Abstract
Previous work in primary cell culture has shown that TNF-α and ceramide are involved in the signaling that induces tolerance to brain ischemia (Ginis et al., 1999; Liu et al., 2000). To validate the in vitro studies, the authors administered cell permeable analogs of ceramides intracisternally or intravenously to examine their effect on neuroprotection after focal cerebral ischemia. Permanent middle cerebral artery occlusion (MCAO) was performed in spontaneously hypertensive rats. Infarct volumes were assessed at 24 hours after surgery. D-erythro-N-acetylsphingosine (C2-ceramide) or its vehicle was infused intracisternally for 1 hour before MCAO. In a second set of studies, D-erythro-N-octanoylsphingosine (C8-ceramide) or its vehicle was injected intravenously 48 or 24 hours before MCAO to mimic preconditioning (PC) and was also injected 5 minutes after MCAO. C2-ceramide infusion significantly reduced infarct volumes by approximately 14% (P < 0.05). C8-ceramide injection reduced infarct volumes approximately 17% compared with controls. This effect was constant and significant compared with controls over the time periods examined (P < 0.01). This work supports findings in primary brain cell cultures that implicate ceramide as a downstream signal that is proximate to development of tolerance to brain ischemia. Because the degree of protection represents approximately 50% of the maximal infarct reduction observed in this model, there are probably additional signaling pathways that subserve tolerance.
Keywords
Brain acquires ischemic tolerance after various sublethal stress stimuli. A well-established model of this tolerance is neuronal survival in the CA1 sector of the hippocampus after sublethal ischemic preconditioning (PC) (Kitagawa et al., 1990; Kirino et al., 1991). Various proteins that promote survival of brain cells have been studied during ischemic tolerance. The expression of bcl-2 family proteins (Shimazaki et al., 1994; Shimuzu et al., 1998), manganese superoxide dismutase (Kato et al., 1995), interleukin-1 receptor antagonist (Barone et al., 1998), heat shock proteins (Kitagawa et al., 1991), and ubiquitins (Kato et al., 1993) have been demonstrated in this state. However, the intracellular signaling pathways that regulate expression of these proteins during induction of tolerance to ischemia have not been fully defined. Activation of the extracellular signal-regulated protein kinase cascade (Shamloo et al., 1999; Gonzalez-Zulueta et al., 2000) and diminished activation of p53 and several of its target genes (Tomasevic et al., 1999) have been reported.
Brief ischemic preconditioning induces tolerance to focal cerebral ischemia (Matsushima and Hakim, 1995; Chen et al., 1996). The authors found that PC with bacterial lipopolysaccharides (LPS) and tumor necrosis factor-alpha (TNF-α) induce a degree of tolerance to local ischemia that is similar to brief ischemic PC (Nawashiro et al., 1997; Tasaki et al., 1997). Lipopolysaccharides and its downstream product, TNF-α, can activate the sphingomyelin pathway for intracellular signaling (Kolesnick and Golde, 1994). Stress stimuli can cause a breakdown of sphingomyelin and generation of ceramide and phosphocholine through activation of various sphingomyelinases. Ceramides, a class of sphingolipids formed by the acylation of sphingosine, are not mere degradation products of sphingomyelin, but have been implicated broadly in cellular stress responses (Hannun, 1996). However, the role of ceramides differs in different cell types. Ceramides are known to play a role in proapoptotic signaling in a leukemia cell line (Jarvis et al., 1994), mesencephalic neurons (Brugg et al., 1996), and synovial fibroblasts (Ichinose et al., 1998). Conversely, the administration of a ceramide analog improved cognitive function after focal cerebral ischemia (Inokuchi et al., 1998) and promoted cell survival in spinal motor (Irie and Hirabayashi, 1998), sensory (Ping and Barrett, 1998), and sympathetic neurons (Ito and Horigome, 1995). Lymphoblasts from patients with Niemann-Pick disease who are deficient in acid sphingomyelinase are resistant to radiation-induced apoptosis (Santana et al., 1996). Other results support a role for ceramide administered exogenously in the survival of cerebellar Purkinje cells (Furuya et al., 1998) and hippocampal neurons (Goodman and Mattson, 1996). These contradictory results on the effects of ceramide are further complicated by the ability of cells to synthesize ceramide de novo by condensation of serine and palmitoyl-CoA. This pathway requires several hours to generate ceramides (Bose et al., 1995).
The authors recently have shown that hypoxic PC in cultured cortical neurons results in up-regulation of intracellular ceramide levels that peak at 24 hours and elicit resistance to a subsequent hypoxic insult (Liu et al., 2000). Addition of fumonisin B1, an inhibitor of de novo synthesis of ceramides, to the cultures reduced ceramide levels and nullified the effect of hypoxic PC (Liu et al., 2000). Based on this result, the authors hypothesized that ceramides could induce tolerance to focal cerebral ischemia and, as a downstream intracellular signaling molecule, reduce the latency for its development. If ceramide was to be the single intracellular signal that regulates tolerance, the authors reasoned that exogenous ceramide should trigger a reduction of infarct size similar to that observed after PC with LPS or TNF-α.
MATERIALS AND METHODS
Reagents
Reagents were obtained from the following sources: D-erythro-N-acetylsphingosine (C2-ceramide, MW 341.5) from Sigma (St. Louis, MO, U.S.A.); D-erythro-N-octanoyl-sphingosine (C8-ceramide, MW 425) from Matreya (Pleasant Gap, PA, U.S.A.); dimethyl sulfoxide (DMSO, pH 7.0) from Sigma. The D-erythro configuration of ceramide is required for ceramide signaling activity. Dimethyl sulfoxide does not induce sphingomyelin hydrolysis (Hannun et al., 1993).
Animals
Adult male spontaneously hypertensive rats (SHR), 13 to 16 weeks, were obtained from Charles River Labs (Wilmington, MA, U.S.A.) and Taconic Farms (Germantown, NJ, U.S.A.). They were maintained in animal quarters with a standard 12-hour light/dark cycle with humidity and temperature controlled at normal levels. Water and food were allowed ad libitum. All procedures in animals were in strict accordance with the NIH Guide for the Care and Use of Laboratory Animals and were approved by the local Animal Care and Use Committee. Rats were fasted 16 hours before surgery, but had free access to water.
Experimental group
Experiment 1: intracisternal administration
Rats were anesthetized initially with 4% isoflurane in 30% oxygen to 70% nitrous oxide gas mixture. Atropine sulfate (0.1 mg/kg; Research Biomedical International, Natick, MA, U.S.A.) was administered intraperitoneally and the rats were intubated transorally. The isoflurane concentration was reduced to 1.0% to 1.5% after intubation and the rats were maintained on a mechanical ventilator (Harvard Rodent Ventilator Model 683; Harvard Instrument, South Natick, MA, U.S.A.). The right femoral artery was cannulated (PE-50; Clay-Adams, Parsippany, NJ, U.S.A.) for blood pressure monitoring and for serial blood gas sampling. Rats were fixed to a stereotaxic apparatus (Stoelting, Wood Dale, IL, U.S.A.). Intracisternal infusion was induced as described previously (Horinaka et al., 1997), with modification. Briefly, after opening of the atlanto-occipital membrane, a silastic catheter (0.31 mm inner diameter × 0.64 mm outer diameter; Helix Medical, Carpinteria, CA, U.S.A.) was inserted 5 mm into the left cerebellomedullary cistern for the administration of either C2-ceramide in a solution of DMSO (n = 8) or its vehicle (DMSO; n = 6) and was fixed at the back of the neck. After fifteen minutes for stabilization, intracisternal infusion was initiated. Infusion was maintained by a microinfusion pump (CMA/Microdialysis, Model CMA/100, Acton, MA, U.S.A.) at a rate of 1.26 μL/min and continued for 1 hour, followed immediately by middle cerebral artery occlusion (MCAO). In a preliminary study, Evans Blue infused in this manner at 1.26 μL/min had reached the MCAO territory. The dosages of C2-ceramide used in this study were selected on the basis of a pilot study in which 100 μg/kg C2-ceramide, but not 0.1, 1, 10 μg/kg, or 1 mg/kg C2-ceramide appeared to be optimally effective in reducing infarct size.
Controls were infused with the same volume of DMSO at the same infusion rate. Middle cerebral artery occlusion was induced as described previously (Tamura et al., 1981), with modification. Briefly, a 1.5-cm skin incision was placed between the left margin of the orbit and the tragus. The temporalis muscle was incised and retracted to expose the squamous portion of the temporal bone. A craniectomy was made at the juncture of the zygomatic process and the temporal bone. The dura was opened with a 30-gauge needle to expose the middle cerebral artery (MCA). The MCA was electrocauterized between the olfactory tubercle and the distal segment of the MCA. The olfactory branch of the MCA, which is consistently present, was preserved. The coagulated MCA segment was then transected with microscissors to verify that the occlusion was permanent. The intracisternal catheter was ligated and left in situ to avoid cerebrospinal fluid leakage. Rats were returned to their cages after surgery and allowed free access to food and water. A single dose of antibiotics (gentamicin, 2.5 mg/kg; Gibco BRL, Grand Island, NY, U.S.A.) was applied topically before closing the surgical sites, and 30% lidocaine cream (Foer's Pharmacy Bethesda, Bethesda, MD, U.S.A.) was also applied to all surgical sites as an analgesic. Laser-doppler cerebral blood flow measurements in a subset of 6 animals pre-MCAO and during the first 60 minutes after MCAO showed no differences between C2-ceramide- and DMSO-infused animals.
Experiment 2: intravenous administration
The methods of induction and maintenance of anesthesia, surgical MCAO, and postoperative care were exactly the same as for experiment 1. Animals were infused with C8-ceramide or DMSO at one of three time points, 48 or 24 hours before MCAO or 5 minutes after MCAO. A catheter (PE-50; Clay-Adams, Parsippany, NJ, U.S.A.) was inserted 2.5 cm into the right external jugular vein for administration of either C8-ceramide in a solution of DMSO or its vehicle (DMSO). The dosages of C8-ceramide used in this experiment were selected on the basis of a pilot study in which 1 mg/kg C8-ceramide, but not 1, 10, or 100 μg/kg C8-ceramide appeared to be optimally effective in reducing the infarct size. A 1 mg/kg dose of C8-ceramide was administered through the right external jugular vein over a 5-minute period at the following time points: 1) 48 hours before MCAO (n = 6 in each group), (2) 24 hours before MCAO (n = 5 in each group), and (3) 5 minutes after MCAO (n = 5 in each group). The catheter was removed after the injection and the vein was ligated. Laser-doppler cerebral blood flow measurements in a subset of 6 animals pre-MCAO and during the first 60 minutes after MCAO showed no differences between C8-ceramide- and DMSO-infused animals.
Physiologic variables
Physiologic variables were monitored to assess the physiologic status of the animals before MCAO and 30 minutes after MCAO. Mean arterial blood pressure was monitored by means of a TA 4000 transducer amplifier (Gould, Valley View, OH, U.S.A.). Pao2, Paco2, pH, and hematocrit were measured with a Corning pH/Blood Gas Analyzer (Model No. 288; Corning Medical, Medfield, MA, U.S.A.) by withdrawal of 100 μL of blood from the indwelling femoral arterial catheter. The same volume of pyogen-free warmed (37°C) heparinized saline (10 U/mL) was injected after each arterial blood sampling. Rectal and temporal muscle temperatures were measured and maintained at 37 ± 0.5°C with a heating blanket (K-20; American Pharmaseal, Valencia, CA, U.S.A.) during all surgical procedures and 2 hours after surgery. In experiment 1, mean arterial blood pressure and the rectal temperature were monitored during intracisternal infusion.
Assessment of infarct size
Twenty-four hours after MCAO, the animals were deeply anesthetized with pentobarbital (65 mg/kg intraperitoneal injection). In experiment 1, the rats were perfused with 200 mL heparinized phosphate-buffered saline (PBS) (10 U/mL, pH 7.4) followed immediately by 400 mL of 4% paraformaldehyde in 0.1 mol/L phosphate buffer (pH 7.43). Their brains were left in situ overnight and then cryoprotected by transfer to 15% (w/v) sucrose in PBS at 4°C. Brains were then cut into 20-μm sections in a cryostat (Jung Frigocut 2800 E; Leica, Nussloch, Germany) at −20°C at 12 predetermined coronal levels (bregma +3.70 mm to −7.30 mm) at 1-mm intervals. Sections were stained with cresyl violet. In experiment 2, the brains were removed and suspended in PBS at 4°C for 30 minutes and sectioned coronally into eight 2-mm slices in a rodent brain matrix (RBM-4000C; Activational System, Warren, MI, U.S.A.). Slices were placed in 2% 2,3,5-triphenyltetrazolium chloride (Sigma) at room temperature for 30 minutes in the dark followed by 4% paraformaldehyde in 0.1 mol/L phosphate buffer (pH 7.43) overnight. Infarct size was determined by means of a digitized imaging system (NIH image, v.1.62). Volumes were calculated by integration of the sequential areas. Volumes of ischemic injury were corrected for brain swelling as previously described (Leach et al., 1993). Perfusion followed by fixation was adopted in experiment 1 to avoid brain injury by the implanted catheter during the removal of the brain. The variation in infarct volumes between experiment 1 and 2 is explained by these methodologic differences.
Statistics
Data are expressed as mean ± SD. Unpaired Student's t-test was used to analyze the differences in physiologic parameters and in infarct volumes in experiment 1. Two-way analysis of variance (ANOVA) was used to analyze the interaction between treatment group and time in experiment 2. If differences reached significance (two-tailed, P < 0.05), post hoc comparisons were made with Tukey's multiple comparison test.
RESULTS
Dimethyl sulfoxide has been reported to influence outcome from cerebral ischemia (de la Torre, 1983). The dose of DMSO used in this study neither reduced nor augmented infarct volumes compared with the same dose of PBS (DMSO: n = 6, 188 ± 8 mm3; PBS: n = 4, 198 ± 32 mm3). Based on this result, DMSO was used as the vehicle control for C2- and C8-ceramides, and PBS as a control for DMSO was not incorporated into experiments 1 and 2.
Physiologic variables
In experiment 1, intracisternal infusion did not cause any changes in mean arterial blood pressure or rectal temperature between the rats treated with C2-ceramide and those that received vehicle (Table 1). Hematocrit 30 minutes after MCAO in rats infused with vehicle was significantly lower than that in rats with C2-ceramide (P < 0.05, unpaired Student's t-test) (Table 1). However, these values were within normal limits. Physiologic variables in experiment 1 did not otherwise differ. In experiment 2, although it was within normal limits, arterial blood pH was significantly higher at 30 minutes after MCAO in the rats treated with C8-ceramide 5 minutes after MCAO than in controls (P < 0.01, unpaired Student's t test) (Table 2). There were no other significant differences in physiologic variables in experiment 2 (Table 2).
Physiologic variables in each experimental group of experiment 1 (intracisternal administration)
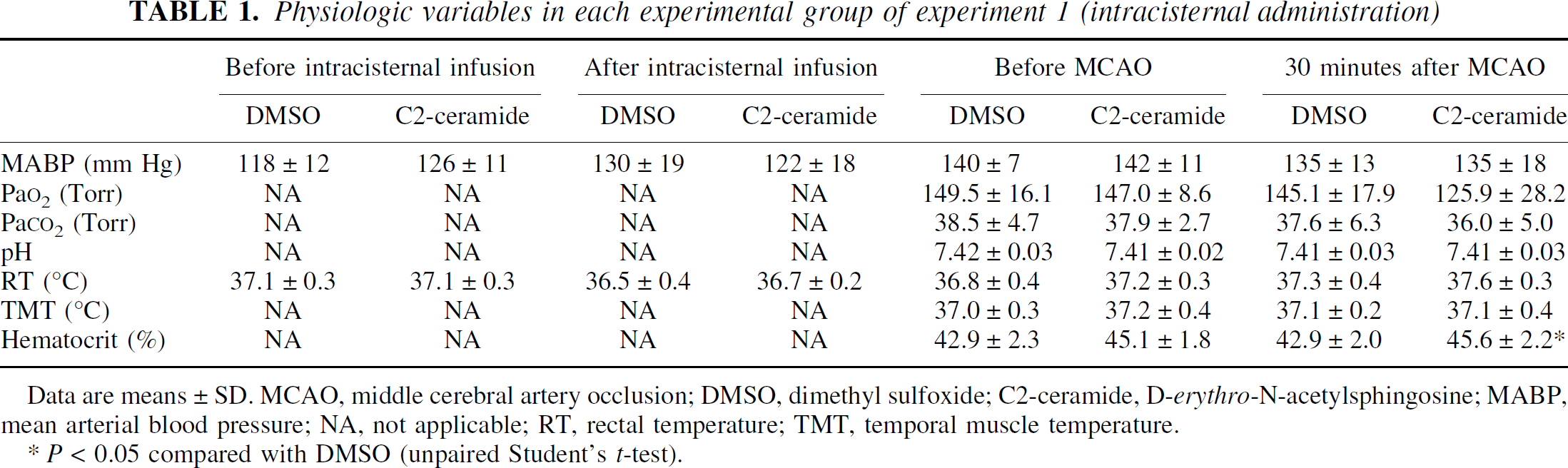
Data are means ±SD. MCAO, middle cerebral artery occlusion; DMSO, dimethyl sulfoxide; C2-ceramide, D-erythro-N-acetylsphingosine; MABP, mean arterial blood pressure; NA, not applicable; RT, rectal temperature; TMT, temporal muscle temperature.
P < 0.05 compared with DMSO (unpaired Student's t-test).
Physiologic variables in each experimental group of experiment 2 (intravenous administration)
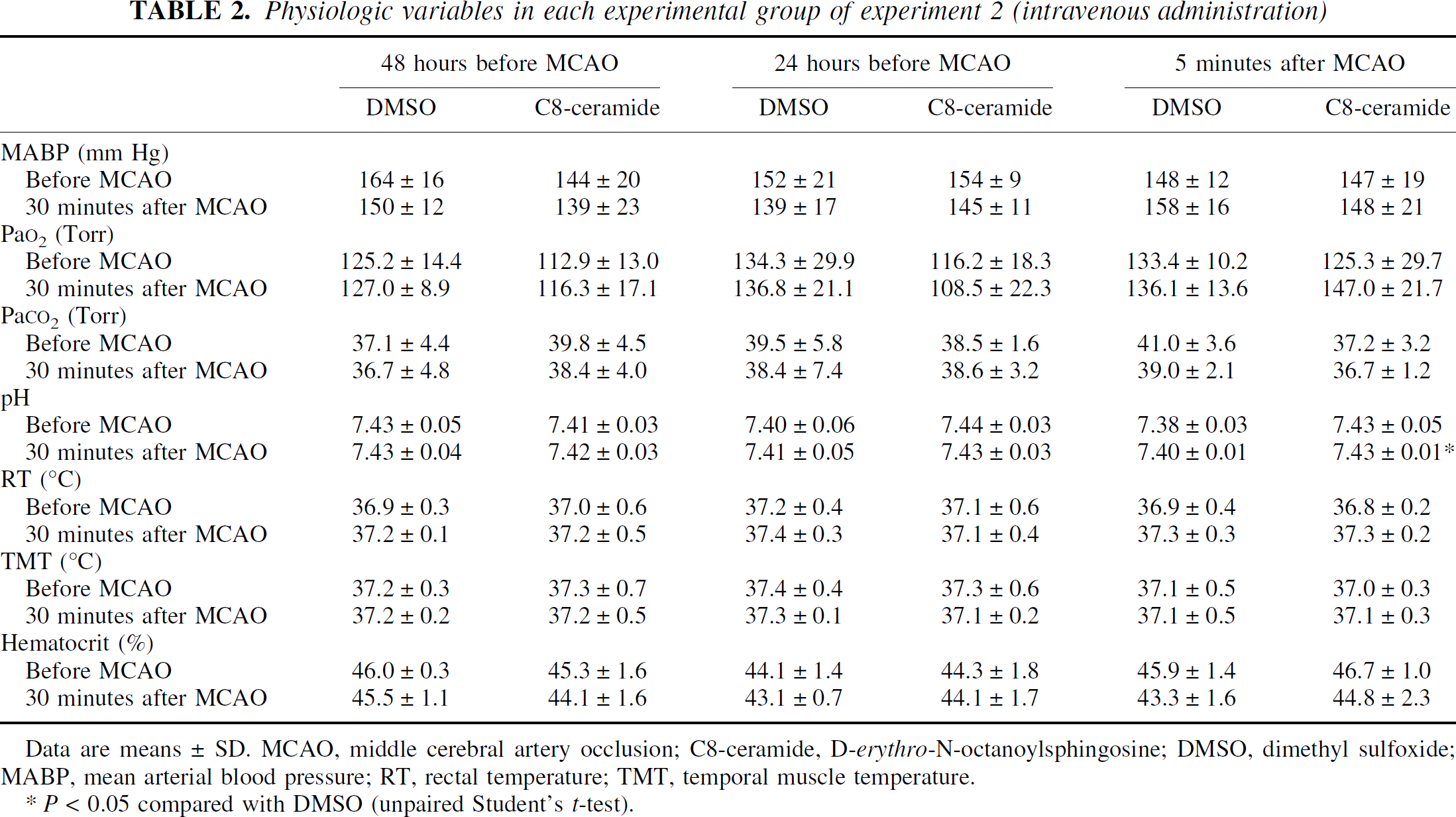
Data are means ±SD. MCAO, middle cerebral artery occlusion; C8-ceramide, D-erythro-N-octanoylsphingosine; DMSO, dimethyl sulfoxide; MABP, mean arterial blood pressure; RT, rectal temperature; TMT, temporal muscle temperature.
P < 0.05 compared with DMSO (unpaired Student's t-test).
Infarct volumes in experiment 1 (intracisternal administration)
Intracisternal C2-ceramide infusion produced a significant reduction in infarct volume assessed 24 hours after MCAO as compared with the values of intracisternal vehicle infusion animals (DMSO: n = 6, 151 ± 18 mm3; C2-ceramide: n = 8, 130 ± 15 mm3; P = 0.03, unpaired Student's t-test). The reduction of the infarct volume was 14% in the C2-ceramide infused group (Fig. 1).
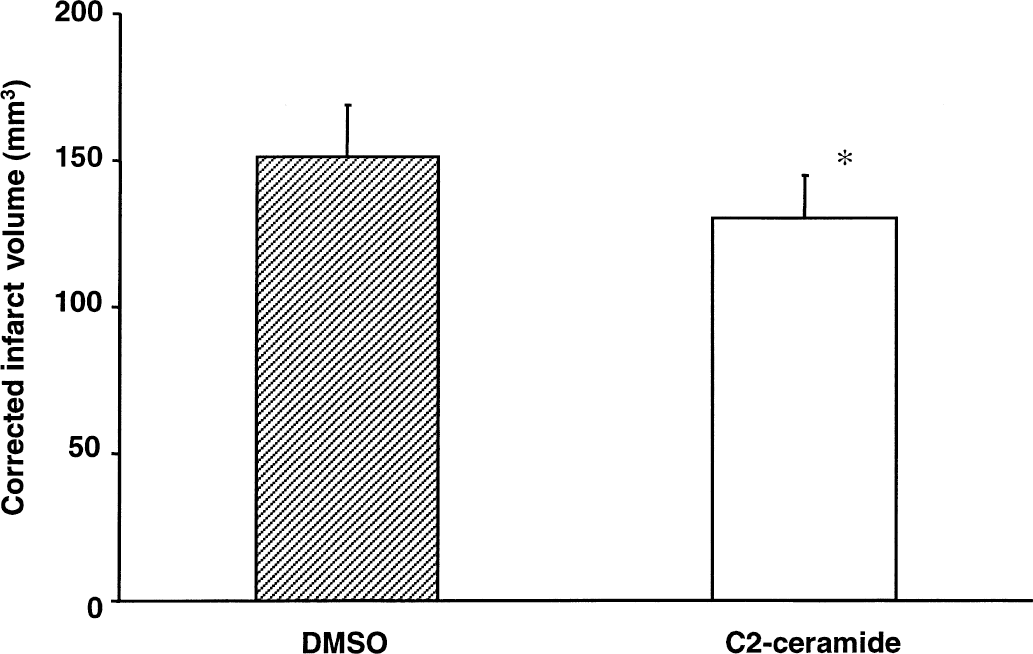
Bar graph showing effects of intracisternal infusion of D-erythro-N-acetylsphingosine (C2-ceramide) on infarct volumes. The values represent group averages (means ± SD). C2-ceramide infusion decreased the infarct size (*P < 0.05 compared with vehicle infusion, unpaired Student's t-test). DMSO, dimethyl sulfoxide.
Time and treatment effect on infarct volumes in experiment 2 (intravenous administration)
Time main effect (48 vs. 24 hours before MCAO vs. 5 minutes after MCAO) on infarct size was not significant (P = 0.767). Interaction of treatment with time also was not significant (P = 0.873). However, the treatment main effect was significant (P = 0.007, two-way ANOVA followed by Tukey's multiple comparison test). This means that the animals treated with C8-ceramide showed a significant decrease in infarct volumes compared with vehicle-treated animals at all time points (48 hours before MCAO: DMSO intravenously, n = 6, 189 ± 21 mm3; C8-ceramide: n = 6, 160 ± 43 mm3; 24 hours before MCAO: DMSO intravenously, n = 5, 206 ± 38 mm3; C8-ceramide: n = 5, 164 ± 23 mm3; 5 minutes after MCAO: DMSO intravenously, n = 5, 193 ± 7 mm3; C8-ceramide: n = 5, 164 ± 43 mm3) (Fig. 2). The average infarct volume reduction in C8-ceramide-treated animals in these experiments was approximately 17%. Overall, the infarct volume reduction in the C2-ceramide and C8-ceramide treatment groups was 16%.
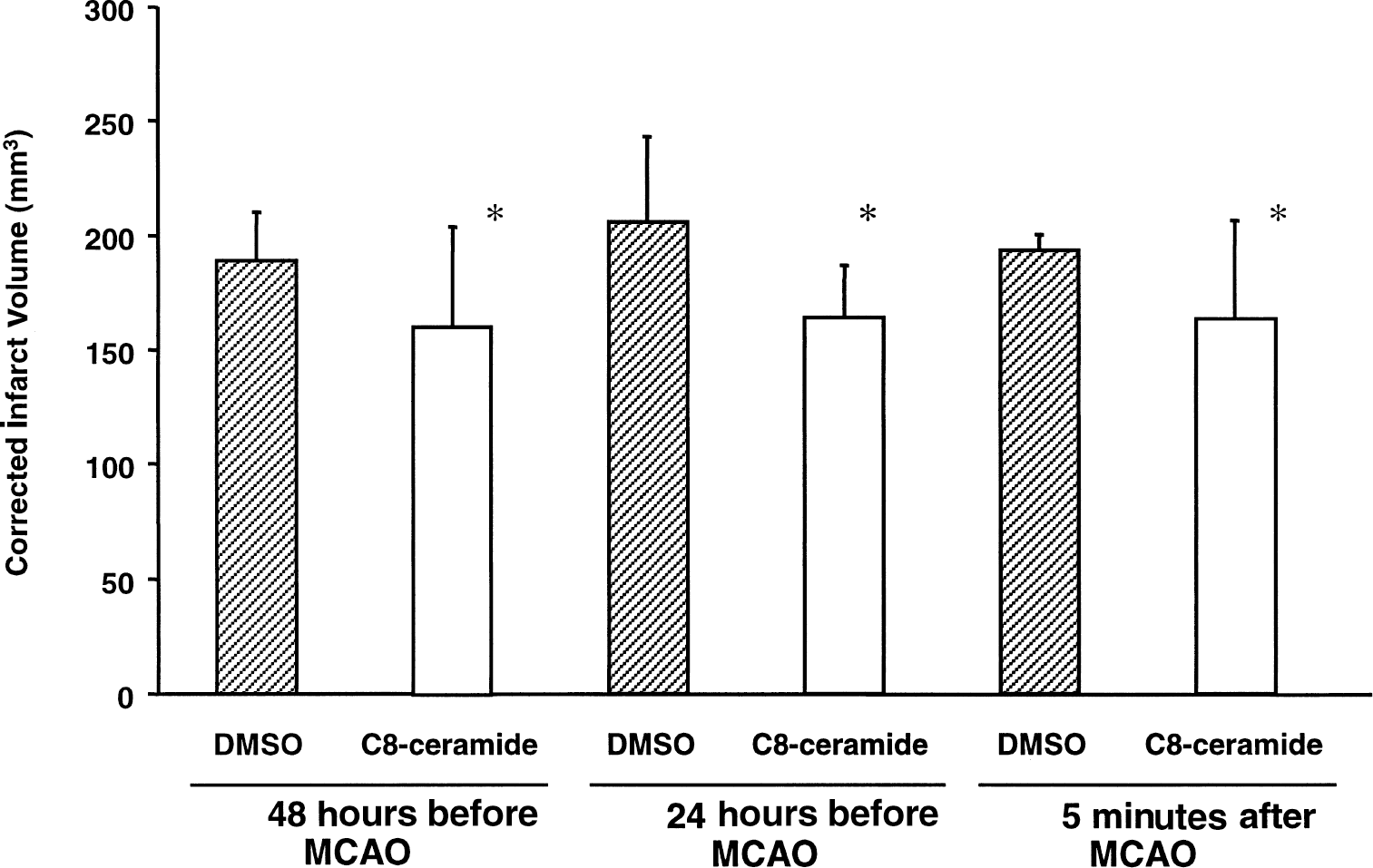
Bar graph showing effect of D-erythro-N-octanoylsphingosine (C8-ceramide) on infarct volumes. The values represent group averages (means ± SD). Preconditioning with C8-ceramide 48 and 24 hours before middle cerebral artery occlusion (MCAO) reduced the infarct size (*P < 0.01, compared with vehicle-treated group, two-way analysis of variance (ANOVA) followed by Tukey's multiple comparison test). Similarly, administration of C8-ceramide 5 minutes after MCAO reduced the infarct size (*P < 0.01, compared with vehicle-treated group, two-way ANOVA followed by Tukey's multiple comparison test). DMSO, dimethyl sulfoxide.
DISCUSSION
The current work demonstrates that cell permeable exogenous ceramides (C2-ceramide and C8-ceramide) administered by two different routes do have a small, but statistically significant effect on reduction of infarct volumes produced by pMCAO in SHR. The overall 16% infarct volume reduction represents approximately 50% of the maximum salvage that has been achievable in this model (Morikawa et al., 1994; Dawson et al., 1999; Tasaki et al., 1997). In experiment 1, the authors adopted an intracisternal route for C2-ceramide administration to assure exposure of ischemic brain to the agent. The timing of the infusion corresponded to in vitro studies, in which ceramide needed to be elevated near the time of the hypoxic insult (Liu et al., 2000). An intraventricular route was not selected because previous studies had shown that fluorescence-conjugated ceramide is retained in the paraventricular area for 24 hours after intraventricular injection (Di Biase et al., 1991). The authors hypothesized that ceramide is a downstream signaling molecule that mediates the cytokine-induced ischemic tolerant state, and thus administration of a cell-permeable ceramide was expected to produce this tolerant state in the absence of LPS or TNF-α preconditioning. In fact, ceramide analogs bypass the receptors and mimic many of the biologic effects evoked by agonists such as TNF-α (Wiegmann et al., 1994). In experiment 2, the authors adopted the intravenous route for ceramide administration at several different time points. They found that by the time these experiments began intravenous administration of fluorescently labeled NBD-C6 ceramide (6-N-(7-nitrobenz-2 oxa-1, 3-diazol-4yl) aminocaproylsphingosine) penetrated the blood-brain barrier as early as 30 minutes after injection and became detectable in brain parenchymal cells. The fluorescent ceramide remained visible for at least 24 hours in the brain.
C2-ceramide is widely used in cell signaling studies. The structure of C2-ceramide is actually closer to sphingosine than to ceramides (Ping and Barrett, 1998). Ceramides are converted to glucosylceramides (GlcCer) by glucosyltransferase (glucosylceramide synthase) and to sphingosine by ceramidase. Sphingosine and sphingosine-1-phosphate have been noted to act in antiapoptotic processes (Cuvillier et al., 1996) and in myocardial protection after ischemia-reperfusion injury (Murohara et al., 1995). Because there was a possibility that the short-chain ceramide would mimic the activity of sphingosine rather than ceramide, the authors used C8-ceramide to minimize this possibility in experiment 2. The overall protective effect of ceramide (16% infarct volume reduction) did not reach the values obtained from the study of LPS tolerant models (≈30% reduction) at any of the time points examined. However, this effect was significant and provides useful information regarding signal transduction in cytokine-induced tolerance to subsequent ischemia. The current results suggest that ceramide plays a partial role in induction of tolerance to ischemia and that additional signaling pathways exist that contribute to the full induction of tolerance by LPS and TNF-α.
Recently, ceramide has been reported to activate the endothelial isoform of nitric oxide synthase (eNOS), which is independent of intracellular calcium concentrations ([Ca2+]i) (Igarashi et al., 1999). Ceramide also reduced phenylephrine-induced vasoconstriction and inhibited elevation in [Ca2+]i in vascular smooth muscle (Zheng et al., 1999). Moreover, the relevance of eNOS to LPS-induced tolerance has been reported. The endothelial isoform of NOS was up-regulated as tolerance developed after PC with LPS, and the protective effect of LPS was reduced by the NOS blocker, nitro-L-arginine methyl ester (Puisieux et al., 2000). In addition, eNOS and sphingomyelin are condensed in caveolae (Michel and Feron, 1997). These findings suggest a close relation among LPS, ceramide, and eNOS in improving cerebral perfusion in tolerant animals (Dawson et al., 1999).
In summary, the current results validate in vitro studies that have identified ceramide as a downstream signaling molecule in the LPS pathway for induction to brain ischemia. Moreover, ceramide analogs have a protective effect and do not need to be administered 24 to 48 hours before a stroke. Ceramide-induced infarct volume reduction is significant, but incomplete, compared with the effects of LPS in the previous study (Tasaki et al., 1997), which suggests that there are some additional signaling pathways mediating the tolerance to subsequent ischemia induced by LPS preconditioning.
Footnotes
Abbreviations used
Acknowledgments
The authors are indebted to Mrs. Mary W. Crawford for preparation of the manuscript. The authors also acknowledge Dr. Naoaki Horinaka, Laboratory of Cerebral Metabolism, National Institute of Mental Health, for critical advice concerning the intracisternal infusion technique.
