Abstract
The effectiveness of angioplasty and stenting in intracranial atherosclerotic diseases is controversial due to high rates of delayed restenosis and hemorrhage compared with extracranial arteries. However, the mechanisms underlying these differences are still unclear, because their pathophysiology is yet to be examined. To address this issue, we established a novel vascular injury model in the intracranial internal carotid arteries (IICAs) in mice, and analyzed the remodeling process in comparison to that of the femoral arteries (FAs). In IICAs, neointimal hyperplasia was observed from day 14 and grew until day 56. Although smooth muscle cells (SMCs) emerged in the neointima from day 28, SMCs in the injured media were continuously lost with eventual extinction of the media. Re-endothelialization was started from day 7 and completed on day 28. Accumulation of macrophages was continued in the adventitia until day 56. Compared with FAs, the following points are unique in IICAs: (1) delayed continuous formation of neointima; (2) accumulation of macrophages in the media on day 14;(3) continuous loss of SMCs in the media followed by extinction of the media itself; and (4) continuously growing adventitia. These pathophysiologic differences might be associated with unfavorable outcomes in percutaneous transluminal angioplasty and stenting in intracranial arteries.
INTRODUCTION
Although angioplasty and stenting have emerged as promising approaches to treating intracranial atherosclerotic disease, the widespread application of these techniques has been set back due to several major complications. Delayed restenosis is one such complication, with the Stenting in Symptomatic Atherosclerotic Lesions of Vertebral and Intracranial Arteries (SSYLVIA) trial showing a 32.4% delayed restenosis rate with bare-metal stent placement. 1 Recent meta-analysis also shows that high incidences of restenosis appear to be a major drawback of intracranial stenting, especially when compared with the relatively high durability of angioplasty and stenting procedures in the extracranial arteries. 2 Self-expandable stents were reported to induce restenosis between 3 and 6 months after injury, whereas balloon-mounted stents caused more delayed restenosis. 2 To overcome this restenosis, drug-eluting stents (DES), such as Taxus (Boston Scientific, Natick, MA, USA) and Cypher (Cordis Corp, Bridgewater, NJ, USA), have been examined, but their efficacy is still controversial. For example, Gupta et al. 3 reported that the restenosis rate at 4 months after injury was 5%, whereas Fields et al. 4 reported 38% at 9 months after injury. 4 Another important complication is the high rate of brain hemorrhage within 30 days of stenting according to the results of the recent Stenting and Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Arterial Stenosis (SAMMPRIS) 5 study, although there were some suggestions of technical limitations in this study. 6
These clinical data raise severe questions about whether the remodeling processes after injury in the intracranial arteries are different from those in the extracranial arteries, where angioplasty and stenting cause lower rates of restenosis and hemorrhage. 7 It is also unclear whether immunosuppressants, such as rapamycin, can prevent neointimal formation in the intracranial arteries. Because the structure of the intracranial arteries is quite different from that of the extracranial arteries, specifically concerning the paucity of elastic fibers in the media, the reduced adventitial tissue, the lack of an external elastic lamina,8,9 and the existence of surrounding cerebrospinal fluid, the intracranial arteries seem to be more sensitive to injuries evoked by percutaneous transluminal angioplasty and stenting than the extracranial arteries. However, there has been no prior study comparing the remodeling process of injured arteries between the extracranial and the intracranial arteries.
To address these questions, we developed a novel vascular injury model in the intracranial internal carotid arteries (IICAs) in mice, and we compared the temporal profile of pathology between IICAs and femoral arteries (FAs). Additionally, we examined the effectiveness of rapamycin in preventing IICA neointimal formation, as the intraperitoneal injection of rapamycin has been shown to prevent neointimal formation in FAs in mice. 10
MATERIALS AND METHODS
Surgical Procedures
All experiments were approved by the Institutional Animal Care and Use Committee of Osaka University and conducted in accordance with the Osaka University Guidelines, which are based on the National Institutes of Health's Guide for the Care and Use of Laboratory Animals, and with the ARRIVE guidelines. Mice were provided with a standard diet and tap water ad libitum. Male 6- to 8-week-old FVB/N mice (CLEA Japan, Tokyo, Japan) were anesthetized with isoflurane (1.4%). Cerebral blood flow (CBF) was measured using a laser Doppler flowmeter (Unique Acquisition software; Unique Medical, Tokyo, Japan) in the right MCA territory (Supplementary Figure 1A). A 5-0 surgical suture, 20 mm long, was coated with epoxide resin solution for a length of 6 mm and 300 μm in diameter from the tip. The suture was inserted from the right external carotid artery and advanced into the IICA until its terminal bifurcation, whereupon CBF was reduced more than 60% from the baseline (Supplementary Figure 1B and 1E). The suture remained in place for 5 minutes to denude and dilate the arteries and was then withdrawn until the CBF recovered. The suture was then advanced again until CBF was reduced. This procedure was repeated 10 times to scratch the arteries. The suture was then withdrawn and CBF was completely recovered (Supplementary Figure 1E). The straight segment of IICAs but not the bifurcation of IICAs was observed in the present study (gray area in Supplementary Figure 1C). In preliminary studies, we examined several sizes, placements, and durations of placements of sutures to find the optimal number of scratches to induce neointimal hyperplasia on day 56 (data not shown). Rectal temperatures were kept at 37.0 ± 0.5 °C during surgery and during the recovery period until the animals regained consciousness.
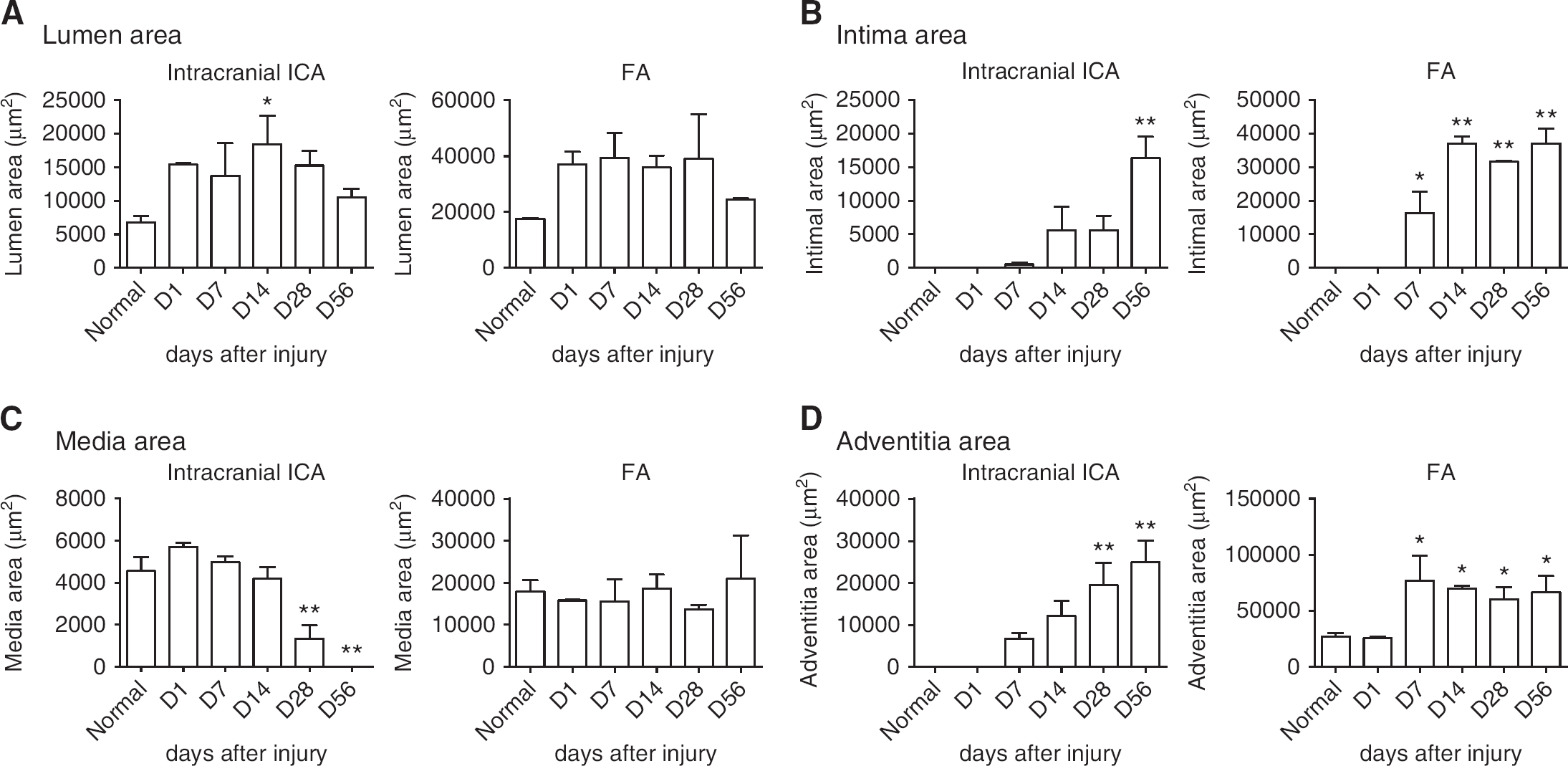
Quantitative analysis of vascular remodeling after injury. Size was calculated as areas in lumen (
For comparison, some 6- to 8-week-old male FVB/N mice were exposed to FA injury as described before. 11 The only differing elements were as follows: a straight spring wire (0.38mm in diameter, No. C-SF-15-15, COOK, Bloomington, Indiana, USA) was inserted into the FAs for more than 5 mm toward the iliac artery, the wire was left in place for 1 minute to denude and dilate the artery, and the wire was then removed to allow blood flow to recover. 11 The straight segment of FAs but not the bifurcation of FAs was examined (Supplementary Figure 1D).
Administration of Rapamycin
In accordance with a previous study showing the effectiveness of intraperitoneal injection of rapamycin in inhibiting the neointimal formation in FAs of mice, 10 intraperitoneal injection of rapamycin (4 mg/kg per day in 0.33% DMSO/0.2% sodium carboxymethyl cellulose per 0.25% polysorbate-80) was started immediately after injury and continued once a day for 56 days.
Histological Analysis
Post operation, mice were euthanized at 3 hours, 1 day, 3 days, 7 days, 14 days, 28 days, and 56 days after the injury. The mice were perfused with 4% paraformaldehyde, followed by post fixation in 4% paraformaldehyde. The brain and FAs were embedded in paraffin. For morphologic analysis, cross-sections (6 μm) including the straight portion of IICAs or FAs (gray area in Supplementary Figure 1C and 1D) were prepared for either HE staining or EVG staining. Images were then digitized by microscope (FSX-100, Olympus, Tokyo, Japan). To quantify each arterial component area, the acquired images were imported into ImageJ (version 1.42q: NIH, Bethesda, MD, USA), and the number of pixels was counted. Each area was defined as follows: the lumen area was defined as the area within the intima; the intimal area was defined as the combined area of endothelial cells, internal elastic lamina, and neointima; the medial area was defined as the area between the internal elastic lamina and the adventitia.
For terminal deoxynucleotidyl transferase dUTP nick-end labeling staining, the cross-sections (6 μm) were permeabilized with 20g/mL proteinase K for 15 min. Terminal deoxynucleotidyl transferase enzyme and fluorescein-conjugated dUTP (Roche Molecular Biochemicals, in situ Death Detection kit, Mannheim, Germany) were added to the tissue sections according to the manufacturer's specifications. Nuclei were counterstained with 4-6-diamidino-2-phenylindole (DAPI; VECTASHIELD Mounting Medium with DAPI, Vector Laboratories, Burlingame, CA, USA). For immunostaining, sections were incubated in anti-CD31 antibody (1:200, Spring Bioscience, Fremont, CA, USA), anti-SMA antibody (1:1000, Sigma-Aldrich, Saint Louis, MO, USA), or anti-F4/80 antibody (1:1000, Abcam, Cambridge, MA, UK). The sections were examined with a Nikon A1 confocal scanning laser microscope, and the images were analyzed using NIS Elements software (Nikon, Tokyo, Japan). All parameters were set in a similar manner when the signal intensity was compared.
Statistical analysis
All values are expressed as the mean ± s.e.m. Multiple comparisons were evaluated by analysis of variance followed by Dunnett's Multiple Comparison Test in the quantitative analysis of remodeling process. Two groups were compared by unpaired t-test in the experiment of treatment with rapamycin. Differences were considered significant at P<0.05.
RESULTS
Temporal Profile of Remodeling Processes in IICAs After Injuries Intracranial internal carotid arteries were successfully dilated after injury without causing hemorrhage or disruption of the internal elastic lamina (Figure 2, Supplementary Figure 2). The lumen of IICAs was dilated to double that of a normal artery on day 1, and the endothelial cell nuclei were almost lost (Figures 1A and 2C). The nuclei in the media were also lost with the thickening of the media (Supplementary Figure 2C). On day 7, cells started to accumulate outside of the vessel wall, and the nuclei had emerged on the intraluminal side of the injured arteries (Supplementary Figure 2E). On day 14, neointimal hyperplasia began to form (Figure 2G, red arrow). On day 28, the neointima started to grow (Figure 2I), and the media was partly indistinguishable from the adventitia (Supplementary Figure 2I). On day 56, the neointima and adventitia were thickened, while the media was extinct (Figure 2K). The cells outside the vessel gradually increased until day 56, with the internal elastic lamina staying intact over the duration (Figure 2K, Supplementary Figure 2K).
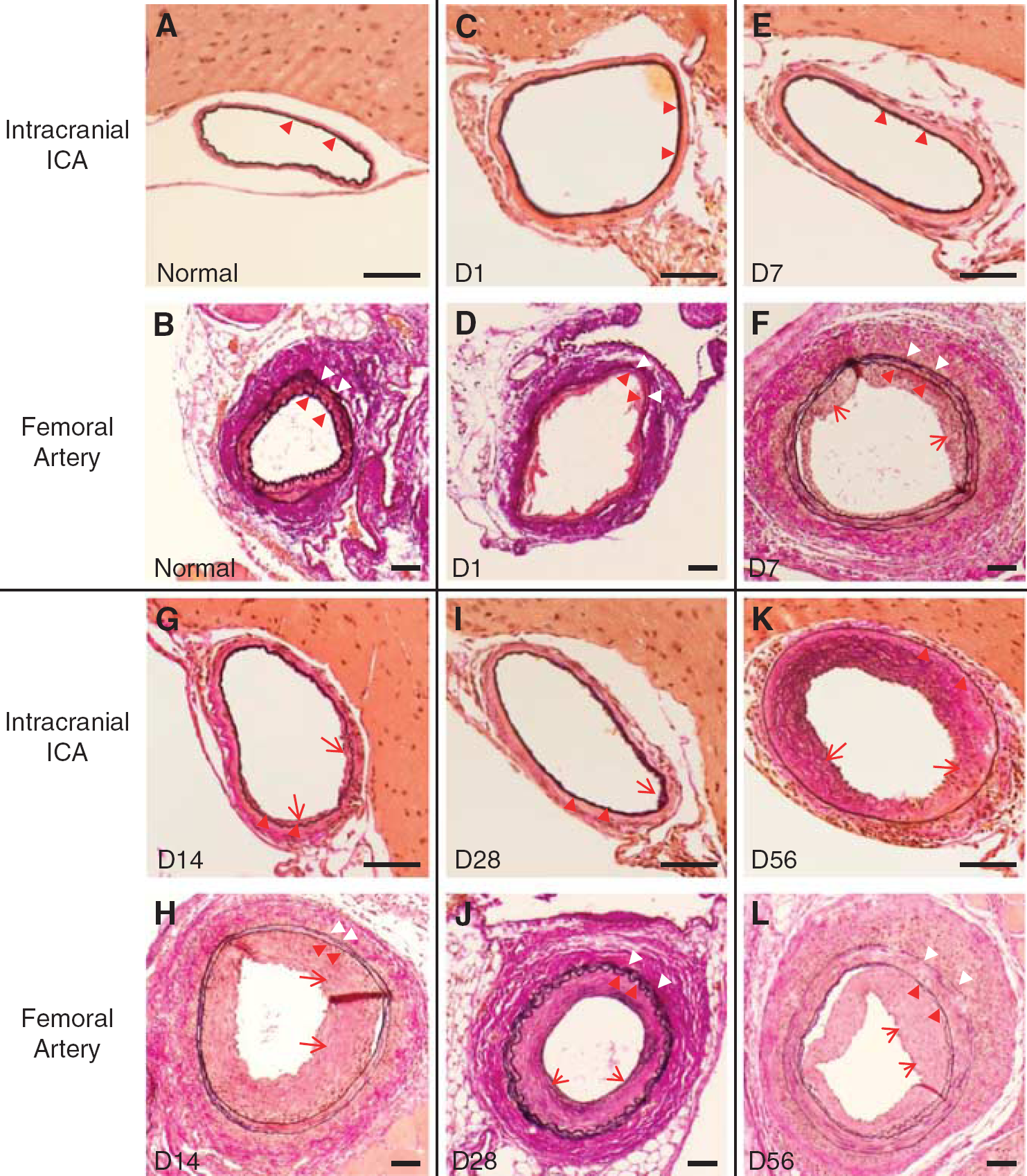
Temporal profile of the remodeling process in intracranial internal carotid arteries (IICAs) and femoral arteries (FAs). Images of Elastic-Van Gieson staining in IICAs (
Quantitative analysis showed the tendency that the size of the lumen in the injured arteries increased at 1 day after injury and remained so until day 28 although a statistical difference was observed just on day 14 (Figure 1A). After that, the lumen gradually decreased until day 56. The neointima grew between days 28 and 56 (Figure 1B). The media was gradually decreased and was completely lost on day 56 (Figure 1C). The adventitia gradually grew for the entire duration (Figure 1D).
Immunohistochemical analysis showed that αSMA-positive smooth muscle cells (SMCs) in the media were lost on day 1 and remained absent until day 56 (Figure 3). Because TUNEL-positive cells were observed in the media at 3 hours and on day 1 (Supplementary Figure 3), the loss of SMCs was partly due to apoptosis. In the neointima, αSMA-positive cells emerged after day 28 (Figure 3C). CD31-positive endothelial cells were completely absent at 1 day after injury (Figure 4A), but were partly observed at day 7 and 14 (Figures 4B and 4C) and had completely recovered by day 28 (Figure 4D). F4/80-positive macrophages were mainly observed in the adventitia from day 7 and continued to be observed until day 56 (Figure 5). Few F4/80-positive macrophages were observed in the neointima (Figure 5C). F4/80-positive macrophages infiltrated into the media on day 14 (Figure 5B arrows) but were absent on day 7 (Figure 5A).
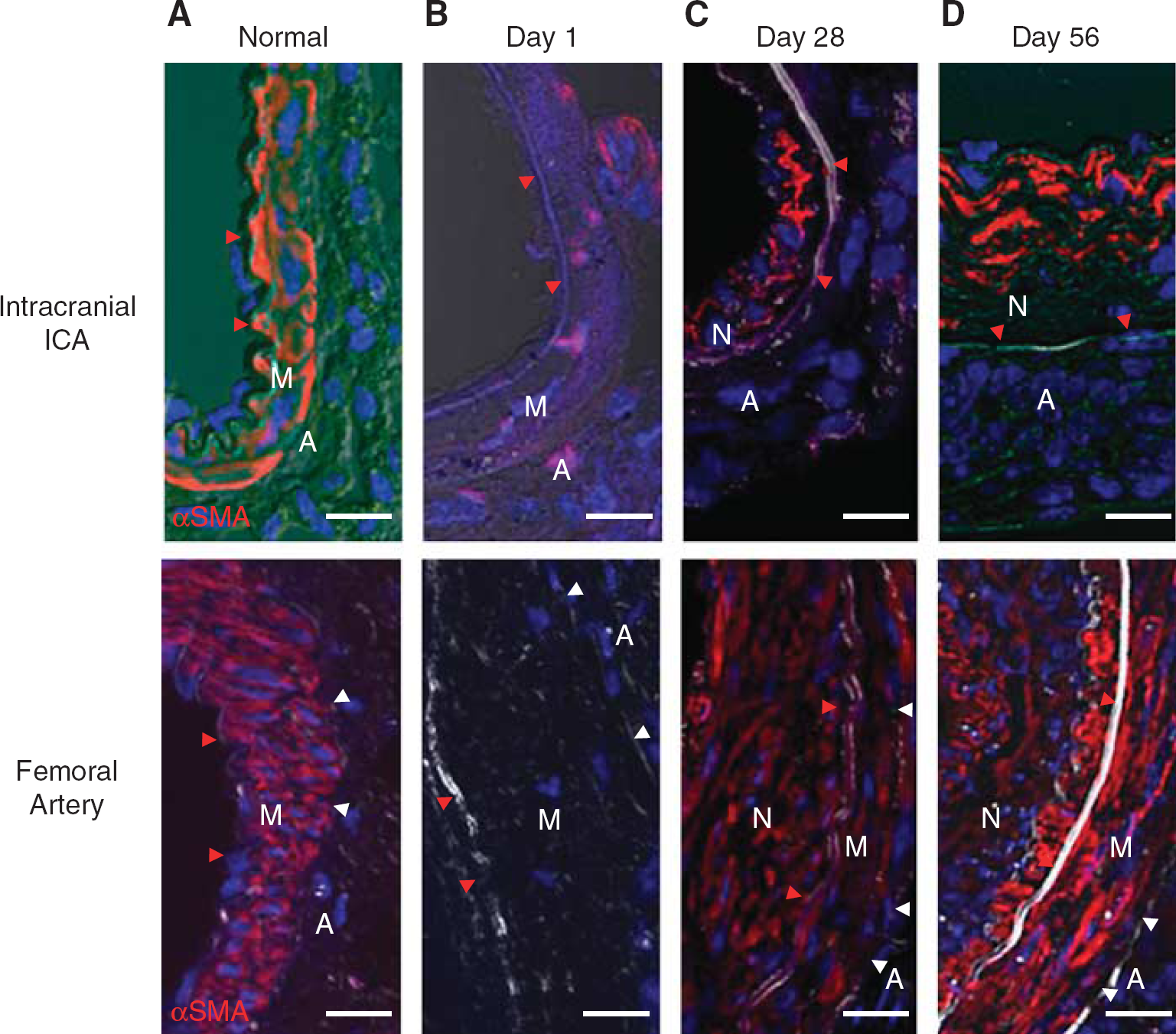
αSMA-positive smooth muscle cells (SMCs) after injury. αSMA-positive SMCs in the media were lost on day 1 in both arteries (
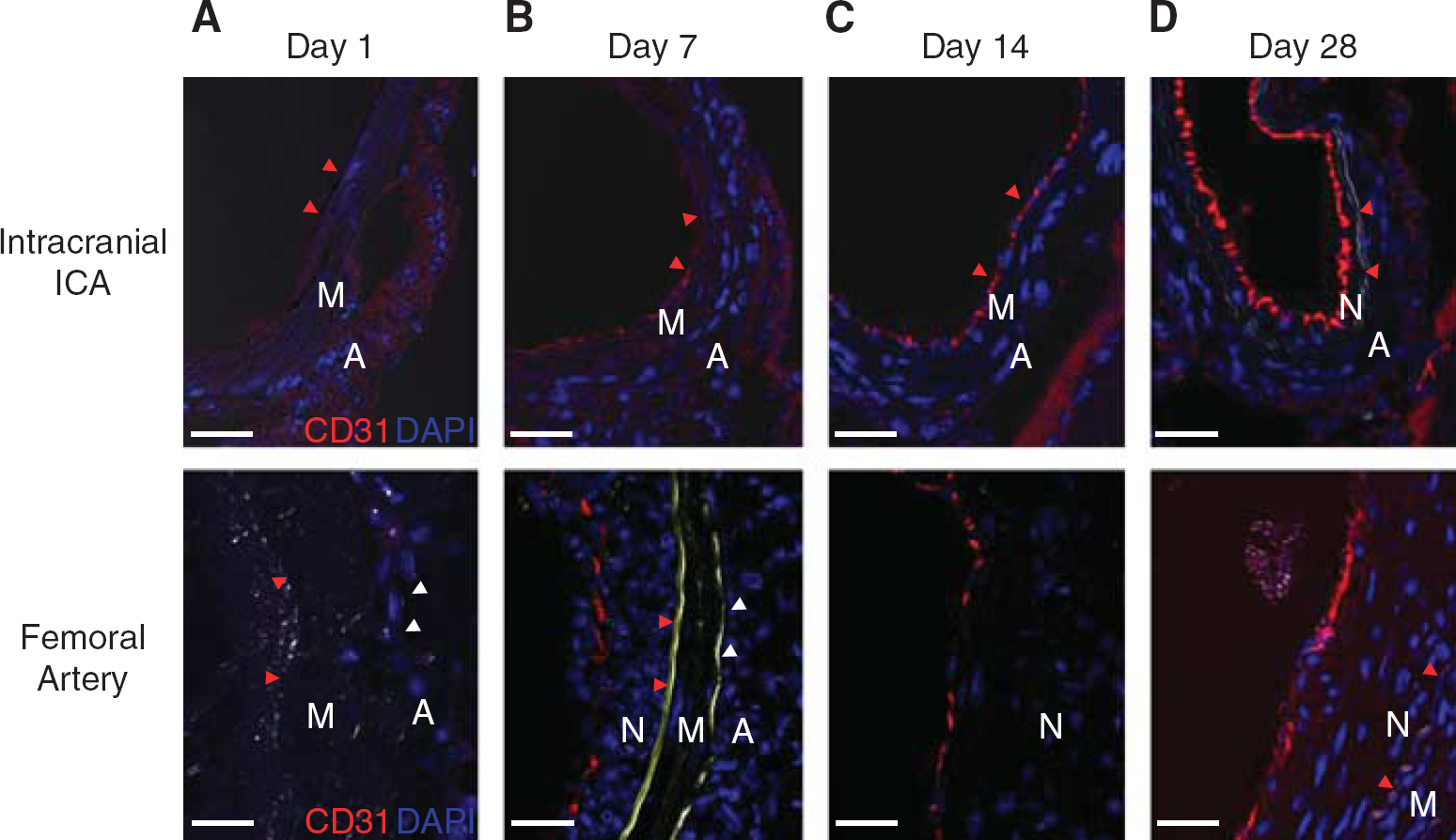
Re-endothelialization after injury. Immunohistochemistry for CD31 showed that endothelia were completely removed on day 1 (
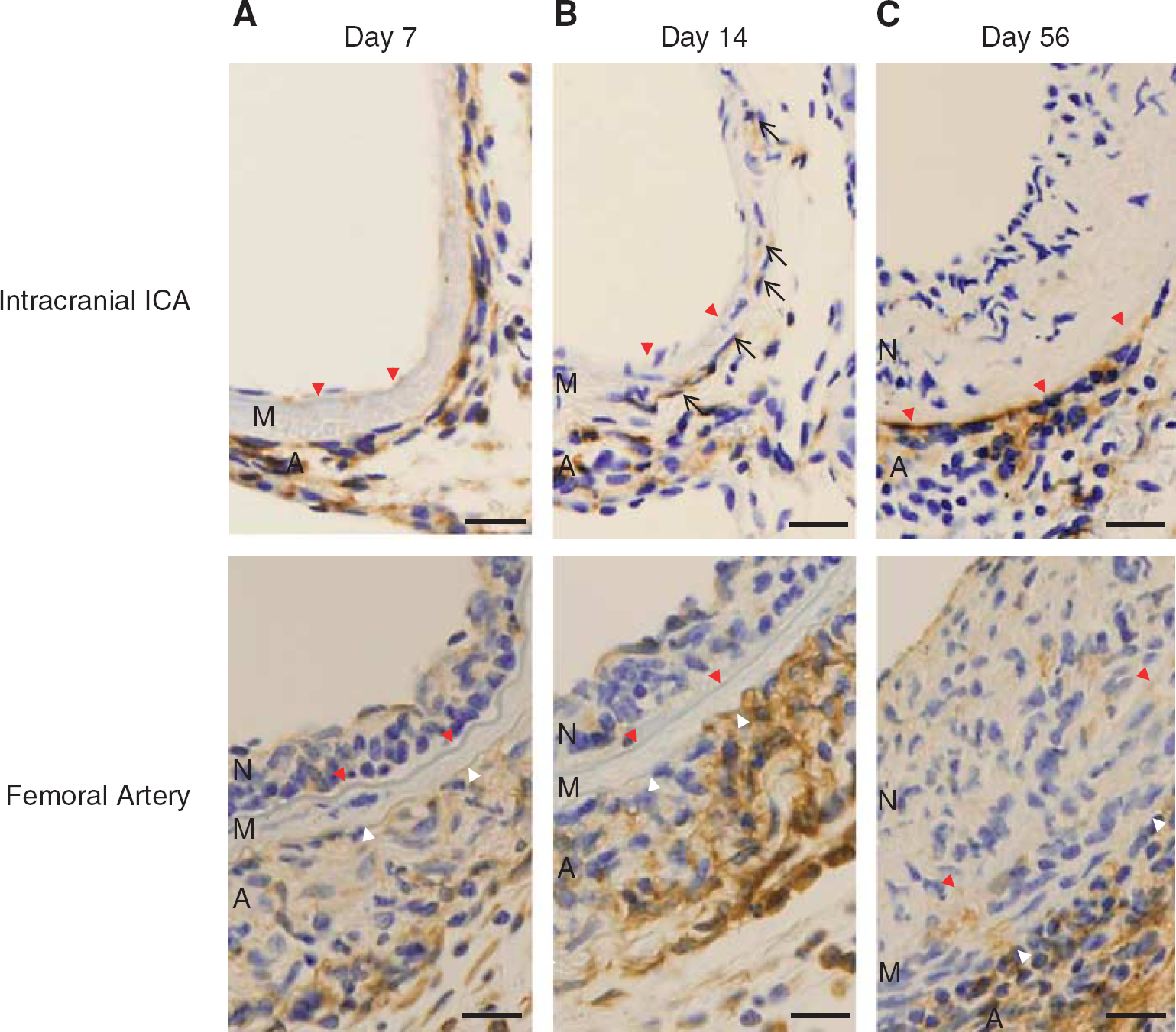
Infiltration of F4/80-positive macrophages after injury. In femoral arteries, F4/80-positive macrophages were observed in the neointima and adventitia (
To assess the influences of injury on the expression of inflammatory cytokines in CSF, the IL-6 level was examined in CSF collected from the cisterna magna on days 1, 3, 7, 14, 28, and 56 after the injury. IL-6 was not increased at these time points (data not shown), suggesting that a widespread inflammatory response in the central nervous system was not evoked in this model.
Comparison of Remodeling Process between Intracranial Internal Carotid Arteries and Femoral Arteries
To compare the process of remodeling after injury between intracranial and extracranial arteries, we selected the FA for comparison, because the technique for injury of the FA is well established 11 and demonstrated to be responsive to intraperitoneal injection of rapamycin in mice. 10 Although there are methodological differences in inducing vascular injury between the IICAs and the FAs, the ratio of lumen size in the injured artery on day 1 to that of a normal artery (2.4 ± 0.3 in IICAs and 2.1 ± 0.1 in FAs) was not significantly different (Figure 1A). Additionally, endothelial cells and αSMA-positive SMCs were completely lost on day 1 in both artery types (Figures 3B and 4A). These findings suggest that both artery types were enlarged to the same extent, and that damage to the endothelium and SMCs was similar as well.
Differing from IICAs, neointimal formation started at day 7 in FAs and grew until day 14 (Figures 1B and 2). Although αSMA-positive SMCs were completely lost in the media on day 1, they had reemerged on day 28 in the media and in the neointima (Figure 3C). Interestingly, F4/80-positive macrophages were observed in both the neointima and adventitia from day 7, although their infiltration into the media was not observed on either day 7 or 14, whereas IICAs exhibited their infiltration on day 14 (Figure 5). The adventitia in FAs grew until day 7 and remained thereafter at the same level (Figures 1D and 2). These results suggest that the remodeling process after injury might be quite different between IICAs and FAs.
Systematic Rapamycin Treatment Prevented Neointimal Formation
Finally, we examined whether rapamycin was effective in preventing neointimal formation in intracranial arteries despite the unique remodeling processes, as the ineffectiveness of DES has previously been reported in clinical studies. 4 Similar to the previous reports showing the effectiveness of rapamycin in FAs in mice, continued intraperitoneal injection of rapamycin prevented neointimal formation on day 56 (Figure 6A and 6B). Additionally, endothelialization was not affected in the group of rapamycin-treated mice (Figure 6A and 6C).
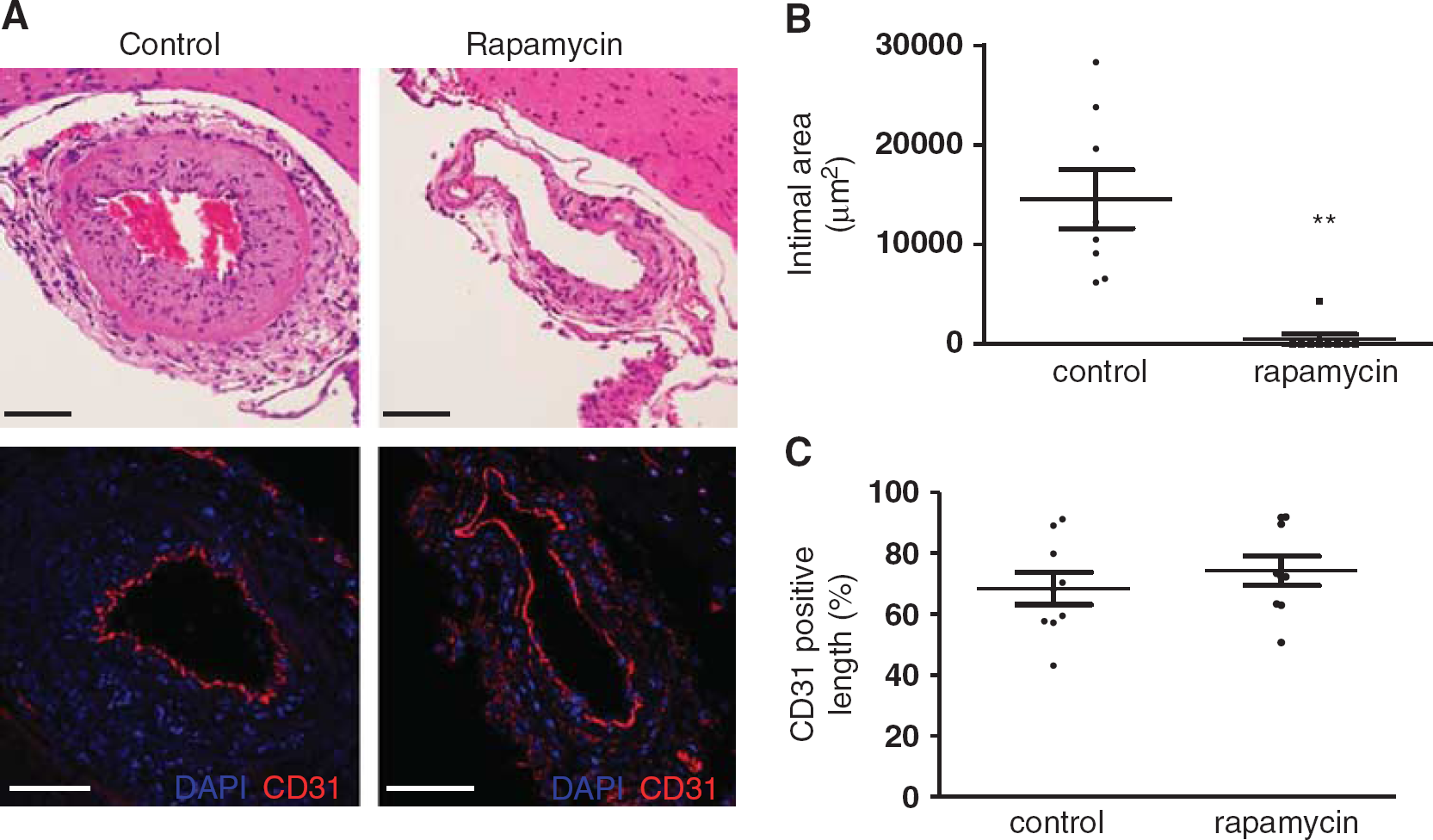
Effects of rapamycin in injured intracranial internal carotid arteries. Images of hematoxylin and eosin staining and immunohistochemistry for CD31 on day 56 (
DISCUSSION
We have successfully developed a novel vascular injury model in IICAs in mice and have shown differences in remodeling processes between IICAs and FAs. These unique remodeling processes in IICAs are characterized by (1) delayed formation of neointima;(2) gradual growth of adventitia; (3) lack of F4/80-positive macrophages in the neointimal hyperplasia; (4) absence of reemergence of αSMA-positive SMCs in the media, and (5) extinction of the media itself, whereas the temporal profile of lumen size and timing of re-endothelialization were the same in both artery types. In spite of such differences in the remodeling process, the neointimal hyperplasia was shown to be successfully inhibited in IICAs with continued rapamycin treatment. This result might suggest that the ineffectiveness of DES reported in a recent study 4 might not be due to the lack of responsiveness of intracranial arteries to rapamycin itself in prevention of neointimal formation. Considering that the neointimal formation gradually grew until day 56, longer-lasting drug releases might be needed to prevent neointimal formation in intracranial arteries. Further studies are also necessary to clarify effects on immunosuppressant-delayed re-endothelialization because DES has been reported to have negative effects, such as delayed re-endothelialization and impaired endothelial function. 12 Other drugs, such as statins, which were reported to be effective in extracranial arteries 13 should be examined in future experiments.
Although the methodological differences might explain these findings, the same degree of extension in lumen size and loss of endothelial cells and SMCs on day 1 suggest different remodeling processes between the two artery types. One of the causes might be the differences of wall-share stress, which affect progression of neointimal hyperplasia, 14 since the site of injury in FAs might be the area of laminar flow due to the lack of branches, but not laminar flow in IICAs due to the circle of Willis 15 (Supplementary Figure 1). The differences in wall shear stress between IICAs and FAs might be supported by the previous studies showing the inconsistent mean wall shear stress along the arterial tree, 16 although there was no study comparing wall shear stress between IICAs and FAs. Another important factor is that the lack of external elastic lamina in IICAs, that might cause differences in cyclic circumferential strain since it reflects elastic modules of arterial material as well as inner radius, pulse pressure, and wall thickness. 17 Because cyclic circumferential strain as well as wall shear stress modulate the endothelium-matrix interactions, which is a critical determinant of the vascular cell fate through matrix metalloproteinase, 18 the different cyclic strain in IICAs might be causative to the loss of reemergence of SMCs in media. Further studies are necessary to clarify the influences of hemodynamic differences on the remodeling process in IICAs.
Also, we speculate that the lack of an external elastic lamina might easily allow macrophages into the injured media given that F4/80-positive macrophages invaded the media on day 14 in IICAs, as shown in Figure 5B, but not in FAs. Infiltrating macrophages might express inflammatory cytokines or produce phagocyte-damaged SMCs, resulting in the inhibition of the regeneration or restoration of SMCs in the media. Further studies are necessary to clarify these mechanisms.
Another interesting finding is that αSMA-positive SMCs emerged in the neointima even though the media was diminished. Although previous studies in extracranial arteries have shown that the proliferation and migration of SMCs from the media towards the intimal layer is one of the key components of a neointima, 19 it is possible that circulating smooth muscle progenitor cells contribute to neointimal formation in intracranial arteries due to the lack of media. Another explanation is that the SMCs in neointima might be originated from the cells in adventitia since previous studies have shown that migration of myofibroblast 20 or resident pericytes progenitor or mesenchymal stem cells in adventitia 21 contribute to the intimal hyperplasia in extracranial arteries. Considering that the pace of thickening of neointima was almost paralleled by the increase in adventitia both in FAs and IICAs and that the pace of thickening of adventitia was slower in IICAs, the delayed migration of cells from adventitia to neointima might cause delayed neointimal formation in IICAs. Further studies are necessary to elucidate the origin of cells in the neointima to explore these hypotheses.
Another important finding is that the injured IICAs from days 1 to 14 exhibited incomplete endothelialization and a lack of reemergence of SMCs in the media, while lacking a thick SMC-containing neointima. Although cerebral hemorrhage was not observed in the present study, this structural fragility in the early timeline of the remodeling process might be one reason why the rate of cerebral hemorrhage within 30 days of treatment was high in intracranial arteries during a recent clinical trial. 5 The protection and regeneration of SMCs and endothelial cells might be important to prevent such complications.
The existence of CSF is a unique characteristic in intracranial arteries. In the case of subarachnoid hemorrhage, inflammatory cytokines, such as IL-6, were increased in CSF in clinical studies and are believed to play an important role in vasospasm. 22 Because the expression of inflammatory cytokines in CSF is yet to be reported in patients with restenosis after angioplasty and stenting in intracranial arteries, it raises the question whether the vascular injury in the present study caused the expression of inflammatory cytokines in CSF and affected vessels beyond the injured region. Because the present study showed that the IL-6 levels in the CSF did not increase over the whole timeline, we believe that the injury might cause limited expression of inflammatory cytokines in the injured region with no influences on other regions. To clarify these points, temporal profiles of other inflammatory cytokines should be examined both in CSF and vessel walls in future studies.
We previously reported an intracranial injury model in Wistar rats. 23 In that study, we showed that a 1-minute placement of a coated suture caused neointimal formation, which grew between days 14 and 28, whereas we did not observe the injured arteries on day 56. However, in that model, the area of neointimal formation was small, and the infiltration of macrophages in the adventitia was limited on days 3 and 7. Additionally, a preliminary trial of 1-minute placement and no scratching in the present study showed insufficient expansion of arteries and removal of endothelial cells on day 1 and no neointimal formation on day 56 (data not shown). This result might be because the size of the coated suture could not be as large as in rats due to the limited size of the carotid canal in mice. Based on these data, we prolonged the duration of suture insertion and added enough scratches to cause sufficient injury to the IICAs in mice.
FVB/N mice were selected in the present model because our previous study showed differences in injury-induced neointimal hyperplasia between inbred strains and revealed stable formation of neointimal hyperplasia in FVB/N mice compared with C57BL/6 and C3H mice. 24 One of the advantages of the establishment of a mouse model is the possible utilization of genetically modified mice whose background is not just FVB/N but C57BL/6 or C3H. Further studies are necessary to clarify that this model could be applied to other strains.
Overall, this study showed that the remodeling processes in IICAs are quite different from those in FAs in their delayed timing of neointimal formation, infiltration of macrophages into the media, and lack of recovery of SMCs in the media. In spite of such differing processes, neointimal formation was shown to be successfully prevented with continued long-term treatment with rapamycin. These findings suggest that intracranial arteries might be more fragile after injury caused by percutaneous transluminal angioplasty and stenting, and that the protection of endothelium and SMCs might be important to inhibit cerebral hemorrhage. Additionally, neointimal formation in intracranial arteries was shown to be susceptible to drug inhibition. Although further studies are necessary, this novel model might help to clarify the pathophysiology and develop new therapies for the prevention of hemorrhage and restenosis in PTA and stenting in intracranial arteries.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
