Abstract
The role of nitric oxide (NO) in the mechanism of hemodilution-induced cerebral hyperemia is unclear. Based on findings in hypoxemia, the authors hypothesize that NO of neuronal origin contributes to an increase in velocity of erythrocytes in the cerebral microcirculation during anemia produced by isovolemic hemodilution. The change in erythrocyte velocity in cerebrocortical capillaries was assessed by intravital fluorescence video microscopy. A closed cranial window was implanted over the frontoparietal cortex of barbiturate-anesthetized, ventilated adult rats. Erythrocytes were labeled in vitro with fluorescein isothiocyanate and infused intravenously, and their velocity in subsurface capillaries was measured by frame-to-frame image tracking. Arterial blood was withdrawn in increments of 2 mL and replaced by serum albumin; arterial blood pressure was maintained at control level with an infusion of methoxamine. Erythrocyte velocity increased progressively, reaching 215% of baseline, as arterial hematocrit was reduced from 45% to 17%. Pretreatment of a separate group of rats with 7-nitroindazole (20 mg/kg intraperitoneally), a relatively selective inhibitor of neuronal NO synthase, abolished the increase in velocity at hematocrits greater than 20%, but the maximum velocity attained at the lowest hematocrit was similar to that in the control group. The results suggest that NO from neuronal source may contribute to the increase in capillary erythrocyte flow during moderate isovolemic hemodilution.
Keywords
Substantial evidence supports that nitric oxide (NO) produced by the neuronal isoform of NO synthase (NOS) plays an important role in cerebral vasodilation to various physiologic, pathologic, and pharmacologic stimuli, including hypercapnia (Wang et al., 1995; Ma et al., 1996b; Okamoto et al., 1997a), neuronal activation (Ayata et al., 1996; Cholet et al., 1997; Ma et al., 1996a), cortical spreading depression (Colonna et al., 1997), excitatory amino acids (Montécot et al., 1997; Yang and Chang, 1998), and volatile anesthetics (Okamoto et al., 1997b). In contrast, the involvement of NO in the hypoxic response of CBF has been controversial (Buchanan and Phillis, 1993; Pelligrino et al., 1993; Reid et al., 1995). We recently found (Hudetz et al., 1998) that acute hypoxemia-induced increases in erythrocyte flow velocity in the cerebral capillary bed were abolished by 7-nitroindazole (7-NI), a relatively specific inhibitor of neuronal NOS. The significance of this finding was that it suggests a role for neuronal NO in the local regulation capillary flow. We wondered if a similar dependence of capillary erythrocyte flow on neuronal NO was present in anemia produced by isovolemic hemodilution.
The effect of hemodilution on capillary perfusion in the brain has not been directly examined. Previously, Rosenblum (1971) studied the velocity of erythrocytes in pial vessels and the velocity of plasma through intracortical vessels of the mouse and reported significant increases in both parameters during isovolemic anemia. Likewise, Lin et al. (1996) demonstrated a reduction of erythrocyte and plasma transit times by isovolemic hemodilution in the partially ischemic rat brain. Todd et al. (1992) measured the effect of isovolemic hemodilution on intracortical erythrocyte and plasma volumes by radioactive labeling and predicted a significant decrease in microvascular hematocrit. None of these studies were based on direct observation of erythrocyte flow in cerebral capillaries.
Recently, Todd et al. (1997) examined the potential role of NO in cerebral vasodilation to anemia and hypoxemia in a rabbit model. They conclude that NO played some role in cerebral vasodilation to hypoxemia but not to anemia. Despite the importance of this study, there are several reasons to further investigate this issue. First, the effect of NOS inhibition on cerebral capillary erythrocyte flow in anemia has not been determined. Studies of the microcirculation demonstrate that during hemodilution, capillary erythrocyte flow may be altered differently from regional blood flow because of changes in perfusion heterogeneity and plasma skimming (Rosenblum, 1987; Tyml, 1991; Pries et al., 1992). Second, the effect of the neuronal NOS inhibitor, 7-NI, on the hemodilution response has not been investigated. Whereas endothelial NO could mediate shear-dependent vasodilation, neuronal NO may play a principal role in the response to hypoxia (Millar, 1995). Third, the experiments by Todd et al (1997) examined the CBF response to severe hemodilution (15% hematocrit) only. We thought that the use of moderate, graded anemic stimulus could reveal the finer adjustments of erythrocyte flow in the capillary network.
The objective of this study was to determine if the neuronal NOS inhibitor, 7-NI altered the cerebral microcirculatory response to graded, isovolemic hemodilution. We hypothesized that 7-NI would attenuate the hemodilution-induced increase in erythrocyte velocity in cerebrocortical capillaries, which would suggest a role for active, neuronal NO-dependent mechanisms in the hyperemic response. To assess the hemodilution-induced changes in capillary flow directly, the study was conducted by intravital microscopic technique using a rat closed cranial window preparation.
METHODS
Experimental preparation
The experiments were performed on nine male Sprague-Dawley rats of 230 to 280 g weight. The animals were anesthetized with sodium-pentobarbital (60 mg/kg intraperitoneally) and tracheotomized, and bilateral femoral arterial and venous lines were placed to measure arterial blood pressure, withdraw blood, and infuse drugs. Anesthesia was maintained with sodium pentobarbital infusion at 7 to 10 mg/h. The head was secured in a stereotaxic apparatus, and a closed cranial window was installed over the right parietal cortex using a technique previously described (Hudetz et al., 1995). The window was equipped with ports for superfusion with artificial cerebrospinal fluid at 37°C and for the measurement of intracranial pressure. The latter was stabilized at 5 to 10 mm Hg. After surgery, the animals were transferred to the intravital microscope, paralyzed with gallamine (80 mg), and artificially ventilated with a mixture of 30% O2 and 70% N2. Arterial blood pressure, end-tidal CO2, and inspired oxygen concentrations were continuously monitored (POET II, Criticare Systems). The rate and volume of ventilation were adjusted to achieve an arterial PCO2 of approximately 35 mm Hg.
Video microscopy
Erythrocytes obtained from a donor rat were labeled in vitro with fluorescein isothiocyanate and injected intravenously in tracer quantities into the circulation of the experimental animal (Hudetz et al., 1995). With the use of epifluorescence and a 40× objective lens, the passage of labeled cells through intracortical capillaries 50 to 70 μm below the pial surface could be visualized in real time and video recorded. The optical magnification was 125× with a field size of 300 × 400 μm. Although the capillary walls themselves were not visible, capillaries could be identified by the presence of single-file erythrocyte flow (implying vessel diameter of less than 5 μm) and by the tortuous and branching pathway of moving cells. The small depth of field (less than 10 μm) limited the study to capillary segments that run at right angles to the camera and are nearly parallel to the brain surface. This minimized the possibility for projection error in the measurement of flow velocity. A typical capillary segment visible in the plane of focus was 60 μm long. To minimize the exposure of the preparation to light, a 50% neutral density filter was used at all times, and continuous illumination was limited to 1-minute periods while the circulation was video recorded. A heat filter and a 455-nm high-pass filter were used to block infrared and ultraviolet irradiation of the tissue.
Experimental protocol
Graded isovolemic hemodilution was produced by the repeated withdrawal of 2 mL of blood, immediately replaced by the same volume of serum albumin (50 mg/mL in 0.7% NaCl, Sigma-Aldrich Co.). A 100-μL sample of blood was used for hematocrit determination, and 400 μL of blood was used for the measurement of blood gases after each hemodilution step. To support the arterial blood pressure, the α1-agonist methoxamine (Sigma-Aldrich Co.) was infused intravenously at a rate between 1 and 15 mg/kg/h, as in prior studies (Hudetz et al., 1998). Intravenous α1-selective adrenergic agonists have been reported to exert no direct effect on the cerebral vasculature in vivo(Sokrab and Johansson, 1989). The rate of infusion of methoxamine was adjusted after each hemodilution step as necessary to maintain MABP at control level. The infusion of methoxamine usually was initiated after the third or fourth hemodilution step.
In each animal, the microcirculation was video recorded in the control state (before hemodilution) and 5 to 7 minutes after the completion of each hemodilution step. This time frame allowed us to stabilize MABP at the control level and was short enough to preserve cardiovascular stability of the preparation for the duration of the experiment. Four animals were treated with 7-NI (ICN Biomedicals, Inc.) at 20 mg/kg intraperitoneally, 45 minutes before the control recording. Three of the control animals were treated with peanut oil, the vehicle for 7-NI, and two received no treatment.
Data analysis
Erythrocyte velocity was measured during video playback by frame-to-frame tracking of the labeled cells using an imaging system developed in our laboratory (Hudetz et al., 1993). Four capillaries typically were chosen for measurement. In each capillary, cell velocity was measured at 3- to 5-second intervals, yielding 15 measurements per capillary, and averaged over the 1-minute recording period. With control plus five hemodilution steps performed, over 3200 velocity measurements were made. Statistical analysis for between-group comparison was performed using multivariate analysis of variance with the type of treatment (7-NI or control) as the factor and the velocity at each hemodilution step as the dependent variable. The effect of hemodilution on velocity in the same treatment group was analyzed with repeated-measures analysis of variance followed by paired t-test. Linear regression was used to estimate the dependence of capillary cell density on arterial hematocrit.
RESULTS
Systemic arterial physiologic parameters are displayed in Table 1. There were no notable differences between the experimental groups except a slightly higher arterial pressure in the 7-NI-treated group (9 mm Hg, not statistically significant). However, all MABP values were within the physiologic range, and they were unchanged during the hemodilution protocol in both groups. All physiologic parameters were stable during the experiments.
Systemic arterial blood parameters in two experimental groups
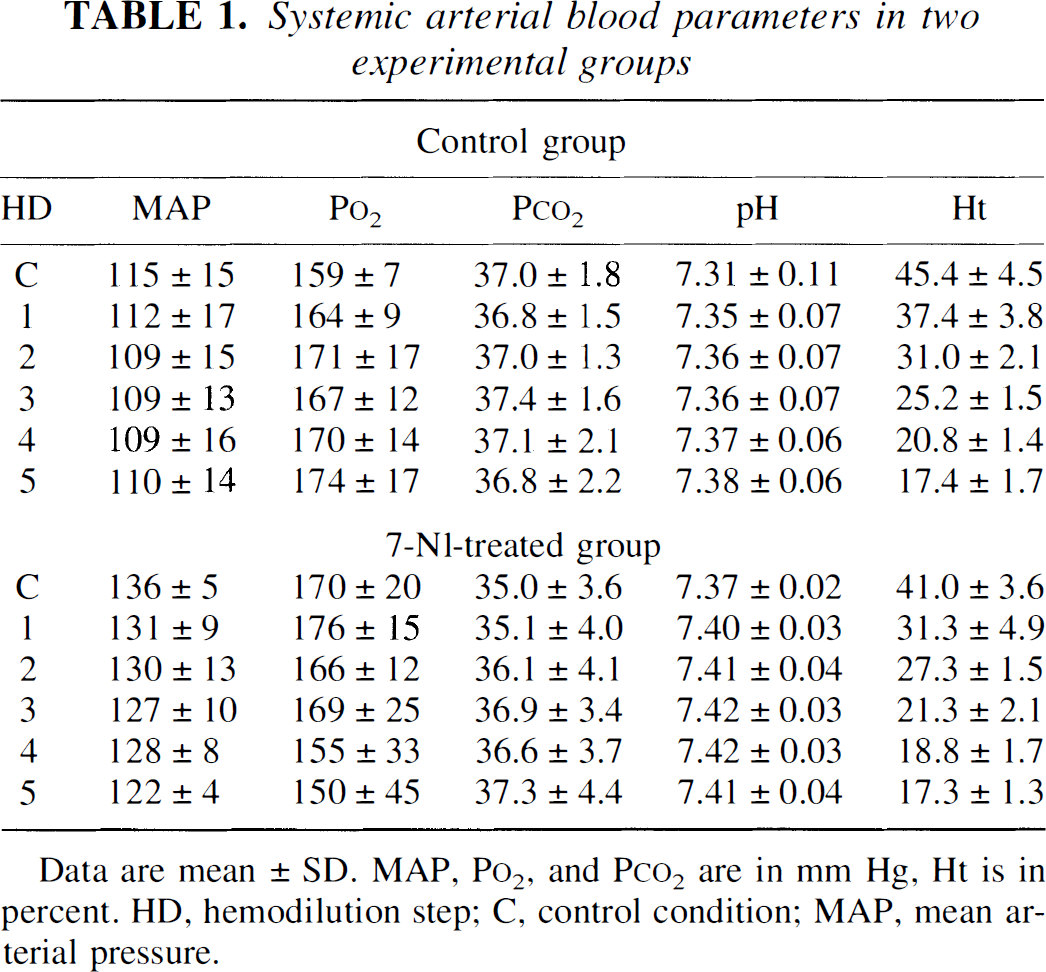
Data are mean ± SD. MAP, P
Average baseline velocity of erythrocytes in the control group was 0.73 ± 0.28 mm/s (mean ± SD). Graded isovolemic hemodilution produced a consistent increase in erythrocyte velocity in every measured capillary. In the control group, a significant increase in velocity was seen with each hemodilution step. The mean velocity attained at the fifth hemodilution step was 1.57 ± 0.40 mm/s. The velocity-hematocrit relationship essentially was linear in this group.
In the 7-NI-treated group, baseline velocity was 0.50 ± 0.23 mm/s, significantly lower than in the controls. This difference was consistent with the expected effect of 7-NI on resting CBF. During hemodilution, erythrocyte velocity attained a maximum of 1.51 ± 0.46 mm/s, which was not different from that in the control group. However, the rise in velocity was considerably attenuated at hemodilution steps 1 through 3 (Fig. 1). Furthermore, in the 7-NI-treated animals, the increases in velocity at 31% and 27% hematocrit were small and not statistically significant at 31% hematocrit.
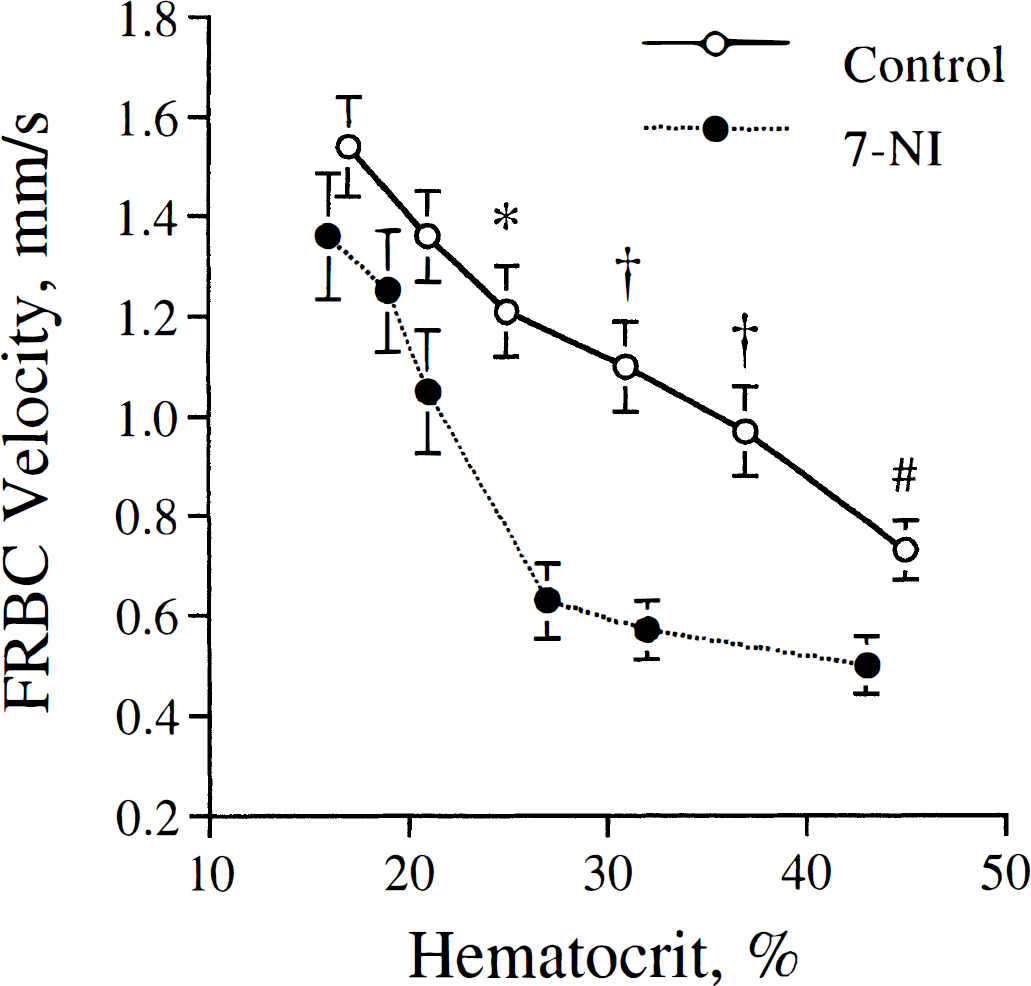
Effect of isovolemic hemodilution on erythrocyte velocity in cerebrocortical capillaries in two groups of animals: control, and treated with 7-nitroindazole (7-NI), an inhibitor of neuronal nitric oxide synthase. Velocity measurements were taken in the order from right to left along the curves, as arterial hematocrit was reduced by stepwise replacement of arterial blood with serum albumin. MABP was maintained constant during the protocol. *P < 0.05, †P < 0.0001, #P < 0.005 versus data at corresponding hemodilution step of the other group.
To estimate the change in capillary hematocrit during hemodilution, the number of labeled erythrocytes passing through each capillary was counted, and lineal cell density was calculated as the cell count per cell velocity. We found that during hemodilution, lineal cell density essentially was unchanged (r = 0.19) in both the control and 7-NI-treated groups. The supply rate of erythrocytes (number of cells traversing the capillary per time) changed in parallel with erythrocyte velocity in both groups.
DISCUSSION
The objective of this study was to determine if the neuronal NOS inhibitor, 7-NI, influenced the response of erythrocyte flow velocity in cerebrocortical capillaries to graded isovolemic hemodilution. We found that 7-NI impeded the hyperemic response to moderate but not to severe hemodilution. These results suggest that NO produced by neuronal (type I) NOS played an important role in the erythrocyte flow response to the moderate anemic stimulus, whereas passive or NO-independent mechanisms played the dominant role under the severe anemic condition.
The observed rise in erythrocyte flow velocity was consistent with the known hyperemic response of CBF to hemodilution. Traditionally, this response has been assigned to a decrease in blood viscosity or to active cerebral vasodilation. Investigators who studied the effect of hemodilution on pial artery diameter found no vasodilation (Rosenblum, 1971; Hurn et al., 1993) or vasoconstriction (Muizelaar et al., 1983; Hudak et al., 1989), supporting the principal role of rheologic factors. However, Todd et al. (1992) report an increase in microvascular blood volume, suggesting the presence of intracerebral vasodilation during isovolemic hemodilution. Subsequently, Waschke et al. (1994) decreased blood viscosity by exchange transfusion with a Newtonian oxygen carrier and saw no change in CBF. These findings suggest that active cerebrovascular adjustment as a function of blood oxygen saturation took place during hemodilution.
The current results are consistent with the role of active vasodilation in the cerebral hyperemic response to hemodilution. However, alternative mechanisms, including blood cell-endothelial interactions in the microcirculation, should be considered. Because arterial PO2 did not fall and capillary erythrocyte flow increased, tissue hypoxia was unlikely and could not play a role in the hemodilution response. This contention is supported by Bauer et al. (1996), who showed that EEG and high-energy phosphates in the brain were preserved at arterial hematocrits as low as 6%. Direct measurements of intracortical PO2 during hemodilution would be made to confirm the absence of tissue hypoxia during isovolemic hemodilution.
Recently, Todd et al. (1997) found that NOS inhibition did not influence the hemodilution-induced hyperemia at an arterial hematocrit of 15%. Our results are consistent with this finding and with the role of NO-independent mechanisms at low hematocrit level. However, with moderate reductions of arterial hematocrit, NO appears to play a significant role, which does not conflict with the results of Todd et al. (1997). Another difference between the study of Todd et al. and ours is that we used 7-NI instead of the nonspecific NOS inhibitor, L-nitro-arginine methyl ester. Although objections to the specificity of 7-NI have been raised, the bulk of investigations suggest otherwise (Handy and Moore, 1998). 7-Nitroindazole has been shown to considerably inhibit brain NOS catalytic activity and cerebral vasodilation to several physiologic stimuli (Wang et al., 1995; Cholet et al., 1997; Colonna et al., 1997; Yang and Chang, 1998). If the minor increase in MABP observed after 7-NI treatment were real, it could be caused by either peripheral or central effects.
In this study, we used a relatively low dose of 7-NI (20 mg/kg) to minimize the chance for nonspecific effects. This dose was chosen based on the study by Connop et al. (1994), who showed that inhibition of NOS in the substantia nigra and the cerebellum was maximal (about 90%) with 20 mg/kg 7-NI. If the degree of NOS inhibition were less, the main conclusion of our study would be unaffected, since the role of NO in the hemodilution response would be, at worst, underestimated.
As noted in Results, resting erythrocyte velocity was lower in the 7-NI-treated group, consistent with the removal of baseline NO and a decrease in resting CBF (Kelly et al., 1995; Wang et al., 1995; Cholet et al., 1997; Okamoto et al., 1997a). It may have been that a lower resting velocity contributed to the different character of the velocity response after treatment with 7-NI. However, this could not be the sole reason for the observed difference because (1) erythrocyte velocities were not different at the lowest hematocrits between the two groups, and (2) the statistically significant difference at moderate hemodilution prevailed after normalizing to or subtracting the respective baseline velocities in each group. We observed that baseline erythrocyte velocity after 7-NI treatment was lower than measured in our previous study (Hudetz et al., 1998). We believe that this difference resulted from the greater care taken to select true capillaries that exhibit the lowest erythrocyte flow rates. Another source of discrepancy could have been the independent group design of the current study.
Whether the impediment of hemodilution response after 7-NI was caused by specific effect on capillary flow or by a more general effect on regional CBF remains to be investigated. We found in a recent study (Hudetz et al., 1998) that the effect of 7-NI on capillary erythrocyte flow in acute hypoxemia was much more dramatic than the corresponding effect on regional erythrocyte flow. These findings suggest that 7-NI interfered with a local mechanism of flow regulation specific to the terminal microvascular bed. It is possible that during hemodilution, capillary erythrocyte flow did not strictly follow the change in CBF. Hemodilution has been implied to influence the microcirculation by increasing erythrocyte flow selectively in poorly perfused capillaries (Pries et al., 1992; Tyml, 1991). Rosenblum (1987) altered hematocrit and plasma viscosity by various means and found that erythrocytes and plasma did not have the same path lengths and influenced the velocities of erythrocyte and plasma in different ways. In this study, we found that erythrocyte density in capillaries did not change with hemodilution after 7-NI, suggesting that a postulated change in microhematocrit was not responsible for the impediment of the hemodilution response. Although lineal cell density changed frequently in individual capillaries, no systematic trend with arterial hematocrit was apparent in either experimental group.
The site of NO's action on capillary erythrocyte velocity is unknown. Capillaries generally are regarded unable to actively change their diameter, although a passive change in diameter with intraluminal pressure is plausible. Detection of a change in capillary diameter was beyond the capability of our imaging technique. As a more likely scenario, terminal feed arterioles or precapillary pericytes may selectively regulate capillary flow; pericytes have been shown to relax in response to exogenous NO (Haefliger et al., 1994). Alternatively, NO may influence capillary flow by modulating leukocyte adhesion (Mitchell and Tyml, 1996).
Whether NO production increased during hemodilution is unclear. In the absence of tissue hypoxia, the signal leading to neuronal NOS activation is elusive. Enhanced production of new NOS protein can be discounted within the time frame of our experiment. Shear-induced NO production in the endothelium by type I NOS (Rosenblum and Murata, 1996) may play a role but needs confirmation. Alternatively, NO may support smooth muscle relaxation without an increase in NOS activity, by a permissive effect (Okamoto et al, 1997a).
In summary, the current study demonstrates that erythrocyte velocity in cerebrocortical capillaries increases as the arterial hematocrit is reduced and that 7-NI inhibits this response at hematocrits as low as 20%. The results suggest that NO from neuronal NOS may play an important role in the regulation of capillary erythrocyte flow during moderate isovolemic hemodilution.
