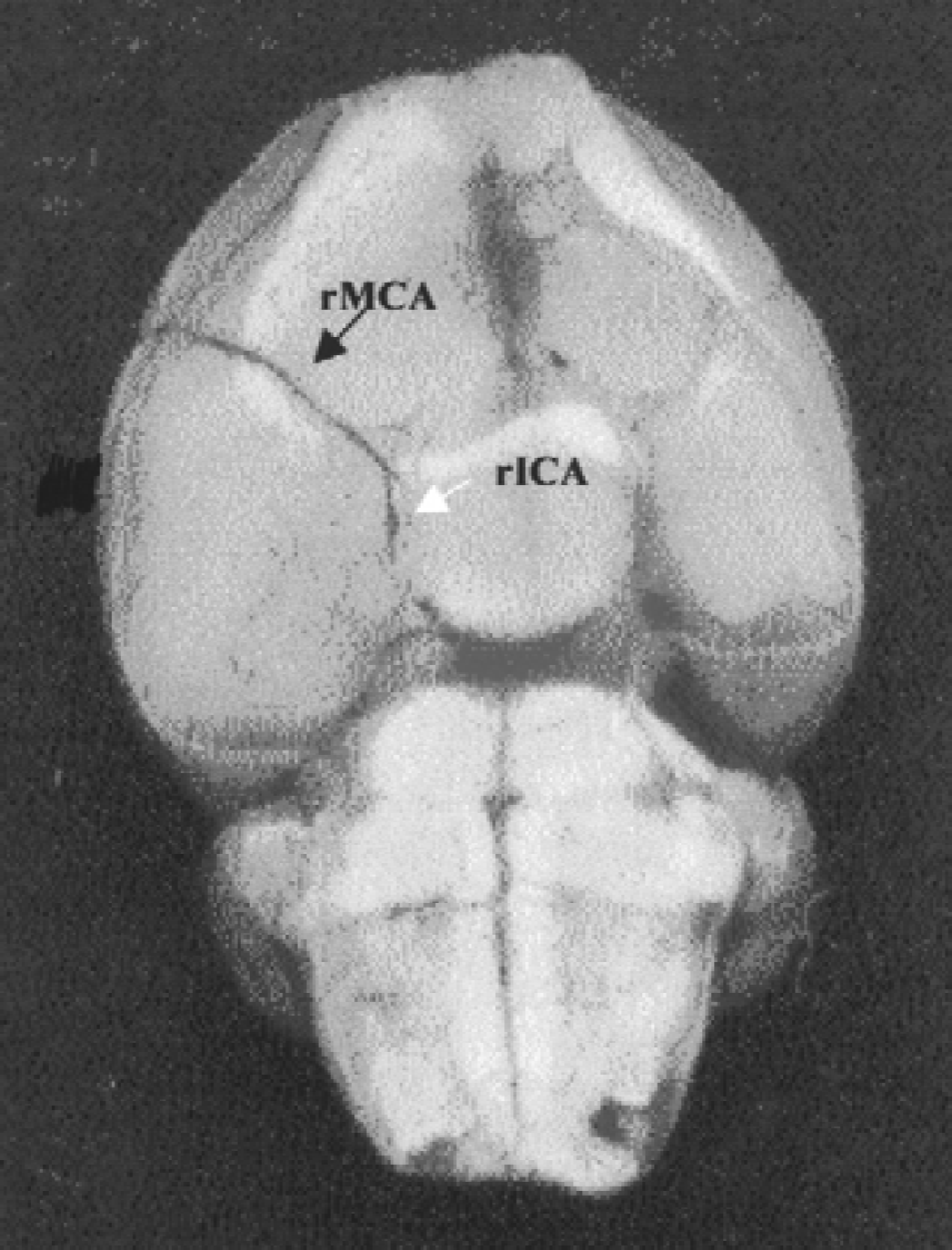
Representative right middle cerebral artery (MCA) angiograms in recanalized animals in high-dose combination group. Patency (contrast filling) of the right MCA was exhibited before injection of thrombus into the right MCA. No contrast medium filling in the right MCA and the right internal carotid artery 15 minutes after injection of the thrombus into the right MCA. Recannulization of the embolized MCA at 3 hours after initialization of high-dose combination therapy. However, this treatment also caused intracranial hemorrhage (as denoted by the arrow). rMCA, right middle cerebral artery; rICA, right internal carotid artery; rPTG, right pterygopalatine artery.
Neurologic deficits
Neurologic deficit scores observed in different groups at 2 hours and 24 hours after focal cerebra ischemia was summarized in Table 1. Animals receiving vehicle treatment, high-dose tPA treatment only, and high-dose combination therapy showed no significant difference in neurologic behavioral outcome between 2-hour and 24-hour observations. In contrast, animals in treatment with 7E3 F(ab′)2 or rt-PA at 10 mg/kg or low-dose combination demonstrated significant improvement of behavioral score at 24 hours.
Neurobehavioral scores observed at 2 hours and 24 hours after cerebral ischemia

Low-comb, low-dose combination group (7E3 F(ab′)2 6 mg/kg + rt-PA 10 mg/kg); high-comb, high-dose combination group (7E3 F(ab′)2 6 mg/kg + rt-PA 20 mg/kg).
Pathologic evaluation
The percentage volume of infarction in the three groups of treated animals compared with controls is shown in Figure 2. Compared with control animals with infarction volume of 42.4% ± 5.6%, animals treated with 7E3 F(ab′)2 showed infarction volume of 24.0% ± 5.1% (P = 0.02). Animals treated with rt-PA showed an infarction volume reduction to 20.7% ± 3.3% (P = 0.01) for 10 mg/kg rt-PA and 19.5% ± 8.2% (P < 0.05) at 20 mg/kg. The combination of 7E3 F(ab′)2 and the lower dose (10 mg/kg) of rt-PA also showed a significant reduction in the volume of infarction to 16.4 ± 5.5 (P < 0.003). Finally, 7E3 F(ab′)2 combined with high-dose rt-Pa (20 mg/kg) showed an infarction volume of 23.7 ± 6.2 (P < 0.05). Additionally, it was also noted that the animals receiving lower lose combination therapy showed significantly more reduction in brain infarction than the single-treatment group with 7E3 F(ab′)2 only (P < 0.048) and the higher dose combination group (P < 0.03). The tendency was seen when compared with other single treatments with rt-PA at higher or lower dose, but the difference did not reach statistical significance (Fig. 2). The larger volume in the high-dose combination group may be in part related to the higher incidence of hemorrhage observed.
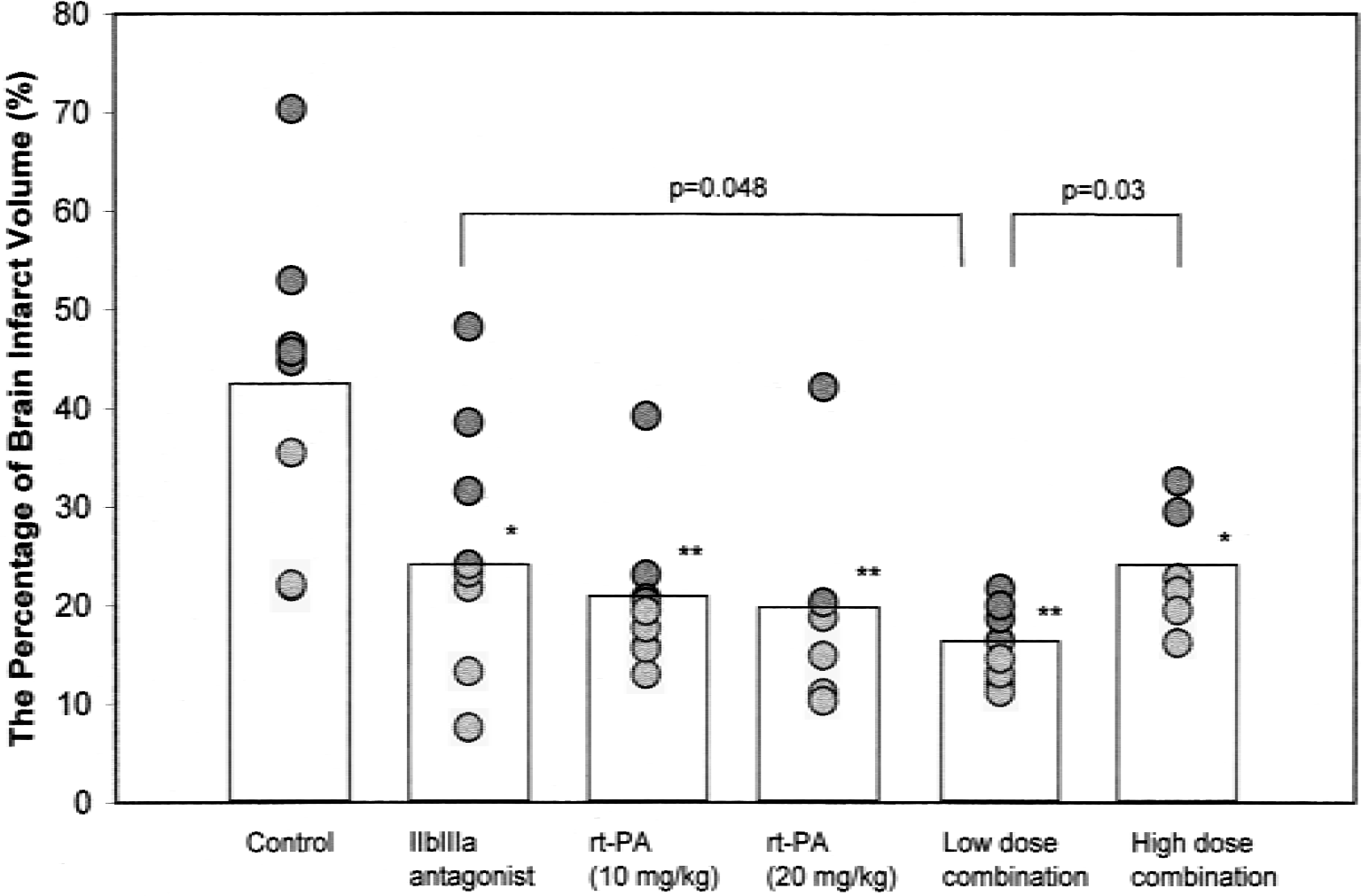
Cerebral infarct volume calculation in different groups. Animals subjected to MCA embolization caused a large cerebral infarction without drug treatment. Postischemia thrombolytic treatment using 7E3 F(ab′)2 demonstrated a significant reduction of neuronal damage after focal cerebral ischemia. Significant protection was also evident in the low- and high-dose tissue plasminogen activator–treated animals. Combination of low dose of the two agents resulted in a similar degree of efficacy. The high-dose combination group showed significant protection but also had a higher rate of hemorrhages (see Results sections for further details). *P < 0.05, **P < 0.01.
Intracranial hemorrhagic incidence
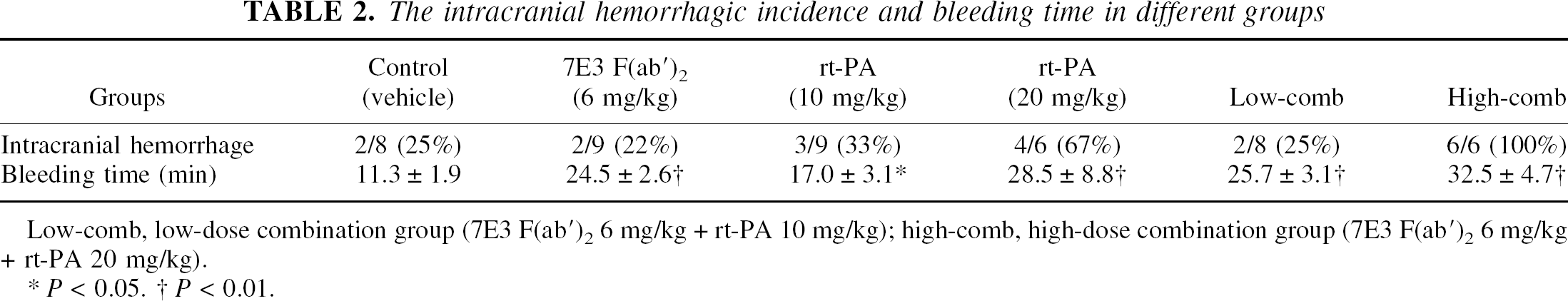
Cerebral hemorrhage was seen in all animals treated with the high-dose combination therapy (6 of 6 animals, 100%) and 4 of 6 (67%) in high-dose rt-PA-alone group. This was evident in the cerebral parenchyma and subarachnoid spaces in all animals. Parenchymal hemorrhage was for the most part restricted to the area of the infarction. In the low-dose combination group, hemorrhage was evident in 2 of 8 (25%) animals. Cerebral hemorrhage was seen in a significantly smaller percentage of animals in all other groups (control, 2 of 8 [25%]; 7E3 F(ab′)2, 2 of 9 [22%]; rt-PA 10 mg/kg low dose, 3 of 9 [33%]; all P < 0.05 when compared with the high-dose combination group) (Table 2).
The intracranial hemorrhagic incidence and bleeding time in different groups
Low-comb, low-dose combination group (7E3 F(ab′)2 6 mg/kg + rt-PA 10 mg/kg); high-comb, high-dose combination group (7E3 F(ab′)2 6 mg/kg + rt-PA 20 mg/kg).
P < 0.05.
P < 0.01.
Tail bleeding time
Bleeding time significantly increased from 11.25 ± 1.9 minutes in the control group to 17 ± 3.1 minutes in the rt-PA group low-dose group (P < 0.05), 28.5 ± 8.8 minutes in the high-dose rt-PA group (p < 0.01), 24.5 ± 2.6 minutes in the 7E3 F(ab′)2 group (P < 0.01), 25.7 ± 3.1 minutes in the low-dose combination group (P < 0.01), and 32.5 ± 4.7 minutes in the high-dose combination group (P < 0.01) (Table 2).
DISCUSSION
The combination of a fibrinolytic and a GP IIb/IIIa antagonist has, to our knowledge, not been previously tested in a focal cerebral ischemia model. Our data show several interesting findings. Although standard doses of 7E3 F(ab′)2 or rt-PA are effective in lysing thrombus, combination of a lower dose of rt-PA with anti-GPIIb/IIIa therapy may also be an effective alternative. Importantly, the efficacy of the combination with low-dose rt-PA was associated with a reduced incidence of cerebral hemorrhage. A higher dose of rt-PA in combination with anti-GPIIb/IIIa therapy, however, was deleterious and produced visually recognizable hemorrhage in all treated animals.
Angiography, done in animals treated with 7E3 F(ab′)2 alone, showed a 30% recanalization rate. Combination of 7E3 F(ab′)2 with rt-PA led to a significantly larger number of animals with arterial opening on angiography. In the combination group, recanalization was evident in 60% of animals in the low-dose rt-PA group and 80% in the high-dose rt-PA group. The increased recanalization with the low-dose combination group may explain the reduced infarction and better neurologic outcomes in this group. In spite of effective thrombolysis, the high-dose combination group did not demonstrate the same benefit, likely because of the hemorrhagic conversion of the infarction.
An occlusive thrombus consists of fibrin, platelets, and thrombin. Fibrinolytic agents such as rt-PA facilitate the dissolution of the fibrin mesh, but do not act on the platelet component of the clot. In addition, release of thrombin that occurs with fibrinolysis activates and recruits new platelets. Such platelet-rich clots are often resistant to thrombolysis and are prone to reocclusion. Acute platelet accumulation and hemodynamic depression may develop shortly after thromboembolic stroke in rats (Stagliano et al., 1997). Data have also suggested that there is significant increased platelet activation in the chronic phase after cerebral ischemia platelets play a role in not only large cerebral vessel occlusion, but also microvascular focal ischemia and infarction (Fisher and Francis, 1990; van Kooten et al., 1999). In our experimental model, the thrombus is lodged at the distal end of the internal carotid artery, leading to cessation of blood flow in the MCA. We have previously shown that immediately after occlusion of the distal common carotid artery, there is cessation of blood flow in the cerebral cortex and the striatum. One hour later, blood flow to the cortex is already becoming established. Three hours after the occlusion, microvessels in the striatum begin to show blood flow. Interestingly, patchy regions of microvascular occlusions may persist for hours after the recirculation (Wang et al., 2001). These microocclusions may be a result of stasis, platelet activation, and clot formation. It is also possible that these occlusions may be secondary to fragmentation of the main clot and its distal propagation. Platelet aggregation, leading to microvascular occlusions in models of focal ischemia, can be suppressed by antagonists of the GP IIb/IIIa receptor complex such as 7E3 F(ab′)2. The relationship between acute ischemic stroke and vascular patency has recently been studied (Abumiya et al., 2000). The investigators showed that platelet activation significantly contributed to small vessel occlusions and that these could be inhibited by Integrilin. Our data shows that combination of such agents with rt-PA may produce maximum therapeutic effects because these agents act through complimentary mechanisms. In this study, beneficial effects were observed with a lower dose of rt-PA combined with standard-dose anti-GPIIb/IIIa. Increasing the dose of rt-PA resulted in producing hemorrhage in all ischemic brains, suggesting that exact dosing of thrombolytic is important to consider when planning combination strategies for clinical trials.
A number of investigators have used embolic models to better replicate the clinical situation. For the last 2 years, we have used a modification of a method initially described by other investigators (Zhang et al., 1997). We believe that this model of ischemia in rats most closely resembles MCA territory infarction in humans. We have shown that the extent of the damage from emboli in the distal carotid can be decreased with the use of thrombolysis and with neuroprotective agents (Shuaib et al., 1998). In a recent study, we evaluated the effects of combining citicoline with urokinase and we showed clear evidence that the combination of the two agents had additive effects (Shuaib et al., 1999). There are several other studies that have used a combination of neuroprotective medications with thrombolytic agents (Zivin and Mazzarella, 1991; Shuaib et al., 1999; Yang et al., 2000) or a combination of thrombolysis with antiplatelet agents (see review article by Diener, 2000). Before our current work, aspirin had been the only agent tested in combination with thrombolysis.
The GPIIb/IIIa receptor plays a crucial role in platelet aggregation and platelet thrombus formation. We demonstrated previously that blockade of GPIIb/IIIa by 7E3 F(ab′)2 either 1 hour or 3 hours after thrombus formation significantly attenuated brain infarction in a focal cerebral ischemia model (Yang et al., 2001). In the present study, the first to our knowledge, we examined the antithrombotic efficacy of combinations of murine 7E3 F(ab′)2 and rt-PA in a clinical relevant and reproducible MCA embolization model. Our results demonstrate that combination therapy can significantly improve neurologic outcome and also improve recanalization. Administration of the GPIIb/IIIa receptor antagonist GPI 562 immediately after reperfusion has been shown to reduce cerebral infarct volumes by 70% in a murine model of reversible focal cerebral ischemia (Choudhri et al., 1998). Because the drug was administered after “re-establishment” of blood flow, the highly significant therapeutic response may be caused by the effects exerted by GPIIb/IIIa receptor antagonist in the microvascular where thrombosis in small vessels may persist despite reestablishment of blood flow. Supportive evidence for small vessel thrombosis was offered by documenting enhanced accumulations of fibrin and platelet on the affected side of cerebral hemisphere after recanalization of the MCA (Choudhri et al., 1998). This postischemic microvascular thrombosis may result in postischemic hypoperfusion and ongoing neuronal damage. Therefore, inhibition of the GPIIb/IIIa receptor could decrease the degree of ischemic damage by inhibiting fibrin and platelet accumulation. In another study (Mousa et al., 1997), preischemia treatment with a GPIIb/IIIa receptor antagonist led to a reduction in platelet aggregation and thrombosis introduced by anodal electrolytic stimulation to the intimal surface of the internal carotid artery in dogs. These findings may provide a possible explanation for the observation in our current study where some murine 7E3 F(ab′)2-treated animals did not demonstrate MCA recannulization but still showed reduced infarction and improved neurobehavioral scores. An accumulation of fibrin occurs in the microvasculature in a time-dependent manner after focal cerebral ischemia and reperfusion (Okada et al., 1994; Iwamoto et al., 1995). A recent clinical pathological study showed that, after a fatal ischemic stroke, there is increased coagulation (formation of microthrombi) in ischemic hemisphere during the stage of fresh infarction. Later, both the ischemic and contralateral hemisphere shows abnormally enhanced coagulation (Heye et al., 1992). Another observation supporting the occurrence of small vessel thrombi formation in the postinfarction period is the reported increase in brain infarction found after ischemic stroke in patients (Pantano et al., 1999). Postischemia treatment with murine 7E3 F(ab′)2 at 1 hour or 3 hours may decrease the progression of postischemia fibrin formation in microvessels of ischemic area. This may in part explain the therapeutic benefits with 7E3 F(ab′)2 in animals in which recanalization was not evident on the second angiogram.
In another study (Choudhri et al., 1998), it was reported that although the GPIIb/IIIa antagonist, GPI 562, caused a dose-dependent increase in tail vein bleeding time, the incidence of intracerebral hemorrhage was not significantly increased at therapeutic doses until at the highest doses tested. This finding is in accordance with our observation. We did not observe any increase in the incidence of cerebral hemorrhage, although the bleeding time was prolonged by more than two times that of control in the treated groups. In our previous study, we did not test the dose effectiveness in relation to development of intercerebral hemorrhage (Yang et al., 2001). However, we did not find the incidence of intercerebral hemorrhage to rise when the GPIIb/IIIa antagonist was given at 1 hour or 3 hours after the MCA embolization. This suggests that murine 7E3 F(ab′)2 has a relatively wide range of safety.
Thrombolysis is an attractive but potentially dangerous therapy for cerebral ischemia (Jaillard et al., 1999; Larrue et al., 1997). Thrombolysis after an acute ischemic stroke increases the risk of cerebral hemorrhage 10-fold compared to controls (Larrue et al., 1997). Risk factors for hemorrhage include time to thrombolysis, the presence of hypertension or coagulation abnormalities, and the dose of the lytic agent (Larrue et al., 1997; Koller and Anderson, 1998; Kochanek et al., 1999; Kristensen et al., 1999). The safety and efficacy of the treatment critically depends on the timing of intervention and on dose of thrombolytic agent used. The present study shows that rt-PA can be combined with murine 7E3 F(ab′)2 and complications are related to the dose of the thrombolytic agent. A lower dose of rt-PA combined with anti-GPIIb/IIIa had potent therapeutic effects without a risk of cerebral hemorrhage that was observed with the high-dose combination. Reperfusion may be the most important factor in tissue salvage after an arterial occlusion.
In conclusion, in this embolic stroke model, we compared the therapeutic effects of rt-PA and 7E3 F(ab′)2 alone and in combination. 7E3 F(ab′)2 significantly reduced the extent of infarction when administered 3 hours after onset of the stroke. Its beneficial effects are similar to those seen with rt-PA. The incidence of cerebral hemorrhage with 7E3 F(ab′)2 was low. When combined with rt-PA, the efficacy and potential for side effects depended on the dose of the thrombolytic. At a low dose, rt-PA combination therapy with 7E3 F(ab′)2 was effective in preventing brain damage from the effects of MCA arterial occlusion. At a higher rt-PA dose, the combination significantly increased the risk of brain hemorrhage. Combining anti-GPIIb/IIIa therapy reduces the dose requirement for thrombolytic for recanalization and thereby increases the safety profile of therapy. Combination therapy also appears to reduce infarct size and improve neurobehavioral outcomes, suggesting additional benefits to this therapeutic approach to treat stroke.
