Abstract
Restoration of local blood supply in the post-ischemic brain plays a critical role in tissue repair and functional recovery. The present investigation explored beneficial effects of recombinant human erythropoietin (rhEPO) on vascular endothelial cell survival, angiogenesis, and restoration of local cerebral blood flow (LCBF) after permanent focal cerebral ischemia in adult mice. Saline or rhEPO (5,000 U/kg, intraperitoneal) was administered 30 mins before ischemia and once daily after ischemic stroke. Immunohistochemistry showed an enhancing effect of rhEPO on expression of EPO receptor (EPOR) of endothelial cells in the penumbra region 3 to 21 days after the ischemic insult. The treatment with rhEPO decreased ischemia-induced cell death and infarct volume 3 days after stroke. Specifically, rhEPO reduced the number of terminal deoxynucleotidyl transferase biotin-dUPT nick end labeling- and caspase-3-positive endothelial cells in the penumbra region. Colocalization of the vessel marker glucose transporter-1 (Glut-1) and cell proliferation marker 5-bromo-2′-deoxyuridine indicated enhanced angiogenic activity in rhEPO-treated mice 7 to 21 days after stroke. Western blot showed upregulation of the expression of angiogenic factors Tie-2, Angiopoietin-2, and vascular endothelial growth factor in rhEPO-treated animals. Local cerebral blood flow was measured by laser scanning imaging 3 to 21 days after stroke. At 14 days, LCBF in the penumbra was recovered to preischemia levels in rhEPO-treated mice but not in control mice. Our data suggest that rhEPO treatment upregulates the EPOR level in vascular endothelial cells, confers neurovascular protection, and enhances angiogenesis. We further show a promoting effect of rhEPO on LCBF recovery in the ischemic brain. These rhEPO-induced effects may contribute to therapeutic benefits in the treatment of ischemic stroke.
Introduction
Cerebral ischemia, induced by the occlusion of one or more cerebral artery/arteries, dramatically decreases local cerebral blood flow (LCBF) and causes cell death, brain atrophy, and functional deficits. Despite the paradox from reperfusion injury, preservation of surviving cells, tissue repair, and functional recovery largely depend on appropriate restoration of local blood supply. Therefore, neurovascular protection and angiogenesis have been an intriguing research area in the investigation of ischemic stroke.
As a member of the hematopoietic cytokine superfamily, erythropoietin (EPO) binds to its receptor (EPOR) in the bone marrow and stimulates erythropoiesis (Genc et al, 2004a). Recombinant human EPO (rhEPO), with high efficacy and safety, has been widely used in the clinical therapy of anemia associated with chronic renal failure (Macdougall et al, 1991). Over the last decade, multiple functions of EPO outside the hematological system have been observed, one of which is a potent neuroprotective action in the central nervous system (Genc et al, 2004b; Brines and Cerami, 2006). Erythropoietin and EPOR are expressed in central nervous system cells, including neurons, astrocytes, oligodendrocytes, and microvascular/capillary endothelial cells (Bernaudin et al, 2000, 1999). Hypoxic and ischemic insults upregulate the expression of hypoxia inducible factor and subsequently stimulate the expression of EPO and EPOR. As a multifunctional protectant, EPO has shown remarkable neuroprotective effects in ischemic stroke models of adult and neonatal animals (Bernaudin et al, 1999; Calapai et al, 2000; Wei et al, 2006). Available data indicate that EPO may be effective and safe in the treatment of human ischemic stroke (Ehrenreich et al, 2002; Brines and Cerami, 2006).
Erythropoietin has a profound antiapoptotic effect mediated by a number of potential pathways (Chong et al, 2003b; Digicaylioglu et al, 1995; Sattler et al, 2004; Siren et al, 2001). Erythropoietin exerts its antiapoptotic effects not only on neurons but also on other cell types. Erythropoietin reduced apoptosis of bovine pulmonary artery endothelial cells (Carlini et al, 1999) and showed an antiapoptotic effect in cerebral vascular cells (Chong et al, 2002, 2003a). The EPO protection is intimately dependent on the activation of protein kinase B (Akt1) and the maintenance of mitochondrial membrane potential (Chong et al, 2002). Several apoptotic events including Bcl-xL expression, cytochrome C release and caspase activation are regulated by EPO in endothelial cells (Chong et al, 2003a).
Recent results suggest that EPO may play a role in stimulating postischemia angiogenesis in the brain (Wang et al, 2004). Seven days after middle cerebral artery (MCA) occlusion, expression of vascular endothelial growth factor (VEGF) in the penumbra regions increased 52% in rhEPO-treated rats compared with the saline group. The increase in VEGF implies that EPO may enhance postischemic angiogenesis (Wang et al, 2004). However, there has been no direct evidence to elucidate whether rhEPO treatment may help to restore LCBF to the ischemic and peri-ischemic regions. In the present investigation, using a new technique of laser scanning imaging, we examined the hypothesis that rhEPO treatment not only protected brain cells including vascular endothelial cells and stimulated angiogenic activity, but also promoted LCBF recovery after permanent cerebral ischemia in mice.
Materials and methods
Middle Cerebral Artery Occlusion in Mice
All animal experiments and surgical procedures were approved by the University Animal Research Committee and met NIH standards. Focal ischemia was induced by right MCA occlusion in adult male C57BL/6 mice (20 to 25 g; Harlan, Indianapolis, IN, USA). Surgical procedures were modified from previous description on rats (Wei et al, 2005). Briefly, animals were subjected to 4% chloral hydrate intraperitoneal (i.p.) anesthesia and the right MCA was permanently occluded by insertion of a 10 to 0 suture (Surgical Specialties CO., Reading, PA, USA). This was accompanied by 8-min bilateral common carotid artery ligation. During surgery and recovery periods, body temperature was monitored and maintained at 37.0°C ± 0.5°C using a temperature control unit and heating pads. Animals were killed different days after ischemic stroke. The brain was immediately removed and mounted in optimal cutting temperature (OCT) compound (Sakura Finetek USA Inc., Torrance, CA, USA) at −80°C for further processing.
Recombinant Human Erythropoietin and 5-Bromo-2′-deoxyuridine Administration
Recombinant human erythropoietin (5,000 U/kg, Amgen Inc., Thousand Oaks, CA, USA) or saline was administered by i.p. injection 30 mins before ischemia and continued once per day after ischemic surgery until the day of killing. For mitotic labeling of cell proliferation, 5-bromo-2′-deoxyuridine (BrdU) (Sigma, St Louis, MO, USA) was administered (i.p.) to animals at a dosage of 50 mg/kg body weight once daily until the day of killing.
Physiological Parameters Measurements
As changes in physiological parameters may affect LCBF, we monitored hematocrit, mean arterial blood pressure, and heart rate at 0, 3, 7, 14, and 21 days after stroke with and without rhEPO treatment. Six animals were used in each group at each time point. Mice were subjected to 4% chloral hydrate anesthesia; heparanized HE-10 polyethylene tubing was inserted into the right common carotid artery and connected to the blood pressure analyzer Model 400 (Digi-Med, Louisville, KY, USA). After stabilization, the systolic, diastolic, and the mean blood pressure as well as the heart rate were recorded every 10 secs for 2 mins. After recordings, heparanized capillary tubing was used for collecting the arterial blood sample. Blood was centrifuged at 12,000 r.p.m. for 5 mins. The hematocrit was calculated by the percentage of the length of packed cells to the whole length.
Measurement of Infarct Volume
The cortical infarct volume was morphometrically measured by triphenyl tetrazolium chloride staining and Nissl staining 3 days after stroke. Animals were killed with an overdose of pentobarbital (100 mg/kg) followed by intracardiac perfusion with 200 mL of 0.9% NaCl. Brains were then sliced into 1-mm coronal sections using a mouse brain matrice (Harvard Bioscience, South Natick, MA, USA). The brain slices were incubated in phosphate-buffered saline (PBS) (pH 7.4) containing 2% triphenyl tetrazolium chloride at 37°C for 20 mins and then stored in 10% neutral-buffered formalin. For Nissl staining, fresh frozen coronal sections (10 μm) were taken every 1 mm from the frontal cortex to the mesencephalon. The infarct ratio and volume were measured by an image analyzer (SigmaScan Pro 5, Systat Software Inc. Point Richmond, CA, USA) and calculated using the indirect method described by Swanson et al (1990).
LCBF Measurement
Laser scanning imaging was used to measure LCBF at seven time points: immediately before ligation, right after occlusion, 3, 7, 14, and 21 days after ischemia. Under anesthesia, a crossing skin incision was made on the head to expose the whole skull. Laser scanning imaging measurements and analysis were performed using the PeriScan® system and LDPIwin 2® (Perimed AB, Stockholm, Sweden) on the intact skull. The scanning region had a center point of ML + 4.1 mm, and the four edges of the infarct area were ML + 2.9 mm, ML + 5.3 mm, AP − 1.5 mm, and AP + 2.0 mm, respectively. In laser scanning imaging, the ‘single mode’ with medium resolution was used to scan the photo image of LCBF. The laser beam was pointed to the center of the ischemic core (ML + 4.1 mm, AP 0 mm), the scan range parameter was set up as 5 × 5 and the intensity was adjusted to 7.5 to 8.0. The conventional ‘duplex mode’ was used to record the Doppler image with the laser beam pointed to exact the same point on the border of the stroke core (ML − 3.5 mm, AP 0 mm). Corresponding areas in the contralateral hemisphere were similarly surveyed as internal controls.
Terminal Deoxynucleotidyl Transferase Biotin-dUPT Nick end Labeling Staining
A terminal deoxynucleotidyl transferase biotin-dUPT nick end labeling (TUNEL) staining kit (DeadEnd™ Fluorometric TUNEL system, Promega, Madison, WI, USA) was used to visualize cell death in 10-μm coronal frozen sections together with the vessel marker glucose transporter-1 (Glut-1). After 10-min fixing by 10% buffered formalin phosphate (Fisher Scientific, Pittsburgh, PA, USA) and pretreatment with −20°C ethanol:acetic acid (2:1) and 0.2% Triton X-100, the brain sections were incubated in an equilibration buffer as instructed by the kit. The TdT enzyme and nucleotide mix were then added at proportions specified by the kit for 75 mins at room temperature. The slides were washed with the provided 2 × SSC washing buffer. To identify endothelial cells, slides were then incubated with the Glut-1 primary antibody (1:1000, Chemicon, Temecula, CA, USA) overnight. After PBS washes and incubation with Cy3-conjugated anti-rabbit IgG (1:1,000, Jackson ImmunoResearch, West Grove, PA, USA), the slides were incubated in Hoechst 33342 (1:20,000, Molecular Probes, Carlsbad, CA) for 5 mins to stain the nucleus before mounting with ProLong Antifade mounting medium (Molecular Probes, Carlsbad, CA, USA) for observation and cell counting (see below).
Immunofluorescence Staining
Coronal fresh frozen sections of 10-μm thick were sliced using a cryostat vibratome (Ultapro 5000, St Louis, MO, USA). After the slides were completely air dried, sections were fixed in 10% buffered formalin phosphate for 10 mins, followed by treatment in a −20°C ethanol:acetic acid (2:1) solution for 12 mins, in 0.2% Triton-100 for 5 mins and washing in PBS three times between each step. Slides were blocked in 1% gelatin from cold water fish (Sigma, St Louis, MO, USA) diluted in PBS at room temperature for 1 h, and subsequently incubated with primary antibodies diluted in PBS overnight at 4°C. The primary antibodies were as follows: rabbit anti-Glut-1 (1:1,000, Chemicon, Temecula, CA, USA), mouse anti-CD31 (1:200, Cymbus Biotechnology, Eastleigh, UK) and rabbit anti-EPOR (1:200, Santa Cruz Biotechnology, Santa Cruz, CA, USA). After rinsing with PBS, brain sections were then treated with secondary antibodies Alexa Fluor 488 anti-rabbit IgG and anti-mouse IgG (1:200, Molecular Probes, Carlsbad, CA, USA) or Cy3-conjugated donkey anti-rabbit IgG (Jackson ImmunoResearch, West Grove, PA, USA) for 1 h at room temperature.
Activated caspase-3-like protein was detected by FAM-DEVD-FMK Caspase 3 Detection Kit (Cell Technology Inc, Minneapolis, MN, USA). The staining procedure was modified from the cultured cell staining provided by the kit. Sections were fixed by 10% buffered formalin phosphate for 3 mins. After gently washing with PBS, slides were incubated in FAM-Peptide-FMK working solution for 30 mins at room temperature, followed by washing using the provided solution.
To double stain vessels with BrdU, slides were postfixed and the BrdU staining procedure was performed with rat anti-BrdU (1:500, Abcam, Cambridge, UK) and Cy3-conjugated goat anti-rat IgG (Jackson ImmunoResearch, West Grove, PA, USA) as the primary and secondary antibodies according to our established technique.
The brain sections were mounted and coverslipped, imaged, and photographed under a florescent microscope (BX51, Olympus, Japan) and laser scanning confocal microscopy (Carl Zeiss Microimaging Inc., Thornwood, NY, USA).
Stereological Cell and Vessel Counting
For systematic random sampling in design-based stereological cell counting, every ninth brain section (90 μm apart) across the entire region of interest was counted. For multistage random sampling, six fields per brain section were randomly chosen in the penumbra region (Whitaker et al, 2006) under × 40 magnification of a light microscope or in confocal images. To quantify the vessels double-labeled with Glut-1 and BrdU, six randomly selected fields in the penumbra region were captured at × 20 magnification. This was repeated in four separate sections per brain. The counting of the blood vessels was based on the morphological vessel-like structures with the positive endothelial cell marker immunoreactions. One vessel-like structure colocalized with EPOR, TUNEL, caspase-3, and/or BrdU was counted as one immunopositive vessel.
Western Blot Assay of Angiogenic Factors
Expression of angiogenic proteins was examined by Western blot. For each group at each time point, three animals were used to collect the ipsilateral penumbra tissue samples and proteins were extracted using the NE-PER Nuclear and Cytoplasmic Extraction Kit (Pierce Biotechnology, Rockford, IL, USA). Protein concentrations were determined by bicinchoninic acid assay (PIERCE, Rock ford, IL, USA). The samples were kept frozen at −80°C until assessment.
Samples of 50 μg proteins were electrophoresed on a 6% to 15% gradient gel in the presence of sodium dodecyl sulfate—polyacrylamide gel electrophoresis in a Hoefer Mini-Gel system (Amersham Biosciences, Piscataway, NJ, USA) and transferred in the Hoefer Transfer Tank (Amersham Biosciences, Piscataway, NJ, USA) to a polyvinylidene difluoride membrane (BioRad, Hercules, CA, USA). Membranes were blocked with buffer (Tris-buffered saline containing 0.1% Tween-20, pH 7.6%, 7% milk) at room temperature for 2 h and incubated overnight at 4°C with rabbit polyclonal Tie-2 (H-176) (1:1,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit anti-mouse Angiopoietin-2 (1:1,000, Chemicon, Temecula, CA, USA), or anti-VEGF (clone JH121) (1:1,000, Upstate, Charlottesville, VA, USA). Mouse β-actin (Sigma, St Louis, MO, USA) was used as the protein loading control. The blots were washed in 0.5% Tris-buffered saline containing 0.1% Tween-20 and incubated with alkaline phosphatase-conjugated anti-rabbit or anti-mouse IgG (Promega, Madison, WI, USA) for 2 h at room temperature. Finally, membranes were washed with Tris-buffered saline containing 0.1% Tween-20 followed by three washes with Tris-buffered saline.
This whole procedure was repeated three times with new samples collected from different animals. The signal was detected by the addition of Bromo-chloro-indoryl phosphate/nitro blue tetrazolium (BCIP/NBT) solution (Sigma, St Louis, MO, USA), quantified and analyzed by the imaging software Photoshop Professional (Adobe® Photoshop® CS 8.0, San Jose, CA, USA). The intensity of each band was first measured and then subtracted by the background. The expression ratio of each target protein was then normalized against β-actin.
Statistical Analysis
Student's two-tailed t-test was used for comparison of two experimental groups. Multiple comparisons were performed using one-way analysis of variance followed by Tukey's test for multiple pairwise examinations. Changes were identified as significant if P was less than 0.05. Mean values were reported together with the standard deviation of mean (s.d.).
Results
Physiological Parameters in Recombinant Human Erythropoietin-Treated Mice After Ischemic Stroke
Adult male C57BL/6 mice were subjected to permanent occlusion of MCA. rhEPO (5,000 U/kg, i.p.) or saline was administered 30 mins before ischemia followed by once daily after the ischemic stroke. No gross adverse effects were seen at this dosage of rhEPO for up to 21 days. The physiological condition of the mice was monitored by hematocrit, mean arterial blood pressure, and heart rate at 0, 3, 7, 14, and 21 days after ischemia. There were no significant differences in these parameters between the rhEPO treatment group and saline control group. No statistical difference was detected for a single parameter at different time points (Table 1).
Effects of rhEPO on physiological parameters in stroke animals
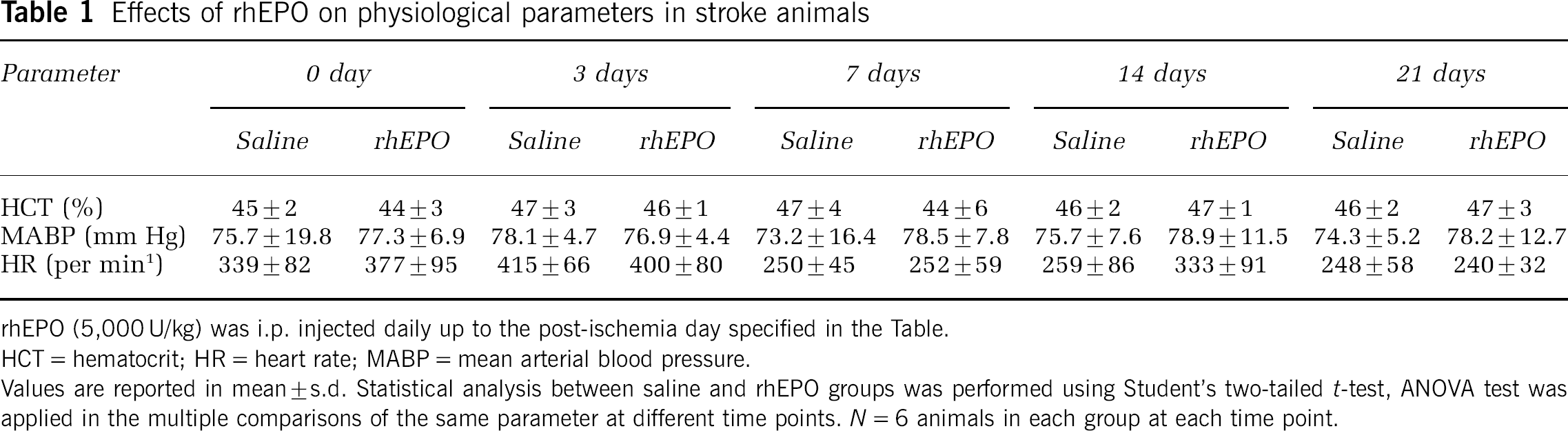
rhEPO (5,000 U/kg) was i.p. injected daily up to the post-ischemia day specified in the Table.
HCT = hematocrit; HR = heart rate; MABP = mean arterial blood pressure.
Values are reported in mean ± s.d. Statistical analysis between saline and rhEPO groups was performed using Student's two-tailed t-test, ANOVA test was applied in the multiple comparisons of the same parameter at different time points. N = 6 animals in each group at each time point.
Recombinant Human Erythropoietin Enhanced the Erythropoietin Receptor Expression in the Ischemic Brain
Erythropoietin receptor expression in microvessels was detected by double immunostaining of anti-EPOR antibody and the endothelial cell antibody CD31 in the penumbra region 3 to 21 days after the onset of ischemia (Figures 1A–1C). Recombinant human erythropoietin treatment significantly increased the number of EPOR-positive vessels compared with saline control (Figure 1D).
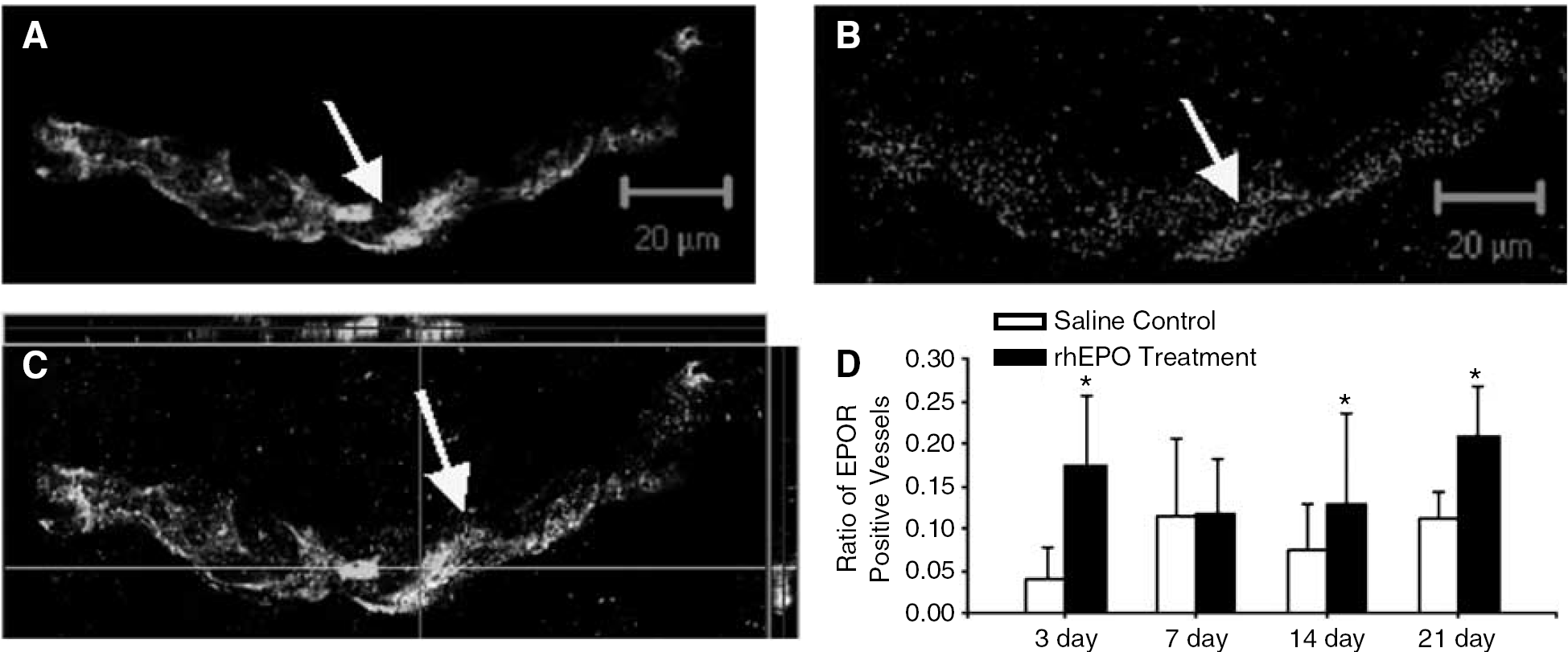
rhEPO enhanced the expression of EPOR on capillary endothelial cells after ischemic stroke. rhEPO (5,000 U/kg, i.p) was administered 30 mins before ischemia and repeated daily after the insult. EPOR expression in the ischemic cortex 3 days after stroke was detected using immunostaining. EPOR expression (red color) was detected in microvessels (green color) in penumbra region. (
Recombinant Human erythropoietin Treatment Attenuated Ischemia-Induced Cell Death and Infarct Size
Three days after ischemic stroke and rhEPO or saline treatment, brain sections were analyzed for cell death and infarct formation. In triphenyl tetrazolium chloride stained brain sections, the pink area between the normal cortex (red) and ischemic core (white) regions represents the bordering penumbra area. Terminal deoxynucleotidyl transferase biotin-dUPT nick end labeling staining was used to detect cell death in the penumbra of ischemic cortex (Figure 2A). The ratio of TUNEL-positive cells versus Hoechst 33342-labeled total cells was reduced by the rhEPO treatment (Figures 2B–2D). Compared with ischemia-saline controls, the infarct volume in mice that received the rhEPO treatment was reduced by 49% 3 days after the onset of ischemia (P < 0.05 compared with saline controls) (Figures 2E–2H).
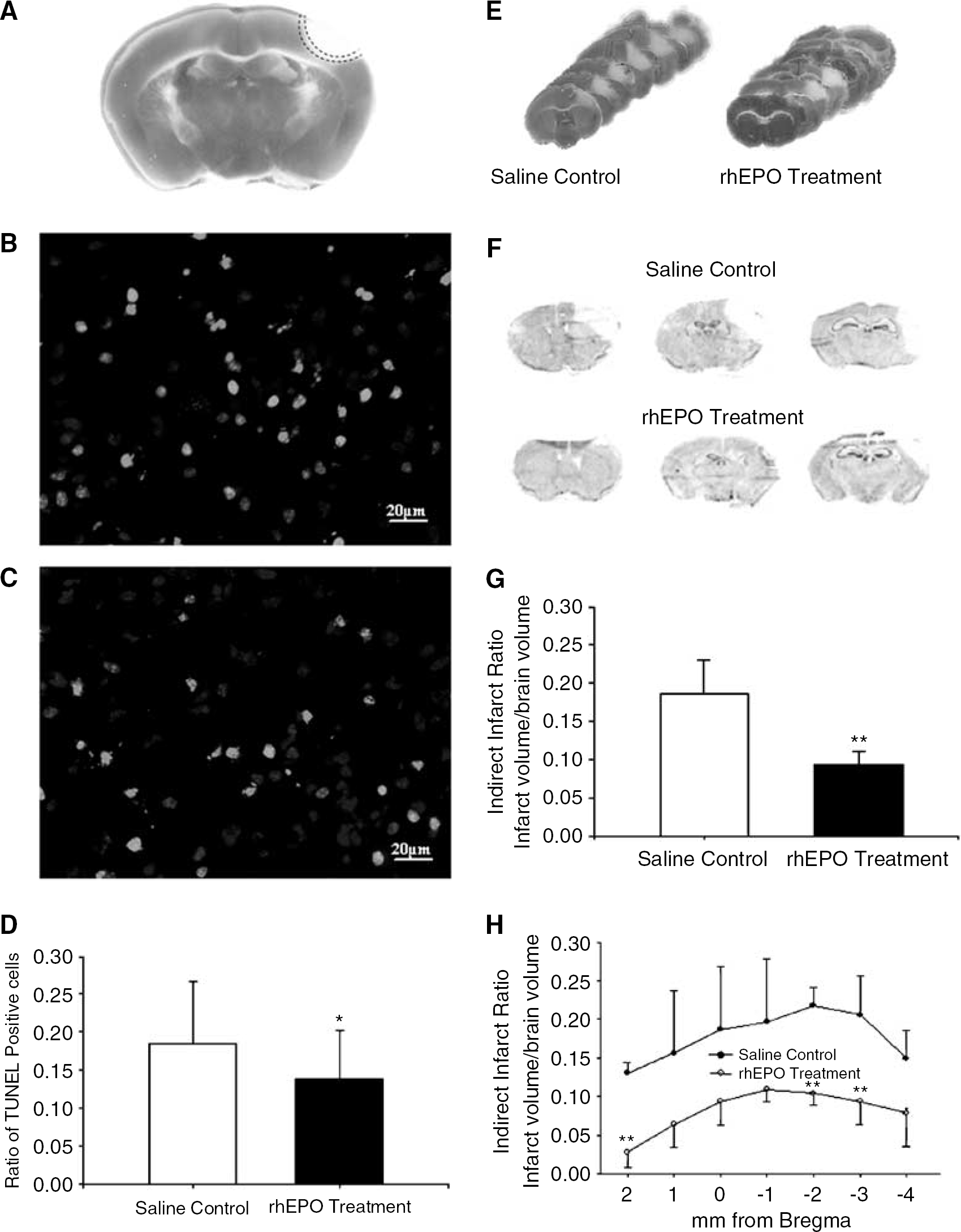
Neuroprotective effect of rhEPO after ischemic stroke. The effects of rhEPO on ischemia-induced cell death and infarct formation were examined 3 days after the onset of ischemia. (
To examine the ischemic injury to vascular endothelial cells, cell death revealed by TUNEL or caspase-3 staining was also assessed in Glut-1-positive cells. Glucose transporter-1 is a specific marker for microvessels (Whitaker et al, 2006) (Figures 3A–3F). The colocalization of TUNEL and Glut-1 showed DNA-damaged endothelial cells, and the colocalization of activated caspase-3 and Glut-1 indicated apoptotic endothelial cells. In both assays taken 3 days after ischemia the rhEPO treatment significantly attenuated ischemic injury compared with that in ischemia-saline control mice (Figures 3G–3H).
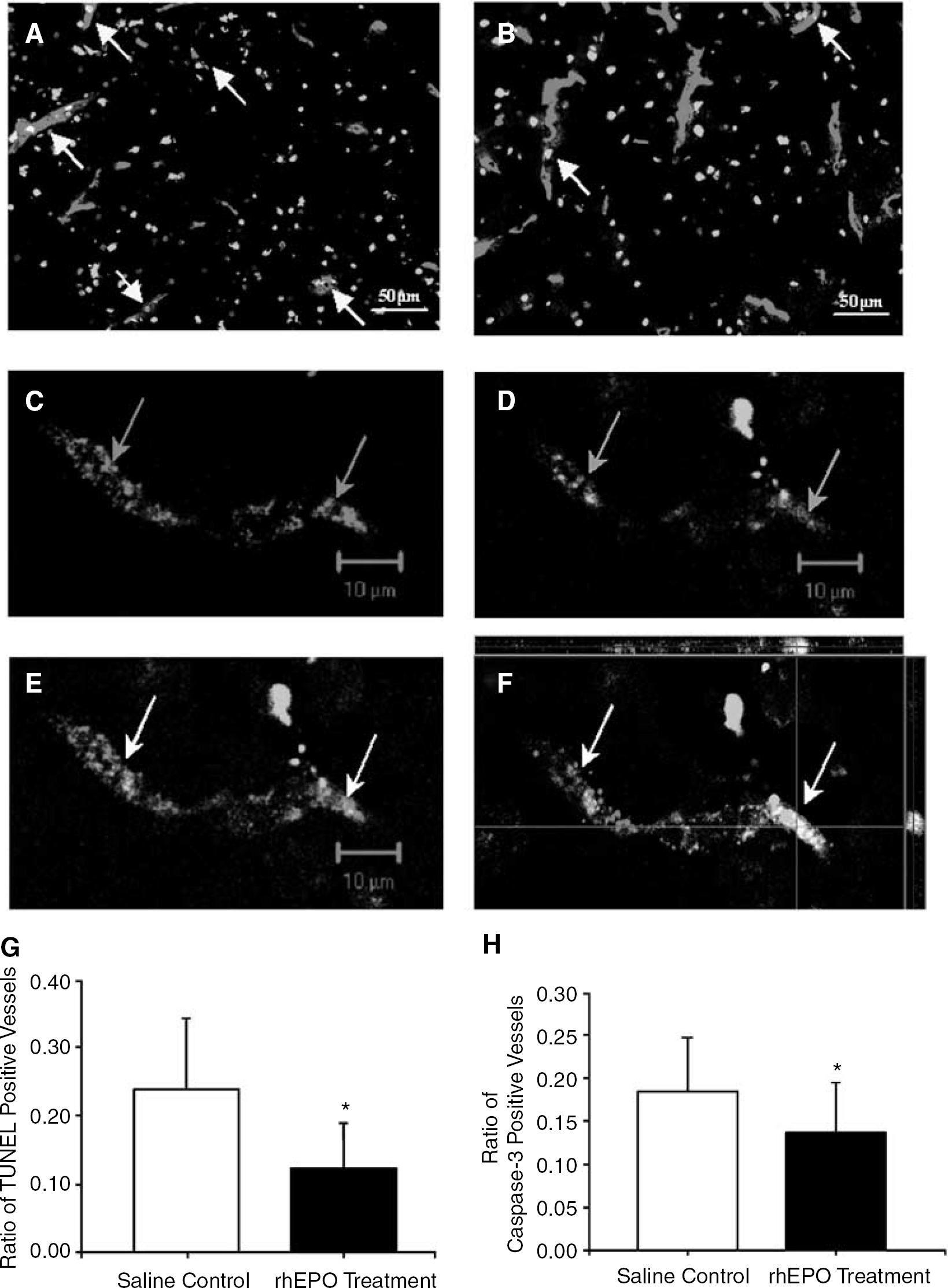
Protective effect of rhEPO on capillary endothelial cells in the ischemic brain. Capillary endothelial cells were visualized by Glut-1 (red) under a fluorescent microscope. Cell death and apoptosis were identified by colabeled TUNEL (green in
Recombinant Human Erythropoietin Enhanced Post-Ischemia Angiogenesis
The mitotic marker BrdU (50 mg/kg, i.p. daily) was injected after ischemic stroke to reveal cell proliferation. Three days after the onset of ischemia, 3.3% ± 0.6% vessels in the saline control group were BrdU-positive, while 7.1% ± 0.7% vessels were BrdU-positive in the rhEPO-treated group, although the difference was not statistically significant (n = 6, P = 0.23). In later time points of 7 and 14 days after stroke, treatment with rhEPO significantly increased BrdU-positive microvessels (Figure 4). The number of BrdU-positive cells in saline-treated mice increased slowly but reached the treatment group value 21 days after stroke (Figure 4).

Effects of rhEPO on endothelial cell proliferation in the penumbra region of the ischemic cortex. Endothelial cell proliferation or angiogenesis was revealed by costaining with Glut-1 (green) and BrdU (red). (
To further show that angiogenesis was promoted by rhEPO treatment, Western blotting was used to analyze the expression of major angiogenic factors Angiopoietin-2, the angiopoietin receptor Tie-2, and VEGF, factors that were likely to contribute to long-term angiogenic activities (Figure 5A). From 3 days after stroke, the expression of Angiopoietin-2, Tie-2, and VEGF was upregulated by rhEPO treatment compared with the saline control group (Figures 5B–5D).
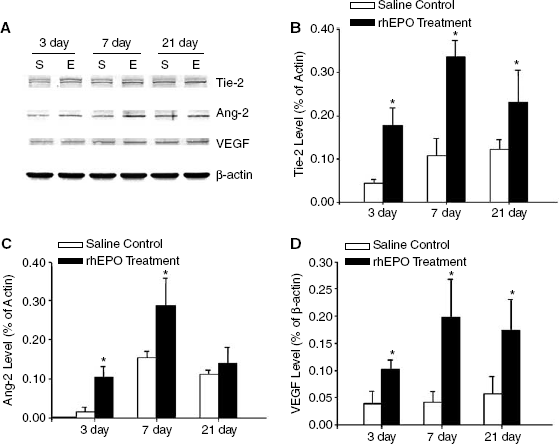
Effects rhEPO treatment on angiogenic factor expression. Angiogenic factors Tie-2, Angiopoietin-2, and VEGF were detected by Western blot. (
Restoration of Local Cerebral Blood Flow in the Post-Ischemic Cortex by Recombinant Human Erythropoietin Treatment
To test whether rhEPO-stimulated angiogenesis had a significant impact on long-term blood circulation, LCBF was measured using the technique of Laser Doppler imaging (Khan and Newton, 2003). Figure 6 shows changes of blood flow in the penumbra region before, immediately after, and 3, 7, 14, and 21 days after MCA occlusion. Local cerebral blood flow in ischemic and peripheral regions decreased markedly in both saline control and rhEPO-treatment groups. Although no significant difference, local blood flow started the trend of recovery 7 days after the onset of ischemia in rhEPO-treated animals but not in saline control mice, which is consistent with the angiogenesis data (Figures 6D and 6E, and see Figure 4). By post-ischemia day 14 and 21, the local blood flow in rhEPO-treated animals almost recovered to pre-ischemia levels (Figure 6D). However, only slight flow recovery was seen in the saline group (Figure 6E). The enhanced and accelerated blood flow recovery was observed with the laser Doppler imaging scanning method and the conventional single point laser Doppler method (Figures 6F and 6G).
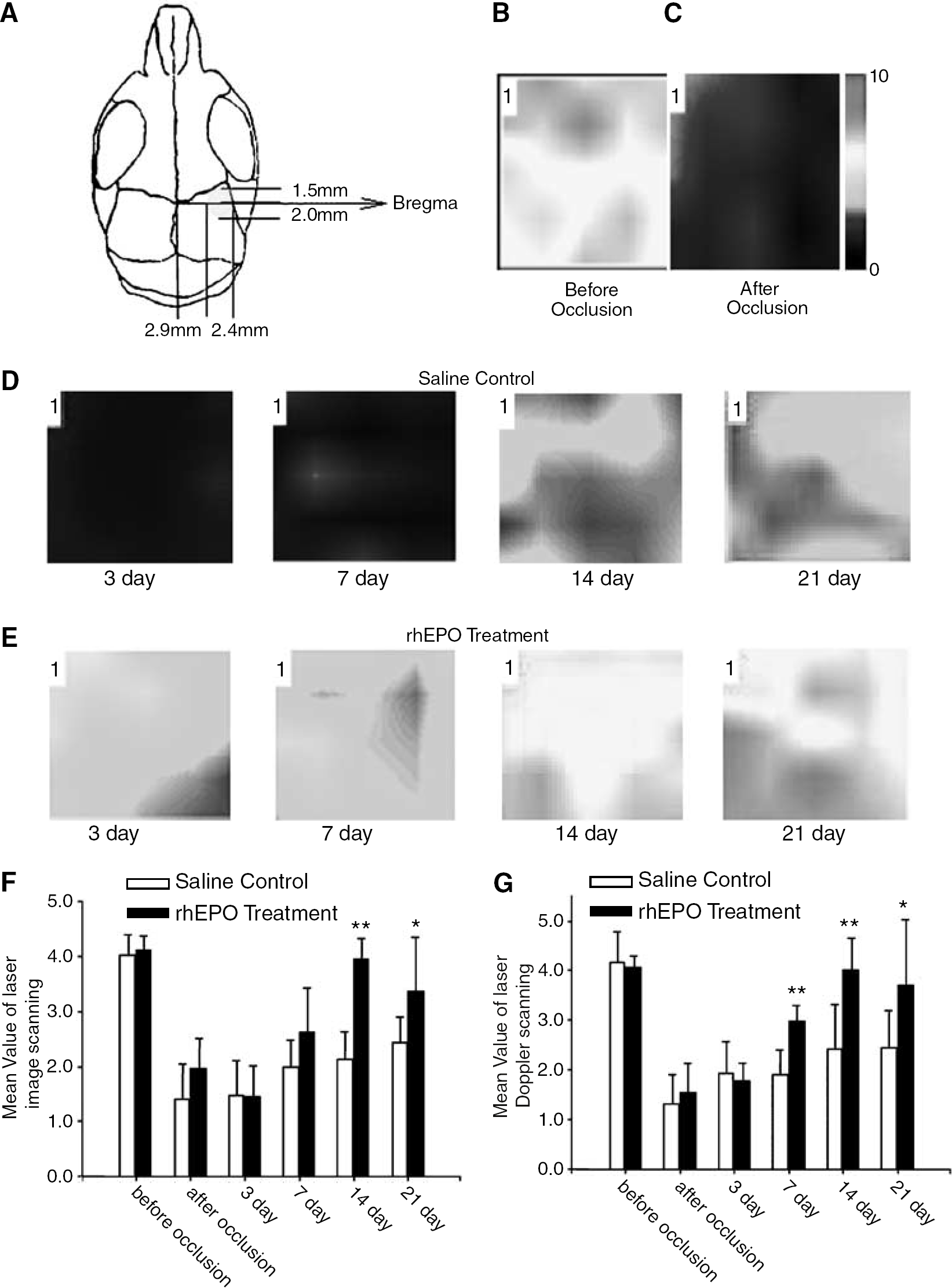
Effects rhEPO treatment on the LCBF in the ischemic brain. Local cerebral blood flow was measured using the PeriScan® laser image scanner. (
Discussion
Although stimulation of angiogenesis is a known effect of EPO, whether the increased angiogenesis might be translated into improved recovery of local blood flow has remained unanswered. In addition to the demonstration that rhEPO treatment reduced ischemia-induced cell death, attenuated the infarct volume, and increased EPOR expression in endothelial cells in the post-ischemic brain, the present investigation showed protective and proliferative effects of rhEPO on vascular endothelial cells. The cell protection and increased angiogenesis were substantiated by early and full restoration of the local blood flow in the penumbra region of the ischemic cortex. The accelerated and enhanced flow recovery may play an important role for preventing delayed cell death, promoting long-term functional recovery, and prolonging the therapeutic window for other treatments.
The safety of using EPO in stroke therapy has been tested in rodents, in preclinical studies and phase I clinical trials; no noticeable adverse effects have being reported (Brines et al, 2000; Ehrenreich et al, 2002; Brines and Cerami, 2006). A primary concern for long-term application of EPO is that, as a hematopoietic factor, EPO may increase hematocrit and induce hypertension. In the present investigation, the rhEPO dosage of 5,000 U/kg was well tolerated in acute and long-term experiments. To identify possible side effects, we used a group of animals to particularly test several physiological parameters. There were no significant changes of hematocrit, mean arterial blood pressure, and heart rate during 3 to 21 days of rhEPO treatment. Thus, daily rhEPO administration at 5,000 U/kg for up to 3 weeks appears to cause no gross adverse effects in the cardiovascular system of adult C57BL/6 mice.
Erythropoietin has antiapoptotic properties in vitro and in vivo (Bernaudin et al, 1999; Sakanaka et al, 1998; Siren et al, 2001; Wei et al, 2006). Compelling in vitro evidence showed that EPO could reduce approximately 50% of the cell death induced by hypoxia and metabolic stress in a number of cell cultures including central neurons (Siren et al, 2001). Erythropoietin treatment resulted in approximately 50% reduction in the infarct volume of the ischemic brain (Bernaudin et al, 1999; Siren et al, 2001; Wei et al, 2006). Previous investigations have focused on EPO protection of neuronal cells; only a few studies examined the effect of EPO on endothelial cells. Tested in cultured endothelial cells, EPO was suggested to be a novel vascular protectant through activation of Akt1 and mitochondrial modulation of cysteine proteases (Chong et al, 2002). The present investigation perhaps provides the first in vivo demonstration that EPO treatment protects against apoptosis of microvascular endothelial cells in the ischemic brain.
Erythropoietin is an angiogenic factor in embryonic development (Kertesz et al, 2004), in female reproductive organs (Yasuda et al, 1998), in tumors (Acs et al, 2001), and in the nervous system (Maiese et al, 2005; Wang et al, 2004). In the central nervous system, angiogenesis results in the restoration of cerebral blood flow in the ischemia penumbra, which contributes to the long-term functional recovery after ischemia stroke (Jaquet et al, 2002; Marti et al, 2000; Ribatti et al, 1999, 2003). Erythropoietin receptor is detected in vascular endothelial cells and its mRNA and protein levels increase after ischemic stroke (Anagnostou et al, 1994; Bernaudin et al, 1999; Yamaji et al, 1996). Erythropoietin receptor expression on the surface of endothelial cells appears much earlier (day 1) than its expression in other cells in the brain after ischemic stroke (Bernaudin et al, 1999). The effect of EPO on capillary endothelial cells and its stimulation of angiogenesis have been speculated to benefit functional improvement of local blood flow in the post-ischemic brain. As expected, we show that EPO administration enhances early angiogenic activity and results in full recovery of local blood flow 2 to 3 weeks after ischemia.
There are at least two mechanisms that may mediate the angiogenic function of rhEPO: (1) enhanced proliferation of endothelial progenitor cells or endothelial cells (Asahara et al, 1997; Bahlmann et al, 2004, 2003; Heeschen et al, 2003; Ribatti et al, 1999) and (2) increased expression of endogenous growth/trophic factors and angiogenic cytokines (Maiese et al, 2005; Wang et al, 2004). The present study shows an enhancement of both angiogenic factors and activities around 3 days after ischemia. This early angiogenesis can be critical for rescue and repair of vulnerable cells and tissues in the ischemic brain. The idea is supported by the functional assay that early and full recovery of LCBF was observed in rhEPO-treated mice but not in saline control mice, even though BrdU-positive endothelial cells in the latter group reached the treatment group values 21 days after ischemic insult.
In the present study, the changes of LCBF during rhEPO treatment on the stroke model was shown in the photographic images, which were measured by a new laser scanning imaging technique. The laser scanning system generates color-coded images of the spatial distribution of the tissue perfusion and can be used to monitor microcirculation by collecting back-scattered light without touching the tissue. Using this tool, changes of blood flow can be accurately displayed in a two-dimension map, which avoids the limitation of single point laser Doppler detection. Furthermore, at each tissue site, the monochromatic laser light penetrates the tissue to a depth of a few hundred micrometers within the scattering volume. Based on those features, the data provided by this technique should represent not the superficial blood flow but the so-called blood flow ‘volume’ (Khan and Newton, 2003).
It is suggested that the combined actions of rhEPO of cell protection and promoting angiogenesis contribute to the early and marked recovery of local blood supply to the ischemic and penumbra brain regions. Our observations also support that rhEPO is a potentially therapeutic compound that acts through multiple intracellular pathways on different cell types and should be further explored for the treatment of ischemic stroke.
