Abstract
Cytokines interleukin (IL)-6 and tumor necrosis factor (TNF)-α can play pathogenetic or protective roles in stroke. They are increased in the brain after experimental ischemia and in the CSF of patients with stroke. However, their presence in the periphery is still controversial. To determine the source and time-course of cytokines in blood of stroke patients, IL-6 and TNF-α release from blood cells and serum levels were determined in 40 patients on days 1 through 2, 4, 10, 30, and 90 after stroke. Twenty healthy age-matched volunteers were used as controls. IL-6 and TNF-α release from stimulated blood cells was increased in stroke patients, compared to controls. A peak response (+224%) was observed at day 4 for IL-6, while TNF-α release was largely and significantly increased (about three-fold compared to controls) from day 1 to 2 until day 90 after stroke. The increase in IL-6 release was significantly higher in ischemic, compared to hemorragic strokes, at days 1 and 4. Circulating IL-6 was increased at each time point. The ischemic processes in the CNS induces a long-lasting activation of IL-6 and TNF-α production in peripheral blood cells, which are a major source of serum cytokines after stroke.
Experimental and clinical evidence indicate that inflammatory-immunologic reactions play a pathogenetic role in ischemic brain damage (Grau et al., 1998; Kochanek and Hallenbeck, 1992). Early (within 15 minutes) induction of mRNA for interleukin (IL)-1 (Minami M et al., 1993; Buttini et al., 1994), TNF-α (Liu et al., 1994) and IL-6 (Wang et al., 1995) has been shown in neurons (tumor necrosis factor [TNF]-α and IL-6β, astrocytes and microglia (IL-1β) in experimental cerebral ischemia. At later times, infiltration of leukocytes and monocytes may be induced by cytokines released from cells within the CNS (Kim, 1996; Clark, 1997).
Despite the abundance of experimental data, very few observations have been made so far in acute stroke patients. Increased expression of TNF-α mRNA has been shown in neurons and glial cells of patients who died between 33 hours and 40 days after stroke (Tomimoto et al., 1996). Early increase (with a peak at 2 and 3 days) of IL-1β and IL-6 has been observed in CSF of acute stroke patients; in the same study IL-6 serum levels appeared increased up to 90 days after stroke (Tarkowski et al., 1995). However, data on peripheral cytokines after stroke are still controversial. In one study (Fassbender et al., 1994) IL-6 serum levels increased within the first hours after stroke, reached a plateau at 10 hours, and returned to baseline by day 7. No change in IL-1β and TNF-α level was observed in the same study. In another study (Beamer et al., 1995) plasma levels of IL-6 and IL-1 receptor antagonist were found to be increased at 4±2 days after stroke. In a more recent study (Kim et al., 1996), IL-6 serum concentration was reported to be increased up to day 7. In some but not all studies, TNF-α serum levels or soluble TNF receptor protein-1 p55 appeared increased after stroke (DeGraba et al., 1996; Elneihoum et al., 1996; Intiso et al., 1997).
Discrepancies of these results might be due to the short half-life of cytokines, to possible high concentrations at the site of release and much lower concentrations after their dilution in blood and to the different types of assay because bioassays measure biological activity of intact cytokines, while immunological assays also measure cytokine fragments or monomers without biological activity.
Moreover, in view of the existance of communication pathways linking the brain to the periphery (De Simoni, 1997), measuring circulating cytokines (in CSF and serum) does not definitely establish their source (central or peripheral) after stroke. To get a deeper insight into the pathogenetic role of cytokines in this condition we wished to determine the source and the time course of peripheral cytokines and to relate them to the type of stroke and to measures of severity and outcome. We obtained blood cells from acute stroke patients (including both ischemic and hemorragic) at various times (from the first day up to 3 months) and investigated IL-6 and TNF-α production after in vitro exposure to lipopolysaccharide (LPS) (ex vivo). Circulating serum cytokines were also investigated at the same time points and all values were correlated with brain lesion size, with stroke severity and outcome, and with peripheral inflammatory indexes.
SUBJECTS AND METHODS
Patients and controls
We enrolled 55 patients with acute stroke (within 24 hours after the onset) who were admitted to the Department of Neurology of the University of Milan at the San Gerardo Hospital, in Monza, Italy, from January 1996 through December 1997. The study was approved by the Ethic Committee of San Gerardo Hospital. Excluded from the study were patients with history of recent (within 3 months before admission) infection, concurrent major cardiac, renal, hepatic, autoimmune and cancerous diseases, recent (within 1 year) history of prior transient ischemic attacks, stroke or head trauma, and signs of acquired infection after admission. Patients taking immunosuppressive or anti-inflammatory drugs were also excluded. Ticlopidine or heparin were admitted for secondary stroke prevention. In preliminary experiments, we tested these drugs and found them not active on cytokine release or cytokine assays (data not shown).
All patients underwent complete physical and neurologic examination at entry and stroke severity was assessed by the National Institutes of Health Stroke Scale (Brott et al., 1989) at the onset of stroke and then at days 2, 4, 10, 30, and 90 after stroke. All patients were examined with computerized tomography of the brain within 24 hours and at 5 days after stroke. The volume of ischemia or hemorrhage was determined from standard computed tomography scans by calculating the volume of an ellipsoid containing the lesion (Pullicino et al., 1997) and also using the “Cavalieri Direct Estimator,” as previously described (Clatterbuck et al., 1997). Both measurements were corrected to in vivo dimensions using the analog scale found on the hard copy computed tomography image, had good interrater agreement and were highly correlated to each other (r = 0.99, P < .0001). Blood cell count, including monocytes and platelets, and analysis of erythrocyte sedimentation rate, C-reactive protein, fibrinogen, protein C and S, antiphospholipid antibodies were performed in all patients at entry and at 10 days after stroke.
Twenty healthy control subjects, without known stroke risk factors (blood donors of the San Gerardo Hospital), balanced for age with the stroke group, were also selected for analysis of cytokine release. Demographic and clinical characteristics of patients and controls are shown in Table 1. Because nine patients were lost for intercurrent infections and six patients died, 40 of 55 (73%) completed the study. Among the studied patients, 29 had ischemic and 11 had hemorragic brain lesions; their data were analyzed both together and as separate groups.
Demographic and clinical characteristics of patients and controls
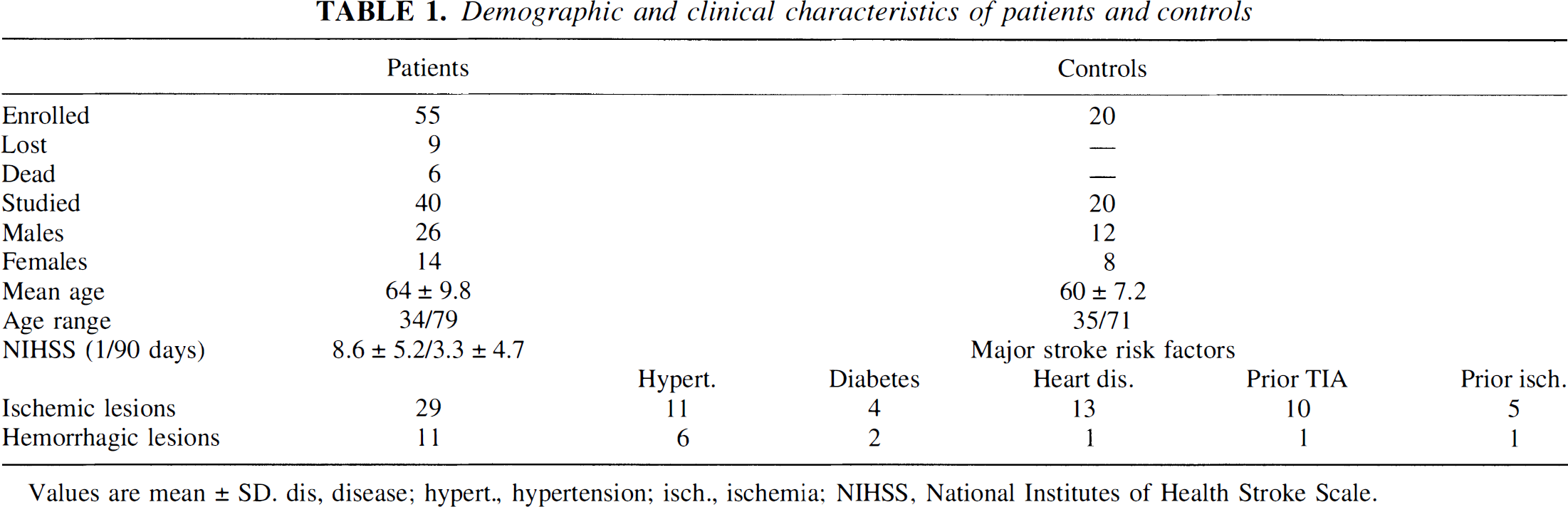
Values are mean ± SD. dis, disease; hypert., hypertension; isch., ischemia; NIHSS, National Institutes of Health Stroke Scale.
For cytokine measurements studies, patients' blood was serially drawn, after informed consent, through the antecubital vein within 48 hours and at days 4, 10, 30, and 90 from stroke onset. Seven milliliters were immediately used for blood cell stimulation studies, 3 mL of serum were also stored for analysis of cytokine levels.
Blood samples from controls were collected with a similar procedure at only one time point because preliminary experiments have shown that in healthy subjects cytokine release remains stable during serial venipuncture.
Whole blood stimulation
Experiments were performed as described by Fantuzzi et al. (1995) with minor modifications. Heparinized whole blood obtained from stroke patients and healthy controls was immediately processed. Blood was diluted 1:2 (v/v) in RPMI 1640 medium (without bovine serum), plated in 96-well tissue culture plates (200μL/well), and incubated for 4 hours at 37°C and 5% CO2, in the presence of 0.01 nmol/L endotoxin (μLPS: phenol-extracted preparation from Escherichia coli 055: B5 [Sigma] dissolved in sterile pyrogen-free saline) or the same volume of pyrogen-free saline. After incubation, whole blood was centrifuged and supernatants collected and stored at -20°C until assay.
Cytokine assays
For each patient, assays were performed when sample collection had been completed (day 90 from stroke). Eight patients and four controls were processed in the same assay and samples were run in triplicate.
Interleukin-6 bioassay. IL-6 was measured in cell supernatant as hybridoma growth factor using the 7TD1 cell line as previously described (De Simoni et al., 1990). Results are expressed as units per milliliter in comparison with a reference curve obtained in each experiment using recombinant human IL-6 (Immunex, Seattle, W A). Reference curves obtained were comparable in all experiments. One unit in the 7TD1 assay corresponded to 1 pg human recombinant IL-6. The sensitivity of the assay was 50 U/mL.
TNF-α bioassay. TNF-α was measured in cell supernatant by cytotoxicity on L929 cells in the presence of 0.001 μmol/L of actinomycin D as previously described (Terrazzino et al., 1997). TNF-α levels were calculated using recombinant human TNF-α (BASF/Knoll, Ludwigshafen, Germany; specific activity 107 U/mg) and expressed as nanograms per milliliter. The sensitivity of the bioassay was 5 pg/mL.
IL-6 and TNF-α immunoassays. For the quantitative measurement of IL-6 and TNF-α in serum we used both the bioassay and the Immulite Automated Immunoassay System (DPC, Cirrus, New Jersey, USA). With this method, a polystyrene bead solid phase (Immulite Test Unit) is coated with a monoclonal antibody specific for human TNF-α or IL-6, with no cross-reactivity to other cytokines. Sensitivity of the assay was 1.7 pg/mL for TNF-α and I pg/mL for IL-6.
Statistical analysis
The significance of cytokine production in patients compared to controls was analyzed by one-way analysis of variance (Dunnett's test). Difference between ischemic and hemorragic patient samples at single time points was evaluated by Student's t-test.
Statistic analysis of correlations between cytokine values and brain lesion volume, stroke severity, outcome and peripheral inflammatory indexes was performed with the Pearson's correlation test.
RESULTS
Figure 1 shows the time course of IL-6 release from blood cells of stroke patients. After stimulation, the release was significantly higher than that from controls' cells at all time points from days 1 to 2 after stroke until 1 month, and it was still 127% of controls after 3 months, although no longer statistically different. The highest release was observed at day 4 after stroke (224%, P < .01 versus controls), but at 30 days it was still about 160% of the controls. IL-6 was not detectable when blood cells were not exposed to LPS.
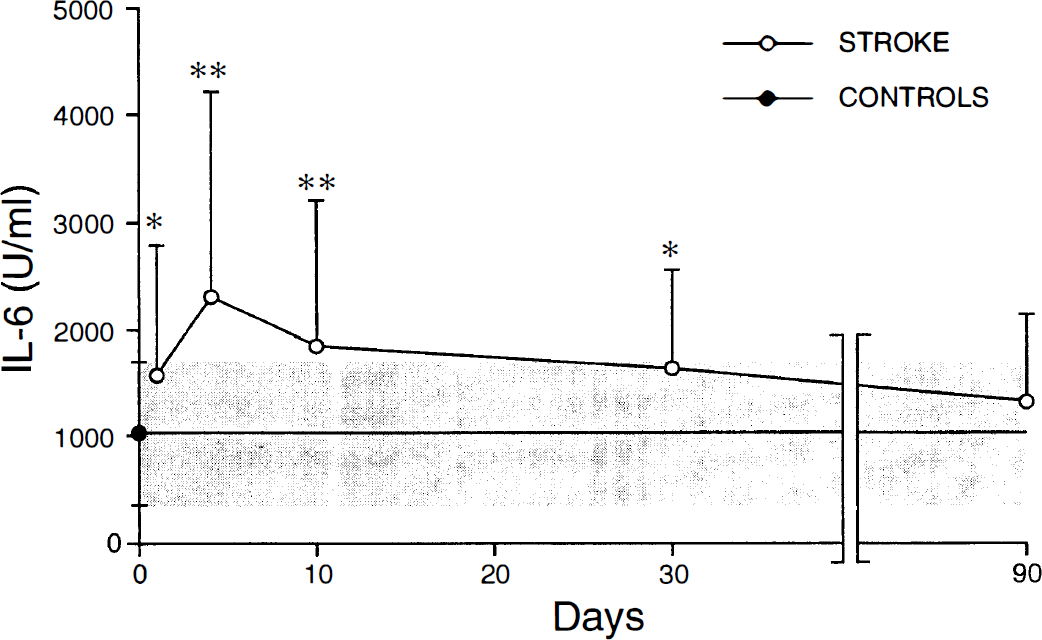
Interleukin-6 bioactivity in supernatants of blood cells stimulated with lipopolysaccharide (0.01 nmol/L at 37°C for 4 hours). Data are obtained from stroke patients (n = 40) and aged-matched healthy control subjects, n = 20) and are expressed as the mean ± SO. *P < .05, **P < .01, Dunnett's test.
TNF-α release, shown in Fig. 2, was significantly increased from days 1 to 2 up to 3 months after stroke. There was no peak response, the mean increase being about three-fold compared to controls from days 1 to 2 through day 30. At 3 months TNF-α values were still 226% of controls. As for IL-6, TNF-α was not detectable in nonstimulated supernatants.
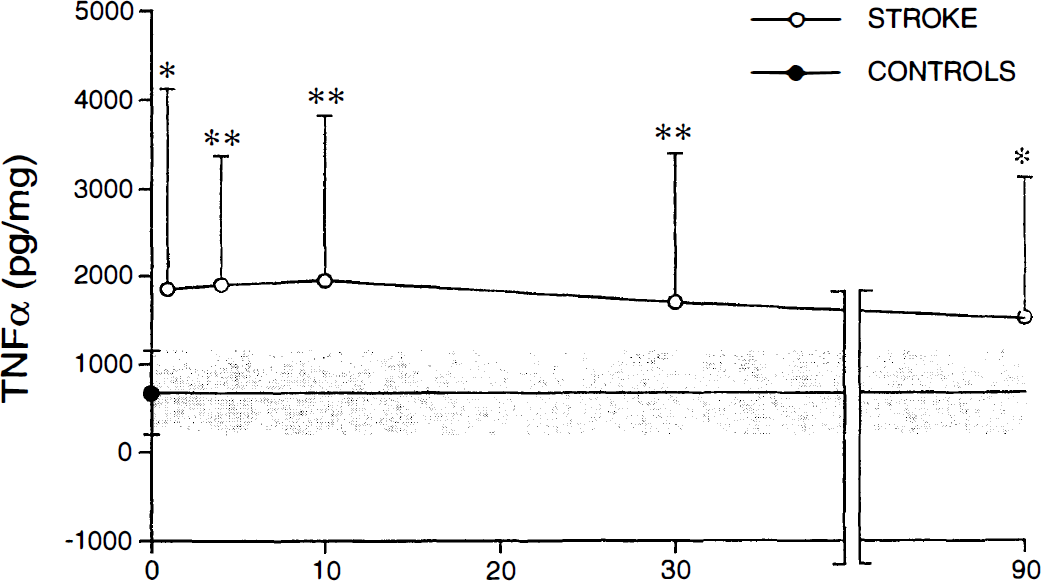
Tumor necrosis factor-α bioactivity in supernatants of blood cells stimulated with lipopolysaccharide. For other details see Fig. 1.
When ischemic and hemorragic strokes were analyzed separately, a difference in the time-course of IL-6 release became evident (Fig. 3). In fact, IL-6 increased earlier in ischemic compared to hemorragic patients, the difference being evident at days 1 to 2 and 4, and disappearing from day 10. Thus, the peak response appeared at day 4 in the ischemic and at day 10 in the hemorragic stroke. No difference could be shown in the time-course of TNF-α release in the two patient groups (data not shown).
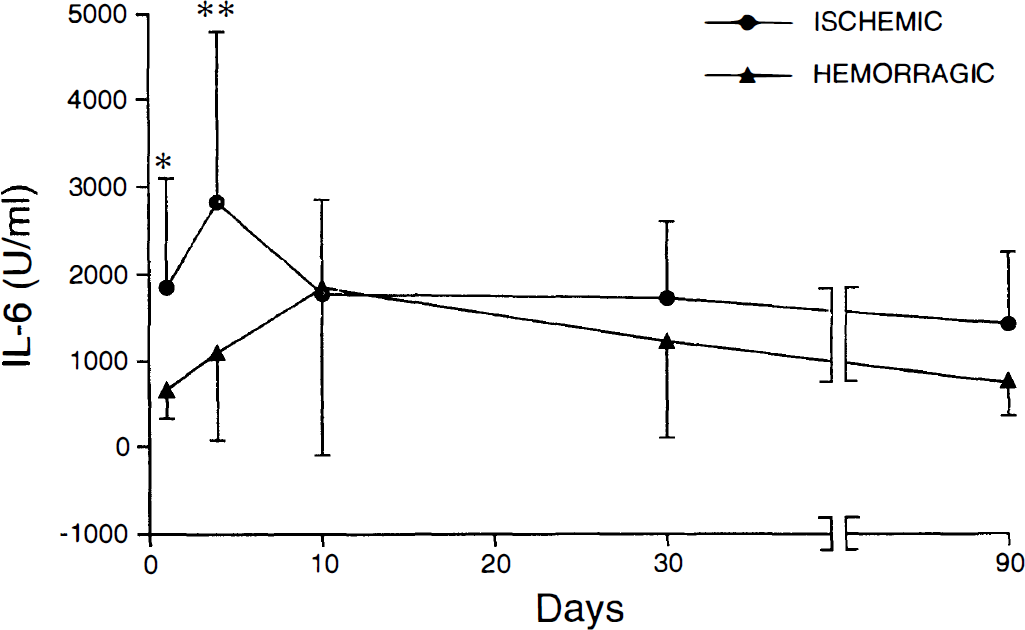
Interleukin-6 bioactivity in supernatants of blood cells stimulated with lipopolysaccharide (0.01 nmol/L at 37°C for 4 hours). Comparison between ischemic (n = 29) and hemorragic (n = 11) stroke patients. Data are expressed as the mean±SD. *P < .05, **P < .01 versus hemorragic patients at a given time point, Student's t-test.
Analysis of possible correlations between the increase of cytokine release and stroke severity at the onset (expressed by the National Institutes of Health Stroke Scale score), residual damage (expressed by National Institutes of Health Stroke Scale score at 3 months) or clinical improvement (measured as a difference of the two scores) did not reveal any significant correlation (data not shown). Moreover, no relation between cytokine increase and the volume of ischemic or hemorragic lesion was observed (data not shown).
Finally, cytokine release was neither related to peripheral indeces of inflammation such as erythrocyte sedimentation rate, C-reactive protein, fibrinogen, nor to the number of leukocytes or monocytes. Antiphospholipid antibodies have been observed in one hemorragic and three ischemic patients; the increase in cytokine release in these patients was similar to that of other patients.
Analysis of serum levels of circulating cytokines by bioassays did not show detectable levels neither in controls nor in stroke patients. On the contrary, immunological analysis by Immulite showed increased concentrations of IL-6 at all time points, with peaks at 4 and 10 days (Table 2). TNF-α serum levels were only slightly and not significantly increased at day 1 to 2, and were similar to controls at later times. No correlation has been found between the release from blood cells measured by bioassay and serum levels of cytokines measured by immunological assay (data not shown). Serum cytokine levels were also unrelated to stroke size and severity (data not shown).
Serum IL-6 and TNF-α concentrtions in stroke patients and controls measured by Immulite
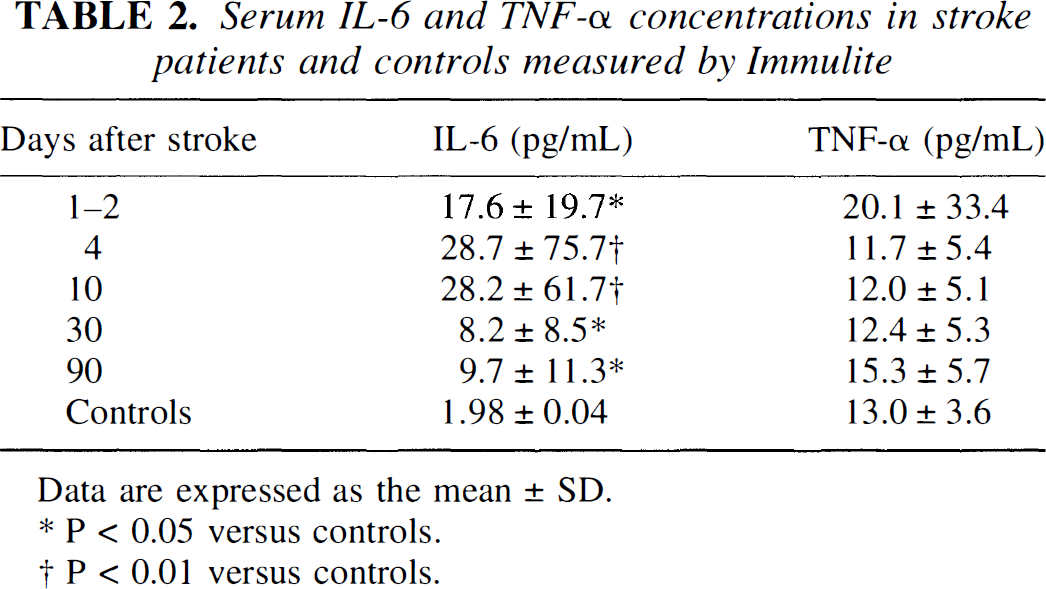
Data are expressed as the mean ± SD.
P < 0.05 versus controls.
p < 0.01 versus controls.
DISCUSSION
In this article we show for the first time that blood cells from patients with acute stroke (ex vivo) release larger amounts of IL-6 and TNF-α after exposure to LPS, a trigger of cytokine production.
This finding may answer some questions and methodologic problems recently raised in the literature. Our results suggest that activated peripheral blood cells should be a major source of cytokines described in various studies (Fassbender et al., 1994; Beamer et al., 1995; Kim et al., 1996; Intiso et al., 1997; Elneihoum et al., 1996; DeGraba et al., 1996) in plasma or serum of stroke patients; accordingly, cytokine leakage from infarcted brain or from CSF should not represent the main contributor to circulating cytokines. This is in line with a previous study showing no correlation between CSF and serum cytokine levels early after stroke, when they are higher in CSF than in serum (Tarkowski et al., 1995); in the same study the correlation appeared only from day 21 to day 90 after stroke, when their gradient indicated a blood to CSF flow (Tarkowski et al., 1995). Our data of protracted release of cytokines from peripheral blood cells favors this interpretation.
Moreover, we observed increased serum IL-6 levels by immunological assay, but not by bioassay, that yielded undetectable concentration for both cytokines. For IL-6, but not for TNFα, this could be due to the different sensitivity of the assays, the immunoassay being more sensitive than the 7TD1 bioassay. The different results obtained with the two assays could also indicate that most of the immunomaterial detected in serum is related to cytokine fragments or monomers without biological activity (Turtinen and Juran, 1998), and that release studies are more reliable than serum measurements.
The normal number of leukocytes and in particular of monocytes (the major source of blood cytokines) and the lack of correlation between monocyte counts and cytokine release indicate that the increased release is linked to blood cell activation and not to monocytosis.
After clearing these methodologic issues, we like to discuss the physiopathologic and clinical relevance of our finding. First of all, we observed that IL-6 release is activated in ischemic and hemorragic strokes with a different pattern. Thus, the different brain tissue reactions may play a specific role in the activation of peripheral blood cells. This is in line with the different expression of cytokine mRNA observed in brain tissue from patients who died at various times after cerebral ischemia or hemorrhage (Tomimoto et al., 1996). These observations indicate that specific brain modifications after stroke trigger peripheral immunologic activation.
The mechanism whereby a CNS lesion such as brain ischemia or hemorrhage activates peripheral blood cells is still unclear, although the existance of a signaling pathway from the brain to the periphery has been clearly shown (De Simoni et al., 1990; 1993; 1995; 1997). Experimental data show that an inflammatory stimulus (such as IL-1 or endotoxin) delivered in the brain efficiently induces inflammatory cytokine production in the periphery (De Simoni et al., 1993; 1995). The present data indicate that a peripheral immune response is elicited by stroke and further underlines the brain-immune system communication pathway in this type of CNS lesion. Because we did not find a correlation between the lesion volume or stroke severity and cytokine release, it is possible that even small lesions may activate peripheral immune system.
On the other hand, it has been repeatedly shown that peripheral cytokines may affect CNS by several mechanisms (De Simoni, 1997). Moreover, leukocytes and monocytes recruited from the periphery have been shown in brain infarcts (Kochanek and Hallenbeck, 1992; Wang et al., 1993), and a direct passage of cytokines from peripheral blood to CNS has been proposed (Banks et al., 1994; Gutierrez et al., 1993). Thus, the possibility that peripherally produced cytokines may in turn affect the brain should also be considered.
The role of cytokines in stroke is intriguing and it is difficult to establish a role of each individual inflammatory cytokine in the pathogenesis of the ischemic process, given their diverse functions and the multiplicity of various cytokine activation. Several experimental studies suggest a role for IL-6 and TNF-α in the pathogenesis of CNS diseases and in neuronal damage, possible mechanisms being the inhibition of glutamate uptake and the facilitation of excitotoxicity (Ye and Sontheimer, 1996; Fine et al., 1996). On the other hand, both cytokines display also protective actions in the brain (Bruce et al., 1996) and possess neurotrophic activity in vitro, an effect which may be direct and/or mediated by neurotrophic factors (Hama et al., 1989; Gadient et al., 1990; Yamada and Hatanaka, 1994; Kossmann et al., 1996).
In line with these neurotrophic activities, our demonstration of a protracted activation of blood cells after acute stroke raises the possibility that cytokines released from these cells are involved not only in the mechanisms of acute reaction to brain infarct, but also in the mechanisms of plasticity and repair. Recently, the importance of cytokines and growth factors as signaling mechanisms not only in stroke recovery, but also as protection from further insults (ischemic tolerance or preconditioning) has been underscored (Chen and Simon, 1997; Hallenbeck, 1997; Mattson, 1997).
We have interpreted the increase in TNF-α levels observed throughout the study as a consequence of the stroke event; however, data in the literature suggest that an alternative explanation should also be considered. Experimental data show that genetically stroke prone/hypertensive rats produce more TNF-α in response to a provocative dose of LPS intravenous than control animals free of these risk factors (Hallenbeck et al., 1991). Thus, high TNF-α levels in stroke patients could be the result of a greater susceptibility of blood cells to inflammatory stimuli present before the stroke event. The data open the possibility that genetic differences and/or stroke risk factors may contribute to the marked and long-lasting TNF-α production observed. Persistent inflammatory response has been recently shown in stroke survivors, associated with increased risk for recurrent vascular events (Beamer et al., 1998).
Finally, our findings show the possibility to analyze the peripheral immunologic activation in patients with brain lesions such as stroke, with the aim of investigating their involvement in the mechanisms of stroke damage and repair.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Drs. Ada De Luigi (Mario Negri Institute) and Elisabetta Raggi (“E. Medea” Institute) for their contributions to part of this study.
