Abstract
The angiopoietin/Tie receptor system may contribute to angiogenesis and vascular remodeling by mediating interactions of endothelial cells with smooth muscle cells and pericytes. The temporal expression of angiopoietin-1 (Angpo-1), angiopoietin-2 (Angpo-2), Tie-1, and Tie-2 mRNA was studied in a focal cerebral ischemia model in rats. The cDNA fragments obtained from reverse transcription polymerase chain reaction amplification were cloned and used as a probe to detect individual genes. Northern blot analysis showed a delayed increase of a 4.4-kb Angpo-1 transcript for up to 2 weeks after ischemia, eightfold higher than the values of the shamoperated controls. A biphasic expression of a 2.4-kb Angpo-2 transcript was noted, peaking at 24 hours (6.4-fold) and 2 weeks (4.6- fold) after ischemia. The expression of Tie-2 mRNA (4.3 kb), a receptor for Angpo-1, and Tie-1 mRNA (4.3 kb) also increased starting 24 hours after reperfusion and remained elevated for up to 2 weeks after ischemia. The temporal profiles of the expression of these genes were different from those of other angiogenic genes such as basic fibrobast growth factor/fibroblast growth factor receptor and vascular endothelial growth factor/vascular endothelial growth factor receptor and proteolytic enzymes (tissue-type plasminogen activator and urokinase plasminogen activator) and their inhibitors (plasminogen activator inhibitor-1). The expression patterns of these genes could be related to progressive tissue liquefaction and neovascularization after ischemia in this stroke model. Differential expression of these angiogenesis genes suggests the involvement of complex regulatory mechanisms that remain to be characterized.
Keywords
The development of vascular supply is a fundamental requirement for organ development and differentiation during embryogenesis as well as for wound healing and reproductive functions in the adult (Folkman, 1995). Angiogenesis has also been implicated in the pathogenesis of a variety of disorders: proliferative retinopathies seen in age-related macula degeneration, tumors, rheumatoid arthritis, and psoriasis. Angiogenic activity reflects a balance of positive and negative regulators of blood vessel growth. The search for these modulators of angiogenesis has yielded several candidates. However, uncertainty remains on the specific roles of endogenous mediators of angiogenesis (Pepper, 1996; O'Reilly et al., 1997; Risau, 1997).
Tie-1 and Tie-2 are receptor tyrosine kinases (RTKs) that are expressed exclusively in the endothelial cell (Mustonen and Alitalo, 1995). The restricted expression in endothelial cells suggests that the signaling pathways mediated by these RTKs play key roles in vascular function. Tie-1 and Tie-2 are encoded by distinct genes on chromosome 4. The deduced amino acid sequences of mouse Tie-1 and Tie-2 contain 1,134 and 1,122 amino acids, respectively. Sequence analysis of these two receptors reveals high sequence conservation in the cytoplasmic region with considerable divergence in the extracellular ligand-binding domain (Dumont et al., 1992; Sato et al., 1993). Engineered mutations of mouse genes coding for Tie-1, Tie-2, and some of their ligands result in distinct vascular defects, demonstrating that the signaling pathways mediated by these RTKs play specific roles in endothelial cell biology (Dumont et al., 1994; Sato et al., 1995; Beck and D'Amore, 1997).
The ligand for the Tie-2 receptor has been cloned and named angiopoietin-1 (Angpo-1) (Davis et al., 1996). However, ligands for the Tie-1 receptor are not presently known. Angiopoietin-1 contains 498 amino acids, including an N-terminal secretory signal sequence. Two regions within the coding sequence display homology to myosin and the C-terminus of fibrinogen, respectively. The pattern of Angpo-1 expression in the embryo suggests its important role in the developing vasculature (Davis et al., 1996; Suri et al., 1996). Mice lacking Angpo-1 display a characteristic vascular defect that is distinguished from that caused by vascular endothelial growth factor (VEGF) gene deletion, but they are reminiscent of Tie-2 gene knockout mice (Sato et al., 1995). They appear grossly abnormal by embryonic day 11 and die by embryonic day 12.5. Even though the total number of endothelial cells seems unaffected, the number of large vessels and their caliber are reduced with fewer and straighter branches than in the wild type. Angiopoietin-2 (Angpo-2) comprises 496 amino acids with a secretion signal peptide and is ∼60% identical to Angpo-1 (Maisonpierre et al., 1997). In contrast to Angpo-1, which stimulates phosphorylation of Tie-2 in a variety of endothelial cell lines, Angpo-2 fails to elicit the same response. In fact, Angpo-2 blocks Angpo-1 activation of the Tie-2 receptor, suggesting that a biological role of Angpo-2 may be to antagonize Angpo-1 activation of Tie-2. The Angpo-2-positive cells are likely to be endothelial cells or pericytes (Hirschi and D'Amore, 1996). Transgenic embryos overexpressing Angpo-2 were found to die on embryonic days 9.5 to 10.5, and the defect in these embryos was similar to that in mice lacking either Angpo-1 or Tie-2 receptor.
The expression of Tie-1 and Tie-2 persists in quiescent adult endothelial cells, although at reduced levels in some organs such as the brain and heart (Dumont et al., 1992). The expression of Tie-1 is increased during maturation of ovarian follicles, wound healing, and tumor angiogenesis (Hatva et al., 1994). However, it is not clear whether the observed Tie-1 expression is a cause or consequence of neovascularization.
Angiogenesis occurs in the ischemic brain, particularly in the ischemic penumbra. The extent of angiogenesis has been correlated to survival in stroke patients (Krupinski et al., 1994). To our knowledge, expression of Angpo/Tie receptor system genes in the ischemic brain has not been previously reported. The purpose of the present study is to examine ischemia-induced expression of Angpo and Tie receptor mRNA in the brain in relation to other genes that may contribute to angiogenesis.
MATERIALS AND METHODS
Stroke model
The focal cerebral ischemia-reperfusion model in the rat has been described previously (Chen et al., 1986; Lin et al., 1993). In brief, male Long-Evans rats weighing 250 to 300 g were anesthetized with chloral hydrate (360 mg/kg body weight, intraperitoneally). The trunk of the right middle cerebral artery (MCA) above the rhinal fissure was identified under a stereomicroscope and ligated with a 10-0 suture. Interruption of blood flow distal to ligation was confirmed under the microscope. Both common carotid arteries were then occluded using nontraumatic aneurysm clips. After 60 minutes of ischemia, the aneurysm clips and the suture were removed and restoration of blood flow in all three arteries was verified. While under anesthesia, the rectal temperature was monitored and maintained at 37.0 ± 0.5°C using a homeothermic blanket (Harvard, Holliston, MA, U.S.A.). In this model, ischemia for 60 minutes consistently resulted in a large infarct confined to the right MCA cortex (Liu et al., 1989). No morphological (Liu et al., 1989) or biochemical evidence of ischemic brain injury was noted in the left MCA cortex (An et al., 1993). After the ischemic insult, rats were kept in an air-ventilated incubator at 24.0 ± 0.5°C for up to 3 weeks and were provided with water and lab chow ad libitum until the end of experiments. At the end of each experiment (30 and 60 minutes after onset of ischemia or 30, 60, 90 minutes, 4, 12 hours, 1, 3 days, and 1, 2, and 3 weeks after reperfusion), rats were killed by decapitation under anesthesia, and the brain was quickly removed to collect the cerebral cortex. The right or left MCA cortices were immediately frozen in liquid nitrogen and stored at −70°C until further processing. The right MCA cortex was also sampled from animals subjected to vascular surgeries without occlusion to serve as sham-operated control. In some experiments, photography of the whole brain was made for gross visual assessment of the extent of angiogenesis. All procedures were approved by our institutional Animal Studies Committee and were in accordance with the Public Health Service Guide for the Care and Use of Laboratory Animals, USDA Regulations, and the Guidelines of the American Veterinary Medical Association Panel on Euthanasia.
RNA isolation and reverse transcription
Total RNA was isolated from frozen cerebral cortex using the single-step acid guanidinium thiocyanate/phenol/chloroform extraction method as previously described (An et al., 1993; Lin et al., 1996). Total RNA (1 μg) was incubated with 200 U of Moloney-Murine Leukemia Virus reverse transcriptase (RTase; Clontech, Palo Alto, CA, U.S.A.) in a buffer containing a final concentration of 50 mmol/L Tris-Cl (pH 8.3), 75 mmol/L KCl, 3 mmol/L MgCl2, 20 U of RNase inhibitor, 1 μmol/L polydT oligomer, and 0.5 mmol/L of each dNTP in a final volume of 20 μL. The reaction mixture was incubated at 42°C for 1 hour and then at 94°C for 5 minutes to inactivate the enzyme. A total of 80 μL of diethyl pyrocarbonate treated water was added to the reaction mixture before storage at −70°C (Lin et al., 1998).
Polymerase chain reaction
Polymerase chain reaction (PCR) primer sequences were chosen from published documents as listed in Table 1. In brief, 5 μL of the reverse transcription (RT) reaction solution was used in the PCR. Polymerase chain reaction was carried out in a final volume of 50 μL containing 200 μmol/L each of dATP, dCTP, dGTP, and dTTP, 5 pmol of each primer, 1.25 U of Taq polymerase (BRL, Grand Island, NY, U.S.A.), 20 mmol/L Tris-Cl (pH 8.4), 1.5 mmol/L MgCl2, and 50 mmol/L KCl. The mixture was incubated in a thermal cycler for 35 cycles using the following profile: 94°C for 7 minutes, then repeat cycles of 94°C for 45 seconds, 55°C for 45 seconds, and 72°C for 90 seconds. Samples were then incubated at 72°C for 7 minutes and cooled to 4°C (GeneAmp 2400, PE, Foster City, CA, U.S.A.) (Lin et al., 1998). Polymerase chain reaction products were run on 2% agarose gel for DNA fragment size verification, then eluted and subcloned (Invitrogen, CH Groningen, The Netherlands) for sequence identity (Fig. 1), and served as probe to detect respective mRNA in Northern blot analysis.
Oligonucleotide sequence of amplification primers for PCR

1. Mol Cell Endocrin (1994) 104:191–200; 2. Endocrinology (1996) 135:589–5596; 3. Oncogene (1994) 9:2683–2690; 4. J Biol Chem (1998) 273:2090–2097; 5. Circ Res (1998) 83:852–859; 6. GenBank (#AF030377); 7. Atheroscleresis (1998) 137:267–275; 8. J Clin Invest (1996) 97:2440–2451; 9. J Cereb Blood Flow Metab (1997) 17:136–146; 10. J Ren Fert (1994) 101:235–240; 11. GenBank (#X71898).
Cloning protocol for direct inserts of polymerase chain reaction (PCR) products into a plasmid vector for sequence identity and generation of sense and antisense probes.
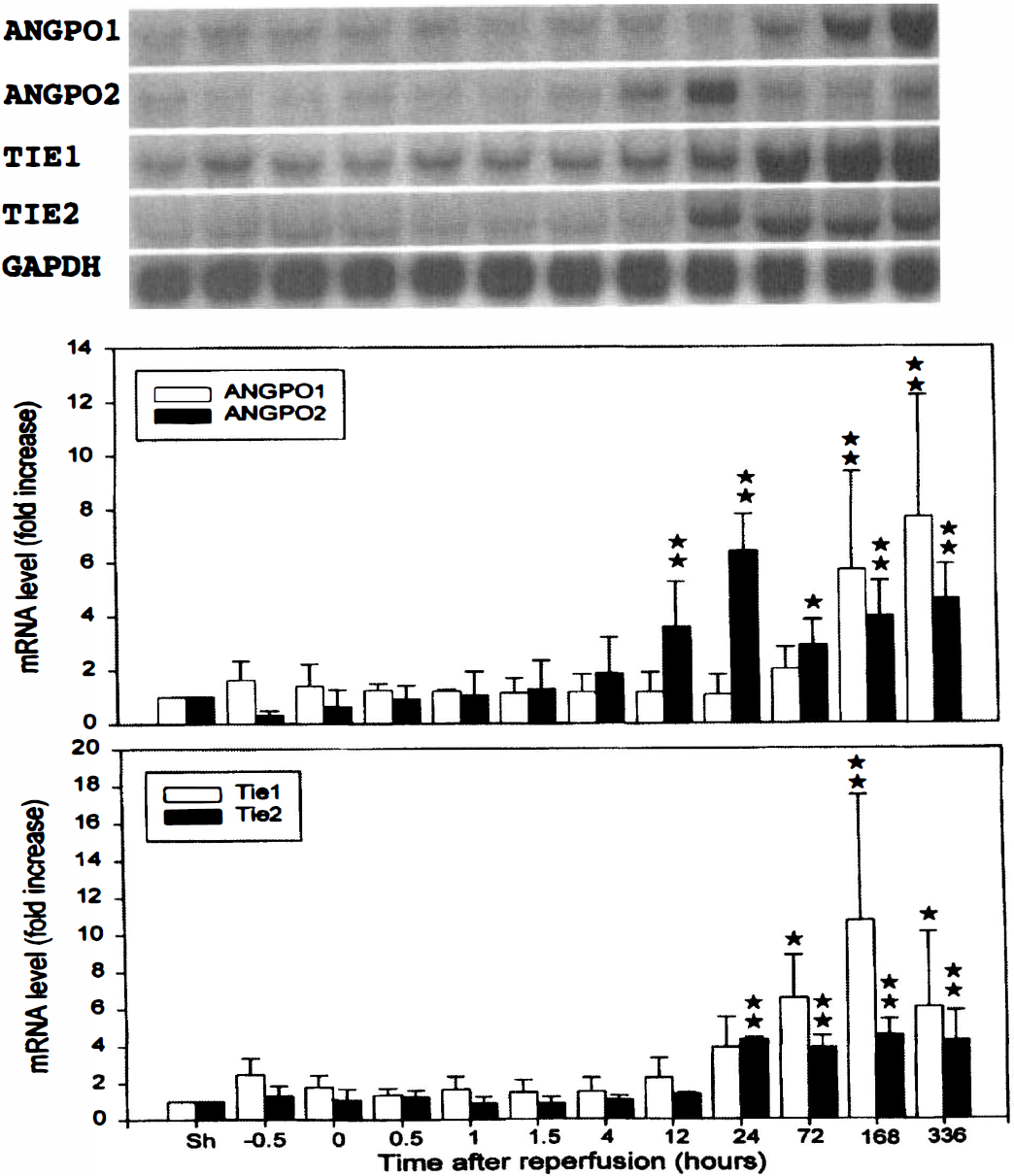
Time courses of angiopoietin-1 (Angpo-1), Angpo-2, Tie-1, and Tie-2 mRNA expression after focal cerebral ischemia-reperfusion. Transient ischemia for 60 minutes resulted in a significant increase in mRNA levels of Angpo-1 (4.4 kb), Angpo-2 (2.4 kb), Tie-1 (4.3 kb), and Tie-2 (4.3 kb) in the ischemic right middle cerebral artery (MCA) cortex. The same blots were subsequently stripped and rehybridized with glyceraldehyde-3-phosphate dehydrogenase to serve as internal controls. The radioactive bands were quantified and normalized with those derived from glyceraldehyde-3-phosphate dehydrogenase mRNA. Value obtained from the sham-operated control was arbitrarily defined as 1. Data were means ± SD from four animals. *P < 0.05, **P < 0.01, significant differences from the sham-operated controls. Sh denotes sham-operated controls and −0.5 and 0 represent 30 and 60 minutes after onset of ischemia or 30 and 0 minutes before the initiation of reperfusion, respectively. 0.5, 1, 1.5, 4, 12, 24, 72, 168, and 336 denote 0.5, 1, 1.5, 4, 12, 24, 72, 168, and 336 hours of reperfusion following 60 minutes of ischemia, respectively.
Northern blot analysis
Northern blot analysis has been described previously (An et al., 1993; Lin et al., 1996). In brief, RNA samples (15 μg/lane) were applied to 1.2% agarose gel in the presence of 2.2 mol/L formaldehyde. After electrophoresis, gels were transblotted onto Nytran membranes (Gene Screen Plus; Du Pont, Boston, MA, U.S.A.). Membranes were prehybridized at 60°C in a solution containing 1% sodium dodecyl sulfate, 1 mol/L NaCl, 10% dextran sulfate, and 100 μg/mL sheared salmon sperm DNA. Reverse transcription PCR-amplified cDNA probes, fibroblast growth factor receptor (FGFR) cDNA (a gift from Dr J. Milbrandt) (Wanaka et al., 1990), and glyceraldehyde-3-phosphate dehydrogenase cDNA probes (Fort et al., 1985) were labeled with [32P]dCTP using the random primer labeling method (Amersham, Arlington Heights, IL, U.S.A.). Radioactive probes (1 × 106 cpm/mL) were added directly to the prehybridization solution. Following overnight hybridization at 60°C, membranes were washed twice in 2× saline/sodium citrate at room temperature for 5 minutes each, followed by two 30-minute washes at 60°C in 2× saline/sodium citrate/1% sodium dodecyl sulfate and two 30-minute washes at 60°C in 0.1× saline/sodium citrate. Membranes were then exposed to Kodak X-Omat/XB-1 film. The radioactive bands were quantified by a densitometer.
Chemicals
All chemicals were of reagent-grade purity and purchased either from E. Merck (Darmstadt, Denmark) or Sigma Chemical Co. (St. Louis, MO, U.S.A.), unless otherwise indicated.
Statistics
One-way analysis of variance was used to compare the temporal expression of mRNAs. The level of significance for differences between groups was further analyzed with post hoc Fisher-protected t tests using statistical software (GB-STAT 5.0.4; Dynamic Microsystem, Silver Springs, MD, U.S.A.). A P value of <0.05 was considered significant.
RESULTS
Every oligonucleotide sequence pair used for PCR amplification resulted in a single cDNA fragment when visualized with ethidium bromide on agarose gel with the respective size shown in Table 1. A representative PCR profile is presented in Fig. 1 (top). The expression pattern of Tie-1 mRNA obtained from RT-PCR was similar to that derived from Northern blot analysis shown in Fig. 2. The cDNA fragments derived from PCR were eluted and subcloned into pCRII, which provides a one-step cloning strategy for direct insertion of PCR products into plasmid vector (Invitrogen). Each amplified cDNA fragment was subjected to DNA sequencer analysis (ABI 377, PE) to establish sequence identity in Genbank. Results indicate a very high fidelity for PCR amplification (data not shown). These cDNA fragments served as probes to detect the respective angiogenesis genes in subsequent studies.
The temporal expression profiles of Angpo-1, Angpo-2, Tie-1, and Tie-2 mRNA following ischemia-reperfusion were examined by Northern blot analysis (Fig. 2). A single species of 4.4-kb Angpo-1 transcript in the adult rat brain was recognized by the PCR-amplified probe. Angpo-1 mRNA could be detected in the right MCA cortex of sham-operated rats. Induction of Angpo-1 mRNA expression in the ischemic right MCA cortex was delayed, with significant increase noted starting 1 week (168 hours) after reperfusion. Quantitative analysis indicated an eightfold increase 2 weeks after ischemia as compared with the sham-operated controls. A single Angpo-2 mRNA species of ∼2.4-kb size was also detected in the right MCA cortex of the sham-operated controls. Ischemia increased Angpo-2 mRNA expression in the right MCA cortex. However, the temporal expression of Angpo-2 mRNA was different from that of Angpo-1. Significant increase in Angpo-2 mRNA expression was observed as early as 12 hours, peaking at 24 hours after ischemia. It declined at 72 hours but remained above basal level. A second peak was noted toward the end of the second week. At its peak, there was a 6.4-fold increase as compared with the sham-operated controls.
The PCR-amplified Tie-1 probe recognized a 4.3-kb transcript in the sham-operated controls. Ischemia induced a delayed increase in Tie-1 mRNA in the ischemic right MCA cortex starting at 72 hours and peaking at 1 week after ischemia (10-fold increase). The Tie-1 mRNA levels remained elevated for up to 2 weeks after ischemia. A similar induction profile was noted for a 4.3-kb Tie-2 transcript in the ischemic right MCA cortex. Significant increase in Tie-2 mRNA levels was observed at 24 hours after ischemia and remained at about the same level for up to 2 weeks with peak increase of approximately fourfold.
To explore the temporal relation of Angpo/Tie expression with other genes involved in angiogenesis, basic fibroblast growth factor (bFGF), FGFR, VEGF, VEGF receptor-1 (VEGFR-1), VEGFR-2, transforming growth factor-β1 (TGF-β1), plasminogen activator inhibitor-1 (PAI-1), tissue-type plasminogen activator (tPA), urokinase plasminogen activator (uPA), and uPA receptor (uPAR) were also probed by Northern blot analysis (Fig. 3). The bFGF mRNA was transiently induced and peaked between 12 and 24 hours after ischemia, whereas the expression of FGFR mRNA was delayed, peaking between 3 days and 1 week after ischemia. Similar induction profiles were obtained using established cDNA or PCR-amplified probes. Unlike bFGF, VEGF mRNA was increased much earlier throughout the ischemic period, extending into the early period of reperfusion. A sharp decrease in VEGF mRNA was noted at 4 hours after ischemia. Interestingly, the level of VEGF mRNA rose again later, peaking at 12 hours after ischemia, and then slowly declined thereafter. It decreased to below preischemic level at 1 week. Two weeks after ischemia, VEGF mRNA was back to the basal level. Very similar temporal profiles were noted for its receptors, VEGFR-1 and VEGFR-2, except that both VEGFR-1 and VEGFR-2 mRNA peaked at 1 day after ischemia. The PCR-amplified VEGFR-1 probe recognized both VEGFR-1 (the upper band) and VEGFR-2 (the lower band) mRNA, whereas the PCR-amplified VEGFR-2 probe recognized only VEGFR-2 mRNA. Ischemia caused a delayed induction of TGF-β1 mRNA, peaking at 1 week after ischemia. The PAI-1 mRNA was transiently increased after ischemia, peaking at 1 day. Similar to VEGF mRNA, biphasic expression of tPA mRNA was noted peaking at 60 minutes and 1 day, respectively. Unlike tPA, the induction of uPA mRNA was delayed. It peaked at 1 week. Its receptor, uPAR, mRNA, however, was induced earlier, peaking at 1 day after ischemia.
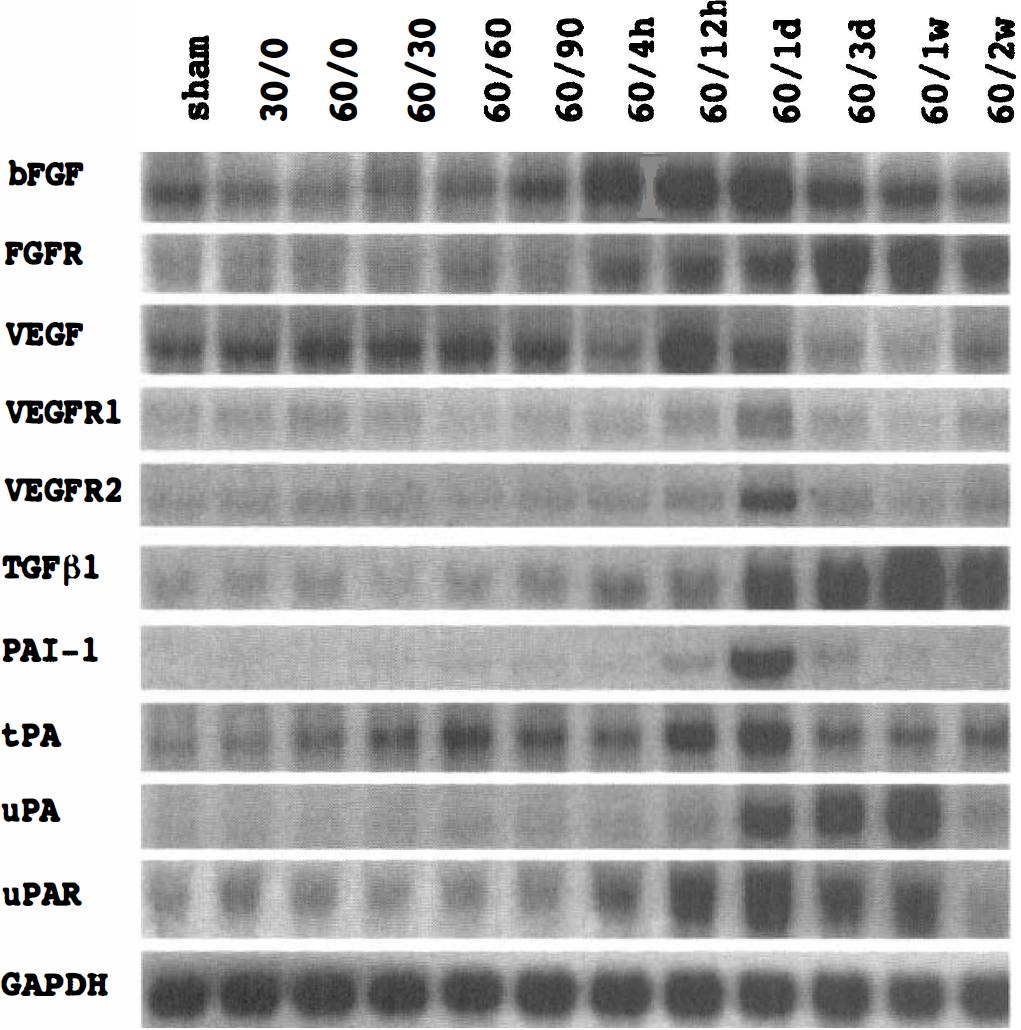
Representative expression profile of basic fibroblast growth factor (bFGF), fibroblast growth factor receptor (FGFR), vascular endothelial growth factor (VEGF), vascular endothelial growth factor receptor-1 (VEGFR-1), vascular endothelial growth factor receptor-2 (VEGFR-2), transforming growth factor-β1 (TGF-β1), plasminogen activator inhibitor-1 (PAI-1), tissue-type plasminogen activator (tPA), urokinase plasminogen activator (uPA), and urokinase plasminogen activator receptor (uPAR) mRNA after focal cerebral ischemia-reperfusion in a time course study as described in Fig. 2. A representative of triplicate experiments with similar results is shown for each gene.
Figure 4 shows the dorsal view of cerebral blood vessels at various time points (12 hours to 3 weeks) after ischemia as compared with a sham-operated control. Vessel dilatation was noted after 12 hours of reperfusion. Blood vessels were more dilated, with increased vascular density in the ischemic side peaking at 1 week. At 3 weeks after ischemia, the increase in vascular density partially resolved. Brain swelling was noted as early as 12 hours after reperfusion and could be detected by comparing the two hemispheres. Brain swelling appeared to peak at 1 day after ischemia and then gradually subsided. No swelling was noted at 1 week after ischemia. The evolution of brain swelling was similar to that in a previous study in which the brain water content (brain edema) was determined from 6 hours to 1 week (Lin et al., 1993).
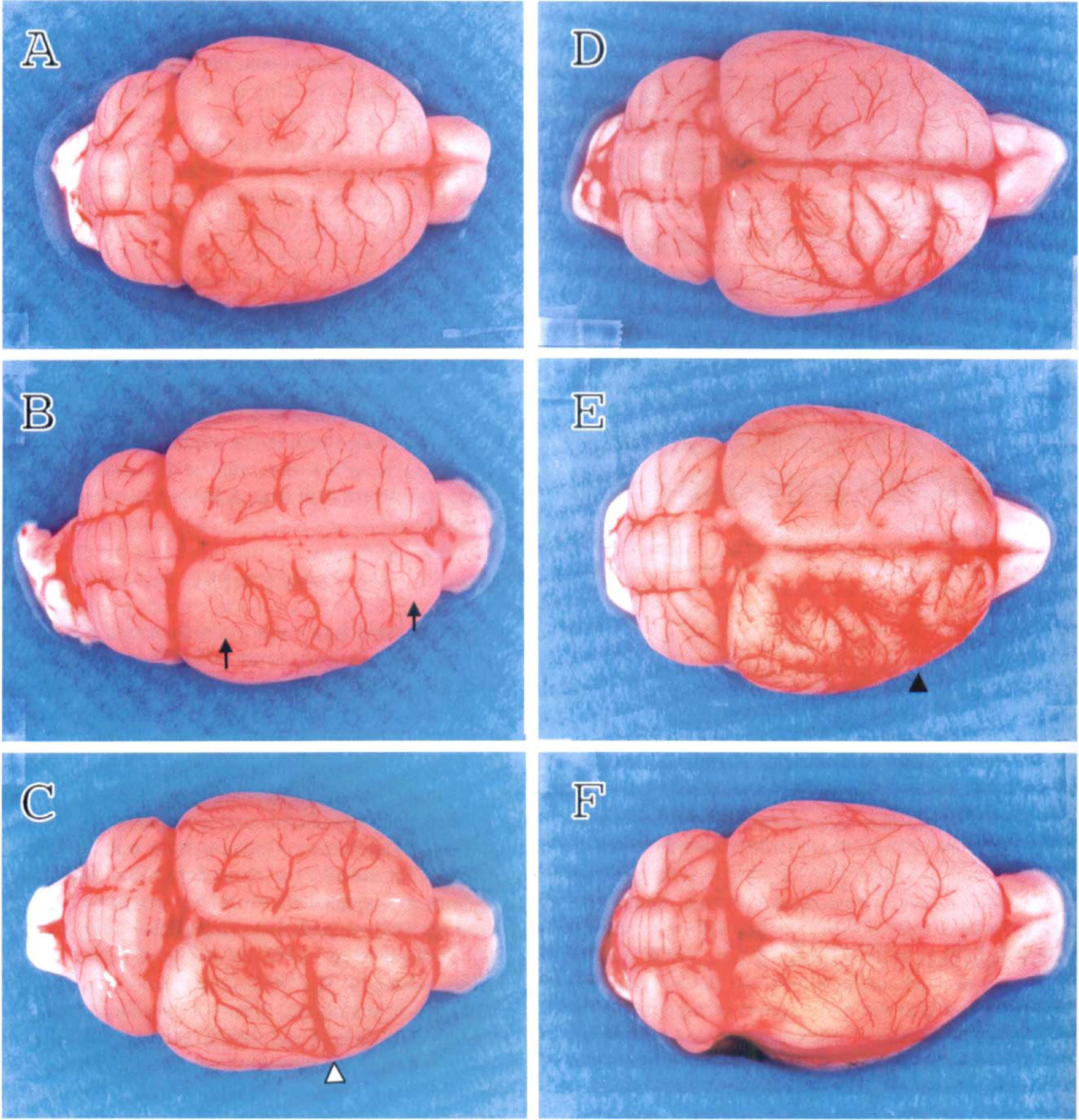
The effect of focal cerebral ischemia-reperfusion on the vascular density. Rats were subjected to 60-minute ischemia followed by 12 hours
DISCUSSION
Ischemic brain injury is a consequence of severe reduction of blood supply to the affected region. The resultant low tissue oxygen tension often leads to compensatory neovascularization to meet the metabolic demand (Plate, 1999). Examples include the development of collateral blood vessels to the ischemic region and angiogenesis in the healing of hypoxic wounds. However, the presumed hypoxia-induced angiogenic factors that mediate this compensatory response have not been fully identified. The putative factors include VEGF and bFGF; both were up-regulated after focal cerebral ischemia with a time course that parallels angiogenesis (Kovacs et al., 1996; Speliotes et al., 1996; Hayashi et al., 1997; Lin et al., 1997; Cobbs et al., 1998). The major cellular origin for bFGF was astrocytes, followed by neurons and endothelial cells (Lin et al., 1997), whereas the cortical neurons and pial cells were the source of VEGF (Hayashi et al., 1997).
In addition to VEGF and bFGF, we demonstrate for the first time that cerebral ischemia resulted in the induction of both Angpo-1 and Angpo-2 genes. However, the temporal profiles of their expression were different. The mechanism underlying the differential induction patterns remains to be determined. During embryogenesis, it has been shown that Angpo-2 may work in concert with VEGF at the front of invading vascular sprouts by blocking the action of constitutively expressed Angpo-1, allowing vessels to remain in a more plastic state in response to the sprouting signal provided by VEGF (Suri et al., 1996; Maisonpierre et al., 1997). In the absence of VEGF, Angpo-2 inhibition of constitutive Angpo-1 signal may contribute to vessel regression. In the present study, the VEGF mRNA induction profile is very similar to that of Angpo-2 mRNA and is in parallel with the evolution of vascular density noted in Fig. 4. These findings suggest that Angpo-2 could interact with VEGF after ischemic insult, similar to that noted during development. Although Angpo-2 is associated with vessel sprouting, Angpo-1 stabilizes the vasculature. The transient increase in Angpo-1 mRNA expression and depression of Angpo-2 mRNA levels during the early phase of ischemia may reflect an initial attempt of the ischemic region to stabilize the vascular integrity. This trend of Angpo-1 dominance was reversed later by a decrease in Angpo-1 mRNA levels and increase in Angpo-2 expression, peaking at 24 hours, in parallel with VEGF expression. The expression of Angpo-2 and VEGF during the same period may provide a drive for sprouting angiogenesis. The dominance of Angpo-2 was reversed later when a progressive increase in Angpo-1 expression was noted late in the angiogenesis process. The dominance of Angpo-1 late in vascular remolding is consistent with a shift in the gene expression profile toward vessel stabilization. Assuming the relative dominance of Angpo-1 and Angpo-2 represents vessel stabilization versus angiogenic sprouting, respectively, the Angpo-1/Angpo-2 ratios over time derived from Fig. 2 (sham-operated control: 1; ischemia 30 minutes: 5; ischemia 60 minutes: 2.23; reperfusion 30 minutes: 1.34; reperfusion 60 minutes: 1.14; reperfusion 90 minutes: 0.89; reperfusion 4 hours: 0.61; reperfusion 12 hours: 0.32; reperfusion 1 day: 0.16; reperfusion 3 days: 0.70; reperfusion 1 week: 1.43; reperfusion 2 weeks: 1.66) appear to reflect very well the evolution of vascular changes noted in Fig. 4, taking into account a lag in mRNA translation.
Five endothelial cell-specific RTKs have been identified (Mustonen and Alitalo, 1995; Beck and D'Amore, 1997), representing two separate classes of receptors for endothelial cell-specific growth factors (Davis et al., 1996). These include Flt-1 (or VEGFR-1), Flk-1 (VEGFR-2), and Flt-4 (VEGFR-3) receptors. The remaining two are Tie-1 and Tie-2 receptors. Results from the present study show that ischemia also led to a delayed and prolonged increase in Tie-2 mRNA levels, which coincides with the induction of its ligands, Angpo-1 and Angpo-2. Interestingly, its sib gene, Tie-1, also shared a similar induction profile, indicating a probable role of this receptor in angiogenesis even though its ligands have yet to be identified. The induction profiles of Tie-1 and −2 lagged behind those of VEGFR-1 and −2. These findings suggest probable differential roles for these two separate classes of receptors at various stages of vessel remodeling. Incidentally, during embryogenesis, the expression of Tie-1 and Tie-2 also follows that of VEGFR-1 and VEGFR-2 (Dumont et al., 1995; Yamaguchi et al., 1993). VEGFR-1 and VEGFR-2 are required at the earliest stages of angioblast differentiation and blood vessel development (Fong et al., 1995).
When the temporal expressions of the three angiogenesis genes (Angpo-2, VEGF, and bFGF) are compared, the fluctuation in mRNA levels of these genes follows different courses in the 2 weeks after ischemia. The molecular mechanism underlying the differential expression patterns remains to be explored. It is noteworthy that the receptors of these three genes also behave differently. The FGFR mRNA was markedly increased at 3 days and 1 week, whereas VEGFR-1 and VEGFR-2 peaked at 24 hours and then declined to below basal level at 1 week. These findings reinforce the contention that Angpo-2, bFGF, and VEGF play different roles in postischemic angiogenesis.
Angiogenic sprouting is accompanied by local degradation of the vascular basement membrane (Risau, 1997). Extracellular proteolysis is required for the degradation of matrix and modulation of cytokine activity during angiogenesis. The expression of TGF-β1 mRNA was delayed, peaking at 1 week. This observation is in agreement with the previously reported TGF-β1 expression in a neonatal hypoxia-ischemia model in rats (Klempt et al., 1992) and a permanent MCA occlusion model in adult rats (Wang et al., 1995). Transforming growth factor-β1 may play a modulatory role in angiogenesis (Pepper, 1997), inflammation, and tissue remodeling (Klempt et al., 1992; Wang et al., 1995) after ischemic insult. The induction of PAI-1 mRNA was transient, peaking at 1 day. Hypoxia stimulates PAI-1 expression in an endothelial cell line (Fitzpatrick and Graham, 1998). High plasma PAI-1 levels have also been noted in patients with a history of ischemic stroke (Margaglione et al., 1994). To our knowledge, the present study is the first on PAI-1 mRNA expression in the ischemic brain. The expression of uPA mRNA was induced during the late reperfusion period and peaked at 1 week. Its receptor, uPAR, was elevated at 12 hours and peaked at 24 hours after ischemia. Although an increase in uPA protein level has been reported in a permanent MCA occlusion model (Rosenberg et al., 1996), results shown are the first for the expression of uPAR mRNA. Surprisingly, uPAR mRNA was induced earlier than its ligand, uPA mRNA. The mechanism for this unexpected time course is not presently known. Membrane depolarization induces tPA (Gualandris et al., 1996). The induction of tPA mRNA after seizure, kindling, and long-term potentiation is reminiscent of immediate early genes (Qian et al., 1993; Tsirka et al., 1995). The tPA levels were elevated after ischemic stroke in patients (Margaglione et al., 1994). The expression of tPA mRNA was increased after ischemia in the present study. The extended expression of proteases tPA and uPA was in contrast to a transient increase in protease inhibitors such as PAI-1. These results suggest an imbalance in the post-ischemic expression of proteases relative to their inhibitors, a probable mechanism for the liquefaction of the infarcted region weeks after ischemia. Pharmacological inhibition of protease activity has many effects on angiogenesis, and ischemic outcomes remain to be explored (Rosenberg et al., 1996, 1998).
Neovascularization is accomplished via the concerted efforts of two major cellular processes. The first is vasculogenesis, a de novo synthesis of endothelial cells from either angioblasts or hemangioblasts. The second is angiogenesis, the growth or derivation of vascular structures from preexisting vessels (Risau and Flamme, 1995). Three forms of angiogenesis have been characterized: (1) remodeling of vessels into smaller ones (intussusceptive vascular growth); (2) endothelialization of a vessel that is growing both in length and in width; and (3) sprouting angiogenesis. Sprouting angiogenesis is the most common process in vascular remodeling. After ischemia, all three forms of angiogenesis were noted in the ischemic cerebral cortex at various times. Angiogenic sprouting appears to be the most prominent feature in the present study. Interestingly, Patt et al. (1997) reported a significant increase in both vessel density and size following severe chronic hypoxia. In the present study, we noted eventual decline in angiogenic sprouting at 3 weeks after ischemia (Fig. 4). Whether enhancing and/or prolonging angiogenic sprouting will affect the ischemic outcome remains to be studied.
In summary, our data demonstrate for the first time that ischemia differentially regulated the induction of Angpo-1, Angpo-2, Tie-1, and Tie-2 at mRNA levels. The Angpo-1/Angpo-2 ratio appears to be a potentially useful index of the activity of vascular remodeling. Although expressions of bFGF, VEGF, and Angpo-2 mRNA were all increased after ischemic insult, each showed a characteristic temporal profile. Furthermore, their respective receptors also exhibited different expression patterns. Together, these three angiogenic factors and their receptors may play different roles at different stages of the postischemic angiogenic process.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Ms. J. J. Chen for her technical support.
