Abstract
Nitric oxide, a product of nitric oxide synthase activity, relaxes vascular smooth muscle and elevates brain blood flow. We evaluated the importance of eNOS to cerebral blood flow augmentation after L-arginine infusion and increases in flow after eNOS upregulation in SV-129 mice. Blood flow was measured by laser-Doppler flowmetry before and after L-arginine infusion (450 mg/kg during a 15-minute period) or measured by 14C-iodoamphetamine indicator fractionation or 14C-iodoantipyrine tissue equilibration techniques. rCBF increased by 26% (laser Doppler flowmetry) after L-arginine infusion but did not change in mutant mice deficient in eNOS expression. After eNOS upregulation by chronic simvastatin treatment (2 mg/kg subcutaneously, daily for 14 days), L-arginine amplified and sustained the hyperemia (38%) and increased absolute brain blood flow from 86 ± 7 to 119 ± 10 mL/100 g per minute. Furthermore, pretreatment with simvastatin enhanced blood flow within ischemic brain tissue after middle cerebral artery occlusion. Together, these findings suggest that eNOS activity is critical for blood flow augmentation during acute L-arginine infusion, and chronic eNOS upregulation combined with L-arginine administration provides a novel strategy to elevate cerebral blood flow in the normal and ischemic brain.
The nitric oxide (NO) precursor L-arginine increases cerebral blood flow in rats and humans after intravenous infusion (Morikawa et al., 1994; Reutens et al., 1997). The mechanism, however, is not well-understood. Studies from our laboratory showed that L-arginine, but not D-arginine, infusion dilates pial blood vessels, and topical nitric oxide synthase inhibitor L-nitroarginine-methyl ester (L-NAME) significantly attenuates this response (Morikawa et al., 1994). Hence, NO or a derivative most likely mediates L-arginine-induced vasodilation. Presumably via a similar mechanism, L-arginine infusion promotes flow-dependent functional recovery in ischemic cortex (Dalkara et al., 1994) and reduces the size of experimental brain infarction in rats (Morikawa et al., 1994) and may therefore be useful therapeutically. However, alternative mechanisms have been put forth to explain the putative linkage between L-arginine administration and vasodilation because under a wide range of physiological conditions, the enzyme eNOS is apparently saturated with substrate in vitro (Morikawa et al., 1994; Harrison, 1997; McDonald et al., 1997; Michel and Feron, 1997).
We recently reported that chronic simvastatin (HMG-CoA reductase inhibitor) treatment upregulates calcium-dependent NOS activity within endothelial cells, enhances basal brain blood flow levels, and reduces experimental brain infarct size in wild-type mice (Endres et al., 1998). However, mice deficient in eNOS gene expression (eNOS-null mice) are refractory to simvastatin treatment (Endres et al., 1998). In this report, we extend evidence implicating NO and its derivatives as mediators of blood flow augmentation after L-arginine and chronic statin administration. We examined whether L-arginine infusion increases flow in genetically-engineered mice deficient in type III NOS (endothelial NOS), and whether increasing NOS protein by chronic statin treatment augments the blood flow response to infused L-arginine during basal conditions and during cerebral ischemia.
METHODS
Drugs
HMG-CoA reductase inhibitor (simvastatin) was purchased from commercially-available sources, purified, isolated, and the activity was verified in vitro. It was chemically activated by alkaline hydrolysis before injection (Endres et al., 1998; Laufs et al. 1997). L-arginine hydrochloride (Sigma, St. Louis, MO, U.S.A.) was dissolved in distilled water and adjusted to pH 7.0 with sodium hydroxide.
General preparation
All experiments were conducted in accordance with National Institutes of Health and Massachusetts General Hospital (MGH) Institutional guidelines. 129/SVEvTacBR mice (male) were purchased from Taconic Farms (Germantown, NY, U.S.A.). Type III NOS mutant mice lacking the eNOS gene were generated at MGH with a mixed background (129/SV and C57BL/6 mice) (Huang et al., 1995). Animals were allowed free access to food and water ad libitum. Anesthesia was induced with 2% to 2.5% halothane and maintained with 1.0% halothane in 70% N2O and 30% O2. Mice were intubated transorally, placed in the stereotaxic frame, and ventilated artificially (SAR-830/P, CWE, Ardmore, PA, U.S.A.). End-tidal CO2 was monitored by a microcapnometer (Columbus Instruments, Columbus, OH, U.S.A.). The femoral artery and vein were cannulated with a polyethylene catheter (PE-10, Intramedic, Becton Dickinson, Sparks, MD, U.S.A.) for continuous arterial blood pressure and heart rate monitoring and for drug infusion. In L-arginine infusion studies, urethane (750 mg/kg intraperitoneally) was administered and halothane was with-drawn gradually after completing surgery and before L-arginine. An additional dose of urethane (500 mg/kg intraperitoneal) was added 30 minutes later. Mice were immobilized with pancuronium bromide (0.4 mg/kg intraperitoneal). There was no blood pressure or pulse elevation in response to tail pinch during anesthesia. Arterial blood gas and pH were analyzed at baseline and after drug infusion in each experiment. Rectal temperature was maintained at approximately 37°C with a thermostatically controlled mat (temperature control, FHC, Brunswick, ME, U.S.A.).
Protocol for laser-Doppler flowmetry
The skull was exposed by a longitudinal skin incision and regional cerebral blood flow (rCBF) was monitored over the intact skull by laser-Doppler flowmetry (BPM 403A, Vasamedics, MN, U.S.A.) using a needle probe (0.8 mm in diameter, p-433) placed over the left parietal cortex. Animals were injected subcutaneously (abdomen) with simvastatin (2 or 20 mg/kg) daily for 2 or more weeks before experimentation. Anesthetized animals were instrumented as described previously. After achieving stable baseline rCBF (5 to 10 minutes), L-arginine (450 mg/kg) or saline vehicle was infused via the left femoral vein at a constant rate of 100 μL/kg per minute for 15 minutes. rCBF was recorded for an additional 40 minutes whereas depth of anesthesia was kept constant.
Measurement of plasma level of amino acids
To determine whether plasma arginine levels differed after amino acid infusion in the presence or absence of simvastatin, blood was sampled before (n = 3 per group) or 40 minutes after L-arginine infusion (n = 4 per group). Plasma was prepared from citrated blood (3.82% adjusted 0.1 vol/vol sodium citrate) and stored at −80°C after centrifugation. Amino acid analysis was carried out by ion-exchange chromatography using a Beckman 6300 amino acid analyzer (Palo Alto, CA, U.S.A.). Samples were deproteinized and half-diluted with 3% sulfosalicylic acid in lithium buffer containing 5% per milligram internal standard (S-2 aminoethyl-L-cysteine).
Absolute cerebral blood flow measurement after L-arginine
The protocol of anesthesia and infusion of L-arginine was the same as described previously. In addition, the jugular vein was cannulated. Thirty minutes after start of infusion of L-arginine or saline, CBF was determined using an indicator fractionation technique described previously (Fujii et al., 1997). Arterial blood was withdrawn continuously from the femoral artery at a rate of 0.3 mL/min with a pump (Stoelting, Wood Dale, IL, U.S.A.). One microcurie of N-isopropyl-[methyl 1,3-14C]-p-iodoamphetamine (American Radiolabeled Chemicals Inc., St. Louis, MO, U.S.A.) dissolved in 0.1 mL saline was injected into the jugular vein as a bolus (<1 second). Twenty seconds after injection, the animal was decapitated and the blood withdrawal terminated simultaneously. The brain was removed quickly and frozen in isopentane chilled with dry ice. After adding Scintigest (Fisher Scientific, Fair Lawn, NJ, U.S.A.) and incubating (50°C overnight), scintillation fluid and H2O2 were added. Twelve hours after shaking, radioactivity in brain and blood were measured by liquid scintillation spectrometry (RackBeta 1209, Pharmacia-Wallac, Gaithersburg, MD, U.S.A.). CBF was calculated according to the method of Van Uitert and Levy (1978) and Betz and Iannotti (1983).
Absolute CBF measurement after middle cerebral artery occlusion and statin pretreatment
To determine the impact of chronic simvastatin treatment on blood flow after permanent middle cerebral artery (MCA) occlusion, male and female SV-129 mice (20 to 25 g) were killed at 4 hours after occlusion for absolute CBF measurement. These mice had been treated with simvastatin (2 mg/kg per day, subcutaneously) or an equivalent amount of vehicle (phosphate buffered saline) for 14 days before the onset of ischemia.
Focal cerebral ischemia was induced by occlusion of MCA using the intraluminal filament technique as described before (ZeaLonga et al., 1989; Huang et al., 1994). Briefly, through a ventral midline incision, the left common and external carotid arteries were isolated and ligated. A microvascular clip (Zen temporary clip, Ohwa Tsusho, Tokyo, Japan) was temporarily placed on the internal carotid artery and the pterygopalatine artery. An 8-0 nylon monofilament (Ethicon, Somerville, NJ, U.S.A.) coated with silicone was introduced into the internal carotid artery via the external carotid artery and advanced 10 mm distal to the carotid bifurcation to occlude the MCA.
Regional cerebral blood flow was measured using the 14C-iodoantipyrine technique described previously in mice (Jay et al., 1988) with some modifications. Briefly, halothane-anesthetized mice were infused with 14C-iodoantipyrine (5 μCi in 100 μL saline) through the left femoral vein during a 1-minute period via a pump, during which time arterial blood samples were collected every 5 seconds (total 50 to 100 μL) onto preweighed filter paper disks. Animals were then decapitated immediately. The whole head was immersed at once in isopentane chilled with dry ice (−45°C) and then stored on dry ice. Radioactivity in blood samples was measured by liquid scintillation spectrometry (RackBeta 1209, LKB) 24 hours later.
The frozen brains were cut into coronal sections of 20 μm thickness using a cryostat-microtome. The sections were thaw-mounted on glass coverslips, immediately dried on a hot plate (60°C), and exposed to Kodak SB-5 autoradiographic film for 3 days in an X-ray cassette along with a set of calibrated 14C-polymer standards from Amersham (Arlington Heights, IL, U.S.A.). Coronal sections and regions of interest (ROIs) were selected as described by Hakim et al. (1992), four sections (1, 2, 3, and 4) corresponding to +1.54, +0.14, −1.94, and −3.88 mm from bregma, respectively. In each section, the density of the autoradiograms covering 4 to 5 brain structures in both hemisphere was measured with a computerized image analyzer (Imaging Research Inc., St. Catherines, Canada). The software converted the optical density to radioactive content and to CBF using the radioactive standards and the 14C-iodoantipyrine blood curve.
Semiquantitative reverse transcription polymerase chain reaction
Aortas were quickly isolated and frozen after sacrifice. Total mRNA isolation, reverse transcription, and semiquantitative competitive polymerase chain reaction (PCR) were performed according to standard techniques as previously described (Endres et al., 1998). The sense (5′-TTCCGGCTGCCACCTGATCCTAA-3′) and antisense (5′-AACATATGTCCTTGCTCAAGGCA-3′) primers for eNOS amplified a 340-bp fragment of murine eNOS that was confirmed by DNA sequencing. Equal amounts of eNOS and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) reverse transcription-PCR products were loaded on agarose gels. Optical densities of ethidium bromide-stained DNA bands were quantified by an image analyzer.
Statistical analysis
Data are presented as mean ± standard error of the mean (SEM). Differences were compared by unpaired two-tailed Student's t-test, by ANOVA preceding Scheffe test (physiologic parameters), by Fisher's protected least-squares difference test (laser Doppler study), or by Student's t-test (absolute CBF measurement). The software StatView 4.0 (Abacus Concepts, Inc., Berkeley, CA, U.S.A.) was used for statistical analysis. P values of <0.05 were considered statistically significant.
RESULTS
Physiologic variables are shown in Tables 1 through 3. There were no group differences in mean arterial blood pressure and heart rate, although values were elevated in eNOS-null mice, as reported previously (Huang et al., 1995). Because blood pressure and heart rate values obtained for every 5-minute epoch during L-arginine infusion were not different by more than 10% of baseline values, only baseline values for each group are given in Table 1. pH values were decreased after L-arginine, consistent with administering the amino acid at pH 7.0.
Physiologic variables during laser-Doppler flowmetry in urethane-anesthetized ventilated wild type, simvastatin-treated and eNOS null mice infused with L-arginine or saline
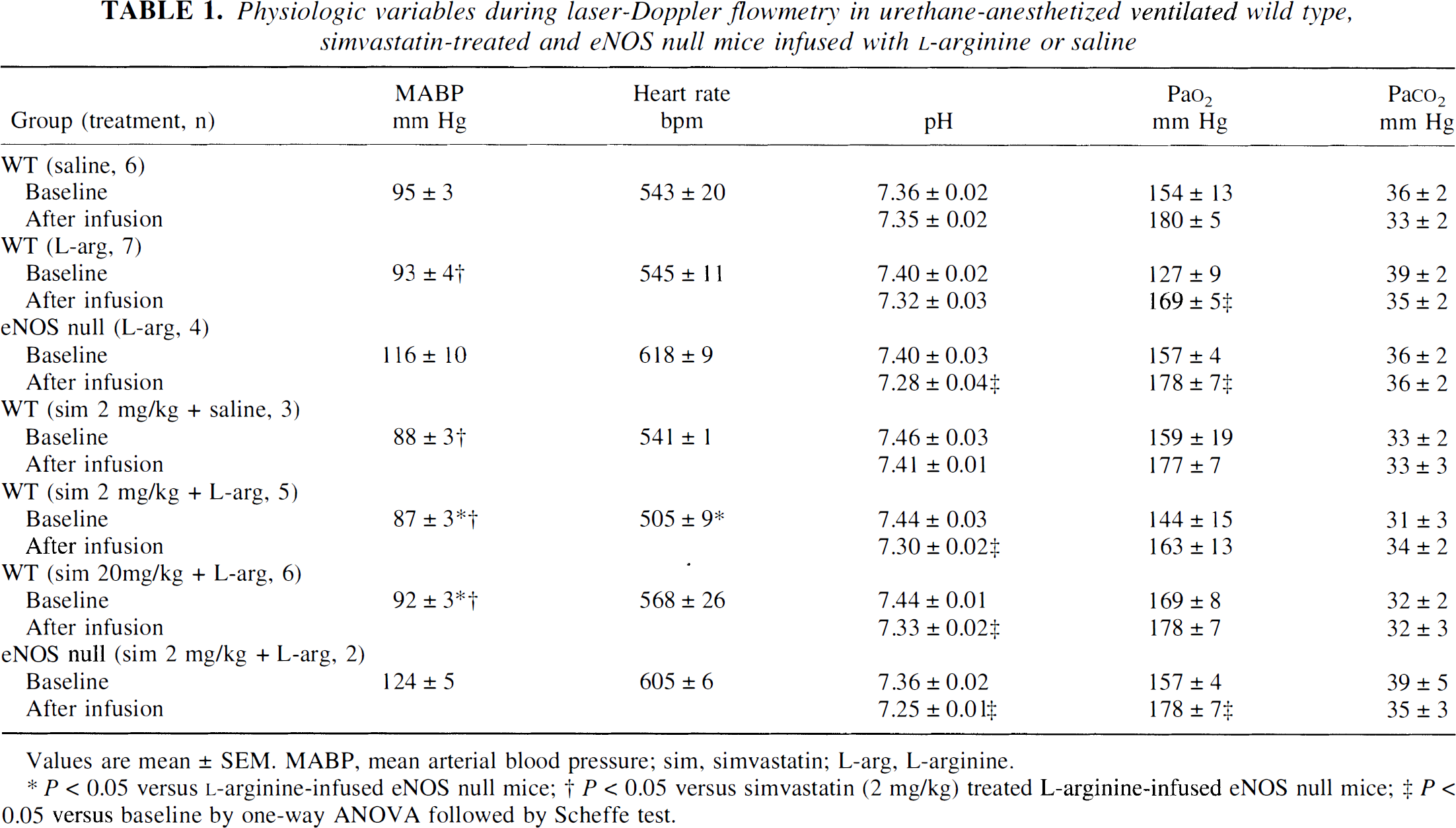
Values are mean ± SEM. MABP, mean arterial blood pressure; sim, simvastatin; L-arg, L-arginine.
P < 0.05 versus L-arginine-infused eNOS null mice;
P < 0.05 versus simvastatin (2 mg/kg) treated L-arginine-infused eNOS null mice;
P < 0.05 versus baseline by one-way ANOVA followed by Scheffe test.
Absolute CBF values detected by [14C]iodoamphetamine indicator fractionation technique and physiologic variables in urethane-anesthetized ventilated vehicle-treated and saline infused, simvastatin-treated saline infused, simvastatin-treated L-arginine infused mice

Values are mean ± SEM. MABP, mean arterial blood pressure; sim, simvastatin; L-arg, L-arginine.
P < 0.05 versus vehicle-treated group by one-way ANOVA followed by Student's t-test.
Physiologic variables before and after MCA occlusion
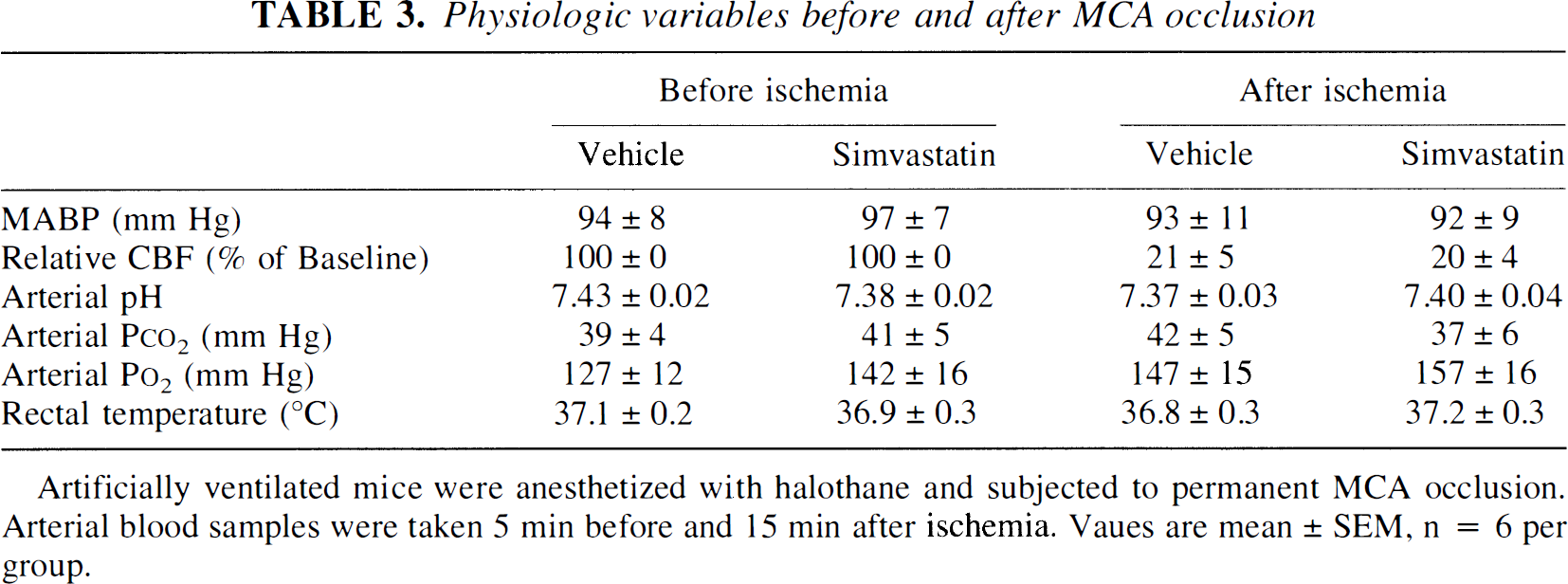
Artificially ventilated mice were anesthetized with halothane and subjected to permanent MCA occlusion. Arterial blood samples were taken 5 min before and 15 min after ischemia. Vaues are mean ± SEM, n = 6 per group.
rCBF response to L-arginine
First, we tried L-arginine 300 mg/kg intravenously because this dose was shown to enhance rCBF in the rat (laser Doppler flowmetry). Only a modest (10%) rCBF elevation was detected (n = 4, data not shown). At a greater dose of amino acid (450 mg/kg intravenously), rCBF increased after 5 to 10 minutes, but was statistically significant after 10 to 15 minutes (Fig. 1). Maximum increases reached 26% at 20 to 25 minutes, after which blood flow declined to control levels. In eNOS-null mice, rCBF did not increase during the 40 minutes after L-arginine infusion; values ranged from −4 to +5% of baseline. In a single experiment, L-arginine (450 mg/kg) increased CBF by 20% during infusion in a C57BL/6 mouse.
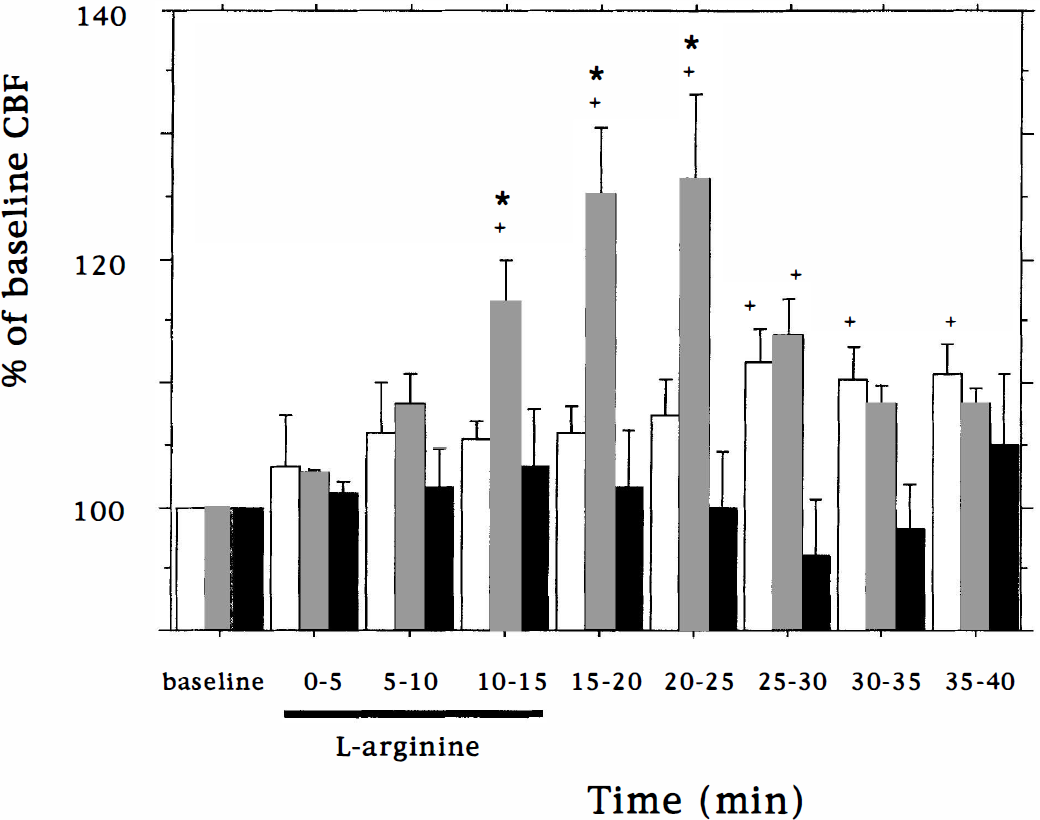
Bar graph showing rCBF changes (laser Doppler flowmetry) in wild-type and eNOS-null mice after L-arginine (450 mg/kg) or saline infusion (100 μL/kg per minute during a 15-minute period). rCBF was measured in urethane-anesthetized ventilated mice. rCBF values for animals infused with saline are represented by clear bars (n = 6), L-arginine infusion in wild-type and eNOS-null mice are shown as grey (n = 7) and black bars (n = 4), respectively. L-arginine significantly elevated blood flow between 10 and 25 minutes. Error bars denote SEM. *P < 0.05 compared with saline infused wild-type; †P < 0.05 compared with baseline control.
As in the rat, L-arginine infusion (450 mg/kg)in mice did not decrease systemic arterial blood pressure in contrast to modest blood pressure decreases or arterial dilation in humans (Giugliano et al., 1997; Nakaki et al., 1990; Smulders et al., 1997; Bode-Büger et al., 1998). An increased cardiac output (not determined) may compensate for vasodilation in animals, although no significant increase in heart rate was observed. Decreases in arterial pH after L-arginine (Reutens et al., 1997) did not change CBF because PaCO2 values did not change (Harper and Bell, 1963).
rCBF response to L-arginine plus simvastatin pretreatment
After chronic daily simvastatin (2 and 20 mg/kg), L-arginine infusion increased rCBF (laser Doppler flowmetry) significantly at the 10 to 15- and 15 to 20-minute epochs during infusion (Fig. 2). Maximum increases of 29% and 31% were measured in groups treated with 2 mg/kg and 20 mg/kg simvastatin, respectively. Increased flow was sustained for at least 20 minutes more than for the response to L-arginine without simvastatin treatment. The maximum increase after acute L-arginine plus chronic simvastatin treatment (31%), however, was not statistically different than for L-arginine alone (26%, Fig. 1). In eNOS-null mice, L-arginine infusion did not increase rCBF significantly after chronic simvastatin treatment (2 mg/kg).
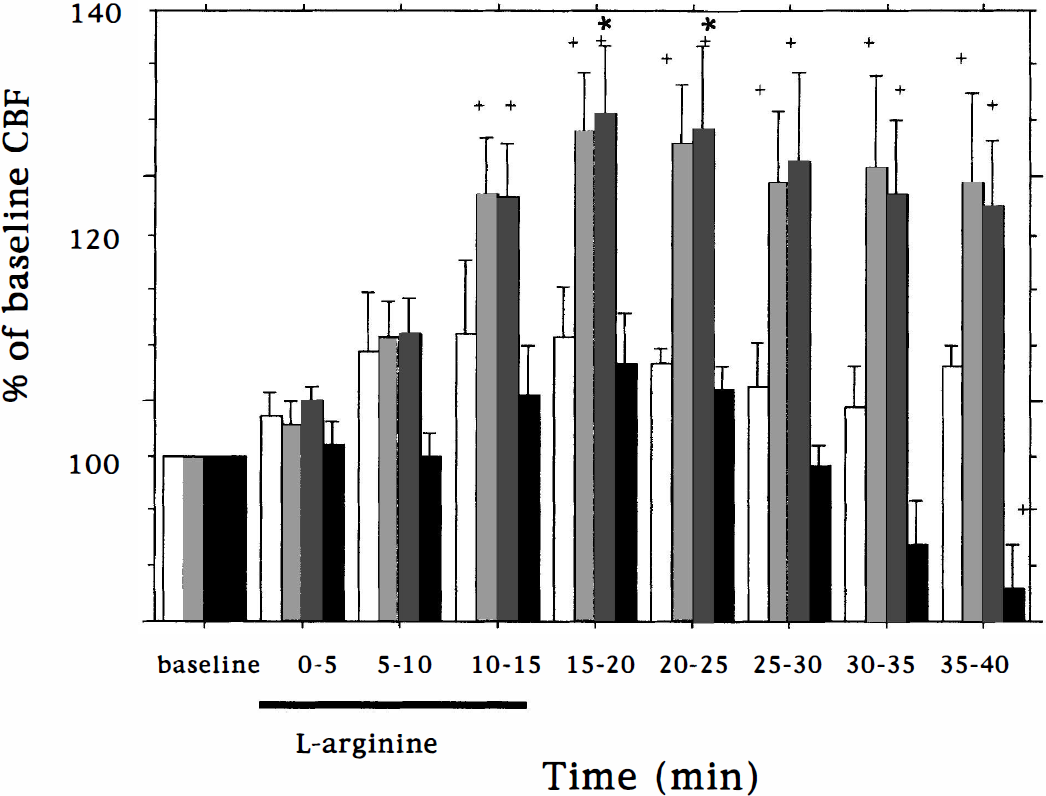
Bar graph showing rCBF changes in simvastatin-treated mice after L-arginine or saline infusion (see legend of Fig. 1). rCBF values for 2 mg/kg-treated animals infused with saline are represented by white (n = 3), L-arginine infusion in 2 mg/kg (n = 5) and 20 mg/kg (n = 6) treated mice are shown as light and dark shades, and 2 mg/kg-treated eNOS-null mice are shown as black bars (n = 2). There was no response to L-arginine in eNOS-null mice, whereas the response to L-arginine was robust and prolonged in mice treated with 2 mg/kg or 20 mg/kg simvastatin. Error bars denote SEM. *P < 0.05 compared with saline infused simvastatin (2 mg) treated mice; †P < 0.05 compared with baseline.
We also detected eNOS expression by semiquantitative reverse-transcription PCR and found that chronic daily simvastatin treatment increased eNOS mRNA levels dose-dependently in the aorta of wild-type mice (Fig. 3). No eNOS expression was detected in aortas of eNOS-null mice.
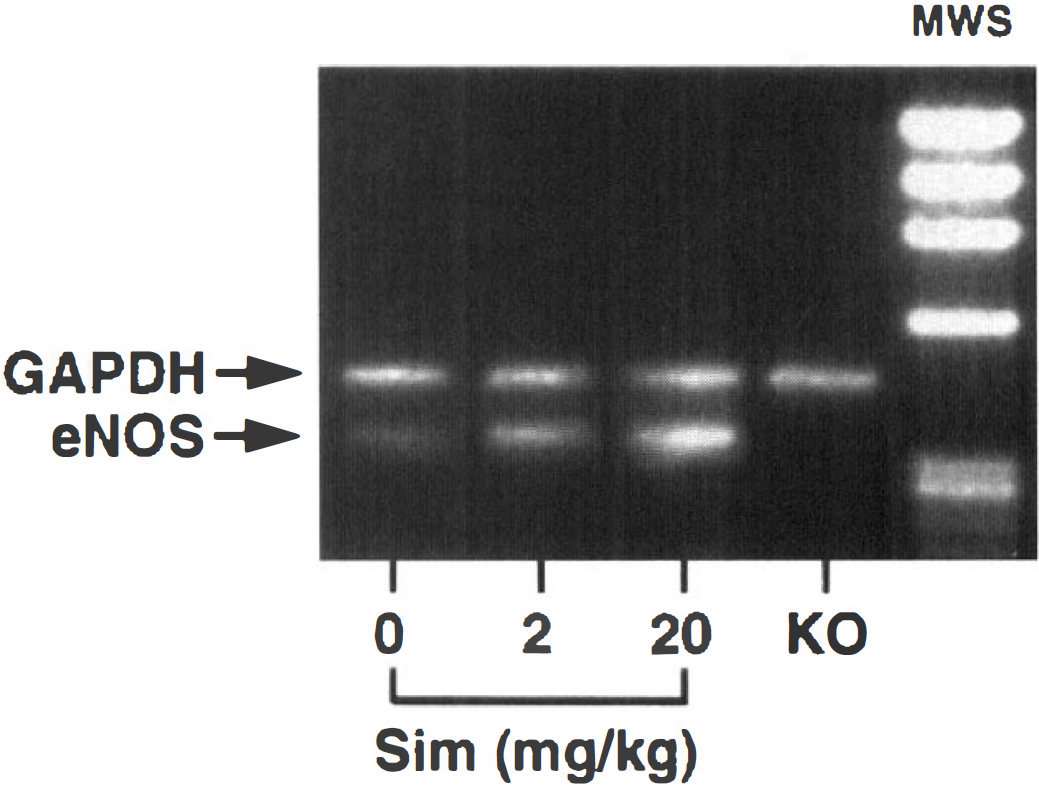
Semiquantitative RT-PCR showing the effects of simvastatin on eNOS mRNA expression in aortas in SV-129 mice after 14 days of treatment. Specificity was determined by the lack of eNOS mRNA in eNOS-deficient mice (KO). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA expression did not change with treatment and was used with an internal standard. MWS, molecular weight standards.
Plasma arginine levels
Plasma L-arginine levels (μmol/L) were 45 ± 6 and 40 ± 7 (n = 3 per group) before infusion. Levels increased to 196 ± 12 and 188 ± 18 (n = 4 per group) 40 minutes after beginning L-arginine infusion in groups treated with or without simvastatin, respectively.
Effects of chronic simvastatin treatment plus acute L-arginine infusion on absolute cerebral blood flow
Chronic simvastatin treatment (2 mg/kg) increased, but not significantly, absolute CBF detected by the iodoamphetamine method by 14% compared with saline-infused, vehicle-treated animals (P > 0.05). However, acute L-arginine infusion (450 mg/kg intravenously) increased absolute blood flow by 38% in simvastatin-treated mice compared with saline-infused, vehicle-treated group (P < 0.05) (Table 2).
Effects of chronic simvastatin treatment on residual absolute rCBF after middle cerebral artery occlusion
Because simvastatin upregulates eNOS activity, augments rCBF, and reduces infarct size (Endres et al. 1998), we determined whether simvastatin (2 mg/kg) augmented regional blood flow in the MCA territory after acute occlusion. Absolute rCBF values were significantly greater than in vehicle-treated animals in nearly all examined brain regions within the ischemic territory using 14C-iodoantipyrine autoradiography (Figs. 4 and 5). This increase was robust in the caudate and putamen. rCBF values also were greater within homologous regions in the nonischemic side. The use of halothane instead of urethane contributed to the greater flow values in normal brain when the tracer iodoantipyrine was administered.
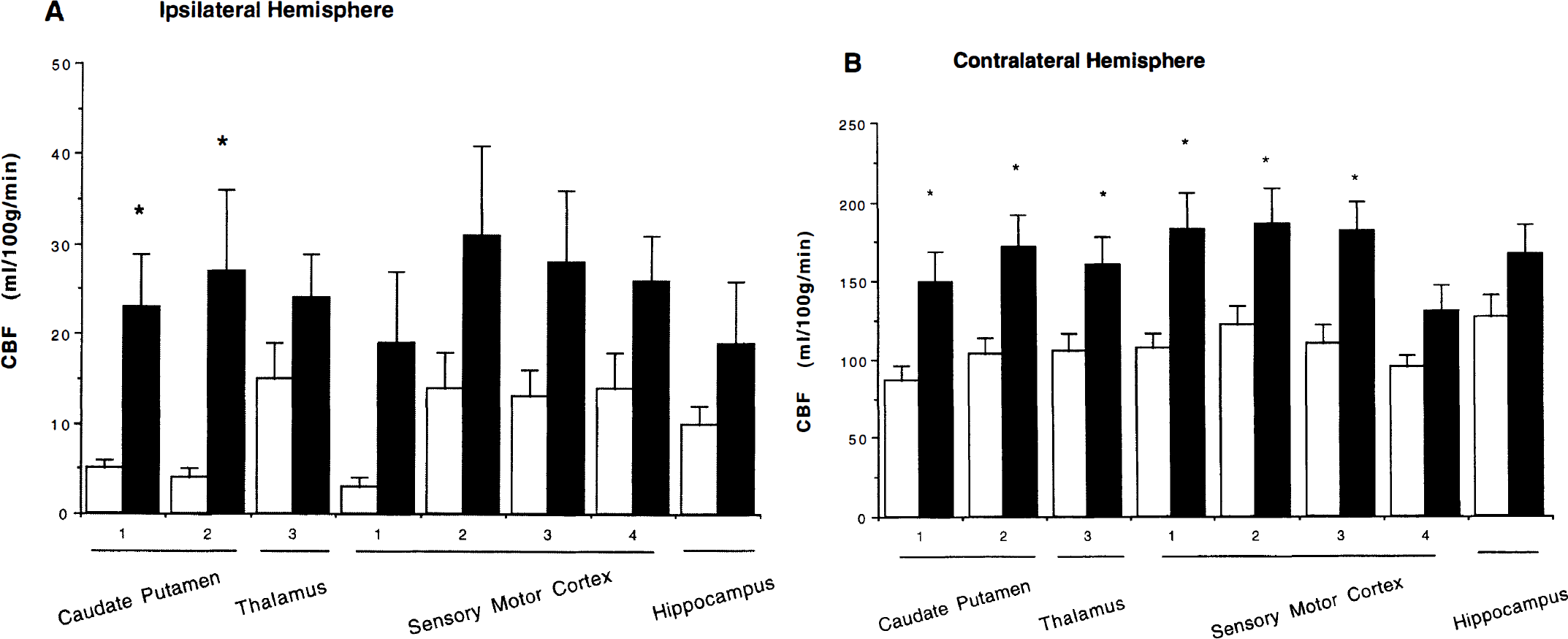
Absolute cerebral blood flow was measured in ipsilateral
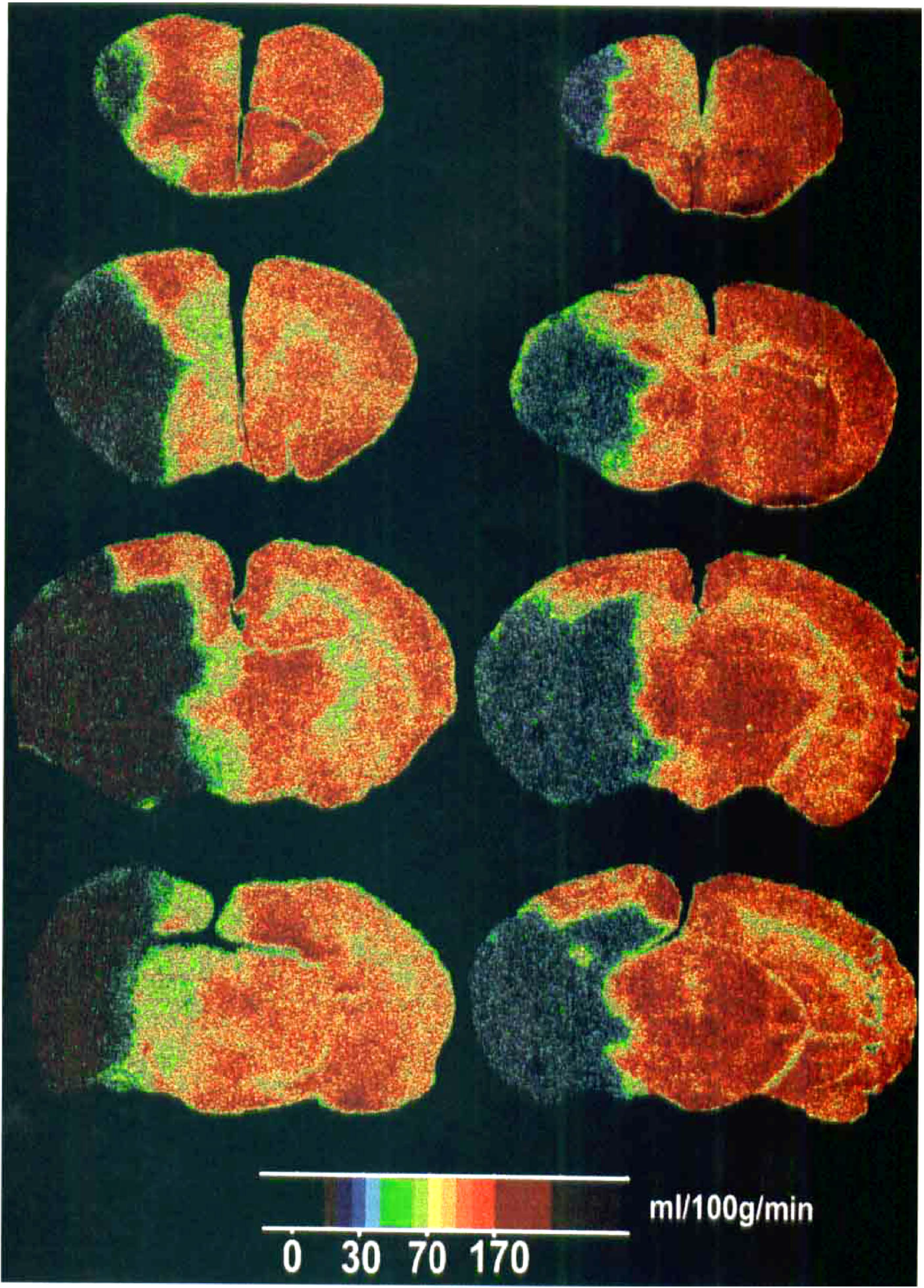
A representative pseudocolor autoradiographic image of absolute cerebral blood flow in brain sections after 4 hours of permanent MCA occlusion in SV-129 mice, showing an enhanced blood flow in all regions of ischemic and nonischemic hemisphere in the simvastatin pretreated mouse (right column).
DISCUSSION
We demonstrate that L-arginine infusion elevates blood flow via eNOS-dependent activity within vascular endothelium. The lack of response in eNOS-null mice is consistent with this conclusion and with previous results showing that topical L-NAME, an inhibitor of NO synthase, attenuates L-arginine-induced dilation of rat pial vessels (Morikawa et al., 1994). The blood flow response to L-arginine following chronic simvastatin treatment further support an association between eNOS activity and rCBF augmentation, and together with results after D-arginine infusion (Morikawa et al., 1994; Dalkara et al., 1994), render unlikely explanations based on flow-dependent osmotic mechanisms during amino acid infusion. We suggest that both the actions of statins and the effects of L-arginine are mediated in part by NO-dependent actions on the brain microcirculation, and possibly by NO-mediated antiplatelet and anti-white cell responses during acute ischemia (Niu et al., 1994).
We previously showed that statins elevate eNOS expression and significantly increase eNOS activity within mouse blood vessels (unpublished observations, Endres et al., 1998; Laufs et al., 1997; Laufs et al., 1998; Laufs and Liao, 1998). The mechanism relates in part to increased mRNA stability and eNOS mRNA translation without affecting nNOS or iNOS gene expression. Statins cause eNOS-dependent cerebral blood flow increases under basal conditions (Endres et al., 1998) and CBF appears to increase further after infusing the eNOS substrate L-arginine. Blood flow was initially assessed using laser Doppler flowmetry as this technique records
The results in eNOS-null mice indicate that the L-arginine response is dependent on eNOS expression, although the mechanism(s) by which L-arginine increases cerebral blood flow is not well-understood. L-arginine may elevate the production of NO by increased substrate availability. However, levels within plasma and cells are substantially greater than the eNOS Km for L-arginine (Arnal et al., 1995; Harrison, 1997; McDonald et al., 1997; Michel and Feron, 1997). The observation that L-arginine drives NO production even when intracellular L-arginine levels are in excess is termed the “arginine paradox” (Arnal et al., 1995; Harrison, 1997; McDonald et al., 1997; Michel and Feron, 1997). Arnal et al. (1995) found that L-arginine reverses L-glutamine's ability to inhibit NO release in bovine aortic endothelial cells and in rabbit aortic rings. In their study, L-arginine dose-dependently enhances endothelium-dependent vascular relaxation only when L-glutamine is present, suggesting a complex interplay between L-glutamine and receptor-mediated activation of NO synthase. The paradox may be influenced by endogenous NO synthase antagonists such as asymmetrical dimethylarginine (Harrison, 1997) or modified by the L-arginine transporter (CAT1) and eNOS at the caveolae in endothelial cells (McDonald et al., 1997; Michel and Feron, 1997). The latter mechanism may facilitate transcellular delivery of substrate to membrane-bound eNOS. The arginine paradox would be unexpected, however, in neurons, axons and synapses where cytosolic nNOS does not reside within membrane bound organelles or, to our knowledge, associate with amino acid transporters. Consistent with this notion, L-arginine infusion does not augment blood flow-associated functional activation in human brain (Reutens et al., 1997), (which in animals is mediated in part by NO release from neurons (Ayata et al., 1996; Ma et al., 1996)), although it does elevate basal cerebral blood flow. Contributions from other mechanisms such as L-arginine-induced insulin release (insulin promotes NO-dependent vasodilation) cannot be entirely excluded but is unlikely since D-arginine, which can also stimulate insulin release, has no effect on CBF (Giugliano et al., 1997; Steinberg et al., 1994; Kurz and Harrison, 1997). Nevertheless, our findings indicate that up- or down-regulation of eNOS protein critically modulates the blood flow response to infused L-arginine.
The effects of L-arginine administration on human vascular beds have been studied also. Intravenous infusion of large doses in awake humans (30 g, approximately 385 mg/kg during a 30 minute period) modestly decreased blood pressure (4%) and total peripheral resistance (10%) but not at doses greater than 6 g (Bode-Büger et al., 1998). In these studies, urinary nitrate excretion and to a lesser extent cyclic guanosine monophosphate (GMP) increased after L-arginine. The authors found that vasodilator response to L-arginine closely corresponded to its plasma concentration and its effects on endogenous NO production. L-arginine decreased peripheral arterial resistance, increased platelet intracellular cyclic GMP levels, and decreased platelet aggregation (Bode-Büger et al., 1994). In hypercholesterolemic subjects, L-arginine, but not D-arginine, improved endothelium-dependent relaxation after methacholine chloride (Creager et al., 1992). In another study, L-arginine infusion augmented nutritive capillary muscular blood flow in patients with peripheral vascular disease (Schellong et al., 1997), consistent with impaired NO synthesis in these patients (Büger et al., 1997). The generation of NO may also mediate the increase in absolute blood flow in the brain after L-arginine infusion (Reutens et al., 1997), suggesting that augmenting eNOS may improve the cerebral microcirculation occurring with conditions of vascular compromise in humans.
Consistent with these findings, L-arginine infusion improves rCBF (as detected by laser Doppler flowmetry) within the ischemic penumbra or perinfarct zone and reduces infarct volume in normotensive and hypertensive rats after middle cerebral artery occlusion (Morikawa et al., 1994). Only modest flow elevations (from 20% to 30% of basal values) improve electrophysiologic activity within ischemic cortex (Dalkara et al., 1994), and elevations of this magnitude can be achieved within the ischemic penumbra or perinfarct zone, as evidenced after chronic eNOS upregulation. L-arginine is reportedly useful in increasing cerebral blood flow after head trauma (Cherian et al., 1999), but not surprisingly, did not decrease damage in models associated with significant endothelial disruption such as ischemia (Prado et al., 1996) and subarachnoid hemorrhage (Pluta et al., 2000).
Because L-arginine and simvastatin increase rCBF, they may also be useful for enhancing the delivery of other coadministered drugs to the hypoperfused brain. Therefore, strategies targeting vascular eNOS activity and generating nitric oxide within vascular compartments provide useful approaches to treat CNS disorders characterized by low flow states (Scandinavian Simvastatin Survival Study Group, 1994; Byington et al., 1995; Sacks et al., 1996; Blauw et al., 1997; Delanty and Vaughan, 1997).
Footnotes
Acknowledgements
The authors thank Victor Nikiforov and Professor Vivian Shih for plasma L-arginine analysis. This work was supported by NIH-sponsored Stroke Program Project 5 P50 NS10828 and also by NIH 1R01 HL6202.
