Abstract
Promoting neural regeneration after cerebral infarction has emerged as a potential approach for the treatment of stroke. Insulin-like growth factor 1 (IGF-1) possesses both neurotrophic and angiogenic properties. The aim of this study was to determine whether postischemic gene transfer of IGF-1 enhances neurovascular regeneration in a mouse model of permanent focal cerebral ischemia. Long-term cerebral IGF-1 overexpression was achieved with adeno-associated viral vector (AAV) by stereotaxic injection at 24 h after a stroke. Adeno-associated viral vector-green fluorescent protein (GFP) or saline was injected as a control. The success of postischemic gene transduction was confirmed by a strong GFP signal and by increased IGF-1 protein expression in the peri-infarct region. Postischemic gene transfer of IGF-1 significantly enhanced vascular density at 8 weeks after a stroke in the peri-infarct and injection needle tract area compared with AAV-GFP or saline treatment, as shown by immunohistochemical staining with the vascular marker lectin. Furthermore, increased vascular density was associated with improved local vascular perfusion. Immunohistochemical staining with the neuronal progenitor marker, DCX (doublecortin), and the cell proliferation marker, BrdU (5-bromo-2-deoxyuridine-5-monophosphate), indicated that AAV-IGF-1 treatment potently increased neurogenesis compared with AAV-GFP injection. These data show that postischemic treatment of IGF-1 effectively promoted neural and vascular regeneration in the chronic stage of cerebral infarction.
Introduction
There is great enthusiasm to repair the brain after stroke injury because attenuating the acute ischemic damage that often becomes irreversible within a few hours of ischemic onset is difficult in clinical applications. Therefore, repairing the damaged brain and promoting functional restoration during the chronic phase of the disease represent attractive alternative approaches. Within this paradigm, insulin-like growth factor 1 (IGF-1) stands out among many growth factors for its wide spectrum of trophic activities. Insulin-like growth factor 1 possesses both neurotrophic and angiogenic effects, and has been shown to protect the central nervous system from experimental ischemic stroke injury (Guan et al, 2001; Leinninger and Feldman, 2005; Liu et al, 2001; Loddick et al, 1998; Schabitz et al, 2001) and cultured primary neurons from various types of insults (Cheng and Mattson, 1992; Dore et al, 1997). Insulin-like growth factor 1 has also been implicated in having a critical role in cerebral angiogenesis during development and in adulthood (Conti et al, 2004; Lopez-Lopez et al, 2004). Stroke patients with poor outcome are associated with low levels of IGF-1, suggesting that endogenous IGF-1 level affects the evolution of cerebral infarction (Denti et al, 2004).
We have reported that adeno-associated viral vector (AAV) mediated IGF-1 overexpression before ischemic stroke enhanced neurovascular remodeling, and improved functional outcome in a mouse model of permanent focal cerebral ischemia (Zhu et al, 2008). Adeno-associated viral vector-IGF-1 was injected before middle cerebral artery occlusion (MCAO) to allow maximum expression of IGF-1. This experimental paradigm minimizes the use of the viral vector-mediated gene transfer in clinical applications. Delivery of trophic growth factors after injury is more relevant to clinical settings, and has therapeutic potential. In this study, we performed postischemic gene transfer of IGF-1 to assess its neural restorative effects.
Materials and methods
Experimental Stroke Model
The study was approved by the University of California, San Francisco Committee of Animal Research and conformed to the NIH Guidelines for the use of animals in research. Adult CD-1 mice weighing 30 to 35 g were subjected to permanent focal ischemia by distal MCAO (Zhu et al, 2008). The left middle cerebral artery was occluded by electrical coagulation just proximal to the pyriform branch. Body temperature was maintained at 37°C ± 0.5°C by using a thermal blanket throughout the surgical procedure. Surface cerebral blood flow (CBF) was monitored during MCAO using a laser Doppler flowmeter (Vasamedics, St Paul, MN, USA). Mice with surface CBF > 15% of the baseline were excluded from the experiment.
Gene Transduction with Adeno-Associated Viral Vector
Viral transduction was performed as described (Zhu et al, 2008). Briefly, 24 h after MCAO, mice were placed in a stereotactic frame (Kopf, Tujunga, CA, USA) under anesthesia, and a burr hole was drilled 2.5 mm lateral to the sagittal suture and 1 mm posterior to the coronal suture. A 10-μL Hamilton syringe (Reno, NV, USA) was slowly inserted into the left caudate nucleus (3.0 mm deep from the dura). A volume of 4 μL viral suspension (AAV-IGF-1 or AAV-green fluorescent protein (GFP)) containing 2 times 1010 vector genomes were injected into the left hemisphere (injection sites are depicted in Figure 1A). Recombinant AAV serotype 2 containing human IGF-1 or GFP was provided by Ceregene (San Diego, CA, USA). Adeno-associated viral vector serotype 2 has been shown to transfect only postmitotic neurons (Burger et al, 2005b). A systemic coadministration of mannitol has been shown to profoundly amplify recombinant AAV serotype 2 transduction efficiency (Burger et al, 2005b). Thus, in our experiments, the mice were injected intraperitoneally with 3 mL of sterile 25% mannitol in 0.9% saline per 100 g body weight 15 mins before intracerebral vector injection.
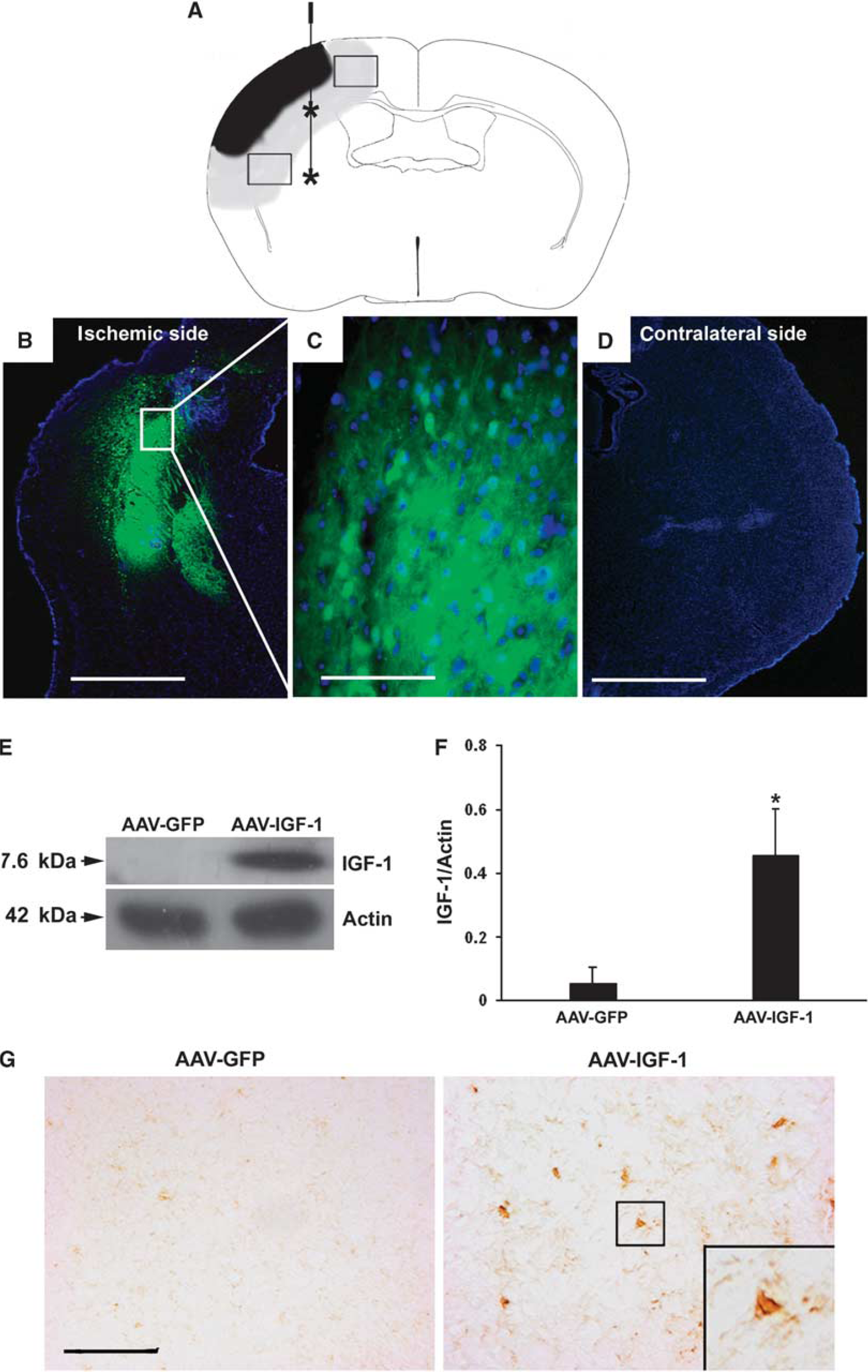
AAV-media ted cerebral gene transduction. (
Administration of 5-Bromo-2-Deoxyuridine-5-Monophosphate (BrdU)
5-Bromo-2-deoxyuridine-5-monophosphate, a thymidine analog incorporated into the DNA of dividing cells, was used to track proliferating cells. Before the killing, mice were injected intraperitoneally twice daily with BrdU (50 mg/kg, Sigma, St Louis, MO, USA) for 7 consecutive days.
Cerebral Blood Flow Measurement
A laser Doppler flowmetry monitor equipped with a small-caliber probe 0.7 mm in diameter was used to measure surface CBF. The laser Doppler probe was in contact with the surface of the mice's skull bone during measurement. Blood flow was recorded in the ischemic penumbra area, which is 1.0 mm lateral to the sagittal suture and 1 mm posterior to the coronal suture. Baseline blood flow was recorded 5 mins before MCAO. Changes in CBF were measured immediately, and thereafter at 2, 4, and 8 weeks after MCAO. Blood flow values were calculated and expressed as the percentages of baseline values (Shen et al, 2006).
Western Blot Assay
The protein concentration was determined using the BCA protein assay kits (Pierce, Rockford, IL, USA). Equal amounts of proteins were loaded on 10% acrylamide gel for electrophoresis, and were electroblotted onto a PVDF (polyvinylidene fluoride) membrane. The membranes were then probed with mouse anti-IGF-1 antibody (1:500; Upstate, Billerica, MA, USA), which reacts specifically with IGF-1 that originates from humans. After the probing, the membranes were then incubated with horseradish-peroxidase-conjugated sheep antimouse IgG (Bio-Rad Laboratories, Hercules, CA, USA). Protein expression was detected using an enhanced chemiluminescence detection system (Amersham Pharmacia Biotech, Piscataway, NJ, USA). The image was scanned with a Canon imaging densitometer (Canon, Lake Success, NY, USA), and protein band densities were quantified using the NIH Image J program. Insulin-like growth factor-1 band intensities were normalized to the band intensities of β-actin, which was used as a loading control.
Immunohistochemical Staining
Immunohistochemical staining was performed as described (Shen et al, 2006). Briefly, sections were incubated with primary antibodies at the following concentrations: mouse anti-IGF-1 (1:100; Upstate); rat anti-CD31 (1:200; BD Bioscience, San Jose, CA, USA); mouse anti-BrdU (1:1,000; Sigma); rabbit anti-doublecortin (DCX) (1:200; Cell Signaling, Danvers, MA, USA); rabbit anti-Tuj-1 (1:2,000; Covance, Princeton, NJ, USA). After incubating at 4°C overnight and washing, the sections were incubated with biotinylated secondary antibody (Vector Laboratories, Burlingame, CA, USA) at a dilution of 1:5,000. The sections were treated using the ABC streptavidin Vector Laboratories, Burlingame, CA, USA) detection system. For dual fluorescent staining, after incubating with primary antibodies, sections were incubated with Alexa Fluor 594-conjugated or Alexa Fluor 488-conjugated IgG (Molecular Probes, Carlsbad, CA, USA) at a dilution of 1:500. Negative controls were performed by omitting the primary antibodies during the immunostaining.
Measurement of Vascular Density
Microvessel counting was performed as described (Shen et al, 2006). Briefly, four brain coronal sections from the lectin-stained brain sections adjacent to the needle track were chosen. Three areas of microvessels, immediately to the left, right, and bottom of the needle track, were photographed using a × 10 objective. Three random areas in the peri-focal region were also photographed. Microvessel and cell counting (BrdU and CD31 dual-labeled cells) were performed on these photographs. Vessels with a diameter between 3 and 8 μm were counted. The number of microvessels was calculated as the mean of the vascular counts obtained from three pictures and the number of cells was calculated in the same manner. The counting was conducted in a blinded manner.
Cell Counting
Quantification of DCX, Tuj-1, or BrdU-positive cells was performed as described (Chen et al, 2005; Zhu et al, 2008). Briefly, DCX, Tuj-1, or BrdU-positive cells in the ipsilateral subventricular zone (SVZ) were digitized under a × 20 objective using a spot camera (Leica, Solms, Germany) equipped with the NIH image J software. To clearly differentiate positively stained cells from the background, the digitalized images were contrast enhanced and a threshold parameter was established to assess the proportion of the immunoreactive region within a fixed field of view. The thresholds were selected with a ‘set-color threshold’ feature in the Image J software, which permits the user to select pixel regions that are considered positive. After establishing these indices, the same parameter was applied to all images acquired under the same magnification and light intensities on slides that were processed identically. The signals were counted using four sections from each mouse and the numbers were averaged. The data were presented as numbers of positive cells per microscopic field. Cell counting was performed by an investigator blinded to the experimental groups.
Statistical Analysis
Data were presented as mean ± s.d. Parametric data from the AAV-IGF-1, AAV-GFP, and saline-treated groups were compared using an one-way ANOVA (analysis of variance), followed by Fisher's protected least significant difference test, as appropriate. A
Results
Postischemic Gene Transduction and Expression with Adeno-Associated Viral Vector
We first examined the levels of GFP expression after intraparenchymal AAV-GFP injection to determine whether postischemic gene transduction was successful. A strong GFP signal was observed adjacent and distal to the injection tract, as well as in the periinfarct region at 8 weeks after MCAO (Figures 1B and 1C). We observed a positive GFP signal as early as 2 weeks after AAV transduction (data not shown). The GFP signal was not noted in the contralateral noninjected hemisphere (Figure 1D). Western blot analysis showed that postischemic AAV-IGF-1 tranduction resulted in increased IGF-1 protein levels compared with AAV-GFP-administered mice (Figures 1E and 1F,
Insulin-Like Growth Factor 1 Gene Transduction Enhances Neovascularization
Insulin-like growth factor 1 has been shown to be a potent angiogenic factor required for cerebral angiogenesis (Lopez-Lopez et al, 2004). We determined whether IGF-1 overexpression would induce neovascularization. Immunostaining with the vascular marker lectin showed increased vascular density in the penumbra of AAV-IGF-1-treated hemispheres (73 ± 14, number of microvessels per field) compared with that in AAV-GFP- (45 ± 15) or saline-injected hemispheres (43 ± 13) (Figures 2A and 2B,
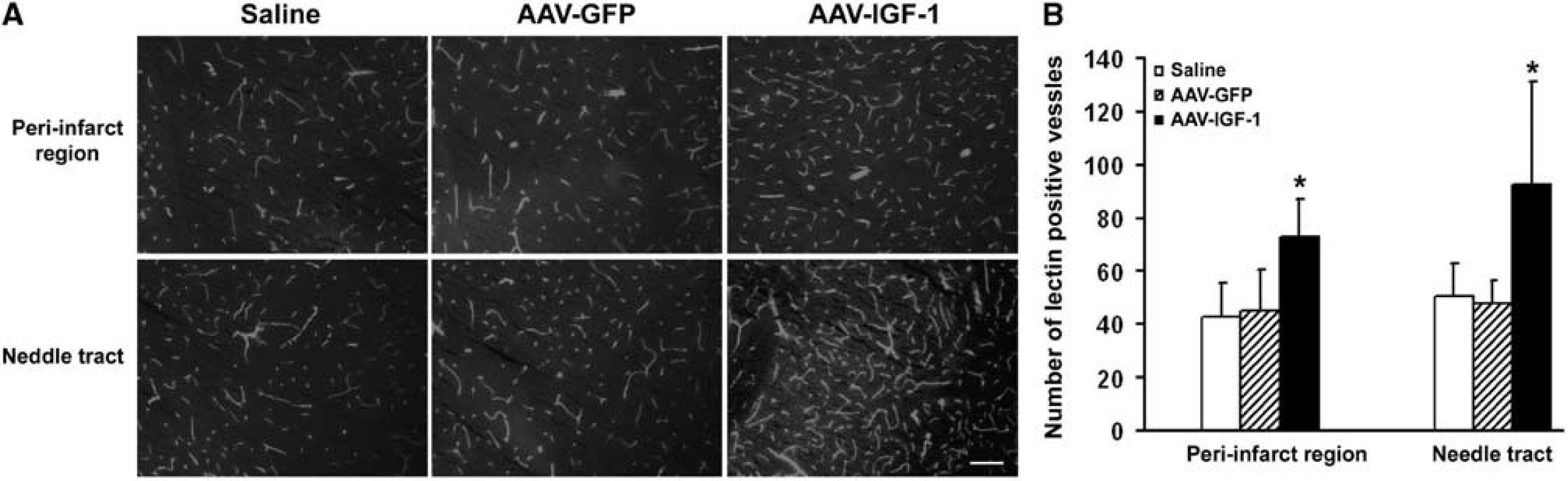
IGF-1 overexpression induces angiogenesis. (
Double immunofluorescent staining showed that BrdU-positive cells colocalized with the endothelial cell marker, CD31, with higher numbers of BrdU and CD31 double-labeled cells in IGF-1-administered groups compared with the AAV-GFP treatment (Figures 3A and 3B, 3.2 ± 2.6 versus 1.4 ± 1.1,
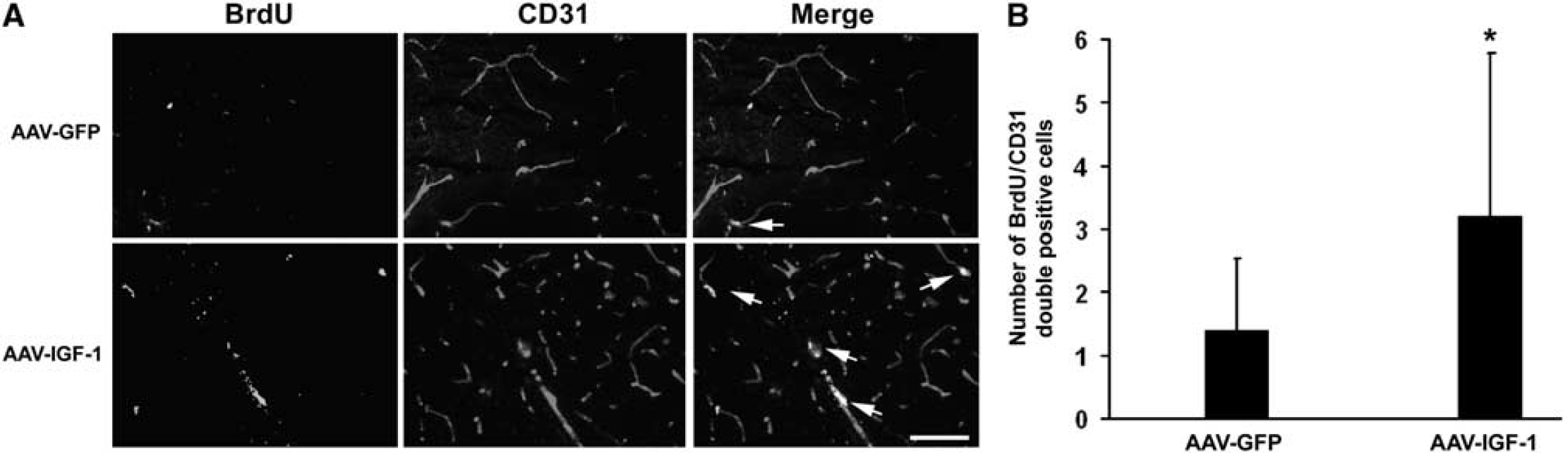
IGF-1 gene transfer increases endothelial cell proliferation. (
Local blood flow in the ischemic cortex
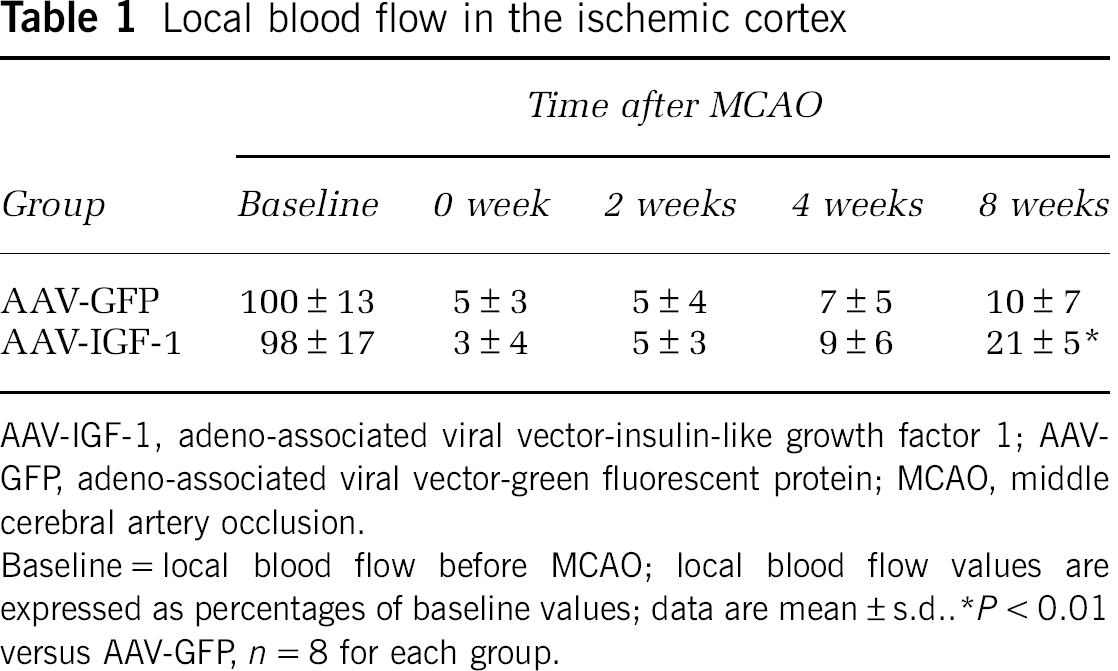
AAV-IGF-1, adeno-associated viral vector-insulin-like growth factor 1; AAV-GFP, adeno-associated viral vector-green fluorescent protein; MCAO, middle cerebral artery occlusion.
Baseline = local blood flow before MCAO; local blood flow values are expressed as percentages of baseline values; data are mean ± s.d.
Insulin-Like Growth Factor 1 Gene Transfer Enhances Neurogenesis
To determine whether the IGF-1 gene transduction promotes neurogenesis, we conducted immunohistochemical analysis with BrdU (a cell proliferation marker) and DCX (a specific marker for neuronal progenitor cells). As shown in Figures 4A and 4B, IGF-1 overexpression resulted in a marked increase of DCX-positive cells in the SVZ compared with AAV-GFP administration (24 ± 13 versus 7 ± 6,
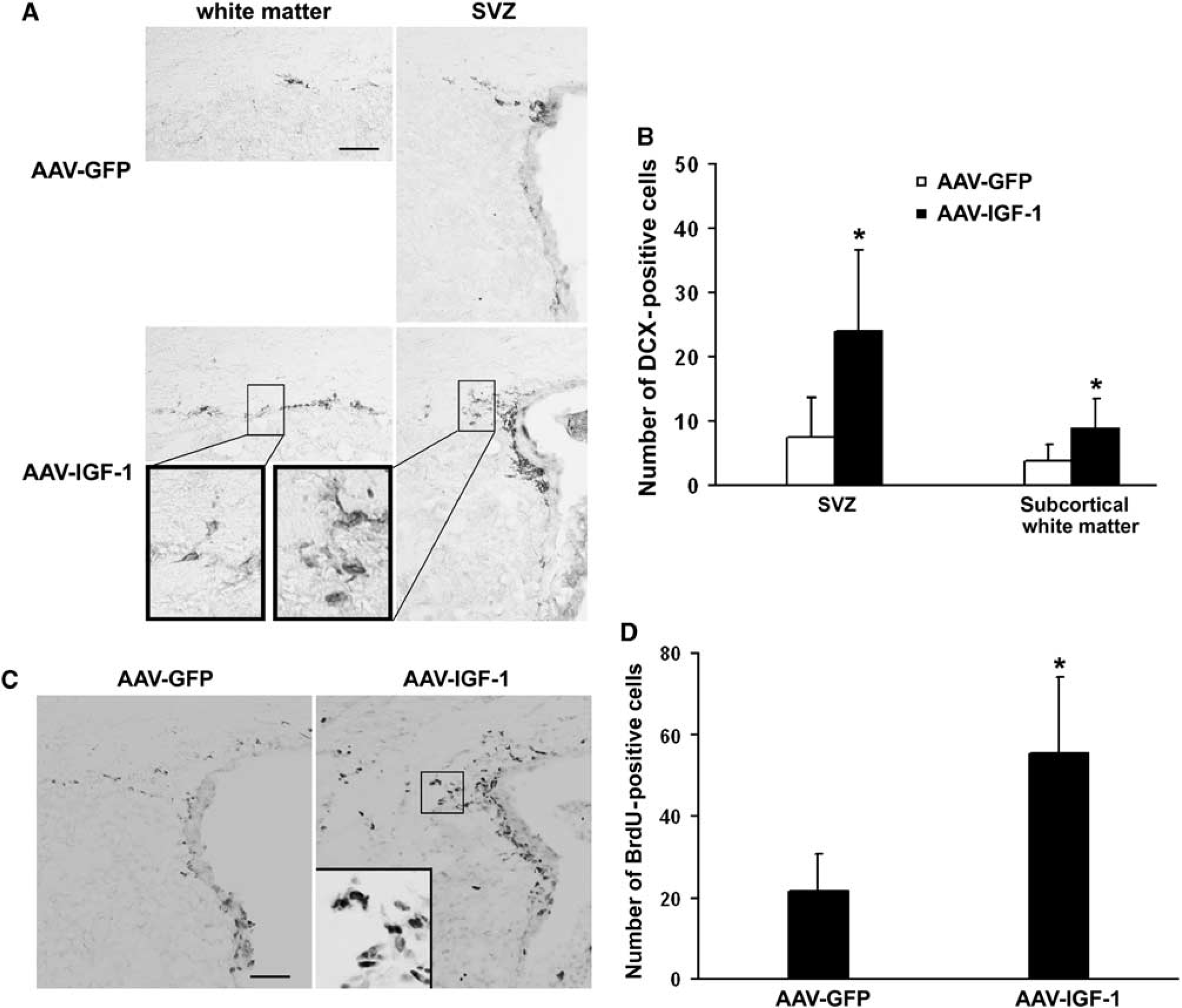
IGF-1 overexpression enhances neurogenesis. (
In the SVZ, there was a significant increase in BrdU-positive cells in AAV-IGF-1-injected mice compared with those treated with AAV-GFP (Figures 4C and 4D, 56 ± 19 versus 22 ± 9,
Using double-fluorescent staining, cells positive for both BrdU and DCX were considerably increased in AAV-IGF-1-administered mice compared with AAV-GFP-injected mice in the SVZ (Figures 5A and 5B, 17 ± 7 versus 5 ± 3,
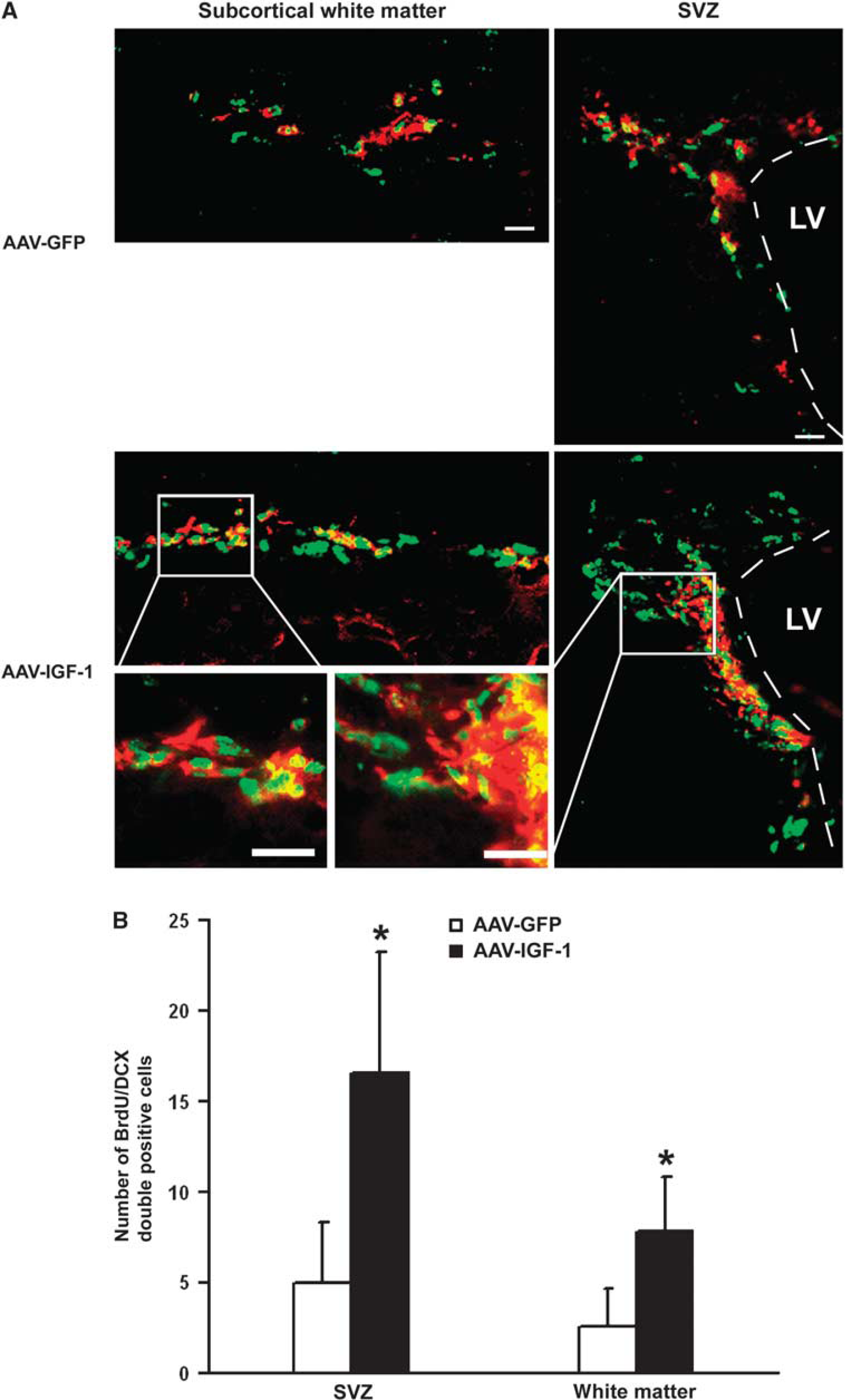
Colocalization of DCX- and BrdU-positive cells. (
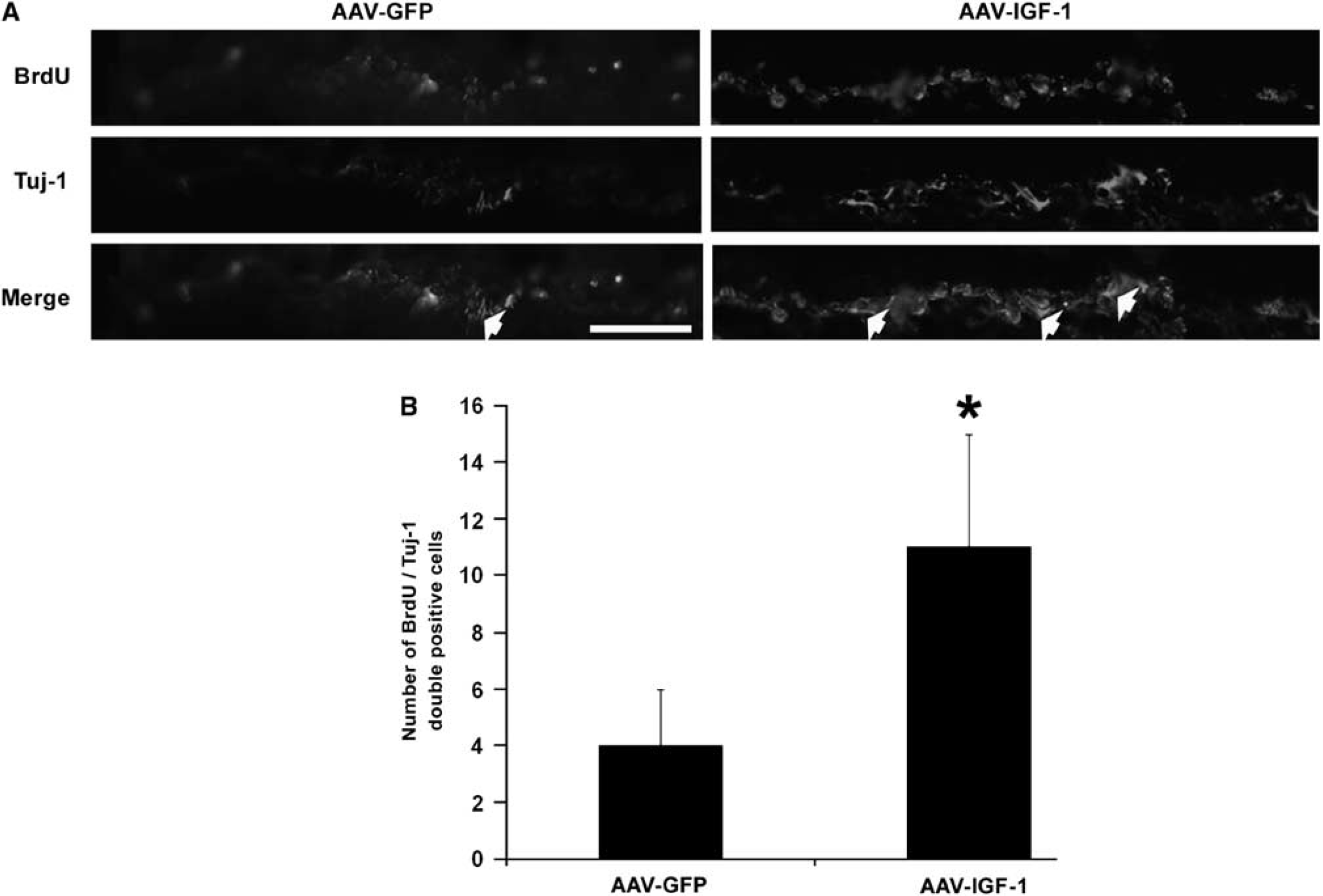
Colocalization of Tuj-1- and BrdU-positive cells. (
Discussion
This study shows that postischemic IGF-1 gene transfer enhanced angiogenesis and promoted neurogenesis. We used AAV to deliver IGF-1 to the ischemic brain. The advantage of gene transfer is that a single injection of vector can lead to efficient production of proteins over a large target area for a sustained period of time. Protein expression after AAV mediated gene delivery follows a well-characterized kinetic pattern that consistently shows a gradual ‘ramp up’ of expression, with relatively low levels for the first several days to 2 weeks after administration and gradually reaching an asymptote at somewhere between 3 and 6 weeks (Burger et al, 2005a). Compared with adenoviral vectors, the AAV vector possesses several advantages, such as low inflammatory potential and the ability to mediate long-term transgene expression (Burger et al, 2005a). Our western blot assay and immunohistochemical staining showed that IGF-1 overexpression was maintained for at least 8 weeks after AAV transfer. On the basis of extensive previous work with AAV vectors and the cytomegalovirus early enhancer/chicken β actin (CAG) promoter, we expect this vector construct to provide a steady expression of IGF-1 for several years. Although we did not measure protein levels for a long term in this study, our previous studies with the same vector and promoter show that maximum levels and volumes of expression are reached at ~1 month, and remain steady thereafter for up to 2 years (Gasmi et al, 2007; Herzog et al, 2008). Whether continuous release of IGF-1 will lead to aberrant vascularization/regeneration is a topic for future consideration through a formal safety/toxicity program, before translating this work to clinical practice.
The acute neuroprotective action of IGF-1 has been well shown (Dluzniewska et al, 2005; Mackay et al, 2003). We have previously reported that the IGF-1 gene transfer before ischemic stroke promoted neurovascular regeneration and improved neurologic functional recovery (Zhu et al, 2008). With pretreatment, the improved neurologic functions could also be contributed by the neuroprotective effects of IGF-1 in the acute phase of a stroke. In this, we focused our effort on assessing the restorative effects of IGF-1 by using postischemic gene transfer. Posttreatment is relevant to clinical settings and has therapeutic use. We showed in this study that postischemic IGF-1 gene transduction, compared with GFP injection, efficiently enhanced neurovascular remodeling assessed at 8 weeks after MCAO.
Our results show that IGF-1 overexpression resulted in increased vascular density in the peri-infarct region. In addition, neovascularization was associated with increased local blood flow. Improved vascular perfusion could lead to long-term functional recovery by supporting neural cell survival, promoting neurogenesis, and facilitating the removal of necrotic debris. Consistent with this view, several reports have shown that enhanced vascular perfusion by neovessel formation leads to functional improvement after a stroke (Gertz et al, 2006; Sun et al, 2003; Wei et al, 2001).
Ischemic injury and various growth factors have been reported to enhance neurogenesis (Gross, 2000). Insulin-like growth factor 1 protein infusion through an osmotic minipump has been reported to increase stroke-induced progenitor cell proliferation in hypertensive rats (Dempsey et al, 2003). Consistent with this finding, we showed in this study that the IGF-1 gene transfer enhanced the number of neuronal progenitor cells in the SVZ area of infarcted hemispheres compared with ischemic-injured hemispheres with GFP treatment. Furthermore, the IGF-1 gene transfer promoted the migration of neuronal stem cells away from the SVZ, as increased DCX-positive cells were observed in the subcortical white matter area. Our data suggest that IGF-1 may promote the healing of the ischemic brain by amplifying endogenous neurogenesis. Angiogenesis and neurogenesis are temporally and spatially coupled processes (del Zoppo, 2006; Ohab et al, 2006). Blood vessels and neuronal fibers develop side-by-side and guide each other to migrate to the target area. Angiogenic vascular cells synthesize and secrete trophic factors, such as VEGF or SDF-1, to promote neural progenitor cells to proliferate and migrate to the ischemic-injured area (Ohab et al, 2006).
Taken together, our data show that postischemic AAV-IGF-1 treatment promotes neurovascular regeneration. We used a permanent focal ischemia model by distal MCAO for establishing the potential efficacy of AAV-IGF-1. This stroke model has been shown to induce reproducible cortical infarcts in a peri-focal penumbra zone with inadequate perfusion (Chan et al, 1993; Chen et al, 1986; Matsumori et al, 2006). The peri-focal area is the ideal therapeutic target for enhancing vascular perfusion and for preventing infarction process. As clinical strokes are heterogeneous, testing this concept in additional models of stroke is a worthwhile endeavor. Furthermore, whether poststroke treatment with AAV-IGF-1 can improve neurologic functions warrants further investigation, as IGF-1 overexpression through AAV-mediated delivery has the potential to be developed into a therapeutic modality for the restoration of the ischemic-injured brain.
