Abstract
With use of magnetic resonance imaging (MRI), the effects of early and delayed treatment of embolic stroke in rat with recombinant tissue plasminogen activator (rt-PA) were investigated. Rats with embolic stroke were treated with rt-PA at 1 (n = 9) or 4 (n = 7) hours after stroke onset or were untreated (n = 15). Diffusion-weighted imaging, perfusion-weighted imaging, and T2-weighted imaging were performed before and after embolization from 1 hour to 7 days. No significant differences were detected in the relative areas with low cerebral blood flow (CBF), apparent diffusion coefficient of water (ADCw), and T2 between the 4-hour treated group and the untreated group. Significant decreases in the average relative areas with low CBF were detected in the 1-hour treated group from 4 to 48 hours after embolization as compared with the untreated group. The increase in T2 in the 1-hour treated group was significantly lower than in the untreated and 4-hour treated groups. A significant increase in ADCw was detected in the 1-hour treated group at 3 and 24 hours after embolization as compared with the untreated and 4-hour treated groups. Secondary embolization was detected by both MRI and laser scanning confocal microscopy. The data suggest that MRI can detect the efficacy of rt-PA treatment and secondary ischemic damage.
Cerebral artery thromboembolism is a major cause of human cerebral infarction. Studies using recombinant tissue plasminogen activator (rt-PA) have demonstrated a significant improvement of neurological outcome in patients with ischemic stroke, although the risk of hemorrhagic transformation increased in patients receiving rt-PA treatment (NINDS, 1995). Diagnostic tools to predict and characterize the efficacy and safety of thrombolysis will greatly improve the management of stroke patients. Diffusion-weighted imaging (DWI), perfusion-weighted imaging, and T2-weighted magnetic resonance imaging (MRI) have been used to characterize the efficacy of acute rt-PA intervention after experimental stroke (Röther et al., 1996; Jiang et al., 1998). The values for cerebral blood flow (CBF) and apparent diffusion coefficient of water (ADCw) obtained 1 hour after injection of rt-PA correlate with histological outcome in the tissue, and the beneficial effect of early rt-PA treatment of embolic stroke is reflected in an increase in CBF and ADCw (Jiang et al., 1998).
Therapeutic intervention with rt-PA is an important breakthrough in the treatment of acute ischemic stroke (NINDS, 1995). However, the majority of stroke patients currently do not present within the required 3-hour time window for rt-PA intervention and so cannot be treated (Barsan et al., 1993). Therefore, it is important to investigate the physiological and biophysical effects of thrombolytic therapy at various durations of ischemia with the ultimate goal of extending the therapeutic window of rt-PA treatment. Previous studies have demonstrated that treatment with rt-PA alone improves neurological deficits and reduces mean infarct volume if it is administered at 1 and 2 hours but not at 4 hours after onset of embolic stroke (Zhang et al., 1999a). However, employing a combination of adhesion molecule antibody and rt-PA treatment at 4 hours after stroke reduces infarct volume. In the present study, we investigated the physiological and biophysical effects of early (1 hour) and delayed (4 hours) rt-PA intervention by means of MRI and histopathology in a rat model of embolic stroke.
MATERIALS AND METHODS
All experimental procedures were approved by the Care of Experimental Animals Committee of Henry Ford Hospital.
Three groups of male Wistar rats weighing 300 to 450 g (n = 31) subjected to embolic stroke were investigated: untreated animals (n = 15), early-treated animals in which rt-PA was administered 1 hour after embolization (n = 9), and delay-treated animals in which rt-PA was administered 4 hours after embolization (n = 7). The method to produce embolic stroke and rt-PA treatment in this study was identical to that previously described (Zhang et al., 1997; Jiang et al., 1998). In brief, an aged white clot (prepared 24 hours before ischemia) was slowly injected into the internal carotid artery to block the middle cerebral artery and to cause ischemic stroke (Zhang et al., 1997).
Magnetic resonance imaging measurements were performed using a 7 T, 20-cm-bore superconducting magnet (Magnex Scientific, Abingdon, U.K.) interfaced to a console (SMIS, Surrey, England). A 12-cm-bore actively shielded gradient coil set, capable of producing magnetic field gradients up to 20 G/cm, and a birdcage radiofrequency coil were used. Stereotaxic ear bars were used to minimize movement during the imaging procedure. During MRI measurements, anesthesia was maintained using a gas mixture of N2O (69%), O2 (30%), and halothane (0.75 to 1%). Rectal temperature was kept constant using a feedback-controlled water bath. A modified fast low angle shot (FLASH) imaging sequence was employed for reproducible positioning of the animal in the magnet at each MRI session (Knight et al., 1991; Jiang et al., 1993). The MRI measurements were performed before ischemia, repeatedly for 4 to 5 hours (for untreated and 1-hour treated groups) or 7 hours (for 4-hour treated group) after onset of embolization, and at 24, 48, and 168 hours after onset of embolization (for all three groups). Methods to set up and perform DWI, perfusion-weighted imaging, and T2-weighted imaging measurements as well as the analysis of MRI data employed in this study are identical to those previously described (Jiang et al., 1998).
Laser scanning confocal microscopy was used to measure cerebral plasma perfusion volume and to identify distribution of clot materials in an additional two animals (one untreated and one treated at 1 hour) using a double-labeled plasma flow method (Zhang et al., 1999c). The embolus was labeled by mixing Evans blue (2% solution in saline, 0.2 mL/100 g of body wt; Sigma Chemicals, St. Louis, MO, U.S.A.) with blood. Animals were administered (intravenously) 1 mL of fluorescein isothiocyanate/dextran (50 mg/mL, 2 × 106 molecular weight; Sigma) immediately after obtaining the last MR image at 4 to 5 hours after embolization, and fluorescein isothiocyanate/ dextran was allowed to circulate for 1 minute. The animals were decapitated, and the brains were rapidly removed and placed in 4% paraformaldehyde for 24 hours. Vibratome coronal sections (150 μm) were analyzed with a Bio-Rad MRC 1024 laser scanning confocal microscope mounted onto a Zeiss microscope (Bio-Rad, Cambridge, MA, U.S.A.). A 10× objective or a 40× oil immersion objective was used for data acquisition. Green (fluorescein isothiocyanate/dextran) and red (Evans blue) fluorochromes on the sections were excited by the laser beam at 488 and 568 nm, respectively, and emissions were acquired with photomultiplier tubes through 522- and 605-nm emission filters, respectively. Histological processing and measurements of brain tissue employed in the current study were identical to those previously described (Jiang et al., 1998).
Statistics
Observations are summarized as means ± SE. The data analysis was carried out using paired t tests for time effects and by two-sample t tests for rt-PA effects. As the effect of rt-PA treatment of thromboembolic stroke is most evident at 24 and 48 hours (Zhang et al., 1997; Jiang et al., 1998) and we have the largest sample size at 24 hours, multiple-comparison issues with respect to rt-PA effects were addressed by considering the 24-hour comparison as the primary test of an rt-PA effect. Unadjusted P values for the secondary tests at other times are also presented.
For tests of time effects (changes from baseline for CBF and ADCw and for normal versus ischemic hemisphere for T2), Bonferroni-adjusted P values were used. There were eight comparisons for CBF and T2 and seven for ADCw. For these tests, P values of 0.05/8 = 0.0065 and 0.05/7 = 0.0071, respectively, were considered significant.
RESULTS
All physiological parameters after embolization were within the normal range. Figure 1 shows temporal profiles of the relative areas with low CBF (relative area containing CBF below 50 mL/100 g/min to the area of the ipsilateral hemisphere within the reference coronal section) in the ipsilateral hemisphere before and after embolization. As a result of embolization, the relative areas with low CBF in the ipsilateral hemisphere increased (P < 0.01) from preischemic control values in all three groups. In the untreated and 4-hour treated groups, relative areas with low CBF in the ischemic hemisphere remained large up to 24 hours. No statistically significant differences were detected in the relative areas with low CBF between the untreated and 4-hour treated groups, although the relative areas with low CBF in the 4-hour treated group decreased slightly compared with the untreated group. The areas with low CBF in the 1-hour treated group gradually decreased after administration of rt-PA. Significant differences in the relative areas with low CBF were detected between the 1-hour treated and the untreated and 4-hour treated groups at 4 hours (with untreated, P =0.008), 5 hours (with untreated, P =0.006), 24 hours (with untreated, P < 0.001; with 4-hour treated, P =0.006), and 48 hours (with untreated, P =0.025; with 4-hour treated, P =0.048) after embolization.
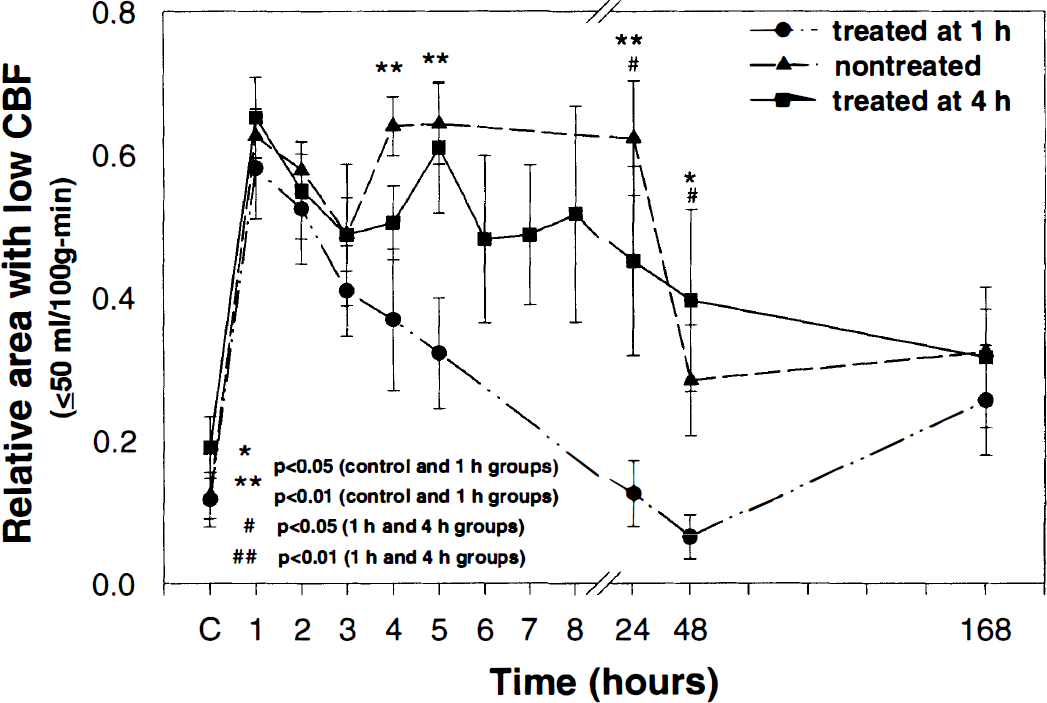
Average relative area with low cerebral blood flow (CBF; relative area containing CBF below 50 mL/100 g/min to the area of the ipsilateral hemisphere) in the ipsilateral hemisphere of treated and nontreated animals plotted as a function of time after embolization. As a result of embolization, the relative areas with low CBF in the ipsilateral hemisphere increased (P < 0.01) from preischemic control values in all three groups. The decrease in the mean values of the relative areas with low CBF was greater in the 1-hour treated group than in the untreated group (4 hours, P = 0.008; 5 hours, P = 0.006; 24 hours, P < 0.001; 48 hours, P = 0.025) and 4-hour treated groups (24 hours, P < 0.026; 48 hours, P = 0.048) after embolization. No statistically significant differences were detected in the areas with low CBF between untreated and 4-hour treated groups.
Figure 2 presents DWI results (b =800 s/mm2) obtained from 1 to 48 hours after injection of the embolus. A hyperintensity is apparent in the DWI of the ipsilateral hemisphere in all three animals after embolization. In the untreated and 4-hour treated animals, a DWI hyperintensity is apparent in the ipsilateral caudate putamen and insular cortex after the onset of ischemia; image contrast and the area of hyperintensity increased as ischemia progressed and were maximal at 24 to 48 hours. In the 1-hour treated animals, changes of DWI hyperintensity were variable after injection of rt-PA. One 1-hour treated animal exhibited a pattern and temporal profile of DWI hyperintensity similar to that for the control animals, whereas three 1-hour treated animals exhibited an early disappearance of DWI hyperintensity (within 24 hours). The remaining 1-hour treated animals exhibited a slower increase in image contrast and area of hyperintensity than the control untreated animals. In the 1-hour treated animals, four of nine animals had redistributed lesion locations from the ipsilateral caudate putamen and insular cortex during acute ischemia to smaller areas in the cortex at chronic time points (24 and 48 hours). In the 4-hour treated animals, two of seven animals exhibited a reduction of hyperintense areas after rt-PA treatment, and the remaining animals exhibited a profile of DWI hyperintensity similar to that of the untreated animals.
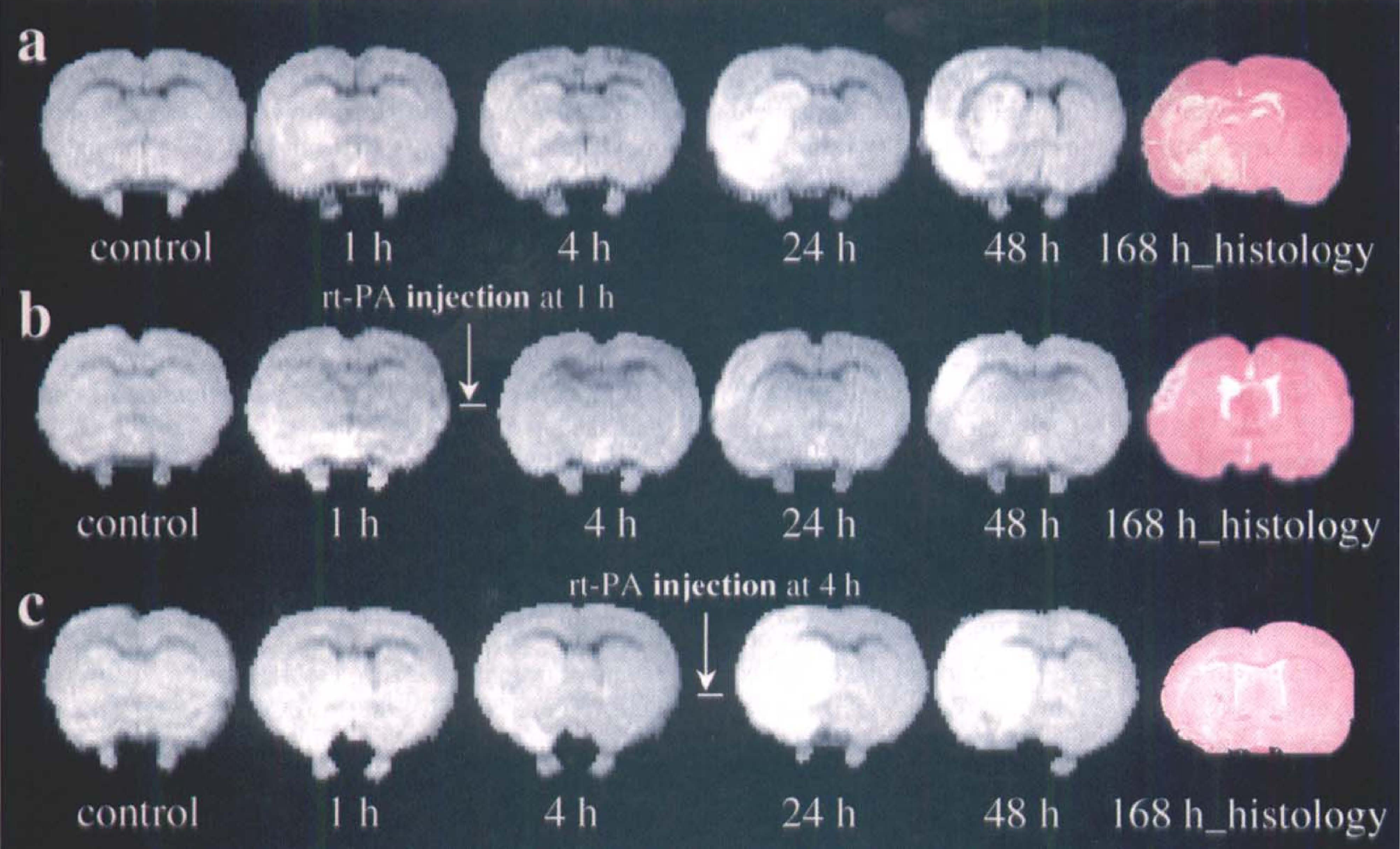
Diffusion-weighted images (b = 800) of coronal sections without
Figure 2 shows the corresponding hematoxylin and eosin-stained coronal sections labeled as 168 h_histology. The damaged area in the hematoxylin and eosin-stained section from untreated and treated animals corresponds closely with the hyperintense area in the DWI obtained 24 hours after embolization. The representative untreated animal exhibited a lesion area incorporating putamen and parietal and insular cortex, whereas the representative 1-hour treated animal exhibited a small lesion area localized to the parietal cortex. The 1-hour rt-PA-treated animals exhibited smaller normalized areas (P =0.04) and volumes (P =0.01) of ischemic damage (34 ± 9%, percent of the lesion area to the area of the contralateral hemisphere within the reference coronal section; 20 ± 11%, percent of the lesion volume to the volume of the contralateral hemisphere) than those in the untreated animals (65 ± 20%, percent of the lesion area to the area of the contralateral hemisphere within the reference coronal section; 43 ± 14%, percent of the lesion volume to the volume of the contralateral hemisphere). The 4-hour rt-PA-treated animals exhibited intermediate areas (63 ± 7%, relative percent of the lesion area) and volumes (36 ± 8%, relative percent of the lesion volume) of ischemic damage.
As illustrated in Fig. 2b, redistribution of lesioned areas was detected by DWI in 44% of 1-hour rt-PA-treated animals. The newly appearing lesions may be caused by incompletely dissolved clots moving downstream to occlude small vessels. To confirm this, laser scanning confocal microscopy was performed to identify the fragments of original clot and cerebral plasma perfusion volume. Figure 3 shows the results from a rat killed at 4 hours after 1-hour rt-PA treatment. A segment of Evans blue-stained clot lodged downstream of the origin of the middle cerebral artery (Fig. 3A, blue). A coronal section shows the Evans blue-stained clot (Fig. 3B, arrow). Figures 3C and D comprises images obtained from laser scanning confocal microscopy of the coronal section in Fig. 3B. Figure 3C shows the xy projection through the stack of 20 optical sections (5-μm-thick section). The clot appears red (arrow) and the perfused cerebral vasculature green (an absence of vascular perfusion was evident around the clot). Figure 3D is a high-magnification image from Fig. 3C (arrowhead) and shows fragments of the parent clot (red) blocking cerebral plasma perfusion (green).
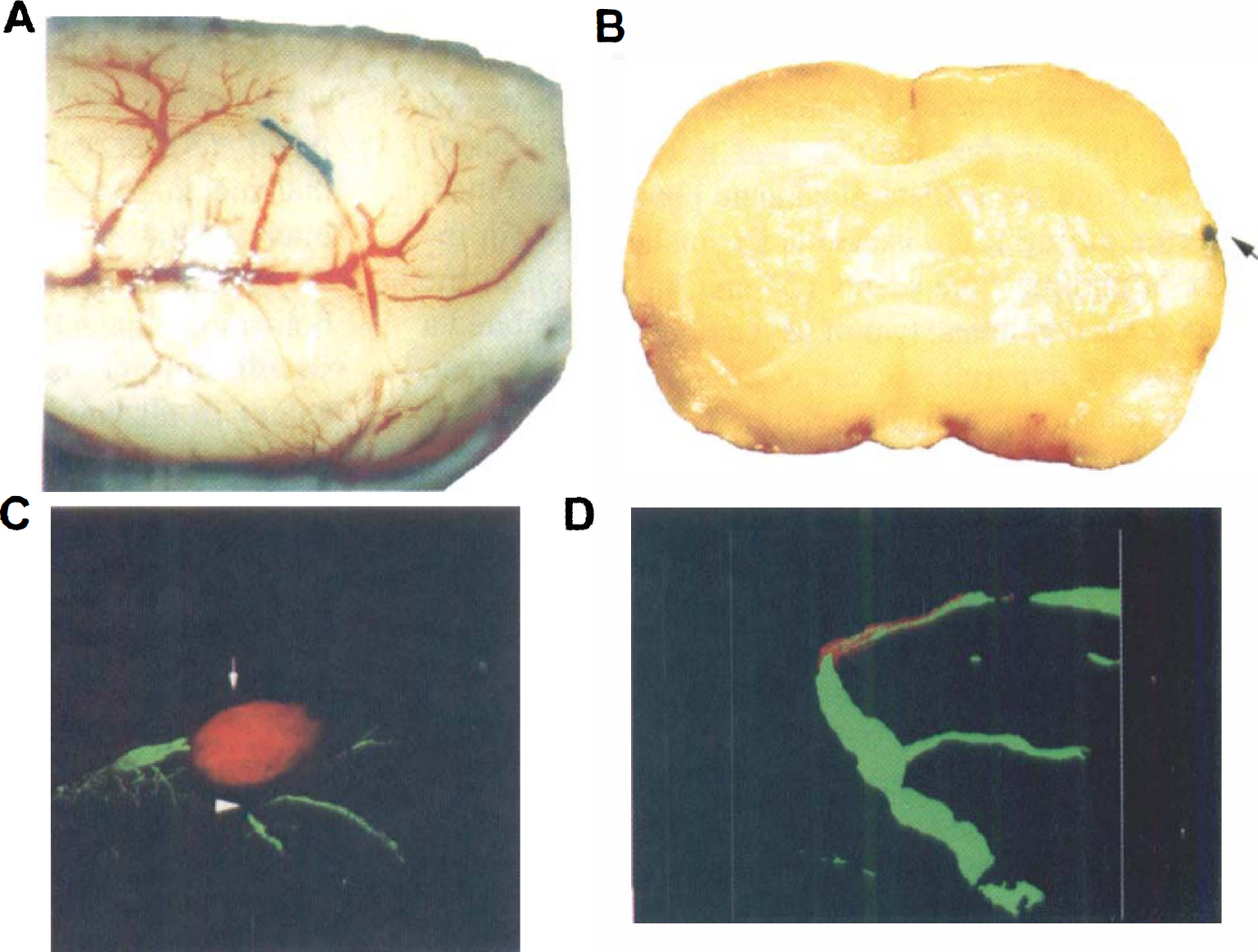
A representative rat killed at 4 hours after 1-hour recombinant tissue plasminogen activator treatment investigated by laser scanning confocal microscopy and histology.
Figure 4 presents the temporal profiles of normalized ADCw (ADCw values for ischemic damaged tissue normalized to preischemic control tissue) for a region of interest localized to the center of the DWI abnormality in the ipsilateral hemisphere in all three groups. After embolization, the ADCw values in the ischemic striatum significantly (P < 0.001) decreased from preischemic values in all three groups. In the untreated and 4-hour treated groups, ADCw in the ischemic striatum remained below preischemic ADCw values up to 24 hours. In the 4-hour rt-PA-treated group, however, ADCw transiently increased after rt-PA injection and then returned to low levels as in the untreated group at 24 hours after middle cerebral artery occlusion. There were no significant differences in the relative ADCw between the 4-hour treated group and the untreated group. In the 1-hour treated group, ADCw gradually increased after injection of rt-PA. Higher mean ADCw values were present from 1 to 24 hours after administration of rt-PA in the 1-hour treated group compared with the untreated and 4-hour treated groups. Statistically significant differences were detected in normalized ADCw values between the 1-hour treated with untreated and 4-hour treated groups at 3 hours (with untreated, P =0.039; with 4-hour treated, P < 0.001), 4 hours (with untreated, P =0.017), and 24 hours (with untreated, P =0.023; with 4-hour treated, P < 0.025) after embolization.
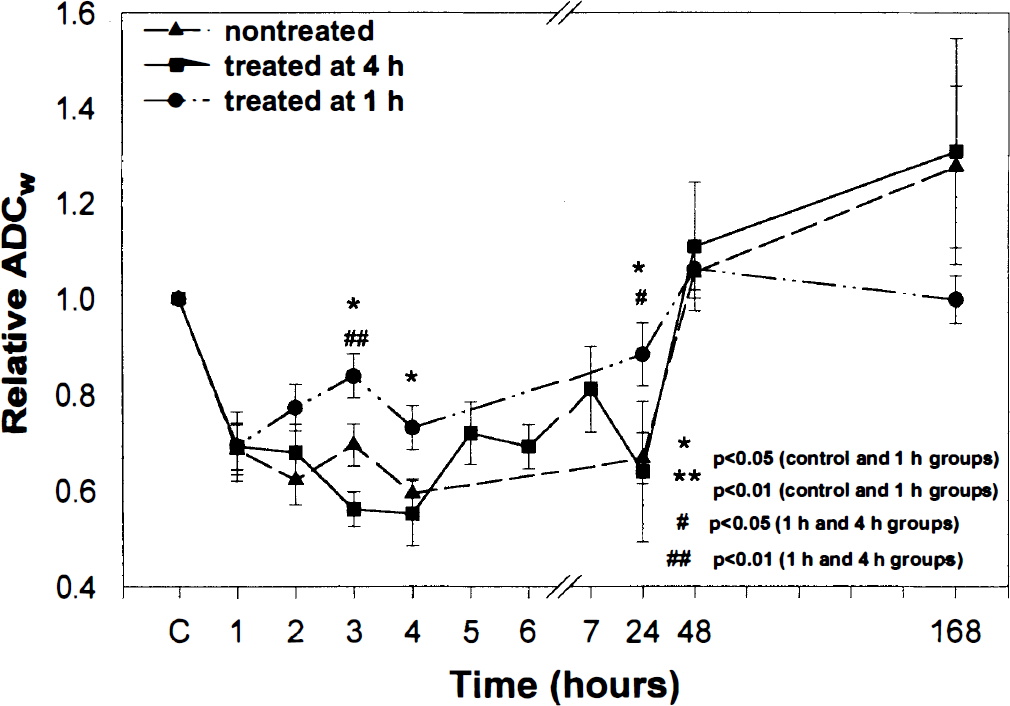
Relative apparent diffusion coefficient of water (ADCw) in the lesion under untreated and recombinant tissuse plasminogen activator-treated conditions plotted as a function of time after embolization. The relative ADCw values in the ischemic area significantly (P < 0.001) decreased from preischemic values after embolization in all three groups. Significant differences were detected after embolization between the 1-hour treated and untreated (3 hours, P = 0.039; 4 hours, P = 0.017; 24 hours, P < 0.023) and 4-hour treated (3 hours, P < 0.001; 24 hours, P < 0.025) groups. No statistically significant differences were detected in the relative ADCw between untreated and 4-hour treated groups.
Figure 5 shows the postembolic temporal profiles of ΔT2 in the lesion (the same region of interest as in ADCw measurements) in the rt-PA-treated and untreated groups. A small increase in ΔT2 was detected within 4 hours; ΔT2 was maximized at 24 hours in the untreated group (P < 0.001) and 4-hour rt-PA-treated group (P < 0.004) and at 48 hours in the 1-hour treated group (P =0.006) and then declined at 1 week after middle cerebral artery occlusion. However, ΔT2 at 1 week remained elevated relative to baseline in all three groups. The 1-hour rt-PA-treated group had a lower ΔT2 than the control and 4-hour treated groups at 24 hours (untreated group, P =0.003; 4-hour treated group, P =0.020) and 48 hours (4-hour treated group, P =0.009) after embolization. There were no significant differences in ΔT2 between untreated and 4-hour rt-PA-treated groups.
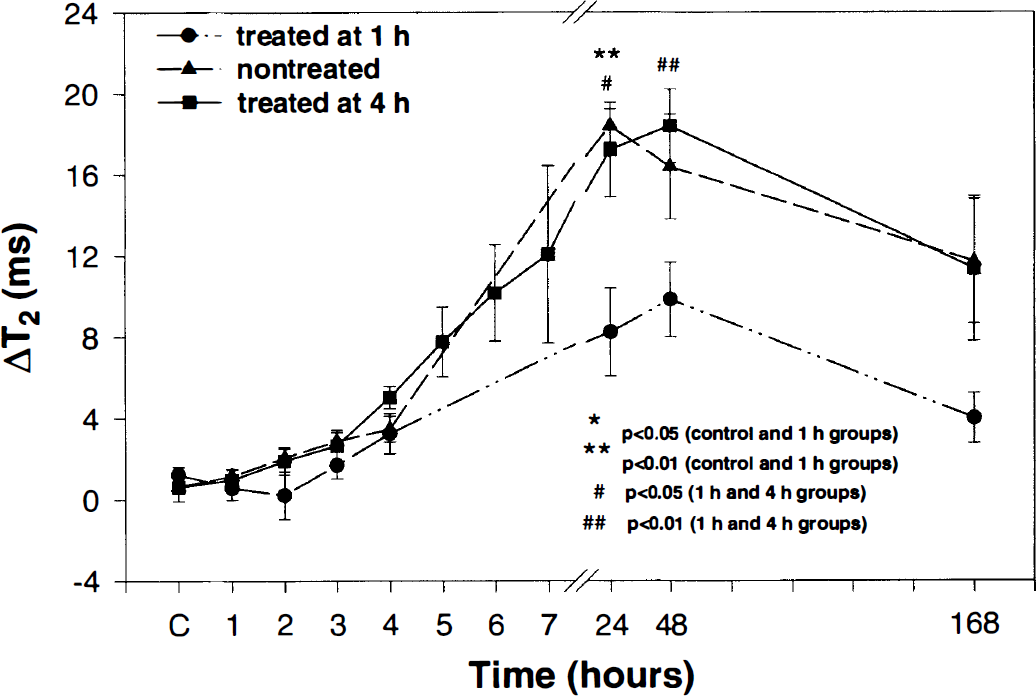
The temporal profiles of ΔT2 (the difference of T2 values in homologous regions of the ipsilateral and contralateral hemispheres) in the lesion before and after embolization in all three treated and untreated groups. A small increase in ΔT2 was detected within 4 hours, and ΔT2 was maximized at 24 hours in untreated (P < 0.001) and 4-hour recombinant tissue plasminogen activator (rt-PA)-treated (P < 0.004) groups and at 48 hours in the 1-hour treated group (P < 0.01) and then declined at 1 week after embolization. The 1-hour rt-PA-treated group had a lower ΔT2 than the untreated and 4-hour treated groups at 24 hours (untreated group, P = 0.003; 4-hour treated group, P = 0.020) and 48 hours (4-hour treated group, P = 0.009) after embolization. There were no significant differences in ΔT2 between untreated and 4-hour rt-PA-treated groups.
DISCUSSION
In this study, we used MRI to investigate 1- and 4-hour rt-PA intervention after embolic stroke. Our data suggest that delayed rt-PA treatment (4 hours) does not significantly alter the MRI parameters of ADCw, CBF, and T2 or reduce ischemic tissue damage as compared with those in control untreated animals. However, early rt-PA treatment (1-hour) reduces ischemic tissue damage, as reflected in a rapid increase in ADCw, a reduction of areas with low CBF, and a reduction of the increase in T2.
Our data indicate that tissue perfusion is not significantly increased when rt-PA treatment is instituted at 4 hours after embolic stroke. We have not performed MRI angiography, and we do not know the status of the clot after thrombolysis. We also cannot exclude the possibility that the clot was effectively lysed but that microvascular plugging occurred downstream of the clot from emboli, from leukocyte adhesion, or from fibrin deposition (del Zoppo et al., 1991). In addition, erythrocyte stasis, reduction of local blood perfusion pressure, increased blood viscosity in vessels, and narrowed lumina due to perivascular glial swelling (Ames et al., 1968; Ginsberg and Myers, 1972; del Zoppo et al., 1991) may also account for the hypoperfusion after 4-hour rt-PA treatment. The cause of low tissue perfusion after 4 hours of rt-PA treatment requires further investigation.
The indexes of MRI measurements of the efficacy of rt-PA intervention in the animal study may have potential for the management of human stroke. Our data indicate that rapid recanalization of an embolically occluded brain artery leads to a reduced lesion. In recent study of rt-PA treatment in patients, Heiss et al. (1998) demonstrated that the percentage of CBF changes measured using [15O]H2O positron emission tomography predicted the degree of clinical improvement achieved within 3 weeks when patients were treated within the 3-hour therapeutic window of rt-PA. Studies by many investigators (Sundt and Waltz, 1971; Pulsinelli et al., 1982; Garcia et al., 1993) using a variety of animal models have demonstrated a critical period of 2 to 7 hours after ischemia in which reversible cell damage transforms to irreversible damage. The time to salvage tissue is related to the degree and duration of CBF reduction, the specific tissue, and the animal species. The efficacy of reperfusion in reducing cell damage strongly depends on the rapidity of reperfusion, as survival of ischemic tissue decreases exponentially with time (Astrup et al., 1981; Jones et al., 1981; Heiss and Rosner, 1983). Thus, in our model of embolic stroke in the rat, early restoration of CBF may be a good indicator of the therapeutic efficacy of rt-PA intervention.
The duration of ischemia and residual CBF are reflected in the evolution of changes of other MRI-measurable biophysical parameters such as ADCw and T2. Busza et al. (1992) have reported that the decrease in ADCw is related to a threshold of CBF. However, ADCw is a function of a number of variables (Cooper et al., 1970; Tanner, 1978; LeBihan et al., 1989; Moseley et al., 1990; Benveniste et al., 1992; Helpern et al., 1992), not only CBF. Benveniste et al. (1992) demonstrated that ADCw declines following direct microdialysis treatment of rat brain with ouabain, a known inhibitor of Na+,K+-ATPase, which acts without changing CBF. Therefore, in the acute ischemic phase, the decline in ADCw may be related to energy failure. Mintorovitch et al. (1994) demonstrated a significant decline in Na+,K+-ATPase activity at 30 and 60 minutes after middle cerebral artery occlusion in the rat. Rapid restoration of CBF induces an early recovery of ADCw, which may indicate the efficacy of rt-PA intervention, as has been demonstrated after cerebral venous embolization and rt-PA treatment of rat (Röther et al., 1996). T2 changes in cerebral ischemic tissue occur much later than changes in ADCw and are related to tissue edema (Knight et al., 1994; Jiang et al., 1997). Previous studies have demonstrated that early recanalization reduces infarction and edema (Overgaard, 1994). Therefore, reduction of the increase in T2 with 1-hour rt-PA treatment reflects the therapeutic efficacy of the thrombolytic intervention.
The efficacy of thrombolysis depends on not only the arterial recanalization but also the secondary effects of thrombolysis such as redistribution of clot material. In this study, our data demonstrate that MRI and laser scanning confocal microscopy can detect the secondary ischemic damage caused by secondary embolization. Secondary ischemic events have long been recognized (Dinsmore et al., 1986), and progressing ischemia extends to previously spared cerebral regions (Haley et al., 1988). However, it is difficult to identify and monitor the processes that cause secondary ischemia in the patient. Early diagnosis of secondary ischemia is of clinical interest. Our study demonstrates that MRI and laser scanning confocal microscopy techniques and the new model of embolic stroke may be useful to investigate secondary ischemic damage.
Extending the time window of thrombolytic intervention would greatly benefit the management of stroke patients. A potentially efficacious treatment strategy to extend the window for thrombolysis would be to employ a combination thrombolytic and neuroprotective therapy (Bowes et al., 1995). A combination of rt-PA and anti-leukocyte adhesion molecule antibody (anti-CD18, anti-ICAM-1) treatment after 4-hour embolic stroke in rat significantly reduces the infarct volume as compared with rt-PA-treated animals (Zhang et al., 1999a,b). In the present study, a reduction in the mean relative areas with low CBF (<50 mL/100 g/min) and a transiently elevated mean relative ADCw were detected after rt-PA treatment in the 4-hour rt-PA-treated group compared with the untreated group, although no improvement in the ischemic outcome was present. We speculate that the CBF and ADCw profiles may provide an index to the window of opportunity for using combination therapy after acute stroke.
In summary, our data indicate that select MRI measurements can distinguish between thrombolytic therapy with rt-PA performed within (1 hour) and outside (4 hours) of the therapeutic window. Early changes in tissue perfusion and ADCw values identify later therapeutic benefit. Secondary ischemic events likely caused by emboli derived from the lysed clot may contribute to the ischemic lesion.
Footnotes
Acknowledgments
The authors thank Upul Senaratne and Jonathan Prostak for data analysis and Denice Bliesath for manuscript preparation.
