Abstract
Acetylcholine is an important regulator of local cerebral blood flow. There is, however, limited information available on the possible sites of action of this neurotransmitter on brain intraparenchymal microvessels. In this study, a combination of molecular and functional approaches was used to identify which of the five muscarinic acetylcholine receptors (mAChR) are present in human brain microvessels and their intimately associated astroglial cells. Microvessel and capillary fractions isolated from human cerebral cortex were found by reverse transcriptase-polymerase chain reaction to express m2, m3, and, occasionally, m1 and m5 receptor subtypes. To localize these receptors to a specific cellular compartment of the vessel wall, cultures of human brain microvascular endothelial and smooth muscle cells were used, together with cultured human brain astrocytes. Endothelial cells invariably expressed m2 and m5 receptors, and occasionally the m1 receptor; smooth muscle cells exhibited messages for all except the m4 mAChR subtypes, whereas messages for all five muscarinic receptors were identified in astrocytes. In all three cell types studied, acetylcholine induced a pirenzepine-sensitive increase (62% to 176%, P < 0.05 to 0.01) in inositol trisphosphate, suggesting functional coupling of m1, m3, or m5 mAChR to a phospholipase C signaling cascade. Similarly, coupling of m2 or m4 mAChR to adenylate cyclase inhibition in endothelial cells and astrocytes, but not in smooth muscle cells, was demonstrated by the ability of carbachol to significantly reduce (44% to 50%, P < 0.05 to 0.01) the forskolin-stimulated increase in cAMP levels. This effect was reversed by the mAChR antagonist AF-DX 384. The results indicate that microvessels are able to respond to neurally released acetylcholine and that mAChR, distributed in different vascular and astroglial compartments, could regulate cortical perfusion and, possibly, blood–brain barrier permeability, functions that could become jeopardized in neurodegenerative disorders such as Alzheimer's disease.
Acetylcholine (ACh) is an important regulator of CBF in man and in many other species (for review see Sato and Sato, 1995; Dauphin and MacKenzie, 1995). Intracortical and basalocortical cholinergic nerve fibers have been shown to intimately associate with microarterioles and capillaries in the rat (Vaucher and Hamel, 1995) and human (Tong and Hamel, 1997) cerebral cortex. Interestingly, cholinergic-mediated increases in local CBF have been observed in the frontal and parietal subdivisions of the cerebral cortex on stimulation of basal forebrain cholinergic neurons and muscarinic ACh receptors (mAChR), some of which, presumably associated with the local microcirculation, have been implicated in this response (for review see Sato and Sato, 1995). Cerebromicrovascular mAChR binding sites have indeed been reported (Estrada et al., 1983), and more recently, multiple mAChR subtypes have been identified in intracortical microvessels and capillaries, including in man (García-Villalón et al., 1991; Linville and Hamel, 1995). However, the absence of highly selective subtype-specific ligands for all mAChR made it difficult to conclusively identify the exact receptor subtype involved. Furthermore, these radioligand binding studies in tissue homogenates could not localize mAChR to a specific cellular compartment of the isolated microvascular fractions (Linville and Hamel, 1995; Moro et al., 1995).
In this respect, in addition to direct interactions with microvascular smooth muscle (SMC) and endothelial (EC) cells, ACh could also affect local brain perfusion and other microvascular functions through perivascular astrocytes (AST), suggested as intermediaries in the neurogenic control of the microcirculation by cholinergic mechanisms (Vaucher and Hamel, 1995). Interestingly, astrocytic processes associated with bovine and rat brain capillaries have been shown to express mAChR (Moro et al., 1995; Luiten et al., 1996) and could thus be involved in the local regulation of cortical perfusion through the release of vasoactive mediators (Alkayed et al., 1997).
Acetylcholine exerts its effects through any one of the five cloned mAChR subtypes, with downstream effects of either inositol phosphate activation (m1, m3, and m5) or attenuation of adenylate cyclase activity (m2 and m4) (for review see Felder, 1995). In large cerebral arteries, activation of mAChR can result in different vascular responses depending on which mAChR subtype is stimulated. Pharmacologic studies have identified the M3 mAChR subtype as the mediator of the ACh-induced endothelium-dependent dilation (Dauphin and Hamel, 1990; Garcia-Villalon et al., 1991), whereas the Ml subtype has been implicated in cerebral vasoconstriction (Dauphin et al., 1991; Shimizu et al., 1993). At the level of the brain microcirculation, ACh induces vasodilation of intracortical arterioles in rat (Dacey and Bassett, 1987), bovine, and human (Elhusseiny and Hamel, 1998). However, the subtypes and location of the receptors involved in this and other putative ACh-mediated responses are not known.
In the present study, we used reverse transcriptase-polymerase chain reaction (RT-PCR) to identify the mAChR expressed in human brain microvessels (MV) and capillaries (CAP). To examine the localization and operational behavior of the mAChR within the vessel wall, we evaluated receptor mRNA expression in cultures of human brain microvascular (i.e., EC and SMC) and astroglial cells, and assayed their ability to couple to their expected second-messenger signaling pathways in cultured cells. The results show that human intracortical MV and CAP are endowed with multiple functional mAChR subtypes that exhibit a distinct cellular localization within the vessel wall. These receptors could mediate vasomotor responses in intracortical MV, either directly by activating receptors on SMC, or indirectly via receptors expressed on EC or perivascular AST.
MATERIALS AND METHODS
Isolated MV and CAP
Human brains (n = 6; average postmortem delay, 16.2 ± 2.2 hours; 4 men, 2 women) were obtained at autopsy from subjects who died from diseases not affecting the CNS, with approval from the Institutional Research Ethics Committee, either from the Brain Bank of the Douglas Hospital Research Centre (Verdun, Québec, Canada) or the department of pathology at the Royal Victoria Hospital (Montréal, Québec, Canada). Intracortical MV and CAP were isolated as described previously (Linville and Hamel, 1995). Briefly, after homogenization and centrifugation over 15% dextran, MV and CAP were harvested by filtration through a 151-µm and subsequently a 53-µm nylon mesh screen, respectively. Previous light microscopic examination of similarly prepared MV and CAP fractions have indicated their high level of purity, as also shown by the enrichment (>30-fold) in the endothelial marker enzymes γ-glutamyl transpeptidase (γ-GTP) and alkaline phosphatase over cerebral tissue (see Linville and Hamel, 1995).
Cell cultures
Cultures of human brain EC and SMC were produced, with approval from the Institutional Research Ethics Committee, from intracortical CAP and MV harvested from biopsies obtained from patients (n = 6) undergoing surgery for the treatment of temporal lobe epilepsy (Stanimirović et al., 1996; Abounader et al., 1999). In short, SMC cultures were generated from cortical homogenates that were sequentially filtered through a 350-µm and 112-µm mesh. The resistance MV were then dislodged from these meshes with cold cell culture medium M199 and dissociated with type IV collagenase (1 mg/mL; 15 minutes). After 4 to 5 weeks in culture, more than 85% of cells stained positively for the SMC marker α-actin, whereas about 10% of cells demonstrated EC morphology and incorporated acetylated low-density lipoproteins (Abounader et al., 1999). Six primary cultures of SMC from different biopsies were used in this study.
Human microvascular and capillary EC cultures were grown from CAP obtained after the 112-µm filtrate had been further resuspended in M199 medium and sieved through a 20-µm mesh screen. Endothelial cell colonies were isolated with cloning rings and grown to confluence. More than 95% of cells in these cultures were immunopositive for factor VIII-related antigen, incorporated acetylated low-density lipoproteins, and expressed high levels of cerebral endothelium-specific enzymes, γ-glutamyl-transpeptidase and alkaline phosphatase. Morphologic, phenotypic, biochemical, and functional characteristics of these EC cultures have been described earlier (Stanimirović et al., 1996). Both primary and passaged (3 to 7 times, n = 6) EC cultures were used in this study.
Cultures of fetal (10 to 18 weeks of gestation) human brain AST (n = 5) were provided by Drs. J. Antel and W. Yong (Montreal Neurological Institute, Montréal, QC, Canada) and prepared according to well-established procedures (for details, see Abounader et al., 1999). Brains were trypsinized (0.25%), homogenized, and filtered through a 130-µm mesh. Cell pellets were resuspended in a 95% Dulbecco's modified Eagle's medium containing 5% fetal bovine serum, and plated onto poly-
Reverse transciption-polymerase chain reaction
RNA isolation and reverse transcription. Total RNA was extracted by homogenization with a polytron (for whole blood vessel preparations) or passage through a syringe (for cell cultures) in the TRIzol Reagent (1 mL per 50–100 mg tissue, Gibco BRL, Gaithersburg, MD, U.S.A.), according to Chomczynski and Sacchi (1987). RNA (2 to 20 µg) was treated with DNase I (3 U, Gibco BRL) to remove residual genomic DNA, extracted with phenol-chloroform, and precipitated with ethanol, and the quantity and purity was assessed by the A260/A280 ratio. Synthesis of single-stranded cDNA was performed by reverse transcription (1 hour, 42°C) in a reaction mixture (final volume, 50 µL) containing 1× reaction buffer (Gibco BRL), 2 mmol/L dithiothreitol, 30 to 60 U RNase inhibitor (Promega, Madison, WI, U.S.A.), random hexamers (200 ng/µg RNA, Gibco BRL), and 1 to 3 U of avian myeloblastosis virus reverse transcriptase (AMV-RT, Promega). To control for genomic DNA contamination, half of the primed RNA was used to generate non-RT samples by incubation in a reaction mixture lacking AMV-RT.
Primers. Specific primers were designed according to published sequences of the cloned human mAChR genes for m1, m2 (Peralta et al., 1987) m3, m4 (Bonner et al., 1987), and m5 (Bonner et al., 1988) and synthesized using Applied Biosystems synthesizer. The m1 and m3 primers were flanked with the T7 (5'-GGT AAT ACG ACT CAC TAT AGG GCG A-3') and SP6 (5'-CTC GGA TTT AGG TGA CAC TAT ATG AAT AC-3') RNA polymerase promoter sequences for use in other experiments. Primer sequences and length of amplified regions were as follows: m1, TGA GGG CTC ACC AGA GAC T (forward 844–862), GTC CAG GTG AGG ATG AAG G (reverse 1218–1236) (Δ393); m2, ACA AGG AAG GAT AGT GAA GCC (forward 808–828), CAT CTC CAT TCT GAC CTG AAG (reverse 1146–1166) (Δ359); m3, AGA TGG ACC AAG ACC ACA G (forward 1178–1196), CAG AGT GGC TTC CTT GAA G (reverse 1572–1590) (Δ413); m4, GAA GGA GAA GAA AGC CAA G (forward 1502–1520), ATC TCA ATG GCT GTC ACA C (reverse 1855–1873) (Δ372); m5, (TCT GTT CAG ATC CTG CTT G) (forward 992–1010), TGC TGG AGA CAG AAG GTA GT (reverse 1327–1346) (Δ355). The primers were tested for specificity by performing RT-PCR in Spodoptera frugiperda (Sf-9) cells infected with recombinant bacculoviruses individually expressing one of the five human mAChR (graciously provided to us by BioSignal Inc., Montréal, QC, Canada).
PCR amplification and sequencing. The PCR amplifications were performed using a final volume of 50 µL containing 3 mmol/L MgCl2, 0.5 mmol/L deoxynucleotide triphosphates, 2.5 µL dimethyl sulfoxide, 0.5 µmol/L of each primer, 3–5 µL of experimental template, and 2 U of Taq polymerase. An MJ Research thermocycler (Fisher Scientific, Montreal, QC, Canada) was used with the following program: an initial denaturation step of 95°C for 5 minutes, 40 cycles (except in three of 20 PCR runs for the m2 subtype in which 35 [n = 1] and 37 [n = 2] cycles were performed) of 94°C for 40 seconds, 56°C for 40 seconds, and 72°C for 60 seconds, and a final extension at 72°C for 5 minutes. The PCR products were electrophoresed on 1.2% agarose gels and photographed under fluorescent ultraviolet illumination. All samples were normally tested in triplicate, but occasionally in duplicate. A positive control consisting of the human cerebral cortex, which contains all five mAChR (Costa et al., 1995), was used to verify the efficiency of the amplification. Reverse transcriptase and non-RT RNA were similarly treated for PCR amplification. The PCR products corresponding to each mAChR were sequenced from representative tissue to verify homology with the cloned human receptor genes. For the m1 (SMC), m2 (EC), and m3 (SMC) mAChR, PCR products were first digested with appropriate restriction enzymes and inserted into PGEM (Promega) or Bluescript plasmids by ligation with T4 DNA ligase. Competent DH5α (pGEM7) and XL1 (Bluescript) cells, respectively, were transformed and prepared for sequencing using mini (5' to 3') followed by midi (Qiagen, Mississauga, ON, Canada) preparations. Nucleotide sequence analysis was performed using the Sanger dideoxynucleotide chain termination method and Sequenase (U.S. Biochemical) in an automated sequencer (ALF, Pharmacia, Montréal, QC, Canada). The m4 (AST) and m5 (AST) PCR products were purified (Qiagen QIAquick) and sequenced directly using direct automated fluorescent sequencing (WM. Keck Foundation Biotechnology Resource Laboratory, Yale University, CT, U.S.A.).
Second messenger assays
Inositol trisphosphate formation. Inositol trisphosphate (IP3) formation in cells subjected to mAChR stimulation was assessed on confluent EC, SMC, or AST cultures grown in Petri dishes and prelabeled with [3H]myo-inositol (1 to 5 µCi/mL) for 24 hours in serum- and inositol-free medium M199, as previously described (Stanimirović et al., 1996). The cells were washed and exposed to ACh (1 mmol/L) alone or after a 15-minute preincubation with the M1-preferring mAChR antagonist, pirenzepine (1 mmol/L) (Dörje et al., 1991), which was shown in preliminary experiments to have no significant effect (104% ± 24% of basal levels) on phosphoinositide production. In some cases, the mAChR agonist carbachol (200 µmol/L) was also used. The stimulation was performed in the presence of 10 mmol/L LiCl (Berridge et al., 1982) for 15 minutes and then terminated by addition of 10% trichloroacetic acid. Cells were scraped, centrifuged, and the supernatants were repeatedly extracted with ethyl ether; IP3 was separated from the aqueous phase by ion exchange chromatography. The columns (DOWEX-AG 1× 8-formate) were washed with 20 volumes of 60 mmol/L ammonium formate–5 mmol/L sodium tetraborate followed by the consecutive elution of inositol monophosphate, bisphosphate, and trisphosphate by 8 volumes of 0.2 mol/L ammonium formate–0.1 mol/L formic acid, 0.4 mol/L ammonium formate–0.1 mol/L formic acid, and 1 mol/L ammonium formate–0.1 mol/L formic acid, respectively. Production of IP3 was expressed as a function of protein content in cell extracts. All results are expressed in mean ± SD, and statistical differences were compared by analysis of variance followed by a Newman-Keuls comparison test (GraphPad Prism).
cAMP attenuation. Inhibition of adenylate cyclase through mAChR stimulation was evaluated by measuring cAMP levels in confluent cells exposed to forskolin (1 µmol/L, 15 minutes) alone or in the presence of either the mAChR agonist carbachol (200 µmol/L, 15 minutes) alone or carbachol and the m2/m4 mAChR antagonist AF-DX 384 (1 × 10−5 mol/L) (Dörje et al., 1991) preincubated with the cells for a 15-minute period. AF-DX 384 was found in preliminary experiments to have no significant effect on cAMP production in cultured microvascular cells (81% ± 23% of basal levels). Levels of cAMP were measured by commercially available enzyme immunoassay kit (Amersham Life Science) and calculated as a function of cell protein content. All results are expressed as mean ± SD, and statistical differences were evaluated by analysis of variance followed by a Newman-Keuls comparison test (GraphPad Prism).
RESULTS
Expression of mAChR in isolated microvascular fractions and cells in culture
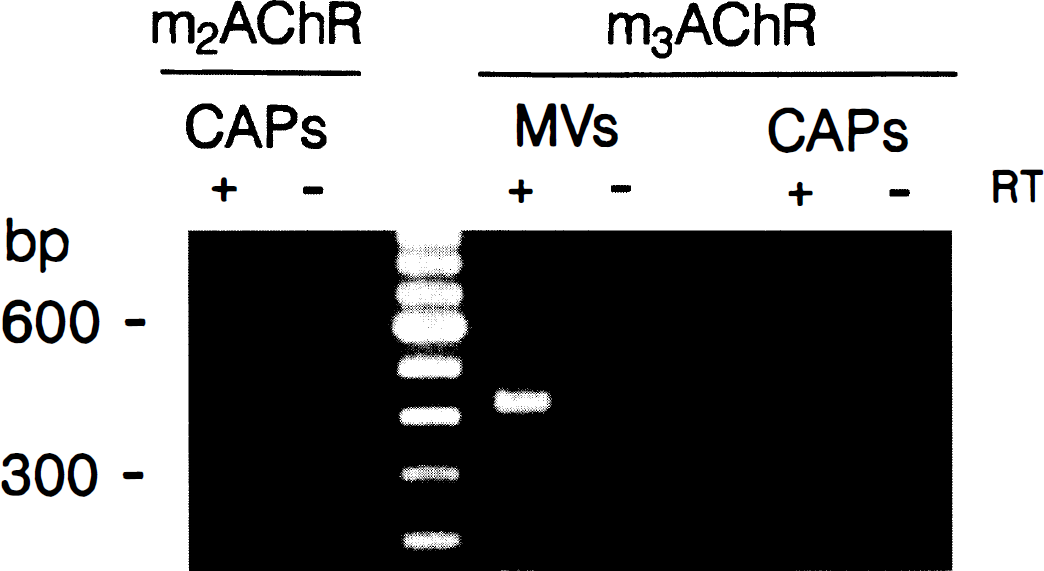
The primers were found to be highly selective and to yield PCR products of the expected size exclusively when the amplification was performed on the Sf-9 cells infected with the corresponding cloned human mAChR (Fig. 1). In all experiments, amplification of PCR products for the various mAChR was not a result of genomic DNA contamination because they were exclusively detected in samples that had been reverse transcribed (+RT lanes in Figs. 1, 2, and 3, see also legends to these figures).
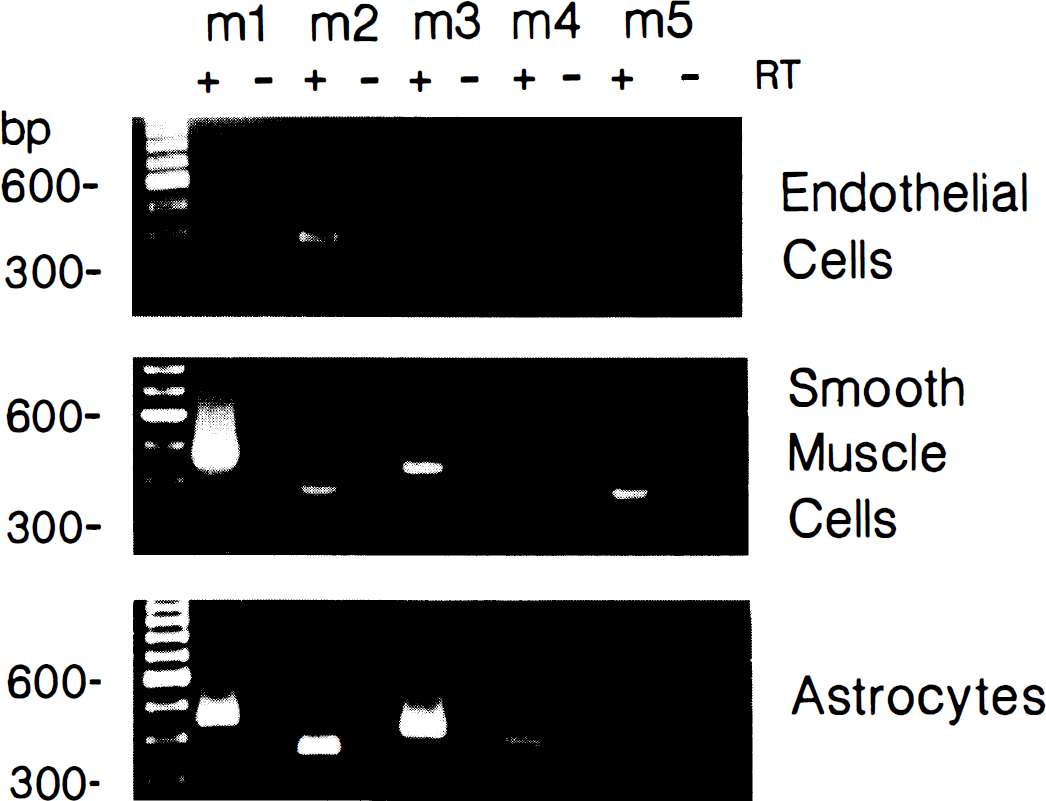
Specificity of selected primers for their respective human mAChR. Agarose gel electrophoresis showing that the selected oligonucleotide primers yield reverse transcription-polymerase chain reaction (RT-PCR) products exclusively in Sf-9 cells infected with the corresponding human mAChR subtype. No products were amplified from the non-RT preparation (not shown). Identification of mAChR in human MV and CAP isolated from postmortem cerebral cortex. Gel electrophoresis of reverse transcription-polymerase chain reaction (RT-PCR) products from representative samples show that the m2 and m3 mAChR are present in a majority of CAP fractions, whereas only the m3 subtype was clearly identified in the MV. Only PCR products present in greater than or equal to 50% of samples tested (n = 6) are illustrated. The PCR reactions were performed on RT (+) and non-RT (–) samples. Representative gel electrophoresis of reverse transcription-polymerase chain reaction (RT-PCR) products for the mAChR expressed in human brain microvascular and astroglial cells in culture. Only products that were present in greater than or equal to 50% of cell cultures are shown. Cultured EC express m2 and m5, SMC express messages for all mAChR except the m4 subtype, and AST, all five mAChR. The PCR reactions were performed on RT (+) and non-RT (–) samples.
Summary of mAChR receptors expression in human intracortical microvessels and capillaries, microvascular endothelial and smooth muscle cells, as well as astroglial cells in culture

Polymerase chain reaction results were obtained from six (or five for AST) separate human donors and are expressed as the amount of samples which showed a positive PCR product, with the following scale: (−): 0; (+/−): 1—2; (++): 3—4; (+++): 5—6. In final results analysis, only receptors that appeared in 50% or more of samples were considered to be conclusively present in the tissues. AST, astraglial cells; CAP, capillaries; EC, endothelial cells; MV, microvessels; SMC, smooth muscle cells.
When studied in microvascular and astroglial cell cultures, PCR products for the m2 and m5 mAChR were invariably amplified in EC (Fig. 3), and message for the m1 receptor subtype was detected in some preparations (Table 1). In contrast, mRNA for the m3 mAChR subtype was not detected in any EC cultures, including the primary EC culture. In primary cultures of microvascular SMC, strong PCR signals could be demonstrated by gel electrophoresis for the m1, m2, m3, and m5 mAChR, but not for the m4 subtype (Fig. 3 and Table 1). In cultures of fetal human brain AST, expression of the five cloned human mAChR was evidenced by their expression level being generally very strong, as evaluated by gel electrophoresis (Fig. 3 and Table 1). Sequence analysis of the selected PCR products showed that they unambiguously corresponded to the cloned human mAChR with the following accuracy: m1 (99%), m2 (98%), m3 (99%), m4 (98%), and m5 (99%). This slight variation in sequence is fully acceptable for single-strand sequence analysis.
Second messenger assays
IP3 assay. Addition of ACh (1 mmol/L) to EC, SMC, and AST cell cultures resulted in a significant increase in IP3 production. The increase over basal levels was comparable in EC (62.6% ± 10.8%; P < 0.01) and SMC (63.5% ± 18.0%; P < 0.05), and somewhat higher in AST (176.2% ± 19.5%; P < 0.01) (Fig. 4). A similar increase (120% ± 34.3%) was obtained with 200 µmol/L carbachol when tested in AST (not shown). In all three cell types, the ACh-induced IP3 production was significantly (P < 0.05) inhibited to near basal levels by preincubation with the mAChR antagonist pirenzepine (1 mmol/L) (Fig. 4).
Stimulation of IP3 production in EC 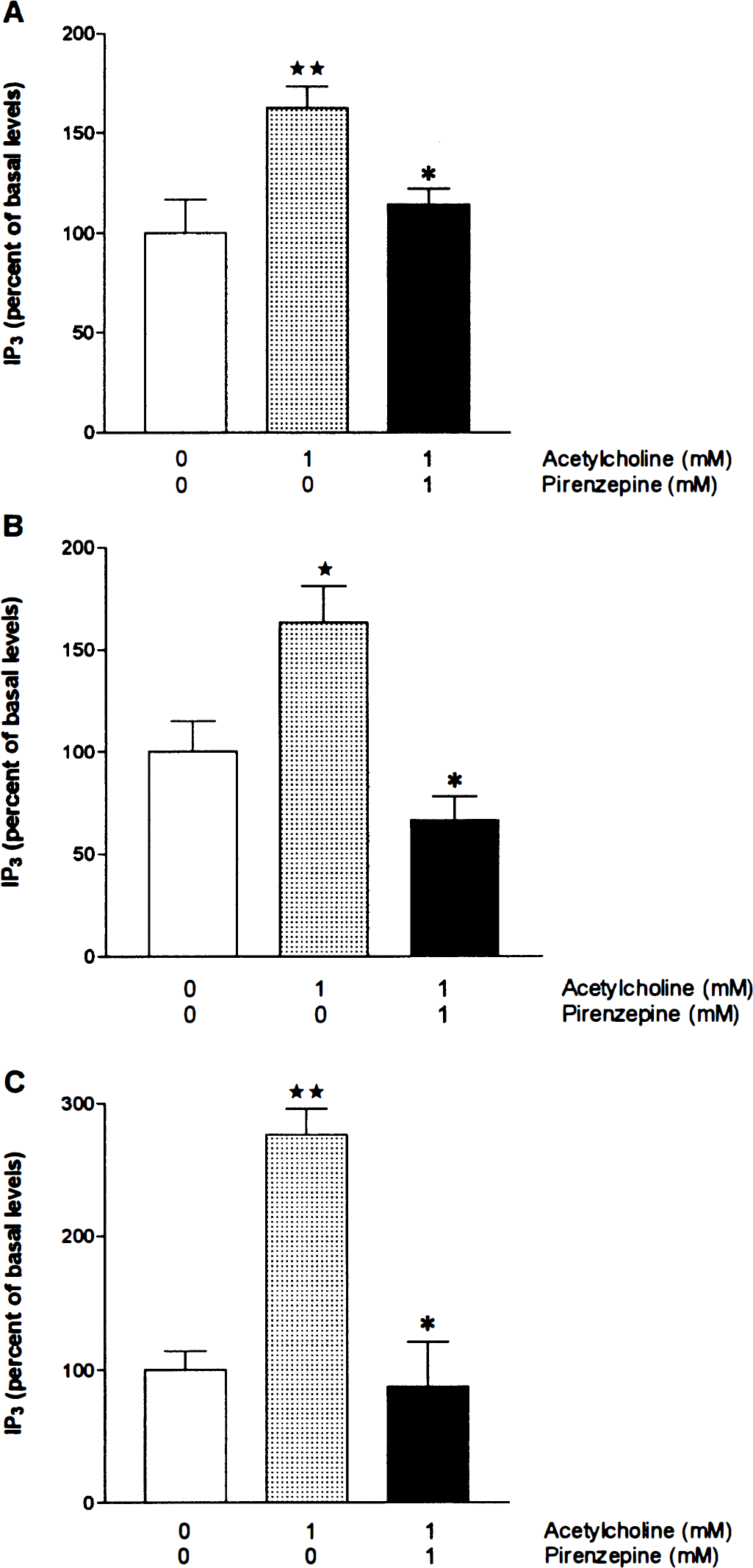
cAMP assay. Forskolin (1 µmol/L) resulted in an 8- to 18-fold increase (P < 0.01) in cAMP levels in EC, SMC, and AST in culture. The cholinergic agonist carbachol (200 µmol/L) significantly reduced the forskolin-stimulated cAMP levels by 50% ± 23.4% (P < 0.05) in EC and 44% ± 10.2% (P < 0.01) in AST. Addition of the mAChR antagonist AF-DX 384 (10−5 mol/L) significantly antagonized the inhibitory effect of carbachol in both cell types, returning cAMP levels to 82.6% ± 7.1% (P < 0.05) and 98.6% ± 11.4% (P < 0.01) of forskolin-stimulated cAMP levels, respectively. In contrast, carbachol and AF-DX 384 had no significant effect on the forskolin-mediated increase in cAMP levels in SMC cultures (n = 3) (Fig. 5B).
Effect of carbachol and AF-DX 384 on the forskolin-induced production of cAMP in EC 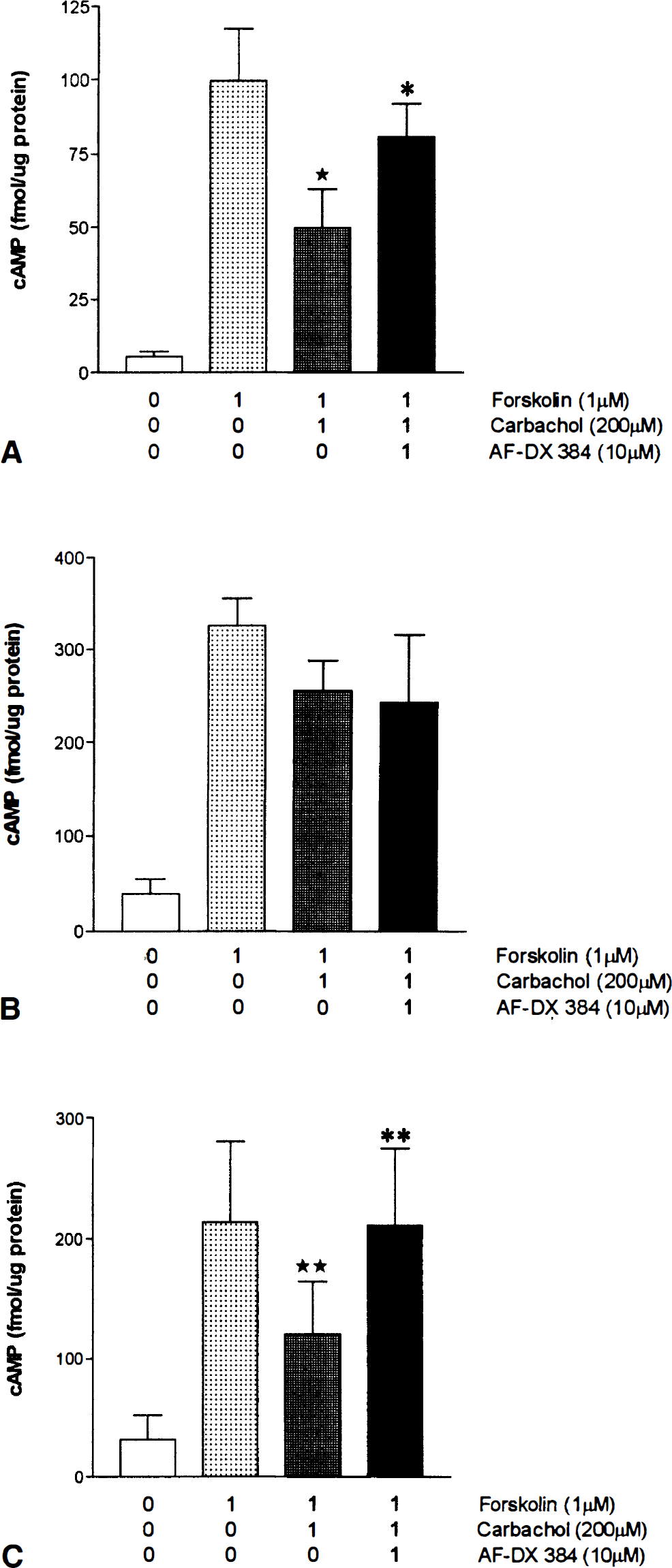
DISCUSSION
This study demonstrates that heterogeneous mAChR subtypes are localized in specific cellular compartments of the human brain microcirculation. Together MV and CAP isolated from postmortem human cerebral cortex were found to express a combination of mAChR, but to lack the m4 subtype. When receptor expression was assessed in more details in microvascular and astroglial cell cultures generated from surgery specimens or fetal brains, the results indicated that human brain EC are endowed with m2 and m5 mAChR subtypes, SMC with m1, m3, and possibly m5 mAChR, whereas all five mAChR were found in human fetal brain AST. The results with mAChR antagonists further indicated that their second messenger coupling efficacy corresponded to that reported previously in EC and SMC cultures from peripheral blood vessels of man (Hawley et al., 1995), or in rat glial cell cultures (Cohen and Almazan, 1994). Altogether, the present results suggest that intracortical MV have the ability to respond to changes in central cholinergic neurotransmission either directly through receptors in the vessel wall itself or indirectly by interacting with receptors located on the perivascular astroglial cells.
The presence of specific binding sites for the mAChR antagonist [3H]quinudidmylbenzilate in collagenase-treated CAP and MV (Estrada et al., 1983) provided the first convincing evidence that mAChR are associated with vascular elements of the brain microrirculation. Although more than one population of mAChR were later suggested (García-Villalón et al., 1991; Linville and Hamel, 1995), there was disagreement as to which mAChR subtypes were present in the brain microvasculature. García-Villalón and colleagues exclusively identified a pharmacologic m4 mAChR in bovine microvascular fractions, but others concluded on the basis of pharmacologic characterization and correlation analyses with the cloned mAChR that the ml, m3, and possibly the m5 receptors were most likely the cerebromicrovascular receptors present, not only in bovine, but also in human cerebromicrovascular fractions (Linville and Hamel, 1995). In the present study, the high sensitivity and selectivity of the RT-PCR approach, together with the demonstration of functional second messenger signaling pathways in the cell cultures, provided unequivocal support for the presence of cerebromicrovascular m1, m3, and m5 mAChR, and further allowed us to identify endothelial m2 mAChR. In contrast, m4 mAChR were found to be exclusively astroglial, a finding that could possibly explain its detection by radioligand binding studies in isolated bovine microvessels (García-Villalón et al., 1991), as these also contain a proportion of attached astroglial cells (for details, see Linville and Hamel, 1995). Most interestingly, the present findings in the human intracortical microvascular bed closely match those reported in a recent RT-PCR study, which showed expression of ml, m2, m3, and m5 mAChR in the rat basilar artery (Phillips et al., 1997). This remarkable similarity in the composition of mAChR between the rat major cerebral arteries and the human intracortical microvessels strongly suggests a high level of conservation for vascular mAChR across species and between intraparenchymal and extraparenchymal blood vessels. It may further imply comparable functions for these receptors in their respective cerebrovascular beds.
The RT-PCR results obtained in the EC cultures showed the presence of mAChR in these cells with a consistent and predominant expression of the m2 and m5 mAChR, a finding that was corroborated by their coupling to the expected downstream signaling pathways but which is at variance with previous studies that could not detect mAChR in brain capillaries, albeit in the rat (Moro et al., 1995; Luiten et al., 1996; Badaut et al., 1997). However, the immunocytochemical demonstration of endothelial m2 mAChR in rat, primate, and human brain capillaries (Levey et al., 1995; Smiley et al., 1998) would argue to the contrary and rather support the present findings. The exclusive mRNA expression of the m2 subtype and the ability of the mAChR antagonist AF-DX 384 (Dörje et al., 1991) to block the inhibitory effect of carbachol on cAMP production (the downstream effect of m2 activation) unequivocally indicated the existence of functional endothelial m2 mAChR. The apparent discrepancy between the present results and the inability of previous radioligand binding studies to detect m2 mAChR in isolated human cerebral microvasculature (Linville and Hamel, 1995) could possibly be explained by the fact that radioligand binding studies, unlike PCR, require that a sizable amount of the receptor protein be present to be detectable. In fact, from the present results, m2 receptors are located in the endothelial but not in the muscular compartment (see below) of the intraparenchymal vascular tissue. It is thus possible that m2 binding sites in a single layer of EC would have been masked by more abundant mAChR subtypes present in both the EC and SMC, or the smooth muscle layers alone. This putative problem of relative abundance between the different mAChR subtypes has been circumvented in the present study by the use of the highly sensitive PCR technique and cell cultures of the different cellular components of the vessels wall.
With regard to mAChR associated with the phosphoinositide pathway, the findings that only the m5 subtype was consistently expressed in human brain EC and that pirenzepine, which at the concentration used will affect not only ml but also the other mAChR subtypes (Dörje et al., 1991), effectively inhibited the IP3 response induced by ACh in these cultures further indicated the presence of endothelial m5 mAChR proteins. No molecular evidence, however, was found in primary and passaged human cerebromicrovascular EC cultures for the m3 mAChR subtype. This finding suggests that its mRNA detection in isolated CAP is probably owing to other cells intimately associated with this isolated microvascular fraction, such as pericytes or AST (see below), rather than to repeated passaging of the cells (Tracey and Peach, 1992). In large cerebral arteries of various species, the m3 mAChR (Dauphin and Hamel, 1990; García-Villalón et al., 1991; for review see Dauphin and Mackenzie, 1995) and the m5 subtype (Hamel et al., 1994; Phillips et al., 1997) have been suggested as possible mediators of the endothelium-dependent, nitric oxide-mediated dilation induced by ACh. Because ACh is also a potent dilator of isolated intracortical arterioles in rat (Dacey and Bassett, 1987), cow, and human, in which nitric oxide-mediated mechanisms appear to be involved (Elhusseiny and Hamel, 1998), the present results suggest that the m5 mAChR is a likely candidate for mediating this response at the microvascular level in man. This receptor is known to share substantial similarities with the m3 mAChR subtype, both in terms of pharmacology (Dörje et al., 1991) and second messenger profile (Felder, 1995). Furthermore, the m5 receptor has been reported to stimulate nitric oxide production when transfected in cells (Wang et al., 1994), a property that could be of interest for a receptor putatively involved in an endothelium-dependent vasodilatory response. Additional studies with m5 mAChR-selective compounds will be of great help in clarifying this issue when they become available.
Isolated MV, which unlike CAP are enriched with vessels with a smooth muscle layer, were found to express ml and m3 mAChR subtypes, a finding corroborated by the expression of these same receptors in microvascular SMC cultures. Although a role for these receptors in human brain microcirculation still has to be defined, both subtypes have been implicated in the ACh-induced vasoconstriction of endothelium-denuded vessels, with the ml subtype being particularly important at the level of cerebral blood vessels (for review see Dauphin and Mackenzie, 1995). In spite of the detection of mRNA for the m2 receptor in SMC, carbachol systematically failed to significantly block the forskolin induced cAMP production in these cells, although it was very effective in EC and AST cultures. This observation suggests that the m2 mRNA is either amplified from the small population of contaminating EC present in the primary SMC cultures, or that functional m2 mAChR exist in a very limited population in SMC, too small to produce a significant effect on the cAMP production in a biochemical assay. The presence of m5 mAChR in both EC and SMC cultures similarly indicate that further investigations will be needed to verify whether the m5 mAChR is genuinely expressed in SMC or whether its detection in these cells is because of the small proportion of contaminating EC. However, as for other microvascular receptors (Riad et al., 1998), it is also possible that the same receptor subtype is expressed in both the EC and SMC compartments.
Our finding that human brain AST express all five mAChR extends previous pharmacologic and molecular studies that showed the presence of m2, m3, and m5 mRNA in human astrocytoma cells (Guizzetti et al., 1996), and ml and m3 in murine astroglial cells in culture (André et al., 1994). In addition to the ml, m2, m3, and m5 subtypes, we further show expression of the m4 mAChR in fetal human brain AST. Its presence in adult human brain AST should be further confirmed, as fetal brain tissue and AST may express different receptor populations than those seen in the adult (Van der Zee et al., 1993; Hohmann et al., 1995), a possibility raised by the absence of m4 mAChR message in either isolated MV or CAP that originated form adult brain. Astroglial cells fulfill a variety of functions within the central nervous system (Van der Zee et al., 1993; Guizzetti et al., 1996) and the multiplicity of mAChR associated with these cells could be a reflection of such a polyvalence. More specifically, perivascular astroglial cells have been involved in the regulation of microvascular tone, metabolic homeostasis, and blood–brain barrier permeability (Tao-Cheng et al., 1987; Tsacopoulos and Magistretti, 1996; Alkayed et al., 1997), all of which could be regulated by ACh because a large proportion of neurovascular cholinergic nerve terminals are in fact associated with the perivascular AST (Vaucher and Hamel, 1995).
PHYSIOLOGIC IMPLICATIONS
This study reports the presence of functional cerebromicrovascular and astroglial mAChR in human cerebral cortex, corresponding in part to those able to mediate vasomotor responses in other cerebrovascular beds (for review see Dauphin and MacKenzie, 1995). Cortical MV and CAP have been identified as a target for basalocortical cholinergic nerve terminals in both rat (Vaucher and Hamel, 1995) and human (Tong and Hamel, 1997). The present results further indicate that these intraparenchymal vessels are equipped with adequate receptors to directly modulate blood flow in response to cholinergic stimulation, in agreement with many physiologic studies involving stimulation of basal forebrain neurons (for review see Sato and Sato, 1995). Furthermore, dysfunctions in this system, like those seen during neurodegenerative disorders such as Alzheimer's disease, could impact on the regulation of the local cortical microcirculation and result in inadequate perfusion, capillary morphology, or blood–brain barrier permeability.
