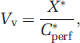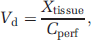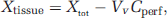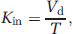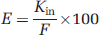Abstract
Uptake1 and uptake2 transporters are involved in the extracellular clearance of biogenic amine neurotransmitters at synaptic clefts. We looked for them at the blood–brain barrier (BBB) and blood–retina barriers (BRB), where they could be involved in regulating the neurotransmitter concentration and modulate/terminate receptor-mediated effects within the neurovascular unit (NVU). Uptake2 (Oct1-3/Slc22a1-3, Pmat/Slc29a4) and Mate1/Slc47a1 transporters are also involved in the transport of xenobiotics. We used in situ carotid perfusion of prototypic substrates like [3H]-1-methyl-4-phenylpyridinium ([3H]-MPP+), [3H]-histamine, [3H]-serotonin, and [3H]-dopamine, changes in ionic composition and genetic deletion of Oct1-3 carriers to detect uptake1 and uptake2 at the BBB and BRB. We showed that uptake1 and uptake2 are involved in the transport of [3H]-dopamine and [3H]-MPP+ at the blood luminal BRB, but not at the BBB. These functional studies, together with quantitative RT-PCR and confocal imaging, suggest that the mouse BBB lacks uptake1 (Net/Slc6a2, Dat/Slc6a3, Sert/Slc6a4), uptake2, and Mate1 on both the luminal and abluminal sides. However, we found evidence for functional Net and Oct1 transporters at the luminal BRB. These heterogeneous transport properties of the brain and retina NVUs suggest that the BBB helps protect the brain against biogenic amine neurotransmitters in the plasma while the BRB has more of a metabolic/endocrine role.
Keywords
Introduction
The trafficking of neurotransmitters within the brain depends greatly on the so-called uptake1 and uptake2 carrier-mediated processes, which help maintain the balance between neurotransmitters in the intracellular and extracellular fluids of the nervous parenchyma. The role of uptake1 or ‘reuptake’ transporters in the clearance of dopamine, serotonin, and norepinephrine at the synaptic cleft has been well established for many years (Daws, 2009; Kristensen et al, 2011). These biogenic amine neurotransmitter transporters are classified according to their affinity/capacity for substrates/inhibitors and sensitivity to ions. Uptake1 are high affinity, Na+/Cl−-dependent unidirectional symporters such as Net (Slc6a2), Dat (Slc6a3), and Sert (Slc6a4). More recently, uptake2 transporters that are lower affinity, polyspecific and Na+/Cl−-independent have been shown to transport some biogenic amine neurotransmitters and xenobiotics in both directions, depending on their concentration gradient. The best known are Oct1-3 (Slc22a1-3) and Pmat (Slc29a4) (Eisenhofer, 2001; Dahlin et al, 2007; Duan and Wang, 2010). We have examined the possibility that the trafficking of biogenic amine neurotransmitters is not restricted to neurons but may also occur at the blood–nerve barriers as is suggested for some peripheral tissues (Catravas and Gillis, 1983; Chemuturi and Donovan, 2007; Eisenhofer, 2001; Ramamoorthy et al, 1993). If this is so, then uptake1/2 transporters could modulate the local availability of biogenic amine neurotransmitters by luminal (blood) and/or abluminal (nerve side) carrier systems. Uptake2 carriers could also transport many drugs and xenobiotics like 1-methyl-4-phenylpyridinium (MPP+) (Koepsell et al, 2007).
We have studied two barriers, the blood–brain barrier (BBB) and the blood–retina barriers (BRB), which have several common features. Although the BBB is primarily formed by the endothelium of cerebral capillaries, there are specific interactions and communication at the abluminal side of the BBB with the other cell components of the neurovascular unit (NVU): pericytes, astrocytes, and neurons (Abbott et al, 2010). The local regulation of mediators like neurotransmitters and their movement across these blood–nerve barriers has received relatively little attention, despite the fact that this could influence and terminate receptor-mediated effects on, at least, the integrity and permeability of the brain capillaries of the NVUs (Leybaert, 2005; Lu et al, 2010).
The eye contains similar NVUs between the blood and the retina (BRB) and the aqueous humor (blood–aqueous barriers, BAB). Because the retina is in contact with the choroid, a highly vascularized tissue with fenestrated capillaries, the BRBs are located at both the retinal pigmentary epithelium (RPE; BRBo), a monolayer of tightly joined cells separating the posterior side of the retina from the choroid, and at the tight endothelium of the retinal capillaries (BRBi) (Pournaras et al, 2008).
These barriers have elaborate tight junctional complexes that efficiently limit the paracellular transport of solutes. But they also have a wide range of transporters that selectively transport solutes and xenobiotics (Abbott et al, 2010). We have used molecular strategies and functional experiments to assess the roles of uptake1, uptake 2, and Mate1 transporters. To do this, we determined whether transport mechanisms are involved in the diffusion of selected biogenic amines at the mouse BBB and blood–eye barriers. We used in situ carotid perfusion (Cattelotte et al, 2008) because this enabled us to control the perfusion time and the composition of the luminal/vascular compartment. The perfusate replaces the blood and so allows the identification and exploration of the kinetics of transporters at the luminal (blood) side of membranes in vivo (Smith, 1996). We find that the mouse BBB does not support carrier-mediated transport of biogenic amines by uptake1, uptake2, or Matel systems. In contrast, we have shown that there is a functional uptake1 carrier, similarly to Net, and an uptake2/Oct1 carrier at the luminal BRB.
Materials and methods
Animals
Male Swiss mice (8 to 10 weeks old) were obtained from Janvier (Genest, France). In some experiments, double knock-out Oct1 and Oct2 Fvb male mice (Oct1,2(–/–)), single knock-out Oct3 Fvb mice (Oct3(–/–)), and triple knock-out (Mdr1a.Mdr1b.Bcrp(–/–)) Fvb mice originally obtained from the Netherlands Cancer Institute (Amsterdam, The Netherlands) were used with Fvb wild-type control mice. The mice were housed in a controlled environment (19 ± 2°C, 55 ± 10% relative humidity) with a 12-hour light/dark cycle, and access to food and tap water ad libitum. All experimental procedures complied with the ethical rules of the French agency for experimentation with laboratory animals, with IMTCE-IFR71 ethics committee (no 86–23) and the facility procedures (Université Paris Descartes, Faculté de Pharmacie), and ARRIVE guidelines.
Drugs and Chemicals
[14C]-Sucrose (588 mCi/mmol), [3H]-MPP+ (70.4.Ci/mmol), [3H]-dopamine (30.3 Ci/mmol), [3H]-serotonin (27.7 Ci/mmol), and [3H]-histamine (13.4 Ci/mmol) were purchased from Perkin-Elmer (Paris, France). GF120918 and PSC833 were gifts from GlaxoSmithKline (Uxbridge, UK) and Novartis (Basel, Switzerland), respectively. Methylene-dioxymethamphetamine (MDMA, ecstasy) was synthesized (purity >99%) in the laboratory of Professor Galons (Faculté de Pharmacie, Université Paris Descartes). All other chemicals were analytical grade.
Antibodies
The following primary antibodies were used: a mouse monoclonal anti-P-gp (C219, Alexis Biochemicals, San Diego, CA, USA, diluted 1:50), a rat monoclonal anti-Bcrp (BXP53, Alexis Biochemicals, diluted 1:50), a mouse monoclonal anti-Net (NET05-1, MabTechnologies, Stone Mountain, GA, USA, diluted 1:600) raised against the N-terminal intracellular domain (residues 5 to 17) and previously characterized (Matthies et al, 2009), a rat polyclonal anti-Dat (Chemicon MAB369; Millipore, Molsheim, France, diluted 1:1,000), a rabbit antiserum against Sert (Immunostar 24330; Euromedex, Souffelweyersheim, France, diluted 1:200), a rabbit antiserum against tyrosine hydroxylase (208020234, Jacques Boy, Reims, France, diluted 1:4,000), a mouse monoclonal anti-Pmat (ab56554, Abcam, Cambridge, UK, diluted 1:100), and a rabbit polyclonal anti-Oct3 (OCT31-S, Alpha Diagnostic, San Antonio, TX, USA) previously characterized (Vialou et al, 2004). Appropriate goat secondary antibodies conjugated to Alexa Fluor-488 or 555 dyes (Life Technologies, Saint Aubin, France, diluted 1:300) were used for detection. Isotype primary antibody controls were from Life Technologies.
In situ Carotid Perfusion
Surgical procedure and perfusion: We assessed the functional role of mediated carriers systems by measuring the transport of selected labeled compounds at the brain and eye of mice by in situ carotid perfusion, in which the blood is completely replaced by an artificial perfusion fluid for a very short time (Cattelotte et al, 2008; Smith, 1996; Takasato et al, 1984). Mice were anesthetized with ketamine-xylazine (140–8 mg/kg, intraperitoneally) and the appropriate vessels were ligated. A catheter was inserted into the right common carotid artery and connected to the perfusion system. The thorax was opened, the heart was cut, and perfusion was started immediately at a constant flow rate (2.5 mL/min) and pressure (Cattelotte et al, 2008; Takasato et al, 1984). [3H]-MPP+ (0.3 μCi/mL), [3H]-dopamine (0.3 μCi/mL), [3H]-histamine (0.5 μCi/mL) or [3H]-serotonin (0.3 μCi/mL) and the vascular marker [14C]-sucrose (0.1 μCi/mL) were added to the perfusion fluid with or without unlabeled ‘modulators’. Perfusion times were 60 seconds except for [3H]-MPP+ which was 120 seconds. The linear time course of the accumulation of [3H]-compounds in the eye and the brain (data not shown) showed that no equilibrium was reached with the selected perfusion times. This ensured that the tissue distribution of [3H]-compounds was governed only by initial transport processes acting at the (blood) luminal side membrane of the BBB and BRB in our experimental condition.
Perfusion was terminated by decapitation and the brain and the right eye were removed from the skull; the brain was dissected out on a cold freezer pack, and the eye on dry ice. The anterior and posterior segments of the eye and the vitreous humor were dissected out in some experiments. The right brain hemisphere, the whole right eye (without the optic nerve) or eye segments, and aliquots of the liquid were placed in tared vials and weighed. Samples were digested with 2 mL of Solvable (Perkin-Elmer) at 50°C, cooled, and mixed with 9 mL of Ultima gold XR (Perkin-Elmer). Dual label counting was performed in a Tri-Carb (Perkin-Elmer).
Perfusion fluid: Unless otherwise specified, the perfusion fluid was Krebs carbonate-buffered physiological saline (mmol/L: 128 NaCl, 24 NaHCO3, 4.2 KCl, 2.4 NaH2PO4, 1.5 CaCl2, 0.9 MgSO4, 9 D-glucose), warmed to 37°C and gassed with 95% O2/5% CO2 to bring the pH to 7.40. Hydrochloric acid or sodium hydroxide was sometimes added to the gassed perfusion fluid to bring the pH to 5.40, 6.40, or 8.40. Acute increases in the intracellular pH (pHi) were obtained by adding NH4+ (30 mmol/L) to the perfusion fluid. This causes NH3 to diffuse into the endothelial cells, where it consumes protons and rapidly increases the pHi.
Chloride and/or sodium ions were iso-osmotically replaced in the perfusion fluid. Cl− was replaced with gluconate and nitrate in the ‘Glu’ buffer. Sodium was replaced with potassium in the ‘K+’ buffer (K+ 154 mmol/L), by lithium chloride in the ‘Li+’ buffer (Li+ 128 mmol/L, K+ 26 mmol/L, Cl− 131 mmol/L), by mannitol in the ‘mannitol’ buffer (mannitol 256 mmol/L, K+ 26 mmol/L, Cl− 3 mmol/L) and by NMDG chloride in the ‘NMDG-Cl’ buffer (NMDG+ 128 mmol/L, K+ 26 mmol/L, Cl− 131 mmol/L). The pH of the perfusion fluid (extracellular vascular pH or pHe) was always adjusted using a digital pH meter (± 0.05 pH) before adding radiolabeled compounds.
Apparent tissue distribution volume and initial transport parameters: The brain and eye vascular volumes (Vv) were calculated from the distributions of radiolabeled sucrose. [14C]-sucrose was used to estimate the tissue vascular volume (Vv; μL/g) as:
The apparent tissue distribution volume (Vd) for [3H]-compounds was calculated from the radioactivity in the right brain hemisphere or right eye corrected for the vascular contamination using:
The relative permeability of the luminal barriers for each compound was measured by comparison with that of [3H]-diazepam, which is freely diffusible. Extraction E (%) values are given by:
Preparation of Tissues for Immunostaining
Anesthetized mice were perfused transcardially at 10 mL/min, first with oxygenated Krebs-bicarbonate buffer warmed to 37°C for 2 minutes, and then with cold (4°C) fixative solution (3% paraformaldehyde in 0.1 mol/L phosphate buffer, pH 7.4). The brain was removed, kept in the same fixative solution at 4°C for 1 hour, stored overnight in phosphate buffer at 4°C. Coronal sections (50 to 100 μm) of brain were cut on a vibratome (VT-1200S, Leica Microsystems, Nanterre, France) and processed immediately for immunostaining or kept at 4°C for up to 1 week.
Cryosections of whole eyes were prepared by perfusion with fixative solution followed by enucleation of the eyeballs and incision of the cornea. Eyes were then postfixed for 4 hours at 4°C, rinsed in phosphate-buffered saline (PBS), cryoprotected in sucrose (15% in PBS) and frozen in Tissue-Tek. Sagittal cryosections (20 μm) were cut and placed on SuperFrost slides (Menzel-Gläser, Braunschweig, Germany).
Immunostaining of Brain Sections and Whole Eye Cryosections
Brain sections were permeabilized by immersion in 0.5% Triton X-100 for 30 minutes, rinsed three times in PBS (5 minutes), immersed in NaBH4 (1% in PBS) for 20 minutes to quench free aldehyde groups, washed three times in PBS (10 minutes) and incubated overnight at 4°C in PBS containing 1% BSA, 10% goat serum, and 0.1% Triton X-100 to block nonspecific binding sites.
The sections were then rinsed three times in PBS, 0.1% Triton X-100 (PBST, 10 minutes) and incubated for 24 hours at 4°C in a mixture of two primary antibodies diluted in PBST. They were washed extensively in PBST and incubated in a mixture of appropriate secondary antibodies for 16 hours at 4°C. Cell nuclei were counterstained with TOPRO-3 (2 μmol/L in PBS for 20 minutes). Brain sections were mounted in 90% glycerol (v/v in PBS) using 0.17 mm thick coverslips (Hecht-Assistent, Igny, France). The specificity of staining was carefully checked by omitting primary antibodies or replacing them with isotypic non-immune Igs.
Whole eye cryosections were treated similarly. They were permeabilized with 0.2% Triton X-100 (15 minutes), free aldehyde groups were quenched with 100 mmol/L glycine in PBS (15 minutes), shorter times were used to block free binding sites (30 minutes), primary antibodies (overnight at 4°C), and secondary antibodies (1 hour at room temperature). The immunostaining signal was increased in some experiment using a tyramide-based amplification kit (TSA Cy3 system, Perkin-Elmer) following the manufacturer's instructions.
Confocal Imaging
Images were recorded on a confocal microscope (TCS-SP2, Leica Microsystems) equipped with a × 40 oil-immersion objective (NA = 1.00). The three channels were acquired sequentially with the following excitation and emission parameters: (488 nm, 500 to 540 nm) for Alexa-488, (543 nm, 555 to 615 nm) for Alexa-555, and (633 nm, 645 to 750 nm) for TOPRO-3. Gains were adjusted to avoid saturation pixel intensities. The three channels were color coded yellow, magenta, and cyan and merged using Image J software, National Institutes of Health, Bethesda, MD, USA. No postimaging treatment was used except for simple noise removal with the despeckle function in Image J. We sometimes acquired z-stacks at various voxel depths and used maximum intensity projections to obtain 3D representations. Imaging was performed on the Cellular and Molecular Imaging facility of the IMTCE-IFR71 (Faculté de Pharmacie, Université Paris Descartes).
Isolation of Mouse Brain Microvessels, Cortex, and Kidney Samples
Brain microvessels were isolated as described by Yousif et al (2007). We used kidney samples as positive controls for our PCR because this tissue has a high concentration of Octs mRNA. Left kidneys, retinas, and brain cortices were each dissected out, frozen, and stored at −80°C.
RNA Extraction and Reverse Transcription
Total RNA was extracted from individual cortices, retinas, and kidneys or pooled brain microvessels using the RNeasy extraction kits according to manufacturer's recommendations (Qiagen, Courtaboeuf, France). The concentrations of RNA were assessed with a NanoDrop ND-1000 spectrophotometer (Labtech, Palaiseau, France). Total RNA (1 μg) was used for Reverse Transcription (RT) as previously described (Marie-Claire et al, 2004). The cDNAs so obtained were stored at −20°C.
Real-Time Quantitative RT-PCR
Real-time quantitative PCR amplification was detected by SYBR Green fluorescence on a LightCycler instrument using the LightCycler FastStart DNA Master SYBR Green I kit (Roche Diagnostics, Meylan, France). The cDNAs were diluted 20-fold and 5 μL aliquots were added to the PCR mix for a total volume of 10 μL. The thermal cycling conditions were 8 minutes at 95°C followed by 40 amplification cycles adapted for each gene (Supplementary data). The primers (Supplementary data) were placed on separate exons or at the junction between two exons whenever possible to avoid amplification of contaminating genomic DNA. We performed no-template controls and no reverse-transcriptase controls (RT negative) for each primer pair; they produced negligible signals (Ct > 40). We used the comparative AACt method to quantify the expression of the Mate1, Pmat, Net, Oct1, 2, and 3 genes. Differences in target gene expression from that of the Hprt gene (reference gene) were determined by the formula: 2−ΔΔCtsample, where ΔCt value of the sample was determined by subtracting the Ct value of the target gene from the Ct value of the Hprt gene and ΔΔCt value was obtained by subtracting the ACt of each sample from the ACt of a sample used as a calibrator. The calibrator was chosen from the five cortex samples.
Statistical Analysis
Data are presented as mean values ± s.d. Student's unpaired t-test or one-way analysis of variance with post hoc test (Dunnett or Tukey) was used to identify significant differences between groups when appropriate. All the tests were two-tailed and statistical significance was set at P < 0.05.
Results
Permeability of the Eye and Blood–Brain Barrier for [3H]-1-Methyl-4-Phenylpyridinium
We first tested for functional biogenic amine transporters at the BBB and BRB using [3H]-MPP+, one of the highest capacity substrates known for Net, Dat, Sert, Oct1-3, Mate1, and Pmat (Amphoux et al, 2006; Buck and Amara, 1994; Duan and Wang, 2010; Koepsell et al, 2007).
We evaluated the relative contributions of the barriers of the anterior (BAB) and posterior (BRB) segments to the distribution of [3H]-MPP+ in the Swiss mouse eye (n = 5). The concentration of [3H]-MPP+ used was higher (0.8 μCi/mL) than normal (0.3 μCi/mL) to get enough signal in the anterior segment. The distribution volume (Vd) of [3H]-MPP+ in the posterior segment (retina, choroid, and sclera) was 81 ± 9% of the total eye Vd, ~ 15% in the anterior segment (cornea, iris, and ciliary bodies) and ~4% in the vitreous humor. Similar results were obtained for [3H]-dopamine (data not shown). We therefore measured the accumulation of [3H]-compounds in the whole eye in all subsequent transport experiments, and considered the Vd measured to mainly reflect transport at the BRB, so avoiding variations in tissue sampling due to manual dissection.
The Kin,brain for [3H]-MPP+ was 0.25 ± 0.08 μL/s per gram and the Kin,eye was 2.1 ± 0.10 μL/s per gram (n = 6; Figure 1) in the Swiss mouse. We compared these kinetic parameters with those for the flow marker diazepam and found extraction values Ebrain ~ 0.6% and Eeye ~ 10.9% (see Materials and methods). The eye was therefore ~18.5 times more permeable to [3H]-MPP+ than the brain (P < 0.00l).
Modulation of [3H]-1-methyl-4-phenylpyridinium ([3H]-MPP+) ocular (BRB, solid bars) and cerebral (BBB, empty bars) transport by biogenic amine and aminergic reuptake inhibitors (
P-glycoprotein (P-gp; Mdr1a) and breast cancer resistance protein (Bcrp), two ATP-binding cassette transporters that allow the unidirectional efflux of many xenobiotics at BBB/BRB, were not involved in [3H]-MPP+ transport since the accumulation of [3H]-MPP+ was not significantly modified by the presence of the specific P-gp inhibitor, 5 μmol/L PSC833, or the P-gp/Bcrp inhibitor, 3 μmol/L GF120918 in Swiss mice (data not shown). These conclusions were also supported in the Fvb strain by the lack of any significant difference between the Mdr1a, Mdr1b. Bcrp(–/–) mice lacking both P-gp and Bcrp and control wild-type Fvb mice to in situ brain perfusion transport experiments with [3H]-MPP+ (data not shown).
[3H]-1-Methyl-4-Phenylpyridinium Luminal Transport: Carrier-Mediated at the Blood–Retina Barrier but Purely Diffusive at the Luminal Blood–Brain Barrier
We used cis-inhibition experiments to determine whether transporter(s) were involved in the retina and brain transport of [3H]-MPP+ in the Swiss mouse (Figure 1A). Perfusion of [3H]-MPP+ together with unlabeled biogenic amines or aminergic reuptake inhibitors resulted in a significant decrease in [3H]-MPP+ transport in the eye but not in the brain (Figure 1A). This suggests that saturable transporters are involved in [3H]-MPP+ transport at the luminal BRB but not at the luminal BBB where it is likely to be purely diffusive. These experiments were conducted using high concentrations of unlabeled transport competitors/inhibitors, where both high (uptake1) and low (uptake2) ‘affinity’ transporters are inhibited, except for histamine which inhibits only uptake2/Octs (Figure 1A). Therefore, these experiments provide no information about the specific transporters involved.
The function of some uptake1 and uptake2 transporters can be modified by changing the pH. Acidifying the extracellular space (perfusion fluid) or reducing the intracellular proton concentration with ammonium ions (see Materials and methods) both decreased the ocular transport of [3H]-MPP+ but not its BBB luminal transport in the Swiss mouse (Figure 1B). Since MPP+ is a quaternary ammonium whose permanent charge is not affected by pH, this suggests that protons have a direct and/or indirect effect on the transporter(s) involved in the retinal transport of [3H]-MPP+. Similar effects of pH have been documented for uptake1 (Ramamoorthy et al, 1993) and uptake2 (Oct1-3, Pmat) transporters, and Mate1 (Koepsell et al, 2007: Rajan et al, 2000; Urakami et al, 1998) and can be interpreted as indicating the presence of a [3H]-MPP+/proton-antiporter mechanism (or proton trans-stimulation effect) at the BRB, although we could not identify the transporter precisely.
Ionic and Pharmacological Modulation of [3H]-1-Methyl-4-Phenylpyridinium Transport Suggesting the Involvement of the Uptake1 Carrier Net at the Blood–Retina Barrier
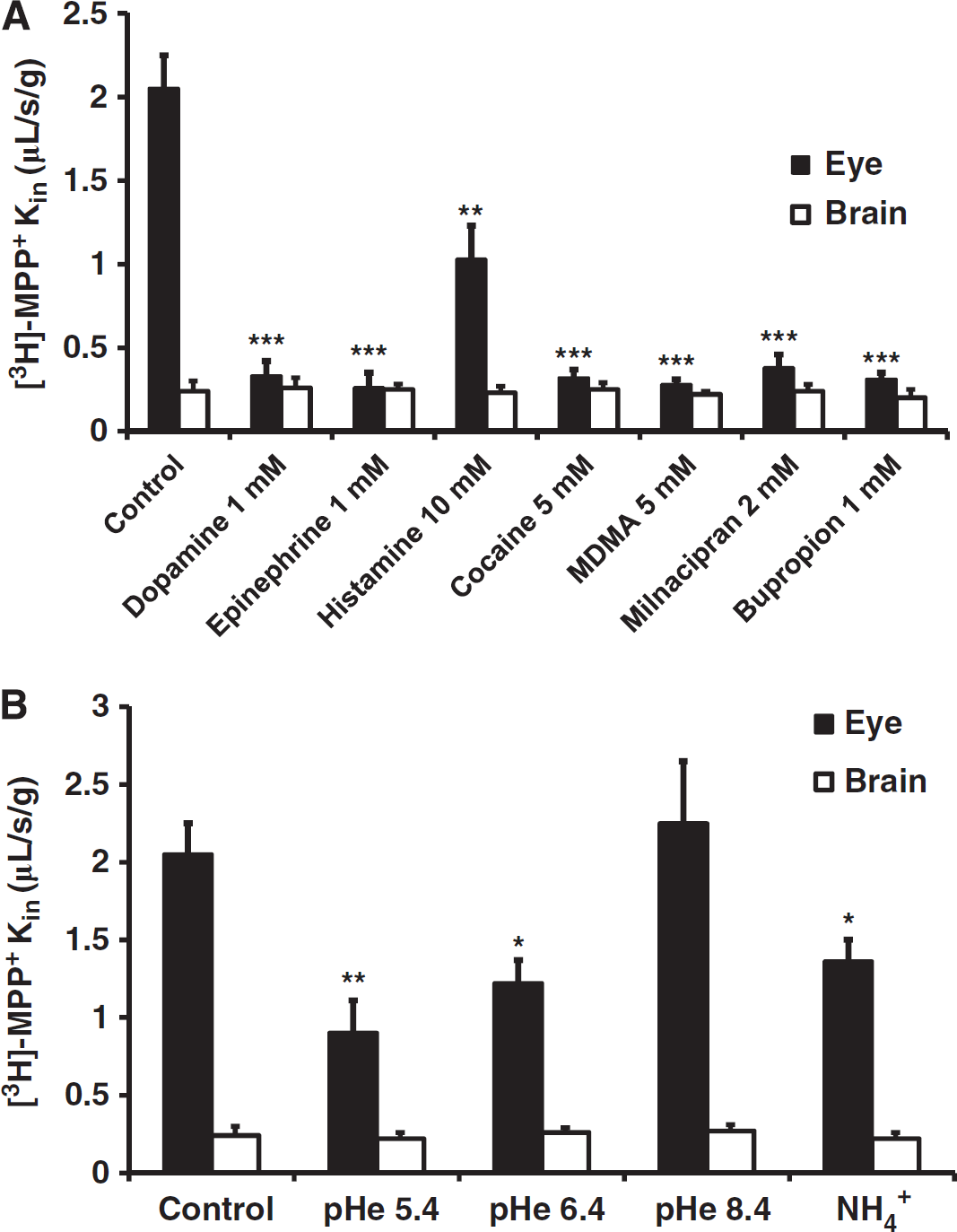
Uptake1 carriers are inhibited by a lack of vascular Cl− and even more by Na+ or Na+/Cl− deprivation, while uptake2 carriers are not. Perfusion with Cl−-free perfusate resulted in a significant 1.9-fold decrease in [3H]-MPP+ transport at the luminal BRB (‘Glu’; Figure 2A; P < 0.0l) and this decrease was even more dramatic (4.0-fold) when Na+ and Cl− were removed (‘mannitol’; Figure 2A; P < 0.00l). This suggests that uptake1 transporters of the Slc6a family (Net, Dat, and Sert) are involved in this transport. In contrast, the cerebral transport of [3H]-MPP+ was not affected by removing Na+ and/or Cl− in the Swiss mouse (Figure 2A).
Uptake1 transporters involved in the ocular (BRB, solid bars) and cerebral (BBB, empty bars) transport of [3H]-1-methyl-4-phenylpyridinium ([3H]-MPP+). Both panels show Kin (μL/s per gram) values obtained after in situ carotid perfusion of Swiss mice with 9 nmol/L [3H]-MPP + for 120 seconds. (
To gain more insight into the specific uptake1 carrier(s) involved in [3H]-MPP+ transport, we perfused the mice with [3H]-MPP+ plus each of the pharmacological transport inhibitors: citalopram, GBR12909, and nisoxetine. We used concentrations of 1 μmol/L, which were low enough to selectively inhibit the transport of Sert (citalopram), Dat (GBR12909), or Net (nisoxetine) (Kristensen et al, 2011). Nisoxetine was the only uptake1 inhibitor that significantly inhibited (3.9-fold; P < 0.00l) the retina transport of [3H]-MPP+ (Figure 2B, solid bars), suggesting that Net, or a pharmacologically similar variant is involved at the BRB. None of these inhibitors significantly altered [3H]-MPP+ transport at the Swiss mouse BBB (Figure 2B, empty bars).
Evidence that Oct1 and/or Oct2 Are the Main Uptake2 Carrier(s) Involved in the Luminal Transport of [3H]-1-Methyl-4-Phenylpyridinium at the Blood–Retina Barrier, but not at the Blood-Brain Barrier
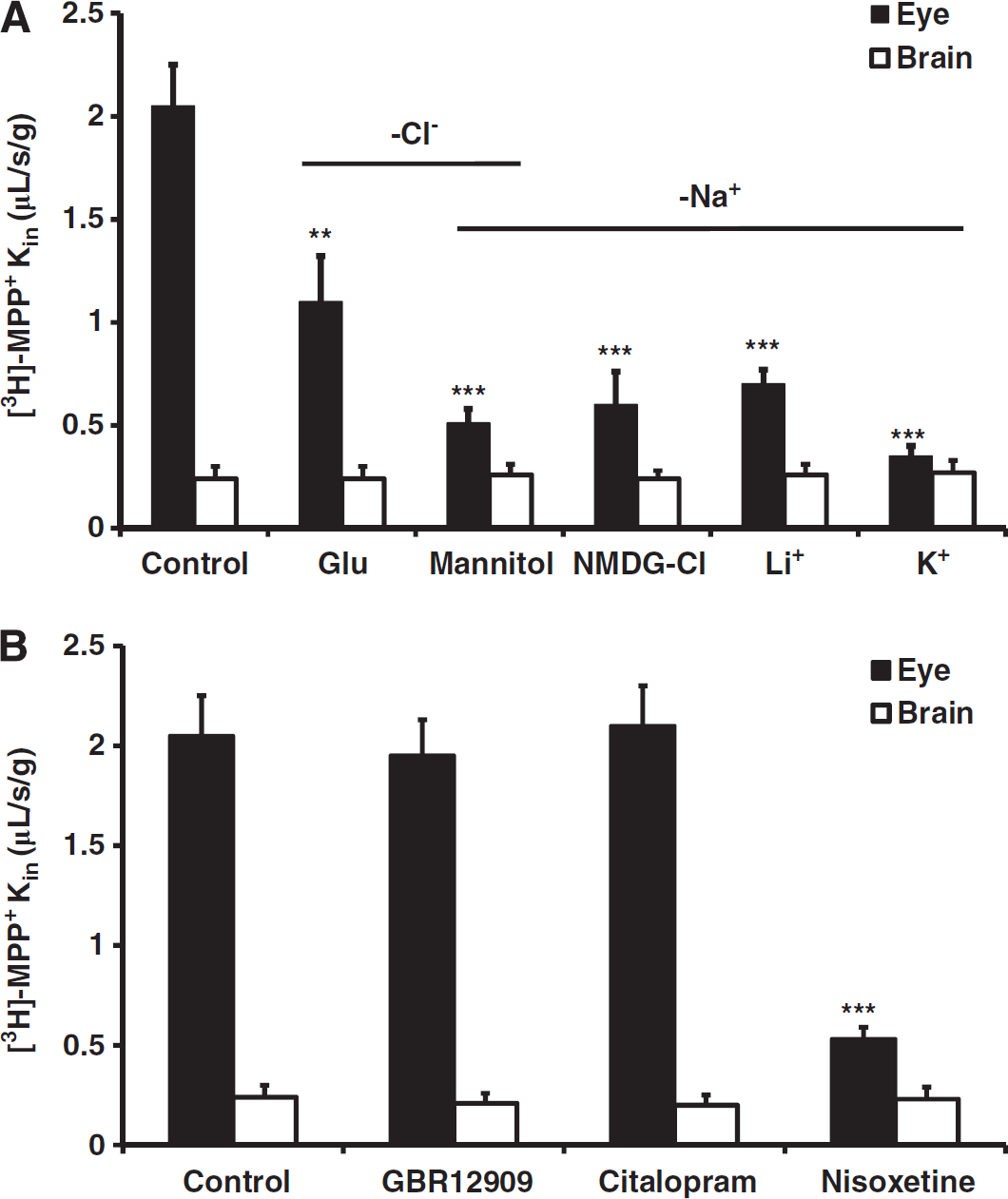
We measured the transport of [3H]-MPP+, which is also a high-capacity substrate for uptake2 carriers and for Mate1, in various Oct-deficient Fvb mice to evaluate the contributions of Oct1-3. The [3H]-MPP+ Kin,eye was significantly (2.1-fold) lower in Oct1,2 (–/–) mice (P < 0.001) than in the corresponding Fvb wild-type mice, but it was not affected in Oct3(–/–) mice (Figure 3A, solid bars). However, the brain transport of [3H]-MPP+ in Fvb wild-type mice was similar to that in Oct1,2(–/–) and Oct3(–/–) deficient mice (Figure 3A, empty bars). Thus Oct1 and/or Oct2, but not Oct3, seem to be involved in the transport of [3H]-MPP+ at the Fvb mouse BRB. However, the brain transport of [3H]-MPP+ in Fvb wild-type mice was similar to that in Oct1,2(–/–) and Oct3(–/–) deficient mice (Figure 3A, empty bars), suggesting that Oct1-3 transporters are not functional at the Fvb mouse luminal BBB. Moreover, the brain [3H]-MPP+ Kin measured in Fvb wild-type mice was 0.30 ± 0.10 μL/s per gram (Figure 3A), which was not different from that measured in Swiss mice (0.25 ± 0.08 μL/s per gram) (Figure 1).
Effects of uptake2, Oct1,2 and Oct3 transporters and uptake2 inhibitors on [3H]-1-methyl-4-phenylpyridinium ([3H]-MPP+) (9 nmol/L) transport (Kin; μL/s per gram) measured by in situ carotid perfusion of Swiss, and Oct-deficient Fvb mice for 120 seconds. (
The involvement of Octs was also assessed in cis-inhibition experiments in the Swiss mouse (Figure 3B). Many compounds inhibit/modulate transporters, although they are not necessarily themselves transported. Thus, Koepsell et al (2007) outlined a general strategy for discriminating between the functions of Oct transporters, but it does not appear to be applicable in vivo due to species differences and lack of extrapolation of transport parameters (Ki/Km/Vmax) usually obtained with transfected heterologous systems. Moreover, a combination of diverse overlapping transport systems with different affinities, such as uptake1 and uptake2, make it difficult to quantify individual transporter components. Therefore, we used broad Octs inhibitors (TEA and corticosterone) and more specific Oct2/Oct1 inhibitors (guanidine, agmatine, and cisplatin) (Koepsell et al, 2007). All these compounds similarly and significantly reduced ~1.7-fold the [3H]-MPP+ eye transport (Figure 3B). By contrast, carnitine, the amphoteric nutrient substrate of Octn transporters did not modulate the transport of [3H]-MPP+ whereas choline only slightly reduced 1.3-fold (P < 0.05) the eye transport (Figure 3B, solid bars). None of these pharmacologic transport inhibitors affected the luminal transport of [3H]-MPP+ at the Swiss mouse BBB (Figure 3B, empty bars). This confirms that [3H]-MPP+ transport at the BRB is partly mediated by Oct1 and/or Oct2 in the Fvb and Swiss mice, while Oct1, 2, or 3 is not involved in the luminal BBB transport of [3H]-MPP+ at the Swiss and Fvb mouse strains.
Evidence from Biogenic Amine Neurotransmitter Transport Experiments that there Are Functional Uptake1 and Uptake2 Carriers at the Luminal Blood–Retina Barrier but not at the Luminal Blood–Brain Barrier
We then examined the luminal transport of selected biogenic amines at the BRB and BBB to verify the results obtained with [3H]-MPP+. The same experiments were also performed at the BBB as ‘control’ experiments, as [3H]-MPP+ transport at this site is not carrier mediated. The tissue distributions of [3H]-histamine (Oct2 < Oct3 substrate), [3H]-serotonin (Sert; Oct2 < Oct3; Pmat substrate), and [3H]-dopamine (Dat < Net; Oct1 < < Oct2 < Oct3; Pmat substrate) were measured after in situ carotid perfusion for 60 seconds (Amphoux et al, 2006; Duan and Wang, 2010; Eisenhofer, 2001; Figure 4). The Kin values obtained from brain and eye in control Swiss mouse experiments (Figures 4A–4C) indicated that the luminal BBB extraction value (Ebrain) calculated (see Materials and methods) for [3H]-histamine was 0.1%, that of [3H]-serotonin was 0.4%, and that of [3H]-dopamine was 3.0%; their corresponding ocular extraction values (Eeye) were 1.1%, 2.7%, and 11.4%, respectively.
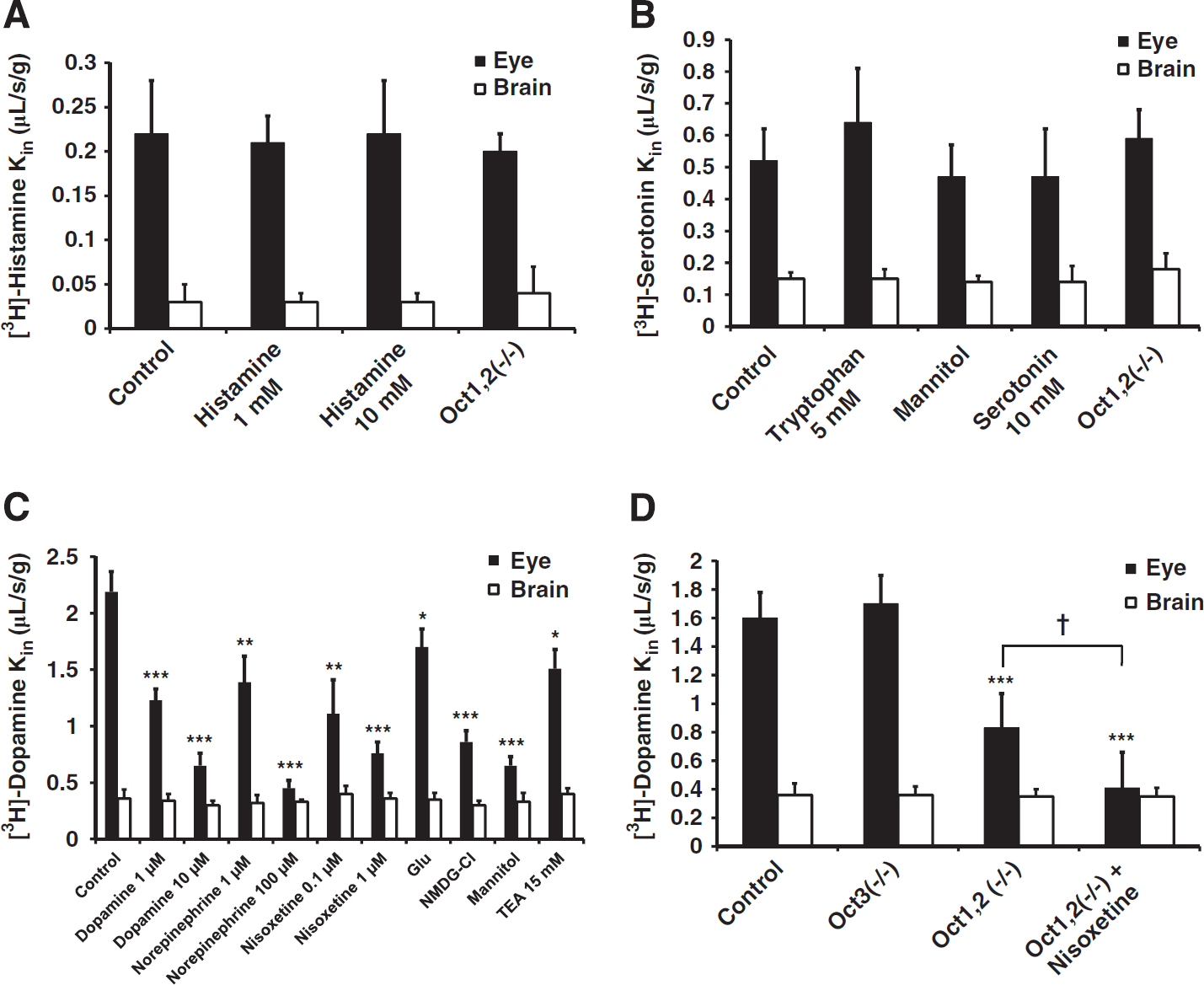
Eye (BRB, solid bars) and brain (BBB, empty bars) transport of [3H]-histamine (
The retinal transport of [3H]-histamine (Kin,eye ~ 0.2 μL/s per gram; Figure 4A) was not inhibited by 1 or 10 mmol/L unlabeled histamine in Swiss mouse and was unchanged in Oct1,2(–/–) Fvb mice (Figure 4A), suggesting that its membrane permeability does not significantly involve the histamine transporters Oct2 and Oct3 in the Swiss and Fvb mice. The retinal transport of [3H]-serotonin (Kin,eye ~0.5 μL/s per gram; Figure 4B) was not inhibited by 5 mmol/L L-tryptophan, 10 mmol/L serotonin or Na+/Cl− depletion, and unchanged in Oct1,2(–/–) mice (Figure 4B, solid bars). This suggests that the transport of serotonin does not involve the amino-acid carrier L (Lat1/Slc7a5), which is known to transport biogenic amines precursors like tryptophan and L-dopa at the BBB. Likewise, Sert, Pmat, Oct2, and Oct3 are not involved in the serotonin transport at the Swiss mouse luminal BRB and BBB (Figure 4C). The ocular transport of [3H]-dopamine (Kin,eye ~ 2.2 μL/s per gram; Figure 4C) was dose dependently inhibited by dopamine, norepinephrine, and nisoxetine, even at very low concentrations (Figure 4C, solid bars). The ocular transport of [3H]-dopamine (Figure 4C) was also decreased 1.5-fold (P < 0.05) by TEA an uptake2 inhibitor, by Cl− depletion (‘Glu’; 1.4-fold; P < 0.05), and strongly reduced (3.4-fold) by Na+/Cl− depletion (‘mannitol’; P < 0.00l) in Swiss mice. The ocular transport of [3H]-dopamine (Figure 4D) is significantly decreased (2.0-fold; P < 0.001) in Oct1,2(–/–) Fvb mice, and by nisoxetine inhibition in Oct1,2(–/–) mice (P < 0.05). Thus, a substantial fraction of dopamine transport at the BRB involves an uptake1 carrier pharmacologically similar to Net, and an uptake2 carrier (Oct1 and/or Oct2), in agreement with the results of the [3H]-MPP+ eye transport experiments in Swiss and Fvb mice.
None of the above inhibitors affected the luminal BBB transport values for [3H]-histamine, [3H]-serotonin, and [3H]-dopamine in Swiss mice (Figures 4A–4C; empty bars), indicating that the luminal transport of these biogenic amines at the BBB is not mediated by uptake1 and uptake2. Similarly, the luminal BBB transport in Fvb mice does not involve Oct1, Oct2, Oct3, and Net carriers (Figures 4A–4D). The brain and eye transport of [3H]-histamine and [3H]-serotonin measured in wild-type control Fvb mice were statistically not different from the Kin values obtained in control Swiss mouse (Supplementary data). This could suggest that the Fvb mice, like the Swiss strain, do not have serotonin transporters like Sert and Pmat at the luminal BBB.
Mate1, Oct1, 2, 3 and Pmat mRNAs in Mouse Brain Microvessels
The functional experiments probed only luminal transport at the BBB and BRB. We measured the amounts of the mRNAs encoded by the Mate1, Oct1, Oct2, Oct3, and Pmat genes in Swiss mouse brain microvessels, brain cortices, and kidney by qRT-PCR to learn more about the global luminal and abluminal synthesis of uptake2 carriers (Table 1A). The kidneys contained much more Mate1, Oct1, and Oct2 mRNAs than did the brain cortices (5,312 times, P < 0.001 (Mate1), 9,329 times (Oct1), P < 0.001 and 3,696 times, P < 0.001 (Oct2)) and all three mRNAs were undetectable in brain microvessels (Ct > 36). Although the amounts of Oct3 mRNAs in the kidney and brain cortices were similar, there was significantly less in brain microvessels (sixfold, P < 0.00l). The amounts of Pmat mRNAs in the brain cortices and microvessels were similar and much higher than in the kidney (50 times, P < 0.00l).
Quantitative RT-PCR data showing the relative amounts of mRNAs encoded by Mate1, Octs1, 2, 3 and Pmat in brain cortices (n=5), brain microvessels (n=3), and kidney (n=4 to 5) from Swiss mice (A) and Net, Octs1 and 2 in brain cortices (n=3 to 5) and retinas (n=5) (B) from Swiss mice
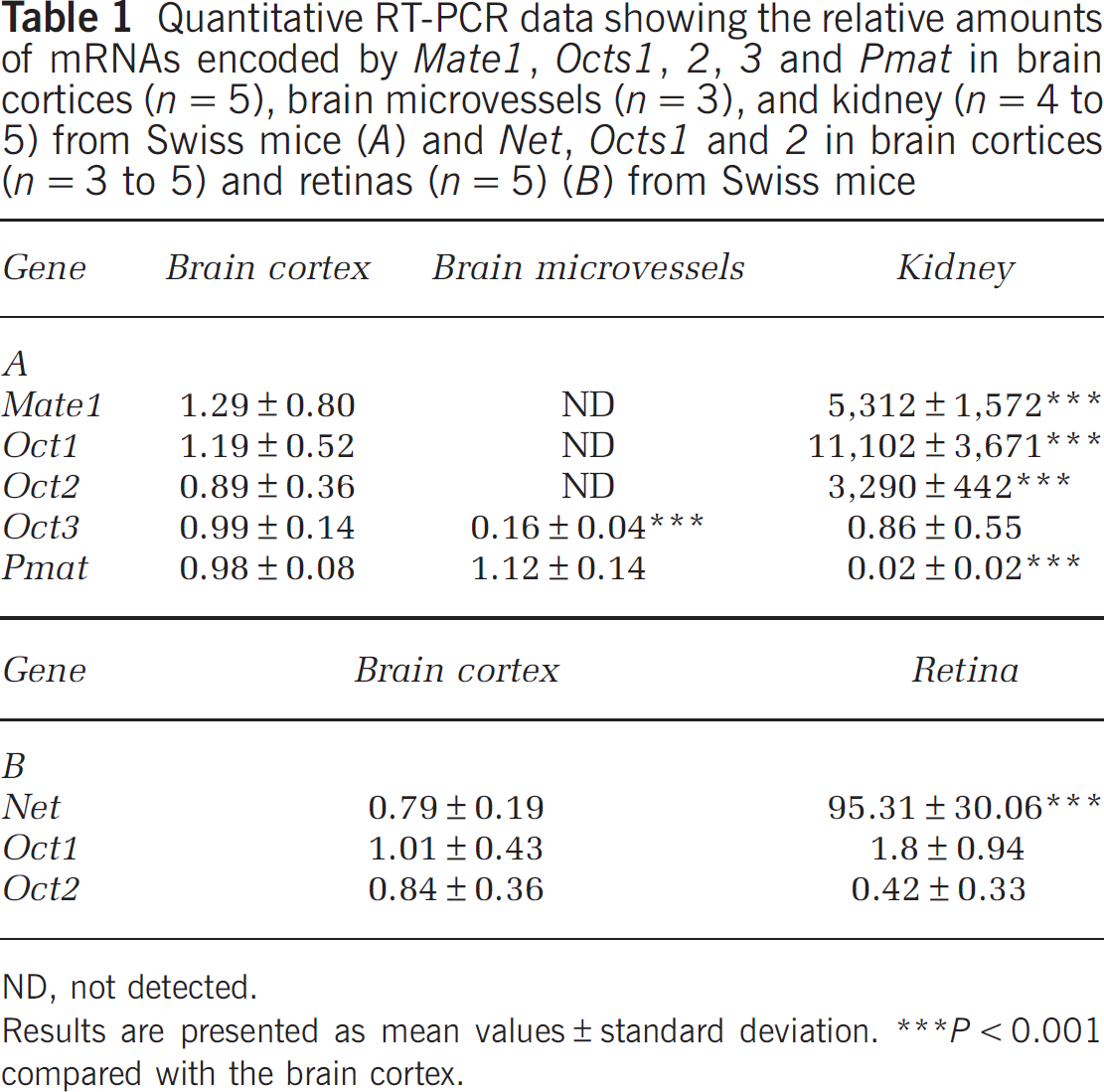
ND, not detected.
Results are presented as mean values±standard deviation.
P<0.001 compared with the brain cortex.
Net, Oct1, and Oct2 mRNAs in Mouse Retinas
We measured the mRNAs of these three genes in Swiss mouse retinas to determine whether the observed transport at the BRB was due to Net, Oct1, or Oct2 (Table 1B). The retinas contained 120 times more Net mRNAs (P < 0.00l) and similar amounts of Oct1 and Oct2 mRNAs than did the brain cortices.
Immunodetection of Transporters at the Blood–Brain Barrier and the Blood–Retina Barrier
We assessed the amounts of Net, Sert, Dat, Pmat, and Oct3 proteins at the Swiss mouse BBB by immunostaining mouse brain sections and confocal imaging. We used antibodies to P-gp (Mdr1a, Abcb1a), which lies at the luminal side of the endothelium, or laminin, a major constituent of blood vessel basal lamina, to identify blood vessels. Dat, Net, and Sert were readily shown in numerous nerve fibers, some of them running parallel and close to capillaries (Figure 5A). However, these uptake1 transporters were not detected in the endothelium, either luminally or abluminally. The anti-Oct3 antibody labeled some fibers in the mouse brain, particularly near the ventricles (Figure 5B), in agreement with previous reports (Amphoux et al, 2006; Vialou et al, 2004). However, we detected no Oct3 in the cerebral vessels, suggesting that this transporter is not present at the BBB. Similar results were obtained in Pmat immunostaining experiments, showing that this transporter is abundant in nerve fibers but undetectable in cerebral vessels, either luminally or abluminally (Figure 5B).
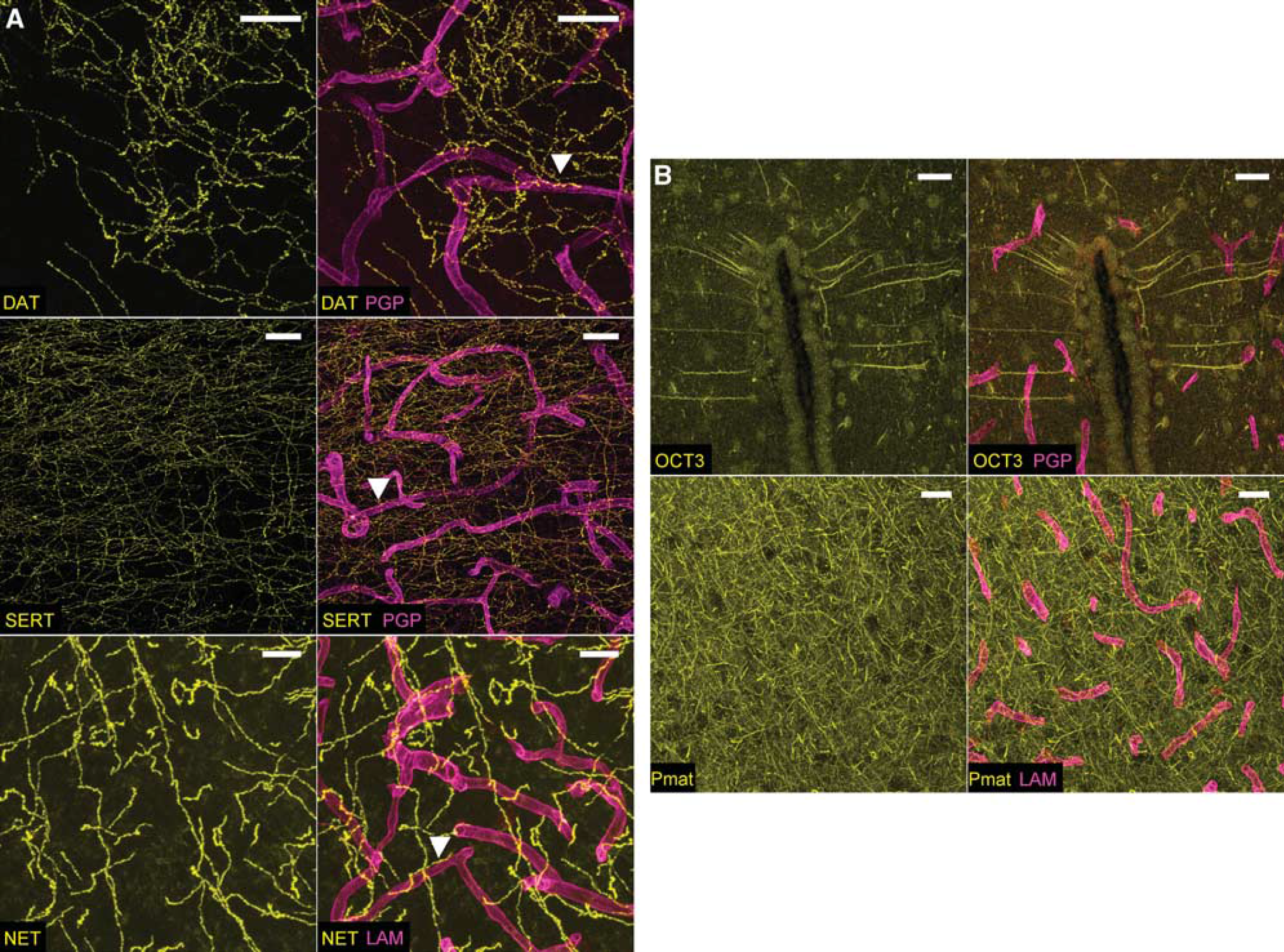
Uptake1 (Dat, Sert, Net) and uptake2 (Oct3, Pmat) immunostaining in the Swiss mouse brain. (
We also looked for uptake1 transporters at the ocular barriers using staining for laminin or actin to delineate the RPE and immunostaining for P-gp or Bcrp (Abcg2) to identify blood vessels in the retina (BRBi) and the iris/ciliary body (BAB) from the Swiss mouse. Dat and Sert were not detected at the BRBi, BRBo, or the BAB (Supplementary data). The anti-Net antibody produced no specific signal in the retina (Figure 6A, left panel) or in the RPE (Figure 6A, central panel) even after tyramide-based signal amplification (data not shown). On the contrary, this antibody gave a strong staining in the iris/ciliary body. This labeling was not in capillaries, but in noradrenergic varicose fibers (Figure 6B, right panel), as confirmed by the complete overlap of immunostaining for Net and tyrosine hydroxylase (Figure 6B).
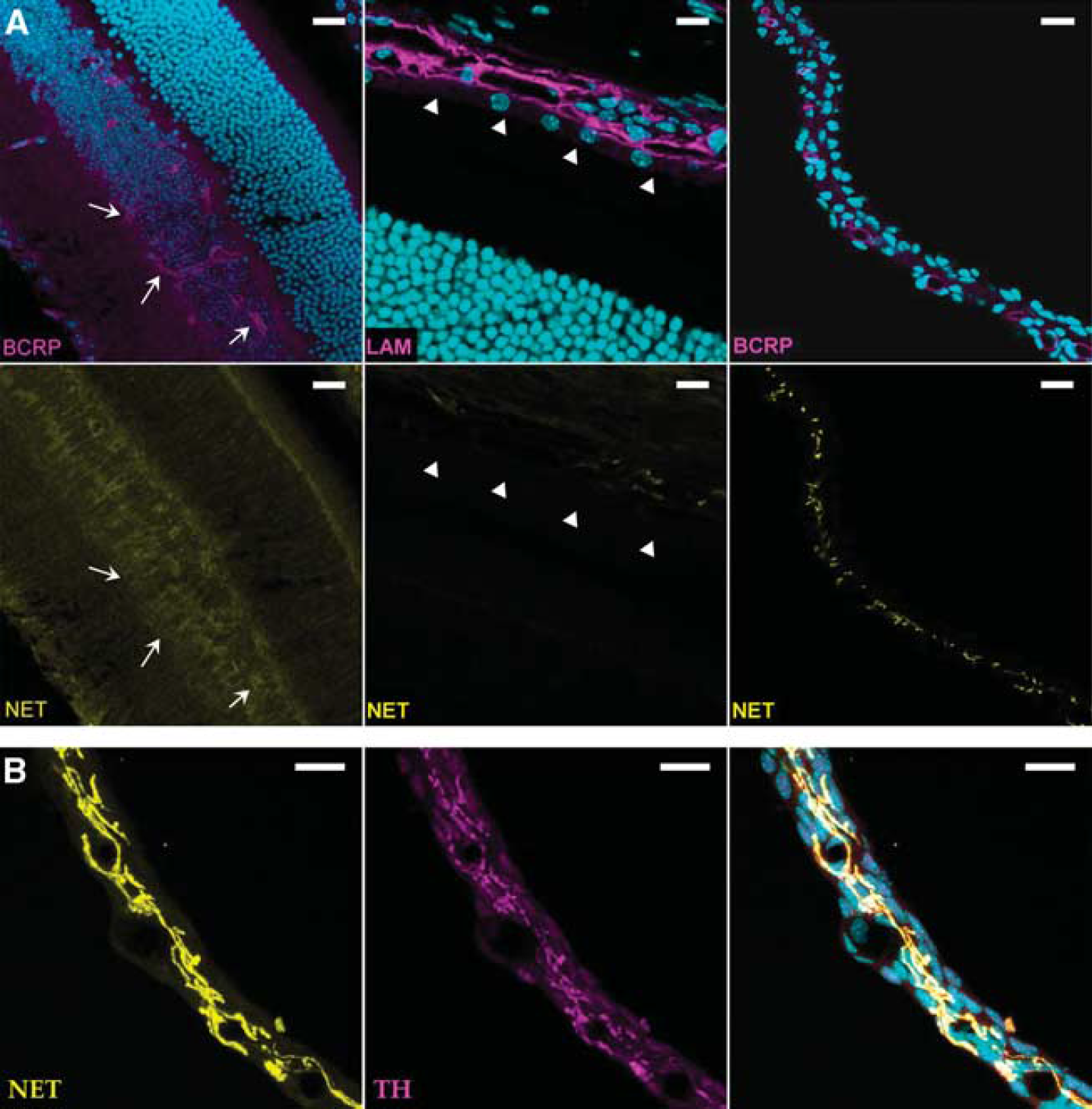
Cryosections immunostained for Net in the Swiss whole eye. Nuclei are stained with TOPRO-3 (cyan). (
Discussion
Blood–nerve barriers like the BBB and the BRB are equipped with specific sets of transporters that facilitate the selective transport of endogenous/hydrophilic compounds from the blood into the nerve parenchyma and the elimination of compounds/metabolites into the bloodstream. At the abluminal side, chemical and ionic mediators permit complex local interplays between the cells of the NVU, as illustrated by the process of coupling responses (Leybaert, 2005). Unlike glutamate and GABA transporters, whose functions at the brain NVU have been convincingly established, the functions of the uptake1 and the more recently identified uptake2 transporters are still debated (Abbott et al, 2010; Lin et al, 2010; O'Kane et al, 1999).
Very few published studies have looked for uptake1 transporters at the BBB. Net and Sert mRNAs have been found at the BBB of mouse TM-BBB4 cells and these proteins were detected immunohistochemically in mouse brain capillaries (Wakayama et al, 2002). Sert was also reported to be functional in RBE4 cells, an immortalized rat brain endothelial cell line (Brust et al, 2000). Molecular and/or functional studies with in vitro BBB models have provided conflicting results about the presence of uptake2 carriers at the BBB; some concluded that Octs are not present (Friedrich et al, 2001). While functional Oct1 and/or Oct2 have been suggested at the BBB using in vitro models (Liou et al, 2007; Dickens et al, 2012) and in vivo by extracellular fluid microdialysis (Lin et al, 2010), these results may not be conclusive. The isolation and culture of endothelial cells for use in in vitro BBB models often leads to the dysregulation of transporters (Lyck et al, 2009). Moreover, brain extracellular fluid microdialysis sampling experiments do not discriminate between the kinetic processes at the BBB and those at the other brain extracellular fluid/intracellular fluid interfaces where these transporters are present (e.g., neurons and ventricles) (Amphoux et al, 2006; Vialou et al, 2004). Our results indicate that the luminal BBB permeability to MPP+, a broad, high-capacity substrate of biogenic amine uptake1 and uptake2 transporters, and the organic cation transporter Matel, is not carrier mediated. The luminal BBBs of Fvb and Swiss mice are similar in that they are both ‘weakly’ permeable to MPP+, serotonin, histamine, and dopamine. These findings are also similar to those of Oldendorf (1971) for Wistar rats. This reflects passive diffusion alone, as these permeabilities were not altered by modulating uptake1 and/or uptake2 transporters, silencing Oct1,2,3 carriers, or ionic manipulations. These experiments confirm that the blood side of the BBB cannot mediate transport by uptake1, uptake2, or Mate1 in vivo. The lack of uptake1 and uptake2 transporters at the luminal BBB limits both the entry of biogenic amines and their clearance. But the metabolic activities of the cytosolic monoamine oxidases and catechol-O-methyl transferase could further reduce the concentrations of these amines in tissues and their extracellular availability (Ghersi-Egea et al, 1988; Shawahna et al, 2011). Few in vivo studies have investigated the presence and role of biogenic amines transporters in clearance and dissipation at the abluminal BBB. Pmat could be involved in the clearance and dissipation of dopamine and serotonin at the abluminal/nervous side of the BBB in addition to its role in their neuronal transport (Dahlin et al, 2007; Duan and Wang, 2010; Okura et al, 2011). But, we could not show either Oct3 or Pmat in endothelial cells by immunostaining or luminal transport experiments. Therefore, the small amounts of the mRNAs encoding Oct3 and Pmat that were detected in isolated cerebral microvessels are probably due to the presence of contaminating nonendothelial cells. Thus, they are unlikely to function at the BBB.
We found differences in the transport capacities of the luminal brain and retina barriers. Biogenic amine transport could be favored at the BRB but not at the BBB. We suggest that Net is responsible for the uptake1-mediated transport of MPP+ and dopamine across the luminal BRB. In agreement, the transport of [3H]-serotonin at the eye was not Na+/Cl− dependent and Dat and Sert were not detected in immunostained eye cryosections. Although qRT-PCR confirmed the presence of Net mRNA in the retina, immunolabeling of whole eye cryosections using the anti-Net antibody (aa residues 5 to 17) detected the transporter only in some nerve fibers of the iris and the ciliary bodies, as previously reported (Laties, 1974). The lack of anti-Net labeling in the retina could be due to its presence as a truncated Net variant not recognized by our antibody, or to the masking of the epitope by an interacting partner (Sager and Torres, 2011). However, additional qRT-PCR experiments using primers designed to reflect the epitope region also detected Net in the retina, thus ruling out the presence of an N-terminal truncated Net variant (data not shown). Moreover, western blotting experiments, in which all noncovalently bound protein complexes are dissociated, successfully detected Net in brain extracts but not in retina extracts (data not shown). We therefore conclude that Net transporter may undergo posttranslational modifications that affect its recognition by the antibody and/or that a functionally similar transporter was involved. Functional strategies do not readily identify variants of transporters (Buck and Amara, 1994), and methods of molecular identification like immunohistochemistry have not been performed for these extraneuronal uptake1 locations (Catravas and Gillis, 1983; Chemuturi and Donovan, 2007; Ramamoorthy et al, 1993).
Another eye kinetic component points to the presence of uptake2 transport mediated by Oct1 and/or Oct2. The reports of OCT3 gene expression in the human RPE cell line ARPE are conflicting (Rajan et al, 2000), whereas another qRT-PCR study detected high amounts of OCT1 mRNA in the human retina, but neither OCT2 nor OCT3 transcripts (Zhang et al, 2008). According to the capacity/affinity of the substrates, our functional studies suggest that the uptake2-mediated transport in the retina is mainly by luminal Oct1 rather than by Oct2. It remains to be established whether these transporters are present at the luminal BRBi and/or the RPE, since our approach cannot kinetically distinguish between these two retinal barrier sites. In vitro studies have shown that dopamine is released from the RPE, although the transporter(s) involved was not identified (Ming et al, 2009). This supports the idea of using these cells in implants to treat Parkinson's disease (Farag et al, 2009).
The pathophysiological implications of the presence of uptake1 and uptake2 at the retina remain to be clarified. Uptake2/Octs transports substrates according to their concentration gradients, whereas uptake1 transporters are unidirectional. The transport of dopamine/norepinephrine from the blood into BRB cells could provide intermediates for the synthesis of melanins and melatonin, which take place mainly at the RPE. Dopamine also has neuromodulatory/endocrine functions and a trophic/regulatory role in organs (Eisenhofer, 2001; Ugrumov et al, 2012). These extraneuronal uptake1 and uptake2 transporters at the BRB could perhaps be involved in ferrying dopamine to some tissue/target sites for endocrine communication and regulation. Similarly, Oct3 in neurons of the circumventricular subfornical organs that lack a tight BBB may act as an indirect endocrine blood ‘sensor’, and are involved in the regulation of the blood osmolarity by controlling the salt intake (Vialou et al, 2004).
We conclude that (1) there is no carrier-mediated transport of biogenic amines at the luminal BBB, (2) there are no detectable uptake1 and uptake 2 transporters at the luminal and abluminal mouse BBB, and (3) the mouse luminal BRB has an uptake1 transporter, Net, or a functionally similar transporter, and an uptake2 transporter, functionally identified as Oct1. These dissimilar transport properties of the brain and retina NVUs suggest that the BBB is more involved in protecting the brain from biogenic amines at the luminal/blood side while the presence of biogenic amine transporters at the blood side of the BRB suggest that it has more of a metabolic/endocrine role.
Disclosure/conflict of interest
The authors declare no conflict of interest.
Footnotes
Acknowledgements
The authors thank Prof. Hervé Galons for generously supplying MDMA. The authors also thank Capucine Besengez for technical assistance and Dr Owen Parkes for editing the English text.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.