Abstract
Repetitive passive movements are part of most rehabilitation procedures, especially in patients with stroke and motor deficit. However, little is known about the consequences of repeated proprioceptive stimulations on the intracerebral sensorimotor network in humans. Twelve healthy subjects were enrolled, and all underwent two functional magnetic resonance imaging (fMRI) sessions separated by a 1-month interval. Passive daily movement training was performed in six subjects during the time between the two fMRI sessions. The other six subjects had no training and were considered as the control group. The task used during fMRI was calibrated repetitive passive flexion-extension of the wrist similar to those performed during training. The control task was rest. The data were analyzed with SPM96 software. Images were realigned, smoothed, and put into Talairach's neuroanatomical space. The time effect from the repetition of the task was assessed in the control group by comparing activation versus rest in the second session with activation versus rest in the first session. This time effect then was used as null hypothesis to assess the training effect alone in our trained group. Passive movements compared with rest showed activation of most of the cortical areas involved in motor control (i.e., contralateral primary sensorimotor cortex, supplementary motor area [SMA], cingulum, Brodmann area 40, ipsilateral cerebellum). Time effect comparison showed a decreased activity of the primary sensorimotor cortex and SMA and an increased activity of ipsilateral cerebellar hemisphere, compatible with a habituation effect. Training brought about an increased activity of contralateral primary sensorimotor cortex and SMA. A redistribution of SMA activity was observed. The authors demonstrated that passive training with repeated proprioceptive stimulation induces a reorganization of sensorimotor representation in healthy subjects. These changes take place in cortical areas involved in motor preparation and motor execution and represent the neural basis of proprioceptive training, which might benefit patients undergoing rehabilitative procedures.
Keywords
Recovery from neurologic deficit has been studied in the last few years with neuroimaging techniques. Profound intracerebral reorganization during the recovery period has been found (Chollet et al., 1991; Weiller et al., 1992, 1993). Many other evidences for brain plasticity also have appeared in the human unlesioned brain in the field of motor learning (Karni et al., 1995; Friston et al., 1991; Grafton et al., 1992, Jenkins et al., 1994).
Proprioceptive repetitive stimulations elicited by passive movements of various limb segments are widely used in many rehabilitation procedures in patients after a stroke. Although clinicians are not far from accepting that rehabilitation does represent an actual therapeutic approach for neurologic function recovery after stroke (Jeffery et al., 1995; Ottenbacher and Jannell, 1993), effective data are lacking, and rehabilitation remains incompletely validated. Moreover, the inclusion of proprioceptive chronic stimulation in the rehabilitation procedures was empirically determined, and little is known about the mechanisms by which proprioceptive stimulation acts.
However, a potential substrate for rehabilitation has been approached in studies using active movement. In monkeys, Nudo et al. (1996) showed that functional reorganization in the undamaged motor cortex was accompanied by behavioral recovery of skilled hand function (Nudo et al., 1996). Using transcranial magnetic stimulation before and after 2 weeks of constraint-induced movement therapy in patients with stroke, Liepert et al. (1998) demonstrated an enhanced neuronal excitability in the damaged hemisphere correlated with an improvement in motor performance (Liepert et al., 1998). Both studies strongly suggest that chronic active training facilitates the reorganization of motor maps.
Modern neuroimaging techniques (positron emission tomography scan and functional magnetic resonance imaging [fMRI]) have allowed the study of the cortical representation of proprioceptive stimulation, and proprioceptive afferences have been shown to project to most of the motor system cortices (Alary et al., 1998; Weiller et al., 1996).
This study determines, with fMRI, if passive proprioceptive training in healthy subjects modifies the cortical representation of passive movements. We hypothesize that chronic passive training induces enhanced neuronal excitability in the sensorimotor cortex of our subjects.
SUBJECTS AND METHODS
Subjects
We studied 12 healthy, right-handed volunteers (7 women and 5 men; mean age 33 years, range 26 to 45 years) with no history of neurologic or psychiatric disease and free of any medication. All subjects gave written informed consent to the study, which was performed with the approval of our Institutional Human Studies Committee.
The subjects enrolled were divided in two groups of six. All underwent two fMRI sessions separated by a 1-month interval.
Activation task
The activation task was the same for both groups during each fMRI session. It consisted of repeated passive extension-flexion of the right wrist. Subjects were instructed to stay motionless with eyes closed. Their arms were restrained to prevent any movement of the shoulder, arm, or elbow. Head motion was minimized by using foam padding with ear pads.
The passive movement was induced by traction on the right hand at a set rate (one complete movement per second) and calibrated amplitude (60°). The task paradigm consisted of eight 30-second epochs alternating between rest and activation, resulting in an acquisition time of 4 minutes per functional time series.
An electromyograph (using surface electrodes on radial muscles) was performed in the same conditions but outside of the MRI to check the absence of muscle activity during passive stimulation. Rest was the control state.
Training procedure
In the first group, after the first fMRI (E1) session, each of the six subjects underwent passive extension-flexion training. It consisted of repeated passive flexion-extension of their right wrist for 20 minutes per day, performed by the same operator (C. C), 5 days per week over 4 consecutive weeks. The rate and amplitude of the stimulation was similar to that used during fMRI acquisitions. A second fMRI (E2) session was performed after training with similar stimulation conditions.
The second group was considered as a control group, and the six volunteers underwent two fMRI sessions separated by 30 days (E'1 and E'2) but received no training procedure (Fig. 1).
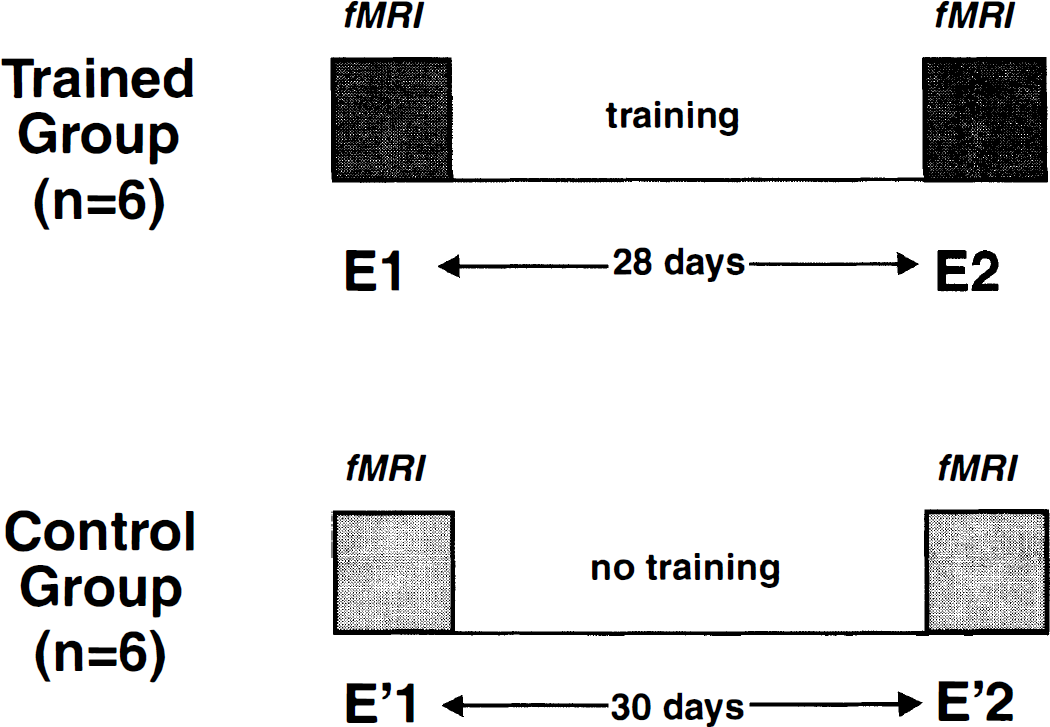
Protocol design.
Data acquisition
The MRI was performed on a 1.5-T scanner (Siemens Vision, Erlangen, Germany) equipped for echo-planar imaging. A three-dimensional, high-resolution, T1-weighted data set of the whole brain (3D MPRAGE; three-dimensional magnetization-prepared rapid acquisition gradient echo) was acquired for each volunteer (repetition time [TR] 15 milliseconds, echo time [TE] 7 milliseconds, inversion time [TI] 300; flip angle [FA] 12°; 30 × 30 cm field of view [FOV]; 256 × 256 matrix; voxel size = 1.17 × 1.17 × 1.17 mm3; 128 slices). After sagittal localization images, nine contiguous, 5-mm thick, axial supratentorial images were obtained, parallel to the bicommissural line of Talairach (AC-PC), and three contiguous images of the cerebellum were acquired (T2-weighted images, TR 418 milliseconds, TE 10 milliseconds, FA 30°, 20 × 20 cm FOV, 128 × 128 matrix, voxel size = 1.56 × 1.56 × 5 mm3, 12 slices).
For fMRI studies, blood oxygen level-dependent imaging was performed using a T2*-weighted single-shot echo-planar imaging sequence (TR 3 seconds, TE 64 milliseconds, FA 90°, 20 × 20 cm FOV, 128 × 128 matrix, voxel size = 1.56 × 1.56 × 5 mm3). Twelve slices (9 + 3) were acquired every 3 seconds, coinciding with the T2-weighted reference images.
fMRI postprocessing
Image analysis was carried out on a SPARC workstation (Sun Microsystems, Surrey, UK) using interactive image display software (Analyze, Biodynamics Research Unit, Mayo Clinic, Rochester, MN, U.S.A.), matlab (Math Works, Inc., Natick, MA, U.S.A.), and SPM96 4 2.c software (Wellcome Department of Cognitive Neurology, London, UK). The first four images of each time series were discarded to eliminate signal intensity variations arising from progressive saturation. Echo-planar imaging images were realigned to the first functional image of each time series to remove residual head movement. The functional images of each subject were coregistred, with the first functional image of one subject taken as a model. Images then were resized into the standard anatomic space defined by the atlas of Talairach and Tournoux (1988) for group analysis. The images were smoothed with a Gaussian kernel of full width, half maximum of 4 mm to reduce the anatomical and functional variability between the subjects.
Next, statistical parametric maps were generated to identify areas of interest that covaried positively with movement (Friston et al., 1995). The differences between baseline and activation were assessed with the t statistic at each voxel for each run of each subject separately. All images were inspected for evidence of misregistration. All subjects displayed activation foci in the contralateral primary sensorimotor cortex (S1M1).
First, we performed a group analysis for each session (E1, E2, E'1, E'2) comparing activation against rest. Tasks were averaged, giving statistical parametric maps of the difference activation minus rest (ACTI-REST). First, these maps were calculated for E1 (n = 6), E2, E'1 (n = 6), and E'2 and thresholded (P < 10−7 for peak height and P < 0.05 for the spatial extent of the activation foci corrected for multiple comparison). They were overlaid onto the anatomical MPRAGE images. Only changes in these activated areas were considered for the following statistics (the conditions E1 > 0 or E'1 > 0 are specified as a mask with SPM software).
Second, a time effect was defined in the control group by the comparison (E'2 – E'1), P < 0.01 for peak height corrected for multiple comparisons, corrected for spatial extent (25 connected voxels).
Third, we performed a direct comparison (E2 – E1) in the trained group whose result combined time and training effects (P < 0.01 for peak height corrected for multiple comparisons, corrected for spatial extent).
Then, to assess the training effect, the comparison (E2 – E1) corrected by the time effect (E'2 – E'1) was performed. The statistic (E2 – E1) – (E'2 – E'1) was made (P < 0.01 for peak height uncorrected for multiple comparisons, corrected for spatial extent, 25 connected voxels).
RESULTS
No muscular activity was detected during passive movements on electromyographic recordings.
One-session activation map
During passive wrist movements, compared with rest, there were strong significant activation in contralateral primary sensorimotor cortex (S1M1), SMA proper, cingulum, ipsilateral cerebellum, and bilaterally inferior parietal lobe (BA40, including supramarginal gyrus) (Fig. 2).
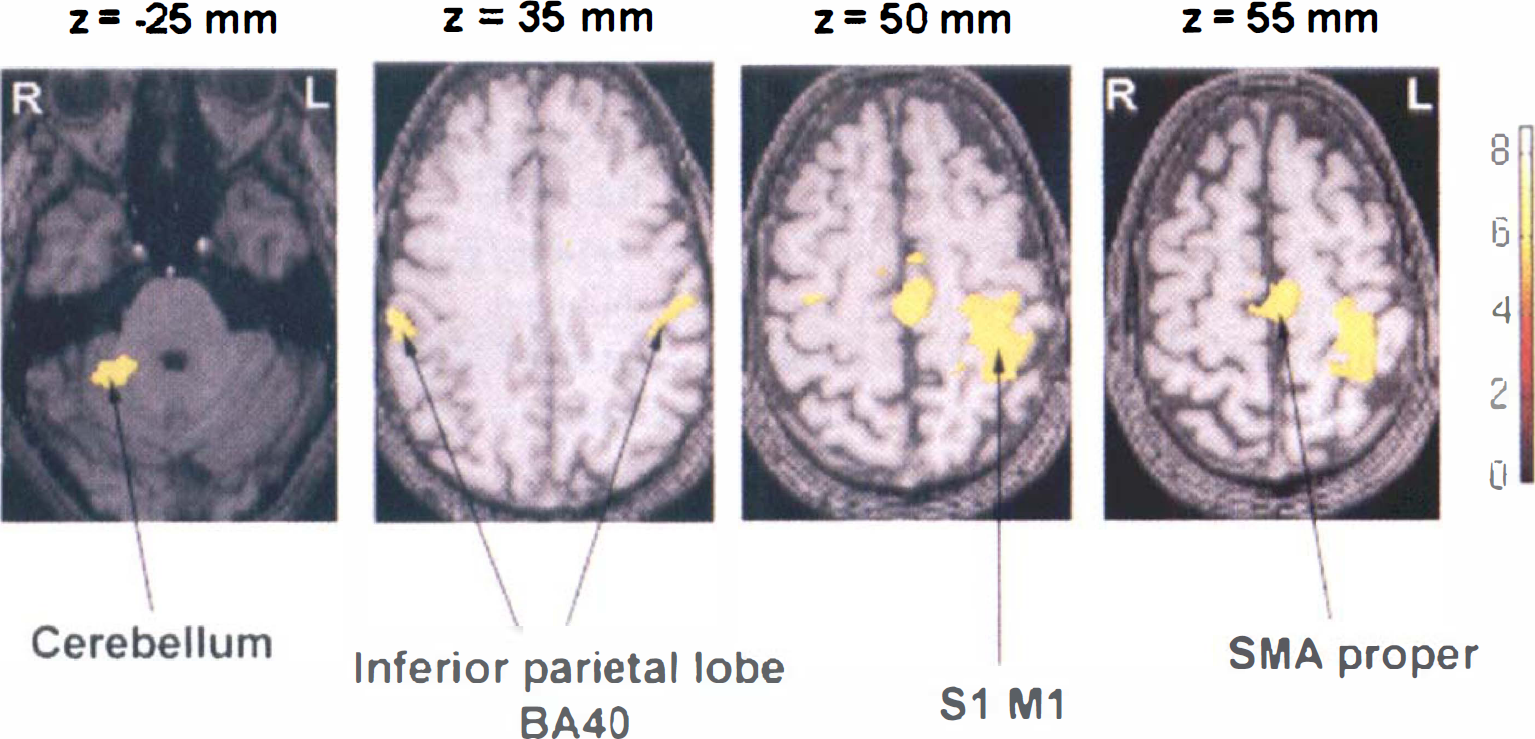
Areas of significant activation during passive movement of the right wrist compared with rest in a group of six normal subjects. The right side of the image corresponds to the left side of the brain. The Z number refers to the distance in millimeters with respect to the intercomissural line, which is at 0 mm. The pixels in yellow show level of statistical significance above P < 10−7 for peak height and < 0.05 for the spatial extent of the activation foci. A corresponding Z score bar is provided.
Interaction between time and movement: time effects
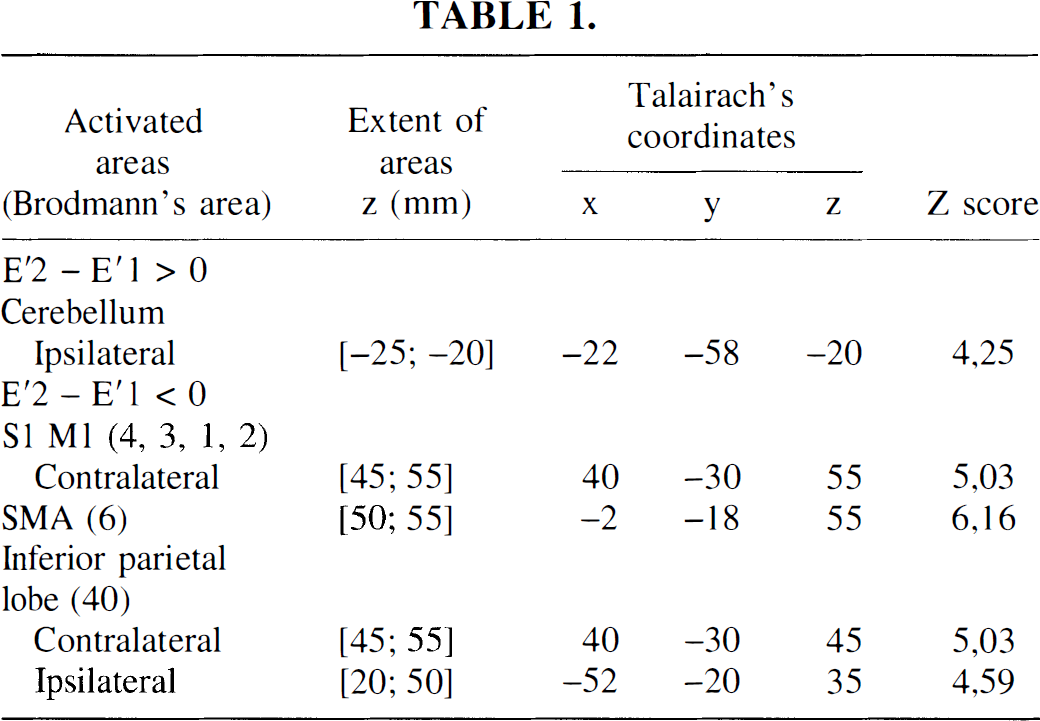
The activation map of the statistical differences between the second and first fMRI examination in the control group (E'2 – E'1) resulted in an increase of ipsilateral cerebellum activation, whereas a decrease of activation was observed for contralateral sensorimotor primary cortex, SMA proper, and bilaterally inferior parietal lobe (BA40) (Fig. 3, Table 1).
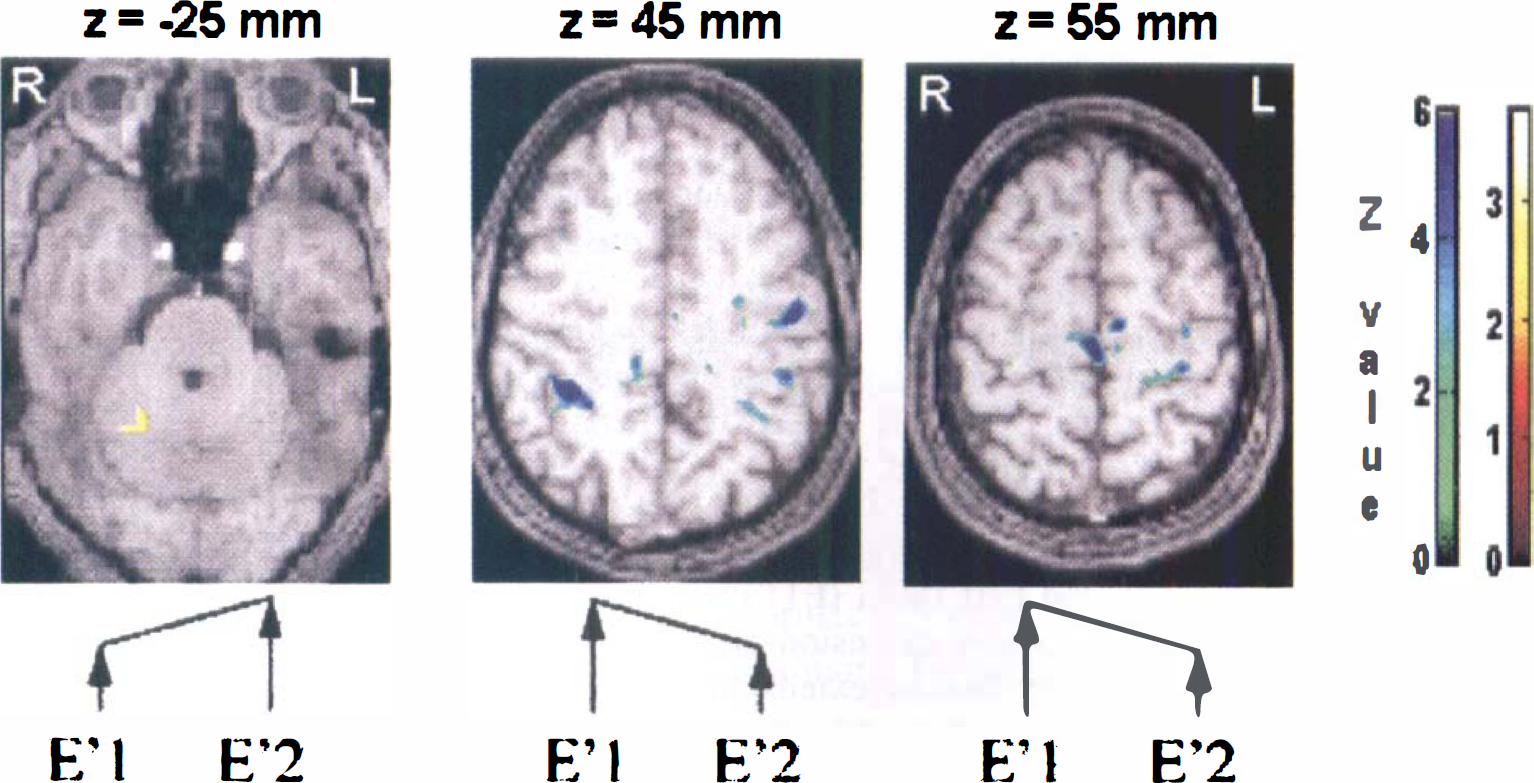
Time effect maps. Activation map of the statistical differences between results of the second (E'2) and first (E'1) functional magnetic resonance imaging (fMRI) examination in the control group of six volunteers. P < 10−5 for peak height corrected for spatial extent.
Interaction between time and training: training effects
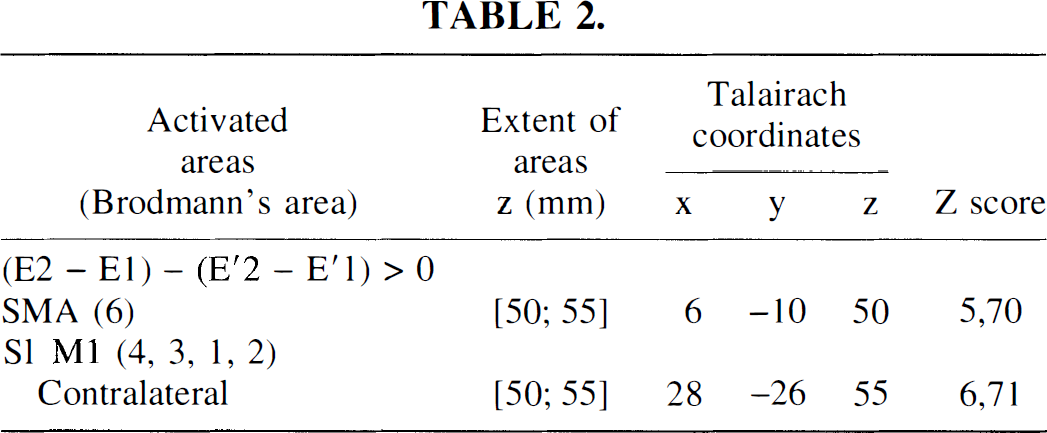
The activation map of the statistical differences between the second and first fMRI examination in the trained group (E2 – E1) showed a decrease of activation in supratentorial recruited areas similar to those observed in “time effects” maps. Moreover, an increase in activation was observed in a rostral part of SMA proper (+4, −12, +50 mm; Z score = 5.31) and in contralateral M1 (+30, −26, +55 mm; Z score = 5.16). Increases in activation had never been observed in supratentorial areas in the control group.
To assess the training effect, the comparison (E2 – E1) corrected by the time effect (E'2 – E'1) was performed, showing a large increase of activation in SMA proper (+6, −10, +50 mm; Z score = 5.7) and in contralateral S1M1 (+28, −26, +55 mm; Z score = 6.71). These increases corresponded to the increases obtained with the direct contrast (E2 – E1), validating a real training effect. All subjects (n = 6) underwent this training effect (Fig. 4, Table 2).
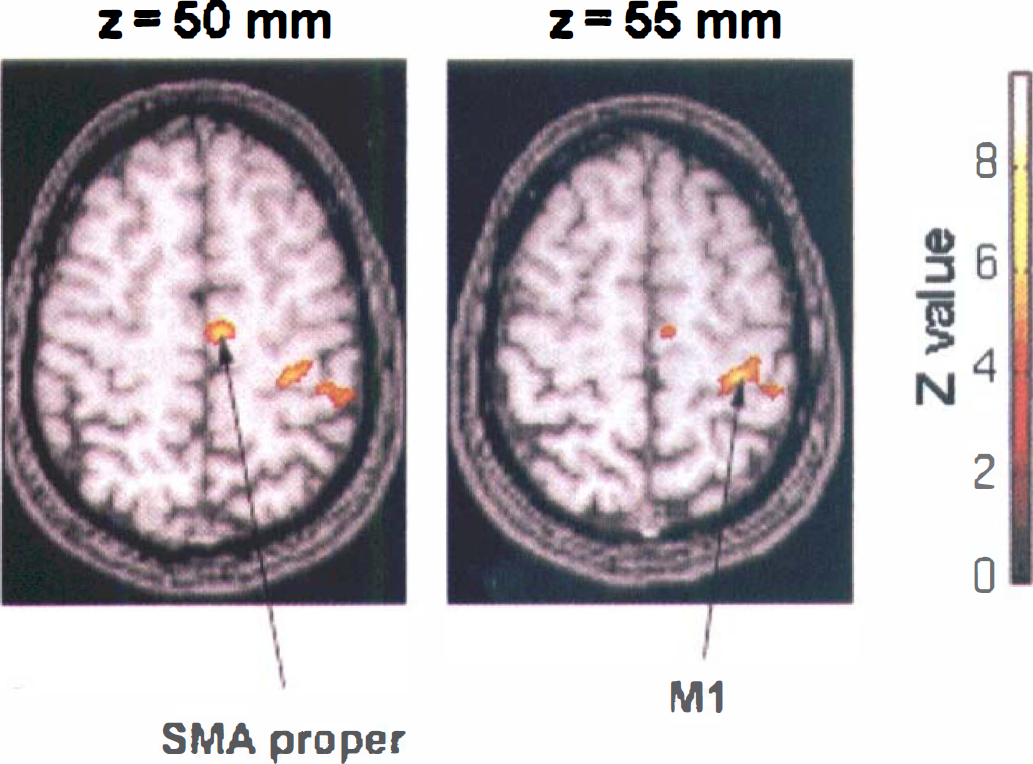
Training effect maps. Activation map of the statistical differences between results of the second (E2) and first (E1) fMRI examination in the trained group of six volunteers corrected by the time effect. P < 10−5 for peak height corrected for spatial extent.
Figure 5 shows the plots of the local maximum signal in contralateral S1M1 through E'1, E'2, E1, and E2 examinations. Results are expressed as a percentage [(x – meanRest)/meanRest], where meanRest is a mean value for E'1, E'2, E1, and E2 rest values. These plots illustrate an increase in signal activation after training: (E2 – E1) – (E'2 – E'1) > 0 (x = 28, y = −26, z = 55; Z score = 6.71). A time effect is visible at this point: E'2 – E'1 < 0 (i.e., [(ACTI – REST)E'2 – (ACTI – REST)E'1] < 0), and training reversed such effect: E2 – E1 > 0 (i.e., [(ACTI – REST)E2 – (ACTI – REST)E1] > 0). Individual pattern is provided for the same spot.
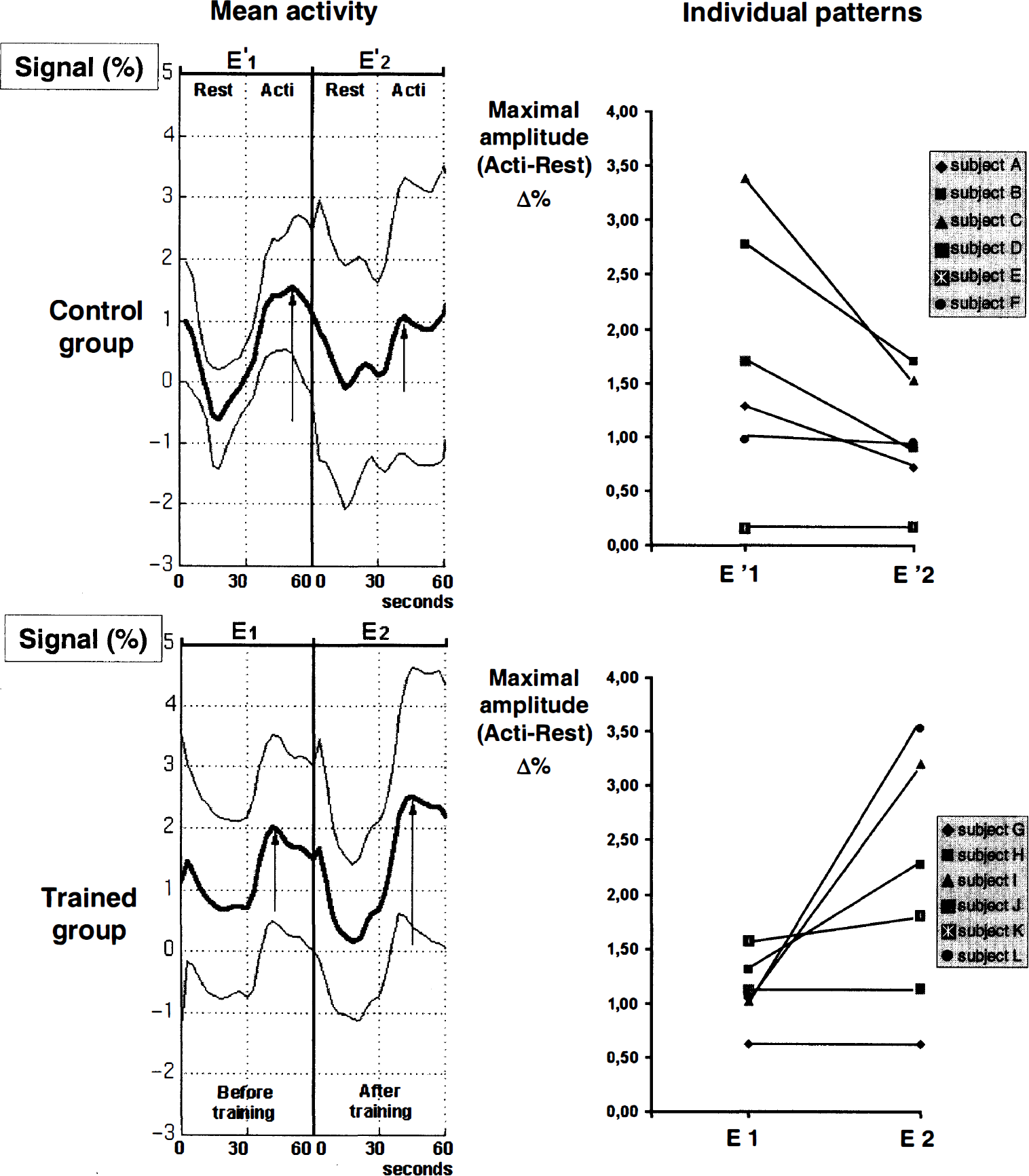
Plots of the local maximum signal in the contralateral sensorimotor cortex S1M1, whose signal changes through E'1, E'2, E1, and E2 examinations (mean ± SD) and individual patterns.
DISCUSSION
We show in this study that prolonged passive training can change the cortical representation of passive movements and can induce over-activation of primary sensorimotor cortex and SMA.
In this study, we used repeated and calibrated flexion-extension of the wrist. This passive movement was chosen because it was recognized as being close to the passive movements performed by physiotherapists during rehabilitation procedures in hemiplegic patients and because it represents a sensory stimulation with high proprioceptive component. The activation maps that we obtained when this movement was compared with rest showed activation of contralateral primary sensorimotor cortex, SMA, and ipsilateral cerebellum, but also BA40 and cingulate gyrus, as previously found with an active movement (Catalan et al., 1998; Colebatch et al., 1991; Rao et al., 1996). Although the movement was not strictly the same, our data are similar to those observed by Weiller et al. (1998) using positron emission tomography scan (Weiller et al., 1996) and by Alary et al. in our group using evoked related potentials and fMRI (Alary et al., 1998). The only difference we found was the activation of the caudal part of cingulate gyrus, which was not present in the passive task of Weiller's study. This caudal part (posterior to the AC line) is activated in motor task execution and in sensory stimulation (Picard and Strick, 1996; Talbot et al., 1991).
Control group and time effect
We thought it was preferable to have a control group with no chronic training in this study. We already found differences between the results of two repeated identical fMRI examinations in a previous study (Loubinoux et al., 1999), and such methodologic aspects seem to be highly important when later assessing the only modifications of cortical activation induced directly by training. We found no published complete studies assessing the reproducibility of fMRI signals between two experiments separated by several weeks. Nevertheless, a reduced extent of cortical primary motor activated areas was demonstrated when 5 to 16 weeks separated the experiments (Ramsey et al., 1996). Loubinoux et al. (1999) found similar reduced cortical and enhanced cerebellar activity in normal subjects doing overlearned active motor tasks 5 hours apart. Karni et al. (1995) also showed differences between a first and second measurement in normal subjects performing active sequential movements (Karni et al., 1995), and they found them consistent with what they considered to be a habituation effect. In learning functional imaging studies, neural responses decreases to repeated stimuli presentations have been found (Büchel et al., 1999). This is believed to reflect a progressive optimization of neuronal responses elicited by the task (Haier et al., 1992). They showed that, in parallel to this adaptation, increase in effective connectivity occurred between distinct cortical systems. “Time effect” in the control group could be indicative of an increase in effective connectivity. Although the data are comparable, the current study differs, first because passive movement stimulation was used, and second because a long time (4 weeks) separated the two sessions. The use of passive movement showed that no active task is needed to observe a habituation phenomenon. This was well demonstrated in early electrophysiologic studies with evoked related potentials and electric stimulation of peripheral nerves (Allison et al., 1962; Mauguière and Fischer, 1990). The effect observed after the long period between the two sessions remains puzzling. Karni et al. (1998) and Brashers-Krug et al. (1996) demonstrated that some gains continue to develop after motor practice has ended and require time to become effective (Brashers-Krug et al., 1996; Karni et al., 1998). This suggests that a long-term memory representation of the stimulation has been implemented into the motor system. Cerebellum that showed greater activation at the second experiment could play a key role, since it would assume a greater role as the task becomes “automatic” (Shadmehr et al., 1997). It would participate in a long-term modulation of cortical activity. It also might be that this effect has little to do with time but with an order effect and remains closer to a test—retest effect than a time effect. Systematic studies of fMRI activations in controlled conditions across time are needed. We postulate that the “time” effect was comparable in our control group and in our trained group.
Training effect
Our trained subjects, when compared with the control group, showed greater activation in primary sensorimotor cortex and in SMA. We think that these changes are related to the 4-week passive training. Several studies about training-induced plasticity of the brain and using positron emission tomography and fMRI appear in the literature. Most studies deal with active training or active learning procedures. It is important to distinguish changes that occur during the training or learning phase from changes detected after this phase when the skill is definitely acquired and has become routine. The former concern working memory systems, whereas the latter concern storage (i.e., long-term memory). We were, in this study, interested in long-term changes occurring after the training period. Increases in activation of primary motor cortex and SMA have been shown during the active phase of motor learning (Grafton et al., 1992). Most of these studies showed that cerebellum, putamen, cingulum, and prefrontal cortex were active during the learning period (Friston et al., 1991; Jenkins et al., 1994; Jueptner et al., 1997; Seitz et al., 1990). We found no significant hyperactivation or hypoactivation of the cerebellum, which was recognized as being activated in many learning experiments. We think that the cerebellum probably is more active during the early phase of training than after. Karni et al. (1995) used fMRI to study the long-term influence of motor learning on the primary sensorimotor cortex of healthy subjects. They found that training and learning resulted in a larger area of activation that persisted for several months. This study suggests a slowly evolving, long-term, experience-dependent reorganization of adult M1, which may underlie the acquisition and retention of the motor skill.
Studies interested in other “sensory training” have been performed (Rauschecker et al., 1999). They show reorganization in cerebral activation with expansion of the areas corresponding to stimuli (Jenkins et al., 1990; Recanzone et al., 1993). Our study differs from others because the training did not correspond to a “classic” learning process. No obvious gain in sensorimotor performance and no new particular skill were acquired during this training. The motor task was passive and could not be qualified as an implicit or explicit task. No attentional component was required during the stimulation, not only in the trained group but also in the control group. Moreover, attention, if any, is expected to decrease in the trained group. Meyer et al. (1991) showed that CBF was increased in the primary sensorimotor cortex of subjects performing a motor task when an attentional component was introduced. Thus, an attentional component cannot explain the increased activity in primary sensorimotor cortex during the second fMRI examination of the trained subjects.
Despite these differences, we observed an increased activity in primary motor cortex after passive-movement training, as Karni et al. (1995) observed in comparable but active-movement training. Our results suggest that proprioceptive inputs probably play a major role in the observed changes in the primary motor cortex. We think that the primary sensorimotor cortex and SMA become overactive after training, however active or passive the training may be.
The maximum activity of the SMA in the training effect maps was located behind the anterior commissural line according to Talairach coordinates (Table 2). The SMA is a complex structure that has been subdivided into multiple parts (Tanji, 1994). Its anterior part, which also is called preSMA, or “cognitive SMA,” is thought to play a role in the initial phases of motor learning and in the initiation of movement, whereas the posterior part, also called “SMA proper,” deals with motor execution of simple and complex sequences (Picard and Strick, 1996). We found that the peak of hyperactivity was located in the posterior part of the SMA. We think that the repeated proprioceptive inputs delivered during the training of our subjects would facilitate the late stages of preparation of movement and its execution, even with a passive movement.
We demonstrate in this study that plastic changes in primary sensorimotor cortex and SMA can be provoked by chronically repeated proprioceptive inputs. It might represent a neural substrate for rehabilitation procedures. Whether these changes can be correlated to clinical recovery in patients remains to be determined.
