Abstract
Uric acid is a natural antioxidant that protects the brain in a model of transient focal ischemia in rats. Here we sought to investigate whether uric acid was protective in a model of thromboembolic brain ischemia in rats, and whether the global benefit of recombinant tissue plasminogen activator (rt-PA) was improved by the combined treatment. Adult male Sprague-Dawley rats underwent either ischemia by thromboembolic middle cerebral artery occlusion (MCAO) or sham operation. Uric acid (16 mg/kg) was injected intravenously (i.v.). 20 mins after mCaO, whereas rt-PA (10 mg/kg) was administered i.v. at 3 h. A group of rats received the combined treatment. Rats underwent two neurologic examinations (30 mins and 24 h after MCAO). At 24 h, infarct volume was measured and brain neutrophil infiltration and protein tyrosine nitration were assessed. Treatment with either uric acid or rt-PA reduced infarct volume versus controls (P < 0.05). The protective effect against brain ischemia was greater after cotreatment of uric acid with rt-PA (P < 0.001), which added further benefit to rt-PA alone (P < 0.05). The neurologic score worsened during the first 24 h in treatment controls, whereas it improved in rats receiving uric acid and/or rt-PA. Uric acid strongly reduced ischemia-induced tyrosine nitration, but it was more effective alone than combined with rt-PA, suggesting that reperfusion enhances nitrotyrosine formation. All treatments reduced postischemic brain neutrophil infiltration. These results show that uric acid administered early after thromboembolic stroke is neuroprotective in the rat brain, as it reduces infarct volume, ameliorates the neurologic function, attenuates the inflammatory response, and extends the benefits of rt-PA.
Introduction
Thrombolysis with recombinant tissue plasminogen activator (rt-PA) administered intravenously (i.v.) within 3 h of symptom onset has shown benefits for patients suffering ischemic stroke in the two linked National Institute of Neurological Disorders and Stroke (NINDS) randomized clinical trials (National Institute of Neurological Disorders and Stroke Study Group, 1995). Nevertheless, a very small percentage of stroke patients receive this therapy given the reduced time window for drug administration (Albers et al, 2001). The risk of rt-PA-related bleeding complications is also of concern, and several neuroprotective strategies have been investigated to increase the safety and efficacy of thrombolysis in experimental animal models (Lapchak and Zivin, 2003), and in patients (Piriyawat et al, 2003).
Several studies support that oxidative stress contributes to ischemic neuronal death (Love, 1999; Watanabe et al, 1994), and various antioxidant therapies have shown encouraging results in acute stroke (Green and Ashwood, 2005). A clinical study reported an association between increased uric acid plasma levels at stroke onset and improved neurologic recovery at follow-up (Chamorro et al, 2002), and systemic administration of the natural antioxidant uric acid was safe in healthy volunteers and increased their serum antioxidant capacity (Waring et al, 2001). In contrast, another study found an inverse association between uric acid levels and stroke outcome (Weir et al, 2003), although the criteria used in this study to define outcome at follow-up were susceptible to confounding factors (Chamorro and Planas, 2004). In experimental studies, uric acid administration showed protective effects against ischemia/reperfusion in a rat model of transient intraluminal middle cerebral artery occlusion in the rat (Yu et al, 1998). Based on this ground, it was recently suggested that uric acid treatment could be beneficial and cost effective in patients with acute stroke, and that further study of this treatment modality was justified (Chamorro et al, 2004).
Here we examine whether treatment with uric acid is protective and can ameliorate the benefits of rt-PA in a rat model of thromboembolic brain ischemia.
Materials and methods
Thromboembolic Ischemia
Thromboembolic ischemia was induced in adult male Sprague–Dawley rats (n = 111) (328±22g, mean ± s.d. body weight). A group of rats was subjected to surgery, but thrombi were not injected (sham operated, n = 6). Rats were housed under a 12-h day/night light cycle and they had free access to food and water. Animal work was conducted with the approval of the ethical committee of our Institution, and in compliance with the Spanish legislation (Real Decreto 1201/2005) and the regulation of the European Community.
Embolization of the right middle cerebral artery (MCA) was carried according to previously described procedures (Busch et al, 1997), with modifications as follows. Heterologous thrombi were formed by rapidly mixing 300 μL of blood obtained through the femoral artery from a donor rat with 75μL of thrombin (30U/mL). Clots were freshly prepared every day. The mixture was immediately injected into a 1-m-long P50 catheter. Fifteen minutes later, the P50 tube was cut into pieces of 5 cm and the clotted blood was removed from the tube by pushing saline through a syringe. The thrombi were extensively washed with saline and pushed through a P10 catheter several times. Washes removed erythrocytes while keeping the fibrin network, which rends clots more resistant to spontaneous thrombolysis (Overgaard et al, 1993, 1994). For latter identification in the postmortem brain, thrombi were stained blue by immersing them in a 1% Blue-Evans solution for 1 min followed by saline washing. The clot pieces showed an irregular diameter, with some zones thicker than others. The thicker zones with a diameter of 0.25 to 0.35 mm resulted from a more compact fibrin network and were the zones selected. These were cut into 1.5-mm-long fragments, 9 of which were introduced in a P10 tube that was inserted into the right external carotid artery of the receptor rat. This rat was previously anesthetized with halothane (1.5% to 2%), intubated through the trachea for controlled ventilation, and the external carotid artery was isolated, as described (Justicia et al, 2001). The ipsilateral common carotid artery was clamped and the thrombi were infused at a speed of 0.45 mL/min in a volume of 150 to 200 μL. The external carotid artery was closed and the clamp on the common carotid artery was removed.
Drug administration: Human rt-PA (Alteplase; Boehringer Ingelheim, Germany) was infused (10 mg/kg of body weight) through the femoral vein by injecting a bolus (10% of the dose) followed by 45-mins infusion (90% of the dose). This dose is 10 times higher than the dose used in humans because of the low affinity of human-derived rt-PA to rat plasminogen (Korninger and Collen, 1981). Recombinant tissue plasminogen activator treatment was initiated 3 h after the onset of ischemia (n = 30). Uric acid was prepared in Locke's buffer (vehicle) as described (Yu et al, 1998), and it was i.v. injected through the femoral vein (16 mg/kg of body weight) 20 mins after the induction of ischemia (n = 32). Treatment controls (n = 30) were untreated or received the vehicle. A group of rats received uric acid at 20 mins and rt-PA at 3 h (n = 19).
Evaluation of brain damage after treatments: A simple neurologic test in a 9-point scale (0 = normal to 9 = highest handicap) was performed. The test was based on classic tests allowing to distinguish between rats with small, medium, and large areas of infarction (Bederson et al, 1986), with modifications (Perez-Asensio et al, 2005). The test was performed twice: 30 mins after embolization and at 24 h. Rats were anesthetized with halothane and were killed by decapitation at 24 h. A visual examination of the cerebral circulatory tree was carried out to identify the presence and location of thrombi. The brain was removed and sliced in 2-μm-thick coronal sections, which were stained with a 1% solution of 2,3,5-triphe-nyltetrazolium chloride (TTC) as described (Justicia et al, 2006). Infarct area in each section was taken as the white or pale zone in TTC staining. Sections were recorded with a video camera (JVC, TK-S350) and analyzed with an image analysis system (AIS, Imaging Research Inc., Ontario, Canada). Infarct areas of all sections were integrated to obtain infarct volume. In addition, the areas of the ipsilateral and contralateral hemispheres were measured, the contralateral area was subtracted from the ipsilateral area, and the difference area in each section was integrated to calculate the volume of brain swelling, as an index of the extent of brain edema.
Assessment of neutrophil infiltration: Myeloperoxidase (MPO) expression was studied by Western blot to assess brain neutrophil infiltration at 24 h, as reported previously (Justicia et al, 2003). In a group of animals (n = 4 to 7 per treatment), one of the 2-mm brain sections (at the level of Bregma) was used to dissect out the ipsilateral and contralateral MCA territory and the tissue was frozen and the latter used for Western blot analysis. Tissue samples were homogenized in lysis buffer containing 50 mmol/L Tris-HCl (pH 7.6), 150 mmol/L NaCl, 5 mmol/L CaCl2, 0.05% Brij-35, 0.02% NaN3, and 1% Triton X-100, and centrifuged at 12,000g for 5 mins at 4°C. All reagents, unless otherwise stated, were from Sigma (Madrid, Spain). Protein content was determined in the supernatant (Bradford assay; Bio-Rad, Hercules, CA, USA). Thirty micrograms of protein were run alongside prestained molecular weight markers (Bio-Rad) in denaturing 10% polyacrylamide gels. Proteins were transferred to membranes (Immobilon-P, Millipore, Bedford, MA, USA), which were incubated overnight at 4°C with a mouse monoclonal antibody against MPO (M1464; Menarini Diagnostics, Barcelona, Spain) diluted 1:500, followed by a 2-h incubation at room temperature with a peroxidase-linked anti-mouse secondary antibody diluted 1:2,500 (Bio-Rad). The reaction was developed with a chemiluminescence's reagent containing luminol. After this first reaction to detect MPO, membranes were incubated with a rabbit polyclonal antibody against β-tubulin (Boehringer Mannheim, Mannheim, Germany) diluted 1:10,000, followed by a peroxidase-linked antirabbit secondary antibody, as a loading control. The peroxidase-linked antirabbit secondary antibody (Bio-Rad) was used diluted 1:2,500.
Tyrosine Nitration
Formation of nitrotyrosine was studied by Western blot. Protein samples were treated as above and electrophoresis was performed in 8% polyacrilamide gels. Membranes were incubated overnight at 4°C with a mouse monoclonal antibody antinitrotyrosine (ab7048; Abcam Limited, Cambridge, UK) diluted 1:1,000. Then, they were incubated with an anti-β-tubulin polyclonal antibody as above. Band intensity was measured with an image analysis system (DC-120 camera and Kds1D, Digital Science System software, Kodak). The ratio between nitrotyrosine band intensity and the corresponding β-tubulin band intensity was calculated to correct for any differences in total protein content per lane.
Statistics
Data groups were assayed for normality with the D'Agostino and Pearson omnibus normality test. Data after a normal distribution were analyzed for differences between two groups with the Student's t-test, and for differences between several groups with one-way analysis of variance (ANOVA) followed by the post hoc Dunnett's multiple comparison test. Data not conforming normality were analyzed with the Kruskal-Wallis test followed by the post hoc Dunn's test. Linear regression analysis was performed to examine the correlation between infarct volume and neurologic score in the same animals. The incidence of events (dead or intracranial hemorrhage) in each group was compared with the Fisher's exact test. Statistical significance was taken below P < 0.05. Statistical analyses were performed with GraphPad Prism software.
Results
Uric Acid and Recombinant Tissue Plasminogen Activator Reduce Infarct Volume and Brain Edema
Mean arterial blood pressure and body temperature were maintained within the physiologic range during surgery, and they did not vary between the groups of rats that were assigned to the different treatments (Table 1). Embolization caused infarction within the MCA territory in 83% of the rats. The percentage of infarction in rats receiving treatments was similar (Table 1). Noninfarcted animals were not used for further determinations or analysis. Mortality within the first 24 h was lower in rats receiving uric + rt-PA, but differences were not statistically significant, as shown in Table 1. Macroscopic signs of intracranial hemorrhage were only observed in one of the surviving rats from the group receiving rt-PA. The incidence of events, that is, death or intracranial hemorrhage, in the rt-PA group showed a nonsignificant tendency to increase in relation to controls (Fisher's exact test P =0.21; odds ratio (OR) = 2.45, 95% confidence interval (CI) 9.24 to 0.65). Cotreatment of uric acid with rt-PA showed a trend to reduce the incidence of events compared with rt-PA alone (Fisher's exact test P= 0.068; OR = 0.14, 95% CI 0.02 to 1.3).
Physiologic parameters and features of the treatment groups
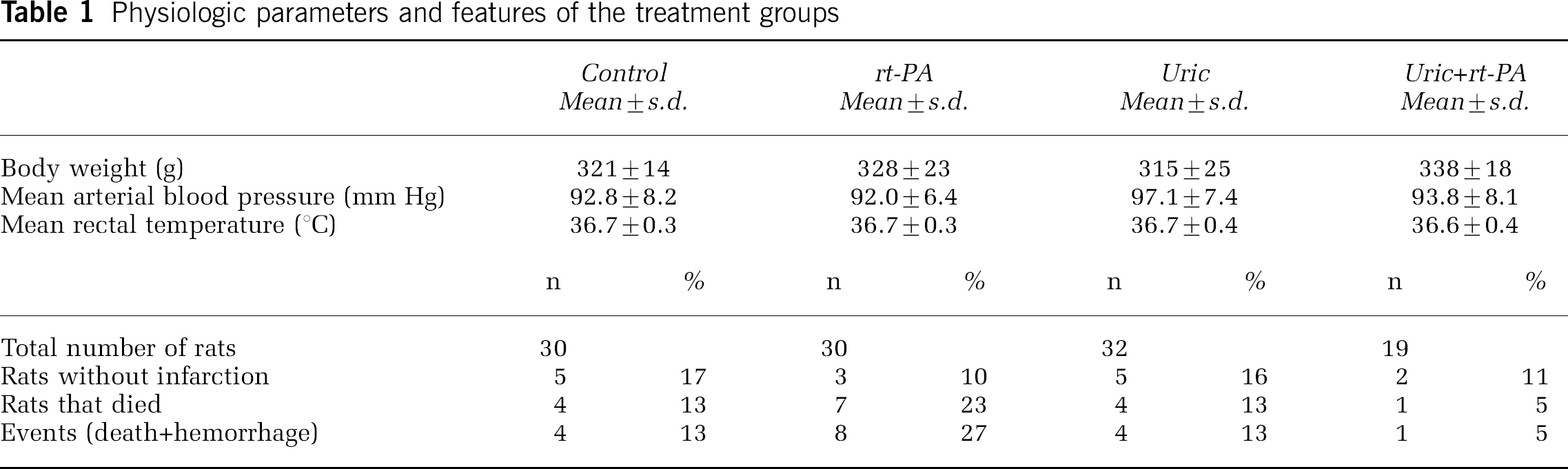
Uric acid reduced infarct volume by 55% in relation to controls (P < 0.05) (mean ± s.e.m.: 131.6 ± 31.8 mm3, in the uric acid group, n = 22 versus 294.8±46.0 mm3, in the controls, n = 21) (Figure 1A). One group of rats was treated with rt-PA alone and another group also received the vehicle of uric acid (Locke's buffer), to verify that the vehicle did not affect the response to rt-PA. Infarct volume (mean±s.e.m.) in rats receiving Locke + rt-PA was 104.6±32.7 mm3 (n =7), which was similar to the volume in rats receiving rt-PA alone (118.9±26.3 mm3; n = 13). These two groups were considered equally responsive to the rt-PA treatment, and were pooled together for statistical testing. Compared with controls, rt-PA reduced infarct volume (114.0 ± 17.4 mm3; n = 20) by 61% (P < 0.05) (Figure 1A). Nonetheless, infarct volume was further decreased in rats receiving uric acid + rt-PA (n = 16) (68.1±9.1 mm3), which showed an additional 17% reduction (P < 0,05) than animals receiving rt-PA alone (Figure 1A). Uric acid and rt-PA, either alone or in combination, reduced brain swelling significantly compared with controls (Figure 1B).
Additive beneficial effect of uric acid and recombinant tissue plasminogen activator (rt-PA) on infarct volume and brain edema. (
Treatments Improve the Neurologic Score
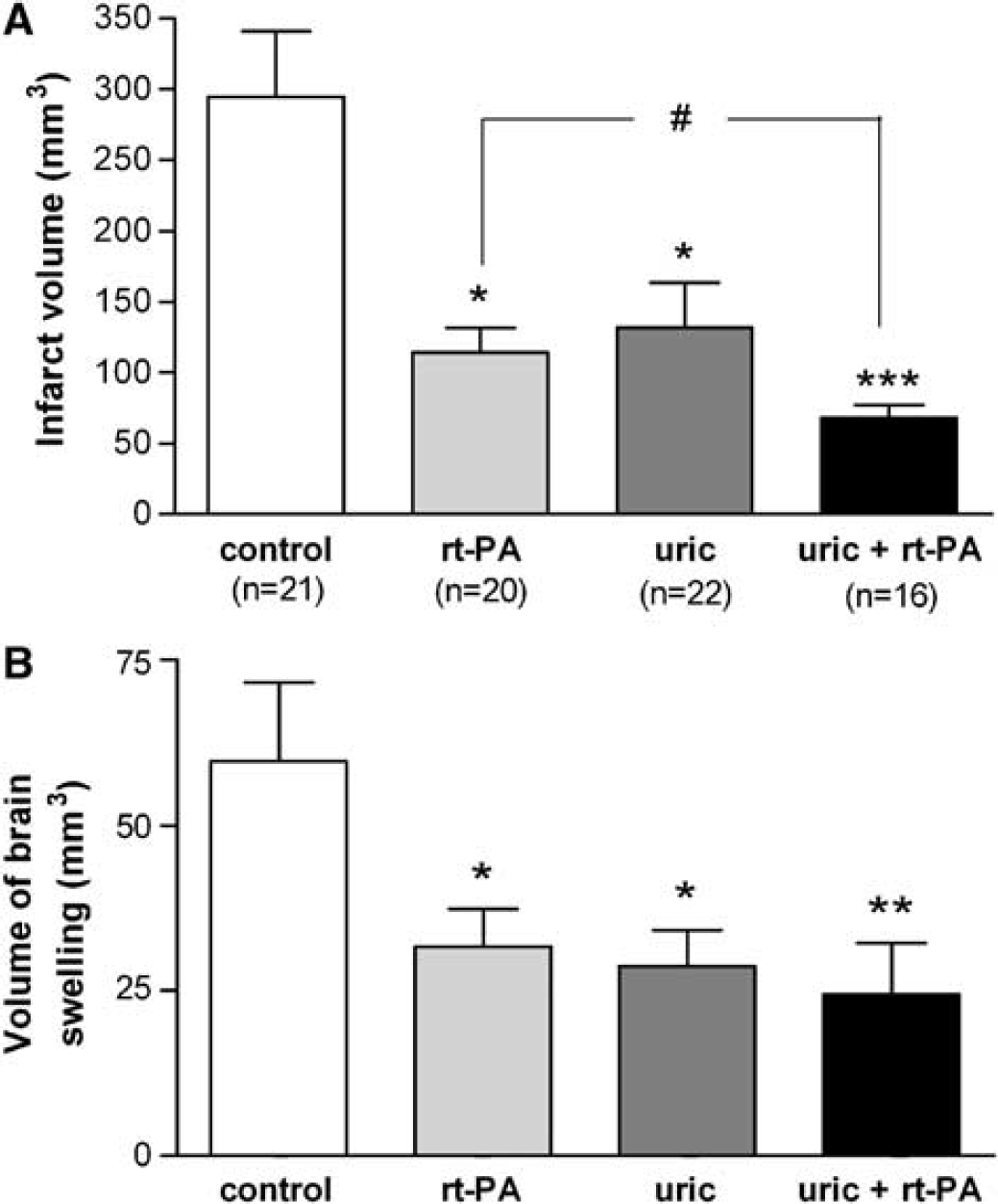
The neurologic score was positively correlated to infarct volume (r2 = 0.453, P < 0.0001) (Figure 2A), although the score was more variable for small than for large infarctions (Figure 2A). Nontreated rats (control) showed worsening of the neurologic function during the first 24 h (Figure 2B). Contrarily, rats receiving treatment (uric acid, rt-PA, or both) improved significantly (P < 0.05) their neurologic score from 30 mins to 24 h, in relation to controls.
Treatments ameliorate the neurologic score. (
Treatments Reduce Tyrosine Nitration in Ischemic Brain Tissue
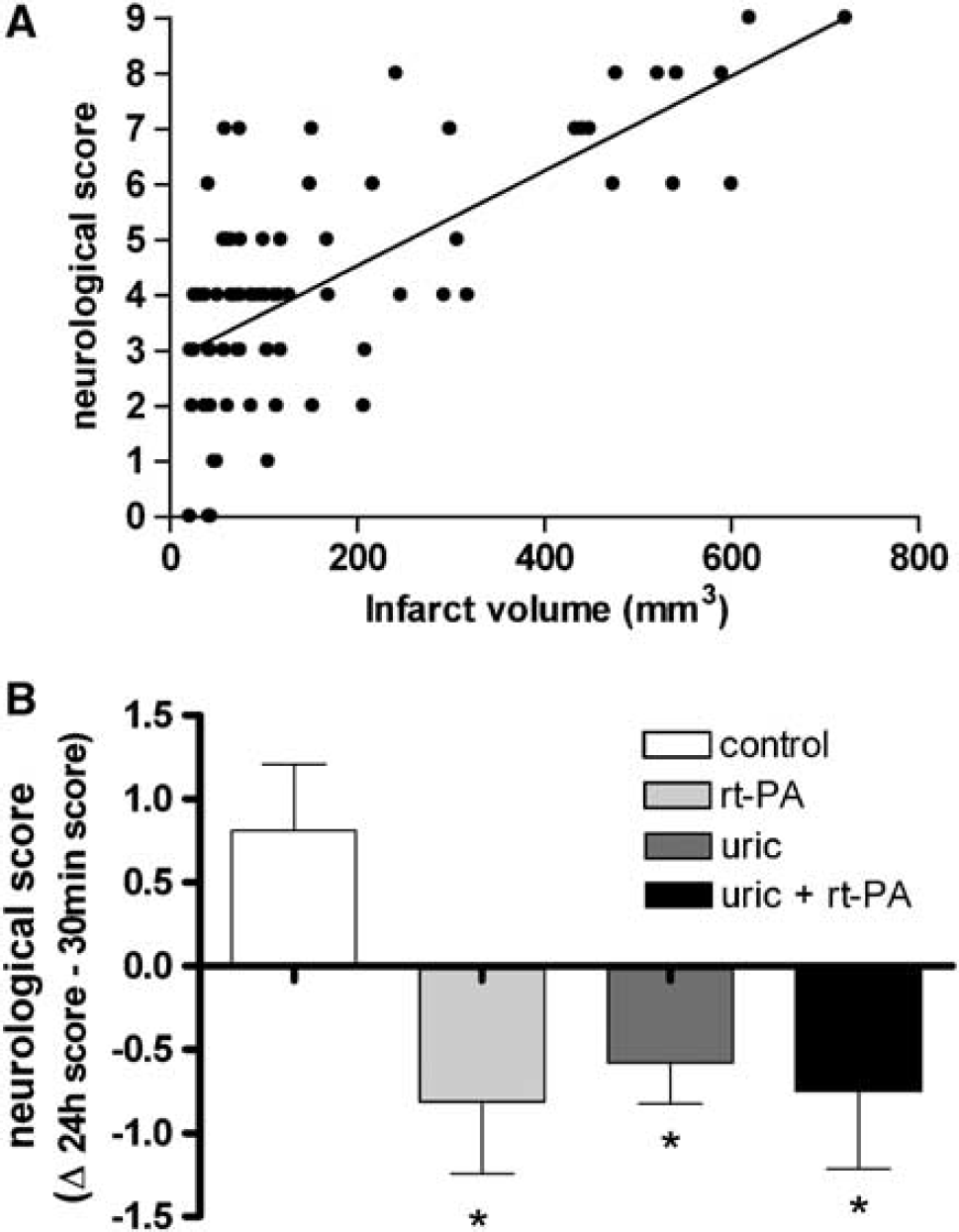
Ischemia induced tyrosine nitration at 24 h, whereas this effect was not observed in sham-operated rats or in the contralateral hemisphere (Figure 3A). Uric acid strongly prevented ischemia-induced protein tyrosine nitration (P < 0.01) (Figures 3B and 3D), whereas rt-PA only showed a tendency to attenuate tyrosine nitration versus nontreated ischemic rats (Figures 3C and 3D). This tendency became significant (P < 0.05) when rt-PA was combined with uric acid (Figures 3C and 3D).
Ischemia-induced brain tyrosine nitration is prevented by uric acid, and reduced by recombinant tissue plasminogen activator (rt-PA). (
Treatments Prevent Brain Neutrophil Infiltration
Ischemia induced neutrophil infiltration at 24 h, as assessed with MPO (Figure 4). Animals with large infarct volumes tended to show higher MPO in brain. Myeloperoxidase was not detected in sham-operated rats (Figure 4), or in the contralateral hemisphere. Recombinant tissue plasminogen activator (Figure 4A) and uric acid (Figure 4B) prevented the increase in MPO induced by ischemia. However, strong MPO was found in one rat of the rt-PA group with intracranial hemorrhage (Figure 4A).
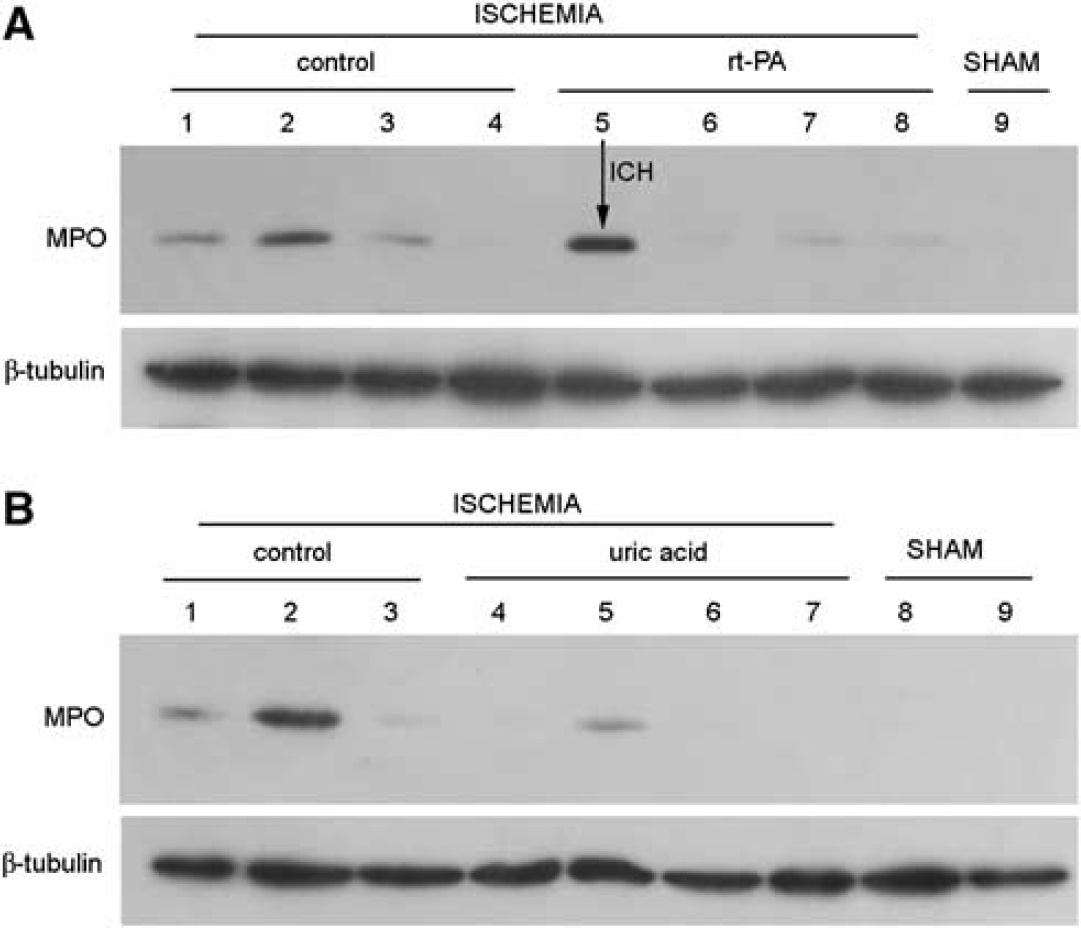
Neutrophil infiltration is reduced by recombinant tissue plasminogen activator (rt-PA) and by uric acid. (
Discussion
We report that uric acid exerts additive neuroprotection to the benefits of rt-PA in a thromboembolic model of stroke in the rat, when this natural antioxidant is administered early after the induction of ischemia. The study also shows that the smaller infarctions attained in animals treated with uric acid were paralleled by an improved animal behavior from the onset of treatment. Clinical improvement and smaller infarctions were also observed in animals treated with rt-PA, in relation to controls, suggesting that reperfusion and cytoprotection explained the main findings of the study.
Uric acid is the end product of purine metabolism and a powerful water-soluble antioxidant and radical scavenger in humans whose generation is increased in situations of oxidative stress, such as stroke (Becker, 1993). In cultured hippocampal neurons, uric acid suppressed the accumulation of reactive oxygen species and lipid peroxidation after exposure to glutamate or cyanide (Yu et al, 1998). In the ischemic rat brain, the administration of uric acid 1 h after reperfusion resulted in neuroprotection and it improved behavioral outcome in a model of intraluminal transient MCA occlusion (Yu et al, 1998). Also, uric acid protects against secondary damage after spinal cord injury by reducing tissue damage, nitrotyrosine formation, lipid peroxidation, activation of poly(ADP-ribose) polymerase, and neutrophil invasion (Scott et al, 2005). Here uric acid strongly reduced neutrophil infiltration in the ischemic brain, indicating that this treatment attenuates the acute inflammatory response induced by ischemia. Furthermore, thromboembolic ischemia caused protein tyrosine nitration. The formation of nitrotyrosine has been previously reported in a model of transient MCA occlusion/reperfusion, where it is believed to contribute to brain injury (Gursoy-Ozdemir et al, 2004). Tyrosine nitration is mediated by reactive nitrogen species, such as peroxynitrite, formed as secondary products of metabolism of the free radical nitric oxide in the presence of oxidants (Radi, 2004). Peroxinitrite-mediated tyrosine nitration is inhibited in vitro by uric acid (Squadrito et al, 2000). In the present study, treatment with uric acid abrogated tyrosine nitration in the ischemic brain, in agreement with the effect of this compound in spinal cord injury (Scott et al, 2005). It is feasible that tyrosine was gradually nitrated during the progression of ischemic brain damage, and that nitrotyrosine formation could further exacerbate injury through proinflammatory mechanisms. Treatment with uric acid prevents this effect and, thus, it might attenuate inflammation. Yet, the observation that uric acid alone was more effective to reduce nitrotyrosine than in combination with rt-PA suggests that reperfusion might further increase protein tyrosine nitration.
In the clinic, the consequences of elevated serum concentration of uric acid at the onset of stroke are controversial (Chamorro et al, 2002; Weir et al, 2003; Chamorro and Planas, 2004). Nevertheless, the studies that reported longitudinal changes in neurologic scores after the stroke found clinical benefit associated to increased levels of uric acid (Chamorro et al, 2002). Our current findings give strong support to the assessment in the clinic of the combined therapeutic effects of uric acid and rt-PA. Yet, a long list of neuroprotectants failed to show benefits in the clinic despite encouraging findings previously observed in the laboratory (Cheng et al, 2004). Several reasons have been put forward to explain the annoying discrepancy between bench and bed, including an excessive delay to treatment onset in most neuroprotection trials, lack of coadministration of a thrombolytic agent together with the putative neuroprotectant, or the use of preclinical models of brain ischemia and treatment protocols not relevant for the human disease (Fisher, 2003). To circumvent these caveats, current experimental designs include the coadministration of a neuroprotectant and a thrombolytic agent (Suzuki et al, 2003), and the development of more adequate animal models (Niessen et al, 2003). In the clinic, advances include the design of clinical trials in which the putative neuroprotectant is administered before hospital admission giving priority to the ‘time is brain’ concept (Saver et al, 2004). Our experimental design accords with the new approaches. However, our study has limitations including (a) the specific nature of the injected clots, which does not model the variety of clot composition in humans, and (b) the neurologic test used, which allows for crude discrimination in motor impairment, but does not involve fine sensorimotor or cognitive tasks.
In conclusion, our findings support that antioxidant uric acid combined with thrombolysis deserves further investigation.
Footnotes
Acknowledgements
We thank Ms Noelia Montoya and Ms Eugenia Gomez for skilful technical assistance.
