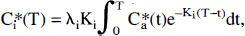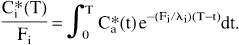Abstract
Autoradiographic measurement of local cerebral blood flow (CBF) with [14C]iodoantipyrine (IAP) is limited in mice by the difficulty in cannulating vessels and the blood loss for repeated blood sampling. The authors modified and validated the method to measure local CBF with [14C]IAP in mice by combining intraperitoneal tracer application with a single blood sampling from the heart at the end of the experiment. Experiments were carried out in male SV129 mice under halothane anesthesia. After intraperitoneal administration of 15 μCi [14C]IAP, arterial blood samples were collected repeatedly and anesthetized animals were immersed in liquid nitrogen. In addition, frozen blood from the heart was sampled to obtain the final blood [14C]radioactivity. Correlation analysis between the sampling time and [14C] radioactivity of the arterial blood revealed a highly significant linear relationship (P < 0.001, r = 0.978) and a lag time of the [14C]tracer in arterial blood of 3.3 ± 0.6 seconds. [14C]radioactivity of the final arterial blood sample (444 ± 264 nCi/mL) was almost equal to that of the heart blood (454 ± 242 nCi/mL), and the absolute difference in each animal was 3.3 ± 4.2% (mean ± SD). The convolution integrals for the CBF calculation were determined either by integrating the radioactivity of individual arterial blood samples or by assuming a linear rise from [14C]tracer lag time after intraperitoneal [14C]IAP injection to the value measured in the blood sample from the frozen heart. Regional flow values calculated by the two methods differed by less than 11 % (not significant). This method allows the quantitative measurement of local CBF in anesthetized mice without any vessel catheterization and will make mutant mice a more powerful tool to elucidate the molecular mechanisms of brain injuries by combining flow studies with molecular-biological methods.
The quantitation of local cerebral blood flow (CBF) using the [14C]iodoantipyrine (IAP) method requires the determination of tissue [14C]radioactivity at the end of the experiment and the time course of arterial blood [14C]radioactivity during the experimental period (Reivich et al., 1969; Sakurada et al., 1978). Local brain [14C]radioactivities can be measured three-dimensionally at high resolution by quantitative autoradiography (Reivich et al., 1969; Kennedy et al., 1972; Hosokawa et al., 1977; Sakurada et al., 1978) and catheterization of blood vessels for intravenous [14C]IAP infusion and repeated arterial blood sampling is practicable in large animals. In small rodents such as mice, however, the cannulation of several blood vessels is necessary, i.e., arterial lines for blood sampling and for blood pressure monitoring during sampling and a venous line for tracer infusion, which demands precise and long-lasting microsurgery under general anesthesia, are required. [14C]radioactivities in arterial blood samples also have to be corrected appropriately for the lag and dead space washout when long and small arterial catheters are used (Jay et al., 1988). Moreover, loss of blood during repeated sampling may lead to hemorrhagic hypotension and derange the physiologic status of the animal. To avoid such complications, we modified and validated the [14C]IAP method in such a way that local CBF in the mouse can be determined quantitatively by combining intraperitoneal tracer application with a single blood sampling from the heart at the end of the experiment.
MATERIALS AND METHODS
Experiments were carried out according to the National Institutes of Health guidelines for the care and use of laboratory animals and approved by the local authorities. Male SV129 mice weighing between 27 and 30 g were used (Harlan Winkelman, Borchen, Germany). Animals were housed under diurnal lighting conditions and allowed access to food and water ad libitum before the experiment.
Anesthesia was induced with 1.5%, and maintained with 1.0% halothane in 30% O2 and the remaining 70% N2O which was delivered via face mask. Throughout the experiment, rectal temperature was kept between 36.5 and 37.0°C using a feedback-controlled heating system (YSI, Yellow Springs, OH, U.S.A.). Laser-Doppler flowmetry was used to monitor possible changes in cortical perfusion during arterial blood sampling. The tip of the fiberoptic probe (PF2B; Perimed, Stockholm, Sweden) was fixed on the intact skull over the brain region supplied by the left middle cerebral artery (2 mm posterior and 6 mm lateral to the bregma). Laser-Doppler flowmetry values were expressed as percentages of baseline before the tracer injection.
The left common carotid artery and the left femoral artery were cannulated with polyethylene tubing (internal diameter, 0.29 mm, Portex, England). The catheter in the left common carotid artery was shortened to 3 cm and used for arterial blood sampling after tracer application. Arterial blood pressure was recorded from the left femoral artery throughout the experimental period. Arterial P
A total of 0.15 μCi 4-iodo-N-methyl-[14C]antipyrine (IAP) (specific activity: 40 to 60 mCi/mmol; Biotrend Chemicals, Cologne, Germany) dissolved in 0.15 mL 0.9% saline was injected intraperitoneally in the anesthetized animal, and arterial blood samples weighing between 10 and 20 mg were collected on preweighed filter paper pieces at 30, 60, 90, 105, and 120 seconds after tracer injection in animals used for the calculation of blood flow (n = 6). Experiments were terminated by immersing the animal in liquid nitrogen immediately after the last blood sampling, and the weight of arterial blood probes was determined. In a separate group of halothane-anesthetized animals (n = 5), the time lag of [14C]tracer appearance in arterial blood was examined after intraperitoneal [14C] IAP tracer injection. For this purpose, arterial blood samples were collected frequently during the first 30 seconds after intraperitoneal [14C]IAP injection. [14C]radioactivities of the various arterial blood samples were measured in a liquid scintillation counter with internal standard calibration (Wallac 1410, Pharmacia, Freiburg, Germany). An assumed gravity of 1.04 g/mL was used to calculate the volume of the blood sample.
Brains were removed in a cold box at −20°C, and blood from the frozen heart was sampled to obtain the final arterial blood [14C]radioactivity. Twenty-micrometer coronal cryostat sections were prepared at −20°C, dried on a hot plate, and exposed for 10 days to X-ray film (Hyperfilm, Kodak, Rochester, NY, U.S.A.) with calibrated [14C]standards. [14C]autoradiograms and [14C]standards were digitized with a CCD camera connected to an image processing system (ImageMG, NIH, Bethesda, MD, U.S.A.), and local [14C]radioactivity was determined by quantitative autoradiography.
Because catheter smearing of [14C]radioactivity can be neglected in the present series of experiments (see Discussion), local CBF in the various brain structures was calculated according to the algorithm described by Sakurada et al. (Sakurada et al., 1978) using the equation
where Ki equal to (fi m) / (λi V). λi Ki can be rearranged to Fi with the IAP diffusion coefficient m equal to 1; Fi is the flow rate fi (milliliters per minute) divided by the tissue weight V (grams); and λi is the tissue:blood partition coefficient for iodoantipyrine equal to 0.8 which cancels. Therefore, the ratio of local tissue radioactivity C*i(T) to Fi equals the flow-related convolution integral in dependency of the time course of arterial blood radioactivities with
Convolution integrals for various blood flow rates were calculated from the two arterial time courses, i.e., from all arterial blood samples collected after intraperitoneal [14C]IAP injection or from heart blood sampling by assuming linearity of [14C]IAP radioactivity between the lag time of [14C]tracer appearance after intraperitoneal [14C]IAP injection and the time of termination of the experiment.
Values are provided as means ± SD. Statistical differences in the mean arterial blood pressure and laser-Doppler flowmetry values were tested by one-way analysis of variance (ANOVA) followed by Scheffé's ad hoc analysis. For other comparisons the Student paired t-test was applied. A P value of <0.05 was considered to indicate a statistical significance.
RESULTS
Arterial blood gases of animals (n = 6) used for cerebral blood flow calculations were within normal ranges, i.e., Pao2: 119 to 143 mm Hg, Paco2: 37.5 to 49.0 mm Hg, and pH: 7.26 to 7.44. Mean arterial blood pressure and laser-Doppler flowmetry values did not change significantly after intraperitoneal [14C]IAP injection indicating steady-state conditions of the animals during arterial blood sampling (Table 1).
Mean arterial blood pressure and laser-Doppler flow measurement during arterial blood sampling
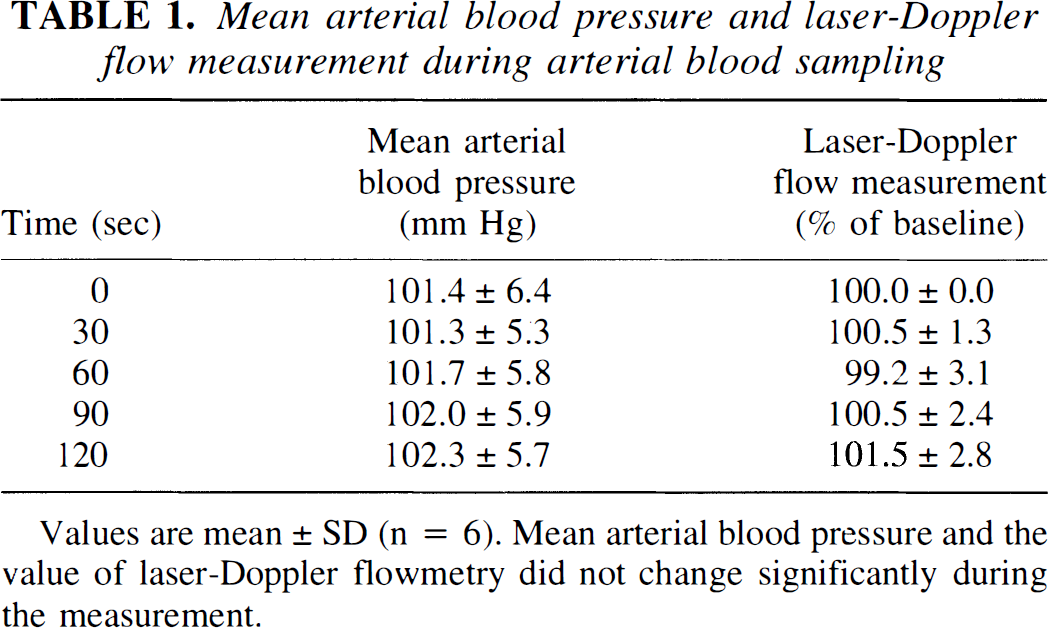
Values are mean ± SD (n = 6). Mean arterial blood pressure and the value of laser-Doppler flowmetry did not change significantly during the measurement.
The lag time of [14C]IAP appearance of arterial blood and the early time course of arterial [14C]radioactivity were determined by frequent blood sampling during 30 seconds after [14C]IAP administration. Intraperitoneal injection of [14C]IAP resulted in a significant linear rise in arterial [14C]radioactivity (y = 0.038× −0.12; r = 0.990; P < 0.001) yielding a lag time of 3.3 ± 0.6 seconds. In animals used for the measurement of CBF, a steady rise of arterial [14C]radioactivity was observed over a time period of 2 minutes after intraperitoneal injection of [14C]IAP (Fig. 1), and correlation analysis between the sampling time and blood [14C]radioactivity (normalized to the final value) revealed a highly significant linear relationship during the initial 120 seconds (y = 0.0085x; r = 0.978; P < 0.001).
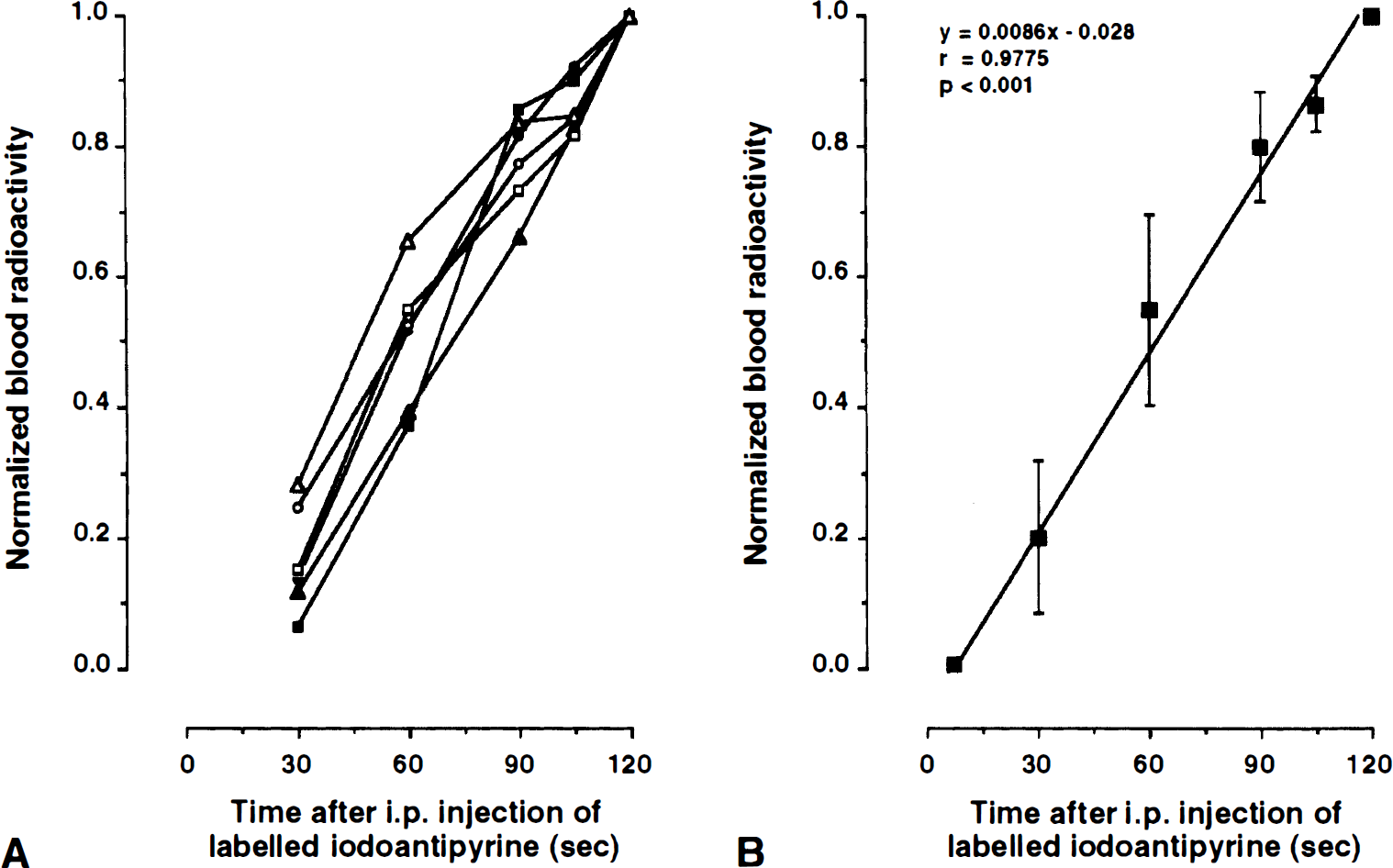
Time course of arterial [14C]radioactivities after intraperitoneal injection of [14C]-iodoantipyrine. [14C]radioactivity was normalized to the final arterial value at 2 minutes.
Because [14C]radioactivity of the final arterial blood sample (444 ± 264 nCi/mL) did not differ significantly from that taken from the heart after in situ freezing (454 ± 242 nCi/mL), i.e., the absolute difference was less than 3.3 ± 4.2%, a linear estimate of arterial [14C]radioactivity from the heart blood sample was assumed to provide a reliable measure of the arterial input function. For this purpose, the convolution integrals of arterial tracer radioactivities (see Eq. [2]) were calculated for a time range from tracer lag time, i.e., 3.3 seconds after [14C]IAP application, to the time of termination of the experiment with all collected arterial blood samples in each animal (final value Ca(T) = 100%) which were compared to those calculated from the final heart blood sample by assuming a linear rise from [14C]IAP lag time to experiment termination (Table 2). The absolute difference between the heart blood convolution integral value and that of arterial blood samples was ≈10% at low blood flow values (0.1 to 0.4 mL/g per minute) and ≈6% at higher flow rates (1.6 to 2 mL/g per minute).
Comparison of convolution integrals calculated from multiple arterial blood samples and the final heart blood sample
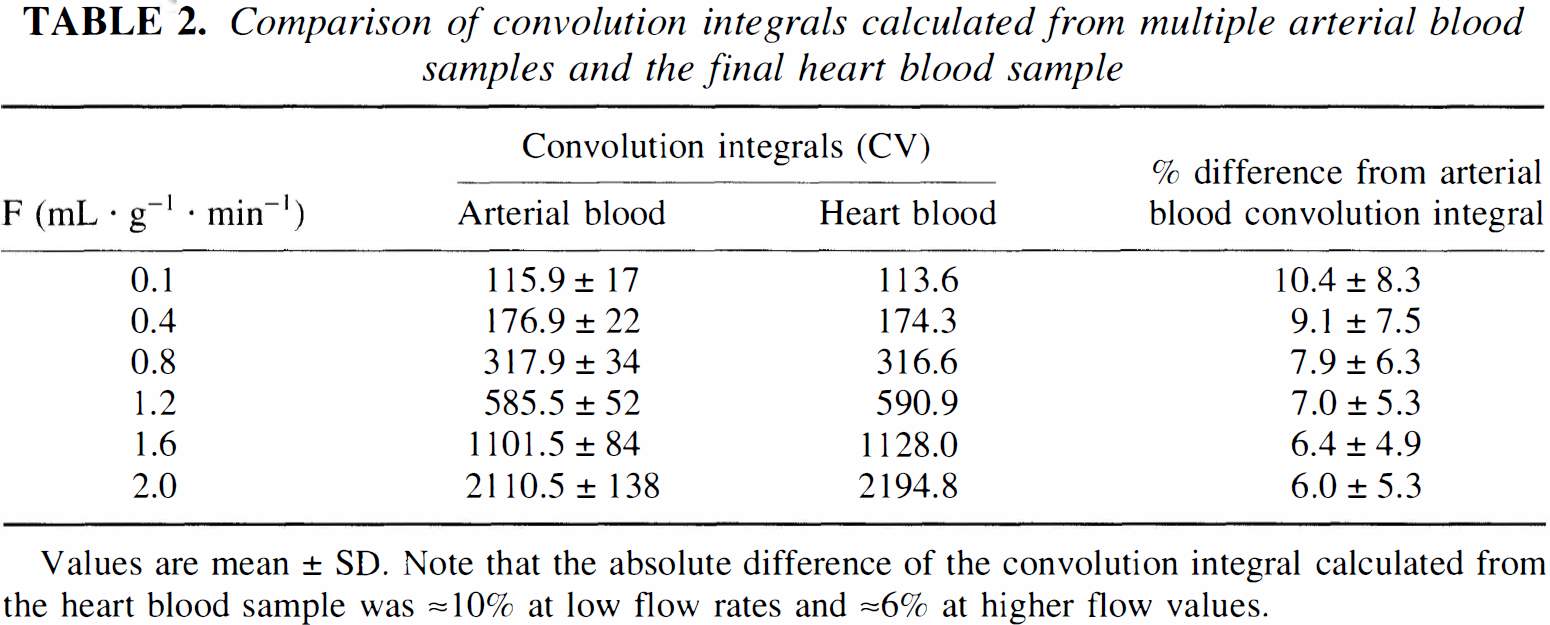
Values are mean ± SD. Note that the absolute difference of the convolution integral calculated from the heart blood sample was ≈10% at low flow rates and ≈6% at higher flow values.
Representative autoradiograms at the levels of caudate nucleus and hippocampus are shown in Fig. 2 suggesting that structural differences in local flow rates are preserved after a tracer circulation time of 2 minutes. Table 3 summarizes local cerebral blood flow rates in various brain structures calculated either with the input from all femoral artery blood probes or from the heart blood sample alone, revealing no statistical differences in regional perfusion when computed with the different tracer input integrals.
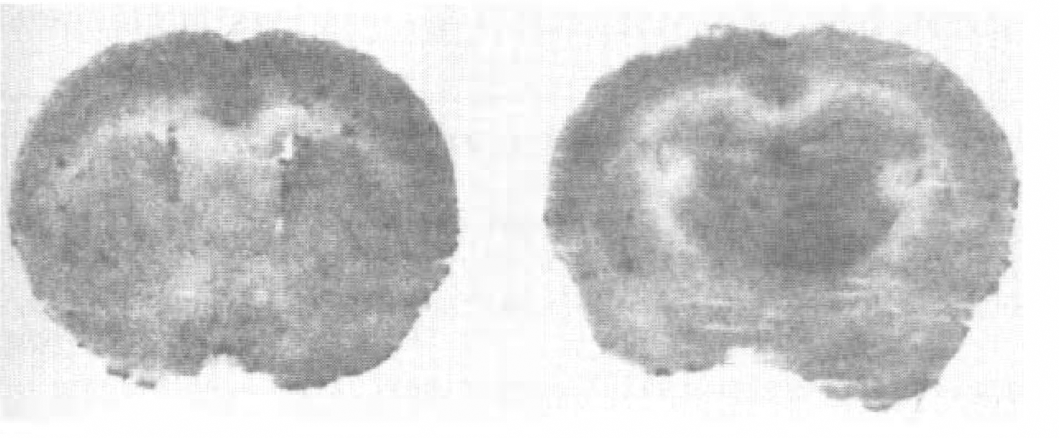
Autoradiograms of the SV129 mouse at the level of caudate-putamen left and hippocampus right. Optical densities of autoradiograms differ in various brain structures, reflecting the differences of local cerebral blood flow.
DISCUSSION
As shown in the present study, local CBF in halothane-anesthetized mice can be measured quantitatively without the need of vessel catheterization by immersing animals in liquid nitrogen 2 minutes after intraperitoneal injection of the labeled tracer. This is possible because the arterial blood radioactivity rises linearly after intraperitoneal injection of [14C]IAP with a short lag time of 3.3 seconds thus allowing a robust estimate of the arterial [14C]IAP radioactivities from a final heart blood sample. Convolution integrals calculated from the heart blood sample differed by less than 11% at low flow rates and less than 7% at higher flow values which introduced an average error in regional flow rates of ≈10%. Regional CBF values as determined with our modified IAP method revealed a flow range from 0.5 to 1.63 mL/g per minute which is comparable to that measured in awake mice (Jay et al., 1988). To our knowledge, there is only the report by Richards et al. (1987) on a modification of [14C]IAP method for rodents without repetitive blood sampling (Richards et al., 1987). The authors used coregistration of [14C]IAP and [99mTc]pertechnetate to determine the time course of [14C]IAP radioactivity in the arterial blood by the continuous external counting of [99mTc]pertechnetate over the chest. However, the authors did not provide any quantitative CBF values.
Two sources of error may compromise the quantitation of local CBF values, namely postmortem diffusion of [14C]IAP in brain tissue and inaccurately measured arterial [14C] IAP concentrations caused by catheter lag time and dead space washout. Diffusion of tissue [14C]IAP can occur when brains are removed from the skull which usually takes about 30 seconds or more. During that time, [14C]IAP in regions with high radioactivity diffuses into adjacent structures with lower levels of [14C]radioactivity in which local CBF will then be artificially overestimated (Patlak et al., 1984; Jay et al., 1988). In this study, [14C] IAP diffusion was minimized by immersing the animal into liquid nitrogen at the end of the experiment which preserves the structural heterogeneity of flow tracer in the autoradiograms. Measurement errors caused by the lag time of the sampled arterial blood and diffusion of the tracer within the catheter dead space should also be minimal. As estimated by Jay et al. (1988), the flow rate in the catheter should be at least 50 times the volume of the dead space per minute. Because we used catheters with a dead space volume of 2 μL, the flow rate in the common carotid artery, from which the arterial blood samples were collected, should then be larger 100 μL/min. In femoral artery catheters, a flow rate ranging from 127 to 258 μL/min was measured (Jay et al., 1988); it is reasonable to assume that such a flow rate is also present in the common carotid artery.
In the original publication on the IAP method (Reivich et al., 1969; Sakurada et al., 1978), an intravenous [14C]IAP ramp infusion over 1 minute was applied to delay or prevent the equilibrium between rapidly perfused tissues with the arterial blood during the period of measurement. As shown in our experiments, [14C]IAP blood radioactivity increased linearly after intraperitoneal [14C]IAP application for 2 minutes after a short lag time of 3.3 seconds and represents a perfect ramp input delivery of the tracer. We, therefore, have chosen a circulation time of the tracer for 2 minutes which results in sufficient brain radioactivity to obtain autoradiograms after intraperitoneal injection of 15 μCi [14C]IAP. It certainly is possible to reduce the period of tracer circulation and/or to improve the quality of the autoradiograms which then requires a larger amount of radioactivity.
In summary, our modified [14C]IAP method can be applied without vessel catheterization to the halothane-anesthetized mouse and reasonable local CBF values are obtained. With the increasing use of mutant mice in stroke research, this method provides the opportunity to combine molecular and/or biologic methods with flow studies to differentiate between hemodynamic and molecular mechanisms of brain injuries.
Comparison of local cerebral blood flow of mice calculated from multiple arterial blood samples and the final heart blood sample
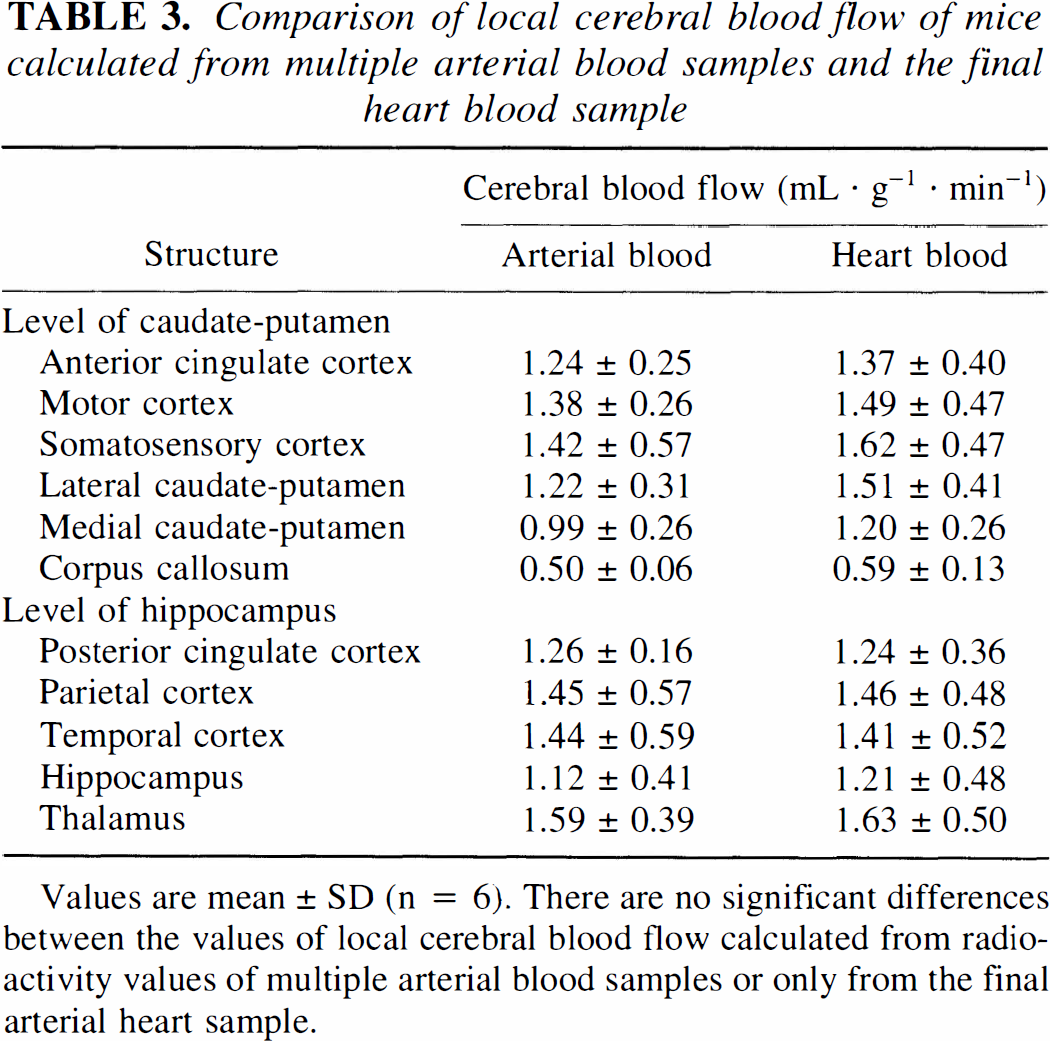
Values are mean ± SD (n = 6). There are no significant differences between the values of local cerebral blood flow calculated from radioactivity values of multiple arterial blood samples or only from the final arterial heart sample.