Abstract
It has been shown recently that matrix metalloproteinases (MMPs) are elevated after cerebral ischemia. In the current study, we investigated the pathophysiologic role for MMP-9 (gelatinase B, EC.3.4.24.35) in a mouse model of permanent focal cerebral ischemia, using a combination of genetic and pharmacologic approaches, Zymography and Western blot analysis demonstrated that MMP-9 protein levels were rapidly up-regulated in brain after ischemic onset. Reverse transcription polymerase chain reaction showed increased transcription of MMP-9. There were no differences in systemic hemodynamic parameters and gross cerebrovascular anatomy between wild type mice and mutant mice with a targeted knockout of the MMP-9 gene. After induction of focal ischemia, similar reductions in cerebral blood flow were obtained. In the MMP-9 knockout mice, ischemic lesion volumes were significantly reduced compared with wild type littermates in male and female mice. In normal wild type mice, the broad spectrum MMP inhibitor BB-94 (batimastat) also significantly reduced ischemic lesion size, However, BB-94 had no detectable protective effect when administered to MMP-9 knockout mice subjected to focal cerebral ischemia. These data demonstrate that MMP-9 plays a deleterious role in the development of brain injury after focal ischemia.
Matrix metalloproteinases (MMPs) form a family of at least 16 zinc-dependent endopeptidases that are collectively able to degrade, or modify, or both, almost all proteinaceous components of the extracellular matrix (ECM) (Nagase and Woessner, 1999). Substrates currently identified include collagen, fibronectin, laminin, and several types of proteoglycans, In addition, other non-ECM substrates may also be targeted, such as myelin basic protein (Chandler et al., 1995), pro-tumor necrosis factor (Gearing et al., 1994), Fas ligand (Mariani et al., 1995), transforming growth factor 1-β (TGF1-β) (Yu and Stamenkovic, 2000), and pro-fibroblast growth factor receptor 1 (Levi et al., 1996). Matrix metalloproteinases play important roles in many physiologic processes, including embryologic remodeling, wound healing, angiogenesis, bone remodeling, ovulation, and implantation (Nagase and Woessner, 1999).
Because MMPs can degrade many components of the ECM, enzyme activities are strictly regulated (Fini et al., 1998). Protein levels are regulated at the transcriptional level of expression. More importantly, however, proenzyme activation and enzyme activities are tightly regulated by tissue inhibitors of MMP (TIMP) and interactions with surrounding ECM molecules. Uncontrolled expression of MMPs can result in tissue injury and inflammation. Recently, abnormal MMP activity has been implicated in cerebral ischemia. In mouse, rat, and baboon models of cerebral ischemia, MMP-2 and MMP-9 expression were significantly increased after ischemic onset (Gasche et al., 1999; Heo et al., 1999; Mun-Bryce and Rosenberg, 1998; Rosenberg et al., 1996). Treatment with MMP inhibitors reduced the formation of vasogenic edema in a rat model of focal cerebral ischemia (Rosenberg et al., 1998), and treatment with an MMP-neutralizing antibody reduced infarct size (Romanic et al., 1998). Taken together, these studies suggest that the MMP family of proteinases participates in the pathophysiology of cerebral ischemia. However, direct evidence for the role of specific MMPs remains to be elucidated.
In the current study, we investigated the hypothesis that MMP-9 (gelatinase B, EC.3.4.24.35) may be a participant in the pathophysiologic cascade of neuronal tissue damage after focal cerebral ischemia. This hypothesis was tested by performing experiments in a mouse model of middle cerebral artery occlusion. First, we documented through zymography, Western blots, and reverse transcription polymerase chain reaction (RT-PCR) that MMP-9 was indeed up-regulated in ischemic brain. Next, we showed that ischemic lesion volumes were significantly reduced in mutant mice with a targeted knockout of the MMP-9 gene. A complementary experiment showed that the broad spectrum MMP inhibitor BB-94 (batimastat) reduced ischemic lesion volumes in normal wild type mice. However, BB-94 had no significant effect when administered to MMP-9 knockout mice. These findings support a deleterious role for MMP-9 in the pathophysiology of brain damage after focal ischemia.
MATERIALS AND METHODS
Animal model
All experiments were performed according to an institutionally approved protocol in accordance with NIH Guide for the Care and Use of Laboratory Animals. Normal male CD-1 mice were used in experiments that documented the ischemic up-regulation of MMP-9 and experiments that tested the cerebro-protective properties of the broad spectrum MMP inhibitor BB-94 (batimastat). For all other experiments, the MMP-9 knockout strain and their corresponding wild type littermates (male and female) were used. The MMP-9 knockout mice were bred from a CD-1 background (Vu et al., 1998). To inactivate the MMP-9 gene, part of exon 2 and all of intron 2 were replaced with a cassette containing the neomycin phosphotransferase cDNA driven by the phosphoglycerate kinase promoter. Homozygous null mice were viable and fertile. The major phenotypic alteration detected was a perturbed pattern of skeletal growth plate development during the first three weeks of life, which eventually remodeled, resulting in an axial skeleton of normal appearance (Vu et al., 1998). In the current study, a macroscopic examination of the knockout mice showed that the major cerebral arteries including the circle of Willis appeared normal, compared with wild type littermates and normal CD-1 mice.
The standard intraluminal middle cerebral artery occlusion method was used. Briefly, mice were anesthetized with 1~0.5% halothane in 30% oxygen and 70% nitrous oxide using a face mask. The right femoral artery was cannulated to record blood pressure and obtain arterial blood samples. Core rectal temperature was maintained at 37.5°C with a thermostat-controlled heating pad. Following a midline skin incision, the right external carotid artery was exposed, and its branches were electro-coagulated. A 7.0 nylon monofilament coated with silicon was introduced into the right internal carotid artery through the external carotid artery to occlude the origin of middle cerebral artery. Sham control animals were subjected to similar operations to expose the carotid arteries without middle cerebral artery occlusion. All animals were assessed with laser—Doppler flowmetry to confirm adequate induction of focal ischemia, and for purposes of comparison of regional cerebral blood flow deficits between experimental groups.
Treatment with matrix metalloproteinase inhibitor
BB-94 (British Biotech Pharmaceuticals, Oxford, England) was used as a broad spectrum MMP inhibitor in focal cerebral ischemia. IC50 values for enzyme inhibition of representative MMP are as follows: S nmol/L for MMP-1, 4 nmol/L for MMP-2, 20 nmol/L for MMP-3, 6 nmol/L for MMP-7, and 3 nmol/L for MMP-9 (Batimastat technical information sheet; British Biotech). Separate experiments were performed to assess the effects of BB-94 in normal CD-1 mice and MMP-9 knockout mice. In all studies, BB-94 was administered intraperitoneally as a suspension of 3 mg/mL in phosphate-buffered saline (PBS; pH 7.2) containing 0.01% Tween-80. The intraperitoneal route has led effective delivery of inhibitor to brain (Paez Pereda et al., 2000). Mice were treated twice, 30 minutes before and 3 hours after the start of the focal ischemic insult (50 mg/kg per dose). The timing of the second dose was based on the estimated 3-hour plasma half-life of BB-94 in mice (Low et al., 1996); similar doses have been shown to be effective inhibitors of MMP activity in mouse tumor models (Goss et al., 1998). Untreated controls received PBS containing 0.01 % Tween-80.
Measurement of neurologic deficits
Mice were tested and scored for neurologic deficits as follows: 0 = no detectable neurologic deficit; 1 = failure to extend left forepaw fully; 2 = spontaneous turning to the left; 3 = spontaneous circling to left; 4 = inability to move or retain normal upright posture. Assessments were made at 2 and 24 hours after middle cerebral artery occlusion.
Measurement of infarct volume
Mice were killed 24 hours after ischemic onset. Eight coronal sections per brain (1-mm-thick) were prepared and stained with 2.3.5-triphenyltetrazolium chloride (Sigma, St. Louis, MO. U.S.A.). Infarct volumes were quantified with a standard computer-assisted image analysis technique. To exclude possible confounding effects of brain swelling, an indirect method was used to calculate the lesion volumes (Lin et al., 1993).
Preparation of tissue extracts
At 1, 3, 6, 12, and 24 hours after the onset of ischemic insult, mice (n = 3 per time point) were deeply anesthetized with halothane and then transcardially perfused with ice-cold PBS (pH 7.4). Sham-operated control mice were similarly perfused at 24 hours. Brains were removed quickly and divided into ipsilateral ischemic hemispheres and contralateral nonischemic hemispheres.
Hemispheric tissue was frozen immediately in liquid nitrogen and stored at −80°C. Brain samples were homogenized in 10x volume lysis buffer (50 mmol/L Tris-HCl, pH 7.4, 150 mmol/L NaCl, 1 % Nonidet P-40, 0.1 % SDS, 0.1% deoxycholic acid), including protease inhibitors (2 μg/mL leupeptin, 2 μg/mL aprotinin, 1 mmol/L PMSF) on ice using a Teflon glass homogenizer. After centrifugation (10,000 g) for 5 minutes at 4°C, the supernatant fluid was collected. Total protein concentration of each sample was determined using the Bradford assay (Bio-Rad Laboratories, Hercules, CA, U.S.A.).
Western blot analysis
To investigate the protein expression patterns in control and ischemic brains, equal volumes (10 μL) of total protein extracts were prepared and normalized for protein concentration (3 μg/μL). Ten microliters of 2x sample buffer (126 mol/L Tris-HCl, 20% glycerol, 2% SDS, 0.005% bromophenol blue) was added to each protein extract. Each sample (30 μg/20 μL) was separated by 4% to 20% gradient Tris-Glycine SDS-PAGE and transferred to polyvinylidene difluoride membrane. The membrane was blocked with 10% nonfat dry milk in PBS containing 0.1% Tween-20 (PBS-T). MMP-9 was detected with rabbit polyclonal antibody (kind gift from Robert Senior) at a dilution rate of 1:4000. This antibody is able to recognize both latent and activated form of MMP-9 protein because it was raised against catalytic domain of mouse MMP-9 protein (Betsuyaku et al., 2000). After washing with PBS-T, the membrane was incubated with peroxidase-conjugated secondary antibody at room temperature for 1 hour. Finally, MMP-9 protein was detected by using chemical luminescence (ECL; Amersham Pharmacia Biotech, Piscataway, NJ, U.S.A.).
SDS-PAGE zymogram
Similarly prepared protein samples (as in Western blot analysis) were loaded and separated by 10% Tris-Glycine gel with 0.1% gelatin as substrate. After separation by electrophoresis, the gel was incubated in renaturing buffer (2.7% Triton X-100 in distilled water) at room temperature with gentle agitation for 30 minutes. The renaturing buffer then was decanted and replaced with developing buffer (50 mmol/L Tris Base, 40 mmol/L HCl, 200 mmol/L NaCl, 5 mmol/L CaCl2, 0.2% Brij 35). After 30 minutes equilibration by the developing buffer, the gel was incubated with fresh developing buffer at 37°C for 24 hours. After developing, the gel was stained with 0.5% Coomassie Blue R-250 for 30 minutes and then destained appropriately.
Reverse transcription polymerase chain reaction
At 12 and 24 hours after the onset of ischemic insult, mice (n = 5 per time point) were killed with a deep dose of halothane anesthesia followed with transcardial perfusion with ice-cold PBS. Sham-operated control mice were similarly perfused at 24 hours. Each brain was removed quickly and ipsilateral hemispheres were immediately frozen with liquid nitrogen. Total RNA was prepared with the RNA isolation reagent (TRIzol Reagent; Life Technologies, Gaitherburg, MD, U.S.A.). One microgram of total RNA from each time point was subjected to reverse transcription polymerase chain reaction (RT-PCR) analysis for MMP-9 mRNA. Reactions were performed using a kit from TaKaRa, Kyoto, Japan. The following conditions were adopted for the RT reaction using oligo-dT primers: 55°C for 20 minutes, 99°C for 5 minutes, 5°C for 5 minutes. The following conditions were adopted for the PCR amplification using specific primers: cDNA products of the RT reaction were denatured for 2 minutes at 94°C before 30 cycles of 94°C for 30 seconds, 60°C for 30 seconds, and 72°C for 90 seconds. Primers for MMP-9 specific amplification were as follows: (5′) primer 5′-GGACAGCGAGACACTAAAGGA-3′ and (3′) primer 5′-AAACCCCACTTCTTGTCAGC-3′ (Perides et al., 1999). Control mouse β-actin cDNA was amplified using: (5′) primer 5′TGGAATCCTGTGGCATCCATGAAAC-3′ and (3′) primer 5′-TAAAACGCAGCTCAGTAACAGTCCG-3′ (Yamamoto and Loskutoff, 1996). The RT-PCR products were separated on 1.8% agarose gel and visualized with ethidium bromide.
Statistical analysis
Quantitative data were expressed as mean ± SD. Ischemic lesion volumes in various groups were compared using analysis of variance followed by Tukey-Kramer tests. For comparing neurologic deficits between groups, the nonparametric Kruskal—Wallis tests were used followed by Mann—Whitney tests. Differences with P < 0.05 were considered statistically significant.
RESULTS
MMP-9 protein expression is up-regulated after permanent focal cerebral ischemia
In Western blot analysis, MMP-9 protein expression was detected as early as 3 hours after the start of the focal ischemic insult, and expression continuously increased up to 24 hours (Fig. 1). Only the 105 kDa latent form of MMP-9 was observed, although the antibody can recognize latent and activated forms of MMP-9 protein. When ischemic brain extracts were incubated with the chemical MMP activator p-aminophenylmercuric acetate (APMA) at 37°C for 3 hours, a clear band became visible at 97 kDa, corresponding with the expected size of activated MMP-9 protein (Fig. 1). No MMP-9 expression was seen in sham-operated brains or normal contralateral brain (Fig. 1).
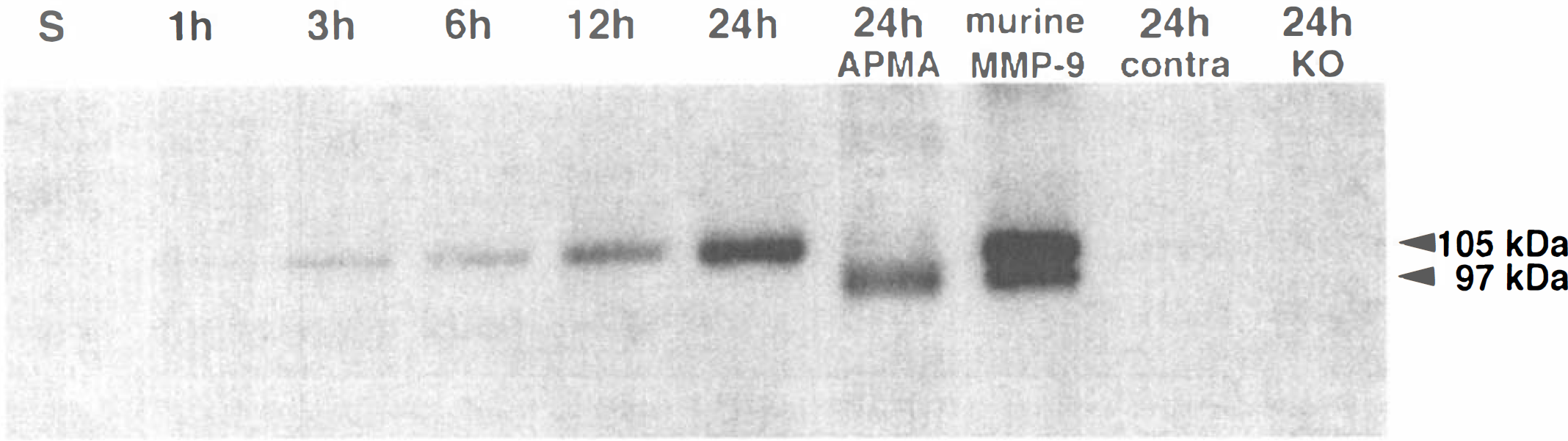
Western blots of MMP-9 demonstrate the up-regulation of protein expression after focal cerebral ischemia. MMP-9 expression was limited primarily to the 105 kDa zymogen, detected as early as 3 hours postischemia, and appeared to increase over time until the last time point measured at 24 hours. Extracts from sham-operated brain (S) did not show MMP-9 up-regulation. (24h APMA) Represents samples from ischemic brain activated by the MMP activator p-aminophenylmercuric acetate (APMA), thus converting the 105 kDa zymogen into the 97 kDa enzyme. Samples (24h contra and 24h KO) from contralateral brain and ischemic brain from MMP-9 knockout mice at 24 hours, respectively. Murine MMP-9 was used as a standard. n = 3 brains per lane were used.
Zymograms of MMP-9 activity was performed using gelatin as substrate. Increased MMP-9 levels were detected beginning as early as 1 hour postischemia, and continuing for up to 24 hours (Fig. 2). Similar to the Western blot findings, MMP-9 was predominantly expressed as the higher molecular weight latent form (105 kDa). However, at 12 to 24 hours, faint bands of activity were also observed at 97 kDa, suggesting that small amounts of activated MMP-9 were present. Incubation of ischemic brain extracts with the chemical MMP activator APMA appeared to convert all the 105 kDa protein into a band at 97 kDa, corresponding with the expected size of activated MMP-9 protein (Fig. 2). MMP-9 was not detected in sham-operated brains or contralateral nonischemic brain (Fig. 2).
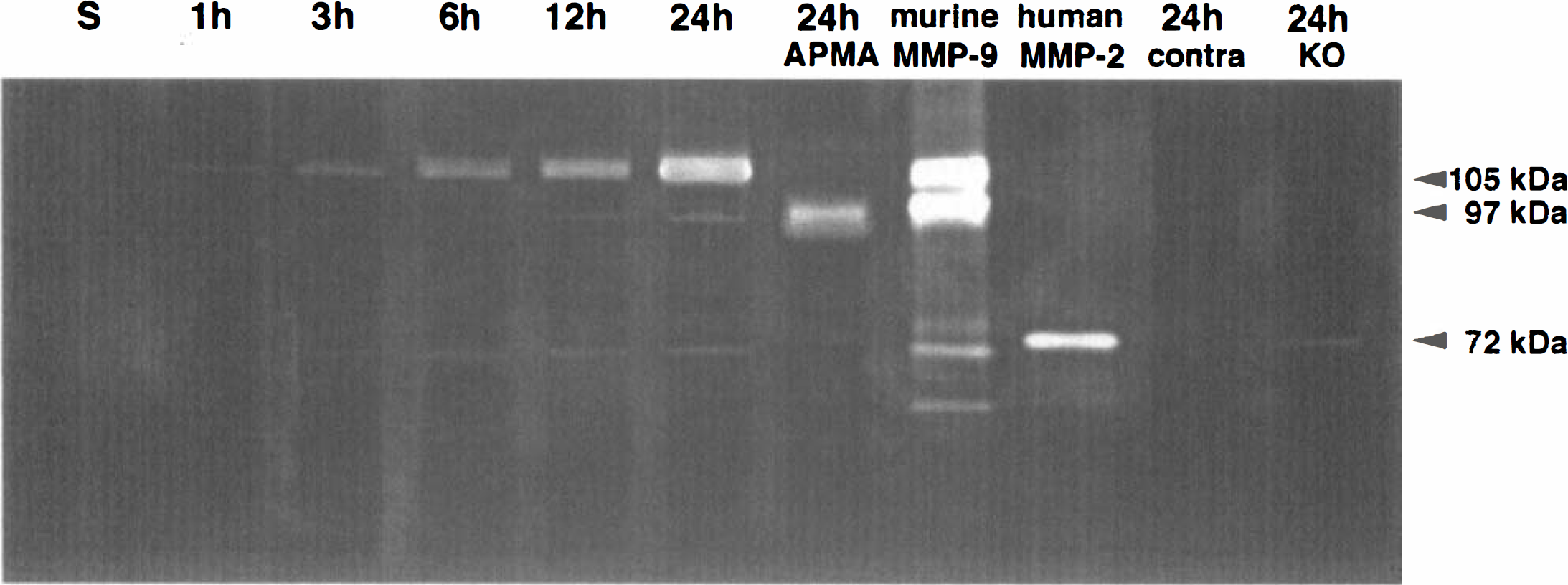
Zymograms demonstrate the up-regulation of MMP-9 after focal cerebral ischemia. Once again, generally the 105 kDa zymogen was detected, although these zymograms also showed some gelatinolytic activity at 97 kDa, corresponding with the cleaved/activated enzyme. MMP-9 was detected as early as 1 hour and continued to increase until 24 hours. Sham-operated brain (S) did not show MMP-9 up-regulation. (24h APMA) Represents samples from ischemic brain activated by the MMP activator p-aminophenylmercuric acetate, thus converting the 105 kDa zymogen into the 97 kDa enzyme. (24h contra and 24h KO) Represent samples from contralateral brain and ischemic brain, respectively, from MMP-9 knockout mice at 24 hours. Subtle gelatinolytic activity was also present close to the molecular weight for MMP-2. Note that under the nonreducing conditions used in the authors' preparations, it is well known that the 72 kDa MMP-2 appears at a lower level (approximately 65 kDa). Murine MMP-9 and human MMP-2 were used as standards. n = 3 brains per lane.
In ischemic brain samples after 12 and 24 hours, faint bands of gelatinolytic activity were detected at approximately the same level as the human MMP-2 standard. These may represent low level induction of MMP-2 after cerebral ischemia in this model; however, these levels were much lower compared with MMP-9.
Transcriptional expression of MMP-9 is increased after permanent focal cerebral ischemia
To determine whether the ischemic up-regulation of MMP-9 protein expression is caused by increased transcriptional activity, RT-PCR was used to analyze levels of MMP-9 mRNA. Low levels of MMP-9 mRNA appeared to be present in sham-operated brains (Fig. 3, top). At 12 and 24 hours after permanent focal cerebral ischemia, MMP-9 mRNA levels were increased (Fig. 3, top). No bands were detected in a negative control reaction without the presence of reverse transcriptase (Fig. 3, top), indicating that detection of the MMP-9 bands was derived from mRNA not genomic DNA. A control experiment using the same RT-PCR condition in samples from 24-hour ischemic brain showed that the amount of final RT-PCR products was proportionally related to the amount of input RNA sample within the range of 0.03 to 1 μg after 30 cycles of amplification (Fig. 3, middle). Intensities of the β-actin band for each time point were equal, demonstrating that comparable amounts of RNA were used for the RT-PCR analysis (Fig. 3, bottom).
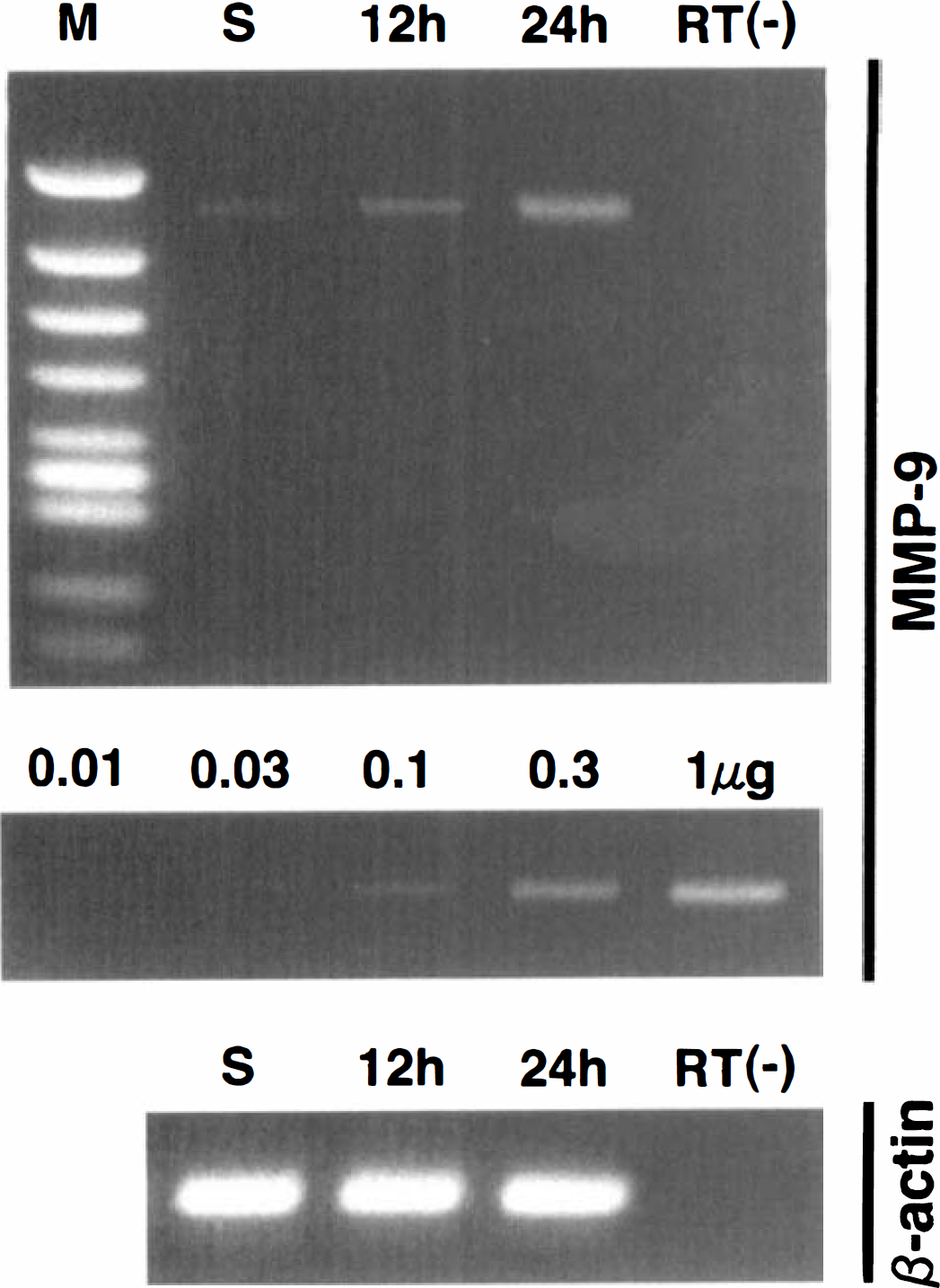
Reverse transcription polymerase chain reaction (RT-PCR) analysis of MMP-9 mRNA.
Ischemic lesion volumes are reduced in MMP-9 deficient knockout mice
There were no differences in physiologic parameters between MMP-9 knockout mice and wild type littermates (Table 1). Furthermore, there appeared to be no differences in the gross vascular anatomy of the Circle of Willis and major cerebral arteries.
Physiologic variables
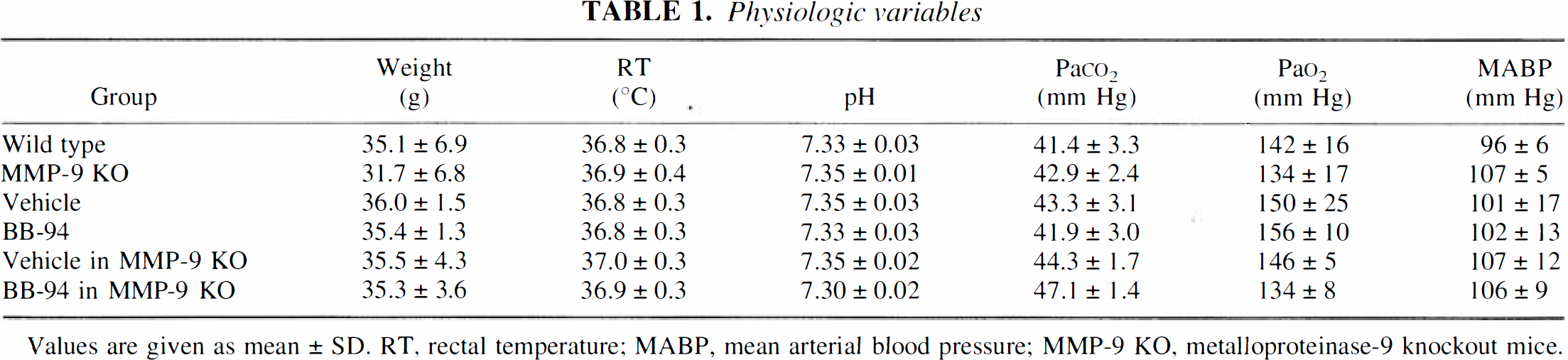
Values are given as mean ± SD. RT, rectal temperature; MABP, mean arterial blood pressure; MMP-9 KO. metalloproteinase-9 knockout mice.
Laser—Doppler flowmetry demonstrated that consistent levels of ischemia were achieved in all animals. There were no differences between MMP-9 knockout mice and wild type littermates in terms of reduction of regional cerebral blood flow (Table 2).
Regional cerebral blood flow
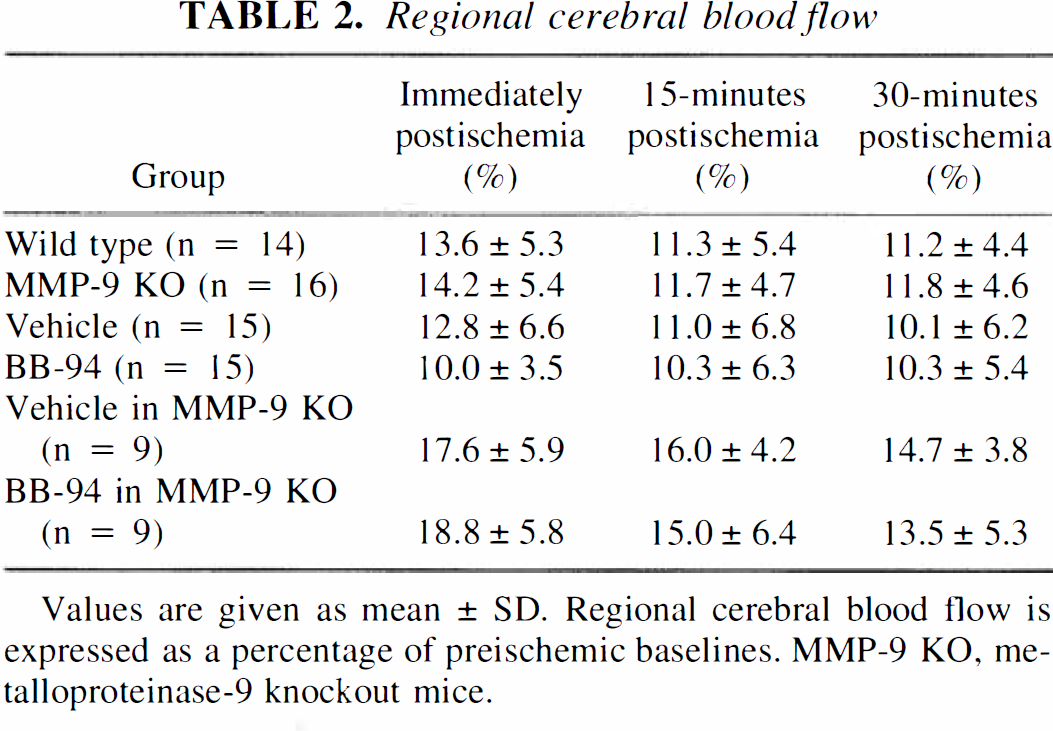
Values are given as mean ± SD. Regional cerebral blood flow is expressed as a percentage of preischemic baselines. MMP-9 KO, metalloproteinase-9 knockout mice.
MMP-9 knockout mice were more resistant to focal cerebral ischemia compared with wild type littermates. To eliminate potential confounds of sex differences, data were analyzed separately in male and female mice. In both sexes, MMP-9 deficiency resulted in significantly smaller ischemic lesion volumes after 24 hours of permanent focal cerebral ischemia (Fig. 4A to 4D). Overall, there appeared to be no differences between males and females. At 24 hours, neurologic deficits were slightly less severe in MMP-9 knockout mice compared with wild types, but the difference did not reach statistical significance (Table 3).
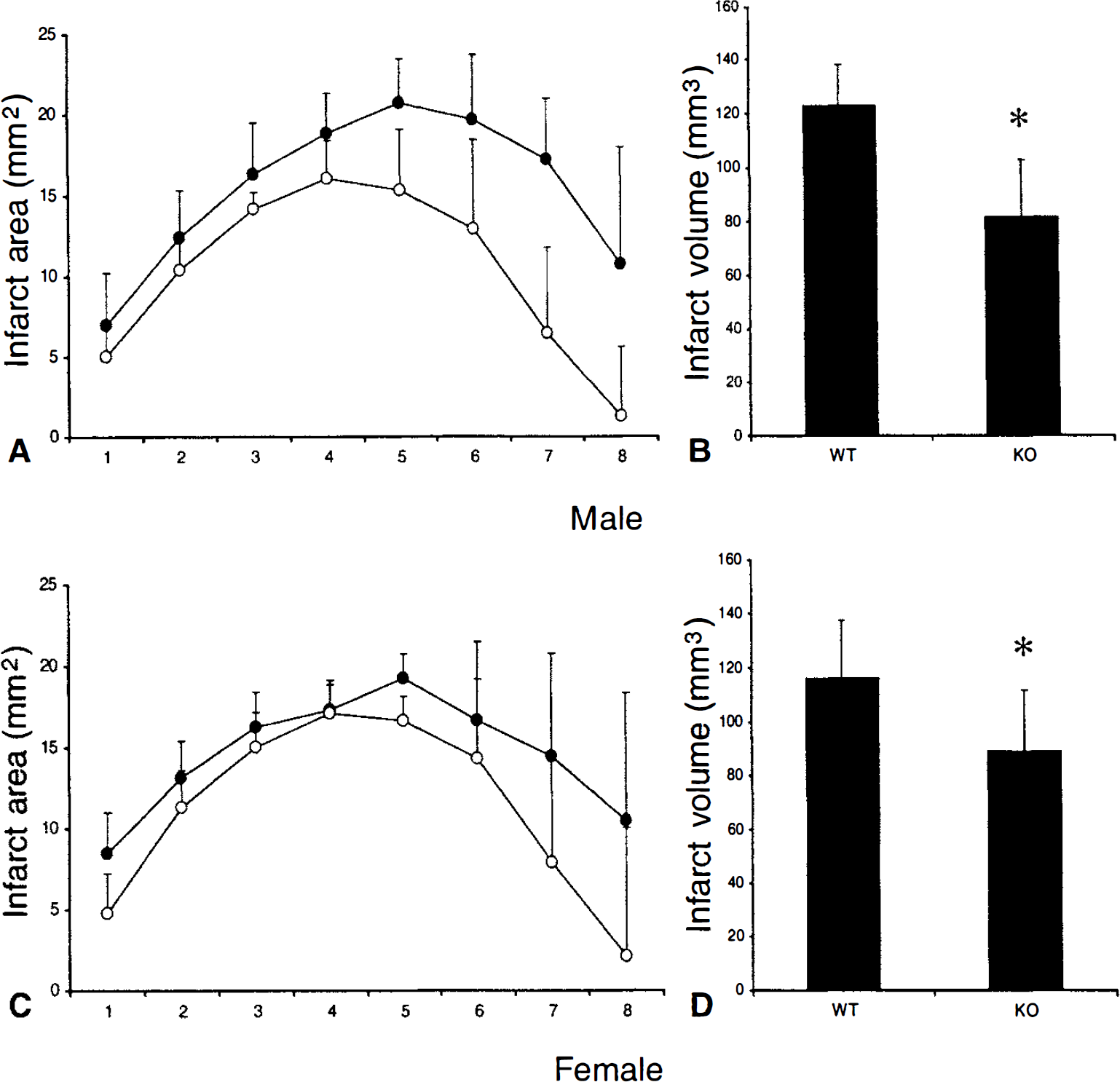
Reduction of 24-hour ischemic lesion volumes in MMP-9–deficient knockout mice (KO) compared with wild type littermates (WT) in males
Neurologic score
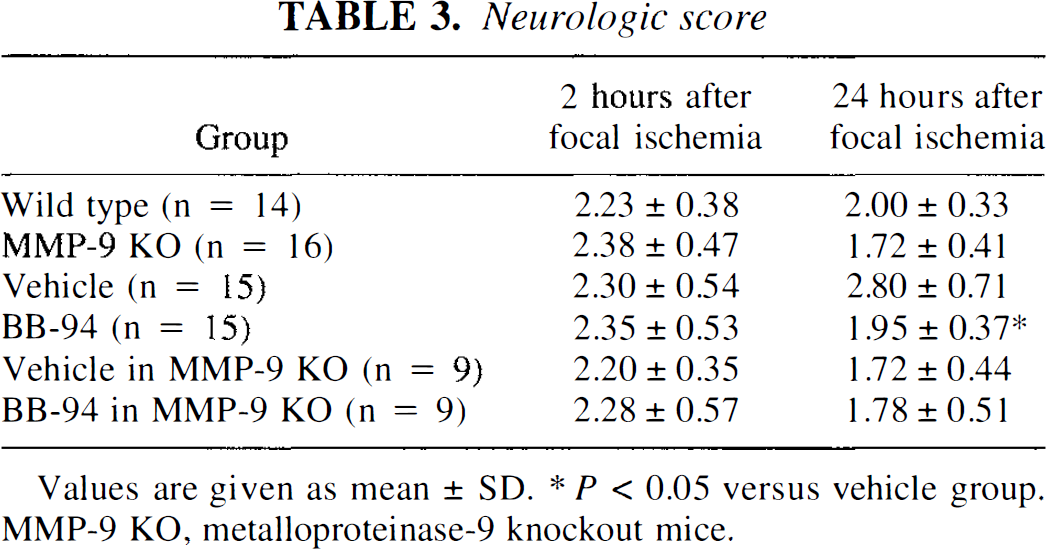
Values are given as mean ± SD. * P < 0.05 versus vehicle group. MMP-9 KO. metalloproteinase-9 knockout mice.
To confirm that MMP-9 was indeed absent in the knockout mice, Western blot and zymographic analysis were performed on brain extracts derived from MMP-9 knockout mice at 24 hours after focal cerebral ischemia. As expected, no expression of MMP-9 protein was detected (Figs. 1 and 2). Interestingly, however, a faint band of gelatinolytic activity was observed at approximately 72 kDa, corresponding with a potential up-regulation of MMP-2 in the ischemic MMP-9 knockout mice (Fig. 2).
BB-94 reduces ischemic lesion volumes in wild type mice
Intraperitoneal administration of the broad spectrum MMP inhibitor BB-94 (50 mg/kg) at 30 minutes before and 3 hours after focal ischemia significantly reduced ischemic lesion volumes at 24 hours (Figs. 5A to 5B). Concomitantly, neurologic deficits were significantly improved in BB-94–treated mice compared with vehicle-treated controls (Table 3). Systemic physiologic parameters including mean arterial blood pressure, rectal core temperature, and arterial blood pH, and gases were not significantly altered by BB-94 (Table 1). There were no differences in regional cerebral blood flow between controls and treated animals (Table 2).
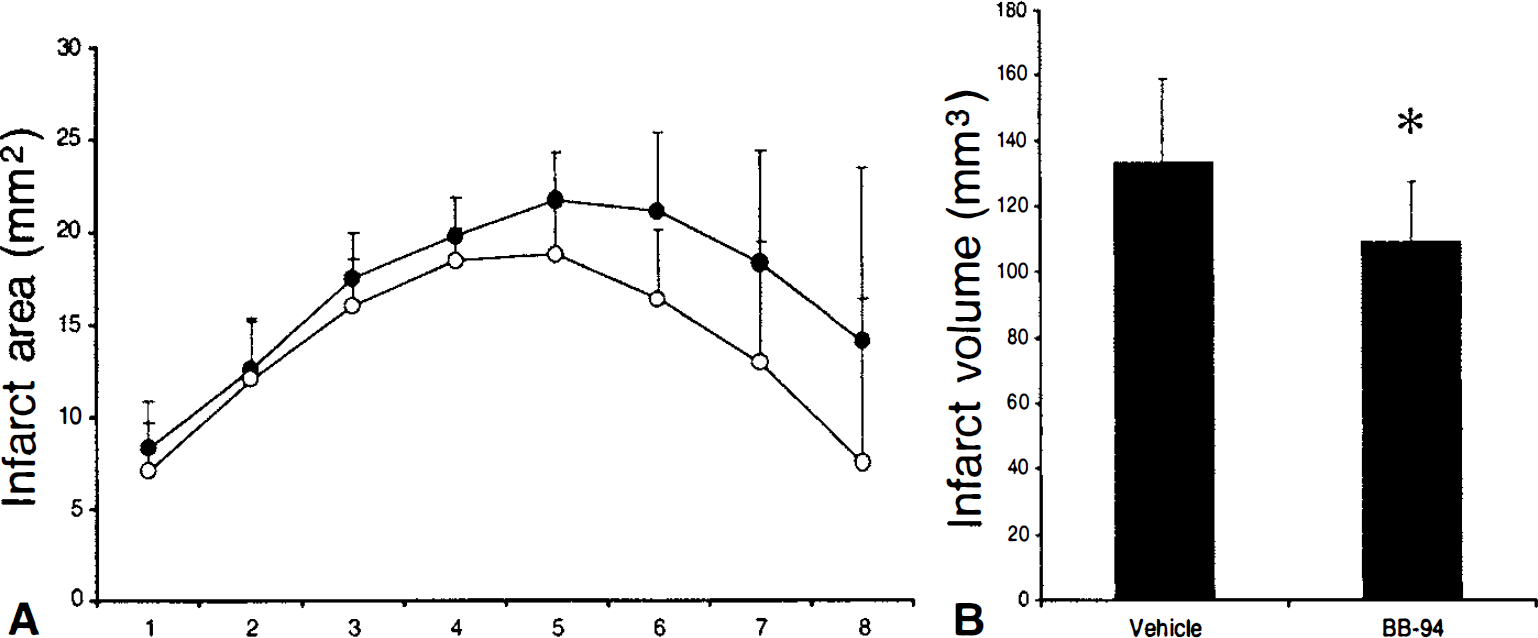
Reduction of ischemic lesion volumes by the broad spectrum MMP inhibitor BB-94 compared with vehicle-treated controls.
BB-94 does not reduce ischemic lesion volumes in MMP-9 deficient knockout mice
MMP-9 deficient knockout mice were subjected to permanent focal cerebral ischemia, and BB-94 treated animals were compared with controls given vehicle alone. Once again, two doses of BB-94 were administered 30 minutes before and 3 hours after ischemic onset (50 mg/kg per dose). BB-94 did not alter physiologic parameters in the MMP-9 knockout mice (Table 1). After induction of focal ischemia, there were no differences in regional cerebral blood flow between controls and BB-94–treated mice (Table 2). At 24 hours, there were no significant differences in ischemic lesion sizes between treated and control MMP-9 knockout mice (Figs. 6A to 6B). Correspondingly, there were also no differences in neurologic outcomes (Table 3).
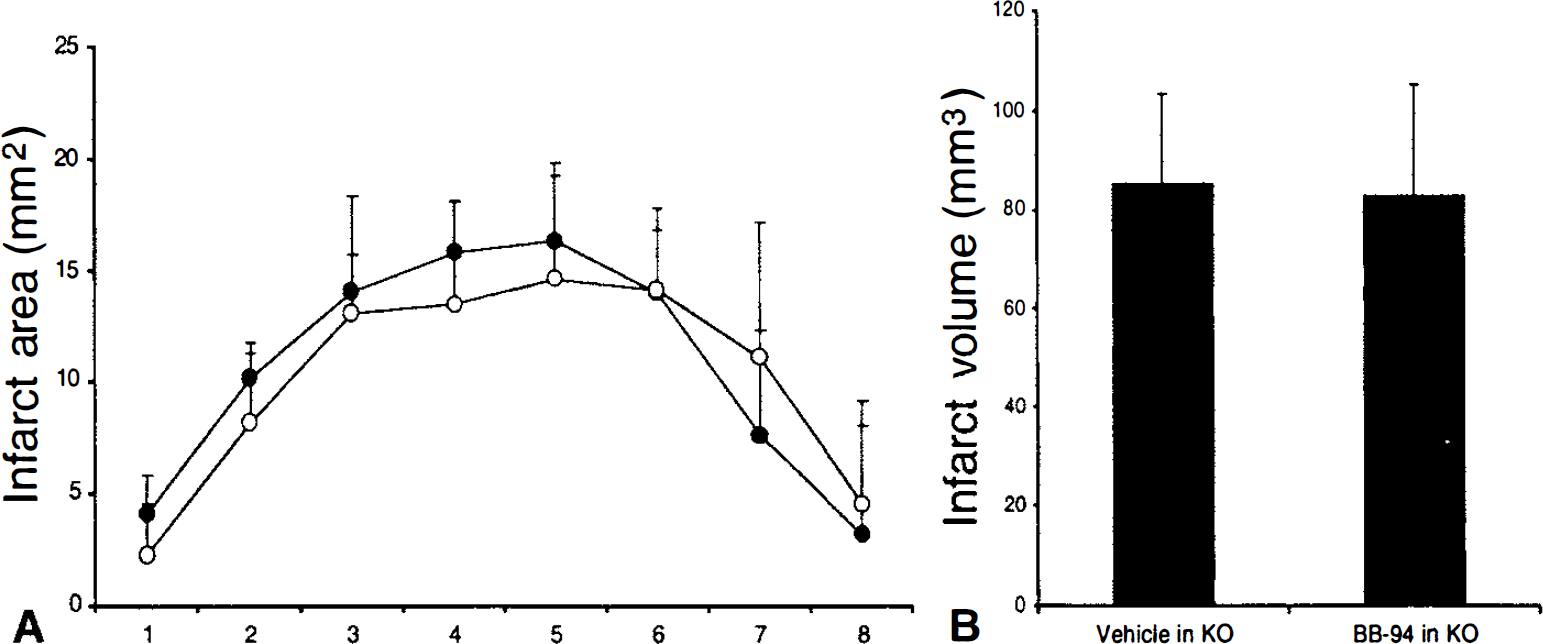
BB-94 did not further reduce ischemic lesion volumes when administered to MMP-9 knockout mice.
DISCUSSION
Recently, there have been several studies documenting the up-regulation of MMPs after cerebral ischemia (Gasche et al., 1999; Heo et al., 1999; Romanic et al., 1998; Rosenberg et al., 1996, 1998). In the current report, we demonstrate that MMP-9 may play a deleterious role in ischemic pathophysiology. Protein and mRNA levels of MMP-9 were up-regulated in mouse brain after focal cerebral ischemia. MMP-9–deficient knockout mice had significantly smaller ischemic lesions compared with their wild type littermates. The broad spectrum MMP inhibitor BB-94 reduced ischemic brain damage in wild type mice, but not in MMP-9 knockout mice.
Precise pathways involved in regulating MMP expression after ischemia remain unclear. Because the gene structures of MMP include API sites, it is possible that immediate early genes such as c-fos and c-jun are involved (Fini et al., 1998; Nagase and Woessner, 1999). It is known that c-fos and c-jun are rapidly up-regulated after cerebral ischemia (An et al., 1993). Another upstream regulating pathway may involve mitogen-activated protein kinase (MAPK) signal transduction (Fini et al., 1998; Nagase and Woessner, 1999). Various MAPKs have been shown to be up-regulated after ischemia—reperfusion (Abe et al., 2000), and inhibition of MAPK pathways have been shown to decrease ischemic brain damage (Alessandrini et al., 1999). In addition, a related mechanism may involve reactive oxygen species. Oxidative stress mediates a large part of the pathophysiology of cerebral ischemia (Chan, 1996). Recently, it has been shown that after brain trauma, MMP expression is reduced in mutant mice overexpressing superoxide dismutase (Morita-Fujimura et al., 1999). Interactions between oxidative stress and the proteolytic cascade subserved by MMPs may ultimately mediate the progression and maturation of the ischemic lesion.
As expected, no significant MMP-9 was detectable in normal brain, which is consistent with the understanding that MMP-9 is not constitutively expressed in mammalian brain (Gasche et al., 1999; Heo et al., 1999; Rosenberg et al., 1996). However, after ischemic brain injury, MMP-9 protein levels increased as early as 3 hours after injury and lasting until at least 24 hours, at which time ischemic were assessed in the current study. Increased MMP-9 protein appeared to be mediated by up-regulated transcription, as shown by elevated mRNA levels. Nevertheless, it will be important to carefully document the temporal profile of MMP-9 levels over longer periods of time in future studies to fully understand its contribution to long-term outcomes. Long-term reorganization of brain after stroke may also involve additional MMP activity. It is possible that a balance may exist between acute deleterious effects of MMPs versus potentially beneficial effects of MMPs that subserve remodeling in the chronic phase of stroke recovery.
In the current study, genetic and pharmacologic approaches were used to make a mechanistic connection between increased levels of MMP-9 protein and brain tissue damage. However, we did not directly measure proteolytic activity and cannot unequivocally exclude the effects of some undefined activity of MMP-9. In addition, the MMP-9 protein detected was primarily the 105 kDa proenzyme zymogen. Very little of the truncated form of the enzyme associated proteolytic activity in vitro was detectable. This in vivo finding is often reported (Fini et al., 1996; Matsubara et al., 1991; Mohan et al., 2000) and is not inconsistent with functionality of MMP-9 as a protease in vivo. A number of studies suggest that enzymatic activation of MMP-9 proenzyme occurs by binding at the cell surface, and activated enzyme is then rapidly degraded to prevent excess activity (Yu and Stamenkovic, 1999). Therefore, only very low levels of active MMP-9 may be present in tissue at any given time.
The current zymographic data show that after ischemic brain injury, the primary gelatinase elevated was MMP-9. Only faint zones of activity were observed at the corresponding band for MMP-2. It is possible that low but important levels of MMP-2 were not detected in the current study because the authors did not use purification schemes that have been used by others to amplify the measurements of endogenous brain MMPs (Gasche et al., 1999; Morita-Fujimura et al., 1999). In a study of cerebral ischemia in the nonhuman primate baboon brain, elevations in MMP-2 activity were observed (Heo et al., 1999).
Although the current data demonstrate that MMP-9 may contribute to brain damage after ischemia, the precise mechanisms that underlie its deleterious effects remain to be defined. In previous studies, an emphasis has been placed on the fact that MMP-9 can digest several matrix proteins present in the vascular basal lamina including collagen, fibronectin, and laminin. Damage to vascular integrity would then lead to disrupted blood—brain barrier function and increased vasogenic edema (Gasche et al., 1999; Heo et al., 1999; Rosenberg et al., 1996). However, matrix proteins such as laminin may also be found in brain parenchyma (for example, hippocampus and cerebellum), and loss of parenchymal laminin may affect cell—matrix interactions and cell survival (Chen and Strickland, 1997). Therefore, it remains conceivable that MMP-mediated matrix protein degradation may disrupt cell—matrix interactions beyond the vascular compartment as well, and further contribute to neuronal cell damage (Heo et al., 1999). A pathophysiologic role for MMPs may extend to other forms of acute brain injury. We recently have shown that MMPs are up-regulated after traumatic brain injury, and MMP-9 knockout mice are similarly protected in terms of both morphologic damage and functional motor recovery (Wang et al., 2000).
There are a few caveats associated with the current study. First, we only measured ischemic lesion volumes at an early time (24 hours). We cannot exclude the possibility that long-term effects may occur that overwhelm the acute protective effects of MMP-9 gene knockout. Second, we do not have data on the cell types that serve as sources for MMP-9. It is well known that macrophages and neutrophils can be an important source for MMPs and other deleterious proteases (Pagenstecher et al., 1998), and inflammatory macrophage and neutrophil infiltration is clearly an important event after cerebral ischemia. However, it has been shown that resident brain cells (neurons, astrocytes, oligodendrocytes) (Gottschall and Deb, 1996; Gottschall and Yu, 1995; Oh et al., 1999; Zuo et al., 1998) and endothelial cells (Herron et al., 1986) can also produce MMPs. It will be important to carefully define the cellular sources of deleterious MMP activity after ischemic brain injury. Finally, the MMP family comprises many other members besides MMP-9. MMPs (along with tissue inhibitors of MMP and plasminogen activators) form a complex protease cascade that may bear some similarity to the caspase cascade in terms of amplifying cell death (Cuzner and Opdenakker, 1999). Much like the case with caspases, where caspase-3 may be a possible final executioner, it has also been proposed that MMP-9 may be a terminal member of the MMP cascade (Cuzner and Opdenakker, 1999). Nevertheless, it will be critical to assess the roles for more upstream signaling pathways using other specific antibodies, and knockout mouse models.
In conclusion, the current study documented the role for MMP-9 in a mouse model of focal cerebral ischemia. Genetic and pharmacologic inhibition of MMP-9 significantly reduced ischemic brain damage. Importantly, the broad spectrum MMP inhibitor BB-94 did not protect ischemic brain in MMP-9 knockout mice, suggesting that MMP-9 plays a critical and deleterious role in the pathophysiology of ischemic brain injury. Further studies are warranted to assess the interactions between MMP-9 and other proteolytic enzymes in ischemia.
Footnotes
Acknowledgments
The authors are grateful to Robert Senior and J. Michael Shipley for permission to use the MMP-9 knockout mice and the anti-MMP-9 antibody. BB-94 was donated by British Biotech. Paul Huang provided advice for the laser—Doppler measurements. Laoti Russo provided expert help with the maintenance of the knockout colony. The authors also thank Xiaoying Wang, Toshihisa Sumii, Tatsuro Mori, and Aneesh Singhal for important discussions, and Michael Moskowitz for his unstinting intellectual support.
