Abstract
Cerebral fractional oxygen extraction (FOE) represents the balance between cerebral oxygen delivery and consumption. This study aimed to determine cerebral FOE in preterm infants during hypotension, during moderate anemia, and with changes in the PaCO2. Three groups of neonates were studied: stable control neonates (n = 43), anemic neonates (n = 46), and hypotensive neonates (n = 19). Cerebral FOE was calculated from the arterial oxygen saturation measured by pulse oximetry, and cerebral venous oxygen saturation was measured using near infrared spectroscopy with partial jugular venous occlusion. Mean ± SD cerebral FOE was similar in control (0.292 ± 0.06), anemic (0.310 ± 0.08; P = 0.26), and hypotensive (0.278 ± 0.06; P = 0.41) neonates. After anemic neonates were transfused, mean ± SD cerebral FOE decreased to 0.274 ± 0.05 (P = 0.02). There was a weak negative correlation with the hemoglobin concentration (n = 89, r = −0.24, P = 0.04) but not with the hemoglobin F fraction (n = 56, r = 0.24, P = 0.09). In the hypotensive neonates, there was no relationship between cerebral FOE and blood pressure (n = 19, r = 0.34, P = 0.15). There was a significant negative correlation between cerebral FOE and PaCO2 within individuals (n = 14, r = −0.63, P = 0.01), but there was no relationship between individuals (n = 14, r = 0, P = 1). Cerebral FOE was not significantly altered in neonates with either mild anemia or hypotension. There were, however, changes in cerebral FOE when physiological changes occurred over a relatively short period; Cerebral FOE decreased after blood transfusion and increased with decreasing PaCO2. As no change in cerebral FOE was seen during hypotension, it was speculated that cerebral oxygen delivery may have been maintained by cerebral blood flow autoregulation.
The adequacy of cerebral oxygen delivery to meet demands is not monitored directly during neonatal intensive care, although it is affected by many routine interventions. For example, cerebral blood flow (CBF) has been shown to decrease after the administration of indomethacin (Edwards et al., 1990) and hyperventilation (Leahy et al., 1980). There are common clinical interventions to improve organ perfusion: Blood transfusions are given to correct anemia, and inotropic support is used for hypotension. However, the effects of these interventions on cerebral oxygen delivery and metabolism are neither fully understood nor monitored.
One approach to a better understanding of cerebral oxygenation is through the measurement of cerebral fractional oxygen extraction (FOE). This represents the amount of oxygen extracted by the brain as a proportion of that delivered. Cerebral FOE can be calculated if the arterial oxygen saturation (SaO2) and cerebral venous oxygen saturation (SvO2) are known and is given by the function FOE = [(SaO2 − SvO2)/SaO2]. A noninvasive method of measuring cerebral SvO2 using near infrared spectroscopy (NIRS) with partial jugular venous occlusion has been validated (Yoxall et al., 1995) and used to study changes in cerebral FOE in children undergoing hypothermic cardiopulmonary bypass (Wardle et al., 1998b) and changes in cerebral oxygen consumption in preterm neonates (Yoxall and Weindling, 1997).
The important factors that influence the amount of oxygen delivered to the brain are the concentration of hemoglobin (Hb), its affinity for oxygen, and CBF, which depends on blood pressure and cerebrovascular resistance. Anemia has been reported to result in significant alterations in CBF, and blood transfusion is presumed to improve cerebral oxygen delivery in preterm neonates (Nelle et al., 1994; Ramaekers et al., 1988). Although cerebral oxygen extraction has not previously been studied in preterm neonates during anemia, it has been generally assumed that it would not change because of compensatory changes in CBF. The first hypothesis of this study, therefore, was that no changes in cerebral FOE would be observed in the presence of anemia.
An important determinant of CBF is the partial pressure of carbon dioxide (PaCO2) (Greisen and Trojaborg, 1987; Hansen et al., 1986; Leahy et al., 1980; Reuter and Disney, 1986; Rosenberg et al., 1988), probably through its effect on cerebrovascular resistance. A fall in PaCO2 results in a fall in CBF; and to maintain the same cerebral metabolic rate for oxygen, an increase in cerebral FOE would be expected. The second hypothesis of this study was therefore that acute changes in PaCO2 would result in changes in cerebral FOE and that cerebral FOE would correlate negatively with PaCO2.
The final part of this study investigated the interaction between blood pressure and cerebral FOE. Hypotension in preterm neonates has been associated with a poor outcome in terms of mortality and morbidity (Bada et al., 1990; Fujimara et al., 1979; Miall-Allen et al., 1987; Szymonowicz et al., 1984; Watkins et al., 1989). The rationale for the therapeutic support of blood pressure is based on the assumption that a low blood pressure is associated with low systemic blood flow (Miall-Allen and Whitelaw, 1989; Report of Joint Working Group, 1992) and therefore impairs oxygen delivery to the tissues. Low systemic blood flow is also associated with changes in the distribution of flow so that perfusion of vital organs such as the brain and the heart is maintained at the expense of flow to peripheral tissues (Finch and Lenfant, 1972; Sidi et al., 1983). This is likely to be true until the blood pressure falls below a limit at which further redistribution of flow is not possible, and at this point oxygen delivery to vital organs including the brain would be expected to be compromised. Provided cerebral oxygen consumption remains constant, a fall in CBF (and cerebral oxygen delivery) would be expected to be compensated by an increase in cerebral FOE. We therefore hypothesized that cerebral FOE would rise during clinically significant hypotension.
METHODS
Cerebral FOE can be calculated from SaO2 and cerebral SvO2: cerebral FOE = (SaO2 − cerebral SvO2)/SaO2. SaO2 was measured using pulse oximetry. Cerebral SvO2 was measured using NIRS with partial jugular venous occlusion (Yoxall et al., 1995) and is briefly described below.
Near infrared spectroscopy cerebral venous oxygen saturation measurements
Measurements were made using the Hamamatsu NIRO 500 (Hamamatsu Photonics, Enfield, U.K.) and a pulse oximeter (Datex-Ohmeda, Helsinki, Finland) in beat-to-beat mode. The NIRO 500 has four laser diodes for measurements with wavelengths of 774, 825, 843, and 911 nm. The light is transmitted through two 1.5-m fiberoptic cables. The monitoring optodes were positioned between 3 and 6 cm apart on the neonate's head in the frontotemporal region as far apart as possible. Brief partial venous occlusions of ∼8- to 10-second duration were made with gentle pressure unilaterally over the jugular vein (Yoxall et al., 1995). Measurements were analyzed off-line using Microsoft Excel by considering the initial rise of the oxygenated Hb and Hb during the first 5 seconds after the start of the occlusion. Cerebral SvO2 was measured five times during a 5- to 10-minute period for each study, and the mean value was used. Approximately 30 seconds was allowed between occlusions. Measurements were made when neonates were quiet and at rest to minimize movement artefact. The pulse oximeter optodes were positioned on the right forearm or hand. The SaO2 was recorded continuously in beat-to-beat mode, and the mean value of SaO2 during each measurement of cerebral SvO2 was used to calculate the cerebral FOE. The SaO2 was maintained in the range of 88 to 94% for neonates at <32 weeks' and above 92% in neonates at >32 weeks' gestation (Cochran and Shaw, 1995). Measurements took ∼10 minutes to complete.
Subjects and measurements
Four groups of neonates were studied.
Group 1: control group. These were stable preterm neonates of any gestational age, nursed in the neonatal unit. They had normal arterial oxygen saturations and blood gases, were not receiving respiratory or inotropic support, and were not considered to need blood transfusions by the clinicians responsible for their care. Blood pressure was not measured invasively in these neonates, who were all assumed to be normotensive. All the neonates in this group had only one measurement of cerebral SvO2.
Group 2: anemic group. Neonates in this group were considered by the clinicians responsible for their care to need blood transfusions on the basis of a standard consensus protocol used routinely on our unit and that has been used in other studies (Shannon et al., 1995). Some neonates in this group were ventilated and were transfused because of a low Hb concentration alone, others were transfused because of symptoms attributed to anemia. Measurements of cerebral SvO2 were repeated 12 to 24 hours after the blood transfusions (20 mL/kg packed cells).
Neonates in Groups 1 and 2 had measurements of Hb concentration and Hb F fraction (HbF) at the time of the initial cerebral SvO2 measurement, except when an insufficient volume of blood was taken. The arterial oxygen content was calculated from the following formula: arterial oxygen content (mL/dL) = Hb × 1.36 × SaO2, where 1.36 is the oxygen capacity of Hb in milliliters per gram.
Group 3: hypotensive group. Neonates in this group were all ventilated and had invasive arterial blood pressure monitoring. Measurements were made when there was a clinical decision to treat hypotension and the mean arterial blood pressure was less than the tenth centile (Watkins et al., 1989). Measurements were first made before any treatment to correct hypotension had been given. Measurements of cerebral SvO2 were then repeated after treatment for hypotension when the blood pressure had become normal. The outcome for the neonates in the hypotensive group was assessed as (1) survival to discharge from the neonatal unit and (2) the presence of significant periventricular hemorrhage (PVH), defined as intraventricular hemorrhage with ventricular dilatation or parenchymal hemorrhage.
Group 4: group with changes in partial pressure of carbondioxide. Cerebral SvO2 was measured before and after changes in ventilator settings that produced a significant change in PaCO2. The changes were made when the PaCO2 was outside its normal range, with the aim of returning the arterial pH to within normal limits (7.25 to 7.40). Some of the neonates included in this group were also included in Groups 2 and 3, but measurements in relation to PaCO2 were not made when the neonates were hypotensive, on inotropes, or anemic.
A measure of the repeatability of measurements was assessed in a subgroup of neonates using methods described by Bland and Altman (1986). Two consecutive measurements (each the mean of five venous occlusions) were made on the same individual within 10 minutes by repositioning of the NIRS optodes before the second measurement.
Statistics
Data were analyzed using SPSS for Windows (version 4). The relationship between variables was assessed using Pearson's correlation coefficients. Differences between groups were assessed using unpaired t tests for parametric data or Mann-Whitney U tests for nonparametric data. Changes in cerebral FOE after interventions (transfusions, treatment for hypotension, and changes in ventilator settings) were analyzed using paired t tests.
This study was approved by the local research ethics committee, and written parental consent was obtained.
RESULTS
There were 43 neonates in the control group, 46 in the anemic group, 19 in the hypotensive group, and 14 in the ventilated group in which measurements were made after changes in PaCO2. Details of the groups are summarized in Table 1.
Physiologic data for the four groups
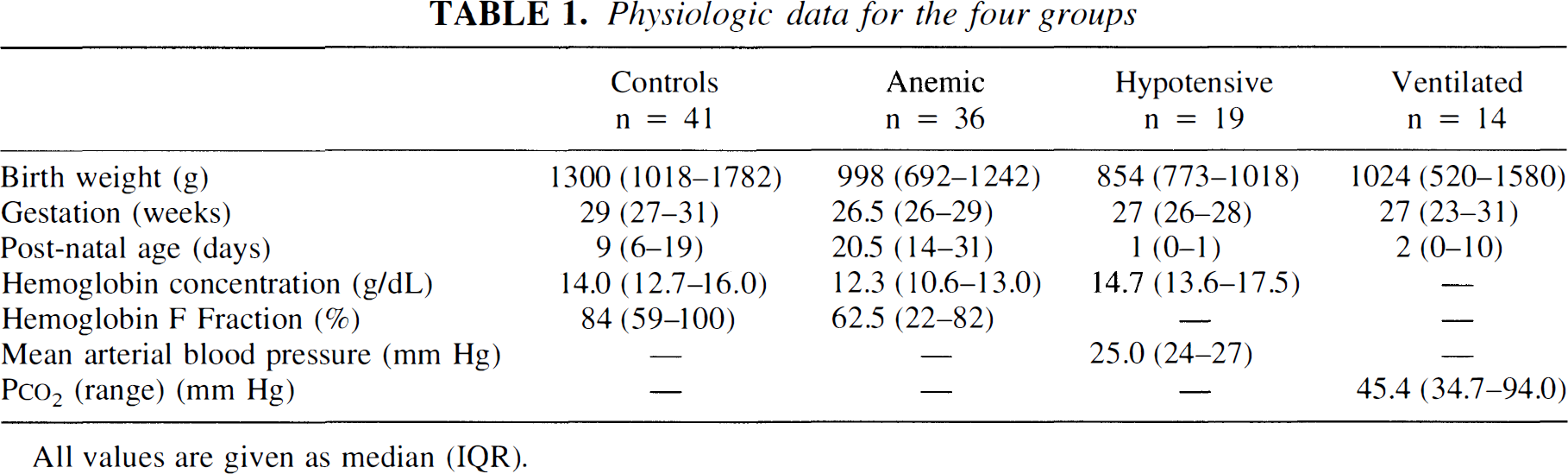
All values are given as median (IQR).
The mean ± SD cerebral FOE in control neonates was 0.292 ± 0.06. Before treatment, the cerebral FOE in anemic neonates was 0.310 ± 0.08 and in hypotensive neonates it was 0.278 ± 0.06; these measurements were not significantly different from those of the control neonates (P = 0.26 and P = 0.41, respectively; Fig. 1). After blood transfusions (20 mL/kg of packed cells) in the anemic group, there was a significant fall in cerebral FOE to 0.274 ± 0.05 (P = 0.02). The mean effect of a transfusion on cerebral FOE (95% confidence interval) was −0.035 (−0.063 to −0.007).
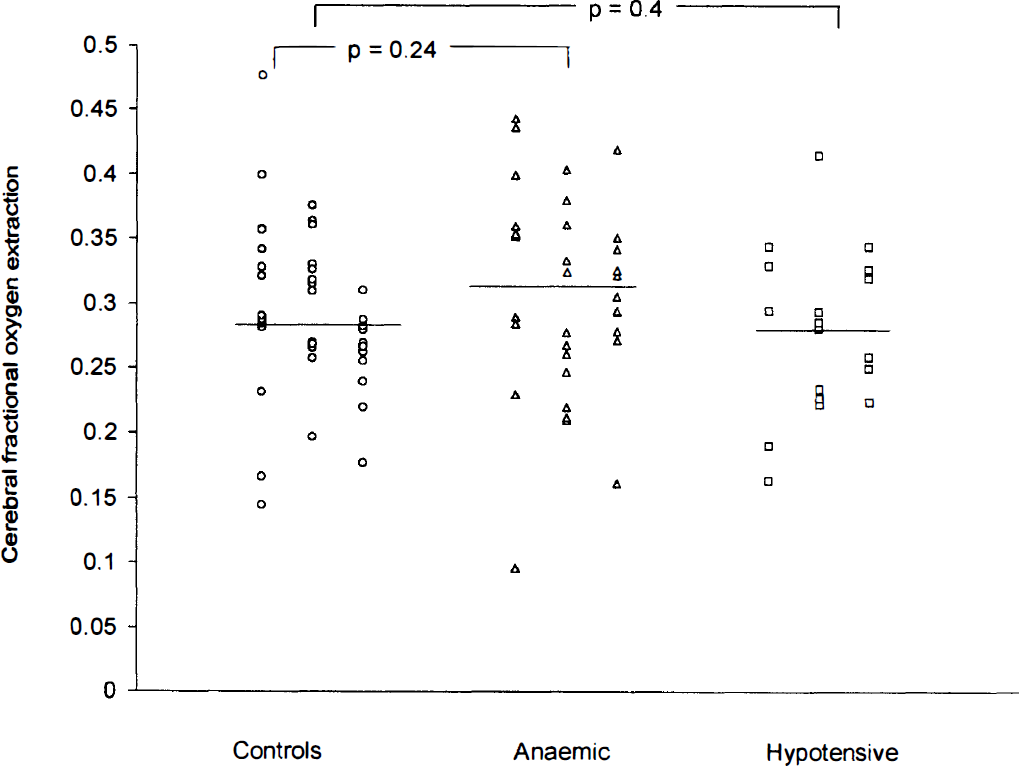
Results of fractional oxygen extraction measurements in all three groups. The bar represents the mean value for each group.
In the anemic group, the median Hb (interquartile range) was 12.3 (10.6 to 13.0) g/dL, which was significantly lower than the control group value [14.0 (12.7 to 16.0) g/dL] (P < 0.0001). After transfusion, median (interquartile range) Hb increased significantly to 15.2 (14.1 to 16.6) g/dL (P < 0.0001). The mean change in Hb (95% confidence interval) was 3.6 (3.0 to 4.2) g/dL. In the anemic group, 18 neonates had symptoms that were attributed to anemia and 36 had no symptoms that were ascribed to anemia but were transfused because of a low Hb level. The mean ± SD cerebral FOE was similar in those with symptoms (0.321 ± 0.05) as in those without symptoms (0.303 ± 0.09) (P = 0.5).
For neonates considered hypotensive, the mean arterial blood pressure was 24.9 ± 2.6 mm Hg. Treatment consisted of 20 mL/kg human albumin solution alone in one baby, a dopamine infusion alone (median dose 5 μg/kg/min, range 5 to 15 μg/kg/min) in nine neonates, 20 mL/kg human albumin solution and a dopamine infusion (median dose 5 μg/kg/min, range 5 to 10 μg/kg/min) in eight neonates, and a blood cell transfusion (20 mL/kg packed cells) in only one baby. Cerebral SvO2 values of neonates considered to be hypotensive were measured again when their blood pressures were in the normal range at a median (range) of 3 (2 to 24) hours after the first measurement. All the neonates in this group were sedated with 25 μg/kg/h of morphine, but none had been given muscle relaxants. In the hypotensive group, there was no relationship between mean arterial blood pressure and cerebral FOE (n = 19, r = 0.34, P = 0.15; Fig. 2). The mean arterial blood pressure after treatment was 34.7 ± 4.2 mm Hg [mean change (95% confidence interval) 9.8 (0.4 to 19.2) mm Hg]. After hypotension was corrected, the mean ± SD cerebral FOE was 0.296 ± 0.06 and was not significantly different from the pretreatment value (P = 0.32). The mean change (95% confidence interval) in cerebral FOE was 0.021 (−0.023 to 0.066).
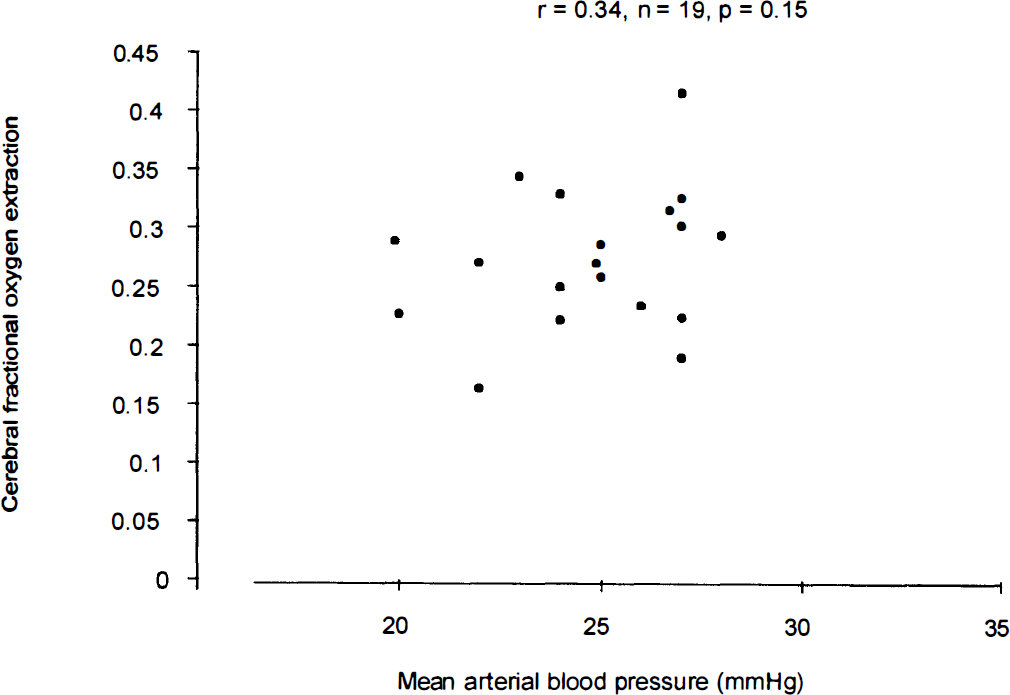
Relationship between cerebral fractional oxygen extraction and mean arterial blood pressure in hypotensive group.
There was a weak negative correlation between cerebral FOE and the Hb concentration (n = 91, r = −0.26, P = 0.02; Fig. 3) in all the neonates studied, and when only the control and anemic neonates were considered (n = 77, r = −0.24, P = 0.04). There was a similar weak correlation between arterial oxygen content and cerebral FOE (n = 91, r = −0.20, P = 0.05). There was no significant correlation between cerebral FOE and HbF (n = 56, r = 0.24, P = 0.08; Fig. 4).
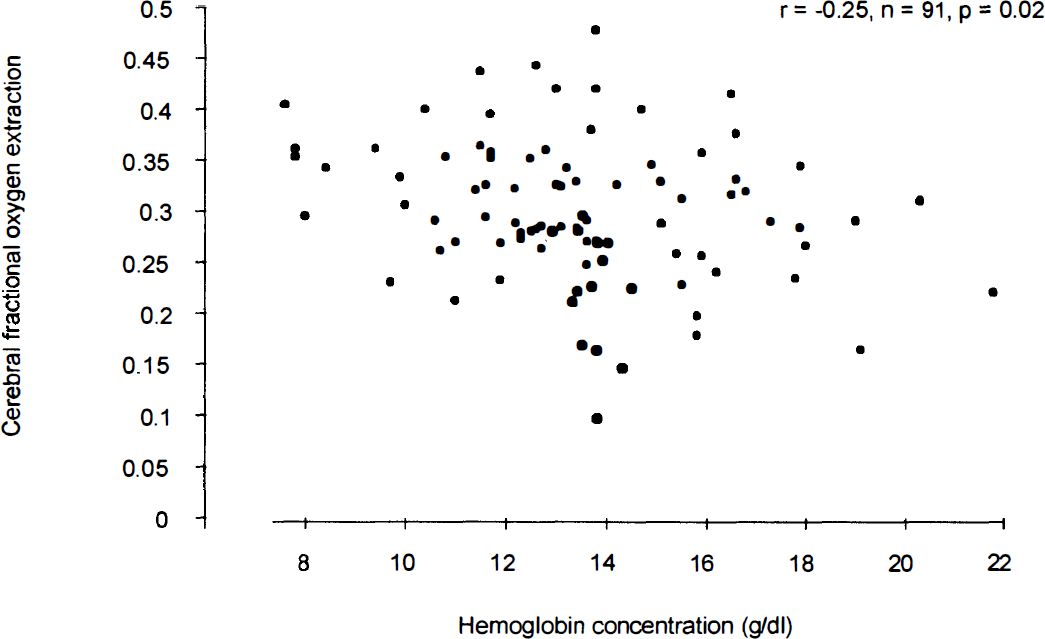
Relationship between cerebral fractional oxygen extraction and hemoglobin concentration.
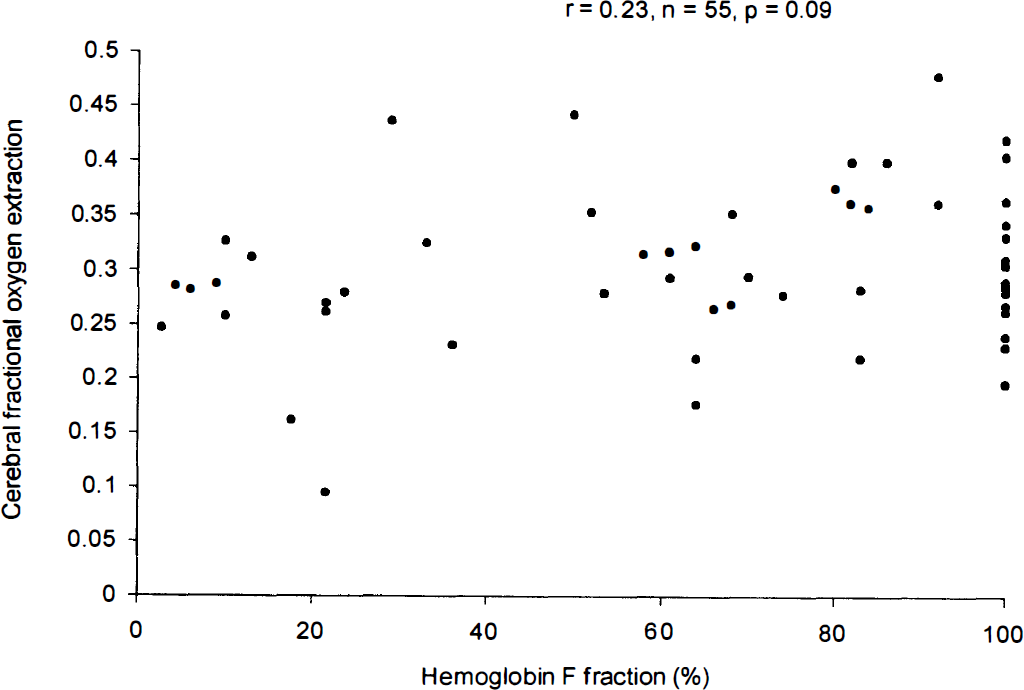
Relationship between cerebral fractional oxygen extraction and hemoglobin F fraction (HbF).
In considering all the neonates, there was no significant relationship between cerebral FOE and gestational age (n = 96, r = −0.01, P = 0.95), postnatal age (n = 96, r = 0.1, P = 0.4), birth weight (n = 96, r = −0.08, P = 0.45), or current weight (n = 96, r = −0.06, P = 0.67).
Fourteen ventilated neonates had a change in PaCO2 between serial measurements of venous saturation. Three of these neonates had also been included in Group 3 and three had been included in Group 2, but they were not hypotensive or anemic at the time of these measurements. In nine neonates, the PaCO2 was decreased, and in five neonates, the PaCO2 was increased. In most of these cases, this involved changing the ventilator rate. The second measurement was made after a median (range) of 1 (0.3 to 6) hours. There was a significant correlation between cerebral FOE and PaCO2 within individuals (n = 14, r = 0.63, P = 0.01) (see Fig. 5). With changes in PaCO2, the mean cerebral FOE change (95% confidence interval) for an individual was 10.8 (3.5 to 29.0) %/kPa. There was no relationship between cerebral FOE and PaCO2 (n = 14, r = 0, P = 1) for the neonates as a group.
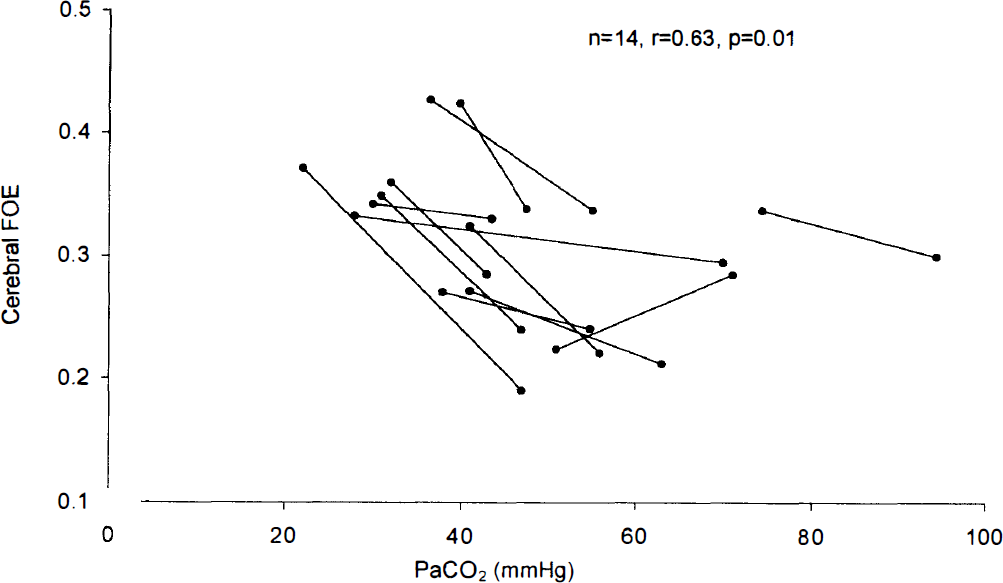
Relationship between cerebral fractional oxygen extraction and PaCO2 within individuals. Measurements within individuals are linked by a line. The mean cerebral FOE change for an individual was 10.8%/kPa.
Outcome
Of the neonates in the hypotensive group (n = 19), 10 survived without evidence of significant PVH or cystic periventricular leukomalacia (PVL). One survived with significant PVH, and eight neonates died. Of the neonates in the anemic and control groups (n = 77), 63 survived without evidence of significant PVH or cystic PVL. Two survived with significant PVH, three had cystic PVL, and nine died. There was no difference in the mean cerebral FOE in the 73 normal surviving neonates (0.298 ± 0.06) and the neonates who died (0.277 ± 0.08) (P = 0.27). Nor was there a difference in the mean cerebral FOE in the six neonates with significant PVH or PVL (0.320 ± 0.11) when compared with the normal neonates.
Variability and repeatability of venous saturation
The mean ± SD cerebral SvO2 for the control population was 68.4 ± 6.2%. The mean difference between the first and second measurements (mean of five occlusions) of cerebral SvO2 was 0.8%, and the intrasubject coefficient of repeatability (± 2 SD differences) was ±9.2%; that is, 95% of repeated measurements of cerebral venous saturation after repositioning of monitoring optodes were within 9.2% of the first measurement. Expressed in terms of the cerebral FOE, the mean difference between the first and second measurements (mean of five occlusions) was 0.005, and the intrasubject coefficient of repeatability was ±0.102.
DISCUSSION
This study has shown a negative correlation between cerebral FOE and Hb and a link between cerebral FOE and PaCO2. There was no relationship between cerebral FOE and HbF or between cerebral FOE and blood pressure over the ranges studied. Cerebral FOE before transfusion for moderate anemia was similar to control values, but it fell significantly after blood transfusions were given.
This study aimed to examine the relationship between cerebral FOE and other physiological variables, but the system is complex and affected by multiple factors, making interpretation of the results difficult. Cerebral FOE varied by a factor of ∼3 in the control group, but this is consistent with the known variability of CBF in “normal” preterm neonates (Skov et al., 1991). Measurements of cerebral FOE in this work have not previously been reported in the preterm human, and the ability to make noninvasive measurements in clinical situations has produced data that have given some insight into the pathophysiological changes that occur in this complex system.
The present study has shown that cerebral FOE correlated negatively with the Hb concentration. This observation is in line with a previous study, using the same technique, which showed a negative correlation between the arterial oxygen content and cerebral FOE in children about to undergo cardiopulmonary bypass operations (Wardle et al., 1998b). This contrasts, however, with observations from animals and adult humans, which have shown little change, if any, in cerebral SvO2 during anemic hypoxia (Borgstrom et al., 1975; Paulson et al., 1973), although cerebral SvO2 has been shown to fall during hypoxaemic hypoxia (Koehler et al., 1982; Paulson et al., 1973; Traystman et al., 1978). Cerebral blood flow studies in adults and term and preterm neonates have all shown a negative correlation between Hb and CBF (Brown et al., 1995b; Ramaekers et al., 1988; Younkin et al., 1981), a relationship that seems to be related to arterial oxygen content rather than blood viscosity (Brown and Marshall, 1985; Ulatowski et al., 1996). A reduction in CBF velocity in the internal carotid artery has also been observed after blood transfusion in preterm neonates (Nelle et al., 1994). The implication of these observations is that cerebral oxygen consumption and cerebral FOE remain constant (Jones and Traystman, 1984). Cerebral FOE has not, however, previously been measured in anemic preterm human neonates. The findings of this study suggest that changes in arterial oxygen content may result in changes in cerebral FOE as well as CBF in preterm humans if the changes are over a relatively short period of time.
This study examined the relationship between changes in Hb and cerebral FOE. There are two important observations. One is the absence of a difference in cerebral FOE between anemic and nonanemic neonates. The other is the decrease in cerebral FOE after blood transfusion. The first of these findings may be explained by the fact that the difference in total Hb between neonates considered to be anemic and those who were not was relatively small: The median Hb in the neonates receiving transfusions was 12.3 g/dL compared with 14 g/dL in the control group. A larger difference may have led to greater differences in cerebral FOE.
There are several possible explanations for the changes in cerebral FOE after transfusion. One is that an increase in Hb is balanced by a fall in CBF, a notion supported by clinical and animal studies (Brown et al., 1995b; Ramaekers et al., 1988; Younkin et al., 1981). In our study, the mean increase in Hb after transfusion was 3.6 g/dL. Assuming that CBF decreases proportionately to accommodate for this, then one would expect a decrease in CBF (95% confidence interval) of ∼29 (24 to 34) %. However, the mean change in cerebral FOE (95% confidence interval) was only 11.3 (2.3 to 20) %. This is a relatively small decrease in cerebral FOE, and if one assumes that cerebral oxygen consumption remains constant, there would need to be an associated increase in CBF. An alternative possible explanation is that the cerebral metabolic rate for oxygen falls during anemia. This explanation seems less likely as it implies that cerebral growth and function are adversely affected during anemia. A third methodological explanation is that the unpaired test used to compare the two groups before transfusion did not have the same power to detect a difference as the paired test used to detect the change with transfusion.
This study has shown no relationship between cerebral FOE and HbF. This further supports the notion that oxygen availability to the brain of these preterm human subjects was maintained by changes in CBF. Two other studies also support this view. The HbF has been shown to correlate positively with CBF velocity (Ramaekers et al., 1988) and CBF (Lipp-Zwahlen et al., 1989) in preterm neonates. Contrasting findings were, however, found in a study of sheep, where increased cerebral FOE was demonstrated when fetal Hb was replaced by adult Hb (Rosenberg et al., 1986). This was achieved by exchange transfusion, which resulted in a fall in arterial oxygen content but no increase in CBF (Rosenberg et al., 1986). There are two probable explanations for the difference between these studies by Rosenberg et al. and our own. The first lies in the differences between sheep and human Hb. In sheep, the difference between the oxygen affinity of adult and and fetal Hb is larger than in humans. The P50 (PO2 value at which 50% of Hb is oxygenated) of adult sheep (HbB) is 5.5 kPa and that of newborn sheep 3.0 kPa, whereas in humans, the relative values are 3.6 kPa for adult Hb and 2.6 kPa for HbF (Battaglia et al., 1970). Thus, changes in the proportion of adult to fetal Hb produce a much larger change in oxygen-Hb affinity in sheep than in humans. Because Rosenberg et al. exchange transfused lambs with adult sheep Hb, there was a rapid decrease in arterial oxygen content as well as a change in Hb oxygen affinity (Rosenberg et al., 1986). In the present study, the arterial oxygen content would be expected to have been approximately constant despite different HbF values, and any small differences in cerebral Hb oxygen affinity leading to changes in cerebral oxygen delivery are likely to have been counterbalanced by changes in CBF.
In this study, the relationship between PaCO2 and cerebral FOE was analyzed using two approaches. First, the effect of a change of PaCO2 on cerebral FOE was considered for individual neonates. This showed a negative correlation between PaCO2 and cerebral FOE, similar to that observed in adult and fetal sheep (Jones et al., 1982). The second approach, which examined the relationship between PaCO2 and cerebral FOE within the whole group, showed no relationship between cerebral FOE and PaCO2. The difference between these two results leads to a view that it is probably the time scale of changes in PaCO2 that affects cerebral FOE. This would be analogous to the observation that when CBF is altered by acute changes in PaCO2, it returns to the normal range as the cerebrospinal fluid pH returns to normal (Lassen, 1974), and neonates with chronically high or low values of PaCO2 do not have abnormal levels of CBF. Changes in cerebral FOE would be expected to be associated with changes in CBF if the cerebral metabolic rate for oxygen remained constant, and the effect of changing PaCO2 on CBF is well established (∼30%/kPa)(Leahy et al., 1980; Greisen and Trojaborg, 1987; Reuter and Disney, 1986; Hansen et al., 1986; Rosenberg, 1988; Pryds et al., 1990). Our data showed that the average change in cerebral FOE with changes in PaCO2 was ∼11%. This is less than the CBF change, but there is probably overlap due to the wide confidence intervals.
Cerebral FOE was not increased in neonates who were considered hypotensive when compared with control subjects, and there was no change in cerebral FOE when the blood pressure returned to the normal range after treatment. There was also no relationship between mean arterial blood pressure and cerebral FOE. Cerebral FOE has not previously been studied in hypotensive preterm neonates, but studies of CBF over the first 3 days of life found it increased independently of mean arterial blood pressure (Meek et al., 1998; Tyszczuk et al., 1998). Unlike in animals (Hernandez et al., 1980; Laptook et al., 1983; Papile et al., 1985; Purves and James, 1969), the limits of autoregulation in the human newborn have not been established. It is likely that for the neonates in this study, the mean arterial blood pressure was consistently above the limit at which oxygen delivery to the brain becomes compromised, despite mean arterial blood pressures below 30 mm Hg. Another possibility is that both CBF and cerebral oxygen consumption were low in these neonates so that FOE remained unchanged. The question of when it is appropriate to treat systemic hypotension in these neonates and how aggressively to treat it remains important and unanswered. Monitoring cerebral FOE during hypotension may yield information of clinical relevance by providing a guide for the treatment for hypotension.
There appeared to be no relationship between cerebral FOE and either gestational age or postnatal age in this study, although consecutive measurements in the same subjects are needed to confirm this suggestion. Cerebral oxygen consumption (mL/100 g) is known to increase in relation to gestational age (Yoxall and Weindling, 1997), and CBF has also been shown to increase with postnatal age (Meek et al., 1998). These effects are likely to be linked to brain maturation and growth and the normal adaptive responses of the cerebral circulation to postnatal life. The results of this study suggest that cerebral FOE generally remains constant and CBF and cerebral oxygen consumption probably increase in parallel with increasing postnatal and gestational age.
Two other methods have been described to measure cerebral SvO2 using NIRS (Skov et al., 1993; Wolf et al., 1997) in preterm neonates. One requires the subjects to be mechanically ventilated (Wolf et al., 1997), and the other requires a head-down tilt of the patient (Skov et al., 1993). In the former, measurements were possible in only 10 of 15 patients studied (Wolf et al., 1997). This technique produced some very low values of cerebral FOE, but this is likely to have been related to the low SaO2 values that were recorded using an NIRS technique that has not been validated, rather than by pulse oximetry (Wolf et al., 1997). Results were obtained in only 9 of 22 patients studied using the tilt technique (Skov et al., 1993). With the partial jugular venous occlusion technique used in this work, measurements were possible in 99 of 102 neonates studied, including neonates who were awake and not ventilated and sick unstable neonates. The mean ± SD cerebral SvO2 was 68.4 ± 6.2% in the control group in this study. This was similar to measurements using the same technique in healthy term newborns (Buchwald and Greisen, 1999). These authors reported a mean SvO2 of 64.12 ± 4.6% in 11 neonates. These data compared with 53.4 ± 15.4% using the tilt technique and 73.0 ± 8.9% using the ventilation technique. The differences in reported SvO2 may reflect the differences between the populations of neonates studied or differences in the techniques, but only NIRS with partial jugular venous occlusion technique has been validated by comparison with invasive measurements (Yoxall et al., 1995).
Measurements of cerebral venous saturation and/or FOE have not previously been reported in preterm humans in relation to anemia and blood transfusions. These results demonstrate that the response by the brain to arterial oxygen content changes is complex and may occur at both a cellular and a vascular level. Changes in cerebral FOE can be a consequence of either altered oxygen delivery or oxygen consumption. Further study using this technique should therefore concentrate on combining measurements of CBF with venous saturation. Studying the peripheral circulation during hypotension (Wardle et al., 1999) and the cerebral circulation simultaneously may generate useful information that will allow interventions to be made earlier than was previously the case. Further studies investigating changes in cerebral and peripheral oxygen delivery and consumption during neonatal intensive care will be needed to clarify if, how, and when systemic hypotension should be treated. These measurements may also allow us to develop more rational interventions for optimizing cerebral oxygenation.
