Abstract
The neurotrophins and the tyrosine kinase (Trk) B receptor may play a protective role in the pathophysiology of cerebral ischemia. In this study, the authors investigated whether reducing endogenous expression of TrkB-binding neurotrophins modifies the susceptibility to ischemic injury after 1-hour middle cerebral artery occlusion followed by 23 hours of reperfusion in a filament middle cerebral artery occlusion model. Mice lacking both alleles for neurotrophin-4 (nt4−/−) or deficient in a single allele for brain-derived neurotrophic factor (bdnf+/−) exhibited larger cerebral infarcts compared to wild-type inbred 129/SVjae mice (68% and 91%, respectively, compared to controls). Moreover, lesions were larger (21%) in nt4−/− mice after permanent middle cerebral artery occlusion. Hence, expression of both NT4 and BDNF, and by inference the TrkB receptor, confers resistance to ischemic injury.
Neurotrophins belong to a family of growth factors that are required for the survival and maintenance of neurons in the peripheral and central nervous system. Members of this family include nerve growth factor (Klein et al., 1991a), brain-derived neurotrophic factor (BDNF) (Leibrock et al., 1989), neurotrophin-3 (NT3) (Ernfors et al., 1990), and neurotrophin-4/5 (NT4) (Ip et al., 1992). The neurotrophins specifically bind to both a receptor tyrosine kinase (TrkA, TrkB, or TrkC) (Klein, 1991a,b; Lamballe, 1991; Soppet, 1991; Squinto et al., 1991) and a common low-affinity p75 receptor (Hallböök et al., 1991; Lee et al., 1994).
Neurotrophins may play a protective role in several neurodegenerative diseases, brain trauma, epilepsy, and cerebral ischemia (Merlio et al., 1993; Lindvall et al., 1994; Kokaia et al., 1995, 1996). For example, the exogenous administration of BDNF or NT4 which both bind to TrkB reduced brain damage in several animal models of cerebral ischemia (Beck et al., 1994; Tsukahara et al., 1994; Chan et al., 1996; Alexi et al., 1997; Cheng et al., 1997; Schäbitz et al., 1997). The precise role of endogenous TrkB receptor agonists in the development of ischemic brain injury, however, remains poorly understood. In this study we determined if a deficiency in endogenous BDNF or NT4 renders the brain more susceptible to ischemic injury. We compared lesion volume and neurologic outcome after middle cerebral artery occlusion (MCAO) in mice lacking either one bdnf allele or both alleles of nt4.
MATERIALS AND METHODS
Knockout mice
The generation of neurotrophin-4 knockout (nt4−/−) has been described before (Liu et al., 1995). NT4−/− mice are viable and fertile and can be crossed to produce offspring (Liu et al., 1995). Brain-derived neurotrophic factor knockout mice (bdnf−/−) die within 2 to 3 weeks after birth, but heterozygote (bdnf+/–) mice are healthy and fertile and were generated and genotyped as described (Ernfors et al., 1994). All nt4−/−, bdnf+/–, and wild-type control mice carried a pure inbred 129/ SVjae background. NT4−/− and bdnf+/– mice have normal appearing brain vasculature and morphology.
Northern blot
Total RNA was extracted from the cortex, subjected to gel electrophoresis, and blotted to the membrane in a standard procedure (Ernfors et al., 1994). Membranes were subsequently hybridized with p32-labeled radioactive nt4 and bdnf probes and exposed to x-ray film (Kodak, Rochester, NY, U.S.A.) for autoradiography.
Model of cerebral ischemia
All animal experiments were performed in accordance with the National Institutes of Health (NIH) guide for the care and use of laboratory animals (NIH publications No. 80-23) and institutional guidelines (Massachusetts General Hospital and Massachusetts Institute of Technology). Gender-matched mice (age, 1.5 to 4 months; body weight, 18 to 26 g) were first anesthetized with the use of halothane (1.5% for induction and 0.8% for maintenance) in 70% nitrous oxide (N2O) and 30% oxygen (O2) administered with a face mask. Focal cerebral ischemia was produced as described (Huang et al., 1994; Endres et al., 1997, 1998b). Briefly, the left middle cerebral artery was occluded with an 8-0 silicone-coated monofilament. For transient ischemia, the filament was withdrawn after 1 hour of ischemia. For permanent ischemia, the filament was kept in place until the animal was killed at 24 hours. In all animals, regional cerebral blood flow was measured by means of a flexible probe and laser-Doppler monitoring (Perimed, Smithtown, NY, U.S.A.) as described previously (Huang et al., 1994; Endres et al., 1998a,b).
Physiologic parameters
Recording of physiologic parameters was essentially performed as previously described (Huang et al., 1994; Endres et al. 1998a,b). Briefly, rectal temperature was controlled and kept constant at 37 ± 0.5°C by means of a feedback temperature control unit (FHC, Brunswick, ME, U.S.A.) and a heating lamp for the duration of ischemia until 1 hour after reperfusion. After 24 hours, rectal temperature was again measured, and no differences were found between groups (<0.4°C, P > 0.05). In randomly assigned animals, the left femoral artery was cannulated using a PE-10 catheter for arterial blood pressure monitoring and blood withdrawal. Arterial blood samples were analyzed for pH and partial pressure of oxygen and carbon dioxide (Corning 178, Ciba-Corning Diagnostics, Medfield, MA, U.S.A.).
Determination of infarct size
Animals were killed at 24 hours by an overdose of pentobarbital (200 mg/kg, administered intraperitoneally). Subsequently, brains were sectioned coronally (2 mm) using a matrix (RBM-2000C, Activational Systems, Ann Arbor, MI, U.S.A.) and stained with 2% 2,3,5-triphenyltetrazolium chloride. Infarction volume was measured quantitatively according to Swanson et al. (1990) or using an indirect method that corrects for brain swelling as described (Endres et al., 1998b).
Histology
Animals were killed at 24 hours as above, and brains were frozen in liquid nitrogen vapor. Frozen coronal sections 1,500 μm in distance and 14 μm thick were cut on a cryostat (Microtome cryostat, Model HM505E, Microm, Walldorf, Germany), thaw-mounted on precleaned glass slides, and stained with hematoxylin and eosin according to a standard protocol.
Immunohistochemistry
Thawed tissue sections were dried completely and postfixed in absolute ethanol at −20°C for 10 minutes. After several washes in 0.1 mol/L phosphate-buffered saline, the sections were incubated with 10% normal goat serum containing 0.3% H2O2 for 1 hour at room temperature. For immunohistochemical staining, sections were incubated with a monoclonal antibody against neuronal nuclear protein (NeuN, Chemicon International Inc., CA, U.S.A., 1:300) at 4°C overnight. After washing in phosphate-buffered saline, the sections were incubated with bodipy fluorescein (BODIPY-FL) conjugated goat anti-mouse IgG secondary antibody (Molecular Probes, Eugene, OR, U.S.A.) for 1 hour at room temperature. After washing in phosphate-buffered saline, sections were dehydrated in ascending ethanol series, immersed in xylene, and coverslipped with Permount (Fisher Scientific, Pittsburgh, PA, U.S.A.)
Neurologic deficits
Scoring of sensorimotor deficits was performed by an observer naïve to the genotype using a scale from 0 (no deficit) to 3 (severe) as described (Huang et al., 1994; Endres et al., 1998a,b).
Statistical analysis
All experiments were performed in a blinded fashion. Values are presented as mean ± standard deviation (SD). Statistical comparisons were performed by unpaired Student's t-test (infarcts), ANOVA followed by Scheffe's test (physiology), or Mann-Whitney rank sum U test (neurological deficits), P values smaller than 0.05 were considered statistically significant.
RESULTS
Bdnf and nt4 mRNA expression in the adult mouse cerebral cortex
Northern blot analysis indicated that bdnf messenger RNA (mRNA) was more abundant than nt4 mRNA in the cerebral cortex (Fig. 1), in accordance with a previous report for adult rats (Timmusk et al., 1993). Bdnf+/–mice expressed reduced (50%) bdnf mRNA levels in the brain (Fig. 1) (see also Ernfors et al., 1994). As previously demonstrated, nt4−/− mice lack nt4 mRNA expression (Liu et al., 1995).
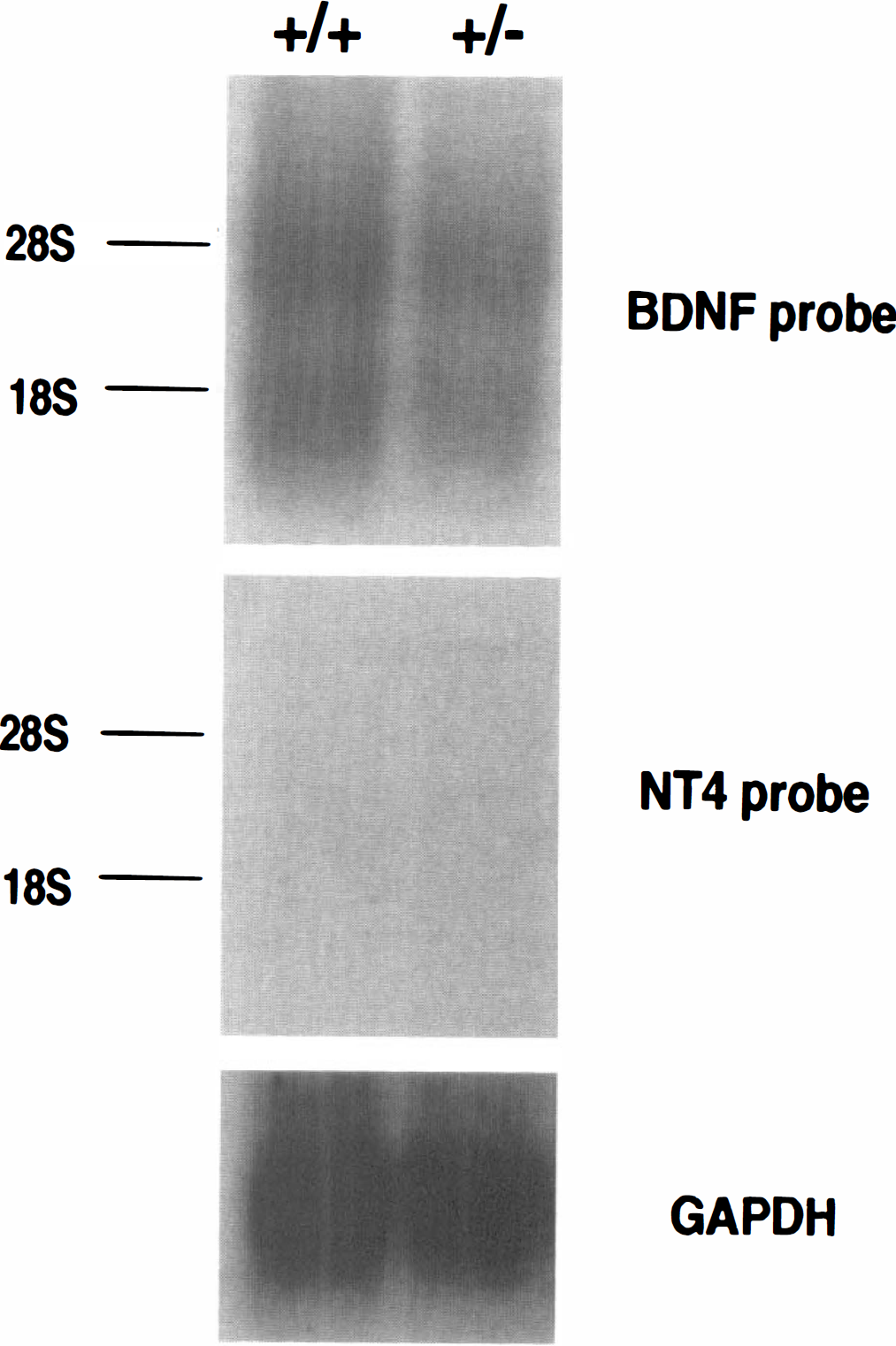
Northern blot with 40 μg of total cortex RNA from wild-type and bdnf+/– mice was sequentially hybridized with bdnf, nt4, and GAPDH cDNA probes. Two bdnf mRNA bands were readily detected at the expected size. In contrast, nt4 mRNA signals were undetectable, even with prolonged autoradiography. GAPDH hybridization showed equal amounts of RNA samples on the blot.
Physiologic parameters
As displayed in Table 1, there were no significant differences in systemic physiologic or regional cerebrovascular parameters between wild-type, nt4−/−, or bdnf+/–mice before, during, or after transient MCAO.
Physiologic parameters in nt4−/−, bdnf+/–, and wild-type control mice (129/SVja)
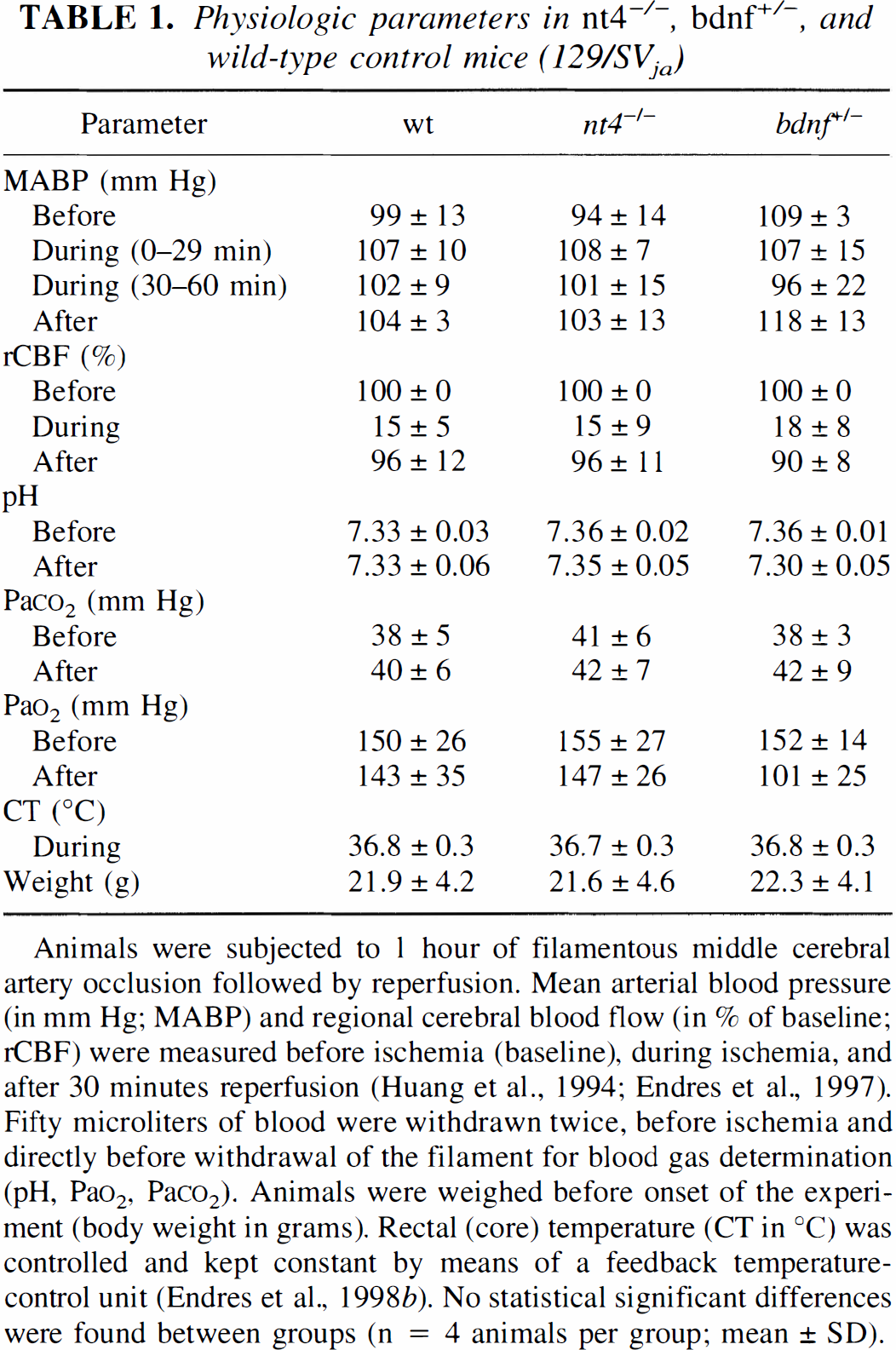
Animals were subjected to 1 hour of filamentous middle cerebral artery occlusion followed by reperfusion. Mean arterial blood pressure (in mm Hg; MABP) and regional cerebral blood flow (in % of baseline; rCBF) were measured before ischemia (baseline), during ischemia, and after 30 minutes reperfusion (Huang et al., 1994; Endres et al., 1997). Fifty microliters of blood were withdrawn twice, before ischemia and directly before withdrawal of the filament for blood gas determination (pH, Pao2, Paco2). Animals were weighed before onset of the experiment (body weight in grams). Rectal (core) temperature (CT in °C) was controlled and kept constant by means of a feedback temperature-control unit (Endres et al., 1998b). No statistical significant differences were found between groups (n = 4 animals per group; mean ± SD).
Cerebral infarcts after transient middle cerebral artery occlusion/reperfusion
After transient ischemia (1-hour MCAO and 23-hour reperfusion), infarcts in nt4−/− mice were significantly bigger (67%) than in wild-type 129/SVjae mice (Fig. 2A). This difference was also significant when lesion volume was calculated after correcting for brain swelling (28.3 ± 16.2 mm3 versus 47.6 ± 15.5 mm3 in wild-type versus nt4−/− mice, respectively, P < 0.01). Moreover, sensorimotor deficits were significantly worse in nt4−/− mice after 24 hours, indicating that larger infarcts were accompanied by diminished central nervous system function (1.1 ± 0.8 versus 2.2 ± 0.8 in wild-type versus nt4−/− mice, respectively, P < 0.01).
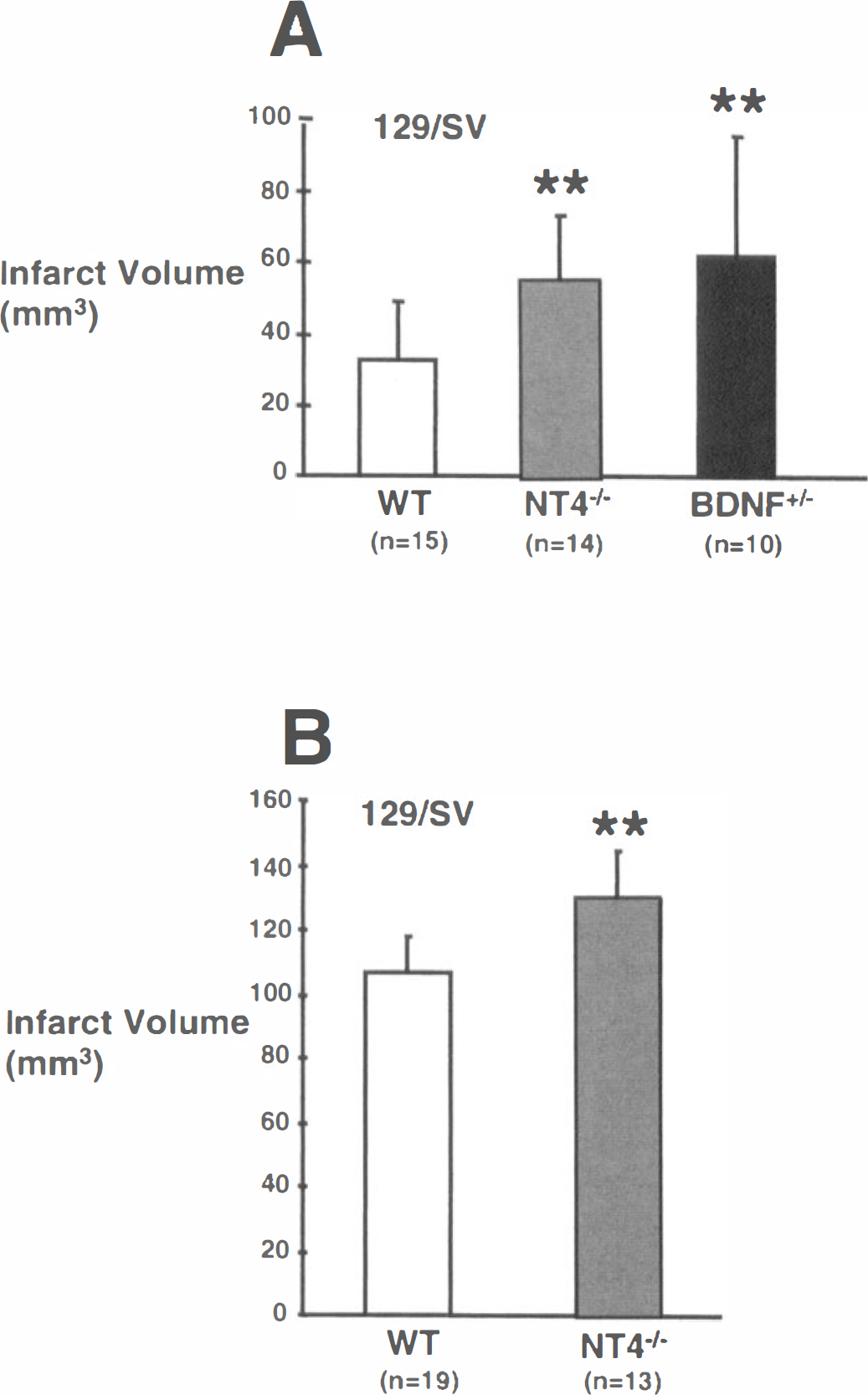
Bdnf+/– mice developed larger infarcts (92%) compared to those of controls when subjected to 1-hour MCAO/ 23-hour reperfusion (Fig. 2A). Differences were also significant when measured by the indirect method (28.3 ± 16.2 mm3 versus 52.8 ± 27.7 mm3 in wild-type versus bdnf+/– mice, respectively, P < 0.05). Neurologic deficits were also significantly worse after 24 hours (1.1 ± 0.8 versus 2.0 ± 0.7 in wild-type versus bdnf+/– mice, respectively, P < 0.05).
In preliminary histologic studies (n = 2 to 3 per group), the same trend was noted (see Fig. 3). Except for differences in infarct area, qualitative differences were not detected in overall histopathology. Nevertheless, in ischemic striatum from wild-type mice, normal appearing NeuN-positive nuclei were found among positive nuclei with a condensed staining pattern; all nuclei showed this abnormal pattern in nt4−/− and bdnf+/– mice (Fig. 4).
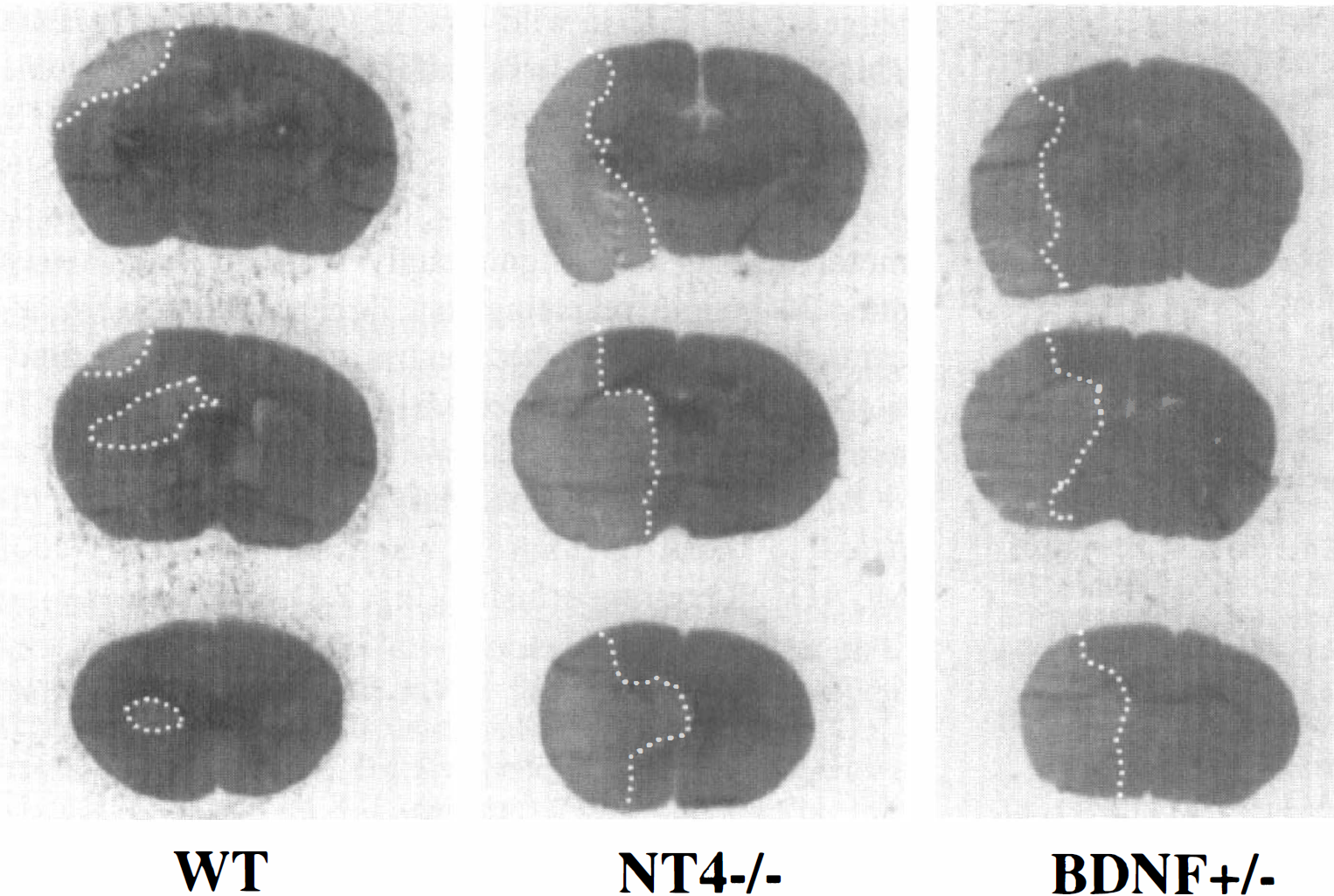
Hematoxylin and eosin staining of coronal sections representative of wild-type, bdnf+/–, and nt4−/− mice after 1-hour middle cerebral artery occlusion and 23-hour reperfusion. The infarcted area (outlined with white dotted line) involves the striatum in all three animals. There is limited cortical infarction in the wild-type (WT) whereas there is extensive cortical involvement in animals lacking one allele of bdnf (BDNF+/–) or both alleles of nt4 (NT4−/−).
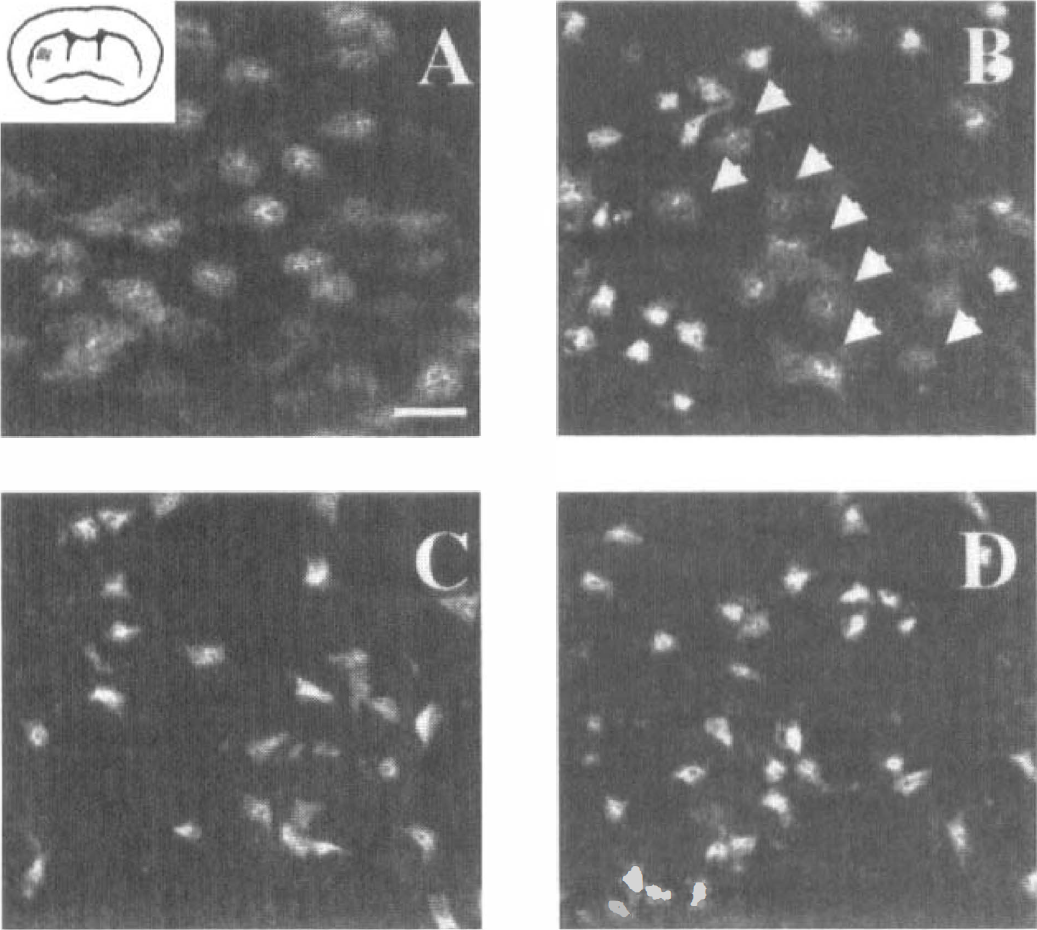
NeuN immunohistochemical staining within striatum. Nonischemic striatal neurons exhibit rounded nuclei
Effects of permanent ischemia in nt4–/– mice
Lesions were significantly larger (21%) compared to wild-type controls in nt4−/− mice subjected to permanent ischemia for 24 hours (Fig. 2B). Differences were also significant when measured with an indirect method (80.1 ± 10.8 mm3 versus 97 ± 17.3 mm3 in controls versus nt4−/−, respectively, P < 0.01, n = 19 and 13 animals).
DISCUSSION
We evaluated outcome after MCAO in transgenic animals either deficient in nt4 gene expression or expressing reduced bdnf levels. We found that these mutant mice were less resistant compared to controls, indicating that nt4 and bdnf may protect from ischemic injury. Bigger infarcts were accompanied by worse neurologic scores in both mutant strains, indicating that the increases in infarct volume corresponded to poorer outcome in sensorimotor function at 24 hours. A more rapidly evolving lesion was also detected in both mutant strains by NeuN immunohistochemistry. In the striatum, nuclei were condensed and irregular, whereas in wild-type mice, normal rounded-appearing nuclei were frequent (Fig. 4). The enhanced susceptibility to ischemia could not be explained by differences in cardio- or cerebrovascular parameters. Interestingly, absolute infarct volumes were larger in bdnf+/– compared to nt4−/− mice (Fig. 2A), indicating that the loss of even one bdnf allele may be functionally more relevant than complete loss of nt4 gene expression. The percentage of increase in infarct size in nt4−/− compared to control mice was less pronounced after permanent MCAO (Fig. 2). This may mean that the neuroprotective effects depend on the severity of the insult as has been suggested by others (Lindvall et al., 1994) or, alternatively, may simply reflect a ceiling effect in the permanent model. Taken together, we infer that nt4 and bdnf confer resistance to brain ischemia when expressed normally. We cannot exclude the possibility that differences in brain development in nt4 and bdnf mutants contribute to the differences in stroke outcome, although gross morphologic appearance of the brain is normal in both mutants.
bdnf nt4, and trkB, their common receptor, are abundantly expressed in the central nervous system especially in cortex and hippocampal subsectors (Kokaia et al., 1993; Timmusk et al., 1993; Yan et al., 1997; Friedman et al., 1998). NT4 and BDNF share a 54% amino-acid identity, and both bind with high affinity to TrkB (Ip et al., 1992). We found that total brain bdnf mRNA levels were more abundant than nt4 mRNA which agrees with the literature (Fig. 1). Although not detectable by analysis of total RNA (Northern blot), widespread nt4 mRNA expression was demonstrated in the brain by RNAase protection assay and polyA+ RNA Northern blotting, as was NT4 protein (Ip et al., 1992; Timmusk et al., 1993; Friedman et al., 1998).
Indirect evidence supports an endogenous neuroprotective role for trkB and its receptor agonists during brain ischemia. After both focal and global cerebral ischemia, neuronal upregulation of trkB (but not trkA or trkC) and bdnf mRNA (in contrast to nt3) was demonstrated (Lindvall et al., 1992; Comelli et al., 1993; Merlio et al., 1993; Takeda et al., 1993; Kokaia et al., 1995, 1996; Friedmann et al., 1998; Narumiya et al., 1998). TrkB and bdnf mRNA expressing neurons are typically located in the penumbral zone (focal ischemia) or CA3 subsector of the hippocampus (global ischemia). Neurons, however, that are likely to die, (e.g., in the ischemic core [focal ischemia] or in the CA1 hippocampal subsector [global ischemia]) do not show enhanced gene expression (Takeda et al., 1993; Kokaia et al., 1996). However, neurotrophins may not always protect ischemic tissue. For example, bdnf may enhance excitatory synaptic transmission which could augment excitotoxic damage and increase the probability of seizures (Knipper et al., 1993; Lohof et al., 1993; Kang and Schuman, 1995). Moreover, truncated TrkB isoforms may block bdnf signaling (Knusel et al., 1994; Eide et al., 1996). In contrast to the adult, expression of truncated TrkB receptors is low in the neonatal period which may explain the more consistent protective effects of BDNF in neonatal in contrast to adult models (Cheng et al., 1997).
The benefits of exogenous administration of several neurotrophins have been reported previously in focal and global ischemia as well as in other brain insults (Beck et al., 1994; Tsukahara al., 1994; Chan et al., 1996; Alexi et al., 1997; Cheng et al., 1997; Schäbitz et al., 1997). For example, exogenous bdnf delivered intracerebroventricularly decreases lesion size after permanent focal (2.1 μg/d for 8 days, beginning 24 hours before ischemia) (Schäbitz et al., 1997) and global cerebral ischemia (0.013 μg/h for 7 days, beginning immediately before ischemia) (Beck et al., 1994) in rats. Similarly, intrace-rebroventricular administration of nt4 1 day before and immediately after occlusion confers protection in a rat permanent MCAO model. Infarcts were reduced by 34% 1 day after ischemia; however, infarction volume was unaltered 4 to 7 days after ischemia (2 × 0.54 μg) (Chan et al., 1996).
In summary, our findings are consistent with the conclusion that endogenous bdnf and nt4 are neuroprotective during MCAO in adult mice and underscore the potential importance of the trkB receptor in conferring resistance to ischemic injury.
