Abstract
The extracellular concentrations of glutamate and its co-agonist for the N-methyl-d-aspartate (NMDA) receptor, glycine, may be under the control of amino acid transporters in the ischemic brain, However, there is little information on changes in glycine and glutamate transporters in the hippocampal CA1 field of gerbils with transient forebrain ischemia. This study investigated the spatial and temporal expressions of glycine transporter 1 (GLYT 1) and three glutamate transporter (excitatory amino acid carrier 1, EAAC 1; glutamate/aspartate transporter, GLAST; glutamate transporter 1, GLT1) mRNA in the gerbil hippocampus after 3 minutes of ischemia. The GLYT1 mRNA was transiently upregulated by the second day after ischemia in astrocytelike cells in close vicinity to hippocampal CA1 pyramidal neurons, possibly to reduce glycine concentration in the local extracellular spaces. The EAAC1 mRNA was abundantly expressed in almost all pyramidal neurons and dentate granule cells in the control gerbil hippocampus, whereas the expression level in CA1 pyramidal neurons started to decrease by the fourth day after ischemia in synchrony with degeneration of the CA1 neurons. The GLAST and GLT1 mRNA were rather intensely expressed in the dentate gyrus and CA3 field of the control hippocampus, respectively, but they were weakly expressed in the CA1 field before and after ischemia. As GLAST and GLT1 play a major role in the control of extracellular glutamate concentration, the paucity of these transporters in the CA1 field may account for the vulnerability of CA1 neurons to ischemia, provided that the functional GLAST and GLT1 proteins are also less in the CA1 field than in the CA3 field. This study suggests that the amino acid transporters play pivotal roles in the process of delayed neuronal death in the hippocampal CA1 field.
Keywords
Transient global ischemia leads to delayed neuronal death in the hippocampal CA1 field of gerbil (Kirino, 1982; Schmidt-Kastner and Freund, 1991). Although the precise molecular mechanisms underlying this phenomenon are as yet unknown, a large body of evidence suggests that glutamate plays a crucial role in ischemic neuronal death (Benveniste et al., 1984; Globus et al., 1988). Exposure of neurons to glutamate causes an increase in the concentration of intracellular free Ca2+ (MacDermott et al., 1986) and initiates the process of the delayed neuronal death (Choi, 1985). In the hippocampal CA1 field, currents through N-methyl-d-aspartate (NMDA) receptor channels are potentiated in response to anoxia or ischemia (Hori et al., 1991; Crépel et al., 1993), in agreement with earlier data demonstrating a postischemic augmentation of Ca2+ influx in CA1 pyramidal neurons (Andiné et al., 1988). These findings indicate that the NMDA receptor plays a key role in early processes of delayed neuronal death of CA1 neurons. Because the NMDA receptor is known to require glycine as a co-agonist (Kemp and Leesson, 1993) and its function is controlled by glycine in the synaptic cleft (Supplisson and Bergman, 1997), extracellular glycine as well as glutamate concentrations may be important determinants of delayed neuronal death.
Extracellular glutamate concentration is regulated by glutamate transporters. To date, five glutamate transporters (EAAT1–5) have been identified in human (Bar-Peled et al., 1997; Arriza et al., 1997). In rat, three transporters that correspond to EAAT1–3 have been cloned: glutamate/aspartate transporter (GLAST, Strock et al., 1992), glutamate transporter 1 (GLT1, Pines et al., 1992), and excitatory amino acid carrier 1 (EAAC 1, Kanai and Hediger, 1992). In addition, glycine concentration in the hippocampus is thought to be regulated by glycine transporter 1 (GLYT1) on the basis of its distribution pattern (Adams et al., 1995). These transporters may participate in the processes of neuronal death or survival in the ischemic brain by regulating extracellular glutamate and glycine concentrations. However, little is known about the normal distributions of these transporter mRNA in the gerbil hippocampus or about changes in the expressions of these transporters in the gerbil hippocampus after transient global ischemia.
Three minutes of forebrain ischemia in gerbil yields a loss of nearly half the CA1 neurons on postischemic day 7 if brain temperature is kept at 37.0° ± 0.2°C (Wen et al., 1995a, 1995b, 1998; Peng et al., 1998; Sakanaka et al., 1998). In this gerbil ischemia model, the degeneration of hippocampal CA1 neurons appears to proceed more slowly than that in the conventional 5-minute ischemia model in which brain temperature is not maintained at 37.0° ± 0.2°C during ischemia (Kirino, 1982). It is speculated that changes in the glutamate and glycine transporters also take place gradually in the hippocampus of 3-minute ischemic gerbils, depending on the stages of neuronal degeneration in the CA1 field. If this speculation is the case, one may easily detect the spatial and temporal profiles of glutamate and glycine transporter expressions within the hippocampus during 7 days after 3 minutes of forebrain ischemia.
In the current study, we investigated the differential expressions of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the hippocampus of 3-minute ischemic gerbils using in situ hybridization histochemistry.
MATERIALS AND METHODS
Occlusion of the common carotid arteries
Male Mongolian gerbils weighing 70 to 80 g (about 12 weeks of age) were used in the present study. The following experiments were conducted in accordance with the Guide for Animal Experimentation at Ehime University School of Medicine. The animals were anesthetized with 1.5% halothane in a 4:3 mixture of nitrous oxide and oxygen and placed in a stereotaxic apparatus. Occlusion of the common carotid arteries was performed as previously described (Sano et al., 1994; Wen et al., 1995a, 1995b, 1996, 1998; Kotani et al., 1996; Lim et al., 1997; Matsuda et al., 1997; Peng et al., 1998; Sakanaka et al., 1998). Briefly, both common carotid arteries were exposed through a ventral midline incision and separated carefully from the adjacent veins and nerves. Immediately after the termination of inhalation anesthesia, the common carotid arteries were clamped for 3 minutes with aneurysm clips.
During forebrain ischemia, brain temperature has been shown to fall differently in individual animals, thereby affecting the number of viable CA1 neurons after ischemia (Mitani et al., 1991). To avoid the effect of unstable brain temperature on ischemic neuronal loss, we kept brain and rectal temperatures at 37.0° ± 0.2°C while clamping the common carotid arteries. This enabled us to induce an invariable neuronal damage in the hippocampal CA1 field even after 3 minutes of ischemic insult (Sano et al., 1994; Wen et al., 1995a, 1995b, 1996; Kotani et al., 1996; Lim et al., 1997; Matsuda et al., 1997; Peng et al., 1998; Sakanaka et al., 1998).
Preparation of complementary RNA probes
Rat EAAC1, GLAST, and GLT1 cDNA fragments were obtained by using the polymerase chain reaction (PCR), and the PCR products were subcloned into pGEM-T vector (Promega, Madison, WI, U.S.A.). These constructs contained the following cDNA fragments: 1,163 through 1,573 of rat EAAC1 cDNA; 1,021 through 1,547 of rat GLAST cDNA; and 320 through 802 of rat GLT1 cDNA (starting codon is 1). For detection of GLYT1, a construct containing full-length mouse GLYT1 cDNA was used (Adams et al., 1995). All cDNA fragments were sequenced and confirmed to be identical with the corresponding genes. Antisense probes were transcribed with the use of T7, T3, or Sp6 polymerase in the presence of 35S-UTP (New England Nuclear, Boston, MA, U.S.A.) on the plasmids linearized with appropriate restriction enzymes. For control experiments, sense probes were also transcribed in the reverse direction. The probes were purified by ethanol precipitation and used for in situ hybridization (see below).
In situ hybridization
On the first, second, fourth, and seventh days after 3 minutes of forebrain ischemia, the gerbils (n = 3 for each group) were decapitated under sodium pentobarbital anesthesia (50 mg/kg, intraperitoneally). Normal gerbils and rats (n = 3 for each) were also decapitated under the same anesthetic condition. The brains were quickly removed and immediately frozen on powdered dry ice. Serial sections 20 µm thick were cut on a cryostat, thaw-mounted onto silane-coated slides, and stored at −80°C until use. The sections were air dried and fixed for 15 minutes in 4% paraformaldehyde in 0.1 mol/L phosphate buffer (PB), washed twice in PB for 5 minutes, and treated with 10 ug/mL proteinase K in a solution containing 50 mmol/L Tris-HCl and 5 mmol/L EDTA for 5 minutes. After a rinse in PB for 5 minutes, the sections were fixed again in the same fixative for 5 minutes, then treated with 0.25% acetic anhydrate in 0.1 mol/L triethanolamine for 10 minutes, washed in PB for 5 minutes, and dehydrated.
Hybridization with radioactive cRNA probes (5 × 106 dpm/mL) was performed at 55°C overnight in a solution containing 50% formamide, 20 mmol/L Tris-HCl, 0.3 mol/L NaCl, 1× Denhardt's solution, 10% dextran sulfate, 500 µg yeast tRNA, and 200 µg/mL salmon sperm DNA. After hybridization, the sections were washed in 5× sodium chloride/sodium citrate (SSC), 1% mercaptoethanol (pH 7.0) at 55°C for 15 minutes, and in high-stringency wash solution containing 50% formamide, 2× SSC, and 10% mercaptoethanol at 65°C for 30 minutes. They were washed three times in RNase buffer (10 mmol/L Tris-HCl, 1 mmol/L EDTA, 0.5 mol/L NaCl) at 37°C (10 minutes for each) and treated with RNase A (1 µg/mL) in RNase buffer at 37°C for 30 minutes. They were then washed again RNase buffer for 10 minutes. High-stringency wash was again performed, and the sections were washed in 2× SSC and in 0.1× SSC at room temperature for 10 minutes each, and dehydrated. The sections were coated with Kodak NTB-2 emulsion for liquid autoradiography (Kodak, Rochester, NY, U.S.A.), exposed for 1 to 4 weeks, and then developed. After washing in water and air drying, they were counterstained with thionin, and coverslipped with Permount. Relative changes in mRNA expression were then quantified by determining, with the NIH image analysis system, the ratio of the optical density of a specified region in the ischemic brain to that in the control brain.
Double labeling
Gerbils (n = 3 per group) were perfused transcardially with 4% paraformaldehyde in 0.1 mol/L PB (pH 7.4). The brains were excised and immersed overnight in 0.1 mol/L PB (pH 7.4) containing 30% sucrose at 4°C. Serial sections 20 µm thick were cut on a cryostat, thaw-mounted onto silane-coated slides, and stored at −80°C until use. First, in situ hybridization was performed until the final 10-minute wash in 0.1× SSC as described above. Then, the brains were processed for immunohistochemistry with an anti—glial fibrillary acidic protein (GFAP) antibody (Sigma, St. Louis, MO, U.S.A.). Briefly, the sections were (I) washed with 0.1 mol/L phosphate-buffered saline (PBS) for 30 minutes and incubated with the anti-GFAP antibody diluted 1:100 with PBS containing 1% normal goat serum and 0.1% Triton-X 100 for 48 hours at 4°C; (2) incubated with biotinylated anti-rabbit IgG and peroxidase-conjugated streptavidin (DAKO, Glostrup, Denmark) overnight at 4°C; and (3) subjected to a modified version of the cobalt-glucose oxidase-diaminobenzidine intensification method (Sakanaka et al., 1987). After GFAP immunohistochemistry, the sections were dried and coated with Kodak NTB-2 emulsion for liquid autoradiography (Kodak), exposed for 1 to 4 weeks, and then developed for visualization of hybridization signals. After washing in water and air drying, they were coverslipped without counterstaining.
Northern blot analysis
For Northern blot analysis, mRNA was isolated from the gerbil brain with the use of Isogen (Nippon Gene, Tokyo, Japan) and Oligotex-dT30 (Takara, Tokyo, Japan). Aliquots (3 µg) of mRNA were separated on 1% agarose formaldehyde gels and transferred onto nylon membrane (Hybond-N; Amersham Corp, Arlington Heights, IL, U.S.A.). DNA fragments for GLYT1, EAAC1, GLAST, and GLT1 were obtained by cutting the plasmids that were used for in situ hybridization with appropriate restriction enzymes, and they were labeled by random priming (Amersham) with [α-32P]dCTP (3,000 Ci/mmol; Amersham). Hybridization was carried out at 42°C overnight in 50% formamide, 5× SSC, 0.1% sodium dodecyl sulfate (SDS), 50 mmol/L sodium phosphate, 5× Denhardt solution, and 100 µg/mL salmon sperm DNA. The blots were washed in the following ways: (1) 5 minutes at room temperature in 2× SSC, 0.5% SDS, (2) 5 minutes at room temperature in 2× SSC, 0.1% SDS, and (3) 30 minutes at 42°C in 0.1× SSC, 0.5% SDS. The nylon membrane was visualized using an image analyzer (model BAS1000; Fuji Photo Film Co, Tokyo, Japan).
Histopathologic study of the hippocampal CA1 region
At the second, fourth, and seventh days after forebrain ischemia or sham operation, gerbils (n = 8 per group) were perfused transcardially with 4% paraformaldehyde in 0.1 mol/L PB (pH 7.4). A brain region including the dorsal hippocampus from 0.5 to 1.5 mm posterior to bregma was removed and kept in the same fixative overnight at 4°C. The hippocampus was embedded in paraffin, and 5-µm serial frontal sections were stained with 0.1% cresyl violet. All neurons with intact morphologic appearance along 1 mm linear length of the CA1 field in six serial coronal sections (1.20 to 1.23 mm posterior to bregma) were counted. The mean number of neurons was calculated in each animal.
In situ detection of DNA fragmentation: terminal deoxynucleotidyl transferase-mediated 2'-deoxyuridine 5'-triphosphate-biotin nick end labeling
On the seventh day after 3 minutes of ischemia, nearly half of the hippocampal CA1 neurons were lost and some of the remaining CA1 neurons appeared to undergo further degeneration. To estimate the number of CA1 neurons degenerating on the second, fourth, and seventh days after ischemia, two additional paraffin sections from each animal in the sham-operated and ischemic groups were processed for in situ detection of DNA fragmentation (terminal deoxynucleotidyl transferase-mediated 2'-deoxyuridine 5'-triphosphate-biotin nick end labeling [TUNEL] staining). Briefly, the sections were (1) deparaffinized in xylene and in a graded series of ethanol; (2) washed two times in 0.05 mol/L PBS for 10 minutes and then incubated with 20 µg/mL of proteinase K (Sigma) for 15 minutes; (3) rinsed with distilled water four times for 2 minutes each and quenched in 2.0% hydrogen peroxide for 10 minutes; (4) rinsed with PBS twice for 5 minutes each and incubated with equilibration buffer (in situ apoptosis detection kit, Oncor, Gaithersburg, MD, U.S.A.), and then incubated with a mixture of terminal deoxynucleotidyl transferase and reaction buffer containing digoxigenin-dUTP in a humidified chamber for 1 hour at 37°C; (5) immersed in wash buffer for 10 minutes and washed with PBS three times for 5 minutes each; (6) incubated with anti—digoxigenin-peroxidase for 1 hour at room temperature; and (7) rinsed with PBS three times for 5 minutes each and exposed to 0.05% diaminobenzidine and 0.02% hydrogen peroxide. All TUNEL-positive neurons along a 1-mm linear length of the CA1 field in the two serial coronal sections were counted, and the mean number of positive neurons was calculated in each animal.
RESULTS
Expression patterns of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the gerbil and rat brains
To investigate the specificity of cRNA probes for detection of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the gerbil brain, we conducted Northern blot analysis (Fig. 1). Each probe detected a single band, and the estimated sizes of the bands were 3.8 kilobases (kb) for GLYT1, 3.7 kb for EAAC1, 4.5 kb for GLAST, and 11 kb for GLT1. These values coincided well with those reported previously in rat (Zafra et al., 1995; Velaz-Faircloth et al., 1996; Strock et al., 1992; Pines et al., 1992).

Northern blot analysis using the probes for detection of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the gerbil brain. Note that each probe recognizes a single band. The positions of
To further confirm the specificity of the probes, in situ hybridization histochemistry on horizontal sections from the rat and gerbil brains was performed with the probes. In the rat brain, GLYT1 mRNA was expressed mainly in fiber tracts such as the corpus callosum and cerebellar medulla, indicating that it is produced in glial cells (Fig. 2A). The distribution pattern of GLYT1 mRNA in the rat brain appeared to be identical with that in the mouse brain (Adams et al., 1995) and the localization of GLYT1 mRNA in the gerbil brain was also quite similar to that in the rat brain (Fig. 2B). These findings suggest that the probe used in this study recognizes GLYT1 mRNA in the mouse, rat, and gerbil brains. The sense probe showed no hybridization signals (Fig. 2C).
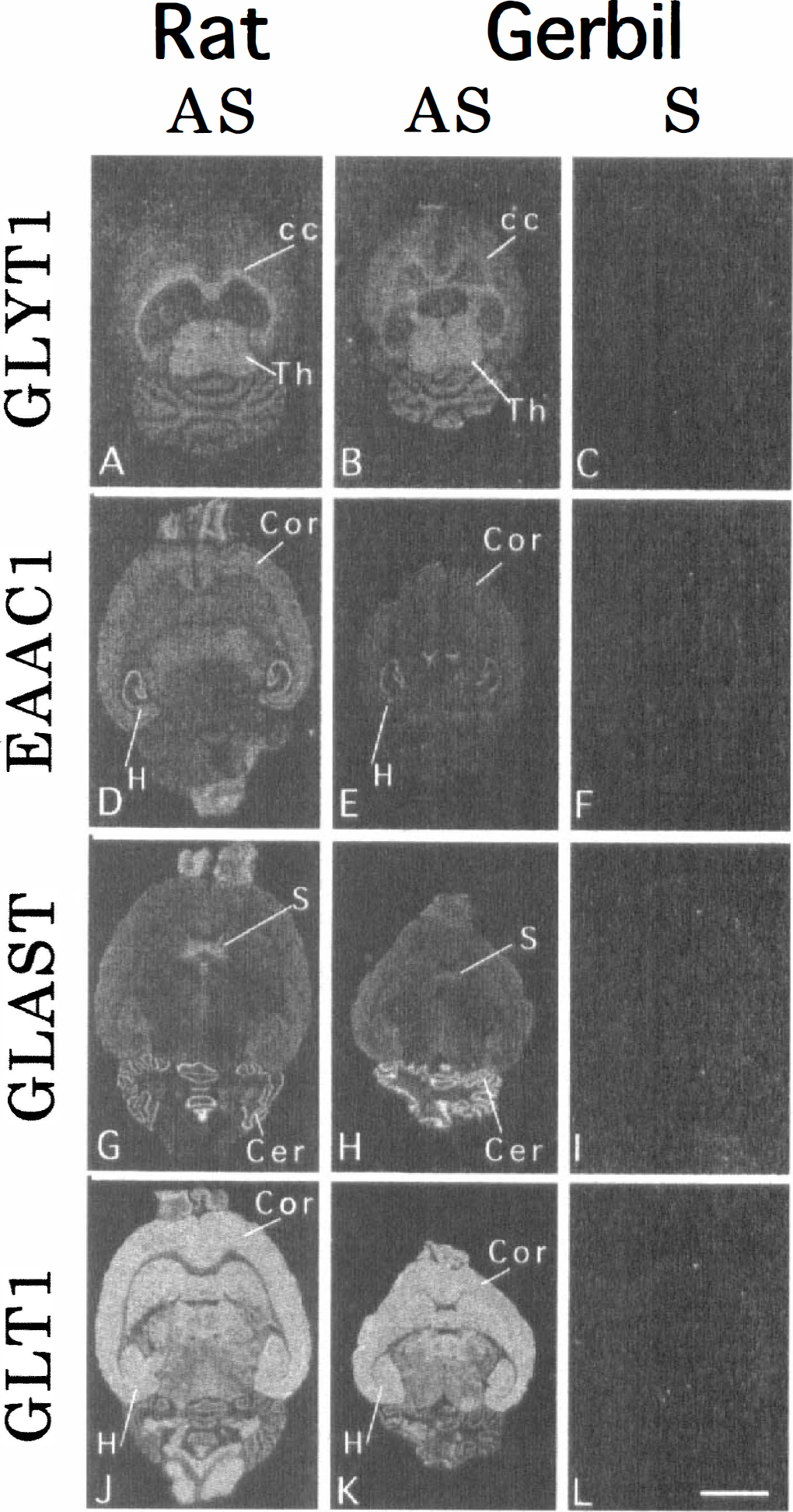
Dark-field micrographs showing the expression of GLYT1, EAAC1, GLAST, and GLT1 mRNAs in the rat brain
The probes for detection of three glutamate transporters enabled us to see characteristic expression patterns of the transporters in the rat and gerbil brains (Fig. 2D, E, G, H, J and K). Intense EAAC1 mRNA expressions were observed in the pyramidal cell layer and dentate granule cell layer of the hippocampus, whereas the cerebral cortex and olfactory bulb exhibited moderate intensities of EAAC1 mRNA expression (Fig. 2D and E). The Purkinje cell layer of the cerebellum, the septum, and the olfactory bulb were intensely labeled with the probe for detection of GLAST mRNA (Fig. 2G and H). Abundant expression of GLT1 mRNA was found throughout the central nervous system of rat and gerbil; intense GLT1 mRNA expressions were observed in the cerebral cortex, basal ganglia, and hippocampus (Fig. 2J and K). Thus, the distribution patterns of the mRNA encoding EAAC1, GLAST, and GLT1 in the gerbil brain were similar to those in the rat brain. The sense probes detected no hybridization signals (Fig. 2F, I and L). These findings suggest that the three antisense probes to localize the mRNA of the rat glutamate transporters can be applied to the gerbil brain.
Expressions of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the normal gerbil hippocampus
Because there is no report on the distribution patterns of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the normal gerbil hippocampus, we first investigated them. In the gerbil hippocampus, GLYT1 mRNA was weakly expressed in glial cells adjacent to pyramidal neurons of the CA1-3 fields and in those adjacent to granule cells of the dentate gyrus (Fig. 3A). In addition, glial cells in the corpus callosum, fimbria hippocampi, and internal capsule were strongly labeled with the antisense probe for detection of GLYT1 mRNA (Fig. 3A). Microautoradiography revealed that silver grains were mainly confined to astrocytelike cells in the corpus callosum (Fig. 3B, arrows). This finding was further confirmed by the simultaneous demonstration of GLYT1 mRNA and GFAP, a specific marker for astrocytes. The double-labeling method showed that abundant silver grains were located on GFAP-positive cells (Fig. 3C, arrows), indicating that astrocytes expressed GLYT1 mRNA.
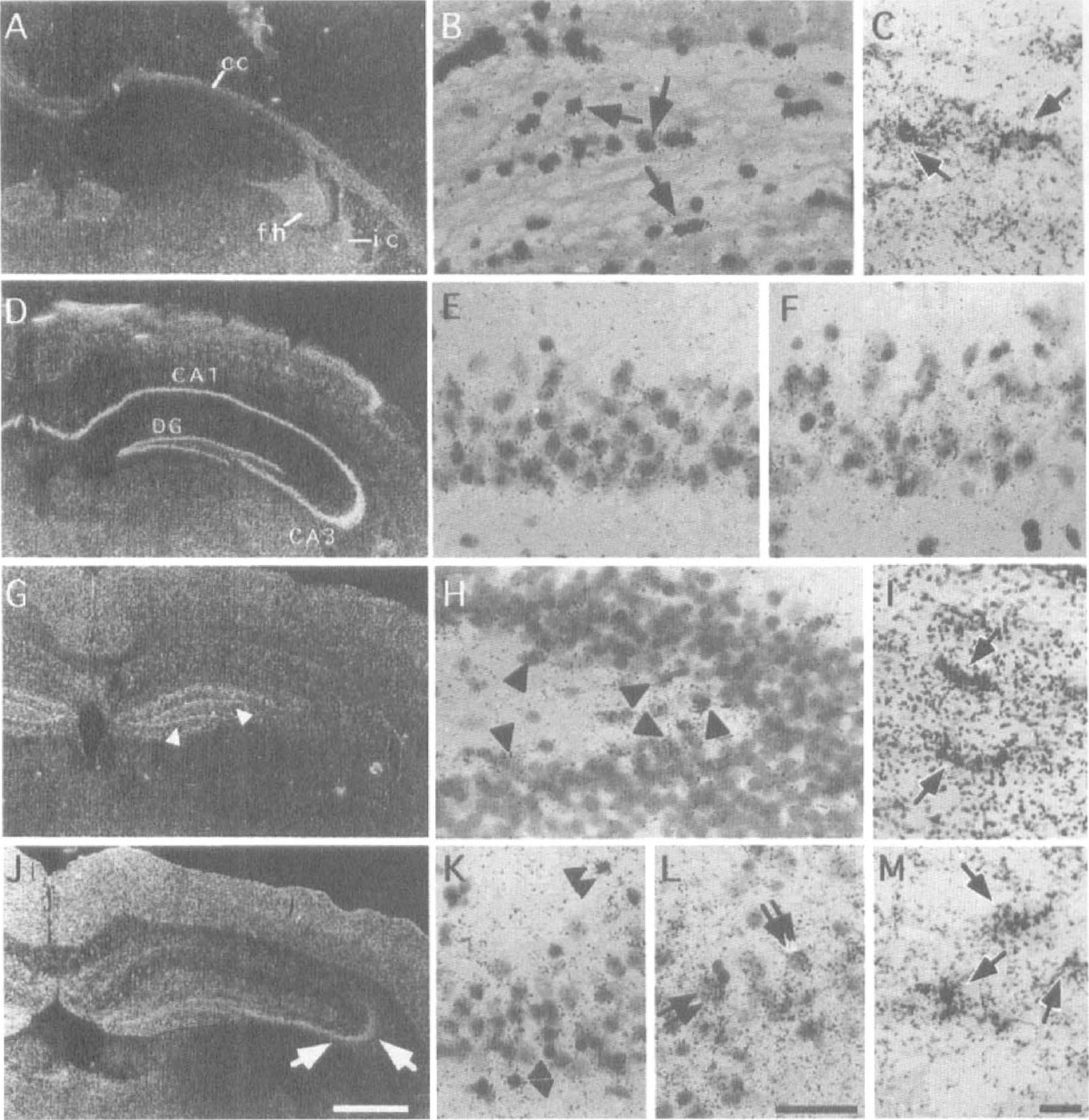
Photomicrographs showing the expression of GLYT1
Strongly expressed EAAC1 mRNA was observed in the pyramidal cell layers of the CA1–3 fields and in the granule cell layer of the dentate gyrus (Fig. 3D). Microautoradiography revealed that pyramidal neurons in the CA1 (Fig. 3E) and CA3 (Fig. 3F) fields expressed EAAC1 mRNA abundantly. Although GLAST mRNA was expressed in many glial cells throughout the gerbil hippocampus, the dentate gyrus expressed more abundant GLAST mRNA than the other parts of the hippocampus (Fig. 3G). An intense GLAST mRNA expression was noted just beneath the granule cell layer of the dentate gyrus (Fig. 3G, arrowheads). By microautoradiography, astrocytelike cells adjacent to dentate granule cells were shown to produce a large amount of GLAST mRNA (Fig. 3H, arrowheads). Expression of GLAST mRNA was also seen in the molecular layer of the dentate gyrus (Fig. 3G). In the CA1 field, glial cells with different amounts of GLAST mRNA expression were scattered (Fig. 3G). The double-labeling method demonstrated that GLAST mRNA was expressed in GFAP-positive astrocytes in the corpus callosum (Fig. 3I, arrows).
A characteristic distribution pattern of GLT1 mRNA was exhibited, with an intense expression of GLT1 mRNA noted in the pyramidal cell layer of the CA3 field (Fig. 3J). The expression level in the CA1 field was weaker than that in the other parts, although astrocytelike cells adjacent to pyramidal cells in the CA1 field expressed GLT1 mRNA abundantly (Fig. 3K, double arrowheads). Observation with a bright-field microscope revealed that CA3 pyramidal neurons as well as astrocytelike cells expressed large amounts of GLT1 mRNA (Fig. 3L, double arrows). This finding is in contrast to the study of Rothstein et al. (1994) who suggested a specific expression of GLT1 protein in astrocytes. The double-labeling method showed that GLT1 mRNA was expressed in GFAP-positive astrocytes in the corpus callosum (Fig. 3M). However, the double-labeling method was not sensitive enough to demonstrate the colocalization of transporter mRNA and GFAP in the hippocampal gray matter.
Histochemical changes in the CA1 field after 3 minutes of ischemia
Before investigating changes in the expressions of the transporter mRNA in the hippocampus of gerbils with 3 minutes of ischemia, we confirmed the morphologic changes in the hippocampal CA1 field on the second, fourth, and seventh days after 3 minutes of ischemia. Nissl staining showed no obvious change until the second day after ischemia (Fig. 4A, B, D and E); however, the number of CA1 neurons started to decrease by the fourth day after ischemia (Fig. 4G and H). On the seventh day after ischemia, the CA1 region exhibited an apparent decrease in viable neurons compared with the CA1 field of sham-operated animals (Fig. 4A, B, J and K); the CA1 neuronal density of the sham-operated animals was 240.3 ± 16.0 cells/mm, whereas that of the ischemic gerbils was 136.9 ± 34.1 cells/mm. There was a significant difference in CA1 neuronal density between the two groups (U = 0, P < 0.01) (Fig. 5). Although there were almost no TUNEL-positive cells until the second day after ischemia (Fig. 4C and F), some TUNEL-positive cells appeared by the fourth day after ischemia (Fig. 4I). On the seventh day after ischemia, TUNEL-positive neurons were more numerous in the ischemic hippocampal CA1 field than in the sham-operated control (U = 0, P < 0.001) (Figs. 4L and 5), suggesting that irreversible neuronal degeneration was in progress at this period.
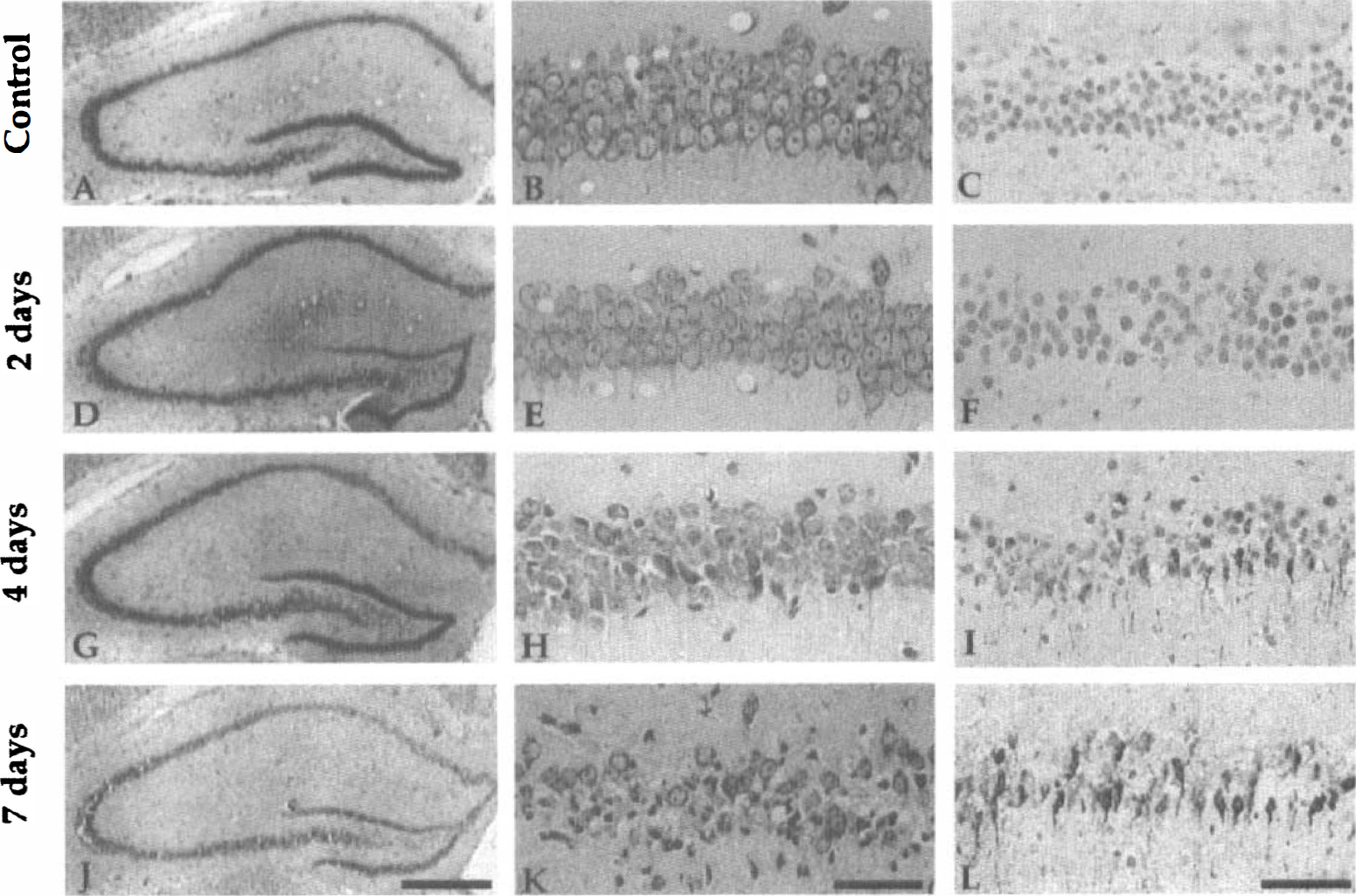
Photomicrographs of the hippocampal CA1 fields of a sham-operated
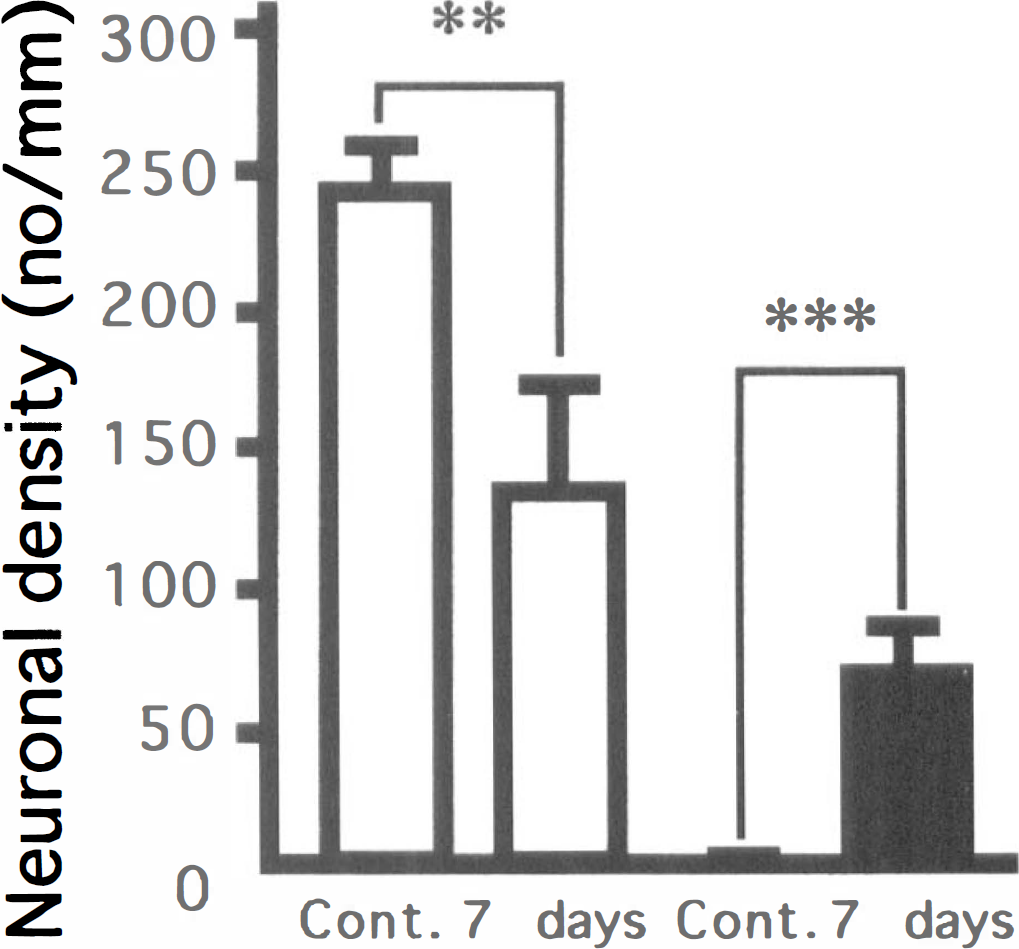
Neuronal density and TUNEL-positive neuron density in the hippocampal CA1 field of sham-operated and ischemic gerbils. ** P < 0.01; *** P < 0.001, significantly different from the sham-operated group (statistical significance tested by the two-tailed Mann-Whitney U-test). (□) Nissl staining; (■) TUNEL staining.
Changes in the expressions of GLYT1, EAAC1, GLAST, and GLT1 mRNA in the gerbil hippocampus after 3 minutes of ischemia
GLYT1. In the sham-operated hippocampus, a weak GLYT1 mRNA expression was observed in the CA1 field (Fig. 6A). After the ischemic insult, GLYT1 mRNA expression was upregulated in the CA1 pyramidal cell layer (Fig. 6B); this increase peaked by the second day after ischemia (Fig. 6C). Thereafter, the expression of GLYT1 mRNA gradually declined (Fig. 6D), reaching the background level by the seventh day after ischemia (Fig. 6E). Microautoradiography revealed that astrocytelike cells in proximity to CA1 pyramidal neurons expressed GLYT1 mRNA (Fig. 6F). Quantitative analysis showed that GLYT1 mRNA expression in the CA1 pyramidal cell layer on the second day after ischemia was almost two times as high as that of the control animals (closed squares in Fig. 6G). No significant changes in the expression of GLYT1 mRNA were noted in the other parts of the hippocampus including the strata radiatum (open squares in Fig. 6G) and lacunosum-moleculare (closed triangles in Fig. 6G) of the CA1 field.
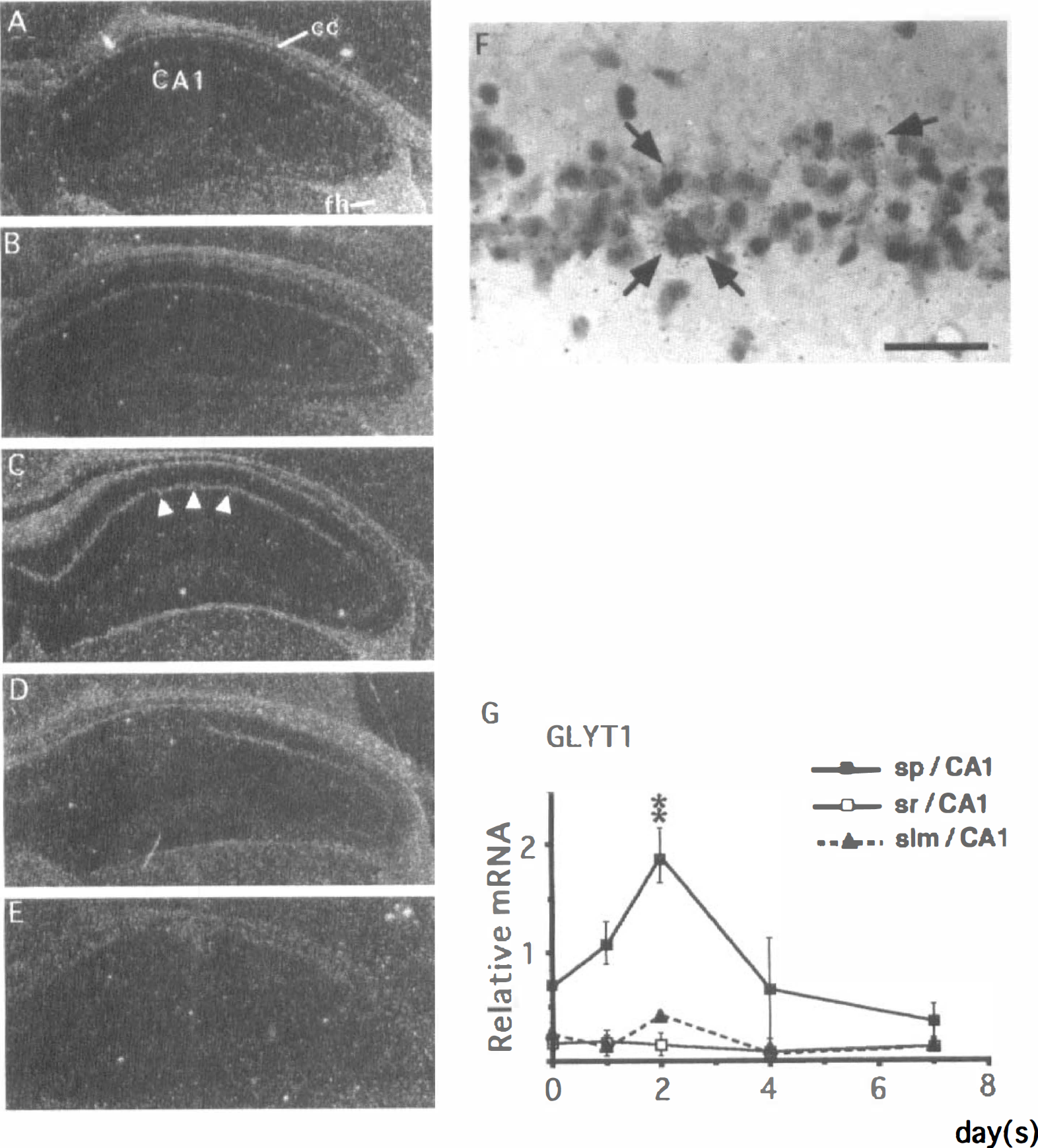
Changes in the expression of GLYT1 mRNA in the gerbil hippocampus after 3 minutes of ischemia.
EAAC1. There was abundant expression of EAAC1 mRNA in the pyramidal cell layers and dentate granule cell layer of the control (sham-operated) hippocampus (Fig. 7A). No significant alteration in EAAC1 mRNA expression was seen in CA1 pyramidal cells until postischemic day 1 (Fig. 7B). After postischemic day 2, EAAC1 mRNA expression in the CA1 pyramidal cell layer started to decrease (Fig. 7C). This decrease became evident on the fourth day after ischemia (Fig. 7D), and the expression of EAAC1 mRNA reached the background level on postischemic day 7 (Fig. 7E). At high magnification, the CA1 pyramidal neurons surviving at the seventh day after ischemia weakly expressed EAAC1 mRNA (Fig. 7F). Quantitative analysis showed that EAAC1 mRNA expression in the CA1 pyramidal cell layer significantly decreased after postischemic day 2 (closed squares in Fig. 7G). In contrast, no significant changes were observed in pyramidal neurons of the CA3 field (open circles in Fig. 7G) and in granule cells of the dentate gyrus (shaded triangles in Fig. 7G).
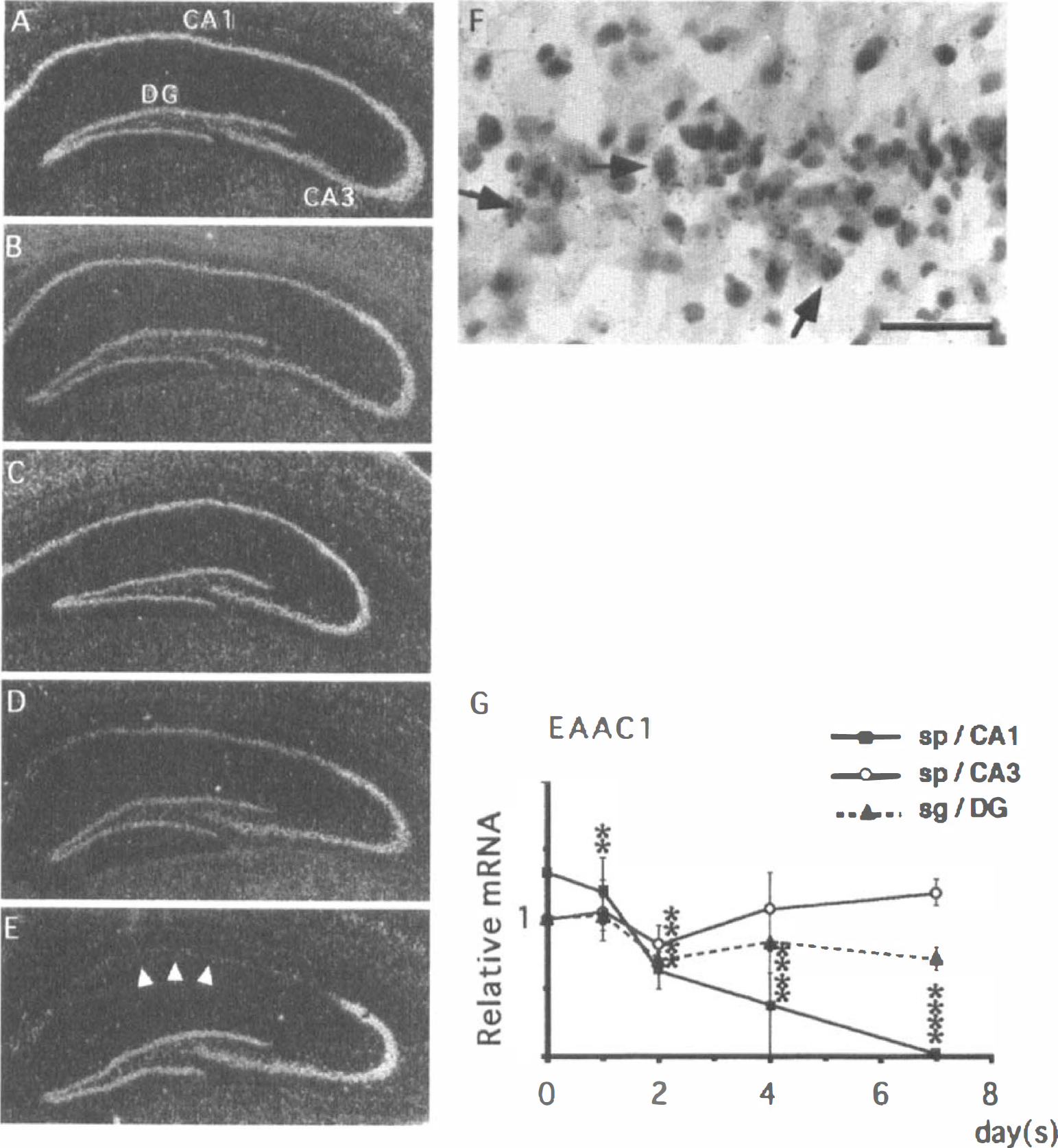
Changes in the expression of EAAC1 mRNA in the gerbil hippocampus after 3 minutes of ischemia.
GLAST. In the CA1 region of the control hippocampus, astrocytelike cells expressing GLAST mRNA were scattered (Fig. 8A). No substantial changes in GLAST mRNA expression were noted until the fourth day after ischemia (Fig. 8A—D and H). However, on postischemic day 7, GLAST mRNA expression significantly increased in intensity in the alveus and stratum lacunosum-moleculare (Fig. 8E). Microautoradiography revealed that astrocytelike cells expressed GLAST mRNA abundantly in both regions (Fig. 8F and G). Quantitative analysis showed that GLAST mRNA expression in the stratum lacunosum-moleculare on postischemic day 7 was approximately four times as high as that of the control animals (closed triangles in Fig. 8H). In addition, GLAST mRNA expression was also increased in the stratum pyramidale of the CA1 field (closed squares in Fig. 8H) after 7 days. There are no significant changes in GLAST mRNA expression within the stratum moleculare of the dentate gyrus (open triangles in Fig. 8H)
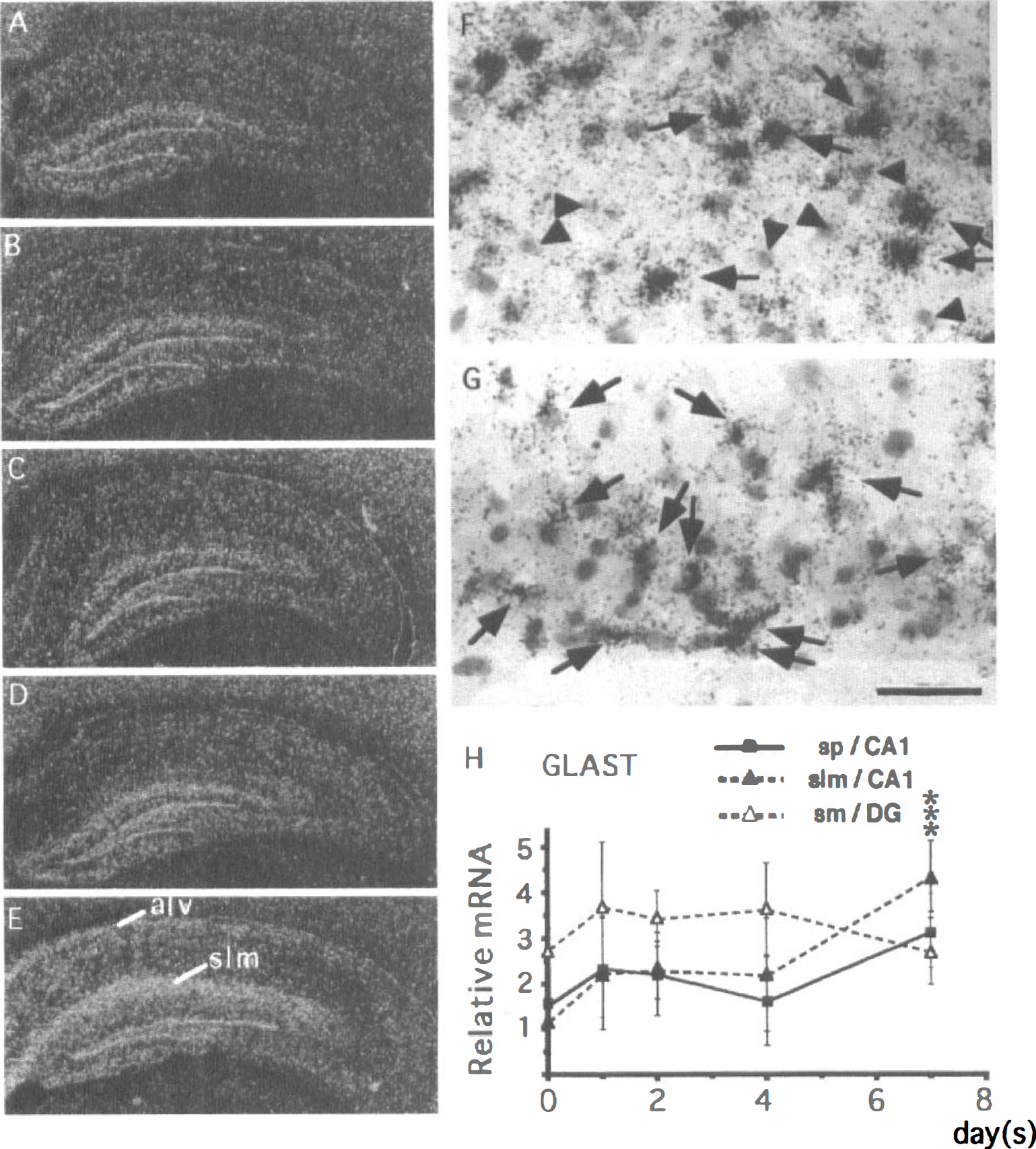
Changes in the expression of GLAST1 mRNA in the gerbil hippocampus after 3 minutes of ischemia.
GLT1. There were no significant changes in GLT1 mRNA expression until postischemic day 2 (Fig. 9A—C). However, after postischemic day 4, GLT1 mRNA expression levels in the strata radiatum, pyramidale, and oriens began to decline (Fig. 9D). By the seventh day after ischemia, GLT1 mRNA expression increased in the alveus and decreased markedly in the strata radiatum, pyramidale, and oriens (Fig. 9E). Microautoradiography showed that in the stratum lacunosum-moleculare, astrocytelike cells abundantly expressed GLT1 mRNA by the seventh day after ischemia (Fig. 9F, arrows). In contrast, astrocytelike cells in the stratum radiatum weakly expressed GLT1 mRNA at this period (Fig. 9G, arrows). Quantitative analysis showed that GLT1 mRNA expression significantly decreased in the stratum radiatum of the CA1 field (open squares in Fig. 8H) on postischemic day 7. In contrast, there were no significant differences in GLT1 mRNA expression within the stratum lacunosum-moleculare of the CA1 field (closed triangles in Fig. 8H) and the stratum pyramidale of the CA3 field (open circles in Fig. 8H).
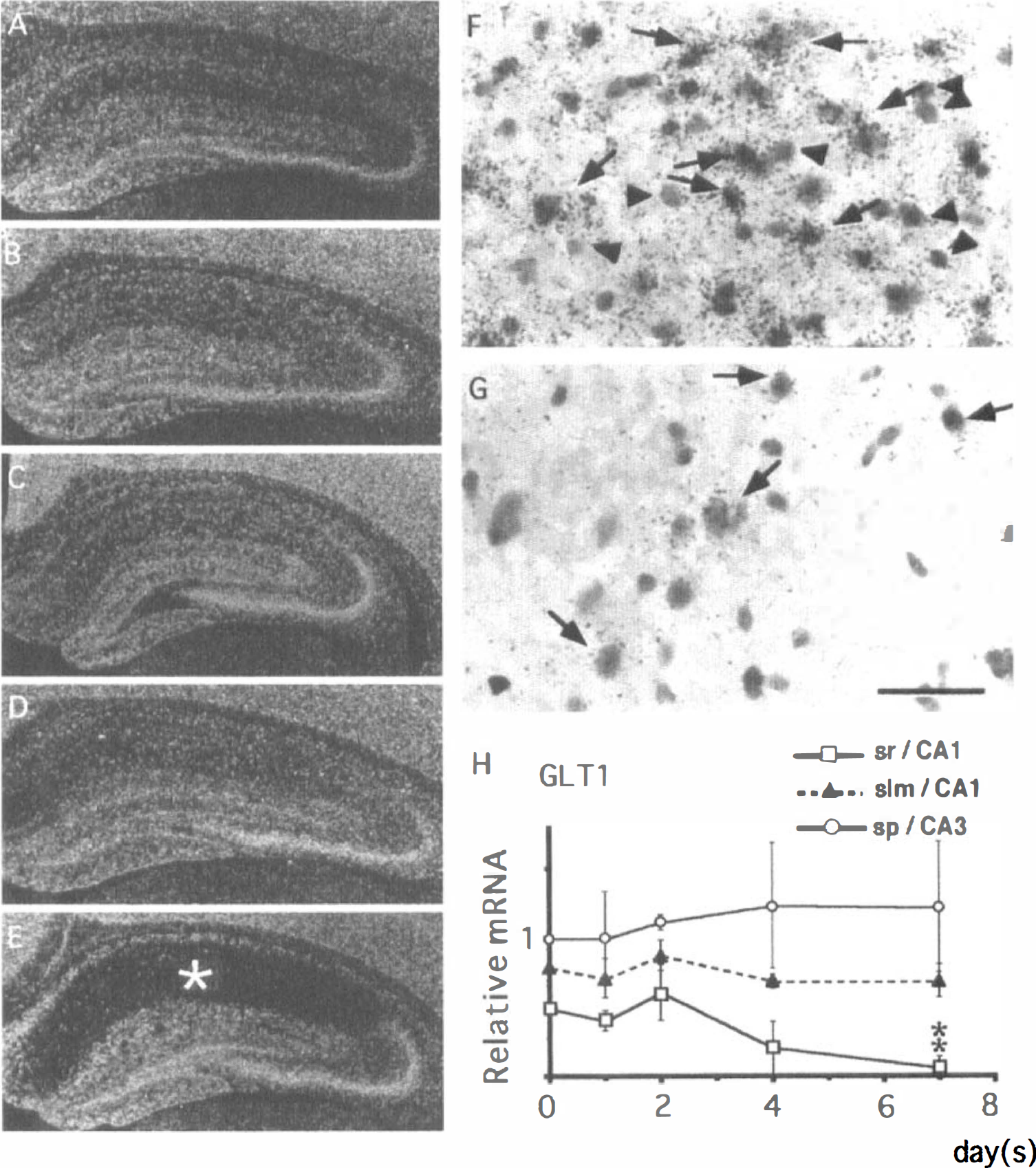
Changes in the expression of GLT1 mRNA in the gerbil hippocampus after 3 minutes of ischemia.
DISCUSSION
The present study focused on the expressions of four amino acid transporter mRNA in the hippocampus of gerbils with 3 minutes of forebrain ischemia. Although changes in the expressions of certain mRNA generally reflect those in the translated proteins, one must take consideration of the fact that in the ischemic brain protein synthesis, but not gene transcription, is frequently impaired (Thilmann et al., 1986; Xie et al., 1989; Widmann et al., 1992; Furuta et al., 1993). Therefore, the changes in the expressions of the transporter mRNA as demonstrated by the present in situ hybridization histochemistry may not be necessarily accompanied by coordinated alterations in the translation of the transporter proteins. With this caveat in mind, the implications of the present experimental findings are discussed.
The transporter GLYT1 has been considered to play an important role in the regulation of NMDA receptor function, which requires glycine as a co-agonist (Kemp and Leesson, 1993). In fact, Virgo and Belleroche (1995) have reported a loss of GLYT1 mRNA expression in the spinal cord of humans with amyotrophic lateral sclerosis, suggesting that the deficit affects synaptic inactivation of glycine and potentially promotes the action of glutamate at NMDA receptor sites. In addition, Supplisson et al. (1997) have shown that glycine uptake by GLYT1 dramatically suppresses NMDA receptor currents by reducing glycine concentrations in extracellular spaces where amino acid diffusion is restricted. They suggest that GLYT1 is capable of desaturating glutamate-mediated neural transmission. The extracellular concentration of glycine in the CA1 field of the rat hippocampus increased approximately three times above the control at 20 minutes after cerebral ischemia (Andiné et al., 1988). Therefore, the significant upregulation of GLYT1 mRNA as observed on the first and second days after ischemia in the present study may contribute to keeping the extracellular concentration of glycine in the CA1 field low and to preventing the activation of NMDA receptor in situ, provided that the increase in GLYT1 mRNA expression leads to stimulation of GLYT1 protein synthesis.
There are two possible mechanisms by which GLYT1 mRNA expression in glial cells is regulated. One is that an increase in extracellular glycine concentration around CA1 pyramidal neurons, as reported by Andiné et al. (1988), directly enhances GLYT1 mRNA expression in the adjacent glial cells. Another possibility is that an unknown factor released from the CA1 neurons after transient ischemia facilitated the GLYT1 mRNA expression. In support of this, Zafra et al. (1997) have shown the existence of a regulatory cross talk between neurons and glia to initiate and sustain the glial expression of GLYT1.
Expression of EAAC1 mRNA in the CA1 field was not upregulated at early postischemic periods when neuronal death or survival is determined in the field, and it began to decline in synchrony with the degeneration of hippocampal CA1 neurons after ischemia. Therefore, either EAAC1 may not play a major role in the process of delayed neuronal death, or a decrease in EAAC1 protein, if preceding the decrease in EAAC1 mRNA as a result of impaired protein synthesis, may contribute to neuronal cell death.
Expression of GLAST mRNA is upregulated in astrocytes within the retina after transient ischemia (Otori et al., 1994) and in the facial nerve nucleus after axotomy (Yamashita et al., 1996). Similarly, we have detected a significant increase in GLAST mRNA in the alveus and stratum lacunosum-moleculare of the CA1 field by the seventh day after ischemia. Microautoradiography has shown that astrocytelike cells express GLAST mRNA in these areas. Because the alveus contains the axons of CA1 pyramidal neurons and nearly half of the CA1 pyramidal neurons degenerate by the seventh day after ischemia, the upregulation of GLAST mRNA expression in the alveus may be involved in the degeneration of these axons. In addition, because the stratum lacunosum-moleculare contains perforant fiber axons and Schaffer collaterals and many synaptic connections between these axons and CA1 neurons are impaired at the seventh day after 3 minutes of ischemia (Matsuda et al., 1997; Peng et al., 1998; Sakanaka et al., 1998), the upregulation of GLAST mRNA in the stratum lacunosum-moleculare seems to take place after degeneration of the projecting axons. We speculate that the upregulation of GLAST mRNA in these regions leads to an increase in glial GLAST protein and to lowering extracellular glutamate concentration, thereby facilitating functional recovery of the remaining neuronal axons.
Expression of GLT1 mRNA in the stratum radiatum markedly decreased by the seventh day after 3 minutes of ischemia. At this period, most of the local synaptic connections, including glutamatergic ones, are impaired in the stratum (Matsuda et al., 1997; Peng et al., 1998; Sakanaka et al., 1998), and there appeared to be little intact glutamatergic synaptic neurotransmission. These results, together with the finding that astrocytes need cocultured neurons to express GLT1 (Swanson et al., 1997), suggest that the expression of GLT1 mRNA depends on intact cross talk between neurons and glia. In contrast, GLAST mRNA expression in the stratum radiatum was not decreased on the seventh day after 3 minutes of ischemia. This result is in agreement with the finding that cultured astrocytes do not need neurons to express GLAST (Swanson et al., 1997). Taken together, the expressions of GLT1 and GLAST mRNA are differentially regulated in the ischemic brain.
This is the first report showing the distribution of GLYT1 and three glutamate transporter mRNA in the gerbil hippocampus, although the distributions of glycine and glutamate transporters in other animal species have been investigated by using immunohistochemistry (Rothstein et al., 1994; Zafra et al., 1995; Lehre et al., 1995; Chaudhry et al., 1995; Schmitt et al., 1996, 1997) and in situ hybridization histochemistry (Adams et al., 1995; Velaz-Faircloth et al., 1996; Schmitt et al., 1996, 1997). Expression of GLYT1 mRNA has been reported to localize exclusively in astrocytes (Zafra et al., 1995; Adams et al., 1995). The present study also showed GLYT1 mRNA expression in astrocytes within the corpus callosum, using a double-labeling method. In line with the studies of Velaz-Faircloth et al. (1996) indicating the neuronal expression of EAAC1 mRNA, we noted an intense EAAC1 mRNA expression in the pyramidal cells and dentate granule cells. Interestingly, GLAST and GLT1 mRNA exhibited unique distribution patterns in the gerbil hippocampus. An intense GLAST mRNA expression was observed mainly in the molecular layer and just beneath the granule cell layer of the dentate gyrus. Expression of GLT1 mRNA is generally considered to occur in astrocytes (Lehre et al., 1995); however, in the present study, we found a strong GLT1 mRNA expression in CA3 pyramidal neurons, supporting the studies of Schmitt et al. (1996) and Martin et al. (1997). In addition, GLT1 mRNA was also intensely expressed in astrocytes in the molecular layer and just beneath the granule cell layer of the dentate gyrus. These findings indicate that GLAST and GLT1 mRNA are abundantly expressed in the dentate gyrus and CA3 field, which are known to be resistant to ischemia. In contrast, GLAST and GLT1 mRNA expressions in the CA1 area were relatively weak. Because GLAST and GLT1 are considered to play a major role in glutamate transport (Rothstein et al., 1996), the reduced expression of GLAST and GLT1 mRNA observed in the CA1 field might account for the vulnerability of CA1 pyramidal neurons to ischemia. However, the expressions of GLAST and GLT1 proteins with or without phosphorylation should be investigated in the future to ascertain this speculation, because the functions of GLAST and GLT1 depend on the local contents of the phosphorylated and nonphosphorylated transporter proteins (Casado et al., 1993; Conradt and Stoffel, 1997).
Footnotes
Acknowledgments
The authors thank Dr. Kazumasa Ikoma for his encouragement during the preparation of this work and Miss Mika Fujimoto for her secretarial assistance.
