Abstract
Tie-1 and Tie-2 are receptor tyrosine kinases (RTKs) that are exclusively expressed in endothelial cells and play important roles in endothelial cell biology. The authors have reported previously the temporal profiles of Tie-1 and Tie-2 mRNA expression after focal cerebral ischemia–reperfusion. In the current study, the localization of Tie-1/Tie-2 mRNA and proteins were further investigated in the same focal ischemia model. In situ hybridization showed that, after 60-minute ischemia and 72-hour reperfusion, both Tie-1 and Tie-2 mRNA appeared as capillary-like structures in the ischemic middle cerebral artery (MCA) cortex. Western blot analysis showed a biphasic expression of Tie-1 protein in the same region. The first peak, spanning the ischemic and early reperfusion period, was of low intensity and short-lived. The second peak was of greater intensity and spanning the period from 72 to 168 hours after reperfusion. Similarly, Tie-2 expression at the protein level also exhibited a biphasic pattern. Immunohistochemical studies, after 72 hours of reperfusion, showed that although Tie-1 and Tie-2 were detected within the ischemic cortex, they actually were expressed in different populations of endothelial cells in different regions. In agreement with the in situ hybridization study, Tie-1 immunoreactivity appeared as capillary-like structures in cortical layers 2 to 4. Similar capillary-like appearance of Tie-2 immunoreactivity was noted in the outer cortical layers. In addition, Tie-2 immunoreactivity also was observed in cortical layer 6b, where de novo large vessel formation was noted. Cellular colocalization experiments revealed that Tie-2 is expressed in proximity to its antagonist, Angpo-2, as well as basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF) in cortical layer 1, where active vessel remodeling was noted. Interestingly, bFGF only partially colocalized with VEGF, suggesting differential roles for these angiogenic factors during vessel remodeling. Tie-1 protein, to a lesser degree, also colocalized with Angpo-2, bFGF, and VEGF in cortical layer 1. Magnetic resonance imaging (MRI) showed increased regional cerebral blood flow (CBF) corresponding to the expression of these angiogenesis gene products. Together, these findings suggest that the evolving expression of angiogenesis genes underlie the robust vascular remodeling after ischemia and reperfusion.
Angiogenesis occurs in the ischemic brain, particularly in the ischemic penumbra. The extent of angiogenesis has been correlated to survival in stroke patients (Krupinski et al., 1994; Plate, 1999). Ischemic brain injury is a consequence of severe reduction of blood supply to the affected region. The resultant low tissue oxygen tension often leads to compensatory neovascularization to meet the metabolic demand. Angiogenic activity is maintained by a balance of positive and negative regulators of blood vessel growth. The search for the modulators of angiogenesis has yielded several candidates; however, the specific roles of endogenous mediators in angiogenesis remain unknown (Folkman, 1995; Pepper, 1996; Rosenberg et al., 1996; O'Reilly et al., 1997; Risau, 1997).
Tie-1 and Tie-2 are receptor tyrosine kinases (RTKs) that are expressed exclusively in endothelial cells (Mustonen and Alitalo, 1995). Engineered mutations of mouse genes encoding Tie-1 and Tie-2 result in distinct vascular defects, suggesting that the signaling pathways mediated by these RTKs play specific roles in the regulation of vascular structures (Dumont et al., 1994; Sato et al., 1995; Beck and D'Amore, 1997). The expression of Tie-1 is increased during maturation of ovarian follicles, wound healing, and tumor angiogenesis (Hatva et al., 1994). However, it is not clear whether the observed Tie-1 expression is a cause or consequence of neovascularization.
Two ligands for the Tie-2 receptor have been cloned, angiopoietin-1 (Angpo-1) (Davis et al., 1996) and angiopoietin-2 (Angpo-2) (Maisonpierre et al., 1997). Angpo-1 is important in the developing vasculature (Davis et al., 1996; Suri et al., 1996). Mice lacking Angpo-1 display characteristic vascular defect that is distinguished from that caused by VEGF gene deletion, but are reminiscent of Tie-2 gene knockout (Sato et al., 1995). Angpo-2 and Angpo-1 share approximately 60% homology (Maisonpierre et al., 1997). In contrast with Angpo-1, which stimulates phosphorylation of Tie-2 in a number of endothelial cell lines, Angpo-2 blocks Angpo-1 activation of the Tie-2 receptor. Transgenic embryos overexpressing Angpo-2 were found to die on E 9.5–10.5, and the defect in these embryos was similar to mice lacking either Angpo1 or Tie-2 receptor. Angpo-1 is likely to play a role in the maturation of the vascular network. Angpo-2, which destabilizes the neovasculature, probably is involved in vascular remodeling.
Recently, Zhang et al. (1999) reported that Tie-1 mRNA was up-regulated in cerebral microvessels after embolic MCA occlusion. The authors previously noted the expression of Tie-1, Tie-2, and Tie-2 ligands, Angpo-1 and Angpo-2 mRNA, after transient cerebral ischemia (Lin et al., 2000). The temporal profiles of the expression of these genes were different from those of other angiogenic genes such as VEGF/VEGFR and bFGF/FGFR. To the authors' knowledge, expression of Tie receptor proteins in the ischemic brain has not been reported. The purpose of the current study was to examine the expression of Tie-1 and Tie-2 receptor at the protein level and their relation to the expression of Angpo-1 and Angpo-2 and other angiogenic factors, such as VEGF and bFGF, in a rat focal ischemia–reperfusion model.
MATERIALS AND METHODS
Stroke model
The focal cerebral ischemia–reperfusion model in the rat has been described previously (Chen et al., 1986; Lin et al., 1993). Briefly, male Long-Evans rats weighing 250 to 300 g were anesthetized with chloral hydrate (360 mg/kg body weight, IP). The trunk of the right MCA above the rhinal fissure was identified under a stereomicroscope and ligated with a 10–0 suture. Interruption of blood flow distal to ligation was confirmed under the microscope. Both common carotid arteries then were occluded using nontraumatic aneurysm clips. After 60 minutes of ischemia, the aneurysm clips and the suture were removed and restoration of blood flow in all three arteries was verified. While under anesthesia, the rectal temperature was monitored and maintained at 37.0°C ± 0.5°C using a homeothermic blanket (Harvard, Holliston, MA, U.S.A.). In this model, ischemia for 60 minutes consistently resulted in a large infarct confined to the right MCA cortex (Liu et al., 1989). No morphologic (Chen et al, 1986; Liu et al., 1989) or biochemical evidence of ischemic brain injury was noted in the left MCA cortex (An et al., 1993). After the ischemic insult, rats were kept in an air-ventilated incubator at 24.0°C ± 0.5°C for up to 2 weeks and were provided with water and lab chow ad libitum until the end of experiments. At the end of each experiment (30 and 60 minutes after onset of ischemia or 30, 60, 90 minutes, 4, 12 hours, 1, 3 days, 1 and 2 weeks after reperfusion), rats were killed by decapitation under anesthesia. Brains were removed quickly to collect the cerebral cortex. The right or left cerebral cortices were frozen immediately in liquid nitrogen and stored at −70°C until further processing. The right cerebral cortex also was sampled from animals subjected to vascular surgeries without occlusion to serve as sham-operated control. In this stroke model, only the right MCA cortex sustained severe ischemia with regional blood flow reduction by 88% to 92% (n = 4). Only very mild ischemia was noted (reduction of blood flow by 10 to 20%) outside the right MCA cortex (An et al., 1993). In some experiments, the whole brain was photographed for a gross visual assessment for the extent of angiogenesis. All procedures were approved by the institutional Animal Studies Committee and were in accordance with the Public Health Service Guide for the Care and Use of Laboratory Animals, USDA Regulations, and the Guidelines of the American Veterinary Medical Association Panel on Euthanasia.
In situ hybridization
In situ hybridization to detect the regional distribution of mRNA signals has been described previously (An et al, 1993; Lin et al., 1997). Briefly, frozen brain slices were sectioned (25-μm-thick) and mounted on poly-L-lysine-coated slides. Brain sections were subjected to 0.001% proteinase K digestion at 37°C for 30 minutes, then immersed in 0.1 mol/L triethanolamine with 0.25% acetic acid anhydride at room temperature for 10 minutes, and subsequently dehydrated in 50%, 70%, 95%, and 100% ethanol (3 minutes each). Hybridization was performed in a solution containing 12.5 mol/L formamide, 10% dextran sulfate, 0.3 mol/L NaCl, 1× Denhardt's solution, 10 mmol/L Tris-Cl 8.0, 500 μg/mL sheared salmon sperm DNA, 100 μg/mL tRNA, 20 mmol/L Dithiothreitol, and 107 cpm/mL of probes at 55°C. Reverse transcription polymerase chain reaction–amplified cDNA probes (Lin et al., 2000) were labeled with [33P]-dCTP using the random primer labeling method (Amersham, Arlington Heights, IL, U.S.A.). After overnight hybridization, slides then were washed sequentially in 2×, 1×, 0.2× and 0.1× SSC at 55°C for 30 minutes each, followed by dehydration in 50%, 70%, 95%, and 100% ethanol for 3 minutes each. Brain sections were exposed to Kodak BioMax-MR-1 film (Kodak, NY, U.S.A.) and autoradiographs were analyzed with Adobe Photoshop 4.0 (Adobe, CA, U.S.A.). In control experiments, sections were incubated with a 100-fold excess of unlabeled probe or pretreated with RNase A (100 μg/mL, 37°C, 30 minutes). These experiments resulted in no or negligible signal.
Western blot analysis
The ischemic (right) cortex was placed in 2 mL ice-cold lysis buffer (0.32 mol/L sucrose, 1 mmol/L EDTA, 50 mmol/L Tris-Cl pH 7.4, and protease inhibitor cocktail [Boehringer Mannheim, Mannheim, Germany]) and then homogenized with a motor-driven glass homogenizer equipped with a teflon pestle. Brain homogenate then was centrifuged at 11,400 g for 10 minutes. Supernatant was obtained and further centrifuged at 45,700 g for 20 minutes. The pellet containing the plasma membrane fraction was collected and washed twice with lysis buffer. The final pellet was resuspended in 1 mL lysis buffer. The supernatant was also collected as the cytosolic fraction. The entire procedures were performed at 4°C. An equal amount of proteins from the plasma membrane fraction (200 μg) was applied to sodium dodecyl sulfate-polyacrylamide gels and electrophoresed under 10% reducing gel conditions. The separated proteins were electroblotted onto Hybond-P:PVDF membranes (Amersham) using a Mini Trans-Blot Cell (Bio-Rad Laboratories, Hercules, CA, U.S.A.) according to the manufacturer's instructions. Membranes were washed once with Tris-buffered saline (TBS), pH 7.2, containing 0.1% Tween-20 (TBS-T) and blocked with TBS-T containing 5% skimmed milk at room temperature for 1 hour. Antibodies against Tie-1 and Tie-2 (1:1000; Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.) were added to the blocking solution and incubated at room temperature for another 2 hours. Membranes then were washed three times with TBS-T, and incubated with horseradish peroxidase-conjugated anti-rabbit IgG (1:3000; Santa Cruz) in TBS-T containing 5% skimmed milk for 1 hour. After four washes with TBS-T, the protein bands were visualized using an enhanced chemiluminescence Western blot analysis system (Pierce, Rockfold, IL, U.S.A.).
Immunohistochemical staining
Frozen brain slices (25-μm-thick) were sectioned and incubated in a free-floating manner. The endogenous peroxidase was blocked with 0.3% hydrogen peroxide for 30 minutes at room temperature. Preincubation with 3% normal horse serum (NHS) containing 0.2% Triton X-100 was performed at room temperature for 60 minutes to block nonspecific binding of immunoglobin G (IgG). Rabbit anti-Tie-1 and anti-Tie-2 antibodies (1:100, diluted in 1% NHS, Santa Cruz) were used in the current study. Sections were incubated with anti-Tie antibodies at 4°C overnight, then rinsed with PBS for 30 minutes, and incubated with biotinylated goat anti-rabbit IgG (1:200 dilution) at room temperature for 1 hour. After several rinses with PBS, sections were incubated with avidin-horseradish-peroxidase complex for 1 hour, then with 3,3′-diaminobenzidine (0.5 mg/mL) in the presence of 0.009% H2O2, and subsequently mounted on gelatinized slides. The specificity of each antiserum was demonstrated by the absence of immunostaining when the diluted primary antiserum was replaced by serum. For additional negative controls, sections were incubated with primary antibody denatured by heat (Lin et al., 1997). The results of these immunohistochemical controls were consistently negative.
Double-staining immunohistochemistry
To ascertain colocalization of various angiogenesis proteins, sections were stained with various combinations of rabbit anti-Tie-1, rabbit anti-Tie-2, goat anti-Angpo-1 (1:100; Santa Cruz), goat anti-Angpo-2 (1:100; Santa Cruz), mouse anti-bFGF (1:100; Merk, Darmstadt, Germany), goat anti-VEGF (1:100; Santa Cruz), mouse anti-TGF-β (1:100; R&D, Minneapolis, MN, U.S.A.), and goat anti-platelet/endothelial cell adhesion molecule-1 (PECAM-1) (1:100; Boehringer Mannheim). The following secondary antibodies were used for the current study: fluorescein isothiocyanate (FITC) linked donkey-anti-rabbit antibody (1:50) for Tie-1/2 detection, donkey-anti-goat-Rhodamine (1:50) secondary antibody for Angpo-1/2 and VEGF detection, donkey anti-mouse-Rhodamine (1:50) and donkey anti-mouse-FITC (1:50) for bFGF detection. After incubation overnight at 4°C, sections were observed under confocal microscopy (Lin et al., 1997).
Assessment of cerebral blood flow by magnetic resonance imaging
Rats were anesthetized with chloral hydrate (360 mg/kg body weight, IP). Each rat was placed in a prone position and was fitted with a custom-designed head holder within the magnet as previously described (Chyi and Chang, 1999). All magnetic resonance (MR) experiments were performed on a 4.7 T Biospec 47/40 spectrometer with an active shielding gradient (5.6 G/cm in 500 microseconds). A 20-cm birdcage coil was used for RF excitation, and a 2-cm diameter surface coil was used for signal reception. The FAIR experiment was implemented with two inversion recovery-fast spin echo sequences (IR-FSE) with and without slice selective gradient during an inversion pulse. Two-slice and nonslice inversion IR-FSE images in an interleaved manner were collected using a repetition time (TR) of 3 seconds, echo time (TE) of 20 milliseconds, and an effective TE of 50 milliseconds with an echo train length of 4, a slice thickness of 2 mm, a field of view of 4 cm, an inversion time (TI) of 1.5 seconds, and a matrix size of 256 × 128. A slab thickness of 5 mm was inverted for the slice inversion IR-FSE images and a hyperbolic secant pulse was used for inversion with a pulse length of 8 milliseconds. FAIR images were generated by the subtraction of nonslice inversion images from their corresponding slice inversion images. FAIR images were obtained using commercially available image analysis software (MRVision, Menlo Park, CA, U.S.A.).
Materials
All chemicals and reagents were purchased from E. Merck (Darmstadt, Denmark) or Sigma Chemical (St. Louis, MO. U.S.A.) unless otherwise indicated.
RESULTS
The authors previously have shown that Tie-1 and Tie-2 mRNA were increased after transient ischemia by Northern blot analysis (Lin et al., 2000). In the current study, in situ hybridization was conducted to further examine the regional expression of Tie-1 and Tie-2 mRNA (Fig. 1). The RT-PCR amplified cDNA fragments, previously used in Northern blot analysis (Lin et al., 2000), were applied here to detect regional distribution of Tie-1 and Tie-2 mRNA. In the sham-operated control, the signal intensity of Tie-1 or Tie-2 mRNA was less than the detectable range. Ischemia–reperfusion led to a marked increase in signal intensity of Tie-1 mRNA in the ischemic (right) MCA cortex. A capillary-like pattern was noted within the ischemic right MCA cortex. Although similar Tie-2 message with capillary-like appearance was also noted in the ischemic area, Tie-2 mRNA did not appear to colocalize with Tie-1 mRNA when expression patterns of these 2 genes were compared in serial brain sections. A substantial increase in signal intensity of the message of these 2 genes was also noted in the periinfarct regions. In the contralateral (left) MCA cortex, the signal intensity of Tie-1 or Tie-2 mRNA was not altered and did not appear different from the basal levels of expressions in the sham-operated control.
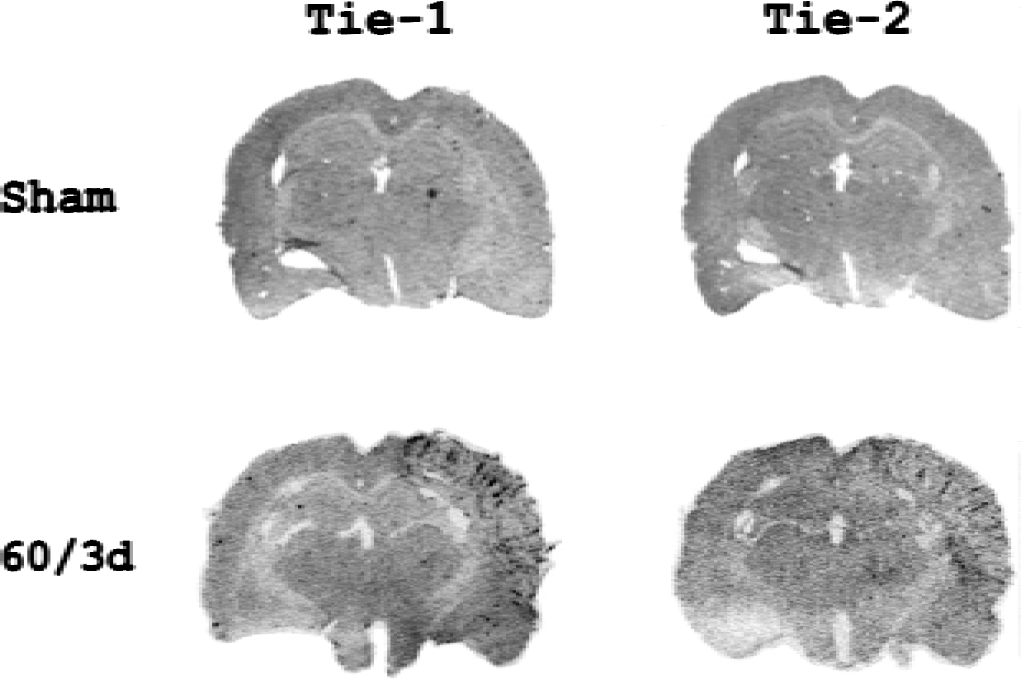
In situ hybridization studies of the regional distribution of Tie-1 and Tie-2 mRNA in the sham-operated and ischemic brains. 60/3d represents 60-minute ischemia and 3-day (72 hours) reperfusion, and sham denotes sham-operated control. Brain slices (25 μm) were hybridized with [33P]-labeled probes as described in the text. Note that an increase in signal intensity of Tie-1 and Tie-2 mRNA was noted in the ischemic right MCA cortex after ischemic insult but not after sham operation. No changes in signal intensity of Tie-1 or Tie-2 mRNA were noted in the contralateral (left) side. Similar results were duplicated in two other sets of animals.
The authors further examined whether increased expression of Tie-1 and Tie-2 mRNA led to the expression at the protein level. The temporal expression profiles of Tie-1 and Tie-2 at the protein level were studied by Western blot analysis (Fig. 2). The specific antibodies used in the current study recognized a 154-kD band of Tie-1 and 147-kD band of Tie-2 protein, respectively. Tie-1 protein was barely detectable in the right MCA cortex of sham-operated rats. The level of Tie-1 protein was transient and slightly increased in the ischemic right MCA cortex 30 minutes after the onset of ischemia and during early reperfusion. A second wave of Tie-1 protein expression of substantially greater intensity was noted at 72 and 168 hours of reperfusion. Similar biphasic expression of Tie-2 protein was noted. However, there were subtle differences in the temporal expression of these 2 gene products. In the first wave of Tie-2 expression, it appeared to be confined only to the ischemic periods and substantially more intense than that of Tie-1. In the second peak, Tie-2 expression appeared to occur earlier and lasted longer than that of Tie-1.
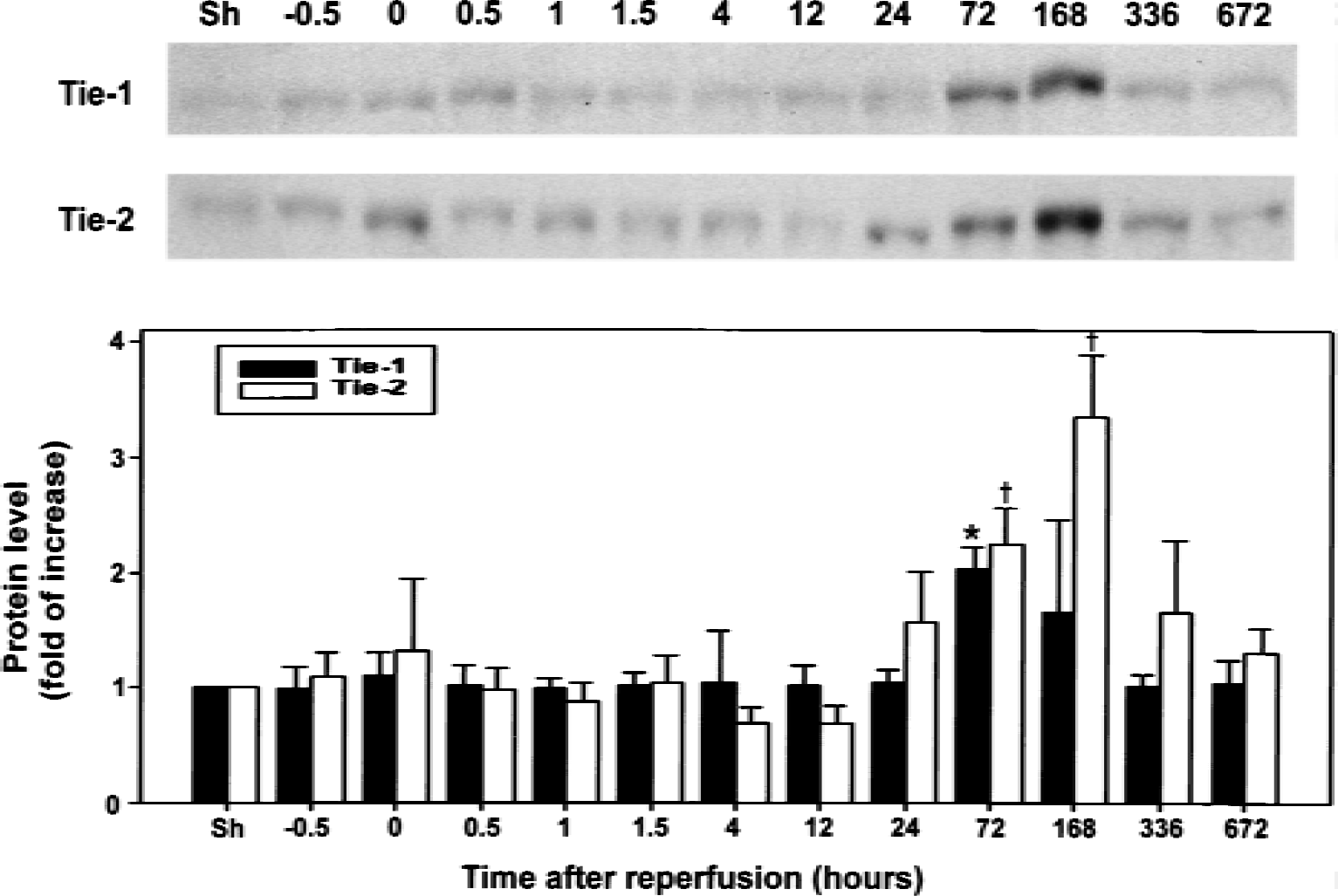
Time course of Tie-1 and Tie-2 protein expression in the ischemic right middle cerebral artery cortex after focal cerebral ischemia–reperfusion. Transient ischemia for 60 minutes resulted in a significant increase in protein levels of Tie-1 (154 kD) and Tie-2 (147 kD) in the ischemic right cerebral cortex. Protein bands were quantified by densitometer and value obtained from the sham-operated control was arbitrarily defined as 1. Data were mean ± SD from 6 animals. Difference from the sham-operated controls was significant at * P < 0.05 and †P < 0.01, respectively. Sh denotes sham-operated controls and −0.5 and 0 represent 30 and 60 minutes after onset of ischemia or 30 minutes and 0 minute before the initiation of reperfusion, respectively. Remaining values denote hours of reperfusion after 60-minute ischemia.
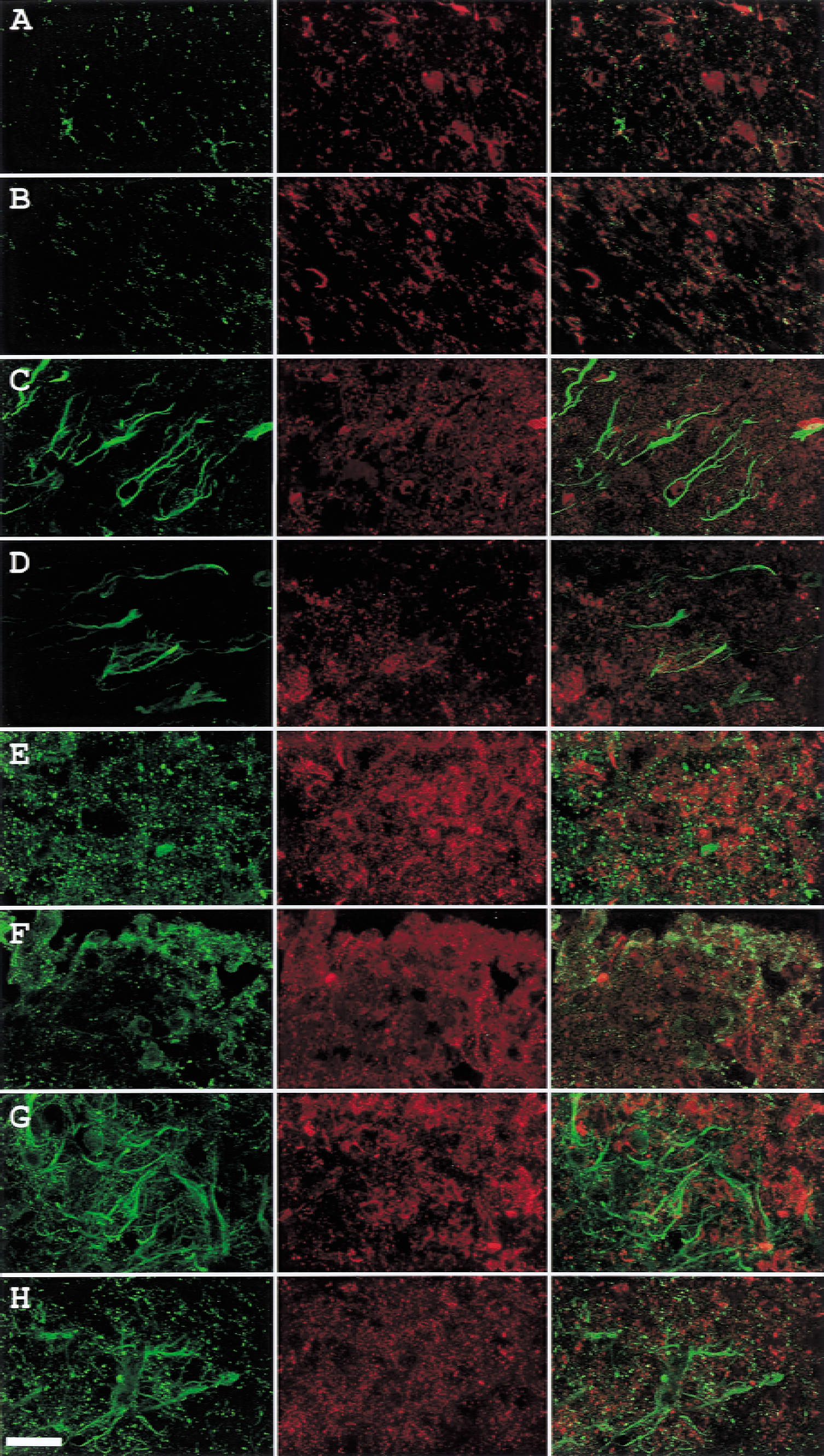
The regional expression of Tie-1 and Tie-2 proteins was examined by immunohistochemistry (Fig. 3). In the current study, coronal brain sections (25 μm) were obtained from rats subjected to 60 minutes ischemia followed by 72 hours reperfusion. All the changes depicted were within the ischemic cortex. The basal level of Tie-1 was low and a substantial increase in Tie-2 signal intensity was detected in large vessels in the contralateral side. Tie-1 immunoreactivity was markedly increased in the ischemic MCA cortex (Fig. 3A). A capillary-like appearance of Tie-1 immunoreactivity was noted in layers 2 to 4 (Fig. 3C). Intense Tie-1 immunoreactivity also was observed in layer 1 (Fig. 3B) and to a lesser extent in layer 6b (Fig. 3D). Tie-2 protein was also markedly increased in the ischemic MCA cortex (Fig. 3E). Although a similar capillary-like Tie-2 immunostaining was noted in approximately the same area (Fig. 3G), subtle differences in regional distribution between Tie-1 and Tie-2 were evident. Cells with Tie-2 immunoreactivity in the cortical layer 1 (Fig. 3F) were morphologically different from those with Tie-1 immunoreactivity (Fig. 3B). Interestingly, a clear vessel-like appearance of Tie-2 immunoreactivity was noted in cortical layer 6b (Fig. 3H), where more diffuse and amorphous Tie-1 immunostaining was detected.
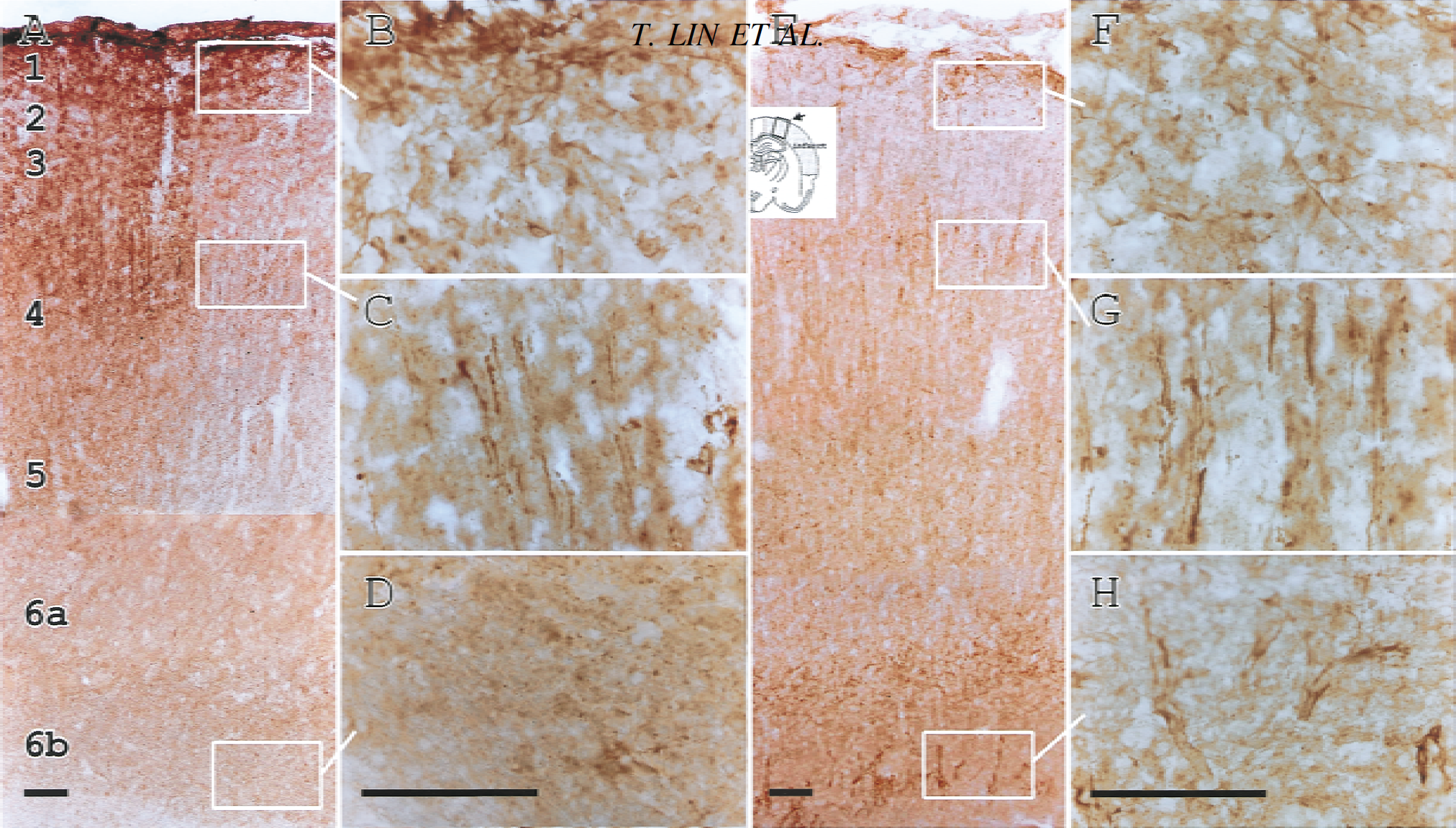
Immunohistochemical studies of Tie-1 and Tie-2 protein expression in the brains of rats subjected to 60-minute ischemia and 72-hour reperfusion. Representative microphotographs highlight the expression in Tie-1
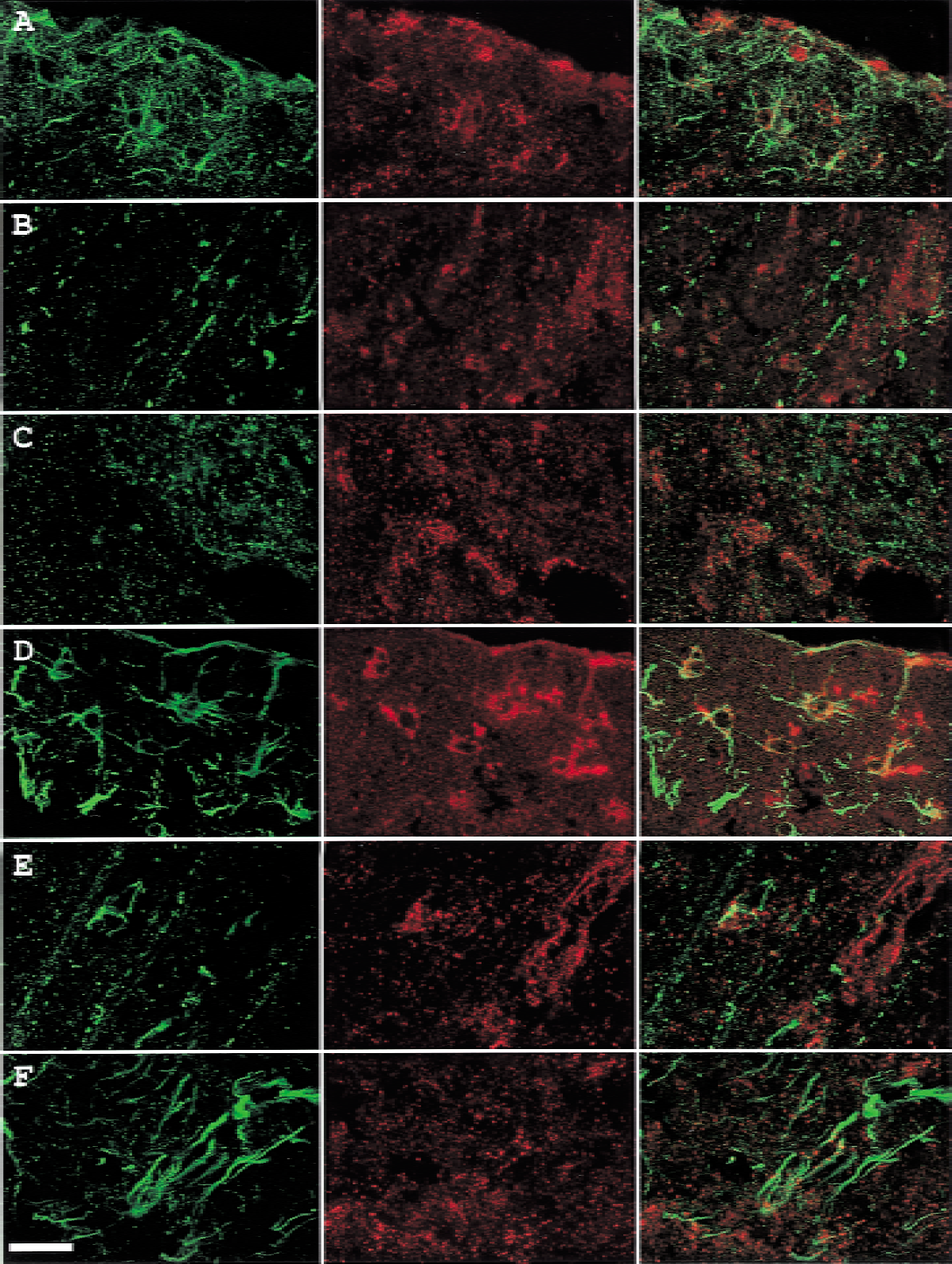
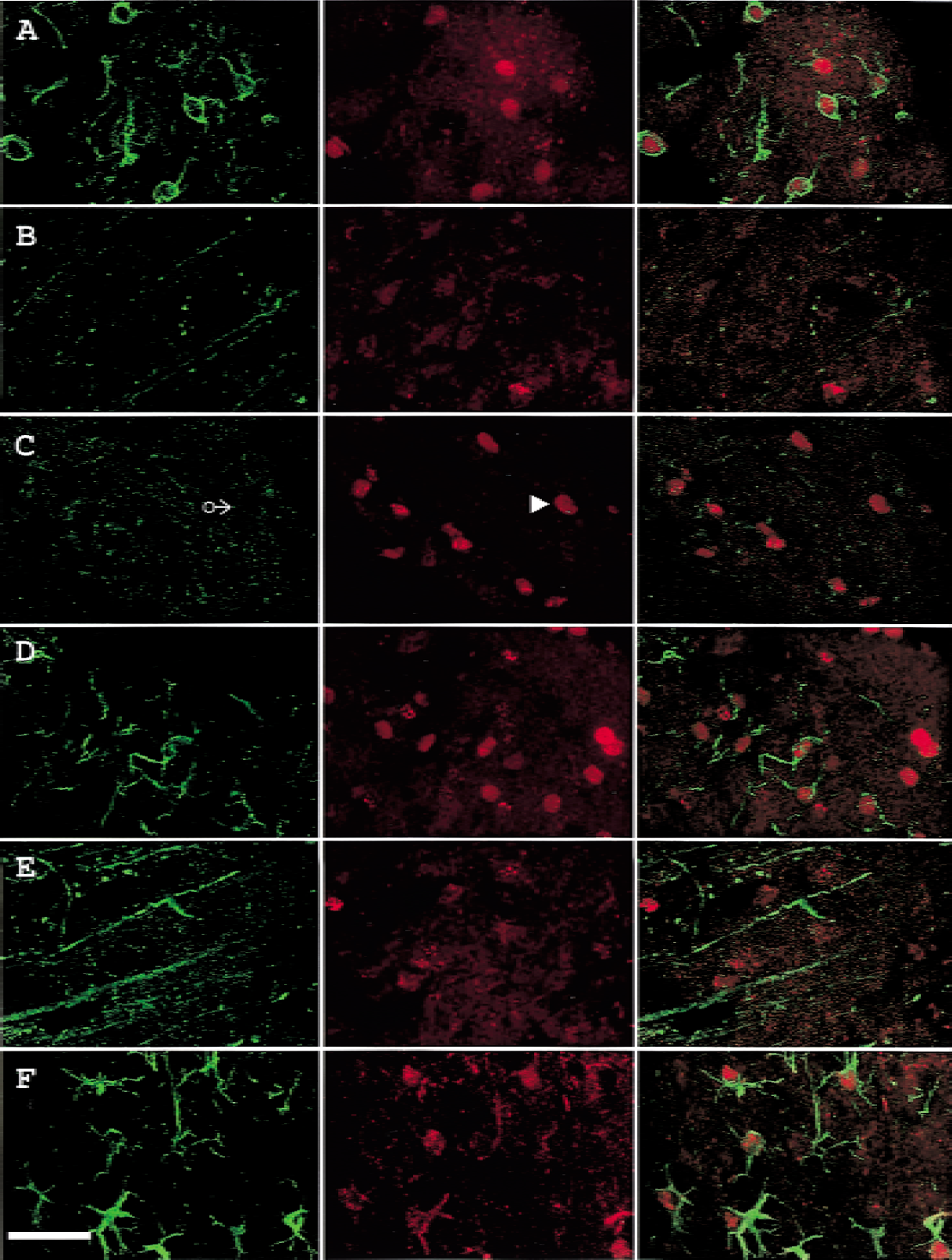
To know whether angiopoietins bound to their receptor, Tie, double-labeling studies were conducted. In cortical layer 1 (Fig. 4A to 4D), Angpo-1 immunoreactivity did not colocalize with Tie-1 (Fig. 4A) or Tie-2 (Fig. 4C). However, some Angpo-2-positive cells expressed Tie-1 (Fig. 4B) and Tie-2 (Fig. 4D) immunoreactivity. A difference in the distribution of Tie-1 and Tie-2 immunoreactivity was again noted. Angpo-2 colocalized with Tie-2 in numerous cells but with Tie-1 only in a few. In cortical layer 4 (Fig. 4E to 4H), the capillary-like Tie-1 (Fig. 4E and 4F) and Tie-2 (Fig. 4G and 4H) immunoreactivity did not colocalize with Angpo-1 (Fig. 4E and 4G) or Angpo-2 (Fig. 4F and 4H). Angpo-2 immunoreactivity formed a dot line-like appearance, reminiscent of capillary structures in this region (Fig. 4F and 4H). In cortical layer 6b (Fig. 5A to 5D), neither Tie-1 (Fig. 5A and 5B) nor Tie-2 (Fig. 5C and 5D) colocalized with Angpo-1 (Fig. 5A and 5C) or Angpo-2 (Fig. 5B and 5D). Even after 168 hours of reperfusion (Fig. 5E to 5H), when Angpo-1 is more intensely expressed than Angpo-2, neither Tie-1 (Fig. 5E and 5F) nor Tie-2 (Fig. 5G and 5H) colocalized with Angpo-1 (Fig. 5E and 5G) or Angpo-2 (Fig. 5F and 5H) in cortical layer 6b.
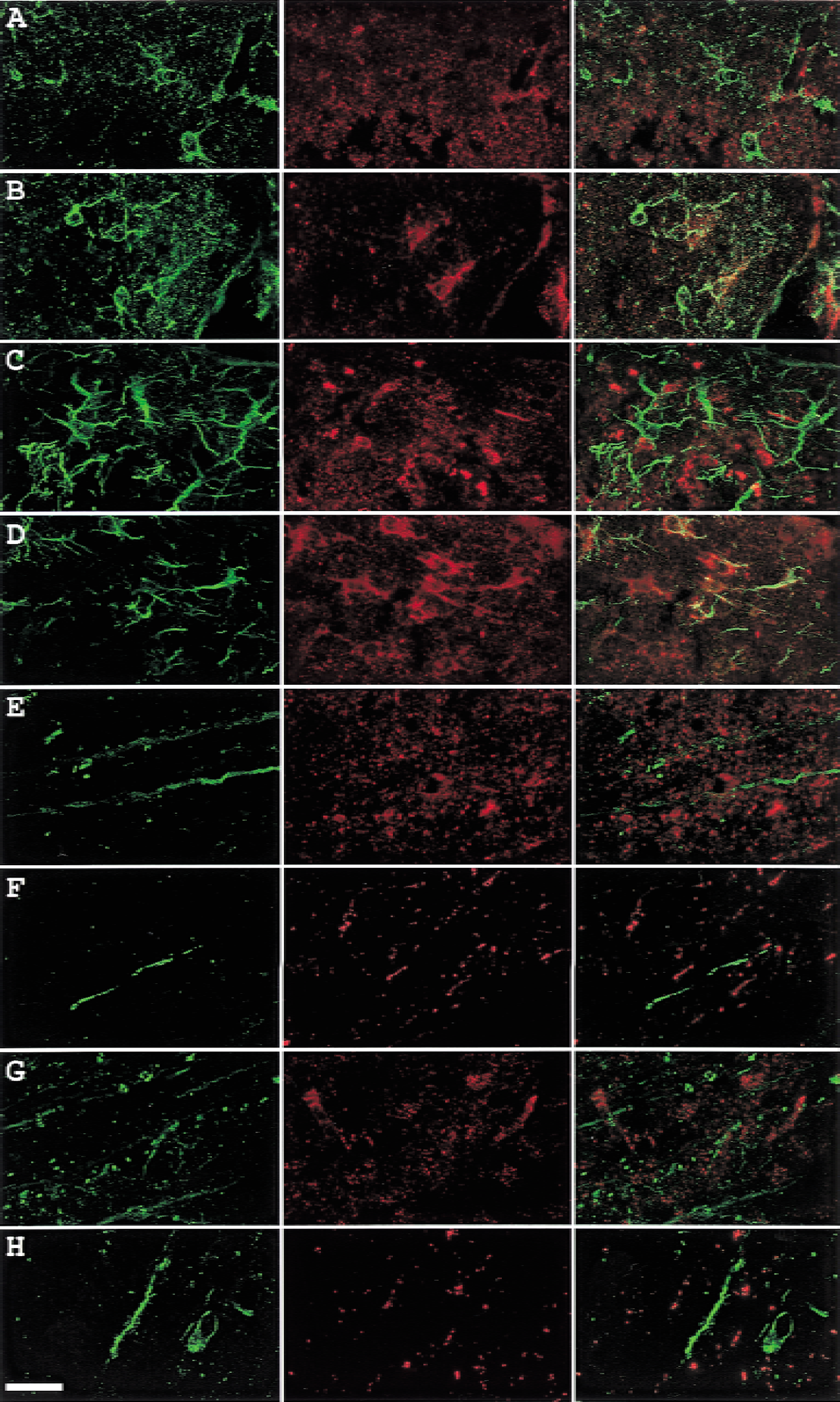
Double labeling of Tie and angiopoietin in the ischemic outer cortical layers of rats subjected to 60-minute ischemia and 72-hour reperfusion.
Double labeling of Tie and angiopoietin in the ischemic cortical layer 6b of rats subjected to 60-minute ischemia and 72-hour
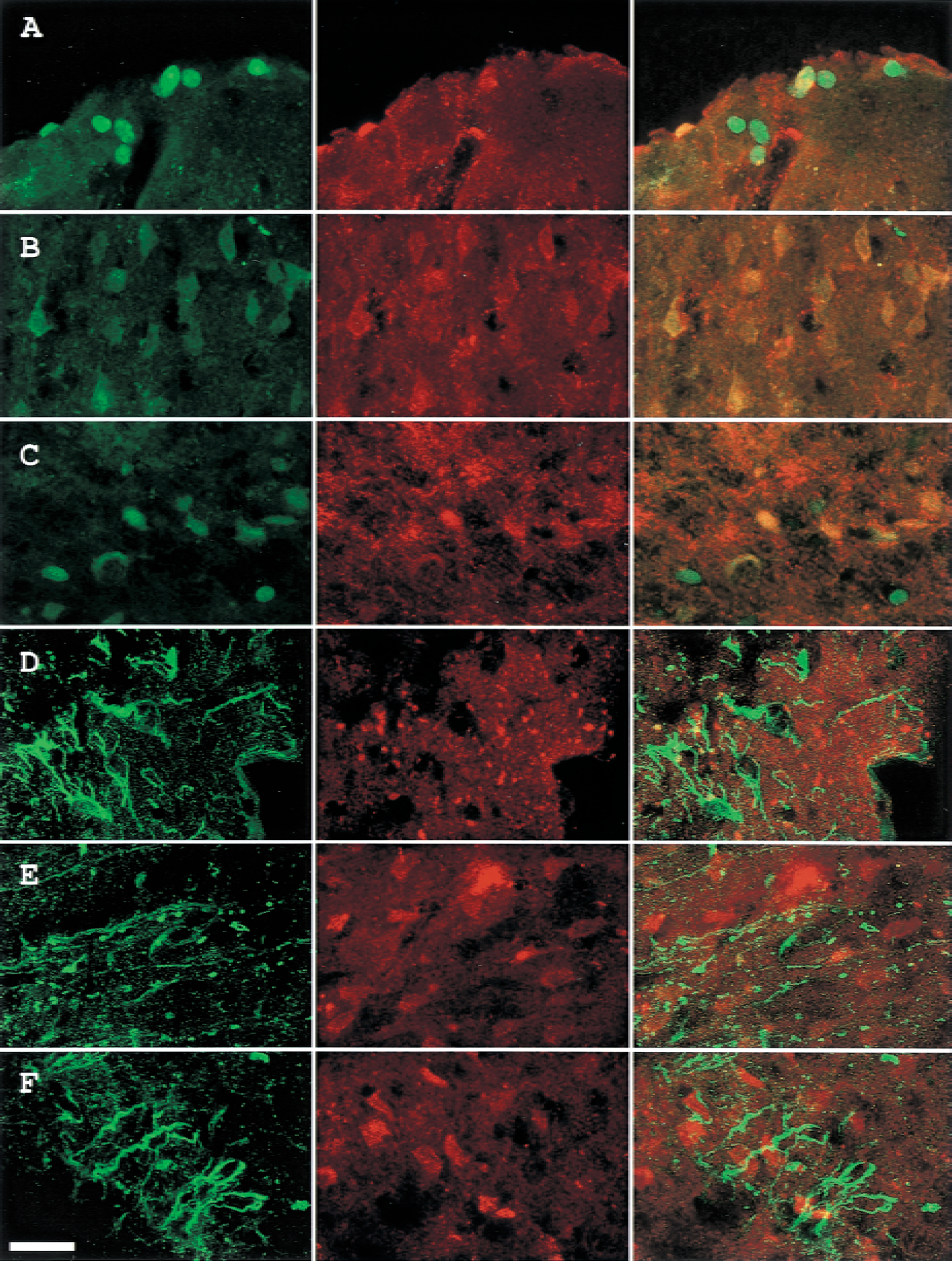
Vascular endothelial growth factor also acts through an endothelial cell-specific receptor tyrosine kinase (VEGF-R) in vascular development. Vascular endothelial growth factor antibody recognized only members of the VEGF-A family, but not the remaining isoforms (VEGF-B to E). Vascular endothelial growth factor immunoreactivity appear to localize with Tie-1 (Fig. 6A) and particularly Tie-2 (Fig. 6D) within the same cells, although VEGF in more in cell body, in cortical layer 1. In cortical layers 4 and 6b, Tie-1 and Tie-2 were not colocalized with cellular staining of VEGF in this region.
Double labeling of Tie and VEGF in the ischemic MCA cortex of rats subjected to 60-minute ischemia and 72-hour reperfusion.
Basic fibroblast growth factor immunoreactivity appears to localize with Tie-1 (Fig. 7A) within the same cells in cortical layer 1, but not layer 4 (Fig. 7B) or layer 6b (Fig. 7C), whereas bFGF immunoreactivity appears to localize with Tie-2 within the same cells in cortical layer 1 (Fig. 7D) and layer 6b (Fig. 7F), but not layer 4 (Fig. 7E). The appearance of bFGF staining pattern suggests that bFGF was distributed mainly within the cell body, whereas Tie immunoreactivity appeared to localize mainly on membrane surface.
Double labeling of Tie and bFGF in the ischemic MCA cortex of rats subjected to 60-minute ischemia and 72-hour reperfusion.
Double staining of bFGF and VEGF (Fig. 8A to 8C) showed various degrees of colocalization in cortical layers 1, 4, and 6b. Basic fibroblast growth factor and VEGF colocalized in a few cells in cortical layer 1 (Fig. 8A), but in larger number of cells in layer 6b (Fig. 8C), and virtually the entire population of visualized cells with polygonal appearance suggestive of neurons in layer 4 (Fig. 8B).
Double labeling of bFGF/VEGF and Tie/TGF-β in the ischemic MCA cortex of rats subjected to 60-minute ischemia and 72-hour reperfusion.
Transforming growth factor-β also affects pathologic angiogenesis and improves the reendothelialization of denuded vessels. Double labeling using antibodies that recognize both Tie-1 and Tie-2 (by adding two individual antibodies together), and an antibody that recognizes TGF-β1, TGF-β2, and TGF-β3 (Fig. 8D to 8F) was also conducted. Results showed that they were not colocalized in layers 1, 4, or 6b (Fig. 8D, 8E, and 8F, respectively). The morphology of TGF-β–positive cells in these three areas was variable. In cortical layer 1, TGF-β immunoreactivity was of a punctuate appearance on an amorphous background, suggestive of a secreted protein. The appearance of TGF-β positive cells, although reminiscent of neurons in cortical layer 4 (Fig. 8E, middle), are suggestive of pericytes and neurons in cortical layer 6b (Fig. 8F, middle).
To further confirm that Tie-1 and Tie-2 are expressed in endothelial cells, the authors conducted a double labeling study showing colocalization of Tie and PECAM-1, a specific cellular marker of endothelial cells. In cortical layer 1, PECAM-1 colocalized with both Tie-1 and Tie-2 (Fig. 9A and 9C), and no colocalization was noted between Tie-1/Tie-2 and glial fibrillary acidic protein (GFAP) (Fig. 9B and 9D).
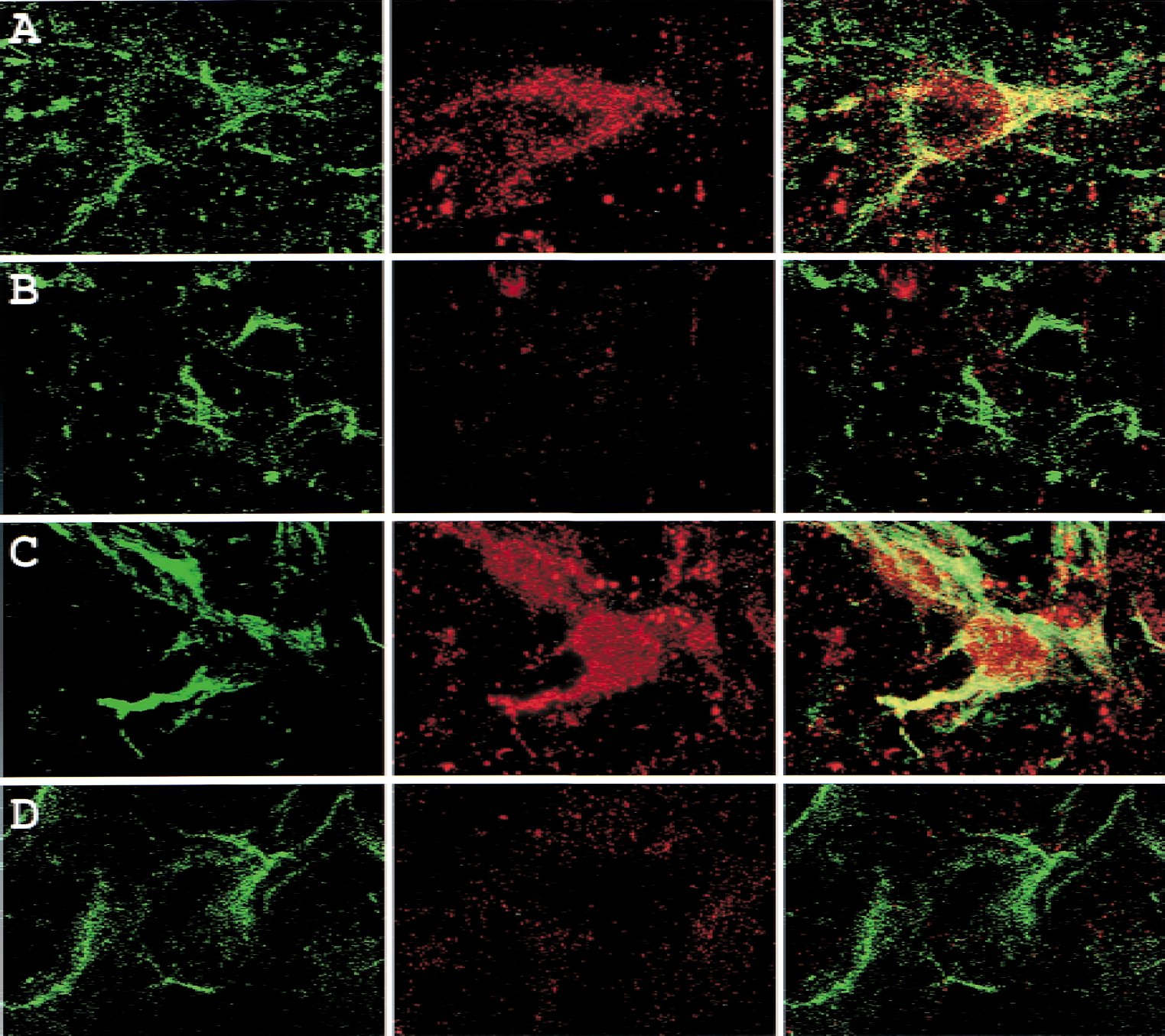
Double labeling of Tie/PECAM-1 and Tie/GFAP in the ischemic MCA cortex layer 1 of rats subjected to 60-minute ischemia and 72-hour reperfusion. (left) Tie-1
The dorsal view of cerebral blood vessels 168 hours after ischemia–reperfusion and sham-operated control were shown in Fig. 10 (top panel). A drastic increase in vascular density was noted in the ischemic (right) MCA cortex as compared with the contralateral side or the sham-operated brain. Figure 10 (bottom panel) shows the corresponding coronal brain sections of MR CBF images measured by FAIR. A substantial increase in CBF was noted in the ischemic cortex as compared with the contralateral cortex or the sham-operated brain. Signal intensity was particularly high in the outer cortical layers of the ischemic MCA cortex corresponding with increased vascular density in the same region.
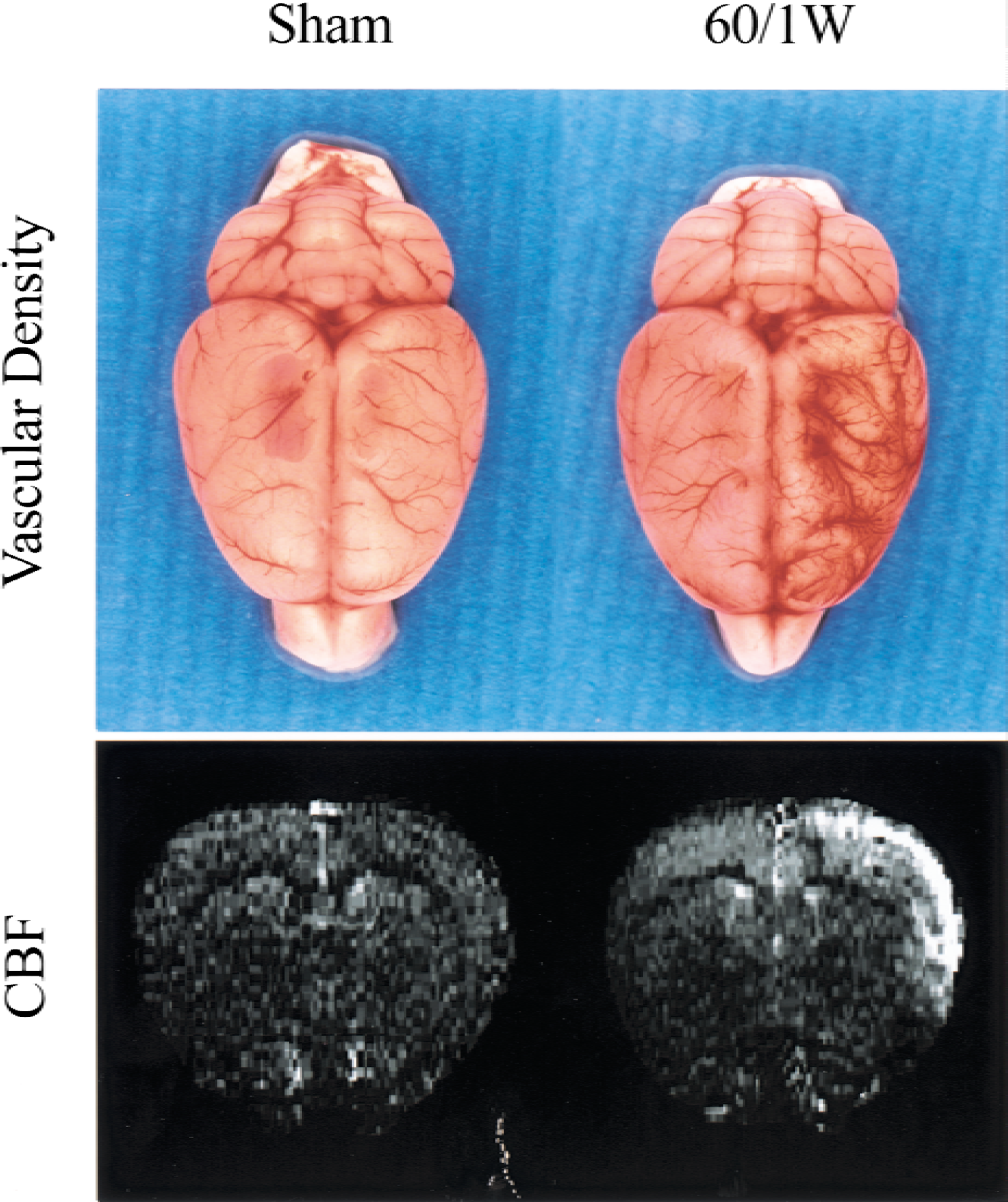
Correlation of vascular density and cerebral blood flow (CBF) after focal cerebral ischemia–reperfusion. Rats were subjected to 60-minute ischemia followed by 168 hours (60/1W) of reperfusion or sham operation (Sham). (top) Dorsal view of cerebral blood vessels. Left hemisphere is the control and right hemisphere is the ischemic side. (bottom) Coronal section of representative CBF imaging measured by FAIR. The highest CBF signal occurred in the outer cortical layers of ischemic region, in which greater vascular density was noted.
DISCUSSION
Angiogenesis, important in maintaining a stable blood supply in the normal physiologic state, is altered in pathologic conditions (Plate, 1999; Griffioen and Molema, 2000; Carmeliet, 2000). The authors previously have reported evolving changes in the expression of Tie-1 and Tie-2 mRNA after focal cerebral ischemia–reperfusion by RT-PCR and Northern blotting (Lin et al., 2000). In the current study, in situ hybridization demonstrated regional distribution of Tie-1 and Tie-2 mRNA and revealed that Tie-1 and Tie-2 mRNA form a capillary-like morphology. The observations based on in situ hybridization are in agreement with those derived from immunohistochemical studies. Western blot analysis strengthened the specificity of Tie-1 and Tie-2 expression at the protein level and indicates transcription of Tie-1 and Tie-2 genes was followed through with the translation of the respective mRNA message. Interestingly, Tie-2 expression at the protein level was rapidly terminated after the onset of reperfusion, suggesting that Tie-2 may be more sensitive than Tie-1 to alteration in oxygen tension or blood flow.
Notably, Tie-1 and Tie-2 each carry characteristic roles in vessel growth and remodeling (Puri et al., 1995; Sato et al., 1995; Partanen et al., 1996). Both Tie-1 and Tie-2 are present in quiescent adult endothelial cell (Dumont et al., 1992; Korhonen et al., 1994). Tie-2 is also present in angiogenic endothelial cells in the adult rats (Wong et al., 1997). In the current study, the regional expression of Tie-1 and Tie-2 immunoreactivity was variable in different cortical layers in the ischemic MCA cortex. It is apparent that Tie-1 and Tie-2 were expressed in different populations of endothelial cells in cortical layers 1 and 6b. This finding is interesting in view of the observations that Tie-1 is more prominent in promoting angiogenic capillary growth and Tie-2 is involved in large vessel remodeling, maintenance of vascular structures, vasculogenesis, and nonsprouting angiogenesis (Puri et al., 1995; Sato et al., 1995; Partanen et al., 1996; Cheung et al., 2000). This may explain, at least in part, why Tie-2 is expressed in cortical layer 6b, where vasculogenesis occurred, and both Tie-1 and Tie-2 are expressed in cortical layer 1, where arteriogenesis (vessels grow both in length and width) and sprouting angiogenesis were noted. Interestingly, Tie-1–positive and Tie-2–positive cells share similar capillary-like morphology in cortical layers 2 to 4. It has been shown that both Tie-1 and Tie-2 are dispensable for the early differentiation of the angioblast, but are needed later for the survival and expansion of the endothelium. Tie-2–null mice lose endothelial integrity and the capillaries appear dilated (Dumont et al., 1994; Sato et al., 1995).
The Tie-1 mutant shows defects in integrity of the microvascular endothelium, resulting in edema and hemorrhaging (Puri et al., 1995; Sato et al., 1995). Therefore, induction of Tie-1 and Tie-2 in this ischemic region probably is to maintain blood vessel integrity (Puri et al., 1999). However, the molecular mechanism and physiologic significance of the diverse expression patterns of these genes in temporal and spatial terms remains to be studied.
Double labeling revealed that Angpo-2, but not Angpo-1, colocalized with Tie-2 in cortical layer 1, where active vascular remodeling took place after ischemia. The intensity of Angpo-2 expression within the ischemic area was much greater than that of Angpo-1 after 3 days of reperfusion; however, this was reversed 1 week after reperfusion (data not shown). This is in agreement with the temporal induction profile of Angpo-1 and Angpo-2 mRNA (Lin et al., 2000). The pathophysiologic significance of a lack of colocalization of Angpo-2 with Tie-2 in cortical layers 4 and 6b, even after one week of reperfusion (Fig. 5, lower half), is not clear. Angpo-2 also colocalized with Tie-1 in some cells in cortical layer 1 but not layer 4 or layer 6b.
To date, at least three families of vascular growth factors that act on respective endothelial cell specific receptor tyrosine kinases have been identified: VEGFs, angiopoietins, and ephrins (Gale and Yancopoulos, 1999). VEGF-A was also up-regulated after ischemia in this MCAO model (Lin et al., 2000). Interestingly, VEGF, like Angpo-2, also colocalized with both Tie-1 and Tie-2 in cortical layer 1 where active vascular remodeling took place, but not in cortical layers 4 or 6b. It has been suggested that Angpo-2 acts in collaboration with VEGF at the front of invading vascular sprouts (Maisonpierre et al., 1997). Because cortical layer 1 is the most active area undergoing vascular remodeling, it is therefore not surprising that VEGF, Angpo-2, and Tie-2 colocalized in this region.
The authors have shown previously that bFGF, another potent angiogenic factor, was also up-regulated with a time course that parallels angiogenesis in this MCAO model (Lin et al., 1997). In the current study, bFGF was colocalized with both Tie-1and Tie-2 in cortical layer 1. However, bFGF only colocalized with Tie-2 in cortical layer 6b. The morphologic differences in bFGF positive cells suggest bFGF plays an important role in both quiescent and angiogenic endothelial cells. No colocalization of bFGF with either Tie-1 or Tie-2 was noted in cortical layer 4.
Although bFGF and VEGF were respectively colocalized with Tie-1 and Tie-2, bFGF and VEGF were only colocalized in selected cells in cortical layers 1 and 6b. These results again suggest the involvement of different populations of endothelial cell during vascular remodeling. However, bFGF and VEGF were well colocalized in cortical layer 4. It has been shown that the major source for bFGF was astrocytes, neuron, and endothelial cells (Lin et al., 1997), whereas the cortical neurons, pial cells, and glial cells were the sources of VEGF (Hayashi et al., 1997; Schratzberger et al., 2000). So it is likely that bFGF and VEGF colocalized in neurons in cortical layer 4. This contention is supported by the neuron-like polygonal morphology of cells with double labeling in this region. It has been shown that even in the infarcted region, some neurons may survive longer than others because of differential vulnerability of different neuronal populations. Indeed, neuron in cortical layer 4 shows the least susceptibility to ischemia-induced cell death (Bermaudin et al., 1998). Recently, Jin et al. (2000) reported direct neuroprotective effect of VEGF in in vitro ischemia. It is very likely that VEGF-positive neuron in layer 4 maybe more resistant to ischemic insult.
Activation of TGF-β can induce change in myo-fibroblasts and pericytes, which may contribute to the formation of quiescent vessels and extracellular matrix, and maintenance of growth control (Risau, 1997; Pepper, 1997). Results of the double-labeling study showed that TGF-β did not colocalize with either Tie-1 or Tie-2 in all cortical layers. The majority of TGF-β–positive cells appeared to be pericytes and neurons.
It has been shown that Tie is a receptor tyrosine kinase expressed exclusively by endothelial cells but not other cell types (Mustonen and Alitalo, 1995). Results of the double-labeling study show that Tie-1 and Tie-2 colocalize with PECAM-1, an endothelial cell-specific marker (Plate, 1999), but not GFAP in cortical layer 1. This is in line with the authors' previous report that no GFAP signal was detected within the ischemic cortex (Cheung et al, 1999). In cortical layer 6b, Tie-2 immunoreactivity also colocalized with PECAM-1, however, neither Tie-1 nor Tie-2 colocalized with PECAM-1 in cortical layer 4 (data not shown). It is obvious that using other cellular marker for various cell types is needed in future studies.
In a previous study (Lin et al., 2000), the authors showed significant increases in vessel sizes and vascular density after transient ischemia–reperfusion. In the current study, MR-based assessment of cerebral blood flow showed that in the same areas, where up-regulation of Tie-1 and Tie-2 were noted, the regional blood flow was substantially increased. Luxury perfusion after ischemic insult has been well documented. The pathophysiologic significance of transient increase in blood flow days to weeks after ischemia remains to be fully delineated. It could serve to increase tolerance of brain tissue to recurrent ischemic insults and may be partially related to a postischemic inflammatory reaction, which may also contribute to trafficking of damaged cellular components.
In summary, the current data demonstrate for the first time that ischemia led to differential expression of Tie-1 and Tie-2, at both the mRNA and protein levels. Tie-2 is colocalized with Angpo-2 as well as bFGF and VEGF in regions of active vessel remodeling. These findings suggest an intimate relationship among these three angiogenic genes. However, bFGF was only partially colocalized with VEGF. Tie-1, to a certain degree, also colocalized with Angpo-2, bFGF, and VEGF. It is likely that Tie-1 and Tie-2 were expressed in different populations of endothelial cells. The molecular mechanisms and pathophysiologic significance in the differential expression of these angiogenesis gene products and variable colocalization in different cortical layers remain to be studied. It is likely that the colocalization of selected angiogenesis gene products in regions of the ischemic cortex is necessary for the postischemic angiogenesis activity reflected by an increase in vascular density and regional CBF.
Footnotes
Acknowledgments:
The authors thank Miss J.J. Chen, Mr. Y.L. Pan, and C.K. Wang for their technical support and Dr. Grace Ku for critical review and editorial assistance.
