Abstract
Cerebrovascular dysregulation during hypotension occurs after fluid percussion brain injury (FPI) in the newborn pig owing to impaired K channel function. This study was designed to (1) determine the role of gender and K channel activation in adrenomedullin (ADM) cerebrovasodilation, (2) characterize the role of gender in the loss of hypotensive cerebrovasodilation after FPI, and (3) determine the role of gender in the ability of exogenous ADM to modulate hypotensive dysregulation after FPI. Lateral FPI (2 atm) was induced in newborn male and female newborn pigs (1 to 5 days old) equipped with a closed cranial window,
Introduction
Traumatic brain injury (TBI) is one of the major causes of morbidity, mortality, and pediatric intensive care unit admissions of children (Rodriguez, 1990). Clinically, it is necessary to understand the pathophysiologic responses in children after TBI to optimize mechanistically appropriate therapeutic modalities. Although the effects of TBI have been well described in adult animal models (Wei et al, 1980; McIntosh et al, 1989), few have investigated these effects in the newborn or the mechanisms underlying such changes. To reproduce some of the biomechanical aspects of TBI, fluid percussion brain injury (FPI) has been used in the adults of several species (McIntosh et al, 1989), but this technique also is thought to mimic some features of shaken impact in infants (Gennarelli, 1994). Fluid percussion brain injury produces an age-dependent reduction in cerebral blood flow, pial artery diameter, and impairment of responses to vasodilator stimuli in the pig, suggesting that the newborn is more cerebrohemodynamically sensitive to the effects of TBI (Armstead, 2005).
The membrane potential of vascular muscle is a major determinant of vascular tone and activity of potassium (K) channels is a major regulator of membrane potential (Nelson and Quayle, 1995). Activation or opening of these channels increases potassium efflux, thereby producing hyperpolarization of vascular muscle. Membrane hyperpolarization closes voltage-dependent calcium channels, causing relaxation of vascular muscle (Nelson, 1993). Although there are multiple subtypes of K channels, pharmacologic studies using activators and inhibitors have provided functional evidence that ATP sensitive (KATP) and calcium sensitive (Kca) channels regulate tone of cerebral vessels
Gender is an underappreciated contributor to outcome after pediatric TBI. For example, boys are disproportionately represented (Langlois et al, 2005). Cerebral autoregulation is often impaired after pediatric TBI (Vavilala et al, 2004) and impaired cerebral autoregulation early after severe TBI has been associated with poor 6-month outcome (Vavilala et al, 2006; Coates et al, 2005). Hypotension can lead to cerebral ischemia when cerebral autoregulation is impaired. Improving cerebral autoregulation and preventing cerebral ischemia during hypotension after pediatric TBI may significantly improve outcome (Tsuji et al, 1998). Hypotension-induced pial artery dilation results from KATP and Kca channel activation (Armstead, 1999a) and impaired cerebral autoregulation during hypotension after piglet FPI may result from impaired K channel function (Armstead, 2005).
Adrenomedullin is a vasoactive peptide structurally related to calcitonin gene-related peptide (CGRP) and is regarded as a member of the CGRP superfamily (Lang et al, 1997). Calcitonin gene-related peptide is an endogenous-selective KATP channel agonist in the piglet cerebral circulation (Armstead, 1997a) whose dilator activity is blunted after FPI (Armstead, 1997, 2005). Cerebrospinal fluid adrenomedullin concentration is increased in infants and children early after severe TBI but its contribution to the regulation of cerebrohemodynamics after TBI is as a function of gender is unknown (Robertson et al, 2001). We speculate that underexpression of CGRP-like peptides such as adrenomedullin may contribute to impaired cerebral autoregulation during hypotension after FPI.
This study was designed to (1) determine the role of gender and KATP and Kca channel activation in adrenomedullin cerebrovasodilation, (2) characterize the role of gender in the loss of hypotensive cerebrovasodilation after FPI, and (3) determine the role of gender in the ability of exogenous adrenonmedullin to modulate hypotensive dysregulation after FPI in the newborn pig.
Materials and methods
Newborn pigs (1 to 5 days old, 0.9 to 1.6 kg, respectively) of both sex were studied. All protocols were approved by the Institutional Animal Care and Use Committee of the University of Pennsylvania. Animals were sedated with isoflurane (1 to 2 minimum alveolar concentration (MAC)). Anesthesia was maintained with a-chloralose (30 to 40 mg/kg supplemented with 5 mg/kg per h intravenously). A catheter was inserted into a femoral artery to monitor blood pressure and to sample for blood gas tensions and pH. Drugs to maintain anesthesia were administered through a second catheter placed in a femoral vein. The trachea was cannulated, and the animals were ventilated with room air. A heating pad was used to maintain the animals at 37°C to 39°C, monitored rectally.
A cranial window was placed in the parietal skull of these anesthetized animals. This window consisted of three parts: a stainless-steel ring, a circular glass coverslip, and three ports consisting of 17-gauge hypodermic needles attached to three precut holes in the stainless-steel ring. For placement, the dura was cut and retracted over the cut bone edge. The cranial window was placed in the opening and cemented in place with dental acrylic. The volume under the window was filled with a solution, similar to CSF, of the following composition (in mmol/L): 3.0 KCl, 1.5 MgCl2, 1.5 CaCl2, 132 NaCl, 6.6 urea, 3.7 dextrose, and 24.6 NaHCO3. This artificial CSF was warmed to 37°C and had the following chemistry: pH 7.33, PaCO2 46 mm Hg, and PaO2 43 mm Hg, which was similar to that of endogenous CSF. Pial arterial vessel diameter was measured with a microscope, a camera, a video output screen, and a video microscaler.
Methods for brain FPI have been described previously (Wei et al, 1980). A device designed by the Medical College of Virginia was used. A small opening was made in the parietal skull contralateral to the cranial window. A metal shaft was sealed into the opening on top of intact dura. This shaft was connected to the transducer housing, which was in turn connected to the fluid percussion device. The device itself consisted of an acrylic plastic cylindrical reservoir 60 cm long, 4.5 cm in diameter, and 0.5 cm thick. One end of the device was connected to the trranducer housing, whereas the other end had an acrylic plastic piston mounted on O-rings. The exposed end of the piston was covered with a rubber pad. The entire system was filled with 0.9% saline. The percussion device was supported by two brackets mounted on a platform. Fluid percussion brain injury was induced by striking the piston with a 4.8 kg pendulum. The intensity of the injury (usually 1.8 to 2.0 atm with a constant duration of 19 to 23 ms) was controlled by varying the height from which the pendulum was allowed to fall. The pressure pulse of the injury was recorded on a storage oscilloscope triggered photoelectrically by the fall of the pendulum. The amplitude of the pressure pulse was used to determine the intensity of the injury.
Protocol
Two types of pial vessels, small arteries (resting diameter, 120 to 160 μm) and arterioles (resting diameter, 50 to 70 μm) were examined to determine whether segmental differences in the effects of FPI could be identified. Typically, 2 to 3mL of artificial CSF was flushed through the window over a 30 secs period, and excess CSF was allowed to run off through one of the needle ports. For sample collection, 300 μL of the total cranial window volume of 500 μL was collected by slowly infusing artificial CSF into one side of the window and allowing the CSF to drip freely into a collection tube on the opposite side.
Five experimental groups were studied for each sex (all
Adrenomedullin Analysis
Commercially available RIA Kits (Phoenix Pharmaceutical, Belmont, CA, USA) were used to quantity CSF adrenomedullin concentration. The CSF samples that were collected were rapidly frozen and stored at −20°C. The radioimmunoassay uses the simultaneous addition of sample, rabbit anti-adrenomedullin antibody, and the 125I-labeled derivative of adrenomedullin. After an overnight incubation at 4°C, free adrenomedullin was separated from adrenomedullin bound to antibody by the addition of goat anti-rabbit immunoglobulin G serum and normal rabbit serum. After centrifugation at 760
Statistical Analysis
Pial artery diameter, mean arterial blood pressure, and CSF adrenomedullin values were analyzed using analysis of variance for repeated measures. If the value was significant, the data were then analyzed by the Student's Newman—Keuls test. An a level of P 0.05 was considered significant in all statistical tests. Values are represented as mean ± s.e.m. of the absolute value or as percentage changes from control value.
Results
Role of Gender and KATP and Kca Channel Activation in Adrenomedullin-Induced Pial Artery Dilation
Topical adrenomedullin (10−9, 10−7 mol/L) elicited reproducible pial small artery (120 to 160 μm) and arteriole (50 to 70 μm) dilation in male and female piglets (data not shown). The onset of the vascular response was within 30 to 60 secs, peaked at 1 to 3 mins, and lasted 8 to 10 mins. Pial small artery dilation to topical adrenomedullin was significantly greater in female compared with male newborn pigs (Figure 1). Adrenomedullin-induced pial small artery dilation was blocked by glibenclamide (10−6 mol/L) in both male and female piglets, with no further effect observed when iberiotoxin (10−7 mol/L) was co-administered with the glibenclamide (Figure 1A). In contrast, adrenomedullininduced pial small artery dilation was unchanged by iberiotoxin in piglets of both genders, but blocked when glibenclamide was co-administered with iberiotoxin (Figure 1B). Similar observations were made in pial arterioles.
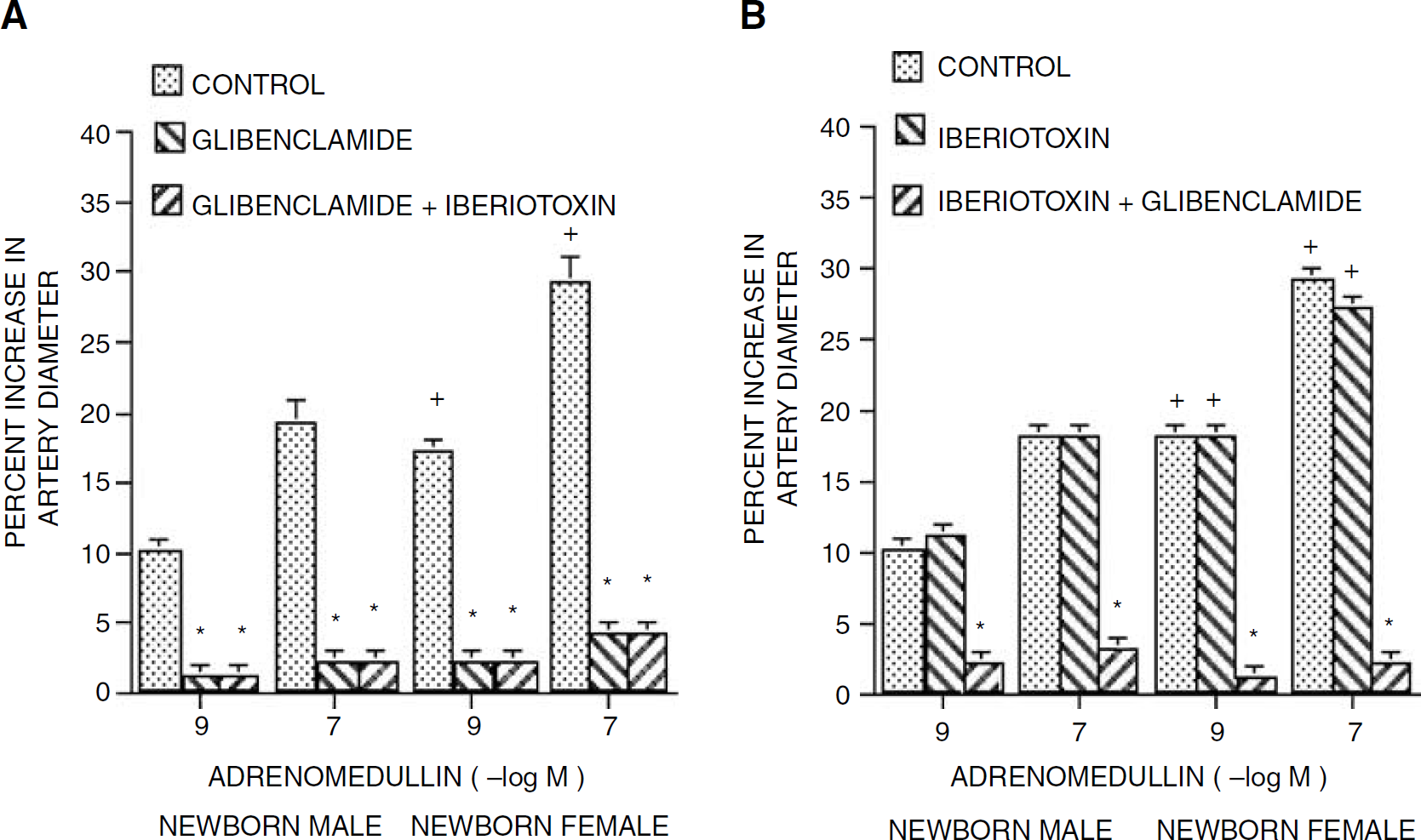
Influence of adrenomedullin (10−9, 10−7 mol/L) on pial artery diameter before (Control), after glibenclamide (10−6 mol/L) and after co-administered glibenclamide and iberiotoxin (10−7 mol/L) (
Gender Selective Effect of Fluid Percussion Brain Injury on Cerebrospinal Fluid Adrenomedullin Concentration
Cortical periarachnoid CSF adrenomedullin concentration was higher in the female compare with the male before FPI (Figure 2). Cerebrospinal fluid adrenomedullin concentration was elevated within 1 h of FPI in the female piglet, with concentrations continuing to increase at 4 h after insult (Figure 2). However, in the male piglet, CSF adrenomedullin concentration was unchanged at 1 and 4 h after FPI (Figure 2).
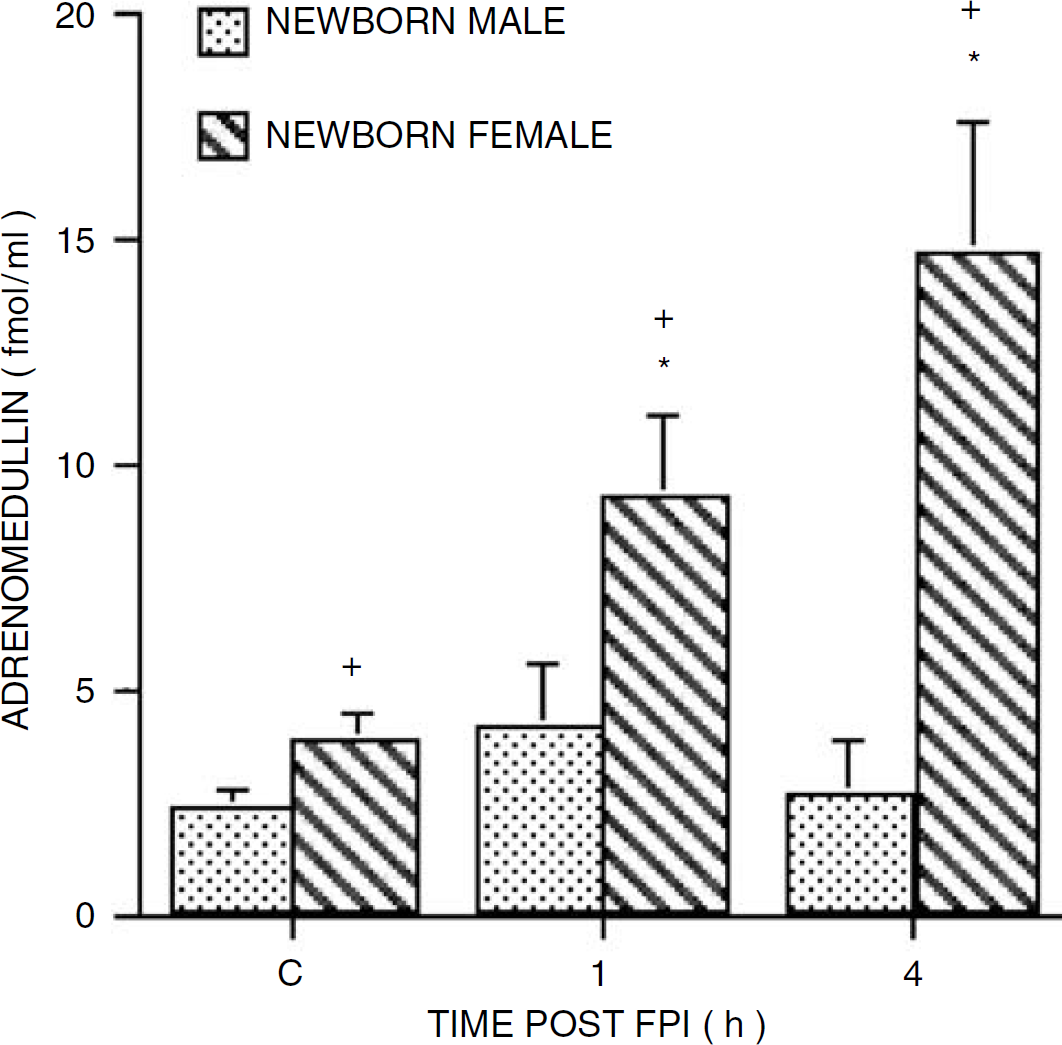
Influence of FPI on CSF adrenomedullin (fmol/mL) as a function of time after insult (h) in male and female newborn pigs,
Gender Selective Effect of Fluid Percussion Brain Injury on Pial Artery Dilation During Hypotension
Two levels of hypotension (moderate and severe) elicited reproducible pial small artery and arteriole dilation (data not shown). Mean arterial blood pressure was 62 ± 1, 47 ± 1, and 36 ± 1 mm Hg in the male, whereas it was 78 ± 2, 58 ± 2, and 43 ± 1 mm Hg in female for normotension, moderate, and severe hypotension, respectively. Nonetheless, on a percentage basis, the decreases in mean arterial blood pressure were equivalent as a function of gender (24 ± 1 and 42 ± 1% in the male; 26 ± 1 and 43 ± 1% in the female). Pial small artery dilation in response to moderate and severe hypotension was greater in female versus male piglets before FPI (Figure 3). After FPI, pial small artery dilation was blunted in both male and female piglets at 1 and 4 h after insult (Figure 3). However, the hypotensive dilator response was blunted to a significantly greater extent in male compared with female piglets at both 1 and 4 h after insult (Figure 3).
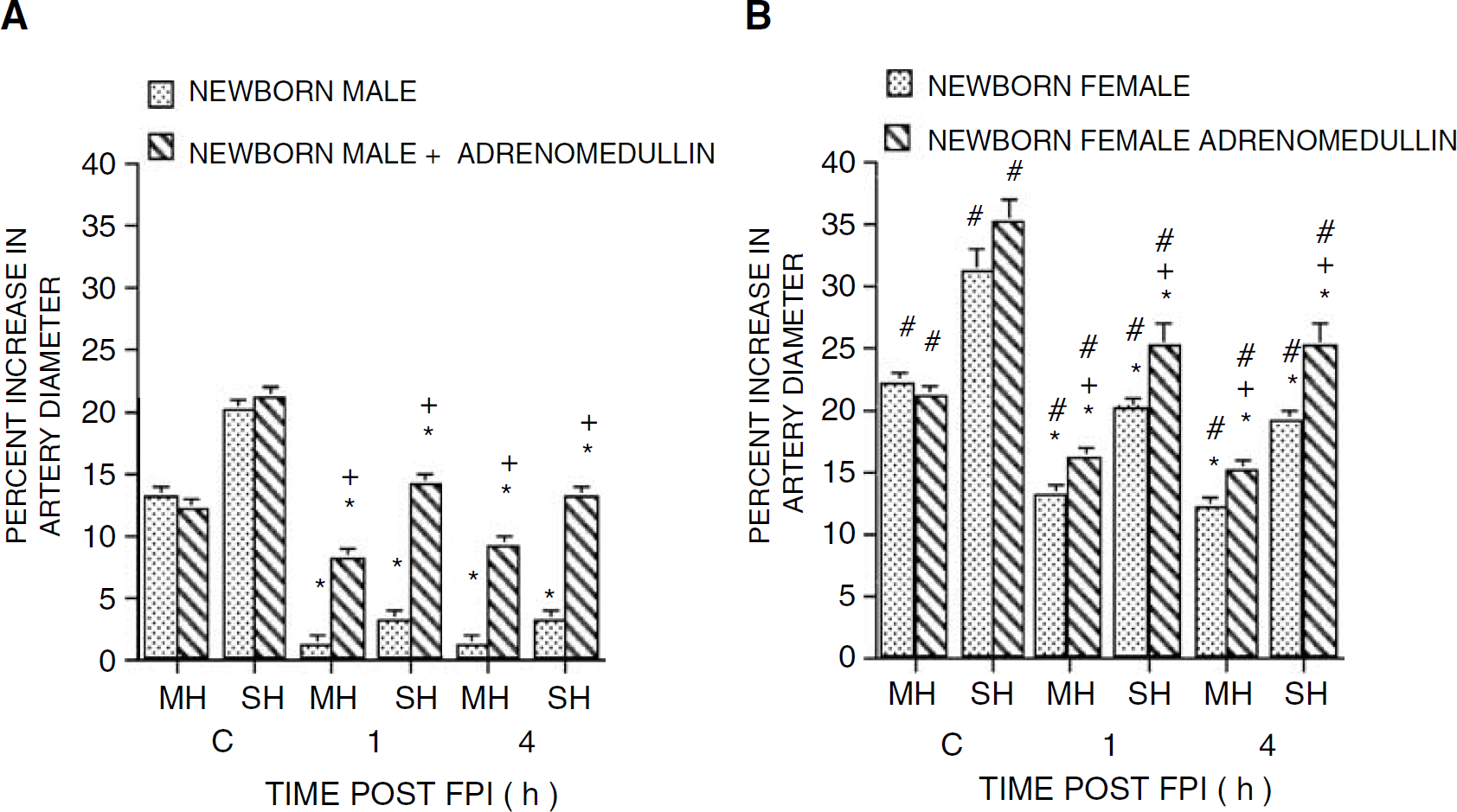
Influence of moderate and severe hypotension (MH, SH) on pial artery diameter before (control, C) and after FPI as a function of time in newborn male (
Gender Selective Effect of Adrenomedullin Pretreatment to Reduce Loss of Hypotensive Pial Artery Dilation after Fluid Percussion Brain Injury
Pretreatment with topical adrenomedullin (10−10 mol/L) 30 mins before injury, reduced the loss of hypotensive pial artery dilation in both male and female piglets after FPI (Figure 3). However, on a percentage basis, the degree of protection was significantly greater in male versus female piglets after FPI (Figure 3). Adrenomedullin (10−10 mol/L) by itself had no effect on pial small artery diameter (2 ± 1% dilation in the male; 3 ± 1% dilation in the female). Similar observations were made in the pial arteriole.
Gender Selective Effect of Fluid Percussion Brain Injury on Pial Artery Diameter
Fluid percussion brain injury produced greater pial small artery vasoconstriction in male compared with female piglets at 1 and 4 h after insult (Figure 4). Adrenomedullin pretreatment blunted FPI-induced pial small artery vasocontriction in both male and female piglets (Figure 4). On a percentage basis, adrenomedullin elicited greater protection in male versus female piglets after FPI. Similar observations were made in pial arterioles.
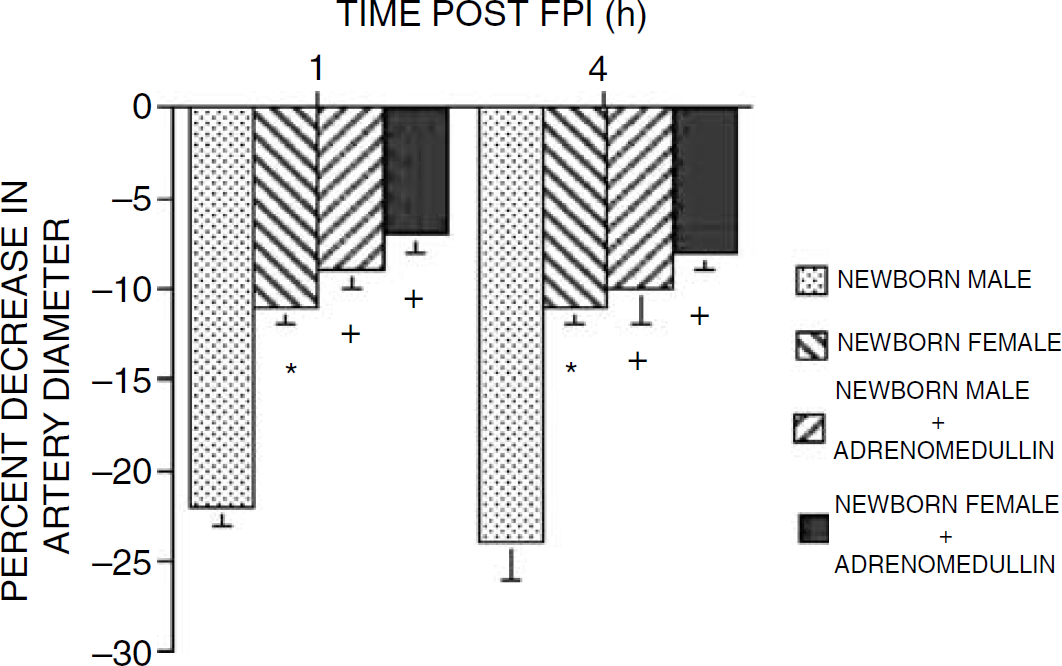
Influence of FPI (h) on pial artery diameter in untreated and adrenomedullin (10−10 mol/L) pretreated newborn male and female pigs,
Effect of Glibenclamide and Iberiotoxin on Pial Artery Diameter
Administration of glibenclamide and iberiotoxin had no effect on pial small artery or arteriole diameter.
Blood Chemistry
Blood chemistry values were collected before and after all experiments. There were no statistical differences between sham control, FPI, and FPI adrenomedullin-treated animals. For example, values were 7.48 ± 0.03, 35 ± 5, and 91 ± 9 versus 7.45 ± 0.03, 36 ± 6, and 88 ± 8 versus 7.46 ± 0.03, 36 ± 8 and 90 ± 10 mm Hg for pH, pCO2, and pO2, respectively in sham control, FPI, and FPI-adrenomedullin animals. The amplitude of the pressure pulse, used as an index of injury intensity, was equivalent in FPI-vehicle and FPI-adrenomedullin animals in both male and female pigs (1.9 ± 0.1 atm).
Discussion
There are several important new findings in this study. First, adrenomedullin produced gender-dependent pial artery dilation, with the vascular response being greater in the female compared with the male. Second, adrenomedullin-induced pial artery dilation in both sexes was blocked by the KATP channel antagonist glibenclamide but unchanged by the Kca channel antagonist iberiotoxin. These data affirm a role in adrenomedullin dilation for K channels in general in the human coronary artery (Terata et al, 2000) and the KATP channel in particular in the rat systemic vasculature (Sakai et al, 1998) and the rat pial artery (Lang et al, 1997). Importantly, a double crossover study design was used presently, which further supports conclusions regarding the relative roles of these two K channels in adrenomedullin-induced pial artery dilation. Since previous studies observed that glibenclamide blocked responses to a KATP channel agonist, whereas responses to a Kca channel agonist were unchanged, glibenclamide appears to be a selective and efficacious KATP channel antagonist in the piglet cerebral circulation (Armstead 1996, 1997b). Similar cross selectivity experiments show that iberiotoxin was a selective and efficacious Kca channel antagonist in the piglet as well. Additionally, both glibenclamide and iberiotoxin had no effect on pial artery diameter by themselves, indicating little tonic KATP or Kca channel contribution to resting vascular tone, similar to previously published observations in the piglet.
Other new results in this study show that FPI differentially increases CSF adrenomedullin concentration as a function of gender, in which cortical periarachnoid CSF adrenomedullin was increased in the female but unchanged in the male. These results extend those of Robertson et al (2001), who did not consider the role of gender in brain injury induced release of adrenomedullin in newborn children. Our results suggest the need for additional clinical study of this area. Equivalent injury insult level was induced in male and female piglets suggesting that unique signaling systems, as yet undefined, couple brain injury to adrenomedullin release in a gender-dependent manner. Male and female brains may thus have both similar, as in KATP channel mediation of adrenomedullin pial artery dilation, and uniquely different (vascular response to adrenomedullin; release of adrenomedullin by FPI) biochemical machinery. An alternative explanation for the gender differences in adrenomedullin vascular responsiveness could be that there is a reduced sensitivity to adrenomedullin/CGRP-like peptides, or that the K channel density and/or the channel gating may be different as a function of gender.
Additional results of this study support the notion that there are gender-dependent differences in the response to a vasoactive stimulus. For example, under noninjury conditions, the pial artery dilator response to hypotension was greater in female compared with male piglets. Mean arterial blood pressure during normotension was higher in the female compared with the male piglet. However, gender-dependent differences in the magnitude of the pial artery response to hypotension do not relate to differences in normotensive blood pressure because, on a percentage basis, the decreases in blood pressure during hypotension were equivalent in male and female pigs. Hypotensive pial artery dilation is dependent on activation of KATP and Kca channels in the piglet (Armstead, 1999a). Because CSF adrenomedullin concentration was greater in female compared with male piglets under noninjury conditions, these data suggest that, in part, the larger vascular response to hypotension in the female could result from a greater tonic activation of KATP channels by adrenomedullin. Nonetheless, other factors are probably involved as well because piglet hypotensive pial artery dilation has also been observed to be dependent on prostaglandins, nitric oxide, and carbon monoxide, the latter of which activates Kca channels to elicit dilation (Kanu et al, 2006; Xi et al, 2004). A limitation to this study design is that the role of adrenomedullin in hypotensive pial artery dilation was not directly tested through the use of an adrenomedullin antagonist such adrenomedullin 22 to 52.
Hypotensive pial artery dilation was impaired to a greater degree in male compared with female piglets after FPI. After FPI (Armstead, 1999b), results of previous studies have observed such hypotensive dilator impairment in the newborn pig. Our new results are the first to show a gender-dependent component to such impairment. Because adrenomedullin concentration was less in male compared with female piglets under non injury conditions and the concentration of this peptide did not increase after FPI in male like it did in female piglets, it was speculated that the gender-dependent greater inhibition of hypotensive pial dilation after insult could result from a suboptimal level in the male of CGRP-like peptides, such as adrenomedullin, which could tonically activate the KATP channel. Therefore, exogenous adrenomedullin was topically administered before FPI in another group of piglets to determine if this peptide could protect the impairment of the vascular response to hypotension after insult. Results show that exogenous adrenomedullin pretreatment did indeed reduce the impairment of hypotensive pial artery dilation after FPI in both male and female piglets, though the percentage protection was greater in the male versus the female. These results affirm previous observations that the prior administration of a KATP channel agonist could be protective in which another KATP agonist, croma-kalim, similarly reduced the impairment of dilation to the opioids methionine enkephalin, leucine enkephalin, and dynorphin after FPI in the piglet (Armstead, 1998). In the latter study, the Kca channel agonist NS 1619, however, had no protective effect, suggesting that this was not an epiphenomenon, but specifically related to the KATP channel. Importantly, in both cases, the concentration of the protectant agent used (10−10 mol/L for adrenomedullin in this study) had no vascular activity by itself. Therefore, physiological vascular action was not a contributor to the protective effect. These data suggest that modulation of distal signaling systems by prior treatment with an activator of the KATP channel may be involved. A limitation to the present experimental design related is that a pretreatment paradigm was used as a proof of principle concept. It is uncertain if a post-treatment paradigm would provide similar outcome, thereby limiting translational relevance. Nonetheless, there are settings in which there is a high probability of cerebral ischemia (cardiopulmonary bypass, for example), where prophylactic administration of adrenomedullin might be beneficial at saving central nervous system tissue/function.
Finally, FPI produced greater pial artery vasoconstriction in the male compared with the female, adding another gender-dependent facet to previously observed brain injury induced changes in cerebral hemodynamics (Armstead, 2005). Pretreatment with adrenomedullin blunted injury-induced pial artery vasoconstriction in both sexes, but such protection was greater in the male compared with the female. These data indicate that there are gender-dependent cerebrohemodynamic differences in the response to brain injury itself as well to vascular stimuli in the uninjured and injured state. It is speculated that signaling systems activated by a subthreshold concentration of adreneomedulllin buffer injury induced changes in cerebrohemodynmics in a gender-dependent manner.
Historically, the paper that revolutionized the field with regard to innate (prepubertal) differences in the gender response to brain injury is from the laboratory of Clark (Du et al, 2004). In those studies, it was observed that XY and XX neurons cultured separately were differentially susceptible to cytotoxic agents and treatments. In particular, XY neurons were more sensitive to nitrosative stress and excitotoxicity compared with XX neurons. Responses to therapies targeting nitrosative stress and excitotoxicity were also gender-dependent. Since nitrosative stress is believed to play important roles in central nervous system disease states such as cerebral ischemia and traumatic brain injury, these findings have important clinical ramifications. In this study, the gender-based differences in physiological responses and pathologic changes with FPI occur in prepubertal animals, affording a window into intrinsic gender cellular differences independent of sex hormones. The biological basis for such differences is unknown. Equally, little is known regarding gender-based differences in factors modulatory of K channel function. A number of experimental caveats arise from such consideration. For example, the amount of damage in response to mechanically equal FPI many not be equal as a function of gender in the developing brain. Oxidative stress mechanisms play a prominent role in pediatric vascular regulation and are well described for developmental traumatic brain injury (Bayir et al, 2002; Fan et al, 2003; Armstead, 2005). Important differences in glutathione levels many underlie some of the differences in neuronal cell death in male versus female neurons (Du et al, 2004). If identical amounts of glutamate induce substantially different amounts of neuronal cell death as a function of gender, then this could influence the vascular compensatory response after insult. Additionally, baseline differences in mean arterial blood pressure between genders were observed to be substantial in this study. Since these blood pressures exist in a narrow range close to the inflection point wherein autoregulation may be lost after FPI, the potential confounding role that gender-based differences in baseline blood pressure may play must be considered as another experimental design caveat. The age range of piglets investigated (1 to 5 days old) could offer a final experimental design caveat in that a similar age range of rats would yield significant maturity level differences in cerebrovascular response and outcome under physiologic and pathologic conditions. However, previous piglet studies suggest that this species matures slower, with the 1 to 5 day and 3 to 4-week-old piglet representing age groupings using distinctly different signal transduction systems as predominant mechanisms for cerebrovasodilation (Willis and Leffler, 1999, 2001; Zuckerman et al, 1996). For example, the prostaglandin system is prominent in the newborn, whereas the nitric oxide system is upregulated in importance with age and consistently observed as a contributory factor in the 3 to 4-week-old age period. Such studies minimize the potential role for gender-dependent differences being related to a perceived wide age range for investigation.
In conclusion, these data provide support for the concept that cerebrovascular responses which result from physiologic and pathologic stimuli are gender-dependent. In particular, these data show that hypotensive pial artery dilation is impaired after FPI in a gender-dependent manner. By unmasking a gender-dependent endogenous protectant, these data suggest novel gender-dependent approaches for clinical intervention in the treatment of perinatal traumatic brain injury.
