Abstract
The diameter of surface microvessels and the erythrocyte velocity and flux through intraparenchymal capillaries in the parietal cortex were measured during transient global cerebral ischemia and reperfusion using laser-scanning confocal fluorescence microscopy in anesthetized rats. The role of nitric oxide (NO) from neurons in the microcirculatory changes was also investigated using 7-nitro-indazole (7-NI, 25 mg/kg, IP). Wistar rats (4 per group) equipped with a closed cranial window were given fluorescein isothiocyanate (FITC)-Dextran and FITC-labeled erythrocytes intravenously to respectively visualize the microvessels and the erythrocytes in the capillaries. Experiments were videorecorded on-line. Forebrains were made ischemic for 15 minutes and then reperfused for 120 minutes under the microscope. Ischemia was associated with a flattened EEG, a low persistent blood flow, and a transient leakage of fluorescein across the arteriole wall. Unclamping the carotid arteries led to immediate high blood flow in the arterioles, but it was not until 5 minutes later that the arterioles dilated significantly (181% ± 27%) and erythrocyte velocity in the capillaries increased significantly (460% ± 263%). Neither nonperfused capillaries nor erythrocyte capillary recruitment occurred. 7-Nitro-indazole significantly reduced the arteriole dilatation and prevented the increase in erythrocyte velocity and flux through capillaries in early reperfusion. 7-Nitro-indazole had no influence on the fluorescein leakage. The current study suggests a partial role for NO released from neurons in the postischemic microcirculatory changes and provides new findings on the timing of arteriole dilatation and blood—brain barrier opening, and on erythrocyte capillary circulation in global ischemia.
Because of a lack of appropriate methods, there was little direct exploration of the capillary intracerebral microcirculation until the last decade. Brain metabolism is mostly oxidative and the O2 carried by erythrocytes is rate limiting. Thus, the erythrocyte flow through capillaries is more important than plasma flow for nutritive perfusion. The problem of nonperfused capillaries after forebrain ischemia has been thoroughly debated, but restricted to plasma flow (Theilen et al., 1993).
Hudetz' group (1997), using epifluorescence microscopy, has described capillary erythrocyte circulation in various physiologic and pathophysiologic conditions. However, no investigation under cerebral ischemia has been reported. The recent development of confocal fluorescence microscopy enabled Villringer et al. (1994) to explore single capillaries in one dimension with erythrocytes appearing as black shadows, but with a reconstruction phase, and only in basal and hypercapnic conditions. These two methods are not suitable for simultaneous investigations of arterioles, venules, and capillaries.
The authors have recently used laser-scanning confocal fluorescence microscopy in a new method (Seylaz et al., 1999) that allows the dynamic, on-line visualization of both microvascular networks and the erythrocytes flowing through parenchymal capillaries. This method was used in the current study to directly assess the magnitude and exact timing of the microvascular changes, the functional dependence of capillaries on arteriolar and venular circulation, the microvessel permeability under 4-vessel occlusion (4-VO) and postischemic reperfusion, and also to address the question of capillary erythrocyte recruitment.
The authors also tested the hypothesis that neuronally-derived nitric oxide (NO) plays a vasoactive role in this model. Nitric oxide is a very simple molecule produced by the activity of very complex enzymes, NO synthases (NOS). Nitric oxide was first identified as endothelium-derived relaxing factor and is a potent vasodilator. The discovery of (neuronal) type I NOS in a dense network of neuron fibers mainly originating from the sphenopalatine ganglia and running in the adventitia of cerebral arteries (Nozaki et al., 1993), as well as the finding of scattered type I NOS-containing neurons close to parenchymal vessels, have led to the hypothesis that NO released from neurons plays a role in the regulation of brain microcirculation. Some data favor this hypothesis because 7-nitro indazole (7-NI), a relatively selective type I NOS inhibitor (Babbedge et al., 1993), reduces basal cerebral blood flow (CBF) without modifying CMRGlu (Kelly et al., 1995) and attenuates the CBF increase in various physiologic and pathophysiologic conditions, including epileptic seizures (Montécot et al., 1997). In addition, using 7-NI, it has been shown that NO from type I NOS contributes to the maintenance of erythrocyte flow through cerebral capillaries and plays a critical role in the selective regulation of cerebral capillary flow during hypoxia (Hudetz et al., 1998). Nitric oxide release has been measured in the ischemic brain by various methods (Tominaga et al., 1994; Olesen et al., 1997), but its role has been explored more in terms of neurotoxicity than of vascular effect. Wei et al. (1999) have demonstrated a progressive accumulation of NO from type I NOS after the onset of global ischemia by using knocked-out mice. In the rat brain submitted to global ischemia, 7-NI reduces the generation of NO by 47% (Olesen et al., 1997) and is neuroprotective (Nanri et al., 1998), which suggests a deleterious role for the NO released from neurons. In gerbils, 7-NI inhibited NO production during global ischemia, but afforded neuroprotection for short periods of ischemia only (Lei et al., 1999). However, laser—Doppler flowmetry revealed no influence of 7-NI on the CBF changes associated with reperfusion (Humphreys and Koss, 1998). In the current study, the authors directly investigated the effect of type I NOS inhibition by 7-NI on cortical microcirculation in transient forebrain ischemia and reperfusion.
MATERIALS AND METHODS
All experimental procedures were performed in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. Experiments were performed under permit No. 02934 from the French Ministry of Agriculture.
Male Wistar rats weighing 260 to 280 g (Seine Maritime, France) were used in this study. Rats had access to food and water ad libitum and were housed in individual cages.
Confocal microscopy
A BioRad Viewscan confocal laser-scanning unit (England) attached to a Nikon Optiphot-2 microscope (Japan) was used. The scanning unit scanned 50 fields per second. A detailed description of the apparatus has been published previously (Seylaz et al., 1999). The light source was an argon-krypton laser (λ 488, 568, and 647 nm). An appropriate filter was used for fluorescence microscopy of fluorescein. Images were recorded at video speed (50 frames per second) with a SIT camera (Hamamatsu, Japan) and a PAL-VHS video recorder (Panasonic, Japan). Images then were digitized and analyzed off-line to determine the vessel diameter and the velocity and flux of fluorescent-labeled erythrocytes through capillaries by means of appropriate software under the Optimas environment.
Preparation of animals for confocal microscopy
Just before the experiment, rats were anesthetized with sodium pentobarbital (Pentothal, 50 mg/kg, IP) and placed in a stereotaxic apparatus (David Kopf). The scalp and connective tissue above the parietal cortex area were removed. Silver electrodes were fixed to the cranial bone with dental cement to record the electroencephalogram (EEG). A closed cranial window was prepared. The cranial bone was thinned to translucency by means of a water-cooled dental drill and carefully removed over an area of 4 mm2. The dura mater was then replaced by a 150-μm-thick quartz microscope coverglass, sealed to the bone with dental cement (Palaferm, Kulzer) to make the preparation waterproof.
Arterial and venous catheters were placed into the femoral vessels for measuring arterial blood pressure, blood gases, hematocrit, and pH and for injecting the fluorescent tracers. Fluorescein isothiocyanate-labeled (FITC-labeled) erythrocytes were prepared in vitro (Seylaz et al., 1999).
Preparation of animals for global ischemia
The rats were prepared for transient global ischemia using the 4-VO model (Pulsinelli and Brierley, 1979). On the day before the experiment, the rats were anesthetized with pentobarbital (60 mg/kg) and both vertebral arteries were electro-cauterized after exposure of the alar foramina under a surgical microscope (Zeiss). On the day of the confocal microscopy experiment, the pentobarbital-anesthetized rats first were equipped with the cranial window and then the bilateral common carotid arteries (CCA) were isolated from surrounding tissues through a midline neck incision, and silk ligatures were placed loosely around them. The duration of the whole preparation was 2 hours from the start of anesthesia to induction of forebrain ischemia.
Experimental protocol
The rat was placed under the confocal microscope and its head was secured in a custom-built stereotaxic device closely fitting the confocal microscope stage. Electrodes were connected to a polygraph (ECEM) for continuous EEG recording. The arterial catheter was connected to a pressure transducer (Statham) for continuous recording of arterial blood pressure (Gould). Rats were ventilated with room air slightly enriched with O2. Arterial blood was regularly sampled to check blood gases and pH (Corning). The hematocrit was measured before and at different times after injecting the tracers. Body temperature was kept at 37°C throughout the experiment by a homeothermic blanket control unit (Harvard Apparatus). To visualize microvessels and delineate their internal walls, 0.5 mL FITC-Dextran (molecular weight = 70,000) diluted in 0.9% NaCl (2.5 mg/mL) was injected into the blood circulation. Fluorescein isothiocyanate—labeled erythrocytes were injected through the same route at a tracer dose (<2%). The whole cranial window was systematically explored with a 10x objective lens (Fig. 1A) to determine the most appropriate field of investigation (one including several arterioles and venules and a dense capillary network) and this was repeated with a 20x objective lens (Fig. 1B). Intraparenchymal capillaries were visualized by progressively changing the focus down to a depth of 200 μm beneath the surface of the brain. The capillaries analyzed in the current measurements were usually located within 50 to 100 μm of the parenchyma.
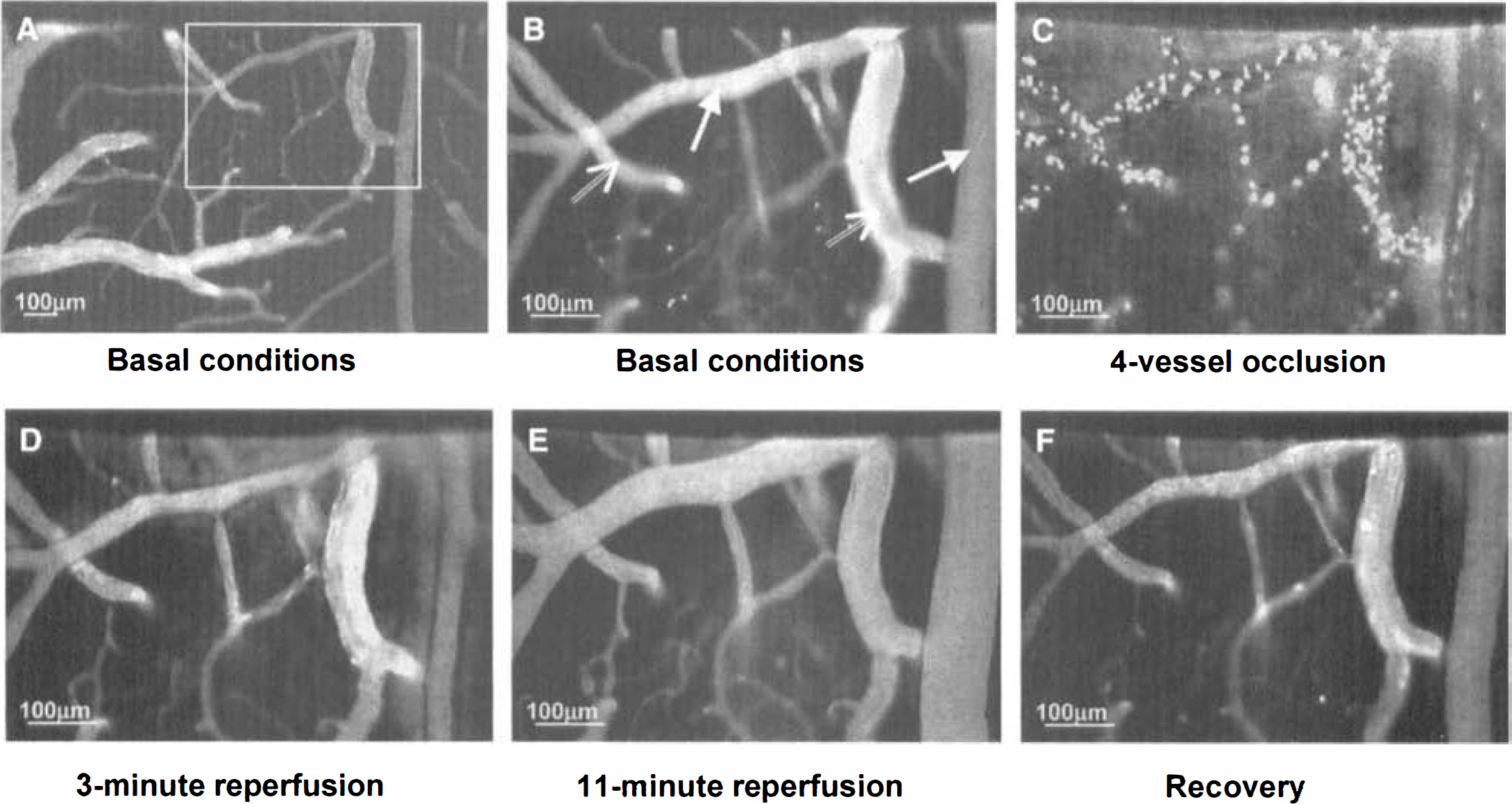
Typical rat microvascular network visualized by laser-scanning fluorescence confocal microscopy through a closed cranial window at key moments of the experiment.
The microcirculation was videorecorded throughout the experiment for periods of 3 minutes every 10 minutes in control conditions and under vehicle or 7-NI treatment, for the whole duration of ischemia and early reperfusion, and for periods of 3 minutes every 5, then 10, then 15 minutes in late reperfusion. These limited sequences of videorecording were used to avoid continuous laser illumination, which could damage the tissue. A control image of the region of interest under basal conditions was stored and permanently displayed on a TV screen to permit the exact repositioning of the microvascular network in case of displacement and to realign capillaries in the field of view.
With the rat head under the confocal microscope, forebrain ischemia was induced 45 minutes after vehicle or 7-NI treatment by occluding the CCA. A flattened EEG confirmed the completeness of ischemia. After 15 minutes of ischemia, reperfusion was performed under the confocal microscope by releasing the clamps. The recovery was analyzed for 120 minutes after CCA unclamping. Then wounds were sutured and infiltrated with xylocaïne, and rats were awakened and returned to their cages. Seven days later, animals were deeply anesthetized and then decapitated. The brain was removed and then frozen during 1 minute in isopentane at −40°C and stored at −80°C. Brains were cut coronally with a cryostat into 15-μm slices at 500-μm intervals. Slices were stained with cresyl violet to attest the success of ischemia and 7-NI treatment, and to check that cortical tissue was undamaged by laser illumination.
Experimental groups
Rats were divided into 2 groups (n = 4 per group). The control group was given intraperitoneal injections of peanut oil, the vehicle of 7-NI, 45 minutes before the carotid clamping and again hourly. The 7-NI-treated group was given 7-NI (25 mg/kg, IP) at the same timing. The dose of 7-NI was selected on the basis of functional studies (Moore et al., 1993) and because the authors previously demonstrated (Nanri et al., 1998) that it is neuroprotective at this dose in similar experimental conditions while inhibiting 70% of brain NOS activity. The repeated 7-NI injections were motivated by the transient effect of 7-NI on NOS activity.
Labeled erythrocyte velocity and flux measurement
The fluorescent erythrocyte velocity (mm/sec) was measured through 2 to 4 capillaries per rat. The measurements were performed in basal conditions and over 19 representative periods: 30 minutes after the injection of vehicle or 7-NI, just before CCA clamping (that is, time 0), 2, 3, 8, and 15 minutes after clamping, and 1, 4, 8, 13, 20, 25, 30, 40, 50, 60, 90, 105, and 120 minutes after CCA unclamping. Velocity was measured over capillary segments of approximately 100 μm. The fluorescent erythrocytes flux through capillaries (number of labeled erythrocytes flowing through a given capillary per unit of time) was measured in the same capillary segments at the same times.
Vessel diameter measurement
Approximately 4 to 5 segments of arterioles and 2 of venules (sometimes from the same tree) were selected from the videorecording of each experiment in control conditions. The internal diameter of the vessels was measured on digitized images using Optimas software throughout the video recorded experiment, at the same times as the fluorescent erythrocyte capillary flow parameters.
Materials
Fluorescein isothiocyanate—Dextran (molecular weight = 70,000) and FITC Isomer I were purchased from Sigma (Sigma Aldrich, Isère, France). 7-Nitro indazole (Sigma Aldrich) was suspended in peanut oil by sonication.
Statistical analysis
Data are expressed as means ± SD. Differences were regarded as statistically significant at P < 0.05. All measurements were taken independently. Within each group, for each variable, the time-related changes were analyzed using a one-way analysis of variance followed by Dunnett's test. The values measured just before CCA clamping were taken as basal values.
At each time point, intergroup comparison was made using bilateral Student's t-test for microvessel diameter and systemic parameters, and using the nonparametric Mann—Whitney U test for labeled erythrocyte velocity and flux changes in capillaries.
RESULTS
Control rats
All rats were in similar physiologic conditions as attested by systemic parameters in basal conditions (Table 1). Two rats presenting electrical activity during the ischemic period were discarded from the study. No change in any systemic parameter occurred at any time during the experiment, except a transient nonsignificant increase in mean arterial blood pressure at CCA clamping, and a nonsignificant decrease in Pa
Mean values of the physiologic parameters in control rats (n = 4) before, during, and after transient forebrain ischemia
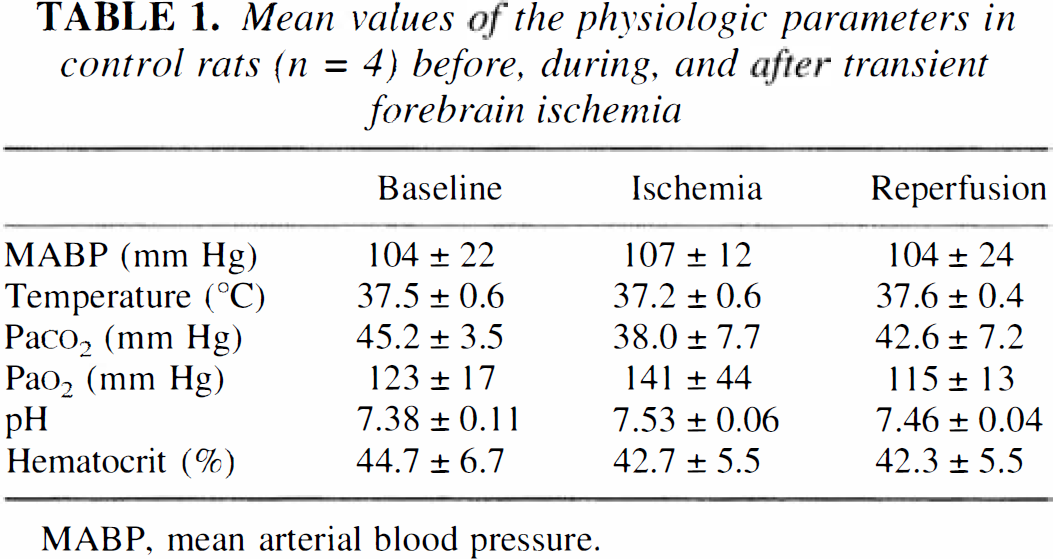
MABP, mean arterial blood pressure.
A typical example of videorecording of the cerebral microcirculation at key moments is shown in Fig. 1B to 1F. The statistical microcirculatory data are summarized in Fig. 2.
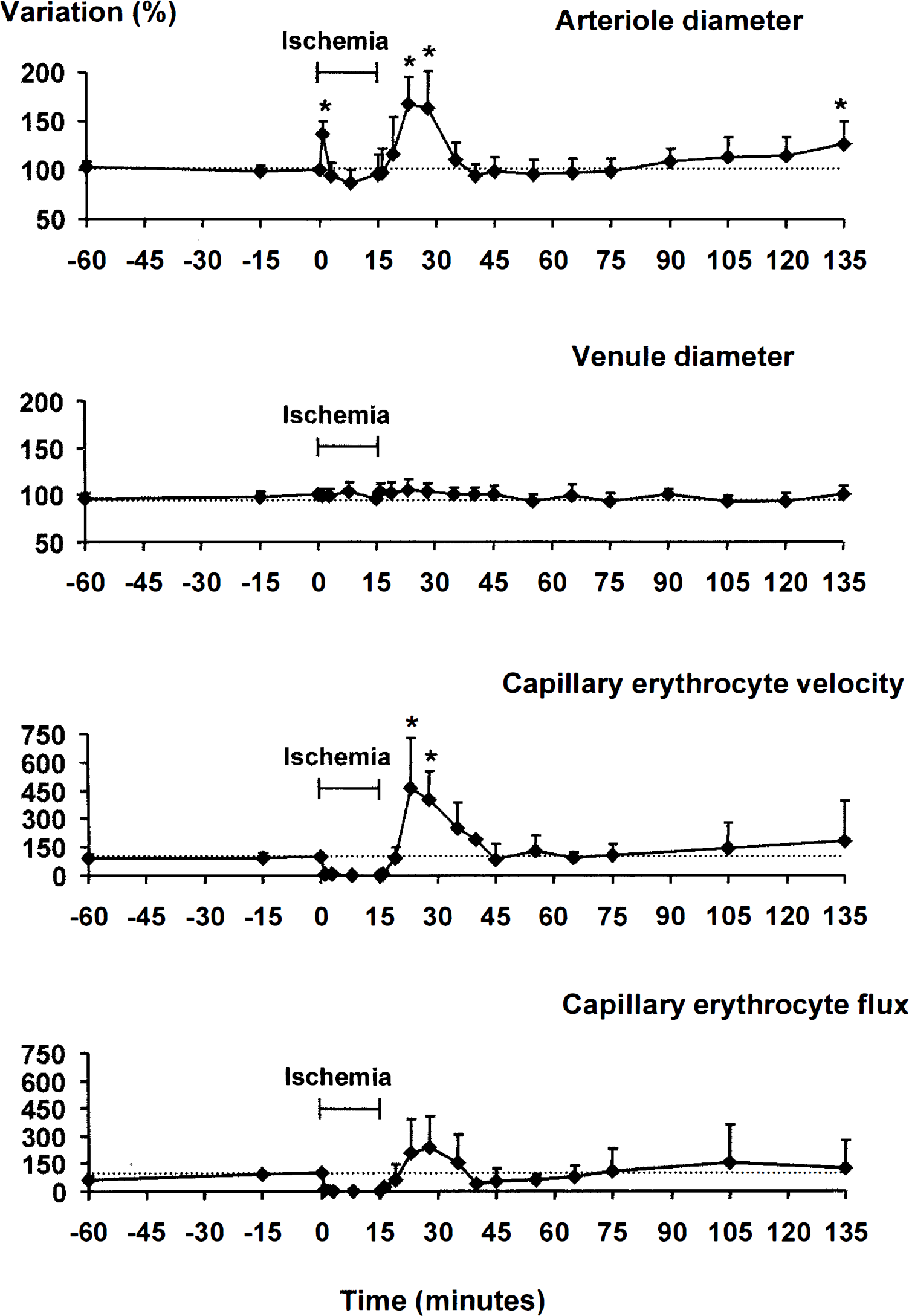
Changes in microcirculatory variables throughout the experiments in control rats. Changes are expressed in percent of the basal value taken at time 0 (Just before CCA clamping) and taken as 100%. The analysis was performed in 19 arteriole segments, 8 venule segments, and 9 capillaries from 4 rats. Forebrain ischemia was induced from 0 to 15 minutes and was associated with a flattened EEG. *Indicates a significant difference from the basal value at P < 0.05.
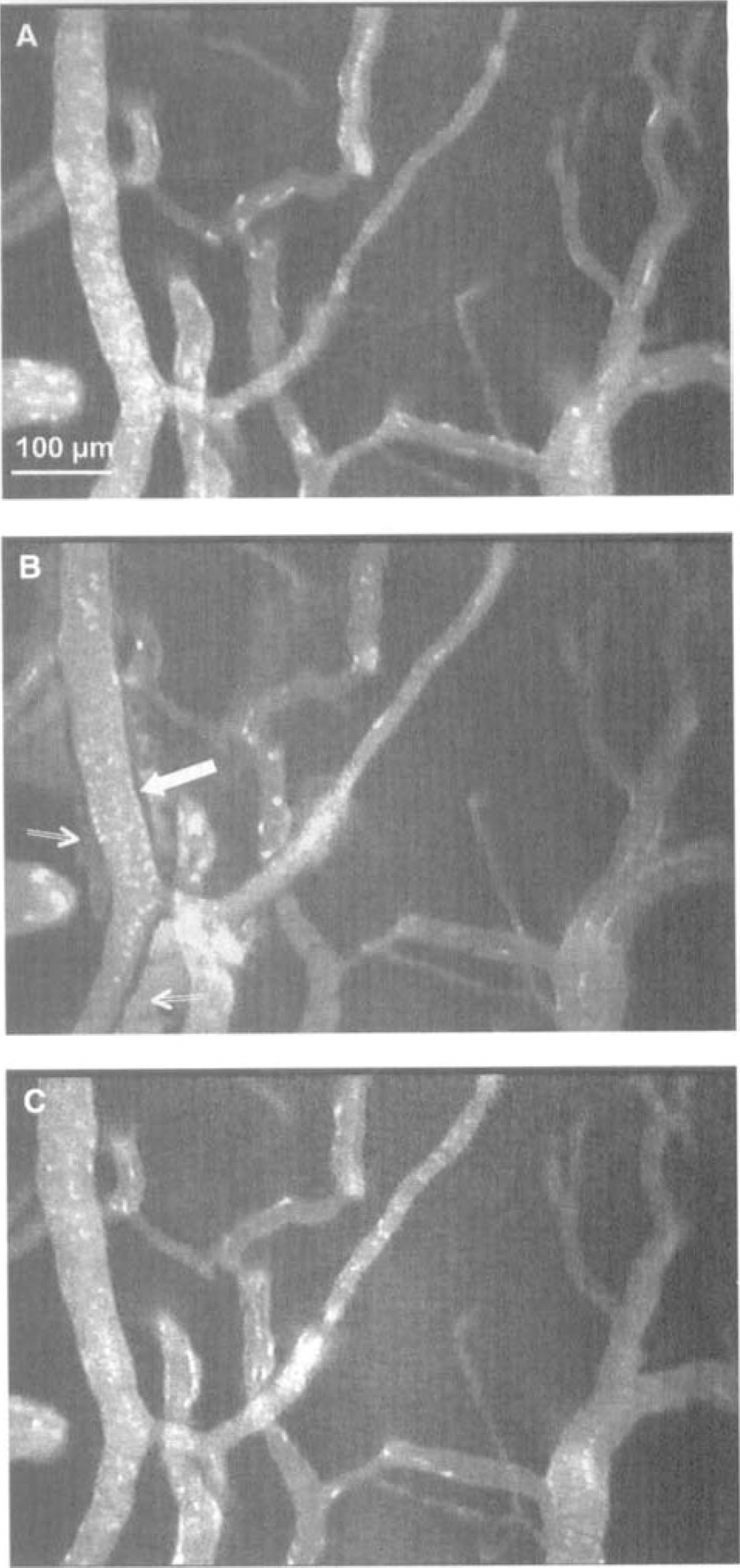
Typical example of an arteriole under basal conditions
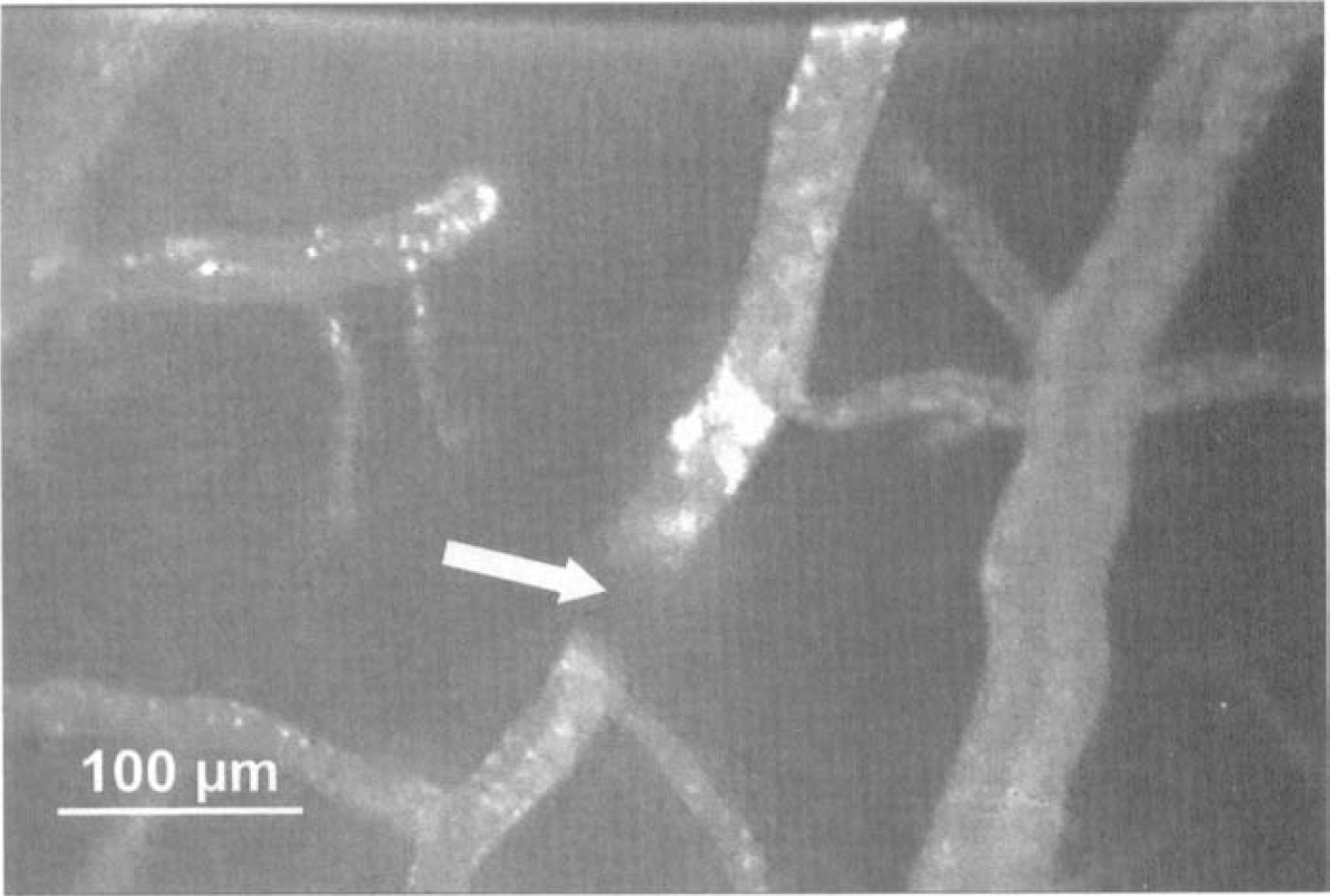
A circulating thrombus (indicated by a white arrow) generating turbulence in an arteriole. Note the upstream accumulation of fluorescein.
7-Nitro-indazole-treated rats
There were no significant differences in systemic parameters for 7-NI—treated rats and control rats at any time during the experiments. 7-Nitro-indazole also caused no significant change in mean arterial blood pressure, indicating its lack of effect on endothelial NOS activity.
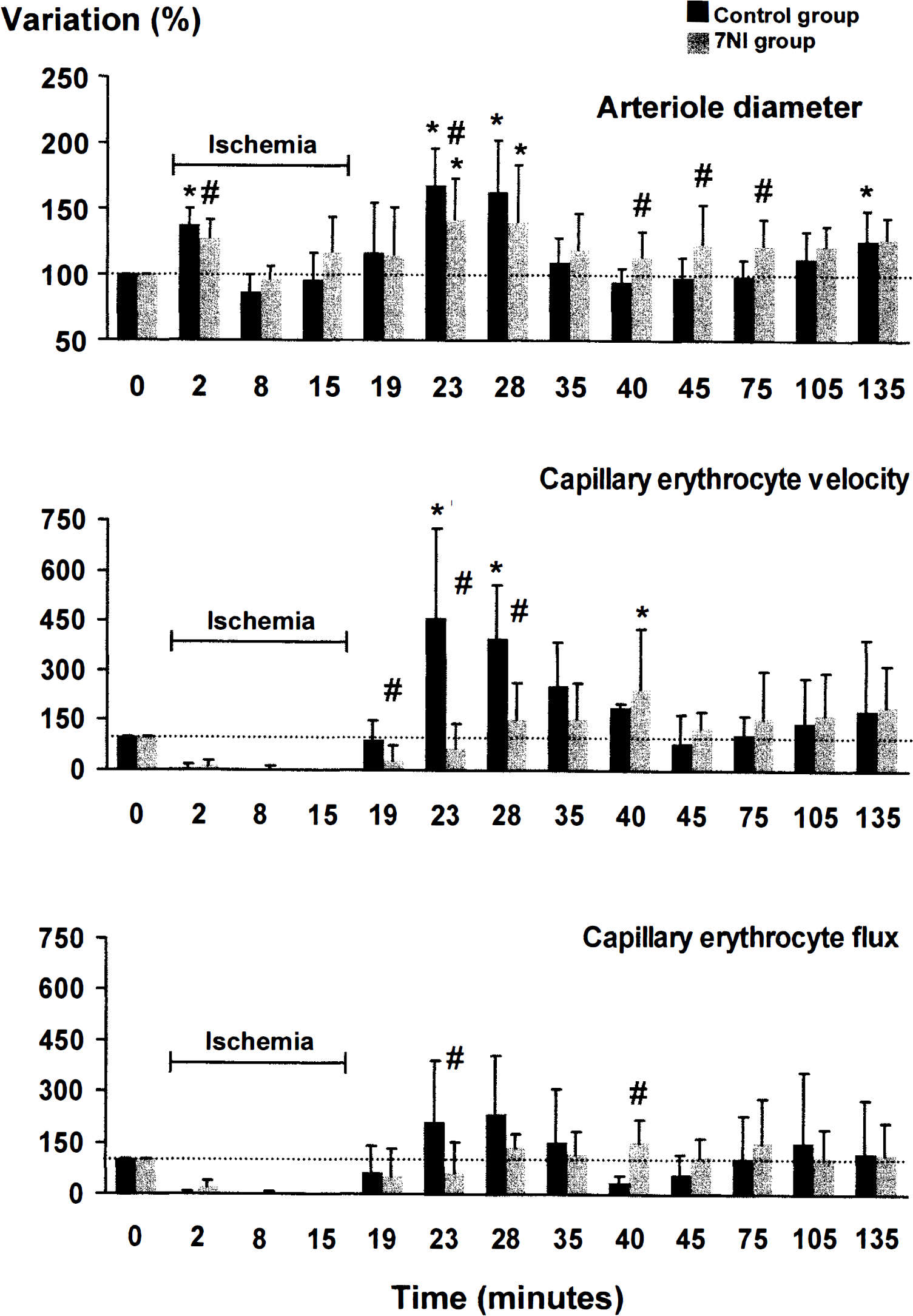
Comparison of cerebral microcirculatory changes between control rats (black columns) and 7-NI—treated rats (grey columns) in 15-minute forebrain ischemia and reperfusion. In the 7-NI—treated group, the analysis was performed in 14 arteriole segments and 13 capillaries. *Indicates a significant difference from the basal value at P < 0.05. #Indicates a significant difference between the two groups at P < 0.05.
Qualitative histologic examination of the brain slices from all rats seven days after the experiment evidenced a better outcome of cortical and hippocampal pyramidal neurons in 7-NI—treated rats than in control rats, as the authors had semiquantitatively shown in a previous study (Nanri et al., 1998).
DISCUSSION
The current study is the first direct dynamic analysis of the cerebral microcirculatory events occurring in the acute phase of the 4-VO model in rats, which has been one of the most widely investigated models of transient global cerebral ischemia for the last 20 years. The pentobarbital anesthesia differs from those commonly used in neuroprotection studies, but it was adapted to induce profound anesthesia, which is a prerequisite for an in vivo study under a microscope. In addition, pentobarbital does not interfere with brain NOS activity, unlike halothane (Tobin et al., 1994). Pentobarbital decreases the velocities of erythrocyte and plasma flows in the cerebral microcirculation (Wei et al., 1993).
The major findings of the current study can be summarized as follows: first, the delay of several minutes in the arteriole dilatation at the CCA unclamping whereas blood reperfusion was instantaneous; second, the close association between the arteriole dilatation and the increase in capillary perfusion; third, the reduction by 7-NI of both the postischemic arteriole dilatation and the increase in capillary erythrocyte circulation; fourth, the lack of capillary recruitment in its classical sense (open vs. closed capillaries) in the postischemic hyperemic phase; fifth, the early transient opening of the BBB during the ischemic insult; and sixth, the generation of venous thromboses and circulating thrombi throughout the arterioles. None of these two latter events were affected by 7-NI treatment.
The methodology is not discussed in the current article because it has been covered in detail in a previous paper (Seylaz et al., 1999). Briefly, the main advantage of laser-scanning confocal fluorescence microscopy is that it makes possible the high-quality visualization of surface microvessels and erythrocytes flowing through intraparenchymal capillaries to a depth of 200 #mUm from the brain surface. Thus, both the blood supply from surface arterioles and the tissue oxygenation can be estimated. The investigation is direct, because it is performed in situ, on-line, and continuously for long periods. Although sources of error cannot be excluded, it has to be mentioned that the hematocrit was not significantly modified by the tracer injection, the tissue was not damaged by the laser illumination, and the vessel reactivity was preserved.
Microcirculation and ischemia
At unclamping, the perfusion pressure was restored, but unlike the response after a 20-second ischemia (Seylaz et al., 1999), the arterioles did not dilate instantaneously, although their smooth muscle cells were relaxed. The delay of several minutes before vasodilatation may be the result of a conflict of signals on the smooth muscle cells, a vasoconstrictory effect caused by the restoration of myogenic tone and a vasodilatory effect caused by the recirculation of blood and the associated shear stress. The delay could also be caused by the time needed for the functional recovery of endothelial and muscular cells. The vascular wall responds to an increase in blood flow under physiologic conditions by an endothelium-mediated dilatation, but the endothelial and muscular mechanisms may be disrupted under reperfusion after 15 minutes of severe hypoxia, as are the coronary cells (Pearson et al., 1990). The transient opening of the blood—brain barrier in arterioles during ischemia, reflecting the loss of one of the major characteristics of cerebral endothelial cells supports this. As the fluorescein leakage through arteriole walls stopped when the arteriole dilated, the endothelial cells may have partially recovered from ischemia at that time. Cerebral smooth muscle cells are also sensitive to hypoxia because the relaxation of a denuded middle cerebral artery under transmural electrical stimulation is inhibited by hypoxia (Okamura et al., 1997).
Under recovery, up to 120 minutes after ischemia, at the time when hypoperfusion has been described (Pulsinelli et al., 1982), capillary erythrocyte flux and velocity were not different from basal conditions, whereas arteriole diameter tended to increase. Such a divergence from classical CBF measurements was also found by Tasdemiroglu et al. (1992), who measured an increase in arteriole diameter for up to 120 minutes after ischemia. These data, in addition to the circulating thrombi that was often seen by the authors, indicate that vessel obstruction is a more likely cause of hypoperfusion than vasoconstriction, although this has been denied for many years (Hossmann, 1997).
The transient leakage of fluorescein through arteriole walls may be interpreted as the opening of the blood—brain barrier to a rather large protein for a short period of time, starting in ischemic conditions. The opening of the BBB in arteriole segments had been reported (Johansson et al., 1970), but under acute hypertensive conditions. This is the first description of such an early process in ischemia, because the BBB is known to be permeable to proteins 2 to 3 hours after ischemia (Del Zoppo, 1994). This finding is important when considering the early delivery of drugs to brain tissue.
In the early reperfusion phase, the mismatch between capillary perfusion and blood flow demonstrated in the current study has also been shown in gerbils with different techniques (Ennis et al., 1990). The delayed concomitant increase in erythrocyte capillary perfusion and arteriole dilatation found in the current study fits well with the classical CBF measurements performed 10 minutes after transient global ischemia. It can also be correlated to the tissue P
The point of capillary recruitment has been extensively discussed, and it is now accepted that there is no capillary recruitment in terms of plasma flow in the post-ischemic hyperemic phase (Theilen et al., 1993). It is clear from the authors' videorecordings that the same is true in terms of erythrocyte flow. No newly perfused capillaries were detected under hyperemic conditions. However, some capillaries were unperfused by labeled erythrocytes for several seconds (usually because one of the erythrocytes was blocked), and then perfused by numerous erythrocytes flowing at a high speed. The erythrocyte capillary circulation was still very heterogeneous during hyperperfusion, and no privileged pathways could be detected over a long period (several minutes). Some thoroughfare channels were sometimes evident, but they were not included in the analysis of capillaries because their functionality was obviously different.
Role of nitric oxide released from neurons
The reduction by 7-NI of the transient dilatation occurring soon after carotid clamping suggests that NO from neurons participates in autoregulation, thus supporting the work of Kovach et al. (1994) in different experimental conditions.
The mechanisms causing arteriole dilatation during reperfusion after global ischemia have been explored, but they are still not completely understood. In the current study, the significant reduction in the increase in pial arteriole diameter when type I NOS was inhibited by 7-NI, indicates that NO released from neurons participates in these mechanisms. The perivascular nitrergic innervation originating from the sphenopalatine ganglia is the most likely source of NO for two reasons. First, functional nitric oxide-synthesizing perivascular nerves have been demonstrated in the rat middle cerebral artery (Ignacio et al., 1997); second, the theoretical distance that NO can cross is too short for the NO released by partenchymal neurons to have a direct action on surface microvessels.
In the late reperfusion phase, the larger arteriole diameter in 7-NI—treated rats than in control rats may be because of a compensatory vasodilatory factor, indicating that an adaptative mechanism to the increased blood supply was triggered somewhat later than in control rats. The current data also suggest that the NO released from neurons is not involved in BBB permeability and thrombus formation under the current experimental conditions and soon after an ischemic insult.
Under reperfusion, the markedly slower erythrocyte velocity through capillaries in 7-NI-treated rats compared with control rats suggests that NO released from parenchymal neurons plays a major role in the local regulation of capillary circulation (that is, at the parenchymal level) in postischemic conditions. This corroborates what has been found in hypoxemia (Hudetz et al., 1998) and moderate hemodilution (Hudetz et al., 2000) and supports the hypothesis that NO from neurons participates in the local regulation of flow specific to the terminal intraparenchymal microvascular bed. The current data strongly support the involvement of NO from parenchymal neurons in the postischemic vasodilatation. Furthermore, as the postischemic vasodilatation is associated with an increase in the flux and velocity of erythrocytes through capillaries, the subsequent oxygen supply to brain tissue favors parenchymal NO formation, because molecular O2 is a substrate of NO synthesis.
The reduction of the postischemic increase in cerebral microcirculation under type I NOS inhibition by 7-NI shown in the current study may be part of the mechanisms by which 7-NI is neuroprotective in transient global ischemia (Nanri et al., 1998), provided that the microcirculatory reactions are similar in subcortex and hippocampus. The combined reduction in oxygen supply to the tissue and NO production limits the formation of cytotoxic oxidants, such as peroxynitrite, and is thus beneficial for the tissue.
In conclusion, the data obtained in this real-time dynamic investigation of cortical microcirculation in transient global cerebral ischemia lead to a better characterization of the role the microvasculature plays during ischemia and reperfusion. This is of importance in view of the recent interest in strategies aiming at cerebral artery reperfusion in thrombotic or embolic stroke. Furthermore, the demonstration of a partial role of NO from neuronal source in the postischemic tissue hyperemia leads to reconsidering the role of neuronally-derived NO in the consequences of carebral ischemia, taking into account its influence on the microcirculation.
Footnotes
Acknowledgments
The authors thank Robert Charbonné and Odile Issertial for excellent technical assistance and Dr. Philippe Méric for fruitful discussion.
