Abstract
The protein kinase Akt/PKB has been implicated in antiapoptosis and neuronal survival. The authors now show that Akt is phosphorylated in the hippocampus during the early reperfusion period after 3.5 minutes bilateral carotid artery occlusion (BCAO) in the gerbil. Repeated sublethal ischemia induces ischemic tolerance, which is known as ischemic preconditioning. Ischemic preconditioning does not affect the amount of Akt protein, but rather decreases the phosphorylation of Akt at Ser-473 after 10 minutes reperfusion after 3.5 minutes BCAO. These results suggest that although Akt may play a role in neuronal survival after ischemia, it may not play a role in ischemic tolerance by preconditioning.
The serine–threonine kinase Akt, also referred to as protein kinase B (PKB) and RAC, is the cellular homologue of the viral oncogene v-Akt (Franke et al., 1995, 1997; Burgering and Coffer, 1995). This protein kinase is activated by insulin and various growth factors, and is mediated by phosphoinositide 3-kinase (PI 3-kinase), a kinase that is inhibited by Wortmannin (Burgering and Coffer, 1995; Franke et al., 1995). Akt is activated by phospholipid binding and activation loop phosphorylation on Thr-308 by phosphatidylinositol (3,4,5)-trisphosphate-dependent kinase 1 (PDK1;Alessi et al., 1996) and within the C-terminus on Ser-473 by integrin-linked kinase (ILK;Delcommenne et al., 1998). Akt has been shown to promote cell survival by phosphorylating and inactivating Bad and caspase-9 on Ser-136 and Ser-196, respectively, thereby inhibiting apoptosis (Kennedy et al., 1997; Datta et al., 1997; del Peso et al., 1997; Cardone et al., 1998).
Although the role of Akt/PKB has been studied extensively with inhibiting apoptosis, there have been limited studies on the involvement of Akt in neuronal survival after ischemia. Kitagawa et al. (1999) reported changes in immunostaining of total-Akt in rat using the permanent middle cerebral artery occlusion model. They reported that Akt immunostaining was transiently increased at 3 to 8 hours in the ischemic penumbral region. Ouyang et al. (1999) demonstrated that Akt is phosphorylated at Ser-473 in the hippocampus during the early stages of the reperfusion period after 15 minutes forebrain ischemia in rats. These studies suggested that Akt has a neuroprotective role after ischemia.
Pretreatment with sublethal ischemia protects the CA1 pyramidal cells from subsequent lethal ischemia, which is well known as ischemic tolerance phenomenon by preconditioning (Kitagawa et al., 1990; Liu et al., 1992). Several molecules, such as heat shock proteins (Kirino et al., 1991; Liu et al., 1992), adenosine, and ATP-sensitive K+ channels (Heurteaux et al., 1995; Kawahara et al., 1998) have been implicated in cerebral ischemic preconditioning
MATERIALS AND METHODS
Ischemia model
Eighty-one male Mongolian gerbils (50 to 70 g) were used in the current study. Gerbils were anesthetized in 2.0% halothane in 70% N2O and 30% O2 using a vaporizer (Halowick, Muraco Medical, Tokyo, Japan). Transient forebrain ischemia was produced by transient bilateral carotid artery occlusion (BCAO) with miniclips in gerbils. Core and temporal muscle temperature during surgery were maintained at 37°C with a thermostat and a heating pad (NS-TC, Neuroscience, Tokyo, Japan; and BAT-12, Physitemp, NJ, U.S.A.). Animals were monitored in a 32°C incubator (Thermocare, Incline Village, NV, U.S.A.) for 3 hours after surgery. Animal protocols followed the National Cardiovascular Center's guidelines for animal care and experiments.
Immunoblotting
To collect brain tissues for immunoblotting, gerbils were deeply anesthetized with overdose of pentobarbital and decapitated. Brain tissues were dissected and quickly frozen in liquid nitrogen. Isolated total hippocampal tissues from both sides were pooled and homogenized in 1 mL of potassium phosphate buffer (10 mmol/L KPO4 (pH 7.05), 1 mmol/L ethylenediaminetetraacetic acid (EDTA), 5 mmol/L ethylene glycol-bis[β-aminoethyl ether] (EGTA), 10 mmol/L magnesium chloride, 50 mmol/L β-glycerophosphate, 1 mmol/L sodium vanadate, 1 mmol/L dithiothreitol (DTT), 0.5% NP-40, 0.1% Brij-35), and centrifuged at 14,000 ×g for 10 minutes. Protein concentration was determined by the Bradford assay (5000–006; Bio-Rad, Hercules, CA, U.S.A.). To evaluate phosphorylation of Akt, 40 μg of total lysate of each sample was loaded on a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel, electrophoresed, and transferred onto Immobilon-P membrane (Millipore, Bedford, MA, U.S.A.). Western blotting was performed as previously described (Alessandrini et al., 1999). The authors used the phospho-specific Akt (Ser-473) antibody (1:1000; New England Biolabs, Beverly, MA, U.S.A.) to detect the phosphorylated form of Akt. Immunoblots were visualized using the ECL immunodetection system kit (Amercham, Arlinton Heights, IL, U.S.A.). To analyze protein levels of Akt, the immunoblot was stripped and reprobed with total-Akt antibody that recognizes Akt independent of the phosphorylation state (1:1000; New England Biolabs). Relative phosphorylation levels of Akt were determined from densitometric scanning of a total of 5 immunoblots film produced by 5 independent experiments. Relative phosphorylation levels in sham control gerbils were given a value of 1. Differences between relative phosphorylation levels over time were determined by one-way analysis of variance followed by Bonferroni's post hoc test. Differences in total-Akt and phospho-Akt levels between preconditioned and sham-operated gerbils (n = 4 in each) were determined by unpaired Student's t-test. P < 0.05 was considered statistically significant.
Immunostaining
The animals were killed by transcardial perfusion with 4% paraformaldehyde in 0.1 mol/L phosphate buffered saline (PBS, pH 7.4). Brains were removed quickly and kept in the same fresh buffer containing 20% sucrose. Brains were cut into coronal sections 40-μm-thick on a freezing microtome (HM400R; Microm, Walldorf, Germany). The sections were successively treated with 0.1% H2O2 in 0.1 PBS, 5% normal goat serum in PBS (pH 7.4) containing 0.1% Tween-20 for 30 minutes, and primary antibody (1:100 for phospho-specific Akt (Ser-473) antibody or 1:500 for total-Akt antibody, respectively) in PBS (pH 7.4) containing 2% normal goat serum and 0.1% Tween-20 at 4°C overnight. After they were washed three times in 0.1 mol/L PBS (pH 7.4), the sections were incubated with biotinylated goat anti-rabbit IgG (Vector Laboratories, Burlingame, CA, U.S.A.) in PBS (pH 7.4) containing 2% normal goat serum and 0.1% Tween-20 for 2 hours at room temperature, and then immunoreactive products were detected by an avidin-biotin-peroxidase complex technique using 0.005% H2O2 and 3,3′-diaminobenzidine tetrahydrochloride (DAB; Sigma, St. Louis, MO, U.S.A.) as chromogen. Four brains were examined in each experimental group, and immunostaining was repeated twice in each brain.
RESULTS
To evaluate the relative changes in total-Akt protein levels and the phosphorylation state of Akt after 3.5 minutes BCAO, the authors performed Western blot analysis using total hippocampal tissues isolated at various reperfusion time points. Akt was dephosphorylated at the end of 3.5 minutes of BCAO, which might be because of decreased ATP production during ischemia. This dephosphorylation was sustained for 1 minute after reperfusion. A significant increase in phospho-Akt was observed by 10 minutes after reperfusion and persisted until 1 hour compared with sham control (Fig. 1A). This was confirmed by densitometric analysis of immunoblots (n = 5 independent experiments;Fig. 1B). Phosphorylation levels returned to control levels by 1 day after reperfusion (Figs. 1A and 1B). Total-Akt protein levels in the hippocampus did not change appreciably (Fig. 1A). The current findings are in agreement with those reported in rats by Ouyang et al. (1999).
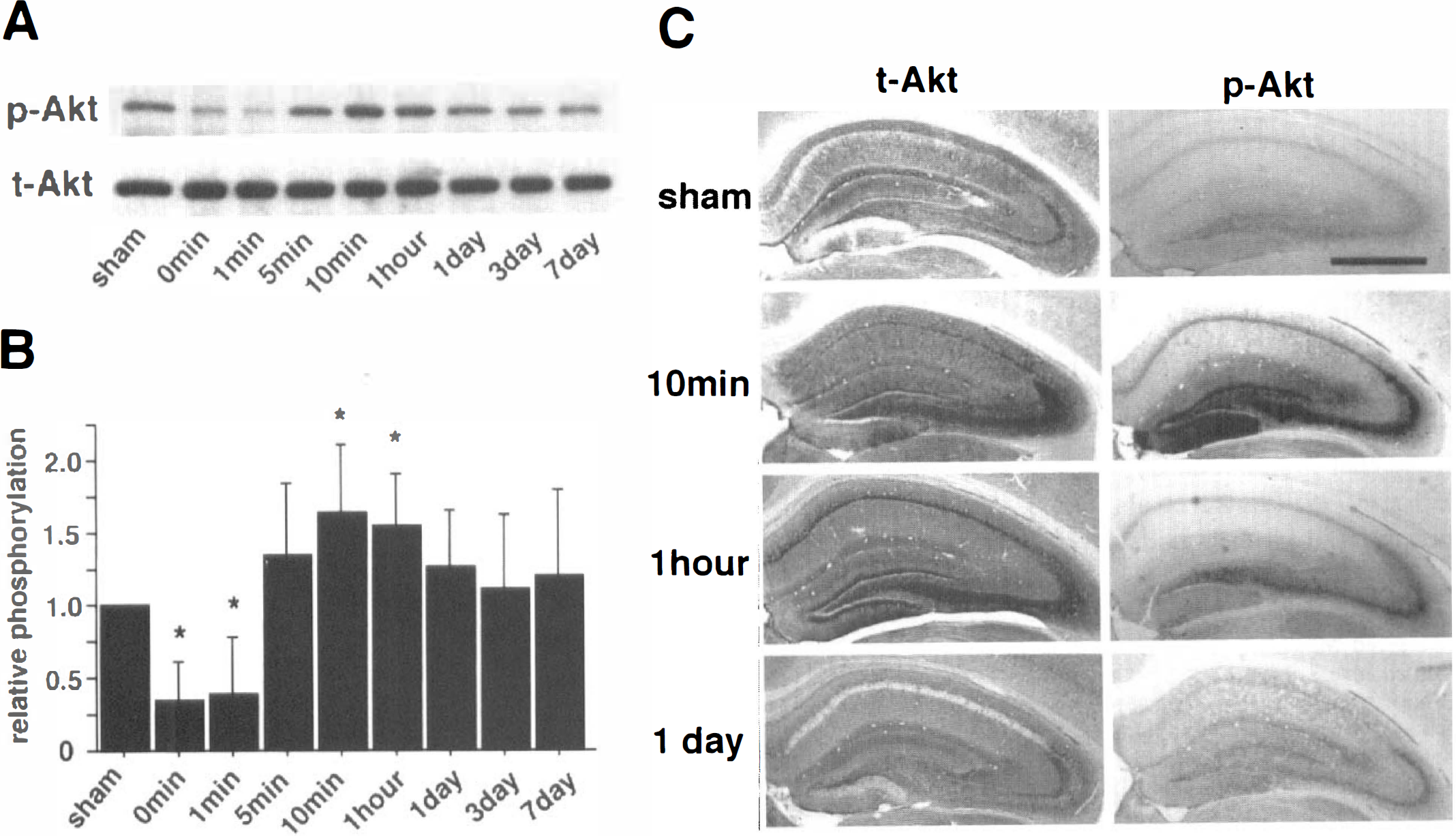
Changes in Akt in the gerbil hippocampus after 3.5 minutes bilateral carotid artery occlusion.
Next, the authors examined the changes in total-Akt immunostaining and phospho-Akt immunostaining in the hippocampus after 3.5 minutes BCAO. Akt immunostaining was seen throughout the hippocampus in sham-operated animals (Fig. 1C). Akt immunoreactivity was increased in the pyramidal cell layer of CA1 10 minutes after reperfusion, remained high at 1 hour, and returned to the control levels by 3 hours (data not shown). This suggests that transient upregulation in Akt protein levels may contribute to CA1 pyramidal cell survival during the initial 1 hour of reperfusion. The authors observed similar changes in Akt immunostaining in the CA3 and dentate gyrus. By 1 day, Akt immunoreactivity was decreased below the control levels in the pyramidal cell layer of CA1 (Fig. 1C). Phospho-Akt immunostaining increased in the hippocampus 10 minutes after reperfusion, which was more dramatic in the dentate gyrus and CA3 than in the CA1. The staining observed in the CA3 and hilar region was high at 1 hour and then returned to basal levels by 1 day (Fig. 1C). Together with the findings from immunoblotting, these results raise the possibility that Akt may delay the death of CA1 pyramidal cells by upregulation of Akt expression and activation of Akt by phosphorylation. This was also postulated by Ouyang et al. (1999).
Finally, the authors addressed whether Akt is involved in ischemic tolerance by repeated sublethal ischemia. Preconditioning has been shown to protect tissue from cell damage after subsequent ischemic insults. Therefore, the authors wanted to study the role of Akt in preconditioning. The authors examined the phosphorylation levels of Akt and expression of total-Akt protein in the hippocampus between sham-operated and ischemic preconditioned gerbils treated by repeated 2-minute BCAO with a 1-day interval between the two 2-minute pretreatments and a 2-day interval between the final pretreatment and the 3.5 minutes BCAO. Because the phosphorylation levels of Akt reached maximum at 10 minutes after reperfusion following 3.5 minutes BCAO (Fig. 1), the authors compared phospho-Akt and total-Akt between sham-operated and preconditioned gerbils by immunoblotting and immunostaining at 10 minutes after reperfusion following 3.5 minutes BCAO. In gerbils previously exposed to repeated 2-minute BCAO, phosphorylation levels of Akt were much less than in control gerbils that were pretreated with sham operations (Fig. 2). Densitometric analysis of immunoblots showed that preconditioning resulted in a 59% decrease (P < 0.05; n = 4) in phospho-Akt when compared with control. However, ischemic preconditioning did not affect total-Akt protein levels (Fig. 2). These results indicate that Akt does not play a major role in ischemic tolerance, and that molecular events either upstream or independent of Akt activation, or both, may be modulated by ischemic preconditioning.
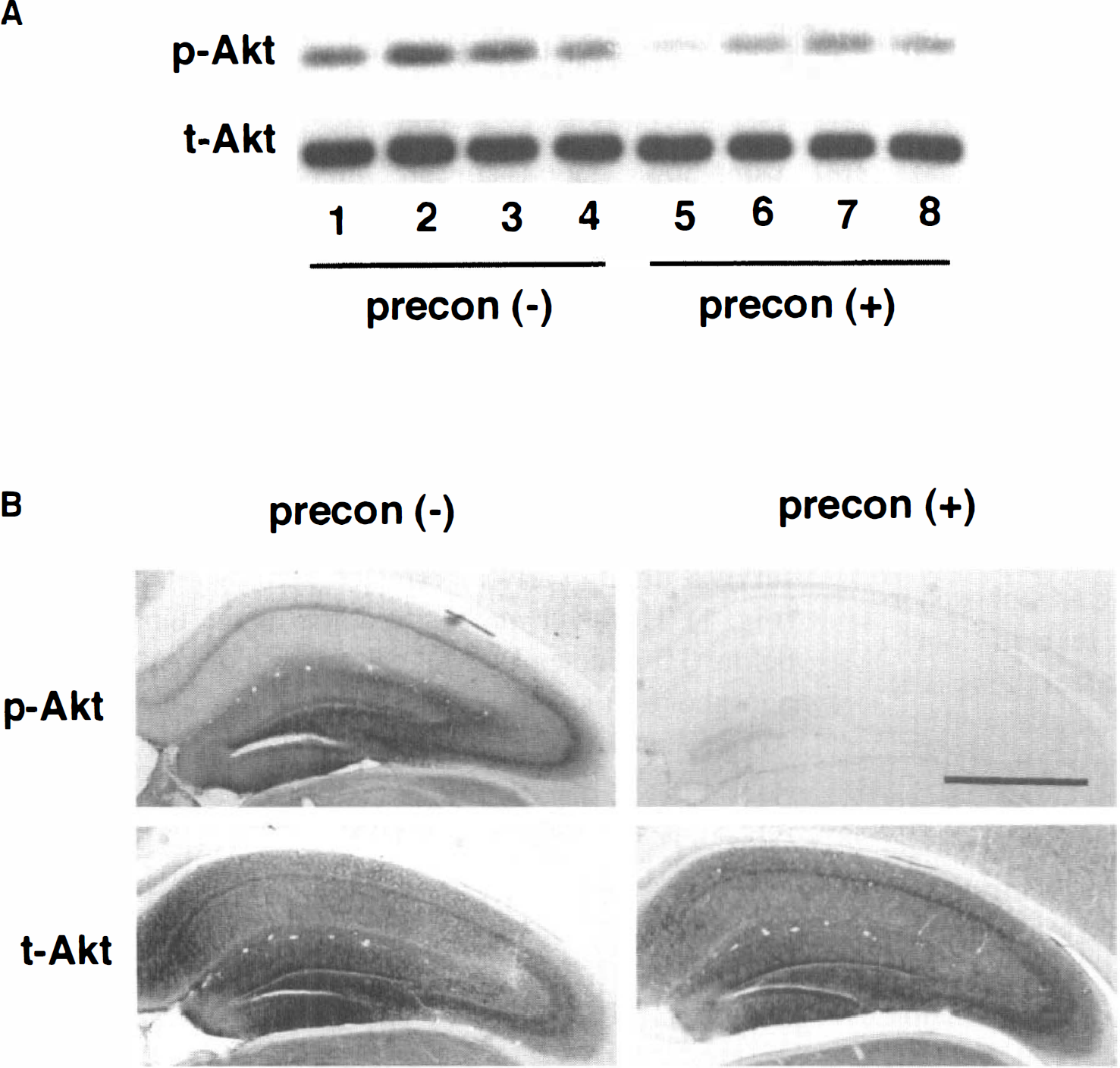
Immunoblotting
DISCUSSION
This is the first report investigating the possible involvement of Akt activation in ischemic tolerance. The authors observed that total protein expression levels of Akt were not affected by ischemic preconditioning, but rather diminished the activation of Akt during early reperfusion periods. The current results indicate that Akt does not play a major role in inducing ischemic tolerance by preconditioning. Unknown molecular events that induce ischemic preconditioning may result in decreased phosphorylation of Akt after reperfusion.
The authors have found that Akt expression and phosphorylation were transiently elevated in the CA1 pyramidal cells during early reperfusion. Enhanced phosphorylation of Akt in the CA1 was also demonstrated in the rat after 15 minutes of global ischemia (Ouyang et al., 1999). It is possible that Akt plays a role in neuronal survival during early reperfusion in the rat and gerbil global ischemia model resulting in delayed death of CA1 pyramidal cells. The authors also demonstrated by immunostaining that elevation of phospho-Akt levels in the CA1 was less than in the dentate gyrus and CA3. This may explain the selective vulnerability of CA1 to ischemia. Furthermore, the authors observed that total-Akt levels in the CA1 pyramidal cell layer were decreased below the control by 1 day after reperfusion, preceding cell death. The causal relation between the decrease in total-Akt levels and cell death is not yet clear; this remains to be determined.
The observed temporal changes in phospho-Akt levels were consistent in immunoblotting and immunostaining; however, the changes in immunostaining for total-Akt using brain sections appeared more prominent than those in immunoblotting. These data reflect methods of detection. Although immunostaining is more sensitive to spatial changes in immunoreactivity, providing information about cellular localization, immunoblotting was performed on lysates from total hippocampal tissues, including not only CA1 but also CA3 and dentate gyrus.
In conclusion, the current study demonstrated Akt activation in the hippocampus after ischemia and reperfusion, suggesting that Akt might be involved in neuronal survival during early reperfusion periods after ischemia (Fig. 1). However, preconditioning of the hippocampus before 3.5 minutes BCAO resulted in a dramatic drop in phosphorylation levels of Akt without affecting the total-Akt protein levels (Fig. 2). This indicates that, with respect to ischemic tolerance, Akt may not play a crucial role in determining cell survival.
