Abstract
The authors evaluated the therapeutic efficacy of tacrolimus (FK506), administered alone or in combination with recombinant tissue plasminogen activator (t-PA), on brain infarction following thrombotic middle cerebral artery (MCA) occlusion. Thrombotic occlusion of the MCA was induced by a photochemical reaction between rose bengal and green light in Sprague-Dawley rats, and the volume of ischemic brain damage was determined 24 hours later. Intravenous administration of tacrolimus or t-PA dose-dependently reduced the volume of ischemic brain infarction, whether administered immediately or 1 hour after MCA occlusion. When tacrolimus or t-PA was administered 2 hours after MCA occlusion, each drug showed a tendency to reduce ischemic brain damage. However, combined treatment with both drugs resulted in a significant reduction in ischemic brain damage. On administration 3 hours after MCA occlusion, tacrolimus alone showed no effect, and t-PA tended to worsen ischemic brain damage. However, the combined treatment with both drugs not only ameliorated the worsening trend seen with t-PA alone, but also tended to reduce ischemic brain damage. In conclusion, tacrolimus, used in combination with t-PA, augmented therapeutic efficacy on brain damage associated with focal ischemia and extended the therapeutic time window compared to single-drug treatments.
Keywords
Stroke is the third leading cause of death in developed countries and leads to significant loss of the quality of life worldwide (Hankey and Warlow, 1999). In most acute ischemic strokes, the underlying cause is a thrombotic or embolic vascular occlusion (Warlow, 1998). Therefore, one strategy for treating stroke is to restore blood flow within the ischemic area by using a thrombolytic agent. Several clinical trials have shown that recombinant tissue plasminogen activator (t-PA) is effective in improving clinical outcomes at 3 months, as long as the patients receive treatment within 3 hours after the acute onset of stroke (Hacke et al., 1995; N.I.N.D.S. rt-PA Stroke Study Group, 1995). However, t-PA is only effective in a limited number of those patients, has a short therapeutic time window for administration (3 hours), and carries a risk of promoting intracerebral hemorrhage. Therefore, there are significant clinical demands for neuroprotective drugs that can ameliorate neuronal death associated with stroke or extend the therapeutic time window in the ischemic penumbra (Kaste, 2001). Combination therapy using t-PA and a neuroprotective agent may achieve better outcomes than using either drug alone.
Tacrolimus (FK506, Prograf; Fujisawa Pharmaceutical, Osaka, Japan) is an immunosuppressant used clinically to prevent allograft rejection. In addition, recent studies have shown that tacrolimus has neuroprotective activity in animal models of cerebral ischemia. After focal cerebral ischemia induced by middle cerebral artery (MCA) occlusion, tacrolimus reduced ischemic injury (Sharkey and Butcher, 1994) and ameliorated neurologic deficits (Sharkey et al., 1996). Tacrolimus also protected neurons against ischemic death after global cerebral ischemia (Yagita et al., 1996). In the present study, we evaluated the therapeutic potential of tacrolimus and t-PA, both alone and in combination, in a photochemically induced thrombotic MCA occlusion model. In this model, generation of molecular oxygen by irradiation of rose bengal leads to injury of vascular endothelial cells, with subsequent thrombus formation at the site of injury (Matsuno et al., 1993; Umemura et al., 1993). Since t-PA has been reported to ameliorate ischemic brain damage in this model (Lekieffre et al., 1997; Sereghy et al., 1993), we used this model to test the hypothesis that combination therapy using t-PA with a neuroprotective agent would achieve better outcomes than using either drug alone.
MATERIALS AND METHODS
Animals
Experiments were carried out using 8- to 10-week-old male Sprague-Dawley rats purchased from Japan SLC (Shizuoka, Japan). Animals were housed at 23°C ± 2°C, 55% ± 5% humidity, under a 12-hour light/dark cycle (lights on at 7:00) for at least 1 week before use and were allowed free access to food and water. Experiments were conducted according to the guidelines of the Laboratory Animal Experimental Committee of Fujisawa Pharmaceutical Co., Ltd.
Photochemically induced thrombotic focal ischemia model in rats
Focal cerebral ischemia was induced photochemically according to the method of Matsuno et al. (1993). Rats were anesthetized with halothane (4% for induction and 1.5% for maintenance) in room air, and the right femoral vein was cannulated with a polyethylene tube (PE50) for administration of rose bengal or test drugs. The scalp and the left temporalis muscle were cut and, under an operating microscope, a sub-temporal craniotomy was performed using a dental drill. A window (about 4 mm in diameter) was opened just anterior to the foramen ovale of the mandibular nerve at the skull base and the main trunk of the left MCA was identified through the dura mater. Photoillumination with green light (wavelength, 540 nm) was accomplished by using a xenon lamp (L-4887, Hamamatsu Photonics, Hamamatsu, Japan) with a heat-absorbing filter and a green filter. While rose bengal (10 mg/kg) was injected intravenously over 1 minute, photoillumination was directed to the MCA for 10 minutes by an optic fiber (3 mm in diameter) mounted on the window at the skull base. Complete photothrombotic occlusion of MCA was visually confirmed through the operating microscope and was ascertained by the decrease in regional cerebral blood flow (rCBF) monitored by laser-Doppler flowmetry. After closure of the wound, rats were returned to their cages. During surgery and rCBF monitoring, rectal temperature was maintained at 37.4°C to 38.6°C with a heating-pad (TR-100, PS-100, Fine Science Tools, North Vancouver, Canada).
Measurement of regional cerebral blood flow
Regional cerebral blood flow (rCBF) in the main trunk of the MCA was monitored by laser-Doppler flowmetry (Omegaflow, FLO-N1, Neuroscience, Osaka, Japan) for 3 or 5 hours after MCA occlusion. A laser-Doppler probe was placed on the dura mater 3 mm distal to the edge of the thrombus. Changes in rCBF were expressed as a percentage of the baseline value.
Volume of ischemic brain infarction
At 24 hours after MCA occlusion, rats were killed and their brains were removed. The cerebrum was coronally sectioned (2-mm thick) using a microslicer from 4 to −6 mm from bregma, and six consecutive slices were stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC; Katayama, Osaka, Japan) at 37°C for 30 minutes. Photographs of the coronal sections were used to calculate the area of ischemic brain damage and the whole area of the cerebrum using a computerized image analysis system. The ischemic brain damage in each animal was expressed as a percentage of the sum of the damaged area compared to the sum of the whole area of the cerebrum, and the volume of ischemic brain damage was calculated according to the trapezoid method (Rosen and Harry, 1990).
Measurement of physiologic parameters
Arterial blood pressure, heart rate (AP-601G, AT-601G, Nihon Kohden, Tokyo, Japan), P
Drugs
The injection formulation of tacrolimus synthesized at Fujisawa Pharmaceutical Co., Ltd. (Osaka, Japan) was diluted with physiologic saline and administered intravenously. For the control group, placebo ampoules synthesized at Fujisawa Pharmaceutical were similarly diluted with physiologic saline and administered. Recombinant t-PA (Alteplase, Activacin; Kyowa Hakko, Tokyo, Japan) was dissolved in distilled water and then diluted with physiologic saline. Twenty percent of the total dose of t-PA was intravenously injected as a bolus, followed by continuous infusion of the remaining 80% over a period of 30 minutes. Physiologic saline was administered to rats in the control group of t-PA experiments. The injection volume was adjusted to 2 mL/kg in all experiments.
Statistical analysis
All values are expressed as mean ± SD. The significance of any difference between groups for ischemic brain damage, rCBF, and physiologic parameters was tested using analysis of variance followed by the Dunnett test. A P value less than 0.05 was considered significant.
RESULTS
Effect of tacrolimus and t-PA on physiologic parameters
Table 1 shows physiologic parameters measured at baseline and 2 and 24 hours after MCA occlusion. Although the blood glucose concentration increased 2 hours after MCA occlusion in all groups, none of the changes was statistically significant. Drugs were administered immediately after MCA occlusion. Administration of tacrolimus (1 mg/kg) did not affect any parameters. Although hematocrit and hemoglobin in the t-PA group decreased slightly at 24 hours after MCA occlusion, these values did not change in the tacrolimus and t-PA combination group. Other parameters including mean arterial pressure, heart rate, P
Effects of tacrolimus and tissue plasminogen activator on physiologic parameters after thrombotic middle cerebral artery occlusion in rats
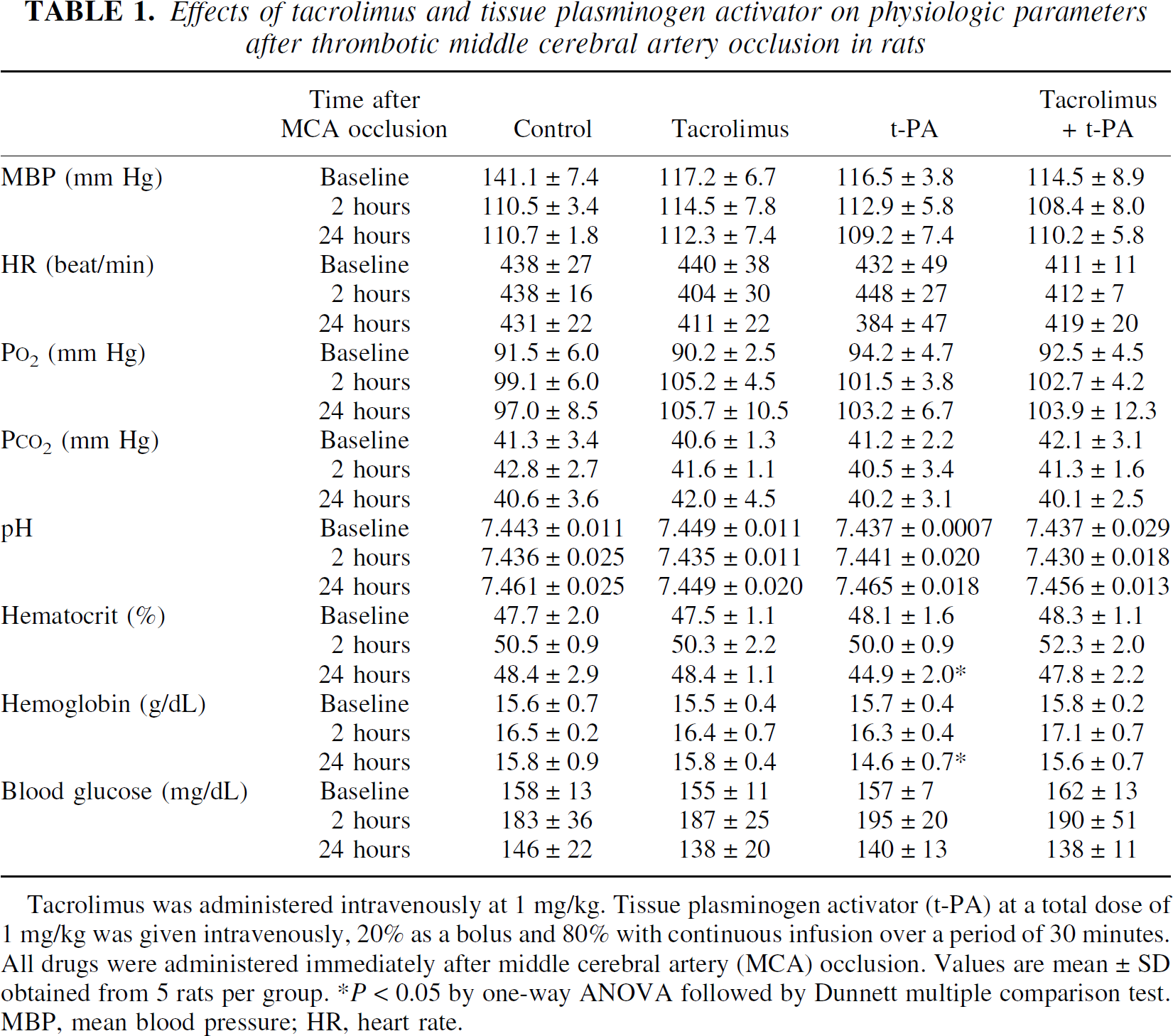
Tacrolimus was administered intravenously at 1 mg/kg. Tissue plasminogen activator (t-PA) at a total dose of 1 mg/kg was given intravenously, 20% as a bolus and 80% with continuous infusion over a period of 30 minutes. All drugs were administered immediately after middle cerebral artery (MCA) occlusion. Values are mean ± SD obtained from 5 rats per group. * P < 0.05 by one-way ANOVA followed by Dunnett multiple comparison test. MBP, mean blood pressure; HR, heart rate.
Effect of tacrolimus or t-PA on ischemic damage after thrombotic focal cerebral ischemia
The dorsolateral cortex and basal ganglia developed infarction after MCA occlusion in the control group. The total volume of ischemic brain damage in the tacrolimus vehicle-treated group was comparable with that in the saline-treated group (202.84 ± 51.65 mm3, n = 11, vs 225.46 ± 42.12 mm3, n = 7, respectively). As shown in Fig. 1A, tacrolimus dose-dependently reduced the size of ischemic brain infarction in the cortex, with a significant effect (24% reduction in ischemic volume) at 1 mg/kg. Administration of t-PA reduced ischemic brain infarction, both in the cortex and striatum, with a maximum effect of almost 50% reduction of the cortical damaged area (Fig. 1B). Intracerebral hemorrhage was not detected in t-PA treated animals.
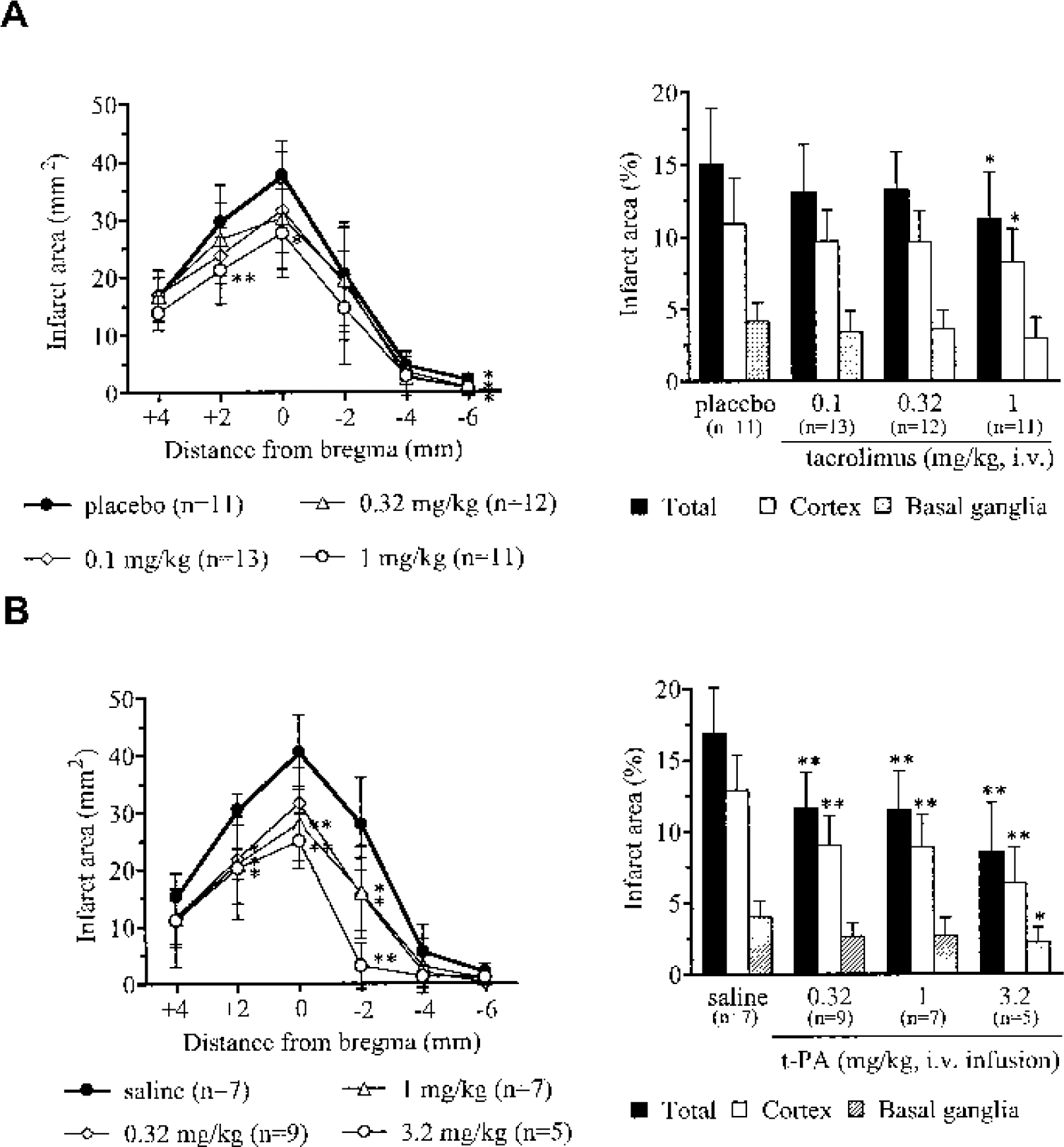
Neuroprotective effects of tacrolimus
Effect of tacrolimus or t-PA on regional cerebral blood flow after thrombotic focal cerebral ischemia
Cortical rCBF decreased within 4 to 9 minutes after the initiation of photoillumination, demonstrating rapid formation of thrombus following the photochemical reaction. In the control group, rCBF decreased to about 20% of the baseline level and remained diminished for at least 3 hours without signs of recanalization. When tacrolimus or t-PA was administered intravenously at the end of photoillumination, tacrolimus (1 mg/kg) alone did not alter rCBF (Fig. 2A). In contrast, intravenous infusion of t-PA (1 mg/kg) alone restored rCBF to 70% of the baseline level (Fig. 2B). Increasing doses of t-PA resulted in more rapid recanalization of the occluded MCA.
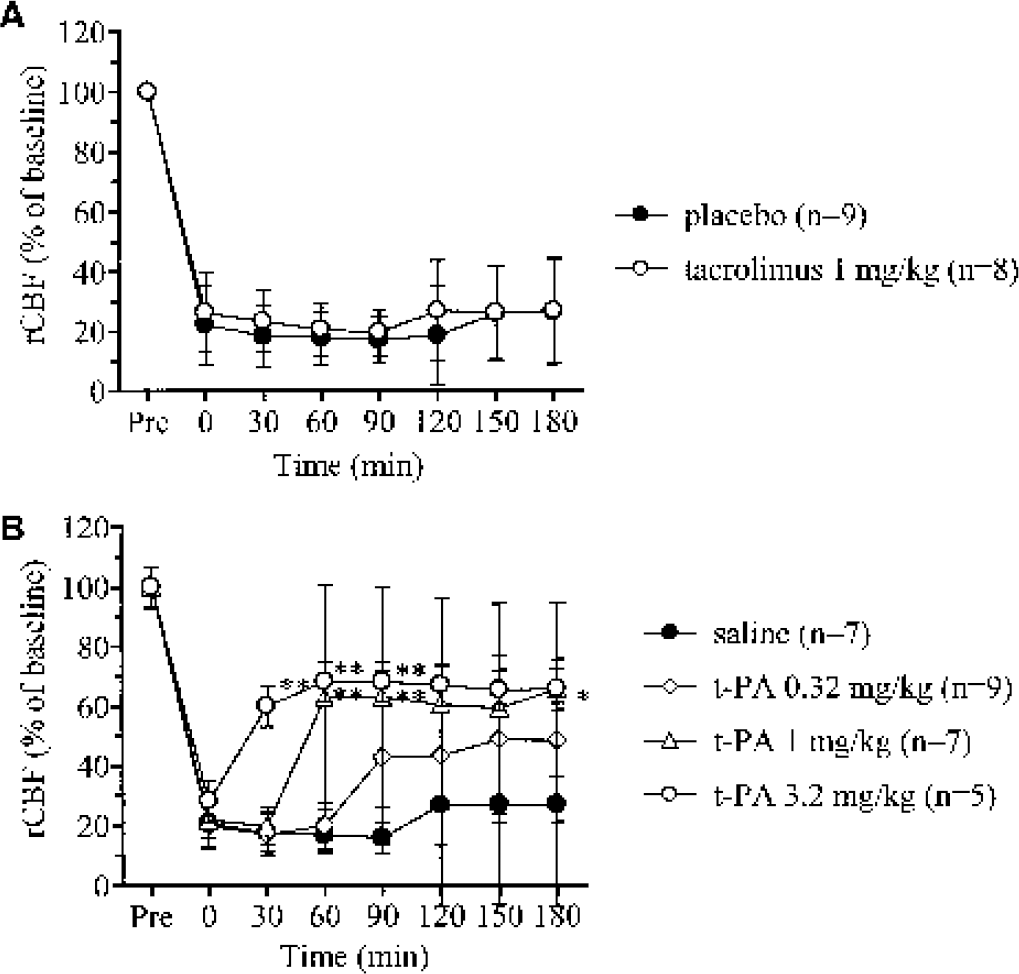
Effects of tacrolimus and tissue plasminogen activator (t-PA) on regional cerebral blood flow (rCBF) induced by photochemical middle cerebral artery occlusion in rats.
Effect of a combined treatment with tacrolimus and t-PA on ischemic damage after thrombotic focal cerebral ischemia
In the control group, there was ischemic damage in the dorsolateral cortex and basal ganglia. The volume of ischemic brain infarction in the control group of each experiment with delayed treatment of 0, 1, 2, and 3 hours after MCA occlusion was 267.47 ± 74.84, 247.40 ± 45.29, 197.12 ± 38.00, and 208.68 ± 72.29 mm3, respectively. The administration of tacrolimus or t-PA immediately or 1 hour after MCA occlusion significantly reduced the infarcted brain area (Fig. 3). The combined treatment with tacrolimus and t-PA reduced ischemic brain damage more effectively when given immediately after MCA occlusion. Both drugs, when administered alone 2 hours after MCA occlusion, tended to reduce ischemic brain damage, but the changes did not reach statistical significance. In contrast, the combined tacrolimus and t-PA treatment reduced ischemic infarction significantly, even when the combination was given 2 hours after MCA occlusion. After a 3-hour delay in administration, tacrolimus showed a minimal effect and t-PA tended to worsen ischemic brain damage. Combination treatment ameliorated the worsening trend seen with t-PA alone and even tended to reduce ischemic brain damage, although the difference was not statistically significant (Fig. 3). No sign of intracerebral hemorrhage was seen after MCA occlusion in any group.
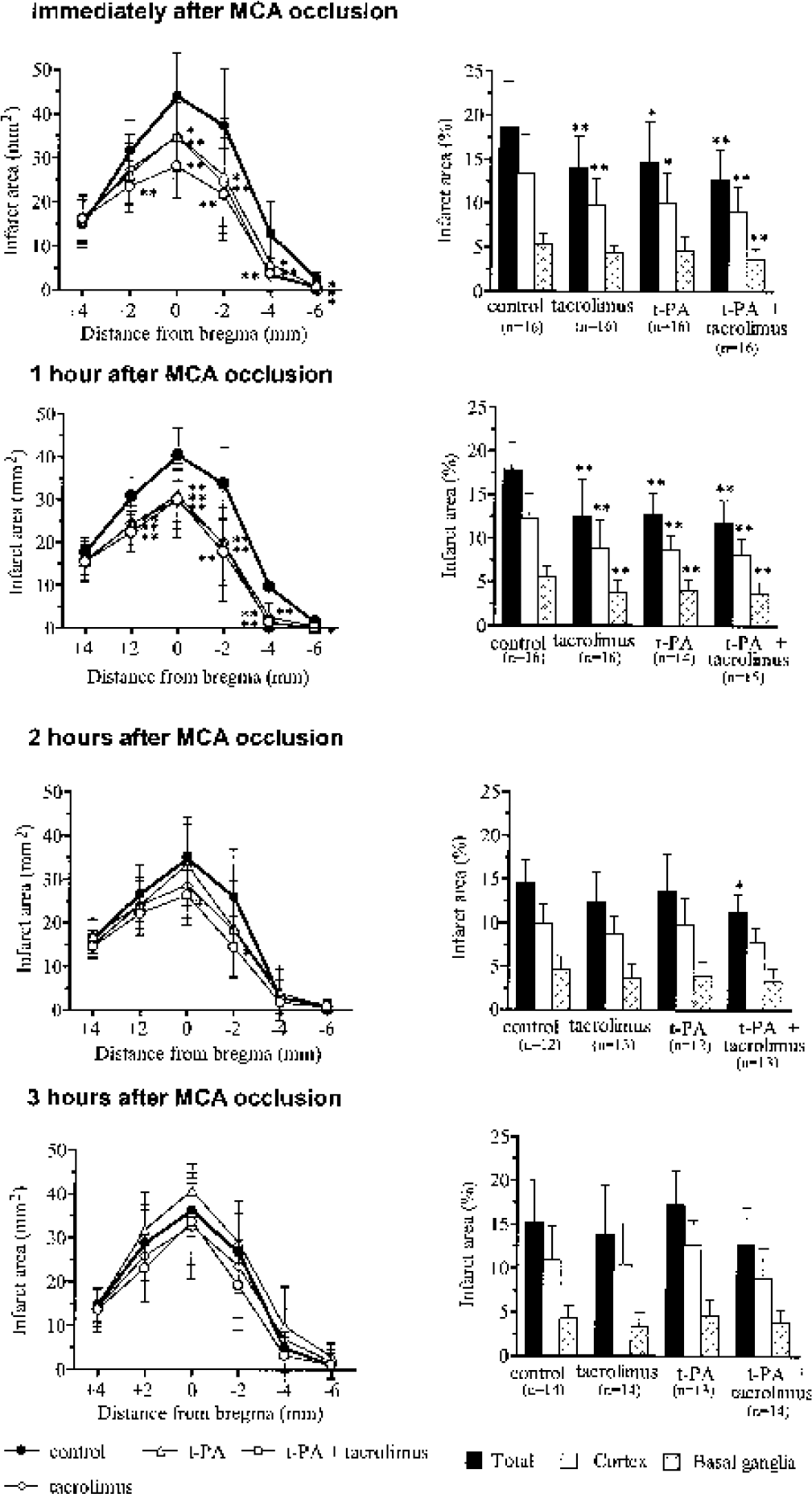
Effects of a combined treatment with tacrolimus (1 mg/kg) and tissue plasminogen activator (t-PA; 1 mg/kg) on ischemic brain infarction in the thrombotic middle cerebral artery (MCA) occlusion model in rats. Values in parentheses are the number of animals. Data are presented as mean ± SD. *P < 0.05 and **P < 0.01 by one-way ANOVA followed by Dunnett multiple comparison test.
Effect of a combined treatment with tacrolimus and t-PA on regional cerebral blood flow after thrombotic focal cerebral ischemia
To clarify whether tacrolimus mediated its neuroprotective effect by altering rCBF, rCBF was measured in animals treated with tacrolimus and/or t-PA 2 hours after MCA occlusion. Tacrolimus did not alter rCBF when administered 2 hours after MCA occlusion. In contrast, t-PA gradually restored rCBF, which reached 70% of the baseline level. Similarly, combined treatment administered 2 hours after MCA occlusion also gradually restored rCBF, indicating that tacrolimus did not alter the ability of t-PA to restore rCBF (Fig. 4).
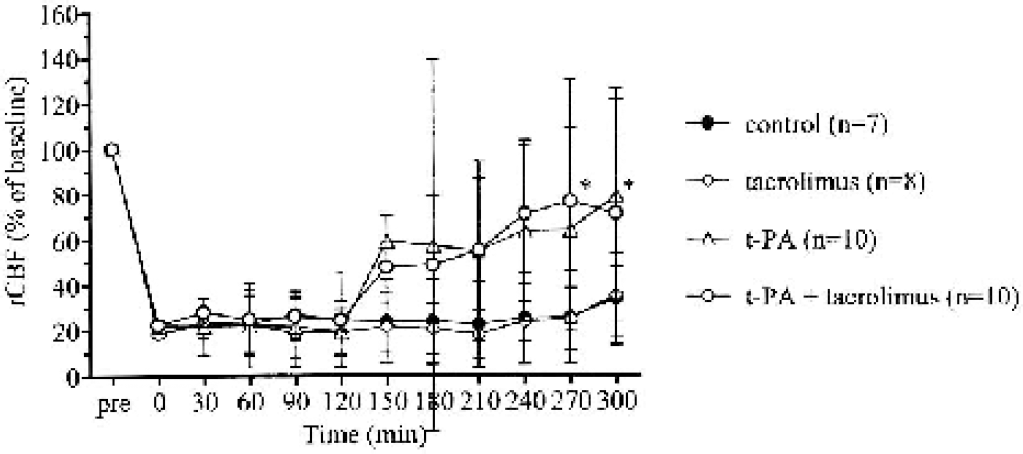
Effects of a combined treatment with tacrolimus and tissue plasminogen activator (t-PA) on regional cerebral blood flow induced by photochemical middle cerebral artery occlusion in rats. Tacrolimus (1 mg/kg) and t-PA (1 mg/kg) were administered alone or in combination 2 hours after occlusion. Values in parentheses are the number of animals. Data are presented as mean ± SD. *P < 0.05 by one-way ANOVA followed by Dunnett multiple comparison test.
DISCUSSION
A photochemical reaction between rose bengal and green light causes vascular endothelial injury, inducing a chain reaction of platelet adhesion and aggregation and leading to the formation of a platelet- and fibrin-rich thrombus at the reaction site (Kawai et al., 1995). Although tacrolimus has consistently been shown to be neuroprotective in animal models of cerebral ischemia (Sharkey et al., 2000), few reports have shown its efficacy in a thrombotic MCA occlusion model. Although Takamatsu et al. (1998) demonstrated the effect of tacrolimus at a dose of 0.32 mg/kg in this model, they did not observe a dose-dependent effect or an effect on rCBF. In the present study, we found that tacrolimus showed a dose-dependent reduction of ischemic brain infarction without affecting rCBF, indicating that tacrolimus exerted its beneficial effect via neuroprotective activity.
In contrast, t-PA dose-dependently restored rCBF, with increasing doses of t-PA resulting in more rapid recanalization of the occluded MCA. Furthermore, t-PA dramatically reduced ischemic brain damage in the cortex in parallel with the restoration of rCBF, indicating that the beneficial effect of t-PA was due to its fibrinolytic activity, as shown by the disappearance of the clot at the time when the tissue could still be salvaged. A dose of 3.2 mg/kg t-PA reduced ischemic brain damage not only in the cortex, but also in the striatum. Although 3.2 mg/kg t-PA rapidly recanalized the occluded MCA, rCBF restored by t-PA was still lower than rCBF before occlusion, indicating that hypoperfusion in MCA perfusion area persisted for at least 3 hours after recanalization. This finding might explain why t-PA treatment showed only a 50% reduction of infarction volume. Although the maximum efficacy of t-PA in reducing the size of ischemic brain damage was greater than that of tacrolimus in this model, this is not surprising because t-PA exerted the beneficial effect through its thrombolytic action. It is unlikely that t-PA possesses neuroprotective activity since there are no reports demonstrating the efficacy of t-PA in cerebral ischemia models other than thrombotic or embolic occlusion models. In fact, we previously observed that t-PA had minimal effects in the permanent MCA occlusion model by electrocoagulation in rats (Furuichi et al., unpublished observation).
The rationale for combination therapy was based on recent data concerning the pathophysiologic mechanisms of ischemic brain damage. Given the complex mechanisms involved in the cascade leading to ischemic brain damage, it seems unlikely that any single therapeutic intervention to modulate the entire cascade (Kaste, 2001). Thrombolytic agents can rescue neurons in the ischemic penumbra by means of recanalization. However, the presence of the ischemic penumbra is dependent on the duration of ischemia (Hossmann, 1994). The longer the hypoperfusion state exists, the higher the residual flow that is needed to promote neuronal survival. Although t-PA has been clinically used for patients with acute stroke, its efficacy is limited and the number of treatable patients is restricted because of its short 3-hour therapeutic time window. We therefore hypothesized that the combination therapy using tacrolimus and t-PA may extend the therapeutic time window.
Tacrolimus showed a neuroprotective effect when it was administered immediately or 1 hour after MCA occlusion, but not when it was administered 2 hours after MCA occlusion. These results indicate that the therapeutic time window of tacrolimus in this model was more than 1 hour but less than 2 hours. Although t-PA at a dose of 1 mg/kg showed the same therapeutic time window as tacrolimus, it is noteworthy that t-PA tended to worsen ischemic brain damage when it was administered 3 hours after MCA occlusion. When t-PA at a dose of 10 mg/kg was administered 3 or 6 hours after MCA occlusion, the size of ischemic brain damage was significantly worsened, even though intracerebral hemorrhage was not detected by visual inspection (data not shown). These results indicate that administration of t-PA 3 hours or longer after MCA occlusion induces harmful events. Administration of t-PA 2 hours after MCA occlusion has been reported to dramatically increase the size of cerebral infarction in wild-type and t-PA-deficient mice (Wang et al., 1998), possibly via plasmin-catalyzed degradation of laminin, an extracellular matrix protein (Chen and Strickland, 1997). In addition, administration of t-PA 4 hours after onset of embolic stroke in rats significantly increased the vascular immunoreactivity of adhesion molecules in the ischemic lesion, implying that t-PA has a direct effect on the expression of adhesion molecules and tends to increase ischemic brain damage (Zhang et al., 1999). These results suggest that the progression of ischemic brain damage in our thrombotic MCA occlusion model is slightly faster than that in the clinical setting and embolic stroke model in rats.
Interestingly, combination therapy using tacrolimus and t-PA significantly reduced brain damage when administered 2 hours after MCA occlusion, and tended to ameliorate the worsening of brain damage when administered 3 hours after MCA occlusion. These results support the hypothesis that the combination of thrombolysis with a neuroprotective agent could decrease or eliminate reperfusion injury. Combination therapy using t-PA and a neuroprotective agent has recently been attempted in embolic stroke models, and encouraging results have been reported (Andersen et al., 1999; Asahi et al., 2000; Zhang et al., 1999). Coadministration of t-PA and aspirin, however, reportedly increase the risk of intracranial hemorrhage in rat and rabbit models of stroke (Clark et al., 1991; Overgaard et al., 1992). In embolic occlusion models, improvement of the neuroprotective activity by combination therapy using t-PA and the glutamate receptor antagonist dizocilpine (Sereghy et al., 1993; Zivin and Mazzarella, 1991) or eliprodil (Lekieffre et al., 1997) has also been reported.
These previous studies, however, did not evaluate the therapeutic time window for combination therapy. In the present study, we found that tacrolimus provided excellent neuroprotection when co-administered with t-PA, particularly in extending the therapeutic time window. Although tacrolimus did not alter the ability of t-PA to restore rCBF, combination treatment significantly reduced the infarction size even though t-PA alone could not. This result suggests that tacrolimus might suppress reperfusion injury resulting from delayed recanalization. The mechanism by which tacrolimus suppresses reperfusion injury presumably involves the suppression of inflammatory responses, since tacrolimus has been shown to attenuate the infiltration of inflammatory cells (Tsujikawa et al., 1998) and suppress expression of ICAM-1 after reperfusion (Wakabayashi et al., 1994).
In conclusion, the present study demonstrated that neuroprotection by tacrolimus was effective in reducing the consequences of MCA thrombosis, and that higher therapeutic efficacy and a longer therapeutic time window were achieved when it was administered in combination with t-PA. Therefore, a combined neuroprotective and thrombolytic therapy may be a valuable approach for treatment of acute stroke in humans.
Footnotes
Acknowledgements
The authors thank Dr. Raymond D. Price for his critical review of the manuscript.
