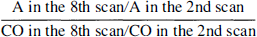Abstract
The aim of this study was to investigate the efficacy of pre-and posttreatment 99mTc-ethyl cysteinate dimer (99mTc-ECD) single photon emission computed tomography (SPECT) for predicting the ischemic outcome of embolic middle cerebral artery occlusion after treatment with local intraarterial thrombolysis. The authors examined 28 patients with a moderately ischemic area (ratio of affected regional activity to cerebellar activity (A/C ratio) of 0.4 to 0.7) determined using pretreatment SPECT, and with complete recanalization within 6 hours. Posttreatment dynamic and static SPECT studies were performed immediately after thrombolysis. The extent of the affected area outlined on pretreatment SPECT was used for the posttreatment SPECT images, and A/C ratios were calculated. The relative retention ratio of 99mTc-ECD in the affected area was also analyzed using posttreatment dynamic SPECT. Fourteen patients either without infarction or with small subcortical and basal ganglial infarction, 11 patients with medium or large cortical infarction, and 3 patients with hemorrhage were identified by follow-up computed tomography. Ischemic outcome correlated with the relative retention ratio of 99mTc-ECD more closely than either the pre-or posttreatment A/C ratios. In particular, a threshold value for the development of hemorrhage was distinct only in the relative retention ratio of 99mTc-ECD. Pretreatment 99mTc-ECD SPECT did not always predict the occurrence of hemorrhagic transformation, whereas dynamic 99mTc-ECD SPECT performed immediately after thrombolysis allowed clear identification of patients at risk for hemorrhagic transformation.
In the treatment of acute ischemic stroke, restoration of cerebral blood flow (CBF) is essential to protect ischemic brain from infarction. Intraarterial thrombolytic therapy has been a useful method for recanalizing occluded arteries (del Zoppo et al., 1988; Hacke et al., 1988; Theron et al., 1989; Zeumer et al., 1993; Jansen et al., 1995; Sasaki et al., 1996; del Zoppo et al., 1998; Ryu et al., 1999; Furlan et al., 1999; Ueda et al., 1999; Kidwell et al., 2000). However, thrombolytic therapy of acute ischemic stroke is associated with significant morbidity and mortality. In particular, the greatest problem reportedly has been hemorrhagic transformation, which may result in rapid deterioration of the patient's neurologic condition (Overgaard et al., 1990; del Zoppo et al., 1998; Furlan et al., 1999; Ueda et al., 1999).
The reversibility of ischemic brain tissue is known to depend on the intensity and duration of ischemia (Astrup et al., 1981; Jones et al., 1981; Weinstein et al., 1986). The former is related to residual blood flow, whereas the latter is related to the timing of treatment. Noninvasive brain perfusion imaging modalities such as single photon emission computed tomography (SPECT), positron emission tomography, and magnetic resonance imaging are expected to have potential value in the assessment of tissue reversibility before treatment (Giubilei et al., 1990; Hanson et al., 1993; Baird et al., 1994; Grotta and Alexandrov, 1998; Heiss et al., 1998; Jansen et al., 1999; Marks et al., 1999; Ryu et al., 1999; Kidwell et al., 2000). Several retrospective studies using SPECT have established the CBF thresholds for the development of infarction or hemorrhage, and the results have suggested that pretreatment SPECT can provide useful parameters to increase the efficacy of thrombolysis by reducing hemorrhagic complications (Ezura et al., 1996; Sasaki et al., 1996; Ueda et al., 1999). However, no studies to date have reported on the ischemic and neurologic outcomes of patients selected based on the pretreatment CBF assessment who underwent local intraarterial thrombolysis. Moreover, studies regarding the relation between ischemic outcome and SPECT findings have exclusively used the tracer 99mTc-hexamethylpropyleneamine oxime (99mTc-HMPAO), which assesses cerebral blood flow (Giubilei et al., 1990; Hanson et al., 1993; Baired et al., 1994; Ezura et al., 1996; Sasaki et al., 1996; Grotta and Alexandrov, 1998; Ueda et al., 1999; Ryu et al., 1999). In contrast, the tracer 99mTc-ethyl cysteinate dimer (99mTc-ECD) reflects not only perfusion but also the metabolic status of brain tissue (Lassen and Sperling, 1994; Nakagawara et al., 1994; Devous, 1995; Shishido et al., 1995; Jacquier-Sarlin et al., 1996). In a study with a time window of 6 hours after the onset of symptoms, 99mTc-ECD SPECT was used to produce a clearer distinction between transient cerebral ischemia and ischemic infarction (Berrouschot et al., 1998). In addition, 99mTc-ECD has been suggested to be a potential agent with which to evaluate neural tissue viability early after reperfusion of cerebral ischemia (Ogasawara et al., 1999).
The aim of the current study was to investigate the efficacy of pre-and posttreatment 99mTc-ECD SPECT for predicting the ischemic outcome of patients in which a moderately ischemic area because of embolic middle cerebral artery occlusion was detected. All cases showed complete recanalization after local intraarterial thrombolysis.
MATERIALS AND METHODS
The authors investigated 34 selected cases involving patients treated at our institution between January 1995, and September 1996, using local intraarterial thrombolysis (LIT) for acute embolic stroke. Of the 34 patients, 28 who had complete recanalization within 6 hours after the onset of symptoms and were studied with both pre-and posttreatment SPECT were enrolled in the current study. The patients consisted of 8 women and 20 men between 40 and 73 years of age (mean ± SD, 59 ± 11 years).
The inclusion criteria for LIT at the authors' institution were as follows: age less than 75 years; no regions of hypodensity observed on computed tomography scan (CT) upon admission; a mean radioactivity in the entire ischemic area obtained by pretreatment 99mTc-ECD SPECT after an initial CT of between 40% and 70% compared with that in the ipsilateral cerebellum; sites of occlusion in the middle cerebral artery suggested by symptoms; a diagnosis of embolic occlusion made according to the guidelines of the Cerebral Embolism Task Force (1986) based on the onset pattern, angiographic findings, and results of cardiovascular examinations such as electrocardiography and echocardiography; the patient was treated within 6 hours after the onset of symptoms; and informed consent was obtained from the patient or relatives. The exclusion criteria were as follows: recent hemorrhagic stroke; critical systemic condition; serious disabling disease; and classic contraindications to thrombolytic therapy (recent surgery, gastrointestinal bleeding, or known bleeding tendency).
Local intraarterial thrombolysis was performed using recombinant tissue plasminogen activator (rtPA). Arterial digital subtraction angiography was performed by the Seldinger method using a 5-French catheter and a 6-French femoral sheath. The tip of a Tracker-18 catheter (Target Therapeutics, Fremont, CA, U.S.A.) was advanced into the thrombus or upstream from the site of occlusion over a 0.356-cm (0.014-inch) Taper Dasher guide wire (Target Therapeutics). RtPA (1,600,000 IU) was dissolved in 20 mL of physiologic saline and then injected manually for approximately 10 minutes. Angiography was performed by injection of contrast material through the Tracker-18 catheter immediately after each infusion. This was repeated until recanalization of the occluded vessel was confirmed. The maximum dose of rtPA was 4,800,000 IU. The patients were managed initially by administration of 20% mannitol (500 mL), phenytoin (500 mg), and vitamin E (500 mg) during the therapy.
Single photon emission computed tomography images were obtained using a multidetector ring-type scanner, of which the detector array consists of 64 NaI crystals in a 38-cm-diameter circle. After tomographic reconstruction, the spatial resolution and slice thickness in the center of the plane were 9 mm and 16 mm full width at half-maximum, respectively, for static imaging and 20 mm and 25 mm, respectively, for dynamic imaging. The energy window in this study was 140 KeV (±15%). Projection data for static and dynamic imaging were processed with Ramachandran's filtered backprojection after introduction of a Butterworth prefilter. A 64 × 64 (99mTc-ECD static) or 32 × 32 (99mTc-ECD dynamic) image matrix was used.
Patients were injected with the tracer in a quiet, dimly lit room while resting with their eyes open. The patient's head was immobilized with a ready-made plastic headholder so that bilateral external auditory meatuses were aligned to the machine indwelling positioning crossed-light beam. To reproduce the same head position during the pre-and posttreatment studies, three markers were affixed on the head under the guidance of a light beam. Of the three markers, two were placed on the anterior edge of the bilateral external auditory meatuses, with the remaining one on the midline of the nose ridge. The three markers lay along a plane perpendicular to the axis of camera rotation.
Pretreatment SPECT imaging with a high-resolution collimator proceeded 10 minutes after intravevous injection of 720 MBq 99mTc-ECD. Tomographic data were continuously obtained over a 20-minute period. Of the six axial slices obtained, the slice most clearly demonstrating the ischemic region was selected. Regions of interest (ROIs) were drawn by hand both in the apparent entire ischemic area based on qualitative inspection and in the entire cerebellar hemisphere on the ischemic side. The mean count was determined in each region of interest. The ratio of affected regional activity (A) to cerebellar activity (C; A/C ratio) was calculated to evaluate the residual CBF in the entire ischemic region before LIT.
Posttreatment SPECT was performed immediately after LIT. Simultaneously with an additional intravenous bolus injection of 900 MBq 99mTc-ECD, dynamic scanning with a high-sensitivity collimator began and continued for a total of 288 seconds (8 scans) with a scan time duration of 36 seconds. Subsequently, static SPECT was performed using the same procedure as the pretreatment study. Before posttreatment SPECT study, baseline static and dynamic SPECT was performed (Fig. 1). After reconstruction, the baseline images were subtracted from the images of posttreatment SPECT study, after correcting for time differences in data acquisition to exclude any influence of the tracer retained in the brain on the pretreatment study and to obtain “true” posttreatment 99mTc-ECD SPECT images. Of the six axial slices obtained, the slice corresponding to the slice chosen in pretreatment SPECT images was selected, and ROIs identical with those of pretreatment SPECT were visually and manually set to the posttreatment SPECT images by two observers blind to the clinical characteristics and CT findings. The mean count was determined in each ROI, and the A/C ratio was calculated in posttreatment static SPECT images. Furthermore, the second and eighth of eight posttreatment dynamic scans were selected. The relative retention ratio in the affected area was calculated as follows:
where A is the mean count in the ROIs on the affected side, and CO is the mean count in the corresponding region on the contralateral side.
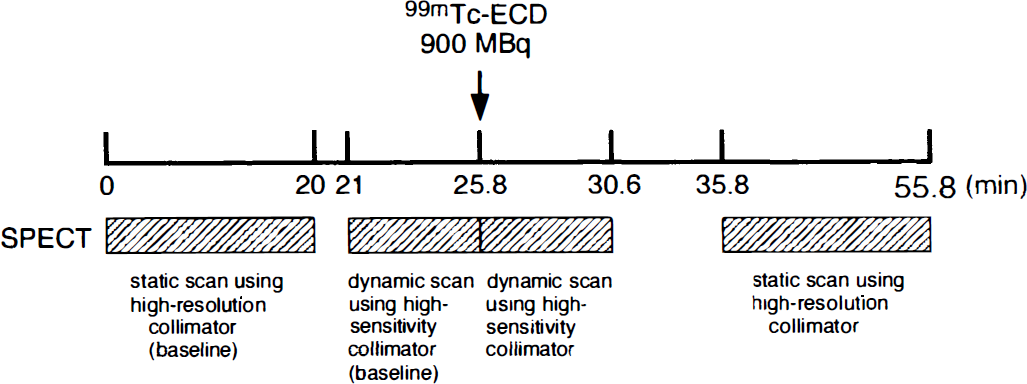
Protocol for 99mTc-ethyl cysteinate dimer (99mTc-ECD) single photon emission computed tomography (SPECT) imaging after thrombolysis.
A CT scan was performed immediately after therapy, the next day, 1 week, 2 weeks, and 1 month after therapy. The patient's neurologic status was evaluated upon admission, the next day, and 1 month after treatment, according to the National Institutes of Health (NIH) Stroke Scale (Brott et al., 1989), which expresses the severity of neurologic status numerically from 0 (normal) to 42. An NIH score of 42 points was assigned to patients who had died within 1 month of treatment.
For statistical analysis, the values were expressed as the mean ± SD, and differences among the three groups were examined by Scheffe's F test. Statistical significance was set at P < 0.05.
RESULTS
Based on the follow-up CT, the patients were classified into three groups: (A) 14 patients with no evidence of infarction, small subcortical infarctions, or basal ganglial infarctions (reversible ischemia); (B) 11 patients with medium-size or large cortical infarctions; and (C) 3 patients with hemorrhagic transformation.
Clinical characteristics, treatment protocol, SPECT findings, and outcomes are summarized in Table 1. The pretreatment NIH score statistically differentiated the hemorrhagic group from the reversible ischemia group and cortical infarction groups. However, the threshold value between the pretreatment NIH score for the development of cortical infarction and that of hemorrhage was not distinct. Neurologic outcome according to the NIH score differed significantly among the three patient groups. The hemorrhagic group were older in terms of mean age and received a larger mean rtPA dose than the other two groups. However, age, duration of ischemia, rtPA dose, and site of occlusion did not differ significantly among the three groups.
Patient data, treatment protocol, SPECT findings, and outcome
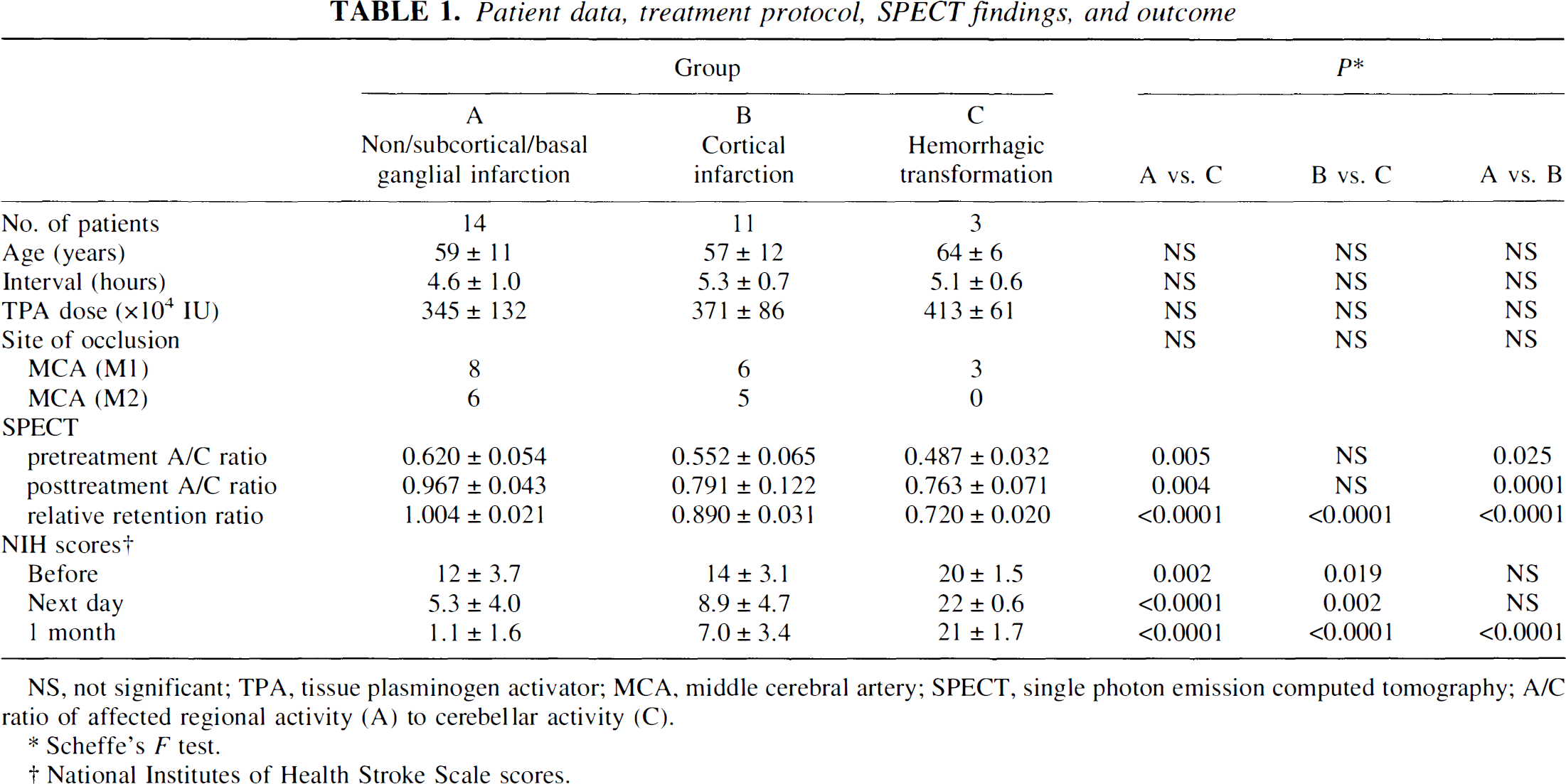
NS, not significant; TPA, tissue plasminogen activator; MCA, middle cerebral artery; SPECT, single photon emission computed tomography; A/C ratio of affected regional activity (A) to cerebellar activity (C).
Scheffe's F test.
National Institutes of Health Stroke Scale scores.
The A/C ratio obtained by pretreatment SPECT statistically differentiated the reversible ischemia group from the cortical infarction and hemorrhagic groups. The mean value was less in the hemorrhagic group than in the cortical infarction group. However, the difference between the two groups in terms of ischemic duration between 3 and 6 hours was not statistically significant. Even when patients were separated into two subgroups according to a duration of ischemia cutoff value of 4.5 hours, the difference in the pretreatment A/C ratio between the cortical infarction and hemorrhagic groups did not reach the level of significance, and it was impossible to identify a threshold value for the development of hemorrhage (Fig. 2).
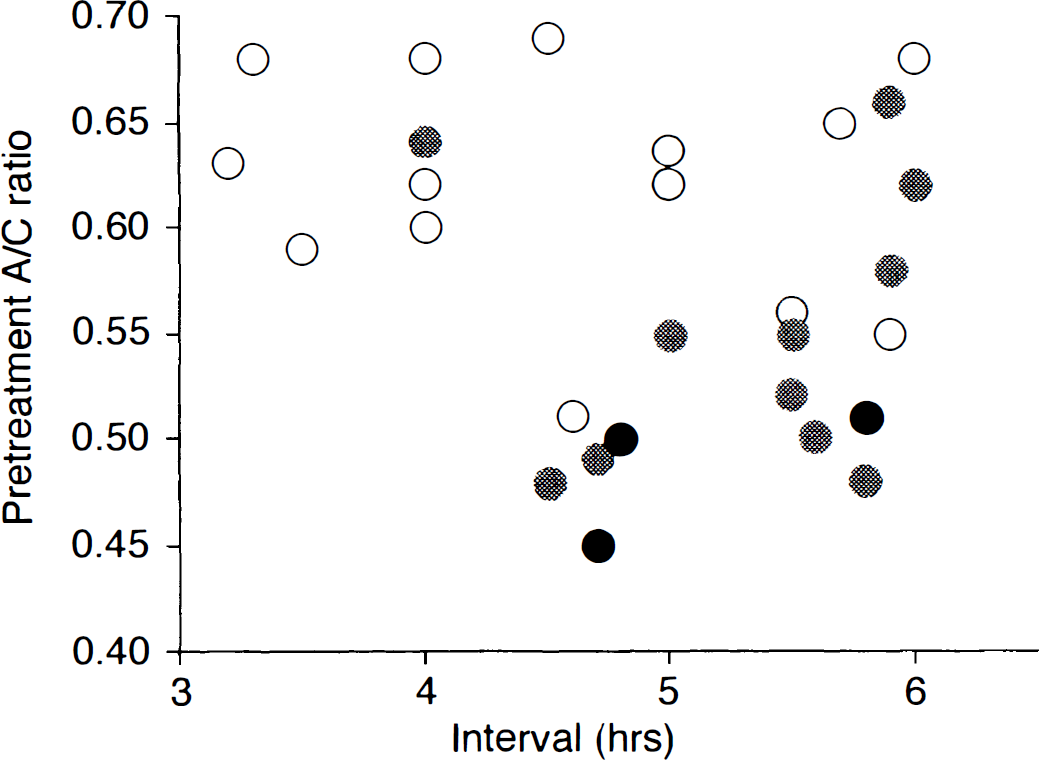
Outcome versus duration and severity of ischemia, which was obtained by pretreatment 99mTc-ECD SPECT. In patients with relatively high pretreatment cerebral blood flow (CBF; ratio >0.55), hemorrhage does not develop. However, in patients with low pretreatment CBF (<55% of cerebellar CBF), the threshold between pretreatment CBF for the development of infarction and that of hemorrhage is not distinct. Open circles represent non/subcortical/basal ganglial infarction. Shaded circles represent cortical infarction. Filled circles represent hemorrhage.
The A/C ratio obtained by posttreatment static SPECT statistically differentiated the reversible ischemia group from both the cortical infarction and hemorrhagic groups. However, the observed threshold value for the development of cortical infarction was not distinct (Fig. 3). Furthermore, the difference between the cortical infarction and hemorrhagic groups was not statistically significant.
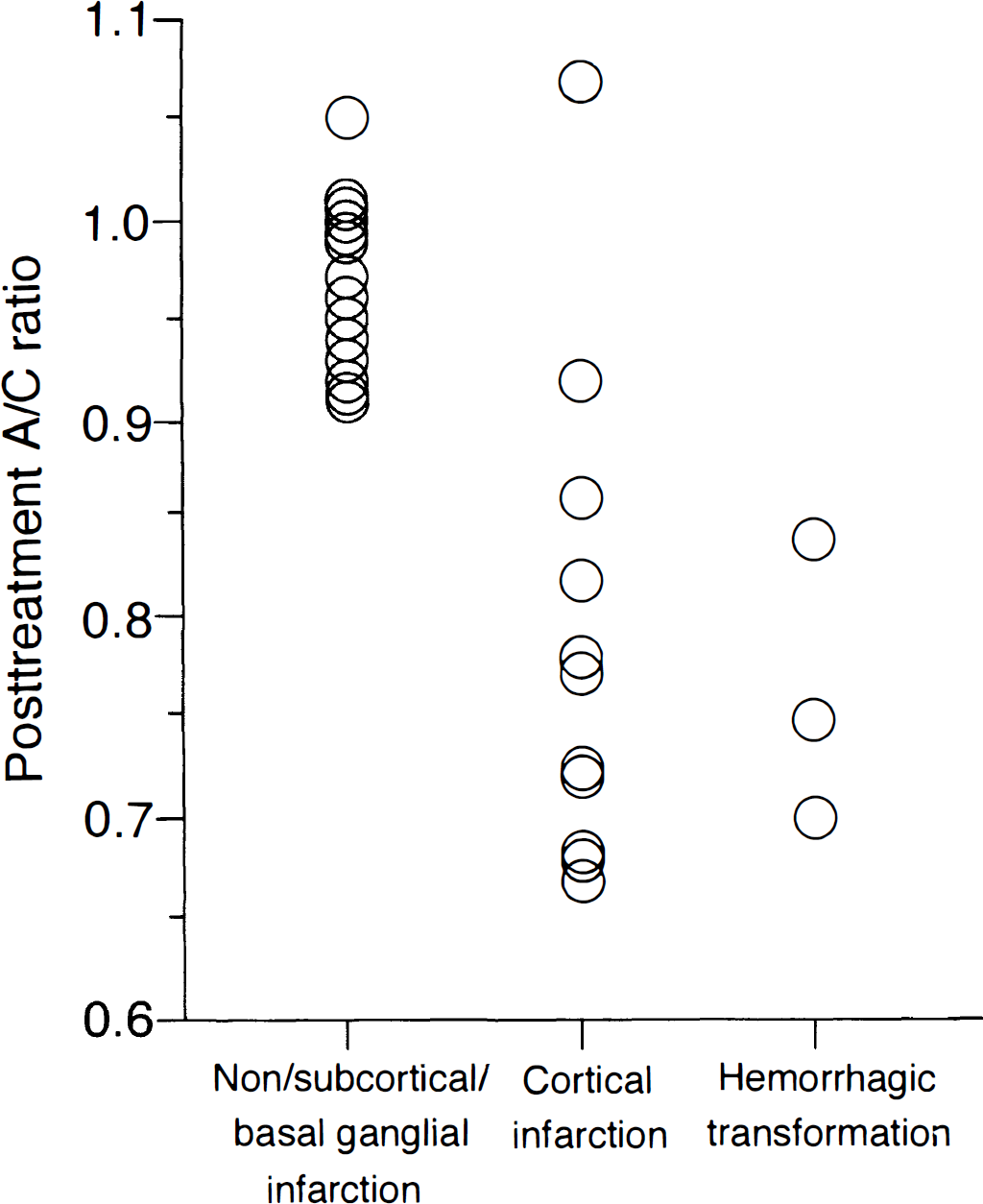
Outcome versus relative counts in the reperfused area, which was obtained by posttreatment static 99mTc-ECD SPECT. The thresholds for the development of cortical infarction or hemorrhage are not distinct.
The relative retention ratio of 99mTc-ECD in the affected area obtained by posttreatment dynamic SPECT statistically differentiated all three groups. Ischemic outcome depended on the relative retention ratio. The thresholds for the development of infarction or hemorrhage were 0.95 and 0.75 of the relative retention ratio, respectively (Fig. 4). Representative images in cases of hemorrhagic transformation are shown in Fig. 5.
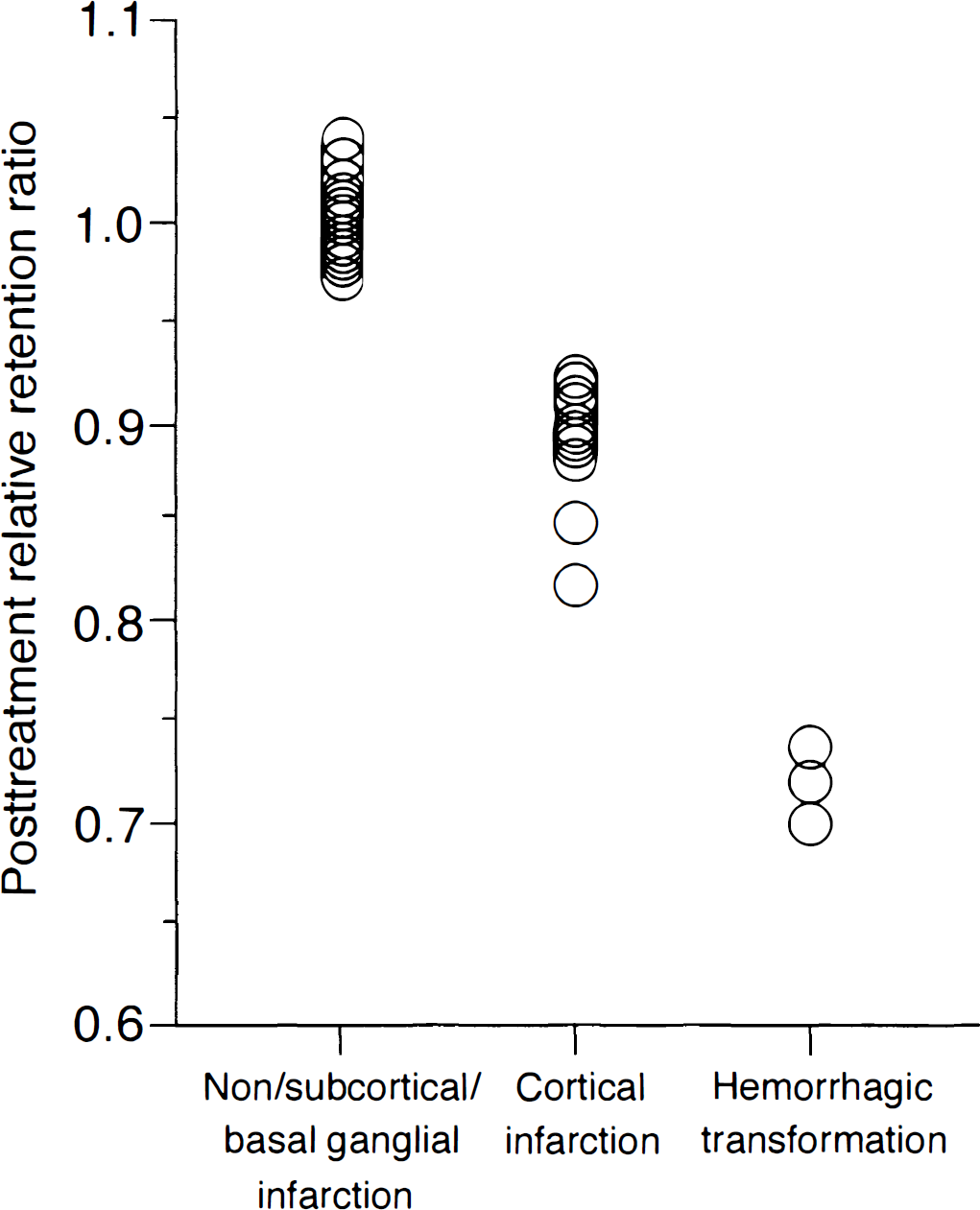
Outcome versus relative retention ratio in the reperfused area, which was obtained by posttreatment dynamic 99mTc-ECD SPECT. Relative retention ratio clearly identifies ischemic outcome. The thresholds for the development of infarction or hemorrhage are 0.95 and 0.75 of relative retention ratio, respectively.
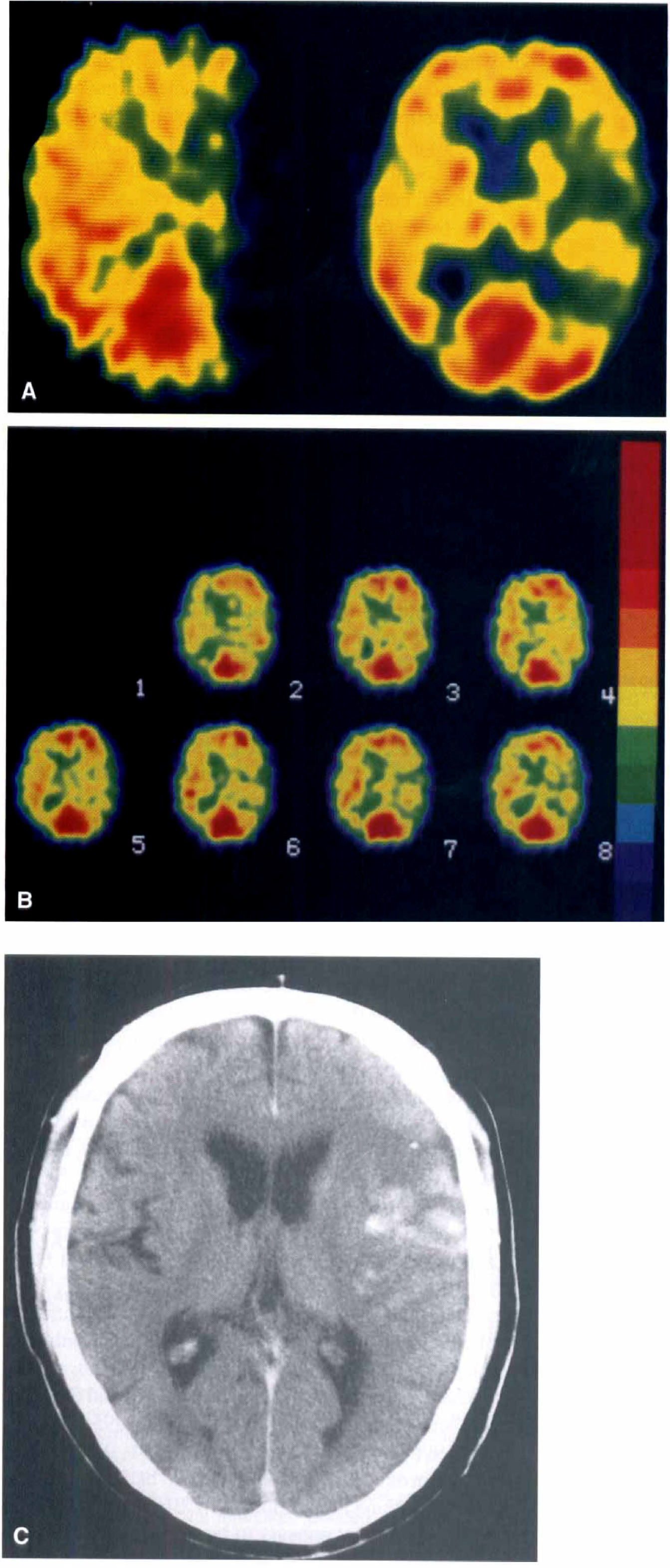
A 70-year-old man with complete recanalization of the left middle cerebral artery occlusion 4.7 hours after onset of symptoms.
DISCUSSION
The current findings demonstrated that CBF parameters obtained by pretreatment 99mTc-ECD SPECT can be used to differentiate patients with reversible ischemia from those with irreversible brain damage. However, it was impossible to identify the threshold value for the development of hemorrhage. The results also showed that ischemic outcomes depended on the relative retention ratio of 99mTc-ECD in the affected area obtained by posttreatment dynamic SPECT, and that the threshold values for the development of cortical infarction or hemorrhage were distinct.
Emergency treatment has to date focused on the first 6 hours after the onset of ischemic stroke symptoms. It is generally believed that the 6-hour period provides a therapeutic window of opportunity during which medical intervention can be initiated to limit brain damage (Ueda et al., 1999). Within this therapeutic window, it is presumed that most ischemic tissue remains viable, and thus theoretically can be revived. However, in recent studies using local intraarterial thrombolytic therapy, not all of the patients had excellent ischemic outcomes, with significant hemorrhagic complications occurring even in those treated within the therapeutic window (Ezura et al., 1996; Sasaki et al., 1996; del Zoppo et al., 1998; Furlan et al., 1999; Ueda et al., 1999). Although many factors, aside from the duration of ischemia, that could possibly influence ischemic outcome have been proposed, the results suggested that the pretreatment CBF of ischemic tissue plays the most important role in influencing ischemic outcome, including development of hemorrhagic complications. In addition, the parameters provided by pretreatment SPECT are useful in assessing the reversibility of ischemic tissue (Ezura et al., 1996; Sasaki et al., 1996; Grotta and Alexandrov, 1998; Ryu et al., 1999; Ueda et al., 1999). Several retrospective studies have demonstrated that within 6 hours after the onset of symptoms, the threshold value for the development of hemorrhage was likely to be approximately 0.4 of residual relative regional CBF (Ezura et al., 1996; Sasaki et al., 1996; Ueda et al., 1999). In the current study, only patients verified to have a moderately ischemic area as determined by an A/C ratio between 0.4 and 0.7, based on the pretreatment CBF assessment, were selected for local intraarterial thrombolytic therapy. The current data showed that hemorrhage occurred in 11% (3 of 28) of patients. The rate of hemorrhagic complication was less (22% to 43%) compared with that of patients who underwent local intraarterial thrombolysis regardless of pretreatment CBF, with complete recanalization obtained within 6 hours after the onset of symptoms (Sasaki et al., 1996; Furlan et al., 1999; Ueda et al., 1999). These findings suggest that pretreatment 99mTc-ECD SPECT provides useful parameters to increase the efficacy of thrombolysis by reducing hemorrhagic complications. However, the authors' result of an indistinct threshold between pretreatment CBF for the development of infarction and that of hemorrhage was inconsistent with previous findings (Ezura et al., 1996; Ueda et al., 1999). Possible sources of this inconsistency are a difference in the tracers used for pretreatment CBF assessment, the methods of establishing ROIs, and the number of enrolled patients. However, the authors consider the most plausible cause of an indistinct threshold between pretreatment CBF for the development of infarction and that of hemorrhage to be the following: the regional CBF, after sudden occlusion of the main trunk of the cerebral artery, is not constant and changes in a nonspecific manner depending on the efficacy of collateral blood supply through the meningo-pial anastomosis (Heiss et al., 2000). In addition, fragmentation and migration of emboli have been documented clinically and experimentally (Bladin, 1964; Liebeskind et al., 1971; Kaneko et al., 1985). Single photon emission computed tomography images can show moderate ischemia in the affected side in a patient with severe ischemia due to embolic internal carotid artery occlusion at the onset, when migration of the embolus into the trunk of the middle cerebral artery has occurred before pretreatment SPECT study. Therefore, CBF data obtained by pretreatment SPECT does not always reflect average perfusion from onset to recanalization. The authors believe that there is a limit to the degree to which they can prospectively identify patients at risk for hemorrhage after local intraarterial thrombolysis using pretreatment SPECT.
Interestingly, in the current study, the threshold value for the development of hemorrhage was not distinct in posttreatment static 99mTc-ECD SPECT images, but was distinct in posttreatment dynamic 99mTc-ECD SPECT images. 99mTc-ECD is transformed by intracellular esterases from a diester into a diacid complex. This polar metabolite does not cross the cell membrane and therefore is trapped in the neural cell (Walovitch et al., 1989; Jacquier-Sarlin et al., 1996). The brain distribution of 99mTc-ECD in static SPECT thus reflects not only perfusion but also the activity of cytosolic esterase, namely the metabolic status of brain tissue (Lassen and Sperling, 1994; Nakagawara et al., 1994; Devous, 1995; Shishido et al., 1995; Jacquier-Sarlin et al., 1996). Although all patients enrolled in the current study had angiographically complete recanalization, the CBF in the reperfused area in each patient appears to be respectively different. Therefore, it is impossible to judge which of the above two factors more strongly affects the focally decreased uptake of 99mTc-ECD in each static SPECT image. However, the relative retention ratio of 99mTc-ECD obtained by dynamic SPECT is presumably influenced only by the activity of cytosolic esterase and the severity of blood–brain barrier breakdown. The extremely decreased relative retention ratio of 99mTc-ECD in the reperfused area thus indicates ongoing irreversible severe brain damage, which in turn leads to hemorrhagic transformation.
Reliable information on the prognosis of an ischemic area after local intraarterial thrombolysis is urgently required as hemorrhagic transformation may result in rapid deterioration of the patient's neurologic condition (Overgaard et al., 1990; del Zoppo et al., 1998; Furlan et al., 1999; Ueda et al., 1999). Specific, but potentially dangerous, treatments such as decompressive hemicraniectomy (Delashaw et al., 1990) or mild hypothermia (Schwab et al., 1997) appear to be more appropriate for those patients. In cases in which a diagnosis of hemorrhagic transformation is confirmed early after thrombolysis, the patient should be transferred immediately to an intensive care unit or to any specially equipped center where decompressive hemicraniotomy or mild hypothermia can be performed (Berrouschot et al., 1998). Dynamic 99mTc-ECD SPECT performed immediately after local intraarterial thrombolysis enables accurate identification of patients at risk for hemorrhagic transformation, which in turn allows clinicians to immediately initiate treatment that may be specific for increasing intracranial pressure.
The procedure used for dynamic SPECT imaging in the current study is accomplished in 10 minutes, including the baseline study, and is not uncomfortable even for critical patients. In recent years, a multihead gamma camera with high sensitivity and spatial resolution has been developed. Such advances will enable dynamic SPECT images to be obtained routinely in many institutions in the near future.
In conclusion, pretreatment 99mTc-ECD SPECT does not always predict the occurrence of hemorrhagic transformation, whereas dynamic 99mTc-ECD SPECT performed immediately after thrombolysis allows for clear identification of patients at risk for hemorrhagic transformation.