Abstract
Alteration of sarcolemmal permeability was evaluated in the cerebral artery after subarachnoid hemorrhage. Significance of membrane dysfunction in the pathogenesis of chronic spasm and contribution of apoptosis were investigated in a canine model. Permeability of the smooth muscle cell (SMC) membrane was assessed by double staining with a hydrophilic (ethidium bromide [EB]) and a lipophilic (Hoechst 33342) DNA-binding dye. Quantitative observations were made with a ultraviolet-fluorescence microscope and a ultraviolet-laser confocal microscope. Occurrence of apoptosis was studied using electrophoresis and TUNEL method. In the normal arteries, nuclei of SMC were stained with Hoechst 33342 but not with EB. In the spastic arteries, SMC in the inner layer of the tunica media were stained with EB. The incidence of EB-positive cells reached maximum on day 7 (45 ± 19%) and decreased in 2 to 4 weeks (13 ± 5.2% and 5.0 ± 2.1%, respectively), in parallel with amelioration of spasm. Electron and light microscopic observations revealed increased density of SMC cytoplasm with widening of the extracellular space. Necrosis was not evident. Apoptosis was not detected by the two methods. These results demonstrate that an augmentation in sarcolemmal permeability takes place during the course of chronic vasospasm and suggest its close correlation to pathogenesis.
Pathogenesis of cerebral arterial spasm after subarachnoid hemorrhage (SAH) has not been fully elucidated despite the clinical and experimental efforts in the past. An integrative theory remains to be provided for the genesis of prolonged arterial narrowing after SAH. The presence of blood clot encasing the cerebral arteries results in constriction of the cerebral arteries, which starts 3 to 5 days after the ictus and lasts for a few weeks (Saito et at, 1977; Yoshimoto et al., 1993; Kim, 1998a) but the exact mechanism is not clear. The chronic cerebral vasospasm is resistant to conventional doses of vasodilatory agents, and despite recent advances in treatment modalities such as use of thrombolytic agents, calcium antagonists, and percutaneous angioplasty, it remains to be a serious problem in neurological and neurosurgical practices (Kim, 1998b).
Numerous studies have been done to study alterations in various properties of the cerebral artery and identify the cause of the vasoconstriction (Kim, 1998a, b ). Changes of the arterial wall, such as alterations in pharmacologic responsiveness (Kim et al., 1988; Nakagomi et al., 1988; Kanamaru et al., 1987), the mechanical properties (Bevan et al., 1987; Kim et al., 1989), immunologic reactions (Peterson et al., 1990), and morphologic changes have been described.
Our previous studies demonstrated that the cerebral artery was in a condition of metabolic failure during chronic vasospasm; contents of high-energy phosphates in the arterial wall were diminished (Kim et al., 1992), and time course of the reduction in ATP, creatine phosphate and GTP was closely correlated with development of angiographic vasospasm (Yoshimoto et al., 1993). Production of cyclic GMP was impaired in the spastic cerebral artery (Kim et al., 1992a). Cyclic GMP is a second messenger playing a pivotal role in vasodilation. Endothelium-dependent relaxations, mediated by the cyclic nucleotide (Furchgott et al., 1989), were impaired in the spastic cerebral arteries (Kim et al., 1988; Nakagomi et al., 1988). However, release of endothelium-dependent relaxing factor from the intima remained unchanged when measured with a bioassay system (Kim et al., 1989a). Mechanical properties of the spastic arterial wall were significantly altered, with a reduced passive compliance and a diminished maximal contractility (Bevan et al., 1987; Kim et al. 1989b).
Recently, we have demonstrated that Ca2+ regulation is impaired in the smooth muscle of the spastic cerebral arteries (Kim et al., 1996). The rise in intracellular calcium concentration ([Ca2+]i] in response to the increase in extracellular calcium concentration ([Ca2+]e) was larger in the spastic arteries. In the normal control arteries, treatment with ionomycin, an ionophore, resulted in an alteration of the [Ca2+]e-[Ca2+]i curve analogous to that observed during spasm. Treatment of the spastic arteries with a calcium channel blocker reversed the changes only partially. These results, together with the previous findings of the cellular derangement in metabolism, suggest that the membrane integrity of smooth muscle may be compromised.
Several histologic studies of the spastic cerebral arteries have been done but not with specific attention to the alterations of the sarcolemma. Several clinical and experimental studies have described smooth muscle necrosis as the characteristic feature (Crompton, 1964; Fein et al., 1974; Hughes and Schianchi, 1978; Tanabe et al., 1978) along with other changes such as subendothelial proliferation and endothelial vacuolization. However, there are studies reporting absence of muscle necrosis both in human vasospasm and experimental chronic vasospasm (Conway and McDonald, 1972; Tani et al., 1978; Mayberg et al., 1978; Eldevik et al., 1981; Pickard et al., 1985). This major controversy has not been solved, and the significance of transformations of smooth muscle in pathogenesis of cerebral vasospasm remains to be clarified.
In this study, we used a novel method of double staining with hydrophilic and lipophilic DNA-binding dyes (Daly et al., 1992) to evaluate alterations in the sarcolemmal permeability during chronic vasospasm. Using fluorescent and confocal microscopes, we studied the spatial and temporal distribution of the changes quantitatively and correlated this with observations by conventional light and electron microscopic study. In addition, possible contribution of apoptosis in the pathologic alterations was investigated using histochemical and electrophoretic methods.
MATERIALS AND METHODS
Animal model
The “double hemorrhage” canine model of SAH was used in the current study (Varsos et al., 1983). Mongrel dogs of either sex, weighing 8 to 13 kg, were randomly assigned to SAH groups (day 7 [n = 6], day 3, day 14, day 28 [n = 3]) and control group (n = 6). Under general anesthesia (pentobarbital, 20 mg/kg intravenously) and mechanical ventilation, vertebrobasilar angiography was performed to measure the size of the basilar artery. The arterial blood gas was monitored to rule out fluctuation of diameter of the cerebral artery due to variation in P
The animals in the SAH groups underwent angiography again on the designated days (days 3, 7, 14, and 28). The control group underwent the angiographic examination on days 0 and 7 without cisternal injections. For quantification of vasospasm, the angiograms were magnified and the images were recorded and digitized using a video tape recording system coupled to a personal computer/image analyzer. Ratio of the cross-sectional area to that of a baseline angiogram taken on day 0 was calculated in each group.
After the second angiogram, the animals were euthanized with an intravenous dose of sodium pentobarbital (50 mg/kg) followed by exsanguination. The brain and the cervical cord were dissected free. Using a stereomicroscope, the basilar artery was separated from the brain stem, and the subarachnoid clot was carefully removed. The artery was cut into rings (about 4 mm long) for DNA staining and fixation for electron microscopic studies. The care of the animals and procedures in the study complied with the Principles of Laboratory Animal Care and the Guide for the Care and the Use of Laboratory Animals (DHHS Publication No. [NIH] 8523, revised 1985) and had been approved by the Institutional Animal Care and Use Committee.
Staining with DNA-binding dyes
Fluorescent nuclear dyes Hoechst 33342 (bisbenzimide, H33342) and ethidium bromide (EB) were used (Daly et al., 1992). Both dyes intercalate with DNA and emit characteristic emission fluorescence on excitation. Hoechst 33342 permeates the normal plasma membrane, binds to DNA, and labels the nuclei with blue fluorescence on excitation. By contrast, EB, a hydrophilic dye, does not normally cross the plasma membrane. It permeates when integrity of the cell membrane is compromised and labels the nuclei with red fluorescence on excitation. Hoechst 33342 has an excitation spectrum from 320 to 380 nm with a peak at 343 nm. When bound with DNA, it has an emission spectrum of 400 to 540 nm with the maximum at 460 nm. The spectrum is shifted 40 nm to the left in comparison with the unbound state (Kapuscinski, 1990). Excitation spectrum of EB is biphasic; one band from 290 to 400 nm with a peak at 320 nm and the other from 450 to 550 nm with the maximum at 510 nm (Le Pecq and Paoletti, 1967). Its emission spectrum is from 580 to 690 nm with the maximum at 616 nm (Crissman and Steinkamp, 1990).
The isolated blood vessels were stained in a standard buffer solution (HEPES 10 mmol/L, NaHCO3 2.7 mmol/L, NaCl 153 mmol/L, and KCl 5.3 mmol/L) containing H33342 (60 µg/mL) for 30 minutes and then in that containing EB (30 µg/mL) for another 30 minutes at room temperature in a light-protected container. Samples taken from the normal control artery and the SAH day 7 group were treated in a paired fashion to safeguard against variability in the staining condition. After the staining process, the ring-shaped preparations were rinsed six times in the buffer solution, cut tangentially, opened, and placed on a slide glass. Observations with fluorescent microscope were made immediately after staining. When observing the smooth muscle cells (SMC) located close to the intima (periintimal portion of the tunica media), the arterial samples were placed with the intimal side facing the cover glass. For observation of those close to the adventitia (periadventitial portion), the preparations were placed with the adventitia in contact with the cover glass. Ultraviolet-grade slide and cover glasses were used.
Observation and image analysis
A Nikon Microphot-FXA microscope fitted with a high-pressure mercury light source was used with a “ultraviolet” filter cassette (excitation 330 to 380 nm with the maximum at 365 nm, dichroic mirror 400 nm, long-pass filter 420 nm). Oil-immersion objective lenses of 40× magnification were used. The numerical aperture was set to 1.3 for the 40× objective lens. The optics gives a limited focal depth of [3.70 to 3.75 µm for the numerical aperture of 1.3; [D (focal depth; µm) = n × λ/(NA)2 + n × 2000/(M × 7 × NA); n, refractive index (1.50 for the optics in use); M, overall magnification; λ, wave length (460 nm for H33, 510 nm for EB); NA, numerical aperture]. Therefore, it was possible to set a thin focal plane on a discrete layer of SMC (cellular diameter approximately 2 to 5 µm). Observation was focused on the periintimal, middle, and periadventitial layers of the tunica media (total thickness approximately 30 to 40 µm). The intima was distinguished with the endothelial cells lined along the axis of the artery. Smooth muscle cells in the periintimal layer of the tunica media were identified as the cells in close vicinity with the furrows of the elastica interna observed at the margin of the focus. Those in the periadventitial layer were identified as the cells close to the fibroblasts, which were seen at the margin of focus. Cell count analysis was performed in each of the layers.
Cells stained with EB were distinguished with red emission of the nuclei, and the cells were stained with H33342 with blue emission. Lipophilic dye H33342 stains nuclei of all normal cells. The incidence of the affected cells were quantitatively measured by counting the number of the cells stained with EB in a field. Cells with increased permeability were stained both by EB and H33342, but the red staining of EB stands out, and the nuclei of the cells appear red. Thus, the proportion of the affected cells was calculated as follows: red nuclei/(red nuclei + blue nuclei) × 100. The SMC were counted in eight different fields (320 × 480 µm) in each layer, and the incidence was calculated for the sample and then averaged for six (or three for SAH days 3 and 14) arteries obtained from each group of animals.
Confocal microscopy
Interactive laser cytometer ACAS570 (Meridian Instruments, Okemos, MI, U.S.A.) equipped with ultraviolet light source was used to perform microspectrofluorometric study and to take a confocal photomicrograph. The apparatus was equipped with a scanning stage, which moves in fixed increments (step size 0.5 µm, 180 steps in × and y axes, scanning speed 0.5 µm/s) under computer control (Wade et al., 1993).
At each point of the scanning, the laser beam of 350 nm was applied for 8 milliseconds, and the fluorescence was measured at the emission maxima for EB and H33342 (590 nm and 450 nm, respectively) simultaneously with two photomultiplier tube detectors. The dual wavelength measurement was done by a dichroic mirror (575 nm short pass) and with two photomultiplier tube detectors equipped with a 605-nm long-pass filter and a 485/45-nm band-pass filter. The two photomultiplier tube detectors were adjusted to obtain equal intensity of fluorescence with EB and H33342 when observing cells that were damaged by mechanical pressure and thus positively stained with EB as well as H33342. Pinhole size of 80 µm was chosen. The image obtained in the protocol consisted of 300 × 300 array of sample points.
Sample preparation for electron and light microscopy
The arteries were bathed in relaxing solution (EGTA 10 mmol/L, HEPES 10 mmol/L, NaHCO3 2.7 mmol/L, NaCl 153 mmol/L, KCl 5.3 mmol/L, and MgCl2 1 mmol/L) for 5 minutes. Then the arteries were perfused with 4% glutaraldehyde in phosphate-buffered saline (PBS; NaCl 150 mmol/L, phosphate buffer 20 mmol/L, pH 7.2 to 7.4), immersed in the solution for 1 hour, and washed for 5 minutes with PBS three times. The fixed samples were further postfixed with 1% osmium tetraoxide in PBS for 1 hour at 4°C and washed in PBS for 5 minutes three times. Block staining was done in 1% uranyl acetate for 25 minutes at room temperature. The specimen was dehydrated with ethanol of concentration of 75% (5 minutes twice), then 95% (5 minutes twice), and 100% (5 minutes twice and 30 minutes) at room temperature. The samples were further dehydrated in acetone (10 minutes three times) and kept in the mixture of 50% acetone and 50% embedding material overnight at 4°C. Finally, the materials were embedded and kept at 60°C overnight for polymerization.
The block was sectioned several microns thick by an ultramicrotome and stained with 1% toluidine blue for light microscopic observations. For electron microscopic study, the ultra-thin sections (50 to 80 nm thick) were stained with lead and observed under JEOL 1200 EX electron microscope.
Observation in the longitudinal section (parallel to the vessel axis) allowed best comparison of the morphologic alterations in the cross-section of the smooth muscle.
Data analysis
Data are expressed as the mean ± SD. For statistical comparisons, analysis of variance (ANOVA) was used. In all cases, P values less than 0.05 were considered to be statistically significant. The number of animals in each set of data is indicated by n.
RESULTS
Angiography: induction and time course of vasospasm
Cross-sectional area of the basilar artery, calculated as the ratio to that of a baseline angiogram taken on day 0, was 100.4 ± 4.3% (mean ± SD) in the control group. In the SAH day 3, 7, 14, and 28 groups, the ratio of arterial cross-section was 66.4 ± 32.3%, 26.3 ± 13.3%, 46.1 ± 19.1%, and 95.2 ± 17.4%, respectively (Fig. 1). The differences were significant each between the control and day 3 groups (P < 0.025, ANOVA), the day 3 and 7 groups (P < 0.01), the day 7 and 14 groups (P < 0.05), and day 14 and 28 groups (P < 0.005).
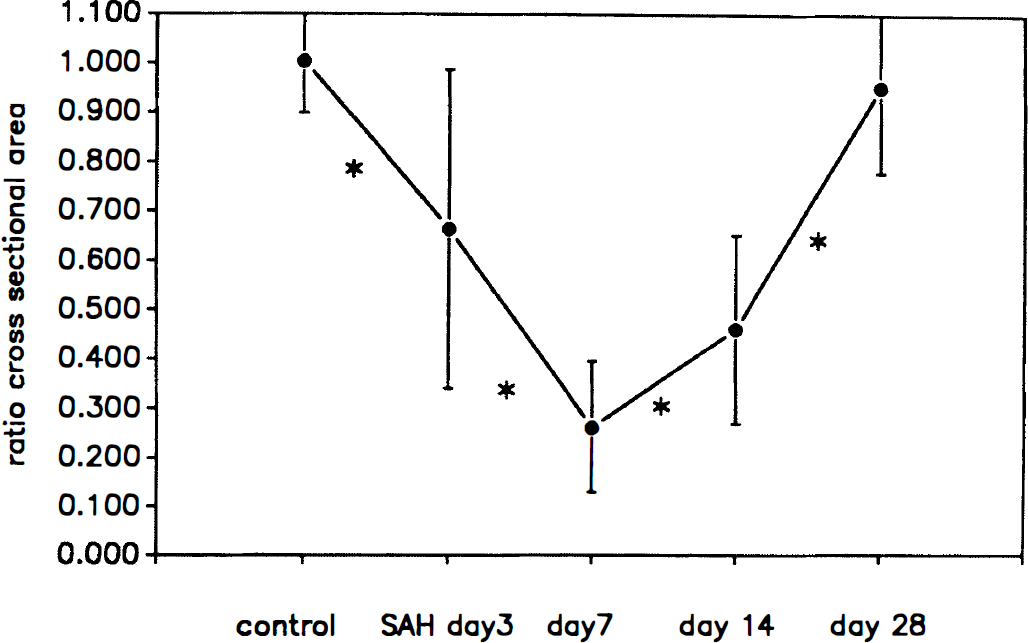
Course of angiographic vasospasm in the canine basilar arteries after subarachnoid hemorrhage (SAH). Ratio of the cross-sectional area to that of a baseline angiogram taken on day 0 was calculated in each group. Data shown are the mean ± SD (n = 6 for control and SAH day 7 groups; n = 3 for SAH day 3, 14, and 28 groups). Asterisk indicates significant difference (P < 0.05, analysis of variance) between two adjacent groups (control versus SAH day 3, SAH day 3 versus day 7, day 7 versus day 14, day 14 versus day 28).
Observations of DNA-binding dye entry
In the normal arteries, H33342, a lipophilic dye, stained the nuclei of the SMC, which are elongated and arranged in a tangential direction (Fig. 2A through C). The SMC nuclei emitted blue fluorescence on excitation. Ethidium bromide did not stain SMC nuclei, either in the inner periintimal layer (Fig. 2A), the middle layer (Fig. 2B), or the outer periadventitial layer (Fig. 2C) of the tunica media. Some of the endothelial cells, lined along the axis of the artery, were positively stained with EB, with the oval nuclei emitting characteristic pink–red fluorescence on excitation (Fig. 2D). The fibroblasts in the adventitial layer also were stained with EB (Fig. 2E).
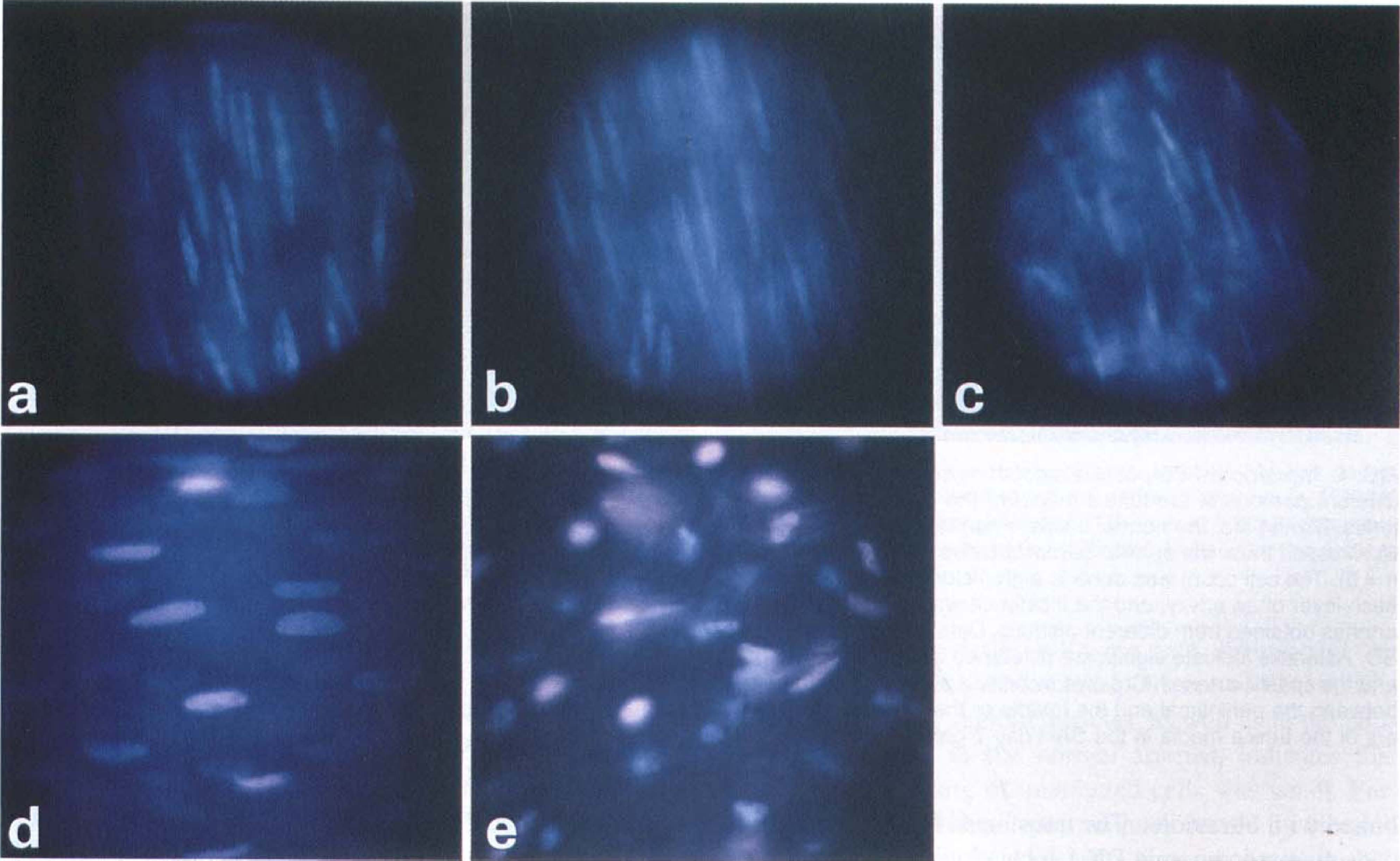
Ultraviolet-fluorescence photographs of a normal canine cerebral artery (control group) stained with two different DNA-binding dyes: ethidiuim bromide (EB) and Hoechst 33342 (H33342). Photographs were discretely focused on thin layers (focal depth 3.7 to 3.75 µm for the numerical aperture 1.3) of periintimal
In the SAH group, numerous SMC were positively labeled with EB; the nuclei were stained with characteristic pink–red fluorescence (Fig. 3). Incidence of EB-positive SMC was far greater in the inner periintimal layer of the tunica media (Fig. 3A) than in the middle (Fig. 3B) or the outer periadventitial portion (Fig. 3C).
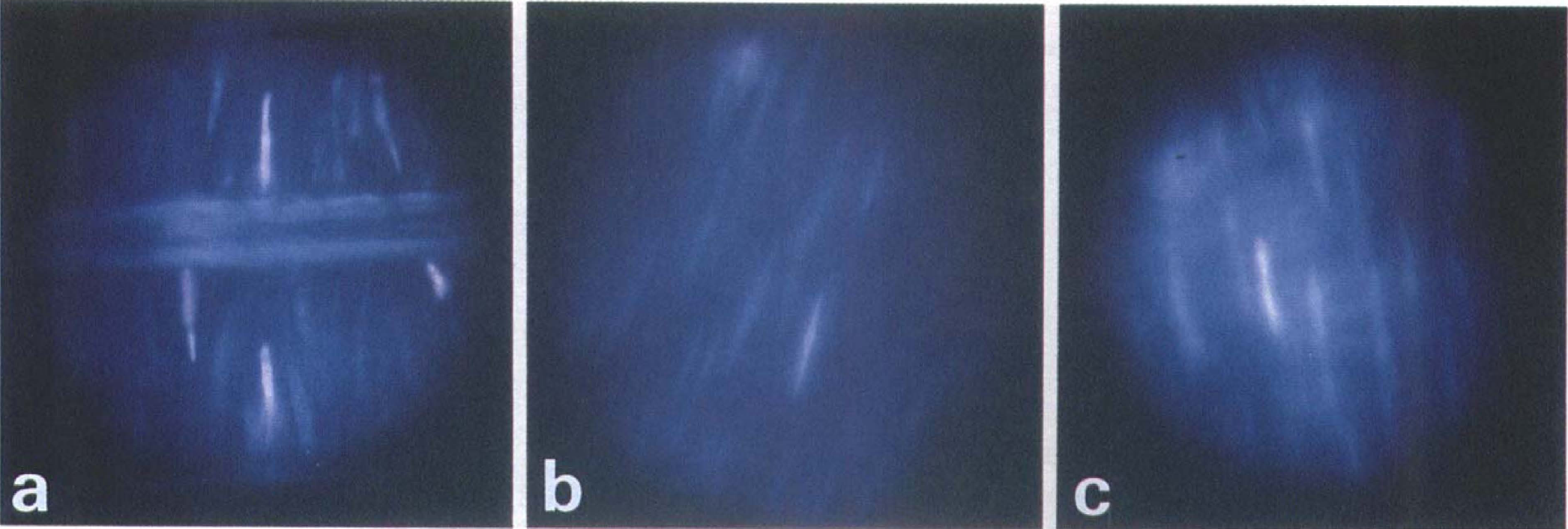
Ultraviolet-fluorescence photographs of a spastic canine cerebral artery (SAH day 7 group) stained with EB and H33342. Photographs were focused on thin layers (focal depth 3.7 to 3.75 pm) of periintimal
Cell counts (in the field of 320 × 480 µm, summation of eight different fields in each artery and averaged for six arteries in each group) were performed to compare the incidence of EB-positive cells quantitatively in the control and SAH groups (Figs. 4 and 5). The total number of smooth muscle cells was comparable between the control group (1081 ± 150, summation in eight fields of 320 × 480 µm, n = 6, mean ± SD) and the SAH day 7 group (1020 ± 172, n = 6, mean ± SD). In the periintimal layer of the tunica media, the incidence of EB-positive SMC was markedly larger in the SAH day 7 group (45 ± 19%) than in the control group (2.0 ± 1.7%, P < 0.0005, ANOVA, Fig. 4). In the middle layer of the tunica media, fewer EB-positive SMC were seen than in the periintimal layer (Fig. 4). The incidence in the middle layer was 8.6 ± 6.6% in the SAH day 7 group and 1.8 ± 3.5% (P < 0.05) in the control group. In the periadventitial layer, incidence of EB-positive SMC was 7.1 ± 3.5% in the SAH day 7 group and 1.1 ± 1.1% in the control group (P < 0.0005). In the SAH day 7 group, the difference between the incidences in the periintimal and middle layers, or that between the periintimal and periadventitial layers was statistically significant (ANOVA, both P < 0.0025).
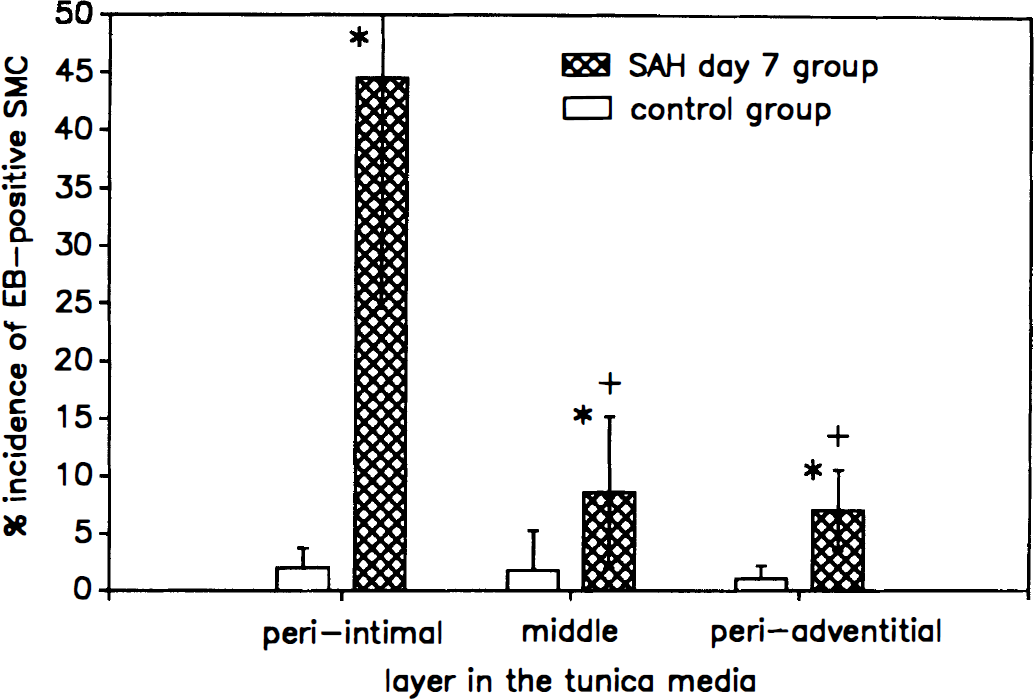
Incidence of EB-positive smooth muscle cells in the three different portions of the tunica media of the canine cerebral arteries. Blank bars: the normal basilar arteries (control group, n = 6). Crossed bars: the spastic basilar arteries (SAH day 7 group, n = 6). The cell count was done in eight fields (320 × 480 pm) in each layer of an artery, and the incidence was calculated for six arteries obtained from different animals. Data shown are mean ± SD. Asterisks indicate significant difference between the normal and the spastic arteries. Crosses indicate a significant difference between the periintimal and the middle or the periadventitial layers of the tunica media in the SAH day 7 group.
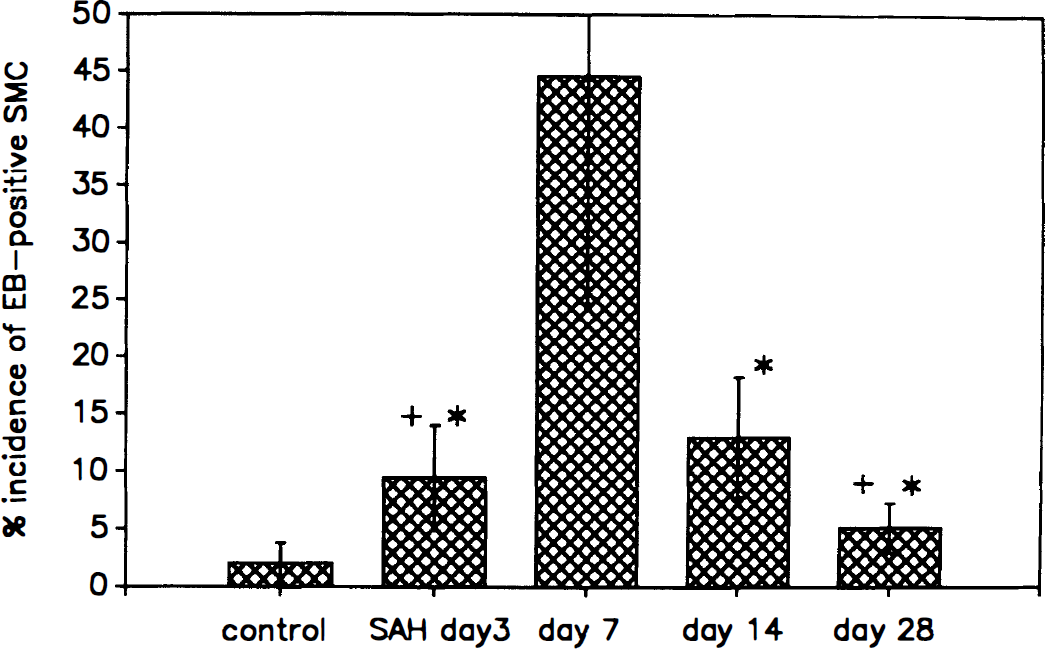
Time course of the alterations of sarcolemmal permeability in the canine basilar arteries after SAH. Incidences of EB-positive smooth muscle cells in the periintimal layer of tunica media are shown in each group (n = 6 for the control and SAH day 7 groups, n = 3 for SAH day 3, 14, and 28 groups). Asterisks indicate significant difference between the the SAH day 7 group and the SAH day 3, 14, or 28 groups. Crosses indicate significant difference between the normal control group and the SAH day 3 or 28 group. The incidence of altered cells in the periintimal layer was maximal at 1 week after SAH and decreased at 2 to 4 weeks after SAH (P < 0.01 between day 7 and 14 groups, P < 0.05 between day 14 and 28 groups).
To assess the time course and reversibility of the sarcolemmal alterations, the numbers of EB-positive cells were compared in the groups of SAH days 3, 7, 14, and 28 (Fig. 5). The incidence of altered cells in the periintimal layer was maximal in 1 week after SAH and decreased in 2 to 4 weeks after SAH (P < 0.01 between days 7 and 14, P < 0.05 between days 14 and 28).
Confocal microscopy
In the control group, intensity of fluorescence in the spectrum characteristic to EB was weak in the tunica media (Fig. 6A) and the incidence of SMC emitting in the spectrum was small in all layers (periintimal, middle, periadventitial) of the tunica media.
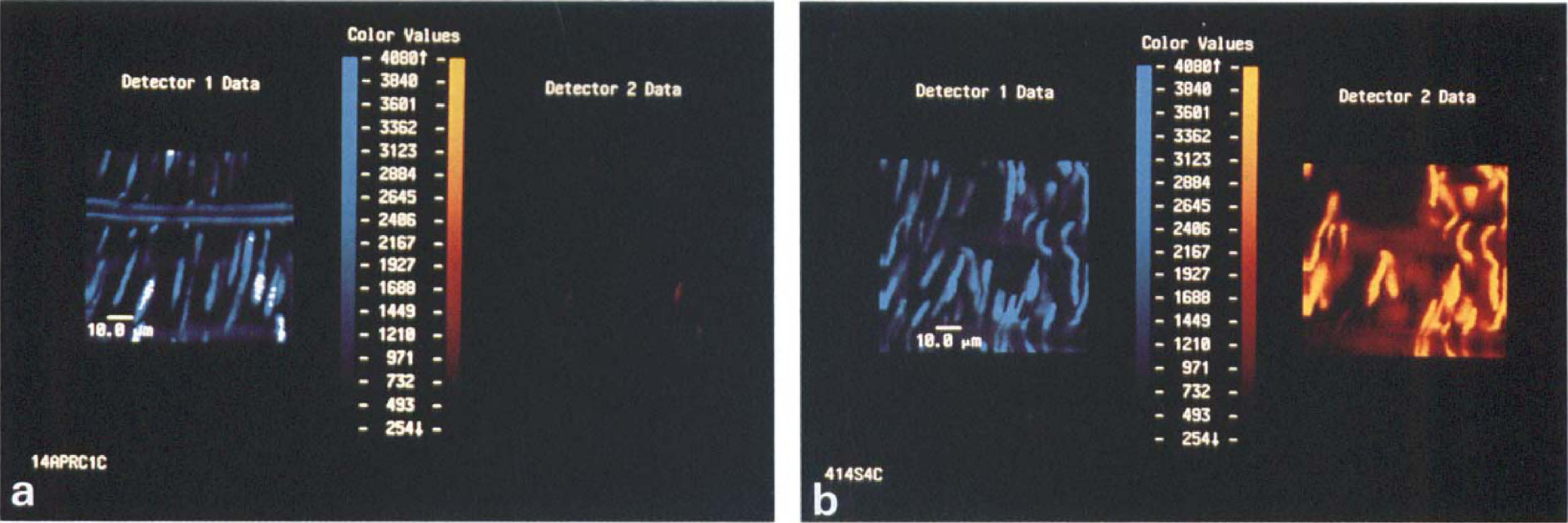
Laser confocal photomicrographs of the periintimal portion of the tunica media of the canine cerebral arteries.
In the SAH day 7 group, numerous nuclei of SMC were stained with EB. The EB-positive cells were frequently seen in the periintimal layer (Fig. 6B), but the incidence in the middle or periadventitial layers of the tunica media was scarce.
Light microscopy
In the control group, the transverse cross-section of the smooth muscles (cut along the vessel axis) appeared round, packed, and uniform (Fig. 7A). By contrast, in the SAH day 7 group (Fig. 7B), cross-section of the cytoplasm appeared shrunken. Cells with condensed cytoplasm were frequently observed. The interstitial space was expanded. These changes were prominent in the periintimal layer of the tunica media. Frank necrosis of smooth muscle was not observed in the spastic arteries.
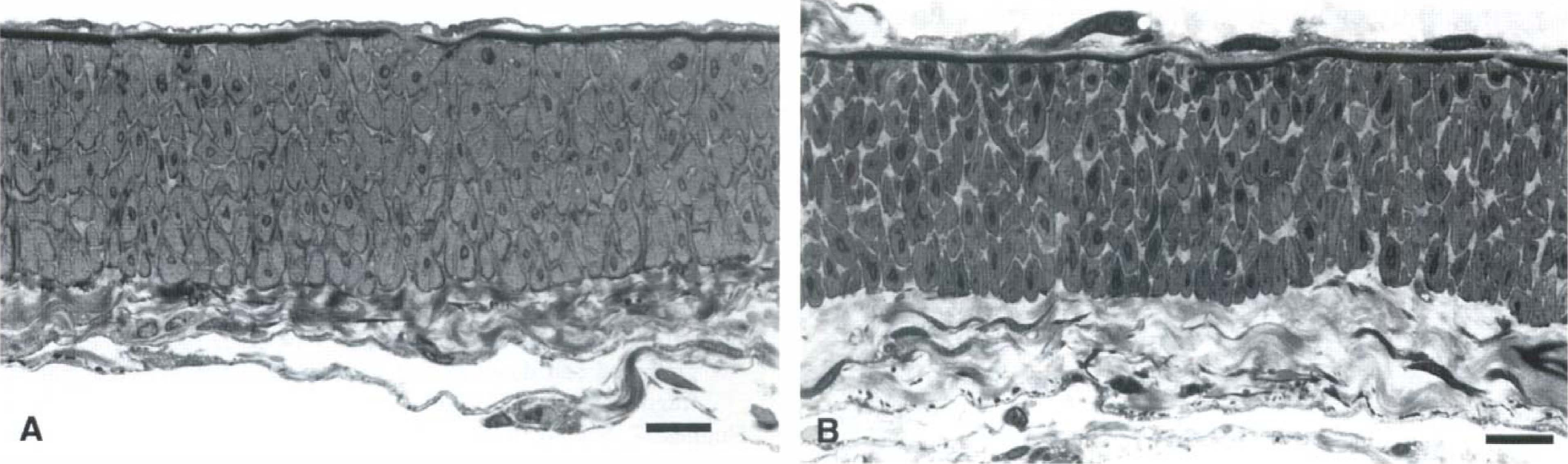
Photomicrographs of the semithin sections of the canine cerebral arteries.
Electron microscopy
Longitudinal section along the axis of the artery allowed best observation of cross-section of the SMC. In the SAH day 7 group, increase in the electron density of the cytoplasm was frequently observed. Size of the section of the cells appeared reduced, and the extracellular space was widened. Deposit of collagen fibers was observed in the space (Fig. 8). Occasionally, secretory-type SMC containing, well-developed intracellular organelles such as endoplasmic reticulum, Golgi apparatus, and mitochondria were seen. These changes were more remarkable in the inner layer of the tunica media but less pronounced in the outer portion. Frank necrosis of SMC was not seen; alterations such as dissolution of myofilaments, disruption of basal lamina, swelling of mitochondria, and disintegration of the cell were not observed. In general, intracellular structures such as dense bodies, sarcolemmal vesicles, and other organelles such as ribosomes, rough endoplasmic reticulum, and Golgi complexes were preserved in the smooth muscle cells of the spastic arteries.
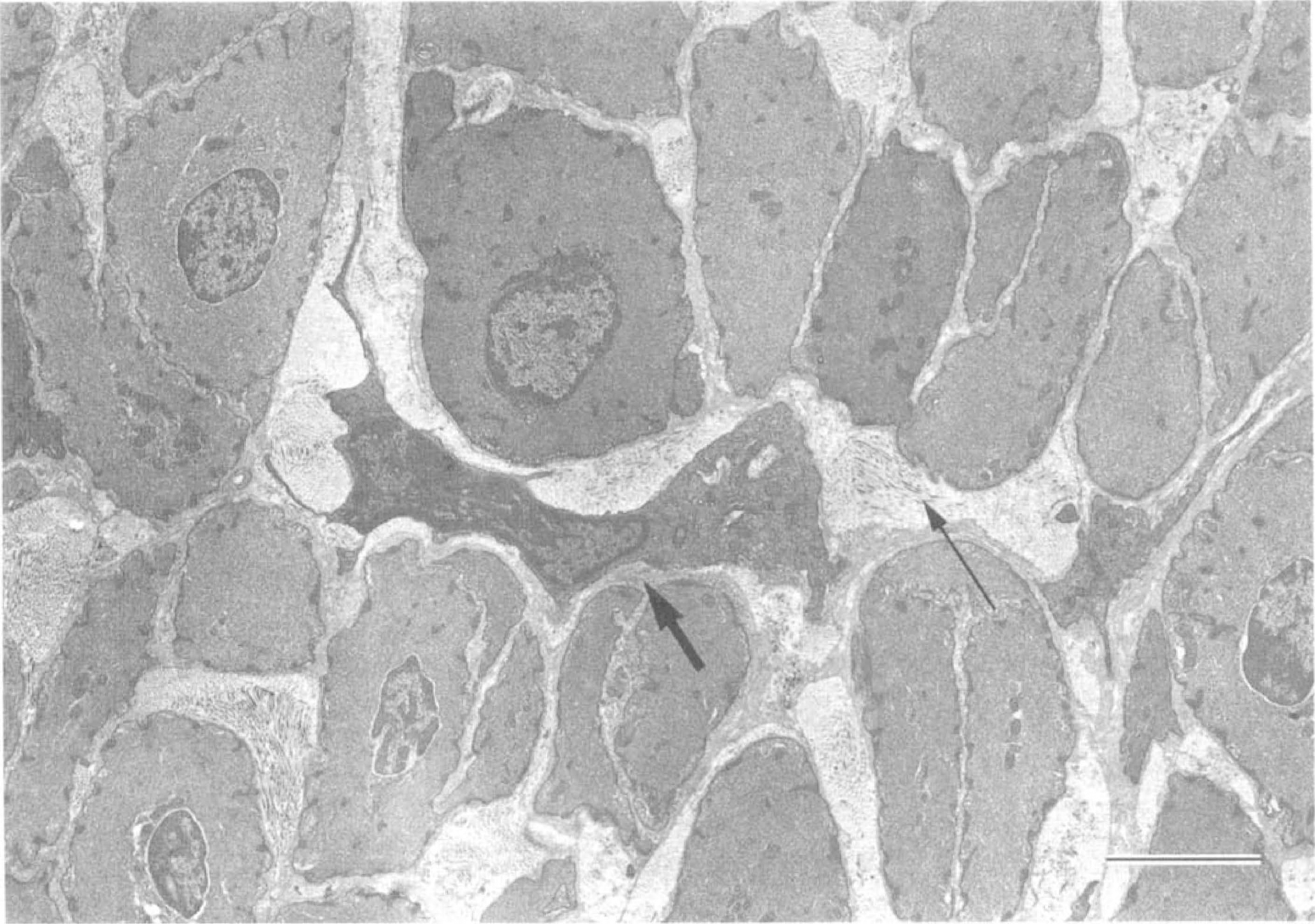
Electron micrographs of the periintimal portion of the medial layer of the canine spastic basilar artery (SAH day 7 group). Longitudinal cross-section (parallel to the vessel axis). Shrinkage and condensation of smooth muscle cytoplasm are characteristically observed. The extracellular space is widened, with deposits of collagen fibers (thin arrow). A secretory smooth muscle cell (thick arrow) is seen with well-developed intracellular organelles. Frank necrosis is not observed. Scale bars = 3 µm.
Detection of apoptosis
Electrophoresis of the lysate of the spastic cerebral arteries (SAH day 7 and 14 groups) was performed to detect DNA fragmentation. Briefly, arterial tissue was lysed in a buffer containing proteinase K and ethylenediamine tetraacetic acid. After heat-inactivation of the enzyme, the lysate was processed with RNase A. Ten micrograms of genomic DNA was electrophoresed on an agarose gel, blotted to a nylon membrane, and cross-linked with ultraviolet. The transferred DNA was probed with digested genomic DNA labeled with 32P. A ladder-like pattern of DNA fragmentation, characteristic to apoptosis, was not observed on the gel plates (data not shown).
For identification of programmed cell death in the tissue section of the spastic cerebral arteries, TUNEL staining (Gavrieli et al., 1992) was performed. Briefly, paraffin-embedded sections of formaldehyde-fixed arteries were deparaffinized and rehydrated. The samples were incubated with proteinase and then with H2O2 to inactivate endogenous peroxidase. After treatment with terminal deoxynucleotidyl transferase, the sections were incubated with biotynylated dUTP. The samples were rinsed, incubated with horseradish peroxidase–conjugated streptoavidine, and reacted with diaminobentidine.
Using TUNEL staining, the spastic arteries obtained from SAH day 7 and 14 groups did not demonstrate SMC undergoing the process of programmed cell death– apoptosis (data not shown). The same protocol positively stained neurons undergoing delayed death after transient ischemia in the hippocampus of gerbil.
DISCUSSION
The current finding of enhanced entry of a hydrophilic dye, EB, indicates an increase in sarcolemmal permeability in the cerebral arteries during chronic spasm. In our previous studies, disturbances in the cellular energy metabolism and alterations in calcium homeostasis of the smooth muscle were evident in the spastic cerebral arteries (Kim et al., 1992; Kim et al., 1996). Contents of high-energy phosphates were diminished with a time course correlated to that of arterial narrowing (Yoshimoto et al., 1993). Calcium homeostasis of the smooth muscle was impaired, with excessive response of [Ca2+]i when [Ca2+]e was increased (Kim et al., 1996). The impairment of the membrane integrity presumably reflects alterations in the cellular milieu and injury to the sarcolemma.
A distinctive spatial localization of the sarcolemmal alterations was observed: 45% of the smooth muscle cells in the inner (periintimal) layer of the tunica media were stained positive with EB, as opposed to 8.6% in the middle layer and 7.1% in the outer (periadventitial) layer of the tunica media. This finding also was confirmed by confocal microscopic study, which showed marked increase in the incidence of smooth muscle cells emitting fluorescence in the spectrum for EB. A logical implication of the observations would be that the causative factors for the alterations exist in a higher density in the inner layers. It has been shown that oxyhemoglobin causes membrane damage, since free radicals are generated in the process of oxidation to methemoglobin (Misra and Fridovich, 1972). The superoxide anions and hydroxyl radicals attack the membrane and cause cellular damage (Chan et al., 1982; Steele et al., 1991). The spastic cerebral arteries are surrounded by blood containing hemoglobin, and if oxyhemoglobin is the primary cause, incidence of affected cells should be higher in the outer layers. A possible alternative mechanism to explain the preference in the inner portion of the sarcolemmal damage is metabolic disturbance secondary to encasement of the artery by the blood clot. The cerebral arteries lack vasa vasorum and instead have a network of channels in the medial layer with pores opened to the subarachnoid space (Zervas et al., 1982). It has been postulated that the pores serve as pathways for the nutrients and metabolites. After experimental SAH, the channels were found to be filled and obstructed with debris (Espinosa et al., 1986). If this condition leads to metabolic disturbances and decreased viability of the smooth muscle cells, it could preferentially affect the deeper layer, namely the inner layer, of the tunica media. The observed distribution of the EB-positive cells is not likely to be caused by differences in exposure to the dye, since the condition of exposure is uniform for the periintimal and the periadventitial sides. The vessels were cut open and exposed in EB solution. In fact in the control group, there was no difference in the incidence of EB-positive cells among the periintimal, the middle, and the periadventitial portions of the tunica media. This indicates that the barrier effect of the endothelium and the lamina elastica is no more than that of the adventitia, which is composed of loose connective tissue, allowing free diffusion of the dye. Simultaneously, the small incidence of EB staining in the periintimal smooth muscles, as well as in the periadventitial layer in the normal arteries, indicates that nonspecific staining of unaffected cells was small. Furthermore, the histologic alterations observed by electron and light microscopic study also were more pronounced in the periintimal layers.
Necrosis of smooth muscle cells has been described in human spastic cerebral arteries (Crompton, 1964; Hughes and Schianchi, 1978) and in that of experimental models (Fein et al., 1974; Tanabe et al., 1978). However, the findings were contradicted by negative reports for such transformation in both clinical (Conway and McDonald, 1972; Eldevik et al., 1981) and experimental vasospasm (Tani et al., 1978; Pickard et al., 1985; Eldevik et al., 1981). The controversy has not been reconciled. Despite the finding of the alterations of sarcolemmal permeability in the periintimal layer, we did not observe frank necrosis of smooth muscle cells in the corresponding location. Changes seen in necrotic process such as cytoplasmic swelling, clumping of chromatin, swelling of mitochondria, membrane disruption, and cell disintegration were not observed in these preparations. Possible contribution of apoptosis to the alterations of smooth muscle properties was examined by histochemical and electrophoretic methods, but no evidence for fragmentation of DNA was obtained in the spastic arteries.
The results therefore indicate that the alteration in the sarcolemma does not lead to acute cell death or programmed cell death. Apoptosis, either as a result or a cause for the alteration, was not detected in the pathologic condition. The prolonged duration of the sarcolemmal alteration and resolution over 2 to 4 weeks coincide with the course of angiographic narrowing. The temporal profile of the phenomenon supports the notion that the membrane alteration may be pathogenetically linked to the development of chronic spasm. Increased sarcolemmal permeability can underlie the increased calcium permeability, which was described in our previous report (Kim et al., 1996). Mild but long-lasting pathologic increase in calcium permeability can cause contraction continuing over a period and refractory to various vasodilatory agents. Increased permeability can lead into decrease in the contents of high-energy phosphates and cyclic GMP, a pivotal substance for relaxation of smooth muscle, including those in response to endothelium-derived relaxing factor, endothelium-dependent relaxing factor, or nitric oxide (Kim et al., 1992a, b ). Therefore the increase in the sarcolemmal permeability may be underlying the “final common pathway” to lead into the sustained and unregulated constriction of the cerebral artery after SAH.
Footnotes
Acknowledgment
The authors thank Dr. Susumu Wakai for reviewing this article and providing helpful comments.
