Abstract
Positron emission tomography scans of patients with head injuries often show discrete areas of increased 18F-fluorodeoxyglucose uptake (“hot spots”) when performed hours to days after the initial ictus. Using quantitative autoradiographic methods, the authors have investigated whether cerebral blood flow and glucose metabolism are uncoupled 2 hours after controlled head injury in an animal model, and whether any “hot spots” are accompanied by changes in cerebral glucose concentration. Experiments were performed on 18 anesthetized, ventilated (1.5% halothane in 2:1 nitrous oxide:oxygen) Sprague-Dawley rats weighing 300 to 330 g. A burr hole was made over the left parietal cortex, and all animals received a piston impact on the intact dura (2 mm in diameter, 2.0 m/sec, 2 mm in depth). All animals remained anesthetized and ventilated for a further 2 hours, after which quantitative autoradiography was used to determine either (1) local cerebral blood flow (LCBF) using 14C-iodoantipyrine, (2) local cerebral glucose utilization (LCGU) using 14C-deoxyglucose, or (3) local cerebral glucose content (LCGC) using 14C-methylglucose. Local CBF, LCGU, and LCGC were measured in five regions adjacent to the contusion, and values then were normalized on the contralateral cortex. Normalized LCBF, LCGU, or LCGC varied in parallel in ipsilateral cortex (no change) and in the ischemic core of the contusion (reduced). However, there were marked changes in the patterns observed in the boundary zone (within 1 mm of the contusion). In all six rats used for LCGU measurement, there were discrete areas of high metabolism, whereas in all six rats used for LCBF measurement, flow was universally depressed in the boundary zone. Of the six rats used for LCGC determination, there was a discrete area of high signal in only one. The authors conclude that there are discrete areas of uncoupling of cerebral blood flow and metabolism after head injury within 2 hours of cerebral contusion in the rat that cannot be explained by changes in cerebral glucose content in the majority of animals.
Keywords
Head injury in humans and animals is often associated with temporal and spatial variations in cerebral metabolism. Studies in humans show that there is a dissociation between regional cerebral metabolic rate of glucose utilization (CMRglu) measured using positron emission tomography (PET) and global cerebral metabolic rate of oxygen (CMRO2), suggesting hyperglycolysis (Bergsneider et al., 1997). More recent studies have confirmed a marked heterogeneity in regional CBF, CMRglu, and CMRO2, all measured using PET techniques (Menon et al., 1999). Early after head injury (<24 hours), this increase in 18F-FDG uptake is associated with hypoperfusion, and later with normo-or hyperperfusion. Positron emission tomography scans are usually performed some time (at least 4 hours) after the initial ictus. A number of mechanisms have been proposed for such FDG “hot spots,” including hyperglycolysis provoked by subconvulsive seizures or glutamate release. In a rat model of head injury, cerebral blood flow and glucose metabolism are uncoupled within 30 minutes (Ginsberg et al., 1997); and in another study, early hypermetabolism (30 minutes postinjury) is followed by a depressed metabolic state 6 hours postinjury (Yoshino et al., 1991). Interestingly, there appears to be a correlation between the duration of metabolic depression and behavioral deficits in the rat (Moore et al., 2000), whereas in humans, there appears to be no correlation between CMRglu and the level of consciousness (Bergsneider et al., 1999).
The authors have investigated cerebral blood flow and metabolism using quantitative autoradiographic methods 2 hours after controlled head injury in an animal model to assess whether metabolic “hot spots” are a reproducible phenomenon, and whether the assumptions required for the use of Sokoloff's equation are valid. Two hours is clinically relevant as a time point before PET data can be obtained from patients with head injuries, and where disturbances in metabolism are because of cerebral damage rather than subsequent inflammation.
MATERIALS AND METHODS
Experiments were performed under the U.K. Animals (Scientific Procedures) Act, 1986. Eighteen male Sprague-Dawley rats (weighing 300 to 330 gm) were anesthetized and ventilated with 1.5% halothane in 2:1 nitrous oxide:oxygen. Arterial blood gases were measured and when found to be outside the ranges 37 to 43 mm Hg Pco2 and 80 to 95 mm Hg Po2, the ventilation parameters adjusted and resampled 10 minutes later. If blood gases were still outside these limits, the animals were not used. A burr hole was made over the left parietal cortex to expose the dura. All animals received an impact on the intact dura using a computer-controlled piston (2 mm in diameter) at a speed of 2.0 m/sec to a depth of 2 mm. The piston tip used was flat, but with the rounded edges. These parameters were chosen to produce a discrete area of damage with minimal disruption to the hippocampus and no disruption to the underlying caudate-putamen. All animals remained anesthetized and ventilated for a further 2 hours. Quantitative autoradiography was used to determine the following: (1) local cerebral blood flow (LCBF) using 14C-iodoantipyrine (Sakurada et al., 1978); (2) local cerebral glucose utilization (LCGU) using 14C-deoxyglucose (Sokoloff et al., 1977); or (3) local cerebral glucose content (LCGC) using 14C-methylglucose (Gjedde and Diemer, 1983).
All methods were fully quantitative, with tissue isotope concentration measured by densitometry and arterial isotope concentrations determined by liquid scintillation counting. Local CBF and LCGU were then calculated using the appropriate operational equation. Local CGC was estimated by dividing the cerebral tissue isotope concentration measured 30 minutes after injection by the integral of the plasma concentration.
Local CBF, LCGU, and LCGC were measured in four regions in the cortex ipsilateral to the contusion—the center of the contusion, the boundary zone (1 mm from the center), and 2 areas of cortex 4 mm either side of the contusion. All values then were normalized on values measured in the contralateral cortex. Comparisons among the three groups were made using one-way analysis of variance.
RESULTS
There was no significant difference between normalized LCBF, LCGU, or LCGC in the ipsilateral cortex (no change) nor in the ischemic core of the contusion (all reduced). However, there were marked changes in the patterns observed in the boundary (within 1 mm of the contusion) (Table 1). In all six rats used for LCGU measurement, there were discrete areas in the boundary zone of the contusion (approximately 1 mm from the central core) of high metabolism (more than 2 standard deviations greater than values in the ipsilateral cortex). All brain sections taken were sagittal, and the areas of high signal were observed lateral to the contusion. In five of the six rats, areas of high signal were observed on both sides of the lesion (Fig. 1), and in the sixth rat, a single area was seen toward the midline only. When normalized on the cortex in the contralateral hemisphere, the values obtained varied from 131% and 264% (mean 174%). In the six rats used for LCBF measurement, the corresponding values were all less than 46% (mean 18%). These data indicate dissociation between flow and metabolism in this area.
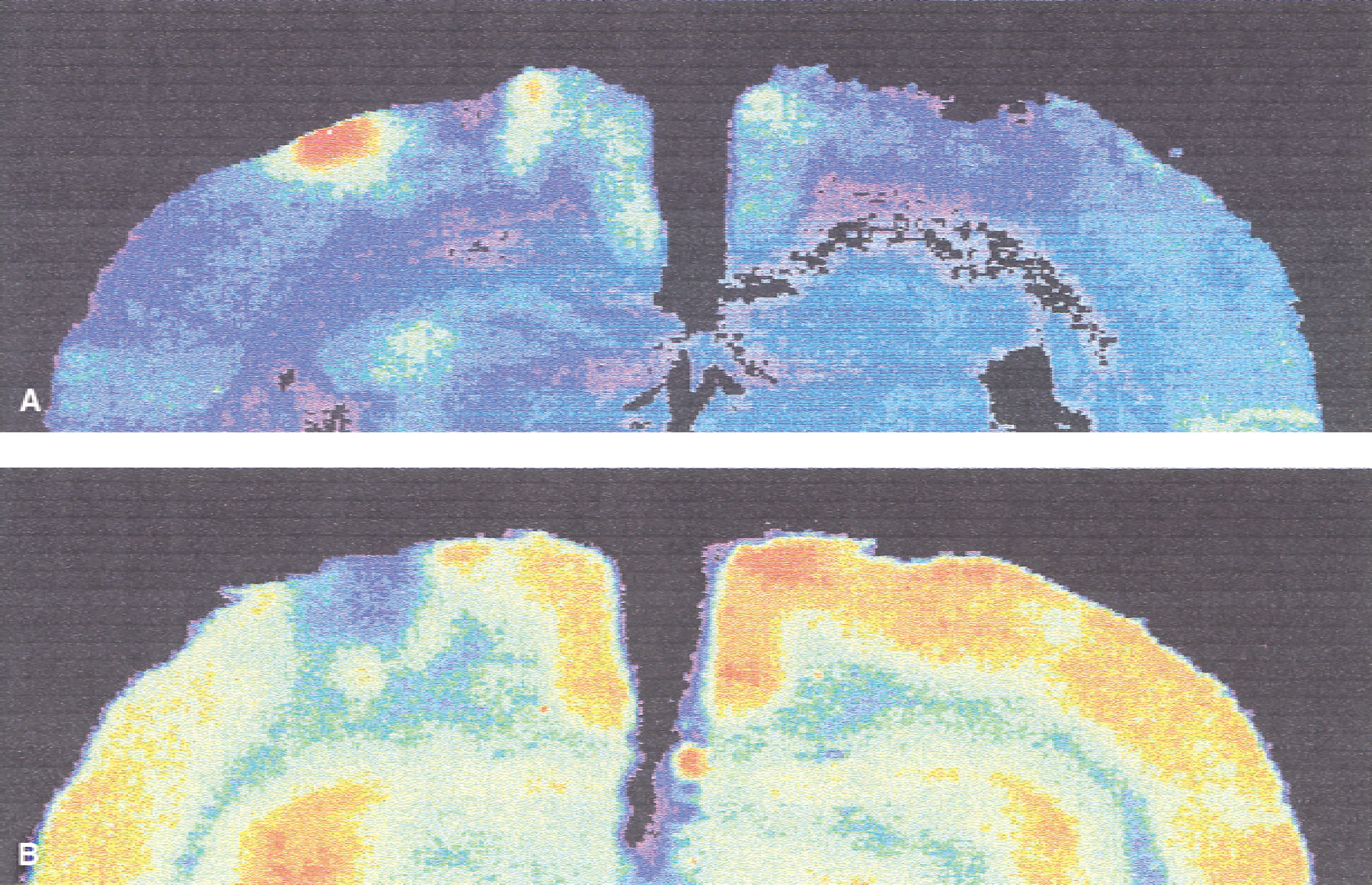
Color-enhanced autoradiograms from two contused animals showing patterns of glucose utilization
Values for LCBF, LCGU, and LCGC as a percentage of values obtained in the corresponding area in the contralateral cortex
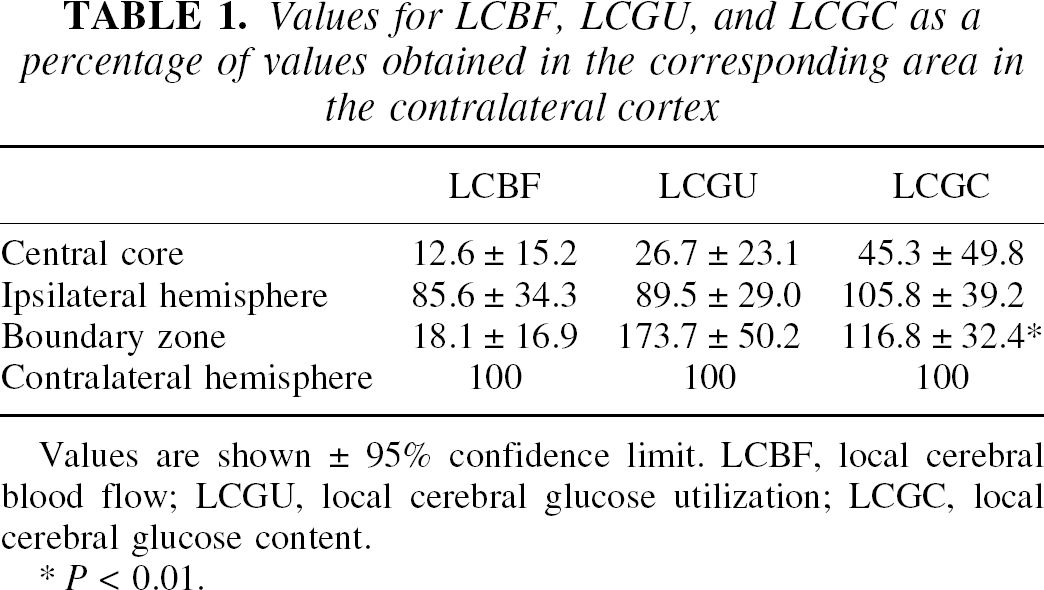
Values are shown ± 95% confidence limit. LCBF, local cerebral blood flow; LCGU, local cerebral glucose utilization; LCGC, local cerebral glucose content.
P < 0.01.
The rats used for LCGC determination showed more variation in their results. Three animals showed a small reduction in glucose content that represented between 70% and 98% of contralateral control values, and 3 animals showed some increase. The increase was relatively small (111%) in one, and in only one was there a substantial discrete area of high signal (124% and 158%), representing values more than two standard deviations greater than values obtained in ipsilateral cortex.
DISCUSSION
The results of the current study in the rat at 2 hours postinjury confirm findings in patients that there is a dissociation between cerebral blood flow and glucose metabolism after cerebral contusion. However, when considering changes in metabolism using the deoxyglucose method, the influence of changing kinetic constants must be considered. In the Sokoloff model, the kinetic constants are grouped together as the “lumped constant” in the operational equation. This lumped constant has been measured in animals and is assumed to be unchanged in a variety of physiologic and pathologic states. The lumped constant may be measured regionally using 14C-methylglucose autoradiography (Gjedde and Diemer, 1983; Kelly et al., 1992). Although in the current experiments the lumped constant has not been calculated, heterogeneity of methyl glucose uptake will reflect heterogeneity in the lumped constant. In four of the six rats where 14C-methylglucose was used, no apparent heterogeneity was observed, but some heterogeneity was seen in two of the animals. Unfortunately, it was not possible to measure methylglucose uptake and glucose metabolism in the same animal, so it is not possible to determine whether these areas coincided with areas of increased glucose metabolism in those animals where there were discrete areas of methyl glucose uptake. However, these focal areas of high signal did correspond to the typical areas where high signal was seen in metabolic maps. Because the magnitude of the increase in local glucose content was less than that in glucose metabolism it is reasonably safe to assume that in many cases hyperglycolysis occurs without a change in lumped constant. However, it is possible that focal changes in lumped constant may occur after head injury. Whether such local variations contribute to observed local changes in glucose metabolism requires further evaluation in patients.
