Abstract
Studies of vascular biology during the past decade have identified an expanding list of agonists and antagonists that regulate local hemostasis, inflammation, and reactivity in blood vessels. Interactions at the blood-endothelial interface are intricate and complex and have been postulated to play a role in the initiation of stroke and the progression of brain injury during early hours of ischemia, particularly in conjunction with reperfusion injury (Hallenbeck, 1996). In the current study of normal and activated vessels in rat brain, immunoreactive tumor necrosis factor-alpha (TNF-α), heme oxygenase-1 (HO-1), and manganese superoxide dismutase (MnSOD) exhibit concentric perivascular rings involving vessel wall and surrounding parenchyma that appear to coincide with one another in serial sections. The ring patterns suggest periodic radial expansion of these molecules released through a process of cyclic activation and inactivation of brain vessel segments. In this process, the rings appear randomly scattered instead of affecting all vessels within a high power field (HPF) synchronously. The average number of vessels per HPF (mean ± SD) with perivascular cuffs of immunoreactive MnSOD increased from 51 ± 28 in Wistar, 72 ± 46 in Wistar-Kyoto, and 84 ± 30 in Sprague Dawley rats (no spontaneous strokes) to 184 ± 72 in spontaneously hypertensive stroke-prone rats (spontaneous strokes). Perivascular immunoreactive cuffs are also increased in spontaneously hypertensive rats by induction of cytokine expression by lipopolysaccharide (64 ± 15 vs. 131 ± 32 /HPF). The patterns of TNF-α, HO-1, and MnSOD in naïve animals are interpreted to indicate that focal hemostatic balance normally fluctuates in brain vessels and influences surrounding parenchymal cells. Perivascular immunoreactive cuffs representing this process are more frequent in animals with lipopolysaccharide-induced endothelial activation or genetic stroke proneness.
Keywords
A panoply of powerful enzyme and cell signaling systems that are usually subject to rigorous control operate at the blood-endothelial interface and can influence the initiation and course of brain ischemia (Hallenbeck, 1996). With respect to hemostasis, potent antithrombotic factors that are induced in endothelial cells concurrently with activation of the prothrombotic factors provide the mutual regulation necessary to maintain fluid blood within the circulation (Stern et al., 1991; Rosenberg and Aird, 1999). Systemic fluctuations in thrombogenic potential have been observed as an overall effect of this form of feedback control. For example, tissue plasminogen activator and plasminogen activator inhibitor-1 undergo diurnal fluctuation such that fibrinolysis is at a nadir in the early morning hours and at a peak in the evening (Angleton et al., 1989). Platelet aggregability is also greatest in the early morning hours (Jovicic et al., 1991) and, correspondingly, strokes tend to occur preferentially at this time (Marler et al., 1989; Labrecque and Soulban, 1991). That hemostatic balance can also fluctuate focally within vessel beds is a relatively recent postulate, owing its origins to the dramatic increase in understanding of endothelial cell biology that has occurred over the last 20 years (Pober and Cotran, 1990; Becker et al., 2000) and to the observation that systemic alterations in hemostatic mechanisms typically give rise to local thrombotic lesions in discrete segments of the vascular tree rather than causing a diffuse thrombotic diathesis with disseminated intravascular coagulation. For example, hypercoagulable states that arise from a general imbalance between procoagulant and anticoagulant forces-such as the antiphospholipid antibody syndrome, Factor V Leiden, mutation of the heparin-binding site of antithrombin III, and prothrombin G20210A mutation-lead to focal rather than diffuse thrombosis by biasing local endothelial cell control of hemostasis. These data have been woven together to support a model for focal development of thrombotic lesions in which signaling pathways specific to a vascular bed are determinate (Rosenberg and Aird, 1999).
Local endothelium integrates various extracellular signals and cellular responses in different regions of the vascular tree according to this model. Some of these diverse environmental cues would include cytokines, chemokines, nitric oxide, endothelin, growth factors, hemodynamic forces, cell-to-cell signaling, and signaling associated with integrins and other adhesion molecules. The ability of endothelium to transduce a given signal is regulated in space and time. The net effect of these variables is to cause differential expression of endothelial cell-derived procoagulant and anticoagulant activities focally throughout the vascular tree. A related paradigm in which there is interplay between focal activation of endothelium and systemic activation of the coagulation or complement systems is the localized Shwartzman reaction (Shwartzman, 1928). Preclinical studies have demonstrated that a localized Shwartzman reaction can produce strokes preferentially in rats with risk factors for stroke (Hallenbeck et al., 1988).
In the current article, the authors studied the distribution patterns of immunoreactivity of several indicators of inflammatory and oxidative stress as well as that of a key antioxidant enzyme in brain sections. The authors found that scattered, cross-sectioned vessels in the brains of normal rats exhibit coincident perivascular halos of immunoreactive tumor necrosis factor-alpha (TNF-α), heme oxygenase-1 (HO-1), and manganese superoxide dismutase (MnSOD). The observed patterns permit the inference that these halos or cuffs undergo periodic radial expansion and fading because of the occurrence of cyclic activation and inactivation of vessel segments in the brains of normal animals. Induction of cytokine expression by lipopolysaccharide (LPS) increases the number of vessels with perivascular cuffs of immunoreactivity. The purpose of the current work was to characterize the perivascular expression of TNF-α (proinflammatory, prothrombotic), HO-1 (oxidative stress marker), and MnSOD (antioxidant) in brain vessels of several rat strains and to evaluate the role of endothelial activation by LPS in the modulation of this protein expression.
MATERIALS AND METHODS
Animals
Male rats (12 to 13 weeks) of the following strains were studied: spontaneously hypertensive rat (SHR) (Taconic Farms, Germantown, NY, U.S.A.), Sprague-Dawley, Wistar, and Wistar-Kyoto (WKY) rat (Charles River Labs, Wilmington, MA, U.S.A.).
Male and female (18 weeks) spontaneously hypertensive stroke-prone rats (SHR-SP) (Kyoto University, Japan) were also studied. Rats were maintained in animal quarters with a standard 12 hour light/dark cycle with humidity and temperature controlled at normal levels. Water and food were allowed ad libitum. The NIH Animal Care and Use Committee approved all procedures in animals.
Histology
The following untreated rats were used in the histologic study: SHR (n = 4), Sprague-Dawley (n = 3), Wistar (n = 3), WKY (n = 3), and SHR-SP (n = 4). Additional SHRs received LPS (n = 10) or saline (n = 5) intravenously (IV) and were killed at different time points. These animals were anesthetized with 2% halothane for induction and 0.8% halothane for maintenance in 30% oxygen and 70% nitrous oxide gas mixture through a facemask. Bacterial LPS (Escherichia coli 0111: B4 phenol extract; Sigma, St. Louis, MO, U.S.A.) was suspended in 0.5 mL of sterile 0.9% saline and administered through the catheter to the femoral vein in a dose of 0.9 mg/kg body weight. Control animals received the same amount of saline through the same route. After the procedure, the animals were returned to their cages with free access to food and water. Animals were divided into five groups according to survival time. There were 2 LPS-injected animals and 1 saline-injected animal in each group and survival times consisted of 8, 24, 48, 72, or 168 hours. After transcardial perfusion with 4% buffered paraformaldehyde (pH 7.43) under deep pentobarbital anesthesia (65 mg/kg intraperitoneal (IP) injection), the brains were left in situ for 1 hour. Brains then were removed and postfixed for another 4 hours in 4% paraformaldehyde and embedded in paraffin. Seven-micrometer-thick sections were cut on a rotary microtome, mounted on Probe On Plus Microscope Slides (Fisher Scientific, Pittsburgh, PA, U.S.A.), and stained with hematoxylin and eosin for morphologic evaluation. Adjacent sections were used for immunohistochemistry.
Immunohistochemistry
Enzyme immunohistochemistry was performed by the avidin-biotin-complex (ABC) method (Vector Laboratories, Burlingame, CA, U.S.A.). Briefly, sections were deparaffinized, rehydrated, and permeabilized with 0.3% Triton in phosphate-buffered saline (PBS) for 1 hour and thoroughly rinsed afterwards. Endogenous peroxidase was inactivated by immersing the slides in a solution of 3% hydrogen peroxide in 10% methanol/PBS. Then slides were washed in PBS 4 × 5 minutes and incubated for 2 hours in 5% normal goat or 5% normal donkey serum in PBS to block nonspecific staining. After blocking, sections were incubated with one of the following primary antibodies diluted in 2% goat serum/PBS overnight at 4°C: rabbit anti-MnSOD in a concentration of 1:200 (a gift from Dr. Kato, Department of Biochemistry, Institute for Developmental Research, Aichi, Prefectual Colony, Kamiya. Kaugai. Aichi 480-03, Japan), mouse anti- 4-hydroxynonenal (HNE) lysine 1:100 (a gift from Dr. Witztum, Immunology Core Laboratory of the La Jolla SCOR Program, University of California, San Diego, CA, U.S.A.), rabbit anti-malondialdehyde (MDA)-modified rabbit serum albumin 1:100 (gift from Hoechst Marion Russel, Inc. Cincinnati, OH, U.S.A.), rabbit anti-HO-1, 1:100 (StressGen, Victoria, Canada), and goat anti-TNF-α diluted 1:40 in 2% rat serum/PBS (R&D Systems, Minneapolis, MN, U.S.A.) and incubated for 48 hours. After washing in PBS 4 × 10 minutes, sections were incubated with the appropriate biotinylated secondary antibody, goat anti-rabbit, goat anti-mouse, or donkey anti-goat (Jackson Immuno Research Labs, West Grove, PA, U.S.A.) in a concentration of 1:1000 for 1 hour at room temperature. Slides were washed as before and incubated for 1 hour in an avidin-biotin-peroxidase complex (ABC-Elite Kit, Vector) at room temperature. Antigen antibody binding was visualized with diaminobenzidine, (DAB Kit, Sigma).
After color development, sections were thoroughly rinsed in PBS again, dehydrated, cleared, and mounted in Permount. For assessment of nonspecific staining, primary antibodies were omitted or replaced by normal mouse, rabbit, or goat immunoglobulin G (IgG). Tumor necrosis factor-α staining was abolished by absorption of antibody with a 50-fold excess of recombinant rat TNF-α.
Quantification of perivascular manganese superoxide dismutase expression
Brain sections were analyzed with a Zeiss Axioplan microscope (Carl Zeiss, Jena, Germany). Images of four different regions (left and right hippocampus, left and right thalamus) were acquired at 50x magnification with the Metamorph Image Processing System (Universal Imaging, West Chester, PA, U.S.A.) and the number of vessels expressing immunoreactive MnSOD cuffs were counted per field of view (39,3216 μm2).
Immunofluorescence
Localization of MnSOD in astrocytes, neurons, perivascular macrophages, endothelial cells, and oligodendrocytes was shown by double-label immunofluorescence with antibodies against markers for these cells. Antibodies and concentrations used: mouse anti-glial fibrillary acidic protein (GFAP, Sigma) 1:400, mouse anti-neurofilament 200 (NF-200, Sigma) 1:400, mouse anti-neuronal nuclei (NeuN) 1:100 (Chemicon International, Temecula, CA, U.S.A.), mouse anti-rat monocyte and macrophage (clone: ED-1; BioSource, Camarillo, CA, U.S.A.), mouse anti-2', 3'-cyclic nucleotide 3'-phosphodiesterase (CNPase, Sigma) 1:500 (marker for oligodendrocytes), and sheep anti-rat von Willebrand Factor, 1:1000 (Cedarlane Labs., Ontario, Canada). Staining for the von Willebrand Factor was performed on frozen sections using fluorescein-conjugated donkey anti-sheep IgG (Jackson Immuno Research Labs) 1:200 as the secondary antibody. All other staining was performed on paraffin sections as follows: sections were deparaffinized, rehydrated, permeabilized in 0.3% triton/PBS for 1 hour, blocked with 5% horse serum/PBS for 1 hour and incubated with primary antibody overnight at 4°C, washed 3 × 10 min in PBS, and then incubated with fluorescein conjugated horse anti-mouse IgG (Vector) 1:100 for 1 hour at room temperature. After thorough washing and blocking for 30 minutes in 5% goat serum, this procedure was repeated with the anti-MnSOD antibody and Rhodamine Red-X-conjugated goat anti-rabbit IgG (Jackson Immuno Research) 1:100. After washing, the sections were coverslipped with Vector Shield Mounting Medium and examined under an epifluorescence microscope (Axioplan, Carl Zeiss) with the appropriate filters. Omission of primary antibodies or their replacement with normal mouse and rabbit IgG resulted in no staining of tissue.
Statistics
Comparison of the MnSOD perivascular immunoreactivity among the groups was by analysis of variance followed by Fisher's Protected Least Significant Difference.
RESULTS
Immunohistochemistry for manganese superoxide dismutase
Manganese superoxide dismutase expression was found in the brains of all animals in a striking pattern of clearly defined cuffs and rings around blood vessels of all sizes. The number of vessels/HPF with MnSOD perivascular cuff expression was variable among untreated rats of different strains. Wistar rats showed the lowest levels of perivascular MnSOD immunoreactivity/HPF (51 ± 28, mean ± SD) and SHR-SP showed the highest levels (184 ± 72) (Fig. 1). Sprague-Dawley (84 ± 30), WKY (72 ± 46), and untreated SHR (64 ± 15) showed values for perivascular MnSOD expression/HPF that did not differ significantly from Wistar. SHR-SP perivascular cuffs/HPF differed from Wistar, Sprague-Dawley, WKY, and SHR at P < 0.001, P < 0.002, P < 0.001, P < 0.001, respectively. Considerable within-group variability was noted among the WKY rats.
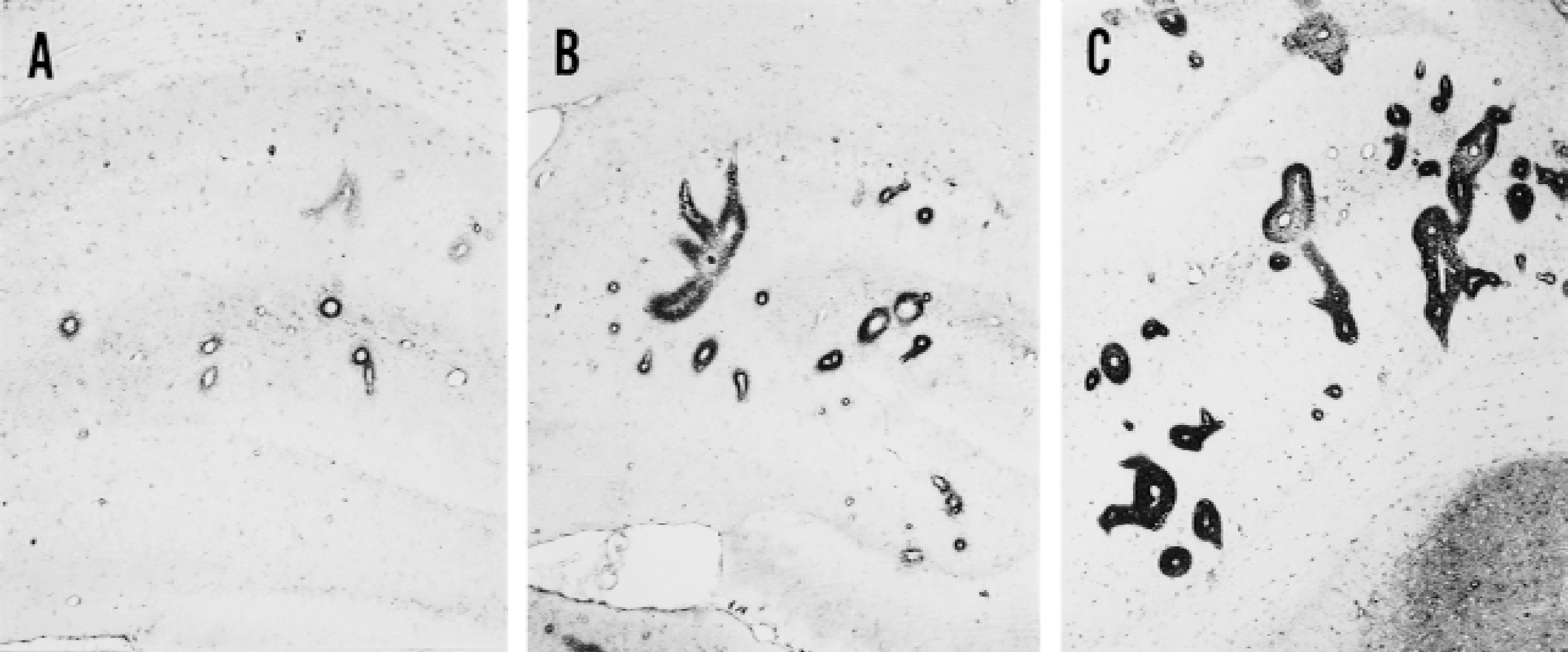
Immunostaining for manganese superoxide dismutase in sections of hippocampus (magnification x50) of
Spontaneously hypertensive rats that received LPS showed increased levels of MnSOD expression (Fig. 1) at all time points. Mean ± SD perivascular MnSOD immunoreactivity/HPF in all SHR exposed to LPS was 131 ± 32 compared with naïve SHR 64 ± 15 (P < 0.02). The stress of anesthesia and femoral artery catheterization was associated with some increase in perivascular MnSOD immunoreactivity in SHR that received saline injections at time points corresponding to LPS injections (107 ± 17), but this was not significantly different than the number of cuffs/HPF in naïve SHR (P < 0.13).
Manganese superoxide dismutase expression staining could be found throughout the brain, but was most prominently expressed around vessels of the hippocampus, striatum, and thalamus. The staining was granular, consistent with the mitochondrial location of MnSOD, and followed several different staining patterns (Fig. 2). Some vessels were surrounded by tightly packed, intensely staining cuffs that did not extend far beyond the vessel wall, whereas others were surrounded by thick cuffs of large diameter relative to the central vessel. The staining intensity of the latter cuffs varied from dense to faint. Adjacent to other vessel walls, the inner area of the cuff was completely unstained with only an outer ring staining for MnSOD. These larger cuffs, which appeared to have expanded radially from the central vessel, could extend to an outer diameter of 50 μm and within some of these expanded cuffs a nascent ring within a ring could be observed. In this pattern, the smaller inner ring hugged the vessel wall and stained intensely for MnSOD, whereas the larger outer ring stained weakly and surrounded tissue in which MnSOD immunoreactivity was faint or absent. Omission of the MnSOD primary antibody eliminated perivascular cuff staining. An inference is that this pattern indicates that MnSOD is induced in wave upon wave originating at the vessel wall and spreading radially from there into the surrounding parenchyma. The antioxidant is induced in cells with the morphology of endothelium, pericytes, and astrocytes, but is most prominent in neurons. Neuronal cell bodies and processes could be detected within the cuffs, with their dendrites often radiating from the cuffs into the surrounding unstained tissue.
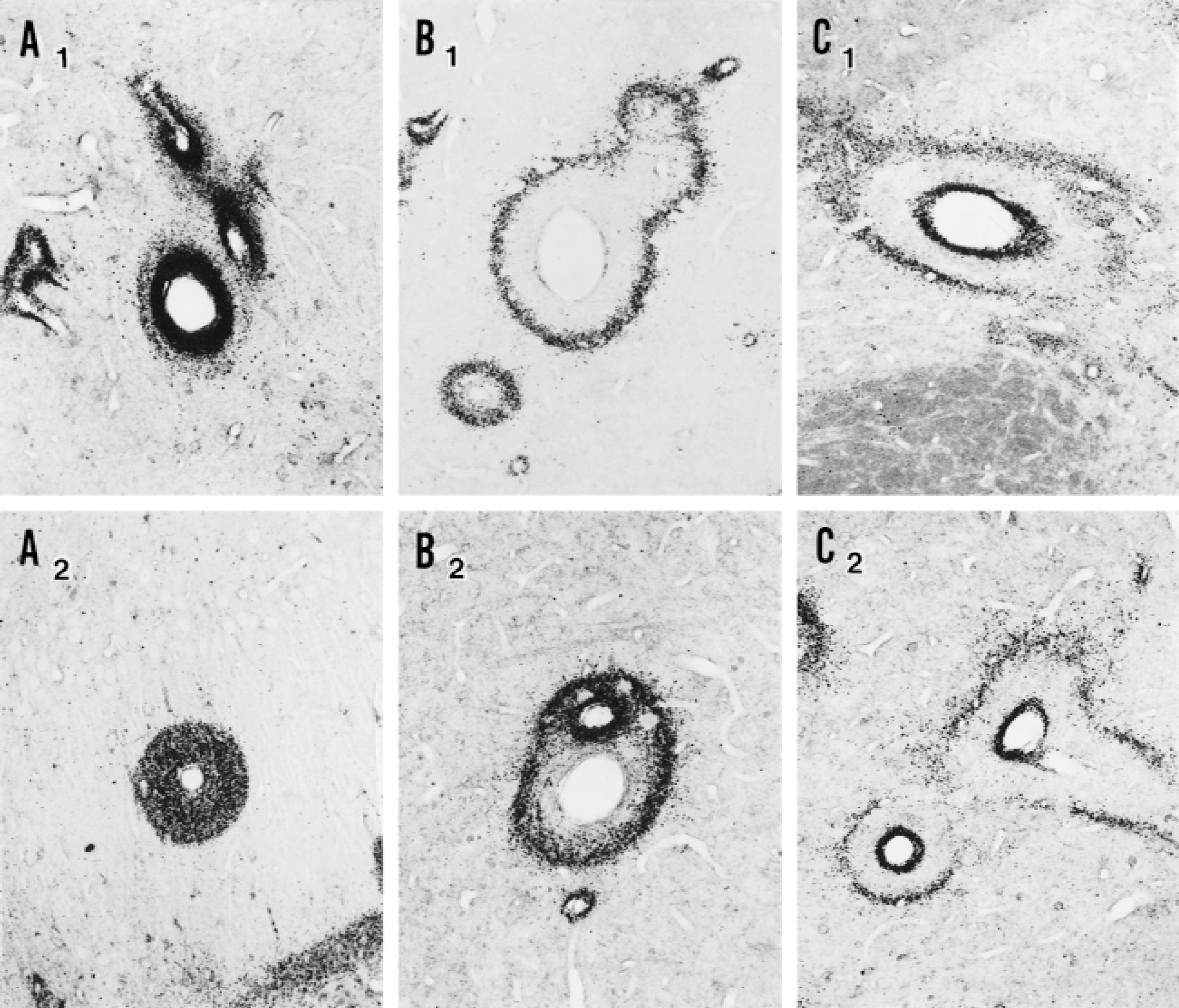
Patterns of manganese superoxide dismutase (MnSOD) expression around brain vessels of spontaneously hypertensive stroke-prone rats.
Double-label immunofluorescence with antibodies against MnSOD, NeuN, NF-200, GFAP, CNPase, von Willebrand Factor, monocytes, and macrophages (ED-1) confirmed MnSOD expression in neurons, astrocytes, oligodendrocytes, endothelial cells, and perivascular macrophages, respectively (Fig. 3).
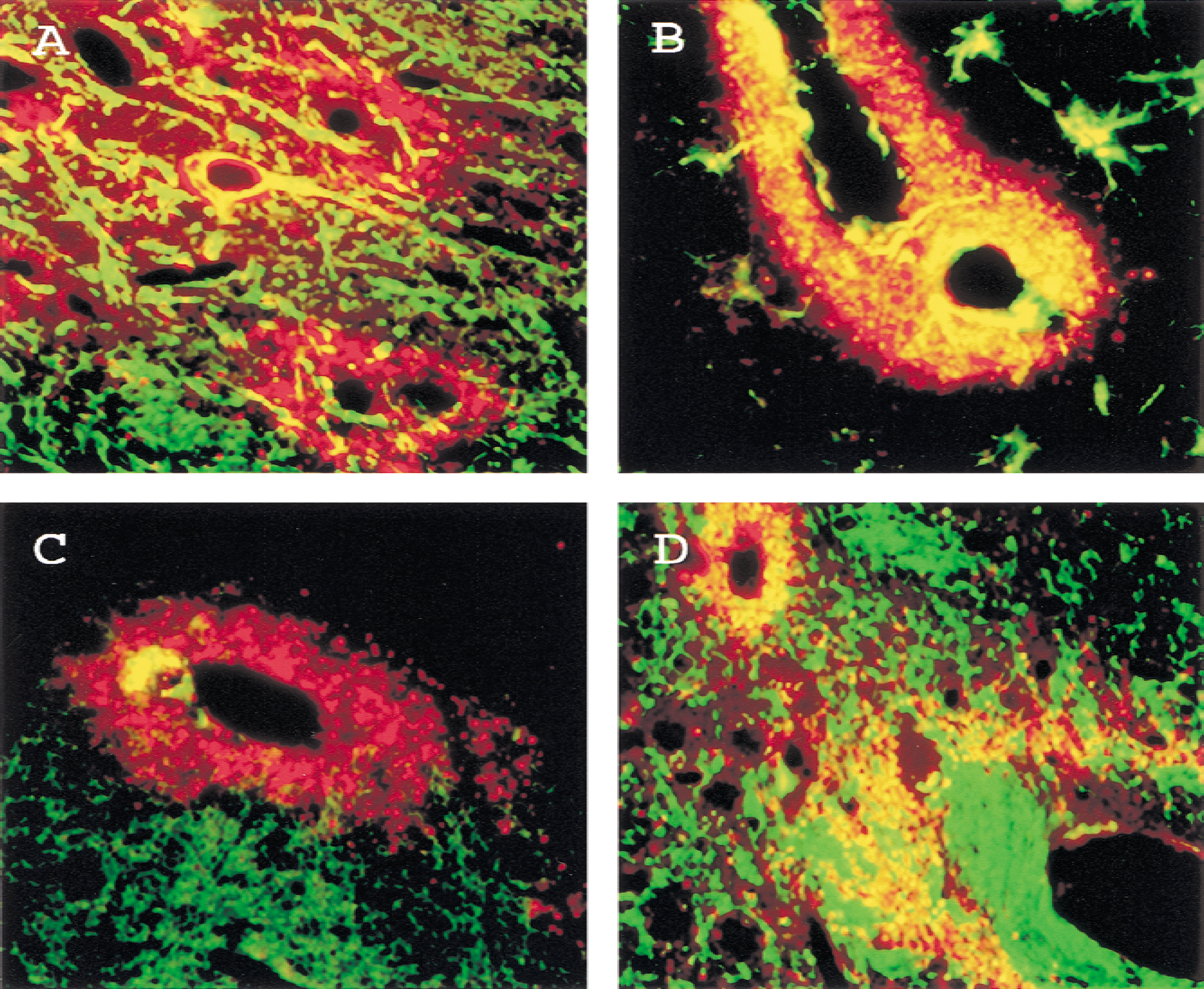
Double Immunofluorescence for manganese superoxide dismutase (MnSOD) and cell type markers in spontaneously hypertensive stroke-prone rats. Images were obtained with a CCD camera connected to the Metamorph Imaging System (Universal Imaging, West Chester, PA, U.S.A.). Brain sections were stained with rabbit anti-MnSOD and Rhodamine Red-X-conjugated goat anti-rabbit IgG (red). Tissue was also stained for the following cell type markers and visualized with fluorescein isothiocyanate conjugated horse anti-mouse IgG (green).
Immunohistochemistry for HO-1
The pattern of immunoreactivity for HO-1 in the brains of all experimental animals corresponded to the observed perivascular MnSOD pattern (Fig. 4). Clearly stained cuffs surrounded scattered blood vessels of all sizes in all areas of the brain, but were most prominent around vessels of the hippocampus, striatum, and thalamus. The staining was robust although not as strong as for MnSOD and was coincident with that antioxidant enzyme. Brains of untreated Wistar, WKY, and Sprague-Dawley rats as well as untreated SHR showed occasional lightly stained cuffing. Brains of untreated SHR-SP showed pronounced cuffing, as did all SHR that received IV injections of LPS. Robust HO-1 expression occurred by the 8-hour time point and persisted through 24 and 48 hours in all injected animals. The enzyme was expressed in cells with the morphology of endothelium, perivascular macrophages, microglia, and astrocytes.
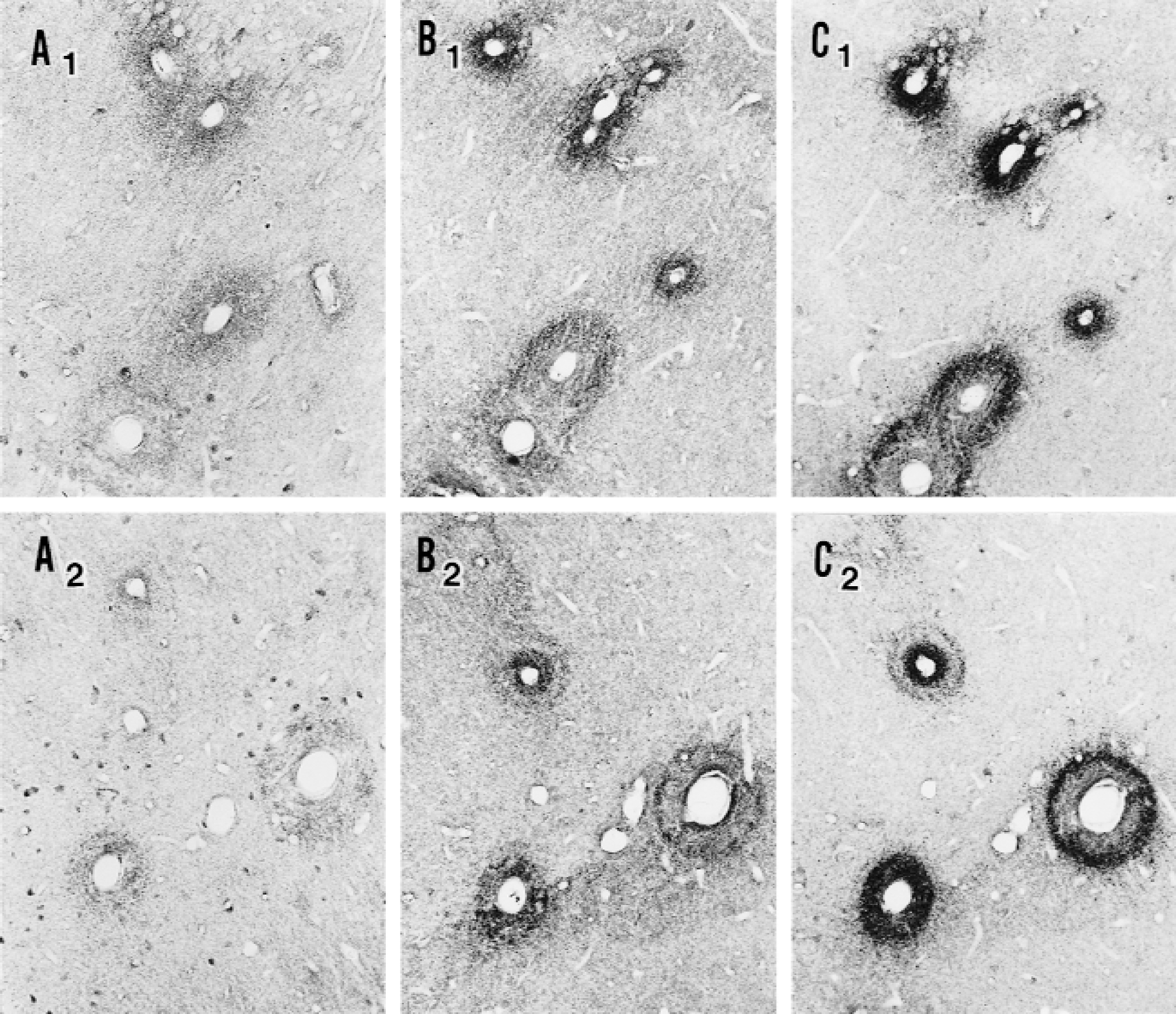
Immunostaining of adjacent vessel segments in adjacent hippocampal sections of naïve spontaneously hypertensive rates.
Immunohistochemistry for tumor necrosis factor-α
Tumor necrosis factor-α showed weak but clear staining in the same peculiar cuffing pattern around vessels of all sizes as described previously for MnSOD and HO-1 (Fig. 4). Its distribution was less extensive and its intensity was lower, but where it was expressed it matched closely the spatial distribution of the other immunoreactive molecules. Brains of untreated animals of all four strains showed only occasional cuffed vessels, whereas brains of untreated SHR-SPs clearly showed an increased frequency of vessels with cuffs. In the brains of animals that had received LPS or saline injection, expression of TNF-α was increased at all three time points over those that had not received any treatment. The most prominent cuffing was found at 24 hours in the animals that had received LPS. Tumor necrosis factor-α was expressed in cells with the morphology of endothelium, pericytes, astrocytes, and some neurons.
Immunohistochemistry for malondialdehyde and 4-hydroxynonenal
Immunohistochemistry for malondialdehyde (MDA) and 4-hydroxynonenal (HNE), cytotoxic aldehydes produced by lipid peroxidation, was generally negative. Only a few isolated vessels stained positively for MDA and HNE and their distribution rarely correlated with the expression of MnSOD.
Hematoxylin and eosin staining
Histopathologic analysis was performed on all brains and showed no pathologic changes or abnormalities. Even the brains and blood vessels of SHR-SP and LPS-stimulated SHR, which showed strong staining for MnSOD, showed no obvious pathology.
DISCUSSION
The current study shows that intraparenchymal brain vessels from normal rats have immunoreactive MnSOD surrounding them. The distribution patterns of this immunoreactivity suggest that an endogenous stimulus for up-regulation of MnSOD originates within scattered microvessel segments and expands centrifugally in cyclic waves. The same patterns were observed for HO-1 at a somewhat reduced intensity and also for TNF-α at still lower intensity. Interleukin-1 (IL-1) immunoreactivity had a distinctly different distribution suggesting that although IL-1 is capable of up-regulating MnSOD (Wong and Goeddel, 1988; Visner et al., 1990), it is not directly responsible for the concentric perivascular distribution noted in these studies. The rare occurrence of MDA and HNE immunoreactivity also followed a pattern that did not correspond to that of MnSOD. This finding indicated that the processes the authors infer to originate from activated brain vessels, expand radially outward, and induce synthesis of antioxidants usually did not lead to clear-cut oxidative damage in the form of lipid peroxidation.
Tumor necrosis factor-α expression has been detected in a number of different cell types in the brain under various experimental conditions. These include endothelial cells (Gourin and Shackford, 1997), perivascular macrophages (Woodroofe and Cuzner, 1993; Seilhean et al., 1997), microglia (Buttini et al., 1996), astrocytes (Lieberman et al., 1989), and neurons (Breder et al., 1994; Liu et al., 1994). After a single stimulus, such as intravenous LPS injection, TNF-α in circulating blood reaches a maximum within 90 minutes to 2 hours and returns to baseline by 6 hours (Feuerstein et al., 1990). In brain, TNF-α peaks at 7 hours and returns to baseline 18 hours after intravenous LPS injection (Buttini et al., 1997). The magnitude of its release in response to LPS is strongly enhanced by risk factors for stroke such as hypertension and advanced age (Sirén et al., 1992, 1993). Tumor necrosis factor-α has a number of proinflammatory and procoagulant effects on endothelium (Pober and Cotran, 1990; Hallenbeck, 1996). It stimulates expression of tissue factor and adhesion molecules for leukocytes, release of IL-1, nitric oxide, Factor VIII/von Willebrand Factor, platelet activating factor and endothelin, suppression of the thrombomodulin-protein C-protein S system, reduction of tissue plasminogen activator, and release of plasminogen activator inhibitor-1. Tumor necrosis factor-α can also stimulate production of reactive oxygen species in endothelium through induction of enzymes such as xanthine oxidase, cyclooxygenase, and NADPH oxidase as well as through other sources (Ishii et al., 1992; Weber et al., 1994; Terry et al., 1999). In addition, TNF-α can directly stimulate production of HO-1 and MnSOD (Visner et al., 1990; Terry et al., 1999). It does not stimulate expression of other antioxidant enzymes such as copper/zinc SOD, glutathione peroxidase, or catalase (Wong and Goeddel, 1988).
Heme oxygenase has three known isoforms. These isoforms consist of the inducible HO-1, the constitutive HO-2, and a recently identified isoform, HO-3 (McCoubrey et al., 1997). Endothelium (Terry et al., 1998), astrocytes, and neurons (Dwyer et al., 1995) produce HO-1 in response to stressful stimuli, particularly reactive oxygen species (ROS). Potential mechanisms advanced for TNF-α stimulation of HO-1 production include activation of protein kinase C and phospholipase A2, changes in intracellular calcium, and inhibition of protein phosphatase-1 and protein phosphatase- 2A (Terry et al., 1999). Heme oxygenase-1 reaches a maximum 4 hours after stimulation and largely disappears by 6 hours (Terry et al., 1998). In addition to providing a signal that indicates preceding cellular stress (Poss and Tonegawa, 1997; Mautes et al., 1998), HO-1 also serves as an antioxidant by cleaving the heme molecule, releasing free iron, and producing biliverdin, a precursor to bilirubin (Tenhunen et al., 1968). Heme oxygenase-1 enzymatic activity makes iron available for sequestration by ferritin, a storage protein that prevents the metal from participating in redox reactions (Balla et al., 1992). The bilirubin that is produced has antioxidant properties (Stocker et al., 1987).
Mammals have three distinct SOD genes that include MnSOD, cytoplasmic copper/zinc-SOD, and a secreted extracellular (EC-SOD) form of copper/zinc-SOD (Janssen et al., 1993). The location of MnSOD is mainly in mitochondria. Copper/zinc-SOD and EC-SOD are expressed constitutively and MnSOD is the only antioxidant enzyme that can be induced by cytokines such as TNF-α or by ROS (Wong and Goeddel, 1988). Mitochondria consume 90% of the oxygen used by cells (Richter et al., 1988; Bandy and Davison, 1990), of which 1% to 4% is converted to superoxide anions by the electron transfer chain (Wallace, 1992; Williams et al., 1998). This renders mitochondria especially vulnerable to oxygen radical attack. Manganese SOD plays a critical role as a first line of defense against superoxide anions that are produced during normal aerobic respiration in these organelles (Williams et al., 1998). It has been shown in the aged brain and heart that mitochondrial mutation frequency increases more than 100-fold (Cortopassi and Wang, 1995) and generation of endogenous oxidants (principally by mitochondria) is hypothesized to contribute to aging (Linnane et al., 1989; Beckman and Ames, 1998). In general, oxidative damage to mitochondrial DNA occurs at 16 times the frequency of nuclear DNA (Wallace, 1992). Some of the reasons for this extreme vulnerability are lack of protective histones, limited DNA repair mechanisms, and low replication fidelity (Bandy and Davison, 1990). These factors render mitochondrial MnSOD a key antioxidant enzyme.
The authors have postulated that perivascular cells can release TNF-α and convert endothelium in the involved vessel segment from an antithrombotic to a prothrombotic luminal surface (Hallenbeck et al., 1988, 1991). In small vessels, perivascular macrophages (Woodroofe and Cuzner, 1993; Seilhean et al., 1997) and endothelium itself (Gourin and Shackford, 1997) are potential sources of TNF-α. Additional cell sources in larger vessels include smooth muscle cells and lymphocytes particularly in atherosclerotic segments (Warner and Libby, 1989; Barath et al., 1990; Kishikawa et al., 1993). In response to a suitably intense LPS stimulus (for example, 1.8 mg/kg intracisternally), rats with identifiable risk factors for stroke such as streptozotocin-induced diabetes, hypertension, advanced age, or genetic stroke proneness release higher levels of TNF-α than stroke risk factor-free controls (Sirén et al., 1992, 1993) and develop signs of focal brain ischemia with an increased frequency (Hallenbeck et al., 1988). This focal vessel activation is viewed as tilting the local hemostatic balance (Rosenberg and Aird, 1999) in a prothrombotic direction and predisposition to thrombotic occlusion or loss of vessel integrity with hemorrhage.
The current findings provide a glimpse of the dynamism involved in focal hemostatic balance in the brain. The authors' interpretation is that under normal conditions, scattered intraparenchymal brain vessels randomly undergo cyclic fluctuation in proinflammatory and prothrombotic potential alternating with antiinflammatory and antithrombotic potential. These cyclic fluctuations originate within the vessels and spread radially outward to involve parenchymal cells and their processes within a perivascular circumference of approximately 50 μm. Periodic activation with local release of TNF-α and ROS tilt the hemostatic balance of vessel segments in a proinflammatory and prothrombotic direction. In response, vascular and perivascular cells exposed to these stimuli synthesize HO-1 and MnSOD to help restore hemostatic balance. Under normal circumstances, these cyclic fluctuations occur within a homeostatic range and local circulatory disturbances and tissue damage do not supervene. Larger fluctuations that exceed some threshold, however, could predispose local segments to thrombosis or hemorrhage. Genetic stroke proneness and activation of endothelium by LPS increase these cyclic fluctuations in brain parenchymal vessels.
Footnotes
Acknowledgments
The authors acknowledge the invaluable suggestions of Drs. Esther Shohami, Anna-Leena Sirén, and Maria Spatz. They also thank Mrs. Mary Crawford for excellent secretarial assistance.
