Abstract
Neuropeptide Y (NPY) is an important vasoconstrictor in the cerebral circulation. Its constrictor response is because of activation of NPY receptors on the vascular smooth muscle (VSM). Little is known regarding the effects of NPY on the endothelium. In the current study, the authors tested the hypothesis that NPY can either constrict or dilate rat middle cerebral arteries (MCAs). Constriction is elicited by stimulating receptors on the VSM; dilation is elicited by stimulating receptors on the endothelium. Middle cerebral arteries were isolated, cannulated with micropipettes, pressurized to 85 mm Hg, and luminally perfused. The extraluminal application of NPY (mixed agonist), [Leu31, Pro34]-NPY (Y1 agonist), or NPY-[13–36] (Y2 agonist) produced concentration-dependent constrictions. BIBP 3226 (Y1 selective antagonist) significantly attenuated the NPY-and [Leu31, Pro34]-NPY–induced constrictions. The luminal application of NPY, [Leu31, Pro34]-NPY, and NPY-[13–36] produced concentration-dependent dilations of MCAs. The maximum dilation produced by the NPY receptor agonists was approximately 40% of the dilation elicited by the luminal administration of 10−5 mol/L ATP. Dilations elicited by luminal NPY, [Leu31, Pro34]-NPY, or NPY-[13–36] were abolished by inhibition of nitric oxide synthase with 10−5 mol/L Nω-nitro-L-arginine methyl ester (L-NAME) or removal of the endothelium. Dilations produced by luminal NPY or luminal [Leu31, Pro34]-NPY were not affected by BIBP 3226. Stimulation of NPY receptors on vascular smooth muscle constricted MCAs. Stimulation of an NPY receptor other than the Y1 subtype on endothelium dilated the MCAs by releasing nitric oxide.
Neuropeptide Y (NPY) is one of the most prevalent neuropeptides in the peripheral and central nervous systems (Lundberg et al., 1996; Michel et al., 1998). It is colocalized and coreleased with noradrenaline (NA) in sympathetic nerves innervating the cardiovascular system (Edvinsson et al., 1987; Lundberg et al., 1989). In addition to constricting peripheral and cerebral arteries by stimulating receptors on the vascular smooth muscle (Edvinsson et al., 1983; Edvinsson, 1985; Pernow et al., 1987; Zukowska-Grojec et al., 1987; Abounader et al., 1995), NPY also potentiates vasoconstriction induced by other contractile agents including norepinephrine (Edvinsson et al., 1984; Macho et al., 1989; Westfall et al., 1995; Fabi et al., 1998). The vasoconstrictor response to NPY is mediated primarily by postjunctional NPY Y1 receptors as shown by reverse transcription-polymerase chain reaction (RT-PCR) and pharmacologic studies (Lundberg et al., 1982; Fuhlendorff et al., 1990; Wahlestedt et al., 1990; Wahlestedt and Reis, 1993; Oellerich and Malik, 1993; Nilsson et al., 1996a,b); however, in some vascular beds, postjunctional Y2 receptors can also mediate vasoconstriction (Grundemar et al., 1992; McAuley and Westfall, 1992; Tessel et al., 1993; Palea et al., 1995; Kotecha, 1998).
Recent studies are now demonstrating an expanding role for NPY in the vascular system. In peripheral vessels, NPY and its mRNA have been reported extraneuronally in endothelium (Loesch et al., 1992; Zukowska-Grojec et al., 1998) and the endothelium appears to possess both the NPY Y1 and Y2 receptor subtypes (Sanabria and Silva, 1994; Jackerott and Larsson, 1997; Zukowska-Grojec et al., 1998). Although little is known about the function of NPY and its receptors on the endothelium, they have been reported to (1) regulate vascular permeability (Noll et al., 1996), (2) control angiogenesis (Zukowska-Grojec et al., 1998), and (3) regulate the release of endothelium-derived constricting factors (Prieto et al., 1995; Fabi et al., 1998). In the current study, the authors have demonstrated for the first time that cerebral vessels possess endothelial NPY receptors mediating vasodilation through an NO-dependent mechanism. Neuropeptide Y was found in the plasma of mammals and was in sufficient concentration to potentially stimulate NPY receptors on the luminal surface of the endothelium (Zukowska-Grojec and Wahlested, 1993).
Evidence has emerged implicating a dilator role for NPY receptors on endothelium (Kobari et al., 1993; You et al., 1995; Torffvit and Edvinsson, 1997). Precontracted renal arteries of diabetic (streptozotocin) and nondiabetic rats relaxed to NPY. The relaxation was attenuated by 40% in arteries from diabetic rats after nitric oxide synthase (NOS) inhibition but not in arteries from the nondiabetic control (Torffvit and Edvinsson, 1997). In the cerebral circulation, an injection of NPY into the carotid artery of the cat transiently increased cerebral blood flow (Kobari et al., 1993). Inhibition of NOS prevented the flow increase, thus implicating the role of endothelium and NO (Kobari et al., 1993). In a study from this group, NPY transiently dilated precontracted basilar arteries isolated from the guinea pig (You et al., 1995). Inhibiting NOS or removal of the endothelium (You et al., 1995) abolished the dilation. However, the concentration of NPY (10−6 mol/L) required to elicit the dilation was at least 100-fold greater (and likely closer to 1000-fold) than that expected in the plasma during the most extreme conditions (Zukowska-Grojec and Wahlested, 1993).
Although the above studies (Kobari et al., 1993; You et al., 1995; Torffvit and Edvinsson, 1997) are provocative, more evidence is needed before it can be firmly established that endothelial NPY receptors have a dilator function in the cerebrovascular circulation. To investigate the role of NPY receptors in the regulation of the cerebrovascular circulation, the authors tested three hypotheses using middle cerebral arteries (MCAs) from rats. Hypothesis 1 suggested that stimulation of receptors on the vascular smooth muscle by NPY constricts the MCA. Hypothesis 2 suggested that stimulation of receptors on the endothelium by NPY dilates the MCA; the dilation occurs in the physiologic concentration range of plasma NPY for the rat (0.1 to 10 nmol/L) (Zukowska-Grojec and Wahlested, 1993). If Hypothesis 2 proved to be true, then the authors would test a third hypothesis involving the dilation. Hypothesis 3 suggested synthesis and release of NO as the mechanism of dilation after stimulation of endothelial NPY receptors. Finally, the authors wanted to provide as much insight as possible as to which NPY receptor subtypes were involved with the NPY-mediated constriction and the NPY-mediated dilation (if any).
MATERIALS AND METHODS
The Animal Protocol Review committee at Baylor College of Medicine approved the experimental protocol. Sixty male Long–Evans rats (270 to 400 g) were anesthetized with isoflurane and then decapitated. The brain of each rat was immediately removed and placed in cold (4°C) physiologic saline solution (PSS). With the aid of a dissecting microscope, both MCAs were carefully dissected beginning at the circle of Willis and extending 5 to 8 mm distally. A section of the MCA (1 to 2 mm in length) was mounted in an arteriograph (Living System, Burlington, VT, U.S.A.) as previously described (Bryan, Jr. et al., 1996; You et al., 1997). Micropipettes were inserted into both ends of each MCA and secured in place with nylon ties. Each MCA was bathed in PSS (37°C) that was equilibrated with a gas consisting of 20% O2:5% CO2 in a balance of N2 (Bryan, Jr. et al., 1995; You et al., 1997). The pH of the bath solution was approximately 7.4, P
Luminal or transmural pressure of the MCAs was maintained at 85 mm Hg by raising reservoirs connected to the micropipettes to the appropriate height above the MCAs (Bryan, Jr. et al., 1996). Luminal perfusion was adjusted to 100 μL/min by setting the two reservoirs at different heights. Pressure transducers on either side of the MCA provided a measurement of perfusion pressure across the MCA and pipettes. The vessels were magnified 600-fold with an inverted microscope coupled with a video camera and monitor. Outside diameters of the MCAs were measured directly from the video screen.
The preparation consisted of two separate compartments—the luminal perfusate and the extraluminal bath. Adding agonists to the luminal perfusate selectively stimulated receptors on the endothelium; adding the agonists to the extraluminal bath selectively stimulated receptors on the vascular smooth muscle (VSM) (Bryan, Jr. et al., 1995, 1996; You et al., 1997, 1999a,b; Johnson et al., 1998; Marrelli, 2000).
After mounting and pressurization, the MCAs developed spontaneous tone (approximately 20% of the initial diameter) over the course of 1 to 1.5 hours. Experimental protocols were not initiated until the MCA diameter was stable over a 15-minute period. Any MCA that did not develop spontaneous tone was excluded. Fewer than 5% of the MCAs were discarded because of lack of spontaneous tone.
NPY (mixed agonist), [Leu31, Pro34]-NPY (Y1 agonist), or NPY-[13-36] (Y2 agonist) was added to either the extraluminal bath or to the luminal perfusate in a cumulative manner. For the extraluminal administration, the VSM was exposed to each concentration of the agonists for 5 minutes before the concentration was increased. The endothelium was exposed to each concentration of NPY or its analogues for ~15 minutes. The steady-state change in MCA diameter was measured at each concentration. Only one concentration-response curve was conducted for each MCA to avoid the risk of tachyphylaxis.
The endothelium was removed in some experiments by passing 20 mL of air through the lumen of the MCAs 30 to 45 minutes before the addition of the agonist (You et al., 1997; Marrelli et al., 1999; You et al., 1999a). Care was taken to ensure that the transmural pressure of the artery did not exceed 85 mm Hg during this process. The absence of a dilation to luminally administered ATP (10−5 mol/L) was indicative of complete denudation (You et al., 1997). After removal of the endothelium, a dilator response to S-nitroso-N-acetylpenicillamine (SNAP), an NO donor, ensured that the VSM was intact.
In some studies, Nωnitro-L-arginine methyl ester (L-NAME; 10 μmol/L), an inhibitor of NOS, was added luminally and extraluminally 30 to 45 minutes before the addition of the agonists. In other experiments, 1 μmol/L N2 - (Diphenylacetyl)-N-[(4-hydroxyphenyl)methyl]-D-argininamide (BIBP 3226), a Y1 antagonist, was added to the extraluminal bath or to both the luminal perfusate and the extraluminal bath 30 to 45 minutes before administration of any agonist.
Drugs and solutions used
Porcine NPY, porcine [Leu31, Pro34]-NPY, porcine NPY-[13–36], L-NAME, and ATP were purchased from Sigma Chemical (St. Louis, MO, U.S.A.). BIBP 3226 was a gift from Dr. Henri N. Doods (Boehringer Ingelheim Pharma KG, Biberach an der Riss, Germany). SNAP, an NO donor, was purchased from RBI (Natick, MA, U.S.A.). Stock solutions of NPY, [Leu31, Pro34]-NPY, NPY- [13–36], and ATP were prepared in O2-free distilled water and frozen. L-NAME and BIBP 3226 were dissolved in distilled water and frozen. The PSS consisted of the following (in mmol/L, Bryan, Jr. et al., 1995, 1996): NaCl 119, KCl 4.7, NaHCO3 24, KH2PO4 1.18, MgSO4 1.17, CaCl2 1.6, EDTA 0.026, and glucose 5.5.
Statistical analysis
All data are presented as mean ± SD and the acceptable level of statistical significance was P < 0.05. For concentration-response curves, results are presented as the percent change of the “baseline diameter”. The “baseline diameter” is defined as the diameter of the MCA immediately before the addition of the NPY receptor agonists. Initially the repeated measure analysis of variance was used to evaluate the concentration-response curves; however, the data did not fit the criteria set by the test. Consequently, responses for a given agonist concentration were compared with the same concentration of agonist of other experimental groups using analysis of variance with a post hoc Dunnett's test (where appropriate). A Bonferroni correction was used because a number of comparisons were made for the different concentrations.
Individual concentration-response dilations were fitted to a hyperbolic curve (Maquardt-Levenberg algorithm) having the formula f(X) = (Emax * X) / (EC50 + X), where f(X) was the dilation of the MCA for a given concentration (X) of the agonist; Emax was the value of the maximum dilation; and EC50 was the concentration necessary to obtain one half of the maximum dilation. For comparison of calculated Emax or EC50s for the different agonists, a one-way analysis of variance with a Fisher post hoc test for multiple comparisons was used.
RESULTS
The mean diameters of MCAs upon initial pressurization and after the development of spontaneous tone were 266 ± 20 μm and 215 ± 20 μm (n = 106 arteries), respectively. The addition of 10−5 mol/L L-NAME, an NOS inhibitor, to the luminal perfusate and extraluminal bath further constricted the MCAs 17% ± 5% (n = 15) from the diameter after the development of tone. Similarly, removal of the endothelium constricted the MCAs 20% ± 6% (n = 15) after the development of tone. The complete removal of the endothelium was confirmed by an absence of dilation to luminally administered ATP (10−5 mol/L) as demonstrated by You et al. (1997). After removal of the endothelium, 10−6 and 10−5 mol/L SNAP, an NO donor, dilated MCAs 17% ± 7% and 40% ± 10%, respectively. The dilation to SNAP was indicative that the VSM was not damaged as a result of removing the endothelium. The addition of 1 μmol/L BIBP 3226, a Y1 antagonist, to either the extraluminal bath (n = 21) or to the luminal perfusate and extraluminal bath (n = 11) did not change the diameter of the MCAs.
Application of NPY and analogues to the extraluminal bath
The concentration-related responses produced by the extraluminal application of NPY (mixed agonist), [Leu31, Pro34]-NPY (Y1 agonist), or NPY-[13–36] (Y2 agonist) are shown in Fig. 1. The maximal constrictions produced by NPY and [Leu31, Pro34]-NPY were greater than that caused through NPY- [13–36]. NPY [13–36] produced weak but statistically significant constrictions (P < 0.05), beginning at a concentration of 10−11 mol/L. The constrictions at the high concentration of NPY [13–36] might have been because of nonspecific effects (Gerald et al., 1996).
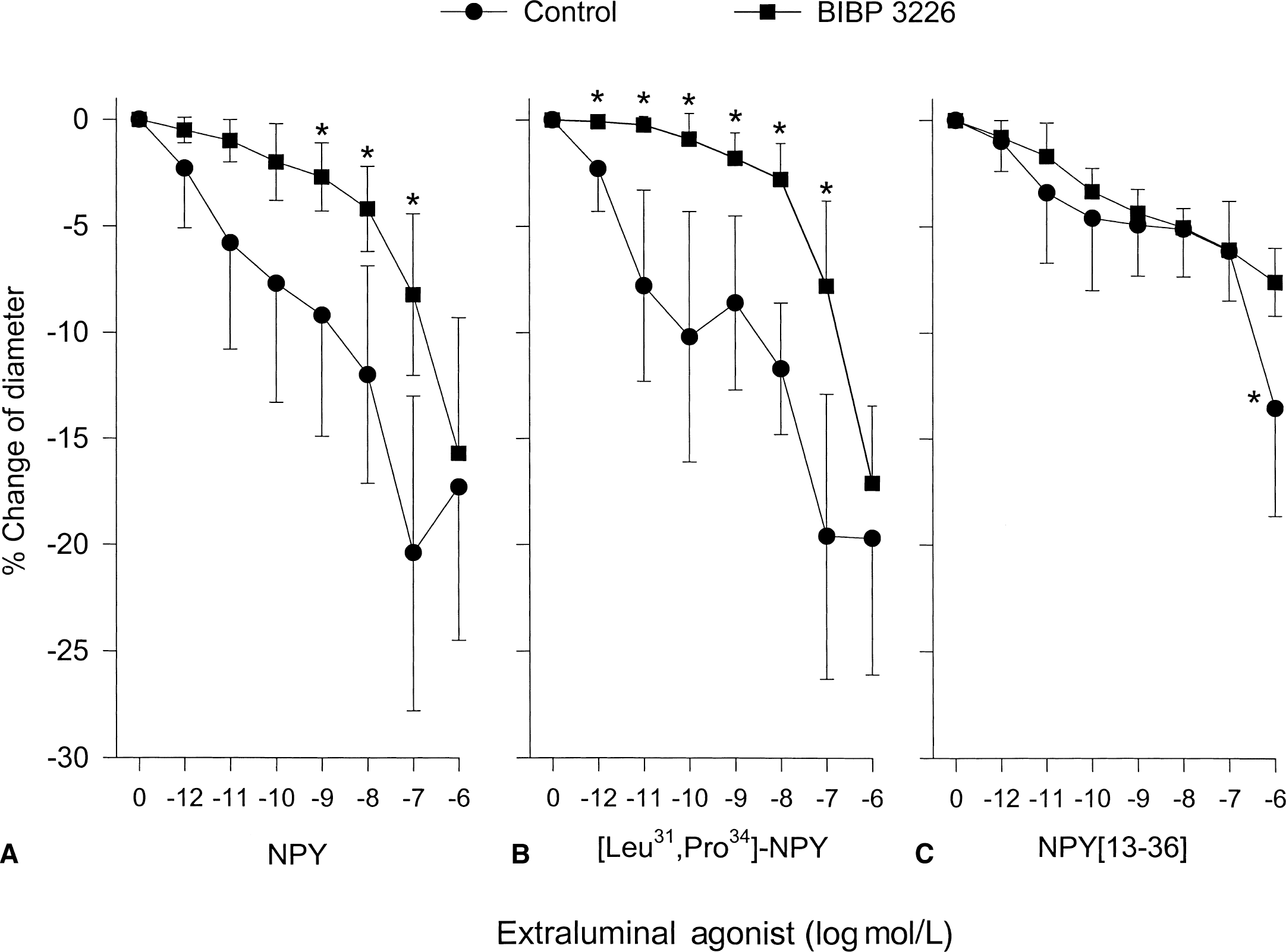
Concentration-response curves (percent change in baseline diameter) to the extraluminal administration of NPY
The antagonist for the Y1 receptor subtype, BIBP 3226, shifted the response curves for NPY and [Leu31, Pro34]-NPY to the right as would be expected by a competitive antagonist (Fig. 1A and 1B). BIBP 3226 significantly attenuated the constriction at 10−6 mol/L NPY [13–36] but had no significant effect at any of the other concentrations studied.
Application of NPY and analogues to the luminal bath
The luminal application of NPY, [Leu31, Pro34]-NPY, or NPY- [13–36], respectively, produced concentration-dependent dilations of MCAs (Fig. 2). The maximum dilations produced by each agonist and the EC50s are given in Table 1. For the purpose of demonstrating the dilator capacity of the peptides, maximum dilations produced by the luminal application of NPY, the two NPY analogues, and ATP, an agonist for P2Y2 receptors (You et al., 1997), were compared. The maximum dilation elicited by NPY or its 2 analogues was approximately 40% of the dilation produced by 10−5 mol/L ATP (P < 0.01) (data not shown).
Dilation induced by NPY and its analogues in rat middle cerebral arteries
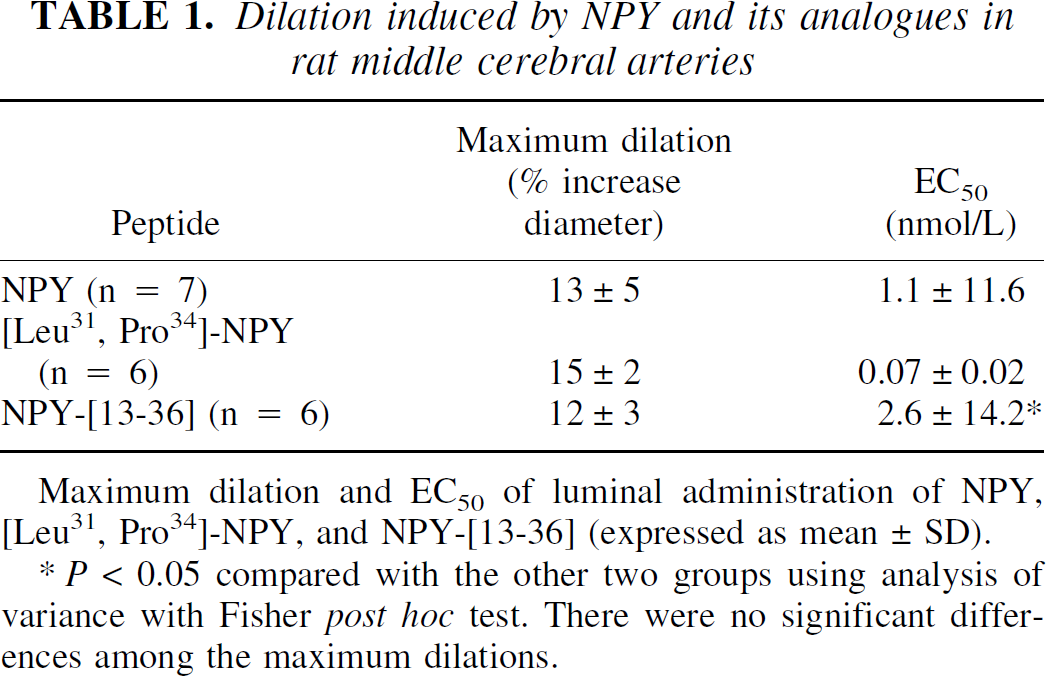
Maximum dilation and EC50 of luminal administration of NPY, [Leu31, Pro34]-NPY, and NPY-[13–36] (expressed as mean±SD).
P < 0.05 compared with the other two groups using analysis of variance with Fisher post hoc test. There were no significant differences among the maximum dilations.
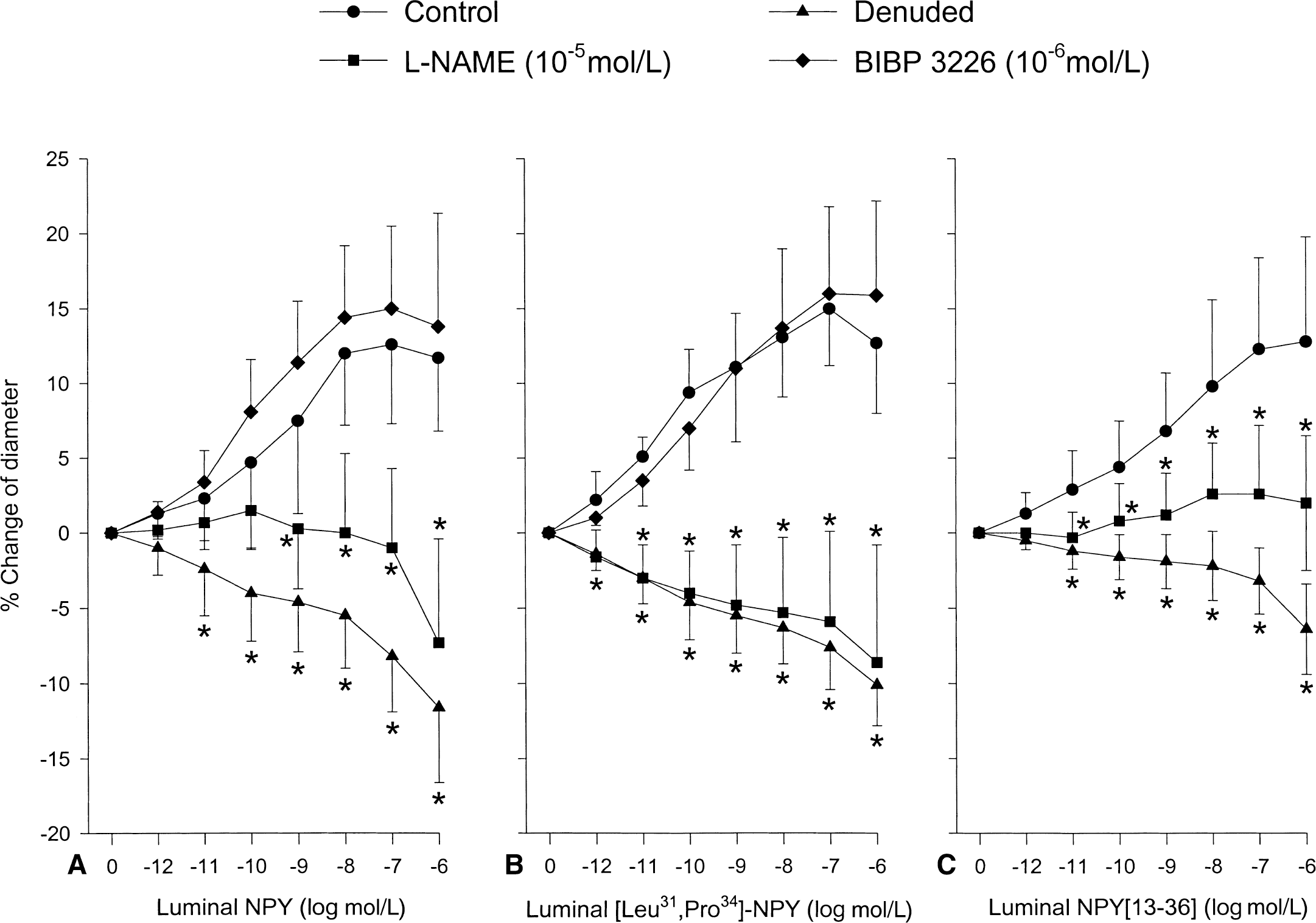
The dilations produced by NPY, [Leu31, Pro34]-NPY, or NPY-[13–36] were abolished by removal of the endothelium or by inhibition of NOS with L-NAME 10−5 mol/L (Fig. 2). After treatment with L-NAME, MCAs dilated 25% ± 11% and 55% ± 10% (n = 6) to 10−6 mol/L and 10−5 mol/L SNAP, respectively (data not shown). BIBP 3226, anY1 antagonist, had no effect on the dilations elicited by the luminal application of NPY or [Leu31, Pro34]-NPY.
DISCUSSION
The current study demonstrated that NPY could dually regulate vascular tone in rat MCAs. Stimulation of Y1 or Y2 receptors on the VSM constricted the rat MCA. Stimulation of an NPY receptor on the endothelia, other than the Y1 subtype, dilated the MCAs by a mechanism that involved NO. The endothelial-mediated dilations elicited by NPY occurred at concentrations considered physiologic for the rat (Zukowska-Grojec and Wahlested, 1993). The major contribution of this study was the establishment of the dilator role of endothelial NPY receptors in the cerebral circulation.
Method for studying isolated middle cerebral arteries
An important aspect of the authors' experimental design was the separate luminal (endothelial side) and extraluminal (VSM side) compartments of the isolated vessel preparation. The authors took advantage of this aspect to selectively stimulate receptors on the VSM or the endothelium (Bryan, Jr. et al., 1995; You et al., 1997; Johnson et al., 1998; Marrelli, 2000). Although the authors cannot be assured that one cell type was exclusively affected independent of the other, the differences in responses with luminal versus extraluminal applications of polar drugs and agonists indicated that a high degree of selectivity could be achieved (Bryan, Jr. et al., 1995; You et al., 1997; Johnson et al., 1998; Marrelli, 2000). Furthermore, the tight junctions of the endothelium appeared to completely confine many polar compounds to the lumen of the vessel (Johnson et al., 1998). Thus, the authors were reasonably assured that the luminal application of the polar NPY agonists used in this study primarily, if not exclusively, stimulated NPY receptors on the luminal surface of the endothelium. The extraluminal application of NPY (or analogues) primarily stimulated NPY receptors on the VSM; however, the agonists should also have access to the extraluminal side of the endothelium.
Problems with identification of NPY receptor subtypes on vascular smooth muscle and endothelium
The NPY receptors have been divided into six subtypes, classified as Y1 through Y6 (Michel et al., 1998). All but the Y3 subtype have been cloned and have met the necessary criteria for full receptor status (Balasubramaniam, 1997; Michel et al., 1998). Evidence for the Y3 receptor is circumstantial at the current time and, thus, is considered only as a putative subtype (Michel et al., 1998).
To the authors' knowledge, only Y1 and Y2 receptor subtypes have been reported on the VSM and endothelium (Fuhlendorff et al., 1990; Wahlestedt et al., 1990; Grundemar et al., 1992; McAuley and Westfall, 1992; Wahlestedt and Reis, 1993; Tessel et al., 1993; Oellerich and Malik, 1993; Sanabria and Silva, 1994; Palea et al., 1995; Jackerott and Larsson, 1997; Kotecha, 1998; Zukowska-Grojec et al., 1998). However, these studies have not ruled out the possibility of other receptor subtypes in vascular tissues and, therefore, it is necessary to consider this possibility when interpreting the results of the current study.
One problem the authors faced was the fact that suitable pharmacologic tools were not available to distinguish the different NPY receptor subtypes (Michel et al., 1998). Of all the subtypes, Y1 has been most studied and has a selective antagonist (Doods et al., 1995; Michel et al., 1998). The authors' conclusions regarding receptor subtypes, other than Y1, are somewhat tentative until more specific pharmacologic tools are developed. They realized at the onset of this study that conclusively determining the receptor subtypes might be impractical. Therefore, their goal has been to provide as much insight into the subtypes as possible given the current constraints.
Stimulation of Y1 or Y2 receptors on the vascular smooth muscle
It has been known over the past 15 years that NPY constricts vessels, potentiates constriction of other agonists, and inhibits relaxation of vessels in the peripheral and central circulation (Edvinsson et al., 1983, 1984, 1987; Edvinsson, 1985; Wahlestedt et al., 1990; Lundberg et al., 1990; Prieto et al., 1995; Abounader et al., 1995; Balasubramaniam, 1997; Michel et al., 1998; Prieto et al., 1998). The constriction of the rat MCA by NPY and its analogues is, therefore, not surprising. However, the important aspect of this study is not the constriction but rather the dual regulation of vascular tone by NPY.
A major component of the constriction elicited by NPY in the rat MCA involved the Y1 receptor subtype. This conclusion is based on the fact that the Y1 agonist, [Leu31, Pro34]-NPY, constricted the MCAs (Fig. 1B), and that the constriction-response curves produced by both NPY and [Leu31, Pro34]-NPY were shifted 100- to 1000-fold to the right by the selective Y1 antagonist, BIBP 3226 (Fig. 1A and 1B). Thus, it appears that Y1 receptors are located on the VSM and stimulation of these receptors constricts the rat MCA. The corroboration between the Y1 agonist, [Leu31, Pro34]-NPY, and selective Y1 antagonist, BIBP 3226, provides strong evidence for this conclusion. The current results agree with previous studies using pharmacologic criteria to define NPY Y1 receptor (Abounader et al., 1995; Nilsson et al., 1996a). Further support for this conclusion comes from the identification of mRNA for NPY Y1 receptors in vessels from human brain (Nilsson et al., 1996a).
In addition, it appears that stimulation of the Y2 receptor subtype can also constrict the rat MCA, but to a lesser degree than stimulation of the Y1 subtype (Fig. 1). This conclusion is based on the fact that NPY [13–36] constricts the MCAs with or without inhibition of the Y1 receptors with BIBP 3226 (Fig. 1C). It has been reported that 10−6 M NPY [13–36] can act receptors other than Y2 (Gerald et al., 1996). The conclusion that Y2 receptor mediated vasoconstriction would be substantially strengthened by confirmation with a selective Y2 antagonist. Nevertheless, there appears to be both constrictor Y1 and Y2 receptors on the VSM of the rat MCA.
Stimulation of an NPY receptor on the endothelium
Previous studies provided evidence that, in addition to constricting vessels, NPY may also dilate vessels through the synthesis and release of NO from the endothelium (Kobari et al., 1993; You et al., 1995; Torffvit and Edvinsson, 1997). In Kobari et al. (1993), the agonist concentration at the receptor site that elicited an increase in cerebral blood flow could not be determined. In the only other study on the cerebral circulation (You et al., 1995), the concentration of NPY necessary to elicit a transient dilation of the guinea pig basilar artery was 10−6 mol/L, a concentration considered to be at least 100-fold greater than that occurring in the guinea pig in vivo (Zukowska-Grojec and Wahlested, 1993). Concentrations of 10−7 mol/L and less did not produce any dilation possibly because of limited access of NPY to the endothelium in the vascular ring preparation, or constrictor responses that overshadowed the dilation when the agonist has access to receptors on the VSM. With the addition of the current study, the authors can confidently state that NPY does dilate cerebral vessels and the dilations are elicited at concentrations that are considered to be physiologic. Basal concentration of NPY in plasma of the rat is approximately 0.1 nmol/L and it can increase during stressful conditions to 10 nmol/L (Zukowska-Grojec and Wahlested, 1993). In the current study, the luminal administration of NPY elicited dilations over this same concentration range (Fig. 2). Perhaps, NPY from nerve terminals elicits constriction by stimulating receptors on the VSM and NPY from endothelium, or circulating NPY elicits dilation by stimulating receptors on the endothelium.
In the current study, the luminal application of NPY, [Leu31, Pro34]-NPY, or NPY [13–36] elicited a dilation by stimulating receptors on the endothelium of the rat MCA (Fig. 2). The maximum dilations produced by NPY or its two analogues were only 40% of the dilation produced by the luminal application of ATP, a P2 receptor agonist that dilates by the release of NO and endothelium-derived hyperpolarizing factor (You et al., 1997) (see Results). The dilator effects of the NPY receptors are thus small in comparison. Perhaps a role of NPY receptors in the cerebrovascular endothelium is to potentiate dilations of other agonist in a similar manner as it does with constrictor agonists that stimulate receptors on the VSM (Edvinsson et al., 1984; Macho et al., 1989; Westfall et al., 1995; Fabi et al., 1998).
The obligate role of the endothelium in the dilations elicited by the peptides became apparent when the endothelium was removed. Dilations by the peptides, when the endothelium was intact, became a constriction after its removal (Fig. 2). Furthermore, the dilations involved NO because they were abolished after NOS inhibition by L-NAME. Thus, NO was involved with the dilation.
Finally, the current results do provide some limited insight as to the receptor subtype or subtypes responsible for the dilations produced by the luminal application of the peptides. First, it can be stated with some certainty that Y1 receptors were not involved with the dilations because BIBP 3226 did not attenuate the dilations to either NPY or the Y1 agonist, [Leu31, Pro34]-NPY. Second, because NPY-[13–36] elicited a dilation, stimulation of the Y2 receptor may have been responsible for the dilation by releasing NO. This conclusion would be substantially strengthened by confirmation with a selective Y2 antagonist. With the identification and cloning of new NPY receptor subtypes, it is now recognized that [Leu31, Pro34]-NPY and NPY-[13–36] are not considered selective Y1 or Y2 agonist as previously thought (Lundell et al., 1995; Gerald et al., 1996; Gregor et al., 1996). Thus, definitive identification of the NPY receptor subtype on rat MCAs must wait until better pharmacologic tools become available.
A picture is beginning to emerge that involves endothelium, NPY, and NPY receptors. Endothelial cells from peripheral vessels synthesize NPY (Loesch et al., 1992; Zukowska-Grojec et al., 1998) and possess NPY receptors (Sanabria and Silva, 1994; Jackerott and Larsson, 1997; Zukowska-Grojec et al., 1998). Furthermore, endothelium can produce NPY[3–36], a Y2 agonist, from NPY by a serine protease, dipeptidyl peptidase IV (Mentlein et al., 1993). The endothelium could release either NPY or NPY[3–36] to act as an autocrine or paracrine agent by stimulating NPY receptors on the endothelium. Alternatively, NPY, or related peptides, or both, in the plasma could stimulate the endothelial receptors. Regardless of the source for the agonist, stimulation of the endothelial NPY receptors could, among other functions (Prieto et al., 1995; Noll et al., 1996; Fabi et al., 1998; Zukowska-Grojec et al., 1998), dilate the vessel.
In conclusion, the authors have demonstrated in the current study that NPY and its analogues can dually regulate the vascular tone in the rat MCAs. The extraluminal administration (smooth muscle side) of NPY and its analogues constricted the MCAs. The luminal application (endothelial side) of NPY and its analogues dilated the vessels through a mechanism involving the release of NO.
Footnotes
Acknowledgment
The authors are grateful to Dr. H. N. Doods for providing BIBP 3226.
