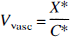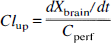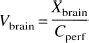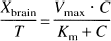Abstract
An in situ mouse brain perfusion model predictive of passive and carrier-mediated transport across the blood-brain barrier (BBB) was developed and applied to mdr1a P-glycoprotein (Pgp)-deficient mice [mdr1a(−/−)]. Cerebral flow was estimated from diazepam uptake. Physical integrity of the BBB was assessed with sucrose/inulin spaces; functional integrity was assessed with glucose uptake, which was saturable with a Km of ∼17 mmol/L and Vmax of 310 mmol · 100 g−1 · min−1. Brain uptake of a Pgp substrate (colchicine) was significantly enhanced (two- to fourfold) in mdr1a(−/−) mice. These data suggest that the model is applicable to elucidating the effects of efflux transporters, including Pgp, on brain uptake.
The blood-brain barrier (BBB) is a physical and functional regulator of solute transport between the brain and systemic circulation. Whereas simple diffusion governs the passage of many solutes across the BBB, carrier-mediated transport (uptake or efflux) can be an important determinant of apparent BBB permeability for endogenous substances and xenobiotics. P-glycoprotein (Pgp) has been identified as an efflux system on the luminal surface of brain capillaries that limits entry of many solutes into brain (Borst and Schinkel, 1998). P-glycoprotein is encoded by a gene family with three isoforms in rodents (mdr1a, mdr1b, mdr2;Ng et al., 1989). In mouse brain, mdr1a is the major isoform expressed; the availability of transgenic and mutant mdr1a Pgp-deficient mice [mdr1a(−/−)] provides a unique opportunity to investigate the importance of this efflux system in the BBB (Schinkel et al., 1994; Lankas et al., 1997; Umbenhauer et al., 1997).
Various methods have been applied to studying the brain uptake of substrates (Pardridge, 1998). Among these techniques, brain perfusion methods represent a highly sensitive means of studying BBB transport without the confounding factor of systemic disposition (Smith, 1996). The composition and flow of the perfusate can be controlled to study the effect of plasma proteins, electrolytes, and transport inhibitors, as well as substrate concentration and perfusate flow, on uptake. The adult rat is by far the most frequently used animal for brain perfusions, but the technique has been applied successfully to other species (Betz and Gilboe, 1973; Zlokovic et al., 1986) or under other conditions. For example, a bilateral in situ brain perfusion was applied to 1-week-old neonatal rats to study acidic amino acid brain uptake (Al-Sarraf et al., 1997). Bradbury et al. (1984) first reported mouse brain perfusion, but this study was not related to BBB transport per se, and the permeability properties of the vessels remained to be characterized. Recently, an in situ mouse brain perfusion (through the left cardiac ventricle) was used to investigate the BBB transport of a β-amyloid binding protein (Shayo et al., 1997). However, this model system has not been further characterized or validated, to our knowledge.
The present study was undertaken to develop an in situ mouse brain perfusion based on an established rat model (Takasato et al., 1984; Smith, 1996) and characterize the BBB transport of selected model substances that undergo passive and carrier-mediated transport across the BBB. The model was then applied to mdr1a(−/−) Pgp mutant mice to examine the influence of Pgp expression on apparent BBB permeability.
MATERIALS AND METHODS
Materials
[3H]Colchicine (61.4 Ci/mmol), [3H]diazepam (83 Ci/mmol), [14C]D-glucose (260 Ci/mol), [14C]inulin (13.2 Ci/mol), [3H]sucrose (10.2 Ci/mmol), and [14C]sucrose (600 Ci/mol) were purchased from NEN Life Science (Brussels, Belgium). [14C]Urea (57 Ci/mol) was purchased from Amersham (Les Ulis, France). D-Glucose (mixed anomers, 99.5% purity) was purchased from Sigma (St. Quentin, France). All other chemicals and reagents were of analytical-grade quality.
Animals
Adult male OF-1 mice (30 to 40 g, 6 to 8 weeks old) were obtained from Iffa-Credo (L'Arbresle, France). Male CF-1 mice genotyped for mdr1a Pgp [mdr1a(+/+) and (−/−), 30 to 40 g, 6 to 8 weeks old] were obtained from Charles River (Wilmington, MA, U.S.A.). Approximately 25% of the CF-1 population is deficient in mdr1a Pgp (Umbenhauer et al., 1997). The Iffa-Credo OF-1 mouse colony originates from CF-1 mice introduced in France in 1967. Animals were maintained under standard conditions of temperature and lighting and given food and water ad libitum. The research adhered to the ethical rules of the French Ministry of Agriculture for experimentation with laboratory animals (law no. 87-848).
In situ brain perfusion
Mice were anesthetized with intraperitoneal ketamine/xylazine (140/8 mg/kg), except in the glucose transport experiments, in which midazolam (5 mg/kg) replaced xylazine, which is known to induce hyperglycemia (Abdel el Motal and Sharp, 1985). The in situ rat brain perfusion technique previously described by Takasato et al. (1984), with modifications (Allen et al., 1997), was applied to the mouse. In brief, the right common carotid artery was exposed and ligated caudally. The external carotid artery was ligated rostral to the occipital artery at the level of the bifurcation of the common carotid artery with the internal carotid. The pterygopalatine artery was not ligated in order to simplify the procedure. The right common carotid artery was catheterized rostrally with polyethylene tubing (0.30-mm inner diameter × 0.70-mm outer diameter; Biotrol Diagnostic, Chennevières-les-Louvres, France) filled with heparin (25 U/mL) and mounted on a 26G needle. If necessary, one-third of the initial anesthetic dose was readministered ∼5 minutes before the perfusion. Immediately before perfusion, the cardiac ventricles were severed via openings in the abdominal cavity and diaphragm. Brains were perfused for 20 to 180 seconds with Krebs/bicarbonate buffer, plasma, or serum gassed with 95% O2 and 5% CO2 for pH control (7.4) and maintained at 37°C at a calibrated flow rate of 2.5 mL/min (Harvard pump PHD 2000; Harvard Apparatus, Holliston, MA, U.S.A.). The perfusate contained at least 0.3 μCi/mL vascular space marker (sucrose or inulin) to achieve at least 1,000 dpm/tissue sample.
The perfusion was terminated by decapitating the animal. The brain was removed from the skull, dissected on ice (Chiueh et al., 1978), digested with 1 mL of Solvable (Packard, Rungis, France) at 50°C, and mixed with 9 mL of Ultima Gold XR scintillation cocktail (Packard). Total 14C and 3H were determined simultaneously in a Packard Tri-Carb model 1900TR liquid scintillation analyzer. Sucrose and inulin spaces were determined in the cerebellum and seven brain regions, in addition to the right and left hemisphere. The intravenous inulin spaces were obtained under anesthesia 5 minutes after administration of a bolus in the right jugular vein. All other data are reported for the right hemisphere, unless specified otherwise.
Calculation of initial uptake clearance and apparent brain distributional volume
Calculations related to the in situ brain perfusion method have been described previously (Smith, 1996).
Vascular volume (Vvasc, mL · 100 g−1) was determined using the following equation:
where X* is the amount of radiolabeled sucrose or inulin in the brain (dpm · 100 g−1) and C* is the perfusate concentration (dpm · mL−1).
A pharmacokinetic definition was used to determine initial uptake clearances (Clup, mL · 100 g−1 · min−1) from the following relationship:
where Xbrain is the amount of radiotracer in the brain (dpm · 100 g−1) corrected for vascular contamination (Xtotal − Vvasc · Cperf) and Cperf is the tracer concentration in the perfusate (dpm · mL−1). In single time point experiments, Xbrain/T replaced dXbrain/dt, where T was the perfusion time (minutes).
Apparent brain distributional volumes (Vbrain, mL · 100 g−1) were calculated from
D-Glucose uptake data at 20 seconds were described with the following relationship:
where Vmax is the maximal transport velocity (μmol · 100 g−1 · min−1), C is the glucose concentration in the perfusate (0.5, 5, 10, 20, and 50 mmol/L), and Km is the concentration (mmol/L) at half-maximal velocity.
Data analysis
Data are presented as mean ± SD for three to six animals, unless specified otherwise. One- and two-way analysis-of-variance techniques or two-sided Student t tests were used to determine the statistical significance of differences between experimental groups. Statistical significance was defined as a P value of <0.05. Estimates of the D-glucose transport parameters (Km and Vmax) were obtained by fitting a Michaelis-Menten type of equation to the uptake rate versus perfusate glucose concentration data by nonlinear least-squares regression (Win-Nonlin 1.1, Apex, NC, U.S.A.).
RESULTS AND DISCUSSION
Success rate for surgical procedure
The success rate for the surgical procedure was >85% for the experiments reported in this communication. All failures could be accounted for by rupture of the right carotid artery during catheterization and cardiorespiratory arrest likely related to anesthesia/blood loss.
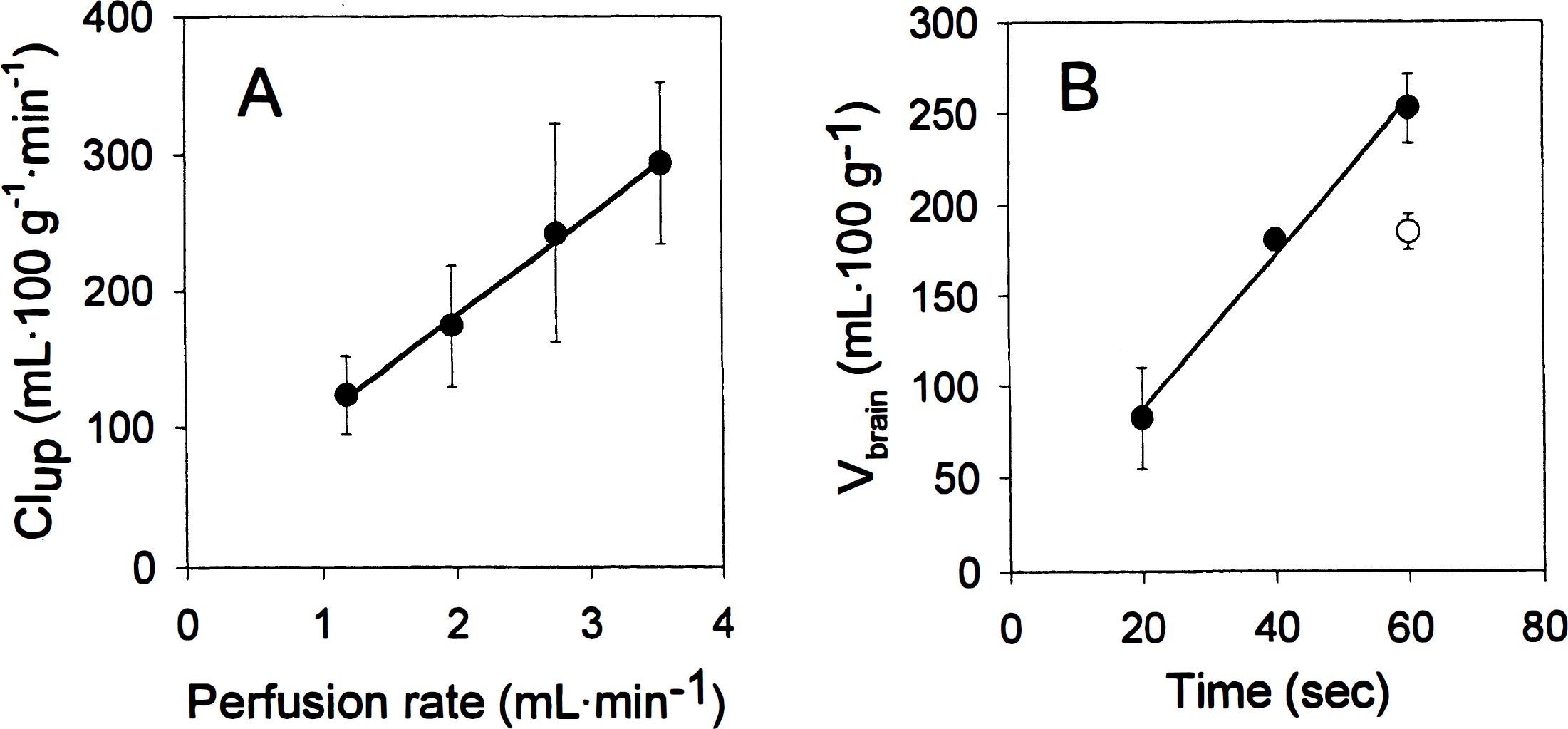
Determination of cerebral flow rate
Diazepam brain uptake is flow limited and can be used to estimate cerebral perfusion rate (Takasato et al., 1984). The modified rat brain perfusion has been used at reduced perfusion flow rates (10 to 12 versus 20 mL · min−1) without compromising the quality of the perfusion (Smith et al., 1990; Rousselle et al., 1998). In the present study, initial selection of perfusion flow was based on the one-to-five brain weight ratio between the mouse and the rat (Davies and Morris, 1993). A perfusion flow of 2 mL · min−1 was selected as a starting value. A linear relationship between diazepam Clup in the right hemisphere at 20 seconds and perfusion flow rate was observed (Fig. 1A). No BBB disruption was observed at the higher flow rates, and uptake in the contralateral hemisphere was twofold lower, on average (data not shown). A perfusion flow rate of 2.5 mL · min−1, which results in diazepam Clup similar to that in previous reports in the rat (Smith et al., 1990; Rousselle et al., 1998), was selected. A separate study at 2.5 mL · min−1 (20-, 40-, and 60-second time points) established that diazepam uptake was linear through 60 seconds with a Clup of 255 mL · 100 g−1 · min−1 (Fig. 1B). Diazepam Clup values determined individually from the discrete time points did not differ significantly (P = 0.77, data not shown). Perfusion of diazepam in rat plasma (∼86% protein binding; Klotz et al., 1976) decreased the Vbrain by 27% at 60 seconds (P < 0.01; Fig. 1B). Despite the high extraction of diazepam, protein binding has been shown to decrease the brain uptake of diazepam (Jones et al., 1988; Fenerty and Lindup, 1989), which is consistent with the present study.
Physical integrity of blood-brain barrier during perfusion
Maintenance of BBB integrity during perfusion was an important aspect of model validation. [14C]- or [3H] Sucrose and [14C] inulin were used as markers of brain vascular volume as these compounds do not cross the BBB significantly during brief periods of perfusion. Vascular volumes were determined from [14C] inulin and/or [14C]- or [3H] sucrose spaces in different regions of the mouse brain after a 60-second perfusion with buffer (Fig. 2). The vascular volumes (Vvasc) obtained in the present study (i.e, from 0.6 to 1.3 mL · 100 g−1, depending on the brain areas) agree with previously reported values in the rat (Takasato et al., 1984; Rousselle et al., 1998). Differences between brain regions and structures, such as a lower vascular space in the striatum and higher values in the hypothalamus (which lacks a BBB), were also in agreement with those in the previous reports. The [3H] sucrose spaces were slightly higher (10 to 15%) than the [14C] inulin spaces, which is consistent with the lower molecular weight and higher permeability of sucrose relative to inulin (Ohno et al., 1978). In a separate experiment, the Vvasc values obtained with perfusion (60 seconds) of buffer (1.25 ± 0.08 mL · 100 g−1) were ∼30% lower than with human serum (1.84 ± 0.29 mL · 100 g−1; P < 0.05). Vascular volumes 5 minutes after intravenous injection of [14C] inulin under anesthesia were even higher (2.58 ± 0.19 mL · 100 g−1). These observations are consistent with the ∼50% decrease in vascular volume with buffer as compared with blood, observed by Takasato et al. (1984). A combination of erythrocyte-mediated mechanical stress on the capillary wall, increased viscosity, and osmotically driven loss of intravascular water to brain tissue due to the absence of plasma proteins during perfusion may explain those results.
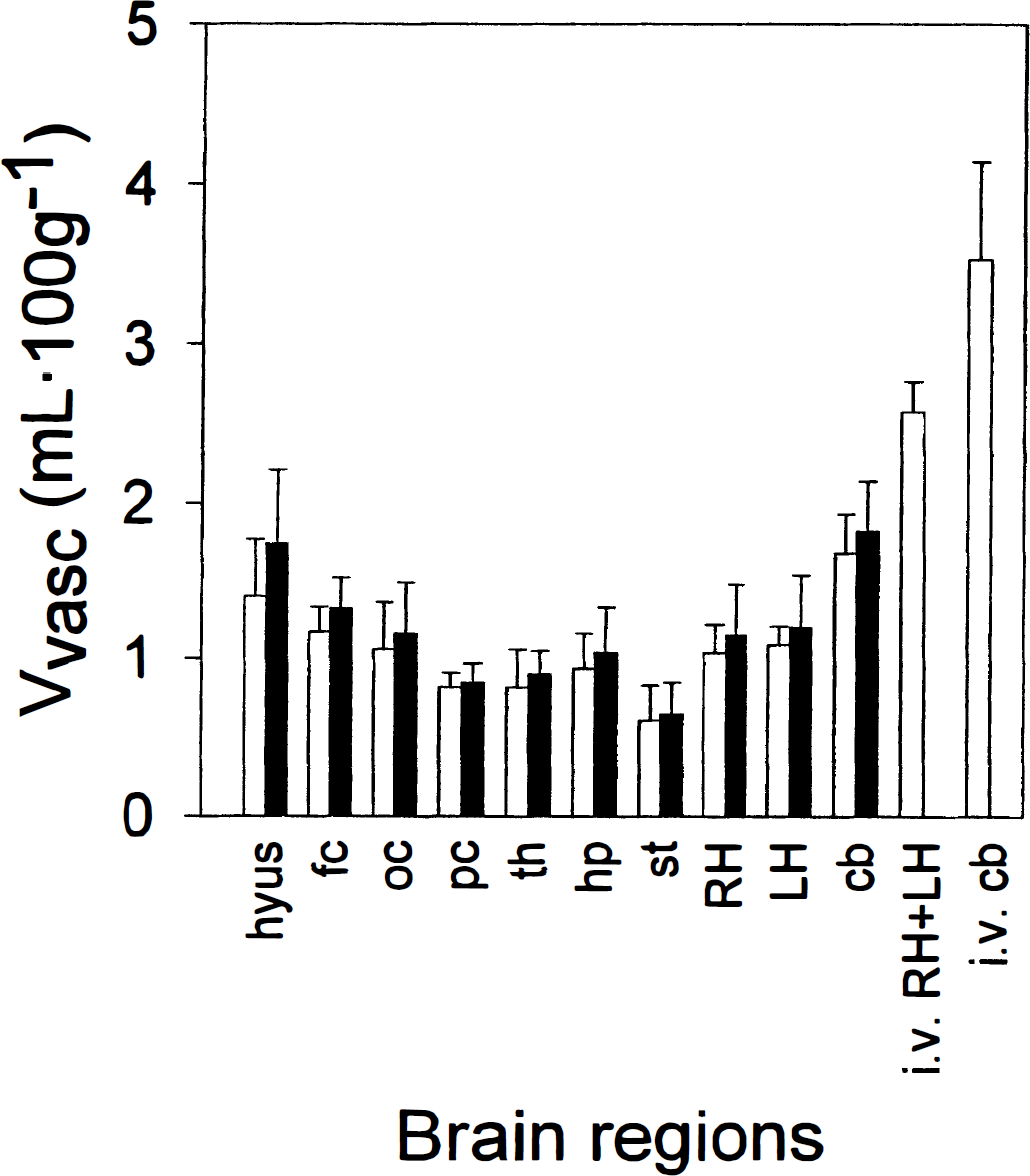
Inulin (open bars) and sucrose (filled bars) (Vvasc) in various regions of the right hemisphere, left hemisphere, and cerebellum after 60-second perfusion or 5 minutes following intravenous injection of [14C] inulin. Data are presented as mean ± SD (n = 5 or 6 for perfusion, n = 4 for intravenous administration). hyus, hypothalamus; fc, frontal cortex; oc, occipital cortex; pc, parietal cortex; th, thalamus; hp, hippocampus; st, striatum; RH, right hemisphere; LH, left hemisphere; cb, cerebellum.
[14C] Urea permeability also was used to assess the physical integrity of the BBB over longer perfusion times. The Clup of urea determined at 180 seconds (0.610 ± 0.083 mL · 100 g−1 · min−1; n = 5) compares favorably with previously reported values of 0.3 to 1.0 mL · 100 g−1 · min−1 (Chikhale et al., 1994; Rousselle et al., 1998). The [3H] sucrose space observed in these experiments was 1.30 ± 0.23 mL · 100 g−1, a value that is similar to the one obtained in the right hemisphere at 60 seconds (Vvasc = 1.10 ± 0.30 mL · 100 g−1).
Functional integrity of blood-brain barrier: D-glucose uptake
The functional integrity of the BBB was assessed by determining the brain uptake of D-glucose, which is transported through the BBB by a saturable carrier-mediated process that is not energy dependent (GLUT1 transporter; Pardridge and Boado, 1993). In the present study, brain uptake of D-glucose after 20 seconds of perfusion was shown to be saturable, with a Km of 17 ± 4 mmol/L and Vmax of 310 ± 29 μmol · 100 g−1 · min−1 (Fig. 3). This represents a Clup (Vmax/Km) of 18 mL · 100 g−1 · min−1, equivalent to a brain extraction of 7% on average. These values are consistent with previous reports in the rabbit (Cornford et al., 1994), rat (Pardridge et al, 1982; Simpson et al., 1999), and mouse (Cornford et al., 1993). However, the Km and Vmax values reported in the present study were higher. The use of different rodent species and methodologies may explain these differences. A diffusional component was not necessary to model the data, as the amount of D-glucose crossing the BBB by passive diffusion over 20 seconds is not significant. D-Glucose uptake (0.5 mmol/L in perfusate) in mdr1a(−/−) CF-1 mice was identical to that observed in OF-1 mice (Clup of 22.1 ± 1.3 versus 22.6 ± 1.7 mL · 100 g−1 · min−1; P = 0.62). In the present study, glycemia was not measured prior to perfusion, and its potential influence on glucose transport parameters was not investigated because the study of the mechanisms of glucose transport at the BBB was not our aim.
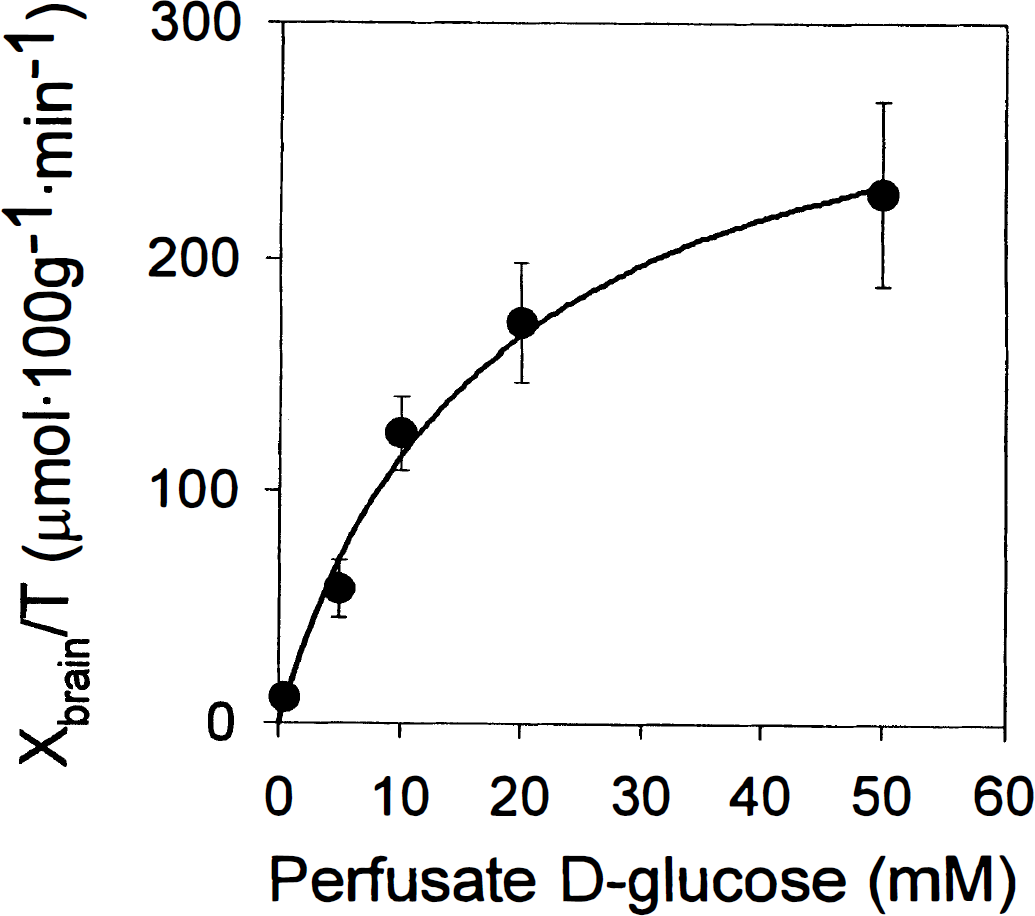
Rate of D-glucose uptake in the right hemisphere (T = 20 s) as a function of D-glucose concentration in perfusate. Data are presented as mean ± SD (n = 3 or 4 per point). The solid line represents a fit of the Michaelis-Menten equation to the data by nonlinear least-squares regression. Parameter estimates for Km and Vmax were 17 ± 4 mmol/L and 310 ± 29 μmol · 100 g−1 · min−1, respectively.
Blood-brain barrier transport of colchicine in wild-type and mdr1a(−/−) mice
P-Glycoprotein was first identified and purified from mammalian cells that were resistant to colchicine because of reduced membrane permeability (Juliano and Ling, 1976; Riordan and Ling, 1979). Colchicine was later shown to interact with Pgp in brain capillaries (Jetté et al., 1995) and the BBB in vivo (Drion et al., 1996; Desrayaud et al., 1997). The distribution of colchicine to the brain of wild-type OF-1 mice was limited over 20 to 90 seconds (Fig. 4) and likely represents sequestration of colchicine in the capillaries with little or no transport to the brain. A two- to fourfold enhancement in Vbrain was observed in mdr1a(−/−) CF-1 mice, and the uptake appeared to be linear in the absence of Pgp. This observation is consistent with the results obtained previously with a potent Pgp modulator in the modified rat brain perfusion model (Drion et al., 1996). To verify that wildtype OF-1 mice were an appropriate control for these experiments, the apparent Vbrain of colchicine was determined in mdr1a(+/+) CF-1 mice and in wild-type OF-1 mice. No significant differences were observed in Vbrain at 90 seconds between the two strains (data not shown). No outliers were observed among the OF-1 mice (n = 24), suggesting an occurrence of the mdr1a(−/−) genotype in this population lower than that reported for CF-1 mice (Umbenhauer et al., 1997). Brain vascular volume in the right hemisphere of CF-1 mdr1a(−/−) mice was similar to that of wild-type OF-1 mice.
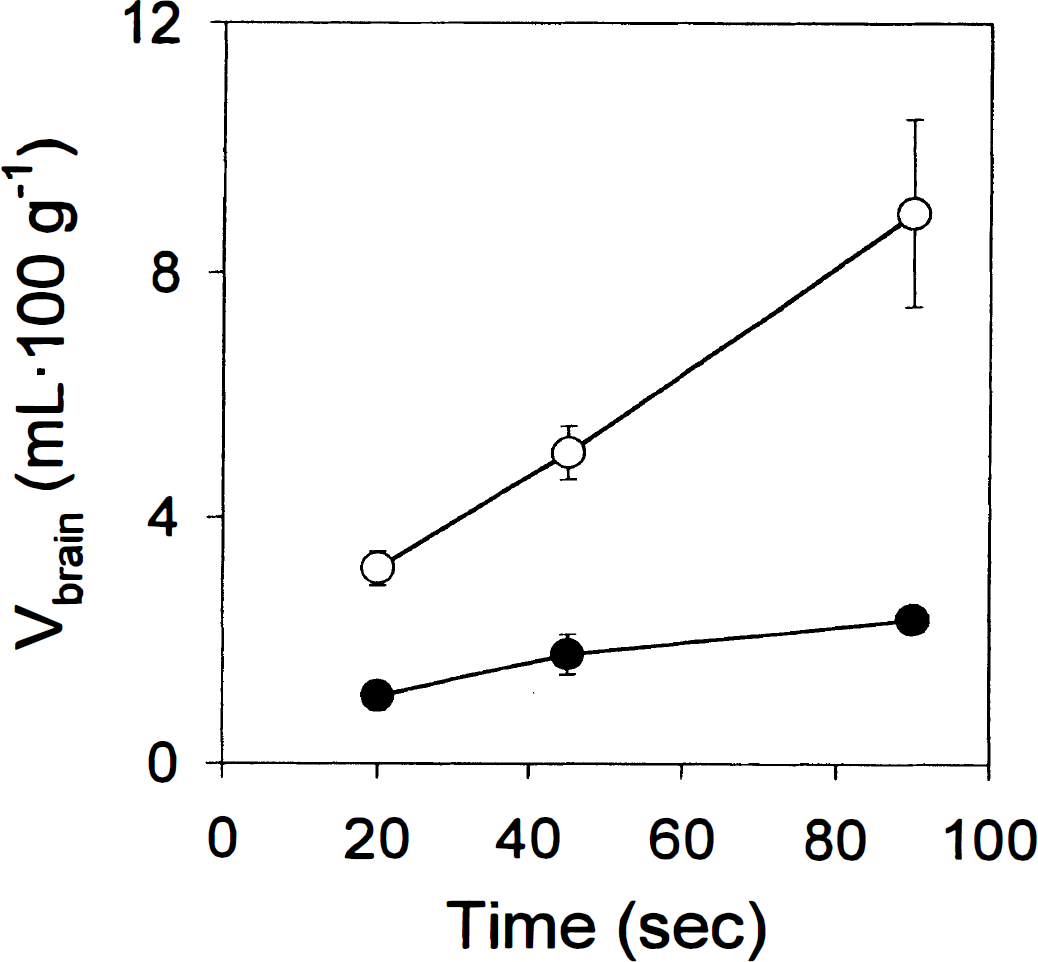
Colchicine brain uptake in the right hemisphere (expressed as apparent brain distributional volume, Vbrain) in wild-type OF-1 (filled circles) and mdr1a(−/−) CF-1 (open circles) mice. Data are presented as mean ± SD (n = 4 per point). A statistically significant difference was observed between the two mouse strains (two-way analysis of variance, P < 0.01).
CONCLUSION
The results obtained in this study demonstrate that the in situ brain perfusion method may be applied to the mouse without compromising the physical or functional integrity of the BBB. Several parameters such as cerebral perfusion flow, brain vascular volume, urea permeability, and carrier-mediated transport of D-glucose were comparable with previously published results and were consistent with the permselective nature of the BBB. These results also suggest that the in situ mouse brain perfusion model can be used to predict the permeability of solutes across the BBB and that it may be useful in assessing the interaction of substrates with mdr1a Pgp or other BBB transporters in vivo as well as BBB transport in transgenic murine models.