Abstract
The biochemical characteristics of white matter damage (WMD) in preterm infants were assessed using magnetic resonance spectroscopy (MRS). The authors hypothesized that preterm infants with WMD at term had a persisting cerebral lactic alkalosis and reduced N-acetyl aspartate (NAA)/creatine plus phosphocreatine (Cr), similar to that previously documented in term infants weeks after perinatal hypoxia–ischemia (HI). Thirty infants (gestational age 27.9 ± 3.1 weeks, birth weight 1122 ± 445 g) were studied at postnatal age of 9.8 ± 4.1 weeks (corrected age 40.3 ± 3.9 weeks). Infants were grouped according to the presence or absence of WMD on magnetic resonance (MR) images. The peak area ratios of lactate/Cr, NAA/Cr, myo-inositol/Cr, and choline (Cho)/Cr were measured from an 8-cm3 voxel in the posterior periventricular white matter (WM) using proton MRS. Intracellular pH (pHi) was calculated using phosphorus MRS. Eighteen infants had normal WM on MR imaging; 12 had WMD. For infants with WMD, lactate/Cr and myo-inositol/Cr were related (P < 0.01); lactate/Cr and pHi were not (P = 0.8). In the WMD group, mean lactate/Cr and myo-inositol/Cr were higher (P < 0.001, P < 0.05, respectively) than the normal WM group. There was no difference in the NAA/Cr, Cho/Cr, or pHi between the two groups, although pHi was not measured in all infants. These findings suggest that WMD in the preterm infant at term has a different biochemical profile compared with the term infant after perinatal HI.
Keywords
Our understanding of the mechanisms of injury to the preterm brain has not kept pace with the technology that can support these infants while they are critically ill. The remarkable advance in neonatal intensive care in the last two decades has improved the chances of survival in very low birth weight infants. Although the incidence of neurodevelopmental sequelae in these infants is less now than in the mid-1980s, it remains unacceptably high (Hagberg et al., 1996). The brain lesion that is the most significantly associated with a poor neurodevelopmental outcome in these preterm infants is periventricular white matter damage (WMD) (Perlman, 1998).
White matter damage is poorly understood but encompasses two main diseases: (1) cystic periventricular leukomalacia (PVL) occurring in 3% to 4% of infants with a birth weight less than 1500 g, and (2) hemorrhagic parenchymal infarction (HPI) occurring in 10% to 15% of infants with a birth weight less than 1000 g (Perlman et al., 1998). Extensive posterior PVL is invariably associated with later spastic diplegia and visual and cognitive defects; depending on the size of the HPI, the prognosis can be poor, with motor and cognitive deficits (Perlman, 1998). Pathologic features of PVL are distinctive and consist of both focal periventricular and diffuse cerebral white matter (WM) necrosis. Cellular aspects include focal coagulation necrosis, axonal rupture, necrosis of oligodendroglia, infiltration and activation of microglia, proliferation of hypertrophic astrocytes, and endothelial hyperplasia (Volpe, 1995). Hemorrhagic parenchymal infarction remains a significant problem in infants weighing less than 1000 g, the pathogenesis being closely intertwined with the commonly associated intraventricular hemorrhage. At a cellular level, there is hemorrhagic infarction with widespread necrosis, microglial activation and infiltration, and astrocyte hypertrophy (Volpe, 1995).
Several nonexclusive mechanisms are thought to lead to WMD in the preterm infant. In some cases of WMD, especially cystic PVL, HI is thought to play a part; the concept of a centripetal and centrifugal arterial supply of preterm WM and a resultant “boundary” zone has added weight to this (DeReuck et al., 1972). This boundary zone theory, however, has been refuted by recent, sophisticated neuropathologic studies (Kuban and Gilles, 1985) showing that these centrifugal arteries were veins. However, despite the nonexistence of centrifugal arteries, the periventricular WM is vulnerable to hypoperfusion and ischemia, as it is at the end of a single arterial supply. Maternal or fetal infection and proinflammatory cytokines also are implicated in the pathogenesis of WMD (Dammann and Leviton, 1997). Markers of intraamniotic infection such as interleukin-6 have been linked to WMD and cerebral palsy (Yoon et al., 1997). Furthermore, at the beginning of the third trimester, the preterm WM appears to be vulnerable to injury. At this stage, the oligodendrocytes have started to produce myelin (Gilles et al., 1983); they have an increased energy demand and an increased susceptibility to oxidative stress because of their low glutathione and high iron content (Juurlink, 1997).
Studies using magnetic resonance spectroscopy (MRS) have characterized the biochemical events occurring in the days and weeks after birth in term infants who develop neonatal encephalopathy after perinatal HI. Term infants with a poor neurodevelopmental outcome after perinatal HI showed a well-described, early progression to “secondary energy failure” within the first week of life (Lorek et al., 1994; Penrice et al., 1997). At that stage, spectral changes included a decline in the phosphocreatine (PCr)/inorganic phosphate (Pi) ratio (Azzopardi et al., 1989), an increased brain lactate/creatine plus phosphocreatine (Cr) (Hanrahan et al., 1999), and an alkaline intracellular pH (pHi) (Robertson et al., 1999a). The severity of these spectral abnormalities correlated with abnormal neurodevelopmental outcome at 1 year (Azzopardi et al., 1989; Hanrahan et al., 1999; Robertson et al., 1999a). In addition, increased myo-inositol/Cr in the first week of life was associated with magnetic resonance imaging (MRI) abnormalities, suggestive of severe injury and a poor neurodevelopmental outcome at 1 year (Robertson et al., 2000).
After these early changes, after 2 weeks of age, there were persisting abnormalities in infants with a poor neurodevelopmental outcome after term perinatal HI; a brain lactic alkalosis persisted for weeks and, in some cases, many months after the insult (Robertson et al., 1999b); and reduced levels of N-acetyl aspartate (NAA)/Cr were demonstrated from approximately 2 weeks of age (Robertson et al., 1999b). Changes in myo-inositol/Cr at this stage have not been documented.
The pathogenesis of preterm WMD is complex. Based on data from the full-term infant in the weeks after HI injury, we hypothesized that there would be a persisting increase in brain lactate/Cr, an alkaline pHi, and a decreased NAA/Cr for weeks after WM injury in preterm WMD. We additionally measured myo-inositol/Cr and choline-containing compounds (Cho)/Cr in the WM. We used localized proton (1H) and whole-brain phosphorus (31P) MRS to define the biochemical characteristics of preterm WMD diagnosed on MRI. The findings in the WM of infants with WMD were compared with infants with no WM abnormality on MRI.
MATERIALS AND METHODS
Permission for this prospective study was granted by the Hammersmith Hospital Research Ethics Committee (no. 93/4047), and parental consent was obtained.
Subjects
The study population consisted of 30 premature infants receiving intensive care either in the Hammersmith Hospital or Queen Charlotte and Chelsea Hospital Neonatal Unit from November 1998 until September 1999. The group had a gestational age 27.9 ± 3.1 weeks with birth weight 1122 ± 445 g. All infants underwent routine cranial ultrasound examination within a few hours of birth and at regular intervals thereafter. The infants were examined using MRI and multinuclear (1H and 31P) MRS at a postnatal age of 9.8 ± 4.1 weeks (corrected age of 40.3 ± 3.9 weeks). Serum electrolytes were sampled from each infant daily as part of their routine care while in the intensive care unit and were noted at the time of the study. Blood lactate levels were measured when these infants were in intensive care and were normal in all infants when they were clinically stable. Because the infants were clinically stable with good perfusion and blood pressure at the time of the scan, it was assumed that the blood lactate was normal. Clinical data are summarized in Table 1.
Summary of demographic details, delivery, neonatal course, and MRI abnormalities
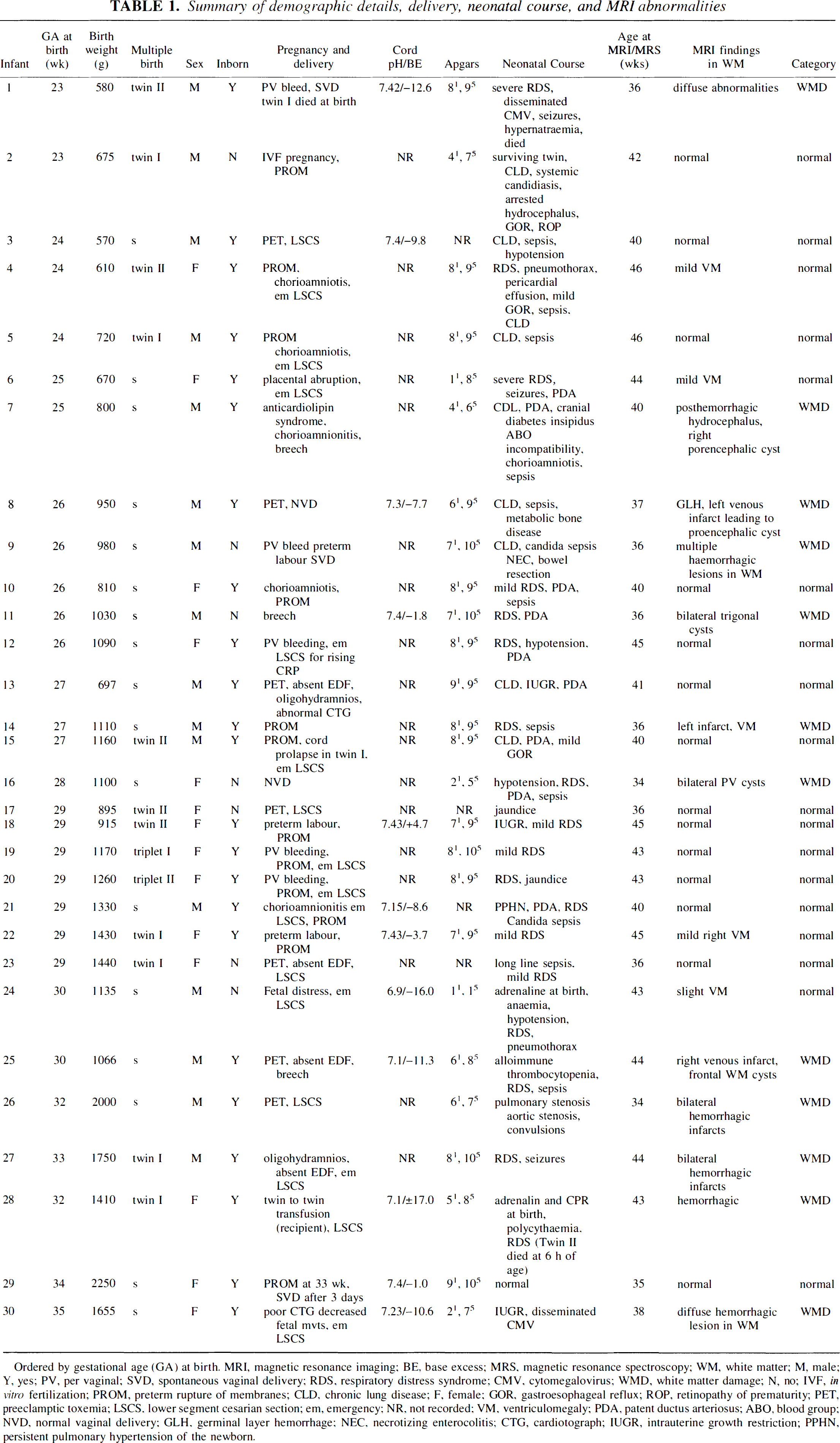
Ordered by gestational age (GA) at birth. MRI, magnetic resonance imaging; BE, base excess; MRS, magnetic resonance spectroscopy; WM, white matter; M, male; Y, yes; PV, per vaginal; SVD, spontaneous vaginal delivery; RDS, respiratory distress syndrome; CMV, cytomegalovirus; WMD, white matter damage; N, no; IVF, in vitro fertilization; PROM, preterm rupture of membranes; CLD, chronic lung disease; F, female; GOR, gastroesophageal reflux; ROP, retinopathy of prematurity; PET, preeclamptic toxemia; LSCS, lower segment cesarian section; em, emergency; NR, not recorded; VM, ventriculomegaly; PDA, patent ductus arteriosus; ABO, blood group; NVD, normal vaginal delivery; GLH, germinal layer hemorrhage; NEC, necrotizing enterocolitis; CTG, cardiotograph; IUGR, intrauterine growth restriction; PPHN, persistent pulmonary hypertension of the newborn.
The MRI and MRS examinations were performed in the same study after sedation with oral or rectal chloral hydrate (50 to 75 mg/kg). Infants weighing less than 1500 g were kept warm by swaddling in prewarmed blankets; additional bubble wrap, hats, and warm gel bags were used as needed. For immobilization and noise dampening, the infant's head was partly surrounded by a bag filled with polystyrene balls from which the air was evacuated. Neonatal intensive care, including mechanical ventilation, intravenous fluids and inotrope administration, was continued as appropriate during the scans, and all infants were accompanied by at least one experienced pediatrician at all times. Heart rate, oxygen saturation, and axillary temperature were monitored throughout the procedure using magnetic resonance (MR)-compatible equipment. Mechanical ventilation or continuous positive airway pressure were provided by a MR-compatible neonatal ventilator (babyPAC Neonatal Ventilator; pneuPAC Limited, Luton, Beds, U.K.).
Magnetic resonance imaging
Cerebral MR data were obtained using a Marconi Medical prototype 1.5-T MRS system (Marconi Medical Systems, Cleveland, OH, U.S.A.) and a double-tuned pediatric bird cage coil. The study protocol consisted of multislice T1-weighted conventional spin-echo (500/15) and dual proton-density, T2-weighted fast spin-echo (repetition time [TR] 4200, echo time [TE] 15/210) transverse images and sagittal T1-weighted conventional spin-echo images. Infants were divided into two groups by analysis of the MR images by a researcher blinded to the MRS results. Infants were categorized as having WMD if they showed cystic regions typical of classic PVL or hemorrhagic lesions typical of HPI. This group of infants was compared with subjects who did not show these WM abnormalities.
Magnetic resonance spectroscopy
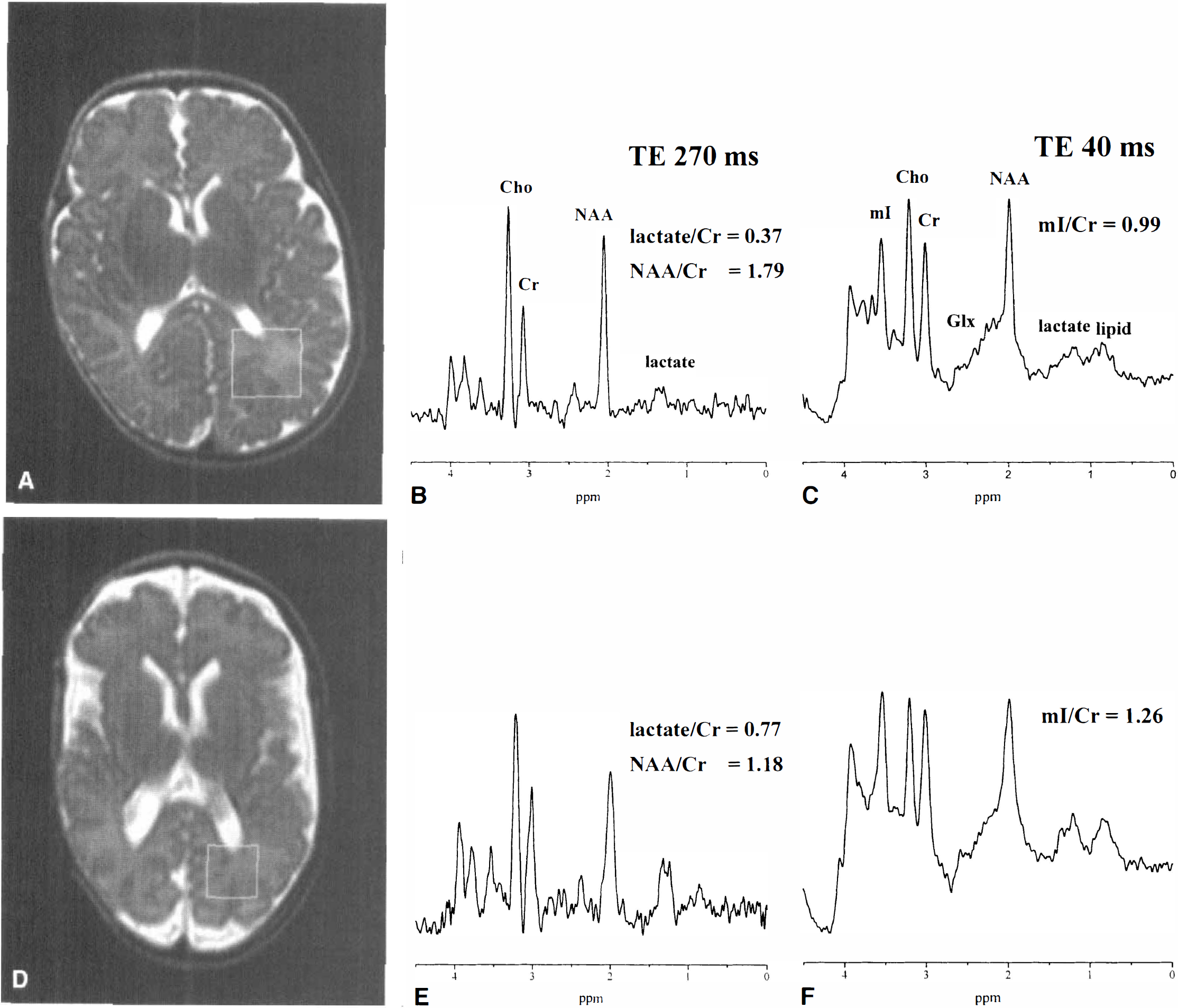
A voxel (2 cm × 2 cm × 2 cm) within the posterior periventricular WM was defined, and 1H MR spectra localized to this voxel were obtained using the point resolved spectroscopy (PRESS) localization sequence (Bottomley et al., 1987) with computer-optimized slice-selective radiofrequency pulses, using TR 2 seconds and 128 data collections, with TE values of 270, 40, and 135 milliseconds. Water suppression was obtained with three initial chemical shift selective saturation pulses. The peak area ratio of lactate/Cr was measured from the spectrum acquired with TE 270 milliseconds, and myo-inositol/Cr was measured from the spectrum acquired using TE 40 milliseconds. The voxel was placed in the posterior periventricular WM on the left side in all cases and not specifically from an area including any lesion. These spectral data therefore reflected both the focal and diffuse component of WMD associated with PVL or HPI. Care was taken to include only WM in the voxel, and although the small size of a preterm infant brain makes it impossible to be sure that cortex or CSF was totally excluded, the contribution of the cortex to the volume of brain tissue in the voxel was minimal in all cases, and the results apply primarily to WM.
The 1H MR data were filtered using a Gaussian filter (line broadening 1.5 Hz), Fourier transformed and manually phased. Peaks were assigned to Cr (3.93 ppm), myo-inositol (3.56 ppm), Cho (3.22 ppm), Cr (3.03 ppm), glutamine/glutamate (2.14 to 2.47 ppm), NAA (2.02 ppm), and lactate (1.33 ppm). Peak area ratios were calculated by fitting the peaks to a combined Lorentzian/Gaussian line shape. For the spectra acquired with TE 40 milliseconds, the maximum peak width of the fitted line shapes was limited to 0.1 ppm.
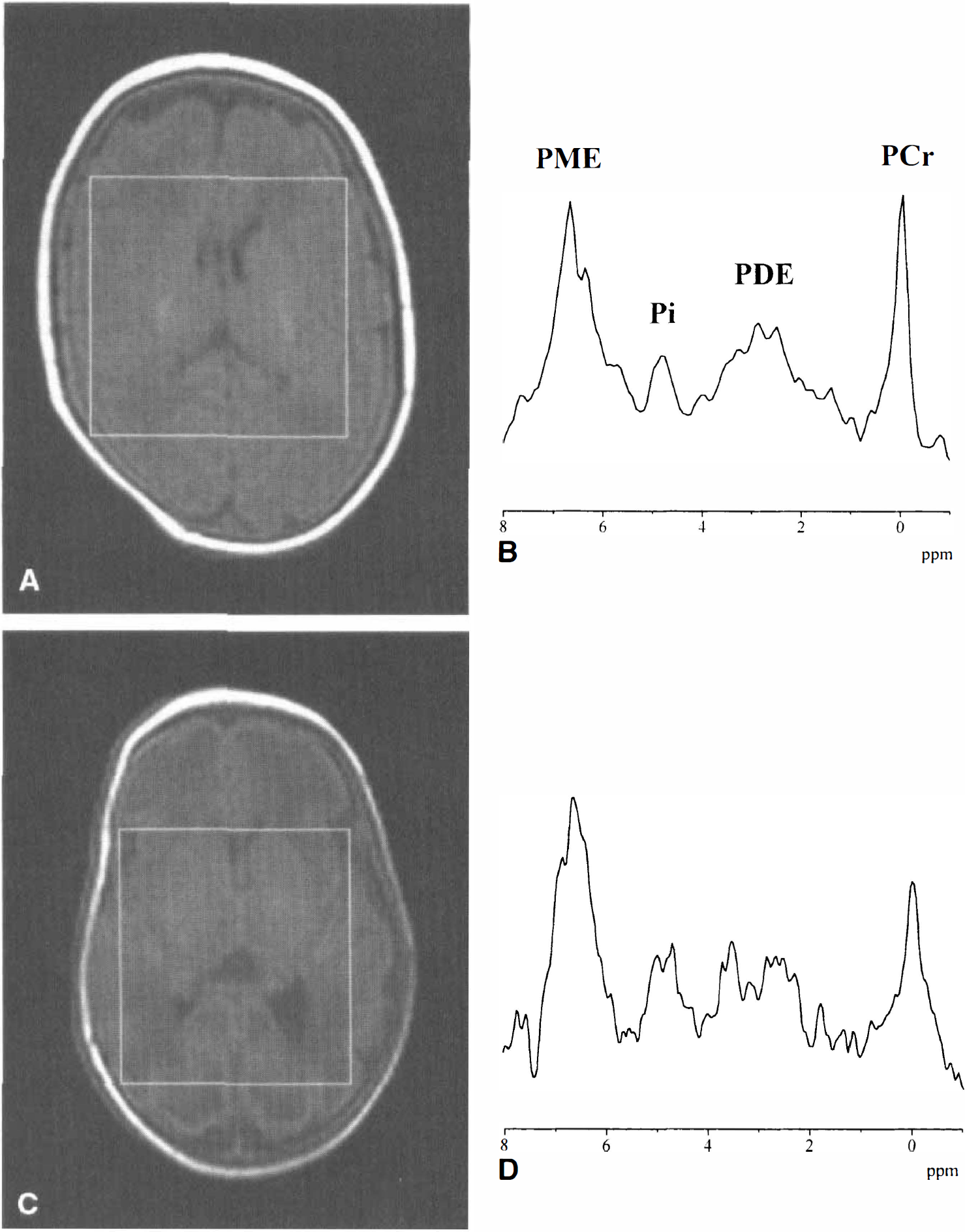
A larger voxel, at least 5 cm × 5 cm × 5 cm, was defined for the 31P MR spectrum. The magnetic field inhomogeneity in the volume of interest was minimized by shimming using the water signal from this defined region and the PRESS localization sequence. A 31P MR spectrum was acquired using the image-selected in vivo spectroscopy localization sequence (Ordidge et al., 1986) with TR 10 seconds, 128 data collections, and an acquisition time of 262 milliseconds or 524 milliseconds. The free induction decay was zero filled to double the number of points and filtered in the time domain using an exponential filter of 0 to 5 Hz followed by Fourier transformation. The small baseline roll resulting from the digital filters was uncorrected. The chemical shift of Pi relative to PCr was measured using the system software (version 10.2B), and from this the pHi was calculated (Petroff et al., 1985). In the cases where the Pi peak shape was complicated and split into a doublet structure, the Pi chemical shift was calculated as an average position of the two peaks.
Statistical considerations
The data distributions were inspected and transformed to normality where appropriate. Student's t-test was used to compare the clinical details of the normal and abnormal MRI groups. Analysis of variance, with age at the time of scan included as a covariate in the analysis, was used to determine if spectroscopy findings in the two patient groups were significantly different. Across the whole sample, Spearman's correlation was used to investigate the amount of common variation between lactate/Cr and myo-inositol/Cr, and between lactate/Cr and pHi.
RESULTS
Representative 1H MR spectra are illustrated in Fig. 1, and representative 31P MR spectra are illustrated in Fig. 2.
Eighteen infants had normal WM on MRI, and 12 infants had WMD. The mean gestational age at birth in the group with normal WM on MRI was 27.4 ± 2.7 weeks, and in the WMD group, 28.6 ± 3.7 weeks (P > 0.05). The mean birth weight in the group with normal WM on MRI was 1042 ± 414 g, and in the WMD group, 1242 ± 478 g (P > 0.05). Two infants were growth restricted (birth weight less than the third centile): one (patient 13) in the normal WM group, and one (patient 30) in the WMD group. The mean corrected age at the time of scan in the group with normal WM was 41.6 ± 3.4 weeks, and in the WMD group, 38.1 ± 3.8 weeks (P < 0.05). The mean postnatal age at the time of scan in the group with normal WM was 13.9 ± 5.4 weeks, and in the WMD group, 9.8 ± 4.1 weeks.
Although there was no significant difference between groups for gestational age, birth weight, and growth restriction, the corrected age and postnatal age at scan were slightly lower in the WMD group; this was taken into account when the MRS data in each group were compared by including scan age as a covariate in the analysis of variance.
For infants with WMD, myo-inositol/Cr and lactate/Cr were significantly correlated (P < 0.01). There was no significant correlation in the group with normal WM (Fig. 3). Spearman's rank correlation was used to assess the magnitude of the relationship between lactate/Cr and myo-inositol/Cr (r = 0.6; i.e., there was approximately a 40% shared variance).
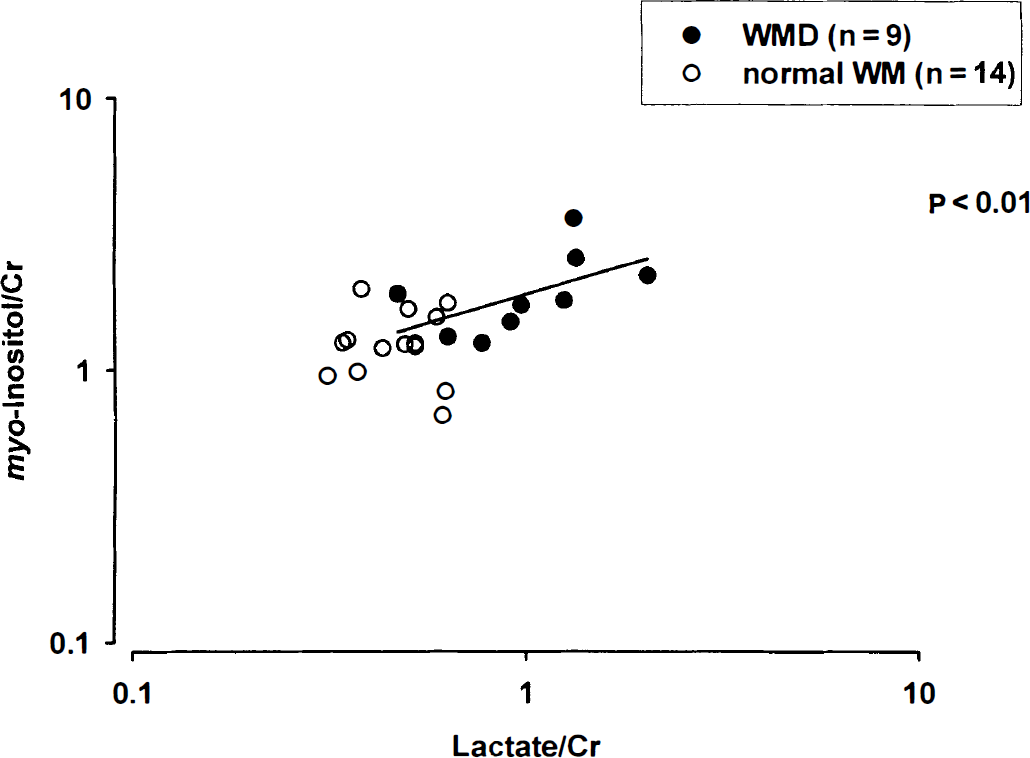
Lactate/Cr versus myo-inositol/Cr for infants with normal white matter (WM) on magnetic resonance imaging and white matter damage (WMD). The regression line for infants with WMD is shown. There is a correlation between lactate/Cr and myo-inositol/Cr in the group with WMD (P < 0.01), but not in the group with normal WM. Lactate/Cr and myo-inositol/Cr are plotted on logarithmic scales. Cr, creatine plus phosphocreatine.
The mean lactate/Cr in the infants without WMD on MRI was 0.51 ± 0.16, and in the group with WMD, 1.16 ± 0.46. The mean lactate/Cr was 110% higher (P < 0.001) in infants with WMD, even when taking into account any confounding bias resulting from age at the time of scan (Fig. 4). There was no significant interaction between the two groups (P = 0.65), implying a constant difference between the groups. Lactate/Cr was, therefore, higher at all ages in the group with WMD.
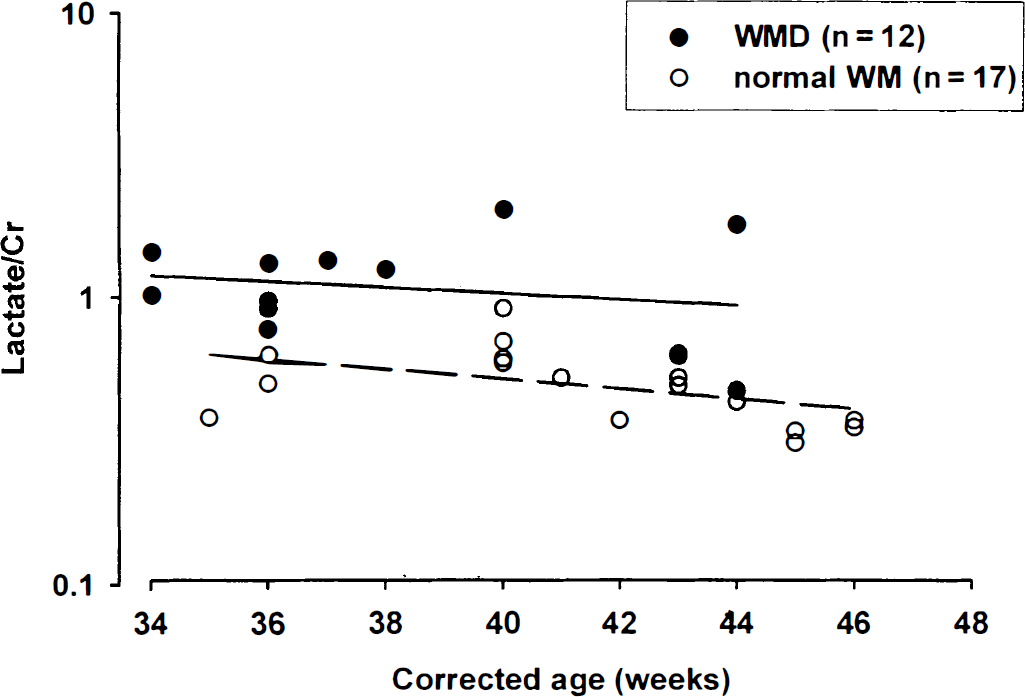
Relation between lactate/Cr and corrected age of scan in weeks for infants with normal white matter (WM) on magnetic resonance imaging and white matter damage (WMD). The regression lines for the group with normal WM (dashed line) and WMD (solid line) are shown. At all ages lactate/Cr was higher in the group with WMD. Lactate/Cr is plotted on a logarithmic scale. Cr, creatine plus phosphocreatine.
The mean NAA/Cr in the infants with normal WM on MRI was 1.59 ± 0.29, and in the group with WMD, 1.53 ± 0.3. There was no difference between groups for NAA/Cr (P > 0.05) (Fig. 5). The NAA/Cr increased with increasing corrected age in both groups.
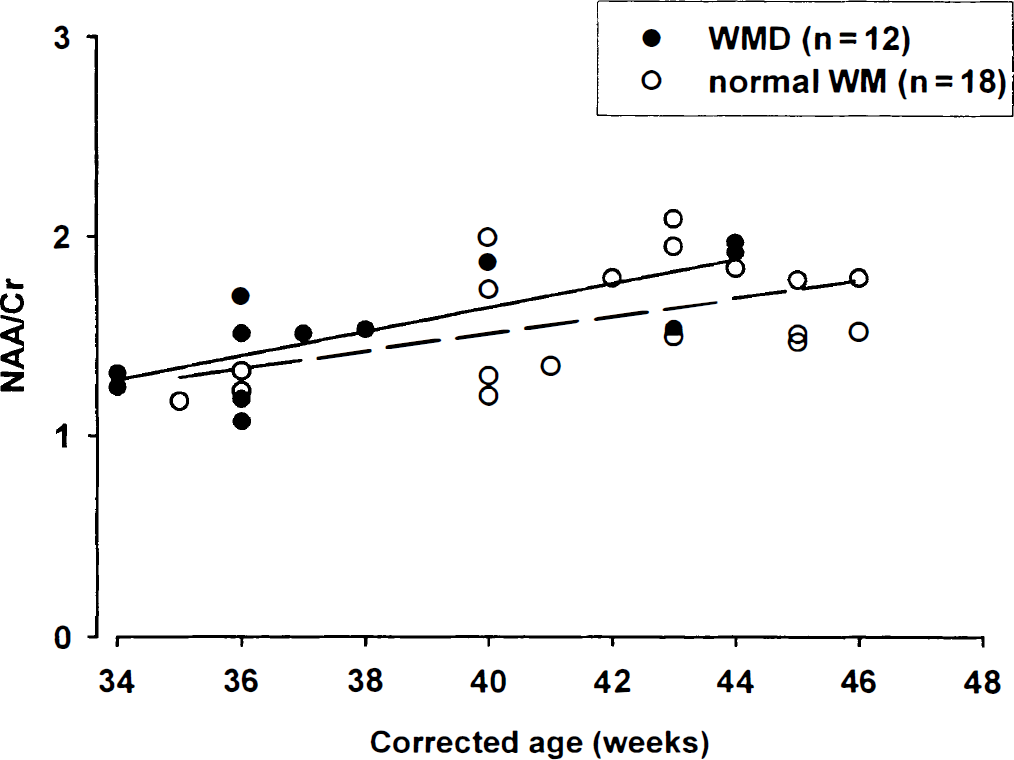
Relation between NAA/Cr and corrected age of scan in weeks for infants with normal white matter (WM) on magnetic resonance imaging and white matter damage (WMD). The regression lines for the group with normal WM (dashed line) and WMD (solid line) are shown. NAA/Cr increased with increasing gestational age (GA) in both groups; there was no difference between the normal WM and WMD groups. Cr, creatine plus phosphocreatine. NAA, N-acetyl aspartate.
The 31P MRS data were obtained in 18 of the 30 infants (60%), 4 in the group with WMD and 14 in the group without WMD. The mean pHi in the infants without WMD was 7.08 ± 0.05 (95% confidence interval [CI] 6.99 to 7.17) and in those with WMD, it was 7.08 ± 0.06 (95% CI 7.04 to 7.10). The mean difference in the pHi between the groups was 0.01 (95% CI 0.05 to 0.07). There was no correlation between lactate/Cr and pHi (P = 0.8) (Fig. 6). There was no relationship between an alkaline pHi and the presence of WMD (P = 0.2).
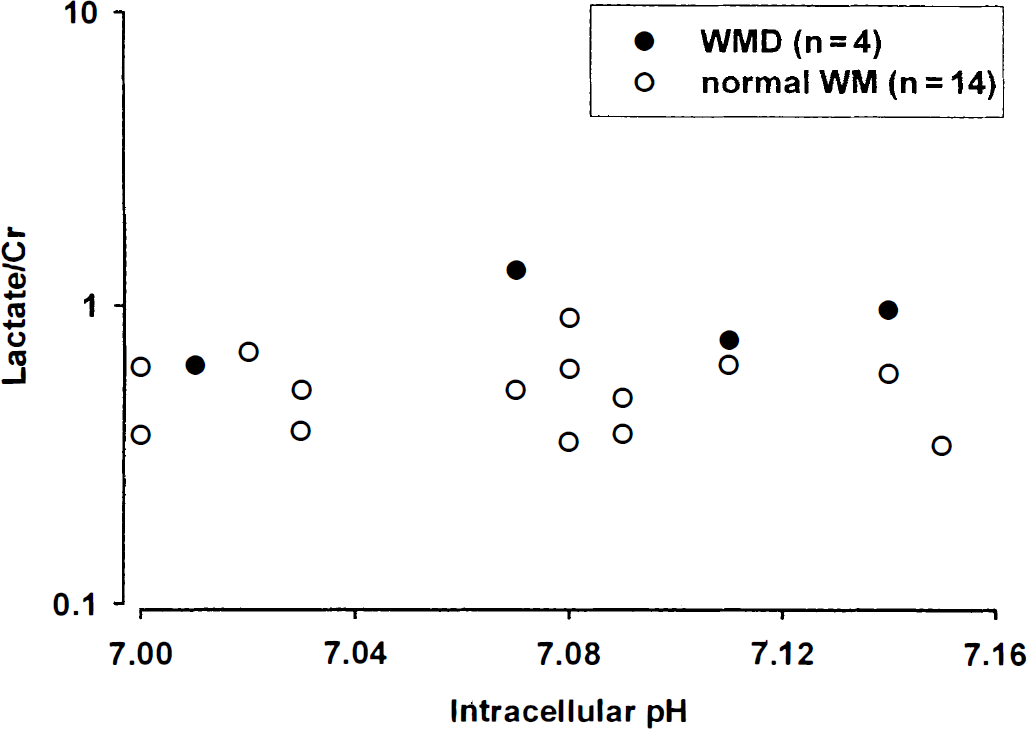
Absence of any relation between lactate/Cr and intracellular pH for all infants (P < 0.05). The normal white matter (WM) and white matter damage (WMD) groups are shown. Lactate/Cr is plotted on a logarithmic scale. Cr, creatine plus phosphocreatine.
In 10 of the infants the Pi resonance was a well-defined peak, and the position of the top of the Pi peak could be robustly determined (Fig. 2A). In eight infants the Pi peak could be resolved into a doublet structure (3 of 4 in the WMD group, and 5 of 14 in the group without WMD) (Fig. 2B). In the group with this pHi heterogeneity, the more alkaline component had a mean of 7.27 ± 0.03. The normal or slightly acidic component had a mean of 7.01 ± 0.04. The mean pHi was calculated from the average position of these two peaks, but there was no association between lactate/Cr and the mean pHi. There also was no association between the pHi of the two components considered separately and lactate/Cr.
The mean myo-inositol/Cr in the infants without WMD on MRI was 1.26 ± 0.35, and in the group with WMD, 2.0 ± 0.7. The mean myo-inositol/Cr was 60% higher (P < 0.05) in infants with WMD, even when taking into account any confounding resulting from age at the time of scan (Fig. 7). There was no significant interaction between the two groups (P = 0.5), implying a constant difference between groups. The myo-inositol/Cr was, therefore, higher at all ages in the group with WMD. The myo-inositol/Cr decreased with increasing postnatal age in the normal WM group (Fig. 8). There was no relationship with postnatal age in the infants with WMD.
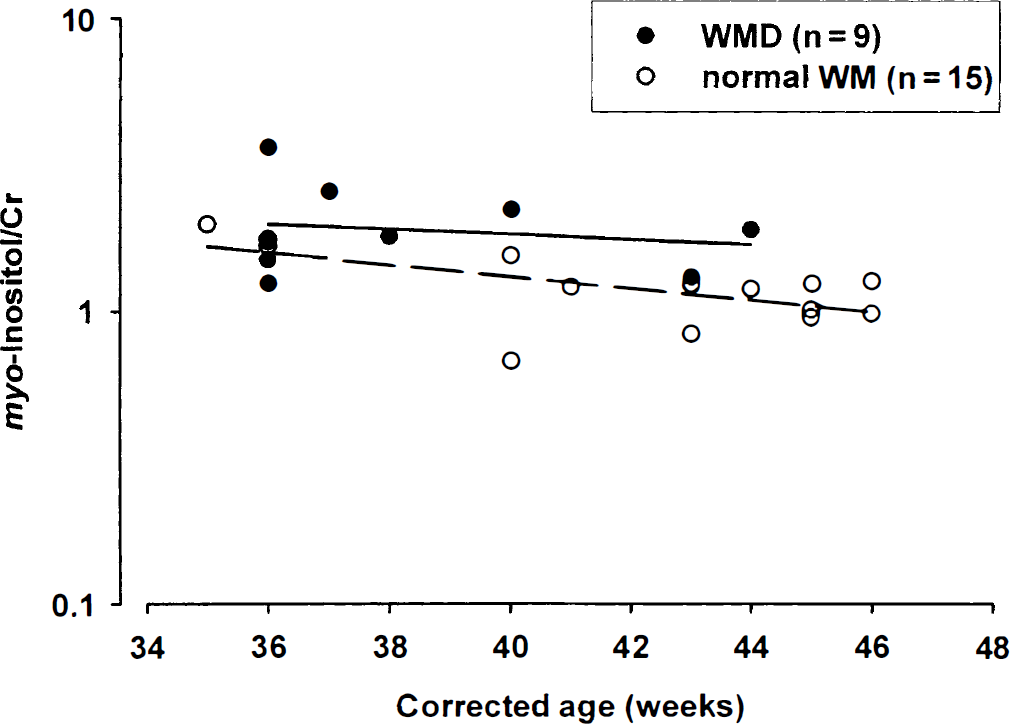
Relation between myo-inositol/Cr and corrected age of scan in weeks for infants with normal white matter (WM) on magnetic resonance imaging and white matter damage (WMD). The regression lines for the group with normal WM (dashed line) and WMD (solid line) are shown. At all ages myo-inositol/Cr was higher in the group with WMD. myo-Inositol/Cr is plotted on a logarithmic scale. Cr, creatine plus phosphocreatine.
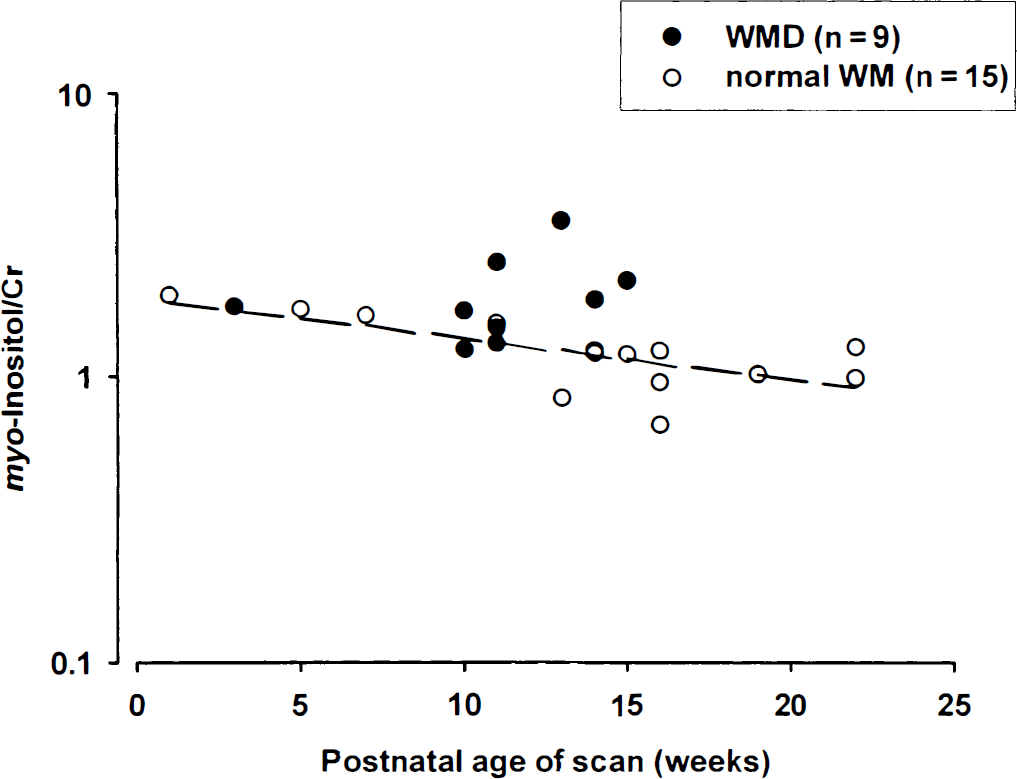
Relation between myo-inositol/Cr and postnatal age of scan in weeks for infants with normal white matter (WM) on magnetic resonance imaging and white matter damage (WMD). myo-Inositol/Cr decreased progressively with time from birth in the normal WM; this relation was absent in the WMD group. The regression line for the normal WM group (dashed line) is shown. myo-Inositol/Cr is plotted on a logarithmic scale. Cr, creatine plus phosphocreatine.
All infants except one (patient 1) had serum sodium concentrations within the normal range at the time of the study and, therefore, in these cases, the myo-inositol/Cr ratio was not affected by changes in extracellular osmolarity. Patient 1 had a brain myo-inositol/Cr of 3.63; this was 2 to 3 days after the plasma sodium concentration had been 179 mmol/L. Data subanalysis was performed excluding this patient; however, this still revealed a significantly increased myo-inositol/Cr in the group with WMD.
The mean Cho/Cr in the infants without WMD on MRI was 2.05 ± 0.41, and in the group with WMD, it was 2.41 ± 0.83. There was no significant difference between groups for mean Cho/Cr (P = 0.12) (Fig. 9).
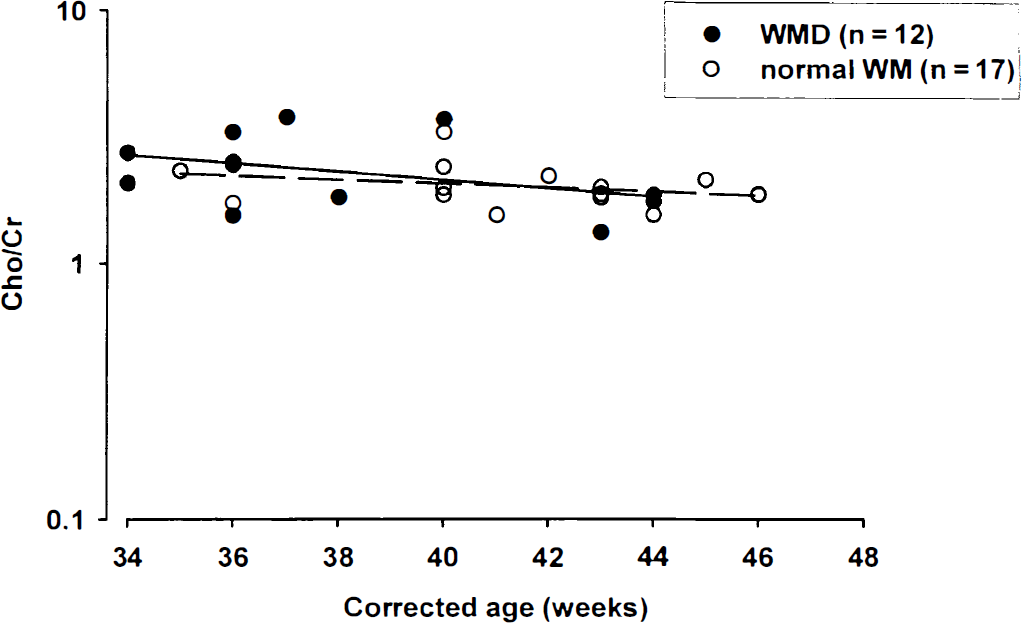
Relation between Cho/Cr and corrected age of scan in weeks for infants with normal white matter (WM) on magnetic resonance imaging and white matter damage (WMD). The regression lines for the group with normal WM (dashed line) and WMD (solid line) are shown. There was no difference between the normal and WMD group. Cho/Cr is plotted on a logarithmic scale. Cr, creatine plus phosphocreatine; Cho, choline containing compounds.
DISCUSSION
In this study, the biochemical characteristics of WMD in the preterm infant in the few weeks after the initial injury included an increased cerebral lactate/Cr without clear evidence of an associated alkaline pHi, a normal NAA/Cr, and an increased cerebral myo-inositol/Cr. These findings were independent of the infant's age at scan. Apart from the increased lactate/Cr, these findings are different from those seen in a term infant studied more than 2 weeks after perinatal HI.
The incidence of PVL in this study was 10%, and of HPI, 30%; this is not representative of the general preterm population. Because of the nature of the patient group, it was not possible to complete the study protocol in all cases. Because the 31P MRS data collection was last in the protocol, these data were the most difficult to obtain, especially in the sick infants with WMD; data were obtained in only 4 of 12 infants for pHi in the WMD group. Observations regarding the pHi in the WMD group therefore must be viewed with caution.
We used Cr as the metabolite of reference for lactate, myo-inositol, NAA, and Cho because Cr concentrations have been reported to be relatively stable after HI (Cady et al., 1997). There may be problems with this in preterm infants because Cr concentrations increase around term (Kreis et al., 1993). To remove any confounding bias by age, we included scan age as a covariate in the analysis of variance.
Lactate
Unlike the adult brain, lactate normally is detectable in the preterm brain using in vivo MRS and increases with decreasing gestational age (Leth et al., 1995). This may be a reflection of the immature stage of enzyme development in the preterm brain (Booth et al., 1980) and is explained by the increasing dependence on glycolysis compared with mitochondrial respiration. The energy requirement or oxygen uptake is low in the preterm brain at 26 to 32 weeks gestation, when it is around one sixth that of the adult brain and one half that of the term brain (Altman et al., 1993); glycolysis appears to be the principle mechanism through which energy requirements are met.
Blood lactate is used as a fuel in the normal preweaning neonatal brain (Vicario C et al., 1991). Transport of lactate from blood to brain is facilitated by monocarboxylate transporters present on the blood–brain barrier (Pellerin et al., 1998). These transporters are detected only in the early postnatal stages of development, their disappearance coinciding with the decrease in blood lactate utilization after weaning (Pellerin et al., 1998). Within the brain, there appears to be a “lactate shuttle” from astrocytes to neurons. Glucose taken up by astrocytes is metabolized to lactate; substances such as glutamate, released by neurons, stimulate this lactate production in an activity-dependent manner (Pellerin et al., 1998).
The lactate/Cr ratio in the posterior periventricular WM was 110% higher than that in the WM of infants with a normal MRI. There are several possible causes, as follows:
Defective energy metabolism.
This elevation of lactate in WMD may result from a defect in energy metabolism at one of several sites in glycolysis, the Krebs cycle, or mitochondria. Damage to the electron transport chain would lead to a reduced ATP flux. This would in turn stimulate glycolysis, promoting a positive feedback system perpetuating lactate production. In term infants after perinatal HI, we have previously hypothesized that the persisting increase in lactate and associated alkalotic pHi resulted from a change in the cellular redox state (Robertson et al., 1999b). Because the same association between lactate/Cr and pHi does not exist in the preterm brain, this alone is unlikely to explain the increase in lactate/Cr.
Persistent WM ischemia.
After ischemia, a decrease in CBF to the WM has been seen to persist despite recovery to other brain areas. This may result from an immaturity in the vasomotor response of vessels supplying the WM in the preterm brain (Cavazzutti and Duffy, 1982). In experimental studies, decreases in blood flow to WM have been demonstrated after cytokine exposure (Kikuchi et al., 1995).
Glutamate receptor–mediated phenomenon.
At physiologic concentrations, glutamate stimulates glycolysis and, hence, lactate production in astrocytes (Pellerin et al., 1998). There is evidence of axonal rupture in WMD, and substantial amounts of glutamate are extravasated into the extracellular space of the WM. This glutamate then may stimulate lactate production in astrocytes.
N-acetyl aspartate
In this study, there was no difference in the NAA/Cr in the group with WMD compared with the normal MRI group. This result is surprising in light of the loss of oligodendrocytes and the later delayed myelination evident on MRI in these infants with WMD (de Vries et al., 1987). Partial volume effects may contribute to our finding, because although the position of the 8-cm3 voxel was carefully chosen to include predominantly WM, in some cases, a small amount of cortex was included.
The preservation in NAA/Cr in our study may result from the plasticity of the cells to repair. The developing brain shows more plasticity after an insult than a mature brain (Murakami et al., 1992), and plasticity of certain pathways has been described in ex-preterm infants with PVL who later developed spastic cerebral palsy (Maegaki et al., 1999). It is possible that this effect is more exaggerated the the earlier the insult.
Intracellular pH
Unlike our findings in the term brain after HI injury, there was no persisting alkalosis and no relationship between pHi and lactate/Cr in these preterm infants with normal WM or WMD on MRI. Because of the small numbers of infants with WMD who had pHi measurements and the possibility of a type II error, we cannot confidently define any relationship of pHi with WMD. However, the relationship between pHi and lactate/Cr appears to differ in preterm infants compared with term infants after perinatal HI; for a given lactate/Cr in a preterm infant, the pHi was never as alkaline as it was in term infants after perinatal HI (Robertson et al., 1999a). A doublet structure on the Pi peak was visualized in 7 of the 18 infants who were examined using 31P MRS in our study. The Pi doublet structure in each case reflected a separate alkaline and normal/acidic pHi. The second more alkaline phosphate pool may result from the extracellular compartment (Portman and Ning, 1990), neuronal/glial compartmentation (Sungawa et al., 1994), or a separate intracellular compartment such as the mitochondrion (Garlick et al., 1983).
myo-Inositol
The myo-inositol peak at 3.56 ppm also may contain contributions from glycine and glucose, although more than 70% generally is from myo-inositol (Ross, 1991). The relative signal reduction seen at TE 270 milliseconds is more typical of the coupling behavior of myo-inositol than a singlet peak showing T2 relaxation typical of glycine.
The myo-inositol concentrations decrease rapidly after birth in the term infant and reach adult levels within the first few months of life. This occurred in our preterm infants with normal WM on MRI but not in the infants with WMD. Intracellular myo-inositol concentrations are higher than plasma or interstitial fluids; the Na+/ myo-inositol cotransporter (SMIT) is thought to be responsible for the maintenance of these gradients.
Function of myo-inositol in the CNS.
myo-Inositol is involved in two main cellular processes in the CNS: (1) intracellular signal transduction (Berridge, 1993), and (2) cell volume regulation during persistent osmotic stress (Thurston et al., 1989). myo-Inositol visualized on short TE 1H MRS reflects free myo-inositol acting as an intracellular osmolyte (Bachelard, 1997).
Clinical studies show that brain cells respond to extracellular hypertonicity by accumulating high concentrations of myo-inositol intracellularly to maintain cell volume and function and minimize the loss of intracellular brain water (Lee et al., 1994). In vitro studies demonstrate that the SMIT mRNA and the transcription rate of the SMIT gene are increased when brain glial cells are cultured in hypertonic medium (Paredes et al., 1992). In our study, part of the elevation of the myo-inositol/Cr in the infant with a plasma sodium of 179 mmol/L may have resulted from this function; however, exclusion of this infant did not change the result.
The myo-inositol/Cr ratio was increased by 60% in the WM of infants with WMD compared with the infants with normal MRI. Possible mechanisms are as follows:
Up-regulation of SMIT.
Induction of SMIT has been reported after brain ischemia and cryogenic injury in neuronal cells (Yamashita et al., 1996, 1997); this may be in response to the influx of cations occurring during membrane depolarization at the start of an insult. This has been demonstrated with neuronal cells exposed to veratidine (causing cellular uptake of Na+); these cells developed an up-regulation of SMIT mRNA levels (Yamashita et al., 1999). Veratidine used with a blockade of myo-inositol uptake demonstrated increased cytotoxicity in neuronal cultures, suggesting a possible protective role of myo-inositol.
Gliosis.
At a cellular level in WMD, microglia become activated to repair the injured brain. In vitro work suggests that myo-inositol is a marker for glia (Brand et al., 1993); clinical studies in adult WM disease attribute raised brain myo-inositol in WM to increased glial activity (Chang et al., 1999). These studies describe a concomitant increase in Cho in gliosis; in our infants with WMD, however, we did not find any significant difference in Cho between the two groups. Recent studies demonstrate that neurons as well as glia are able to maintain high intracellular concentrations of myo-inositol (Novak et al., 1999), suggesting that reactive gliosis may not be enough to explain the persisting increase in myo-inositol seen in preterm WMD.
In conclusion, we demonstrate a different metabolic profile of preterm WMD compared with that seen more than 2 weeks after term HI in that there was an increased lactate/Cr and myo-inositol/Cr in the WM of infants with WMD, but not an associated alkaline pHi or reduced NAA/Cr. Both the type of injury and response of the extremely preterm brain to an insult may differ from that in the term brain; these data add weight to the concept that preterm WMD has a complex pathogenesis.
