Abstract
HMG-CoA reductase inhibitors (statins) are associated with improved stroke outcome. This observation has been attributed in part to the palliative effect of statins on cerebral hemodynamics and cerebral auto regulation (CA), which are mediated mainly through the upregulation of endothelium nitric oxide synthase (eNOS). Several animal studies indicate that statin pretreatment enhances cerebral blood flow after ischemic stroke, although this finding is not further supported in clinical settings. Cerebral vasomotor reactivity, however, is significantly improved after long-term statin administration in most patients with severe small vessel disease, aneurysmal subarachnoid hemorrhage, or impaired baseline CA.
Keywords
INTRODUCTION
Long-term treatment with HMG-CoA reductase inhibitors (statins), which are principally prescribed as cholesterol lowering drugs, has been found to reduce stroke incidence and improve stroke outcome. These results are most likely attributable to the statins' pleiotropic effects—especially to the upregulation of endothelial nitric oxide synthase (eNOS) with a subsequent increase in nitric oxide (NO) bioavailability—and not to their cholesterol lowering properties. 1 Several animal models and clinical trials suggest that statin administration augments cerebral blood flow (CBF) and improves cerebral vasomotor reactivity (VMR) by increasing NO bioavailability. 2
Vasomotor reactivity indicates the counterpoise constrictive or dilatory capacity of distal cerebral arteries to a vasoactive stimulus (hypoventilation, hyperventilation, CO2 inhalation, or injection with acetazolamide), while cerebral autoregulation (CA) is the homeostatic mechanism that maintains normal cerebral perfusion against low amplitude arterial blood pressure shifts within normal range perfusion pressures.3–5 In clinical practice, impaired VMR reflects a reduced cerebrovascular reserve, constituting a risk factor for ischemic stroke in settings such as carotid artery disease or delayed cerebral ischemia after subarachnoid hemorrhage.6–8
Herein, we review the effects of statins on cerebral hemodynamics derived from epidemiological, pathophysiological, and clinical evidence. Sources included MEDLINE and EMBASE (last search update performed on 9 January 2012). The search strategy was based on the combination of the following terms: cerebral vasomotor reactivity, cerebral hemodynamics, cerebral autoregulation, cerebral blood flow, statins, and HMG-coA reductase inhibitors. References of the retrieved articles were also screened.
STATIN PRETREATMENT AND CEREBRAL BLOOD FLOW AFTER INDUCED CEREBRAL ISCHEMIA IN LABORATORY TRIALS
In an ischemic stroke model, parenteral administration of high dose (100 mg/kg) simvastatin 2 weeks before middle cerebral artery (MCA) occlusion decreased the infarct volume and ameliorated both perfusion and neurologic deficits in rats, suggesting that simvastatin pretreatment promotes microvascular reperfusion. 9 Long-term statin pretreatment was also related to higher absolute CBF, more intense CBF response to L-arginine, enhanced eNOS activity, and higher eNOS mRNA levels after MCA occlusion in normocholesterolemic mice. These results were independent of serum cholesterol levels and they were not reproduced in eNOS-deficient mice, verifying that statins enhance CBF by a direct upregulation of the eNOS pathway.10,11 Likewise, parenteral mevastatin treatment with 20 mg/kg previous to MCA occlusion in mice for 14 days also resulted in higher eNOS mRNA, eNOS protein, and CBF levels. These effects were absent in the group treated with 2 mg/kg mevastatin and in eNOS-deficient mice, independent of the treatment duration. 12 Finally, results from another laboratory trial showed enhanced CBF in rats with posttraumatic ischemia when simvastatin (20 mg/kg) or atorvastatin (20 mg/kg) administered subcutaneously. 13 Interestingly, in this study eNOS RNA was not increased, suggesting that other eNOS-related mechanisms may be in play including posttranslational modification, or statin effects on increasing the half-life of circulating eNOS. Another study using a daily oral treatment with 20 mg/kg lovastatin for a month did not normalize the lower limit of autoregulation in spontaneous hypertensive rats. 14 Direct effect on CBF was not tested, nor were specific relationships with eNOS activity.
Apart from eNOS upregulation, chronic statin pretreatment may improve outcomes through other pleiotropic, NO-independent mechanisms. For example, higher mRNA levels of endogenous tissue plasminogen activator were seen after induced MCA occlusion in wild-type mice, and also to reduced ischemic lesion volume in eNOS-deficient mice. 15 In summary, observations from animal models suggest that statins exert their protective effects by increasing blood flow through NO-dependent and NO-independent mechanisms.
STATINS IMPROVE IMPAIRED CEREBRAL VASOMOTOR REACTIVITY, WITHOUT A DOCUMENTED EFFECT ON CEREBRAL BLOOD FLOW, IN PATIENTS WITH CEREBROVASCULAR DISEASE
In contrast to animal models, human studies suggest that the protective effects of statins are more related to vasoreactivity than to CBF per se. For example, a retrospective analysis of CBF measurements obtained from a masked prospective study of patients with internal carotid artery occlusion (ICAO) revealed that the 19 patients who were treated with statins did not have higher global or regional baseline CBF values ipsilateral or contralateral to the occluded carotid artery, even though they were younger. Therefore, the diminished rate of cerebrovascular events observed after statin treatment was unlikely to be attributable to CBF augmentation. 16 In another study, statins failed to decelerate the decline in CBF with increasing age, as demonstrated with gradient-echo phase-contrast magnetic resonance imaging of both internal carotid and vertebral arteries. The lack of statin CBF effect held even when the age-related attenuation in CBF was adjusted for the brain parenchymal volume loss over time. 17
In agreement with the considerations above, 4 months of atorvastatin (40 mg) administration had no effect on the resting CBF or mean transit time of cerebral perfusion, measured with arterial spinning labeling and dynamic susceptibility contrast perfusion respectively in a randomized, double-blinded, controlled study. However, the statin-treated group had an improved regional cerebral vasoreactivity, revealed with blood oxygen level-dependent functional magnetic resonance imaging during a recognition memory task, compared with the placebo group. 18 Early reperfusion in the setting of acute stroke was assessed in another mean transit time study protocol at both 4.5 and 6 hours after stroke onset, and was found to be 2- to 3-fold greater in patients who were on statin treatment before stroke onset compared with those who were not. Statin pretreatment was also related with greater neurologic improvement at 1 month follow-up, supporting the hypothesis that statins improve stroke outcome through cerebral hemodynamic amelioration. 19 Daily administration of 40 mg atorvastatin improved L-arginine cerebrovascular reactivity more than systemic vascular response in patients with arterial hypertension and other cardiovascular risk factors. 20 Moreover, in the same study cerebrovascular reactivity to L-arginine was equalized between hypertensive patients and healthy control group after a 3-month treatment with atorvastatin.
In another study, short-term administration of pravastatin (40 mg) in 25 healthy volunteers for 14 days, significantly increased VMR, which was estimated with bilateral transcranial Doppler sonography of both MCA. The effect was greatest on the seventh day of treatment and was most pronounced in those with lower initial VMR values. 21 Likewise, long-term treatment with high-dose atorvastatin (80 mg) improved VMR in an open-label, self-controlled, interventional study of 36 patients with hypercholesterolemia and controlled hypertension. This enhancement in VMR persisted for up to 45 days after discontinuation of therapy.22,23 Enhanced cerebrovascular collateralization was related to a long-term statin administration before ischemic stroke in a retrospective analysis of 96 patients with a major artery occlusion. 24
Finally, in a placebo-controlled, double-blinded, crossover study of 16 healthy students, pravastatin withdrawal was correlated with an acute and significant decrease in CBF velocity. 23 This negative feedback phenomenon, attributed to a deciduous suppression of eNOS production, is thought to have a major contribution to the adverse cerebrovascular events observed after abrupt statin withdrawal. 1
STATINS AND CEREBRAL VASOMOTOR RESPONSE IN PATIENTS WITH SMALL VESSEL DISEASE
Although VMR measurements are most commonly applied to large vessel disease, several studies show that statins improve cerebral hemodynamics in small vessel disease (SVD). For example, administration of 5 mg simvastatin improved VMR in 10 patients with history of ischemic stroke at least 6 months earlier, even though regional and overall CBF at rest were not affected. 25 The improvement in VMR was more pronounced in lacunar infarctions than in large vessel disease. In another study, impaired cerebrovascular reactivity to L-arginine in patients with lacunar infraction was significantly improved after daily administration of 40 mg atorvastatin for 3 months. 26 In a longitudinal exploratory pilot study, administration of 20 mg pravastatin for 2 months was related to a significant improvement in VMR in 16 patients with cerebral SVD, especially in those who initially exhibited a more severe VMR impairment. However, pravastatin treatment was neither related to an increase in mean CBF volume nor to the decline of serum cholesterol levels, again consistent with the theory that statins influence cerebral hemodynamics with a cholesterol-independent mechanism. 27
Not all statin studies showed improved hemodynamics. Results from a randomized, double-blinded, placebo-control study of 94 patients with severe SVD and a history of lacunar infraction indicate that treatment with 80 mg atorvastatin for 3 months had no positive effect on severely impaired VMR. 28 In a nonatherosclerosis cohort, neither resting mean flow velocity nor VMR were improved after atorvastatin administration in 24 patients with cerebral autosomal dominant arteriopathy with subcortical and leukoencephalopathy. 29
STATINS ALSO HAVE EFFECTS IN SUBARACHNOID HEMORRHAGE
Immediate oral treatment with 40 mg pravastatin was related with a briefer period of impaired CA after aneurysmal subarachnoid hemorrhage (aSAH), mainly in the ipsilateral side of the aneurysm, when compared with placebo-treated group. In this phase II randomized, placebo-control trial, improved CA also correlated with reduced incidence rates of vasospasm, severe vasospasm and delayed ischemic deficits after the onset of the aSAH, observed in the statin-treated group.30,31 The clinical outcomes above were reproduced in an experimental laboratory setting, in which male mice were subcutaneously injected with simvastatin (20 mg/kg) or vehicle for 14 days after the SAH onset. Western blot analysis revealed augmented eNOS expression in the SAH mice groups that were pretreated or posttreated with statin, supporting the assumption that statins improve cerebral hemodynamics by upregulating the endothelial NO synthase. 32
A double-blinded, placebo-control, randomized clinical trial evince that daily simvastatin (80 mg) administration for up to 14 days had no effect on vasospasm, delayed ischemic deficit, or outcome. Considering that simvastatin-treated patients were reported to receive vasoactive medication more frequently, either for hypertension or for hypervolemia, compared with placebo group, this confound could be responsible for the elimination of the beneficial effect of statins. 33
In a large retrospective cohort study of 514 patients with SAH who received statins there was a higher risk for total vasospasm, although statin administration was neither related to higher rates of symptomatic vasospasm nor to worse outcome. Abrupt statin withdrawal was related with a higher incidence of total and symptomatic vasospasm further supporting the effect of statins on cerebral hemodynamics in this population. 34
DISCUSSION
In the present literature review, we considered the evidence for the relation between statin administration and cerebral hemodynamic response. We retrospectively reviewed data from trials with wild-type mice, eNOS-deficient mice, healthy adults and patients with arterial hypertension, dyslipidemia, ICAO, ischemic stroke (lacunar or major artery occlusion), and aSAH.
Our main finding is that whereas many studies show better outcomes due to pleiotopic effects of statins, these effects appear to be related to absolute increases in CBF in animal studies, but to improved VMR in human studies (Table 1).
Statins and cerebral hemodynamics in human studies
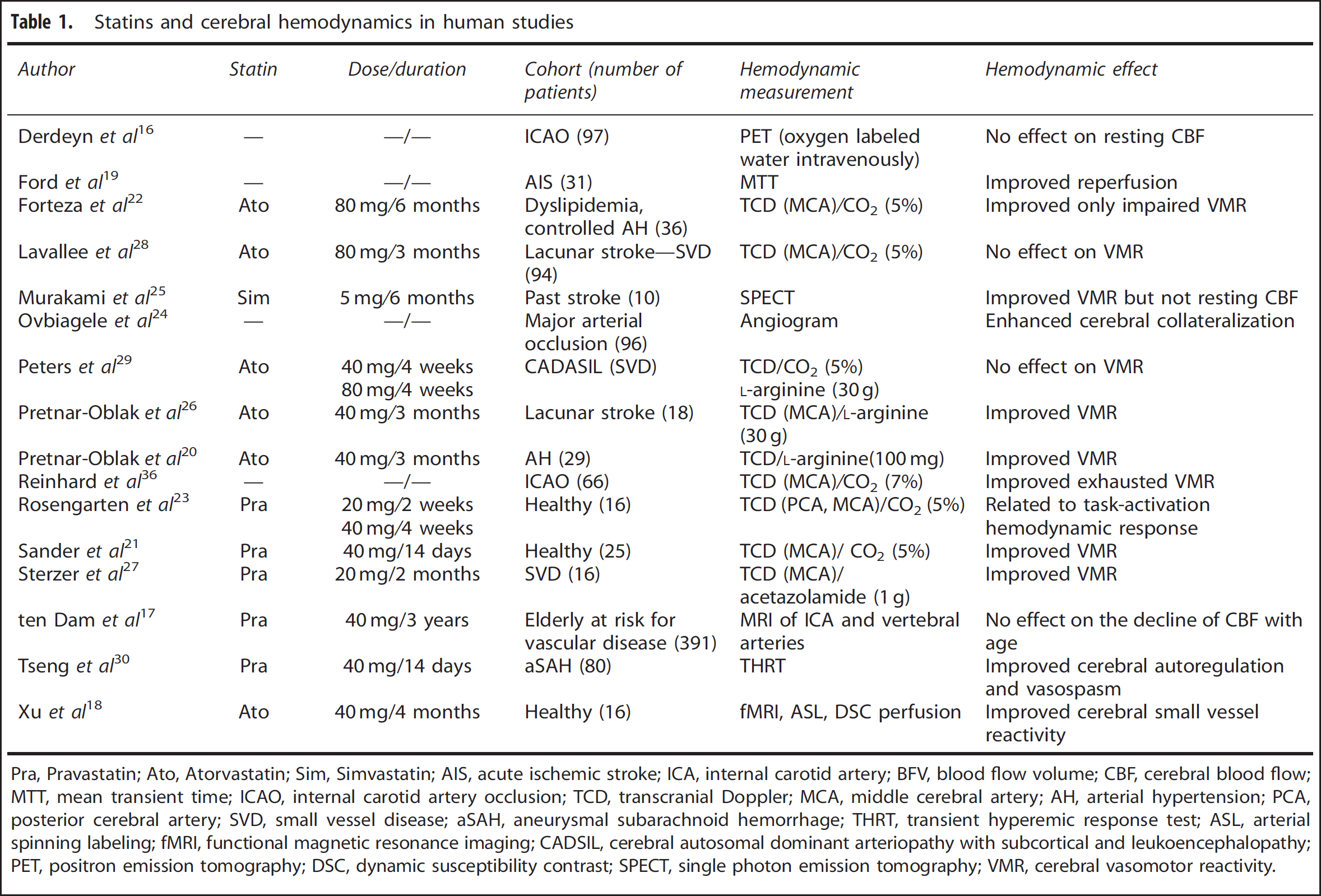
Pra, Pravastatin; Ato, Atorvastatin; Sim, Simvastatin; AIS, acute ischemic stroke; ICA, internal carotid artery; BFV, blood flow volume; CBF, cerebral blood flow; MTT, mean transient time; ICAO, internal carotid artery occlusion; TCD, transcranial Doppler; MCA, middle cerebral artery; AH, arterial hypertension; PCA, posterior cerebral artery; SVD, small vessel disease; aSAH, aneurysmal subarachnoid hemorrhage; THRT, transient hyperemic response test; ASL, arterial spinning labeling; fMRI, functional magnetic resonance imaging; CADSIL, cerebral autosomal dominant arteriopathy with subcortical and leukoencephalopathy; PET, positron emission tomography; DSC, dynamic susceptibility contrast; SPECT, single photon emission tomography; VMR, cerebral vasomotor reactivity.
Although this dichotomy was robust in our review, it is possible that it is due to both preference and experience of researchers in reporting cerebral hemodynamics measurements in animals versus humans.
Statin treatment was found to enhance CBF in all animal experiments, except for the one in which the drug was orally administrated. 14 Therefore, the fact that CBF augmentation was not confirmed in clinical trials, could also be due the lower doses used and the nonparenteral route of drug administration, which are currently not clinically available applicable in humans. Future research should include the development of parenteral administrated formulas, their possible side effects and the determination of the maximum dosage that can be safely used in humans.
Limitations of this review also include the small sample size, nonblindness, nonrandomization, selection bias, absent placebo control group and retrospective study designs present in many of the clinical trials cited in this paper. Moreover, the variability of the study methods of cerebral hemodynamic measurement methods, statin dosage, and study duration impedes comparison and therefore should temper our conclusions. Even though statin administration was related to VMR enhancement in 8 out of 10 clinical trials, this effect was more pronounced in patients with initially impaired VMR and in patients with SVD (Table 1). It is notable that there are two negative studies.28,29 Finally, when assessing the outcomes above we should take into consideration that different HMG-CoA inhibitors exhibit different lipophilicity, penetration of the blood–brain barrier, and upregulation of the eNOS.1,12
In a recent experimental setting, intraperitoneal administration of simvastatin before transient MCA in mice had no significant effect on CBF or infarcts size. However, when combined with the phosphodiesterase inhibitor dipyridamole both CBF increased and infarct size decreased significantly. These effects were absent in coadministration of aspirin and in eNOS-deficient mice, suggesting that dipyridamole beyond platelet inhibition enhances the regulatory role of statins on endothelium via the NO pathway. 35 Combination of drugs that augment eNOS, like dipyridamole, could amplify the effect of statins on VMR and thus constitute a novel therapeutic approach after stroke either by direct reperfusion or by enhancing the delivery of coadministrated drugs in the ischemic tissue.10,11
In conclusion, the CBF enhancement observed after long-term statin treatment in laboratory settings was not confirmed in clinical trials. However, cerebral vasomotor reactivity in many clinical protocols of patients with SVD, large vessel disease, or aneurysmal SAH was significantly improved with statin pretreatment. These effects are attributed to the cholesterol-independent pleiotropic effects of statins, most likely to eNOS augmentation. Although the reasons that animal studies differ from human studies in the manifestation of blood flow effects are still unclear, the literature overall is consistent in showing statins' positive impact on CBF regulation, and can thus be offered as a plausible explanation for improved clinical outcomes after their use. Additional randomized controlled trials using a variety of blood flow measurements are required to confirm the findings above.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
