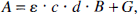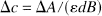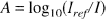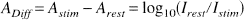Abstract
In this study the authors used a whole-spectrum near-infrared spectroscopy approach to noninvasively assess changes in hemoglobin oxygenation and cytochrome-c oxidase redox state (Cyt-Ox) in the occipital cortex during visual stimulation. The system uses a white light source (halogen lamp). The light reflected from the subject's head is spectrally resolved by a spectrograph and dispersed on a cooled charge-coupled device camera. The authors showed the following using this approach: (1) Changes in cerebral hemoglobin oxygenation (increase in concentration of oxygenated hemoglobin, decrease in concentration of deoxygenated hemoglobin) in the human occipital cortex during visual stimulation can be assessed quantitatively. (2) The spectral changes during functional activation cannot be completely explained by changes in hemoglobin oxygenation solely; Cyt-Ox has to be included in the analysis. Only if Cyt-Ox is considered can the spectral changes in response to increased brain activity be explained. (3) Cytochrome-c oxidase in the occipital cortex of human subjects is transiently oxidized during visual stimulation. This allows us to measure vascular and intracellular energy status simultaneously.
The energy requirements of brain tissue at rest are almost entirely met by oxidative metabolism of glucose. Cytochrome-c oxidase (Cyt-Ox) is the terminal electron acceptor of the mitochondrial electron transport chain and responsible for more than 90% of the cellular oxygen consumption. The energy generated in this process is used to synthesize ATP. The mechanism and control of the enzyme have been discussed in recent reviews (Cooper, 1990; Brown, 1992).
In sections of brain tissue Cyt-Ox activity and concentrations of the enzyme can be measured with different approaches. Such measurements are well established, and Cyt-Ox activity has been shown to be an endogenous metabolic marker of long-term neuronal activity (Wong Riley, 1989).
Because Cyt-Ox has a characteristic light absorption pattern in the visible (Keilin, 1925) and near-infrared parts of the electromagnetic spectrum (Wharton and Tzagoloff, 1964), it is in principle feasible to measure changes in the Cyt-Ox redox state in vivo (Jöbsis, 1977; Ferrari et al., 1990; Cooper et al., 1994). Early data on the Cyt-Ox and NADH redox states in isolated mitochondria showed a transient oxidation during increased cellular activity (Chance and Williams, 1956), thus indicating that the in vivo assessment of the Cyt-Ox redox state offers an approach to evaluate transient changes in the cellular energy metabolism that occur during changes in neuronal activity. Indeed, from experiments on exposed brain tissue of rats, it has been reported that changes in the redox state of Cyt-Ox may occur during local electrical stimulation (Lockwood et al., 1984), i.e., Cyt-Ox may also serve as a marker of transient brain activity.
Near-infrared spectroscopy (NIRS) is an approach to noninvasively measure changes in cerebral hemoglobin oxygenation in response to increased brain activity in human subjects (for a recent review, Villringer and Chance, 1997). Because the spectral resolution of most of the currently used NIRS devices is poor (they measure at only three to five wavelengths), Cyt-Ox measurements have been very controversial (for recent reviews, Cooper et al., 1997; Cooper and Springett, 1997). In this study we used a charge-coupled device (CCD) camera-based whole-spectrum NIRS system that offers superior spectral resolution over the wavelength range from 700 to 1,000 nm. This improved spectral resolution may not only give a better spectral separation of different chromophores (such as hemoglobin and Cyt-Ox), but may also be used to quantify chromophore concentration changes (Matcher et al., 1994), which is not possible with current commercially available NIRS devices. Using this approach, we addressed the following questions:
Does the high spectral resolution CCD approach allow us to measure cerebral oxygenation changes in the human occipital cortex during visual stimulation quantitatively?
Can the spectral changes during functional activation be completely explained by changes in hemoglobin oxygenation, or are there indications for changes in the Cyt-Ox redox state?
If the latter is the case, what are the changes in Cyt-Ox oxidation during brain activity?
SUBJECTS, MATERIALS, AND METHODS
Principles
Near-infrared spectroscopy uses light in the wavelength range between 700 and 1,000 nm. Near-infrared light can penetrate biologic tissue, and even bone, quite well, which makes transcranial measurements through the intact skull and scalp feasible (Jöbsis, 1977; Villringer and Chance, 1997).
A modified Lambert-Beer law is used to determine changes in the local concentrations of oxygenated hemoglobin ([Oxy-Hb]), deoxygenated hemoglobin ([Deoxy-Hb]), and the Cyt-Ox redox state on the basis of variations in light attenuation. The method has been described in detail elsewhere (Cope and Delpy, 1988; Cope et al., 1989; Matcher et al., 1994). Briefly, the attenuation of light by tissue depends on scatter and absorption. In a highly scattering medium like brain tissue, the optical attenuation A can be expressed as follows:
where ε is the specific extinction coefficient of the absorbing compound measured in liters per micromole per centimeter, c is the concentration of the absorbing compound measured in micromoles per liter, and d is the interoptode spacing measured in centimeters. B, the differential path length factor (DPF) (Delpy et al., 1988; Duncan et al., 1996), is defined as the gradient of the attenuation with respect to the absorption coefficient of the tissue. It is wavelength dependent and a nonlinear function of both the tissue absorption and scattering properties. The additive term G accounts for scattering losses. Under the assumption that d, B, and G remain constant during the measurement period, concentration changes (Δc) can be calculated from changes in attenuation (ΔA)
Changes in concentration of a number of chromophores can be computed simultaneously from the changes in attenuation at a number of wavelengths using a least-squares regression algorithm incorporating the relevant extinction coefficients for each wavelength and chromophore (Wray et al., 1988). For known DPF, the concentration changes can be expressed in units of micromoles per liter.
A drawback of previous activation studies using NIRS was that the DPF was not known for the individual volunteers.
To give quantitative results instead of trend information only, we used second differential spectroscopy of the tissue attenuation spectra to estimate each individual's DPF at 830 nm as described elsewhere (Matcher et al., 1994). We assumed that the individual DPF did not change significantly during the experiment.
Instrumentation
In this study a halogen lamp emitting a continuous spectrum from 400 to 1,100 nm is used to illuminate the subject's head. The light is guided to the head through an optical fiber bundle, a so-called optode. The reflected light that has traveled through the tissue is collected by a second optode and guided through the entrance slit of a grating imaging spectrograph (focal length, 0.275 m; SpectraPro 275, [Acton Research, Aston, MA, U.S.A.]). The light is spectrally resolved and collected by a Peltier cooled CCD detector (Princeton Instruments) with a 1024 × 256-pixel resolution. The resulting intensity spectra are read out to a computer for further analysis.
To increase temporal resolution only a part of the CCD image pixels was read out, limiting the spectral range to 700 to 1,000 nm (474 pixel). Although the resulting pixel bandwidth was 0.6 nm, the effective spectral bandwidth was limited to approximately 10 nm by the finite entrance slit of the spectrograph. The spectra were collected with a temporal resolution of 100 ms.
Subjects
Ten healthy, right-handed adults were examined (5 women and 5 men; mean age, 22.5 years). Each subject gave informed consent to participate in the study.
Optode localization
The NIRS probes were placed over the right occipital region at the level of the calcarine sulcus according to a previous study (Wenzel et al., 1996). The interoptode distance was between 2.6 and 3.0 cm (mean, 2.85 cm). Theoretical investigations of the photon path in a head model indicate that the gray matter is part of the sampling volume even at interoptode distances as short as 2 to 2.5 cm (Okada et al., 1997). The optodes were secured in place and covered with dark cloth to prevent external light reaching the detector.
Stimulation protocol
All measurements were performed while the subjects were sitting in a comfortable position in a dark, quiet room. After dark adaptation the subjects were exposed to a green-red checkerboard reversing at 10 Hz during the stimulation period (10 seconds) and nonreversing (i.e., stationary) during the rest period (30 seconds). This stimulation cycle was repeated between 11 and 33 times (average, 26.8).
Data analysis
For the data analysis we followed the work of Matcher and coworkers (1994). The measured intensity spectra (I) were averaged time-locked to 5 seconds (50 spectra) before the onset of the stimulation. From the first 5 seconds and the last 2 seconds of the 40-second stimulation cycle an average spectrum acquired under resting condition (rest intensity spectrum Irest) was calculated.
This was then used to convert the intensity spectra I into tissue attenuation spectra A by correcting for the reference intensity Iref
Here the reference intensity spectrum Iref was acquired by placing the light-delivering and -detecting fibers against each other. In this way the spectra were corrected for the wavelength dependence of the lamp irradiance, the system transmission, and the detector sensitivity. The spectra were smoothed over 18 data points, i.e., the effective spectral resolution was 10.8 nm. To compensate for the wavelength dependence of the optical photon path length in tissue, the spectra were divided by the wavelength-dependent DPF given in the literature (Essenpreis et al., 1993). The concentration changes of the chromophores were subsequently calculated with a least-squares multiple regression procedure.
We performed a preliminary analysis of the difference spectra during brain activation to choose temporal windows of maximal Cyt-Ox changes (fit for Oxy-Hb, Deoxy-Hb, and Cyt-Ox over 740 to 900 nm, data not shown) for each subject. Based on this preliminary analysis, we determined the temporal window (width, 2 seconds) showing the largest presumable Cyt-Ox amplitude for each subject. From this temporal window an average intensity spectrum was calculated (stimulation spectrum Istim). Irest and Istim were then converted into attenuation spectra Arest and Astim according to equation 3.
Analysis of activated-rest difference spectra. The average spectra acquired under resting and stimulated conditions were used to compute a difference attenuation spectrum.
To test whether the spectra obtained in the state of increased brain activity can be explained by changes in [Oxy-Hb] and [Deoxy-Hb] alone, or whether additionally [Cyt-Ox] changes must be considered we further analyzed this difference spectrum.
For this analysis we assumed that there is no change in the water signal with visual stimulation, i.e., water was not considered in the further analysis of the difference spectra.
Fitting procedures. We used six different procedures (Table 1) to fit changes in the chromophore concentrations to the attenuation spectra. The contribution of Cyt-Ox to the near-infrared spectrum is most significant above 780 nm, and in neonatal pig studies the change in the Cyt-Ox signal is critically dependent on the wavelength range over which the fit is performed (Cooper et al., 1999). We applied the least-squares fitting procedure to different spectral ranges.
One fitting procedure including only the spectra of Oxy-Hb and Deoxy-Hb was applied over the range 740 to 900 nm, thus including the prominent Deoxy-Hb feature at 760 nm (Fig. 1). Only Oxy-Hb and Deoxy-Hb were included in the fit over the range 780 to 900 nm covering the same region as most of the published discrete wavelengths algorithms (for a performance comparison see Matcher et al., 1995). A fit using the discrete wavelengths 778, 825, 850, and 904 nm (Fig. 1) as published by Cope and Delpy (1988) was applied. Only Oxy-Hb and Deoxy-Hb were included as parameters. In addition to Oxy-Hb and Deoxy-Hb the difference spectrum of oxidized minus reduced Cyt-Ox (Cyt-Oxox/red) was incorporated. The fit was performed over 740 to 900 nm, compare with 1. Same as 4, applied over 780 to 900 nm, compare with 2. Same as 3, including Cyt-Ox as additional component.
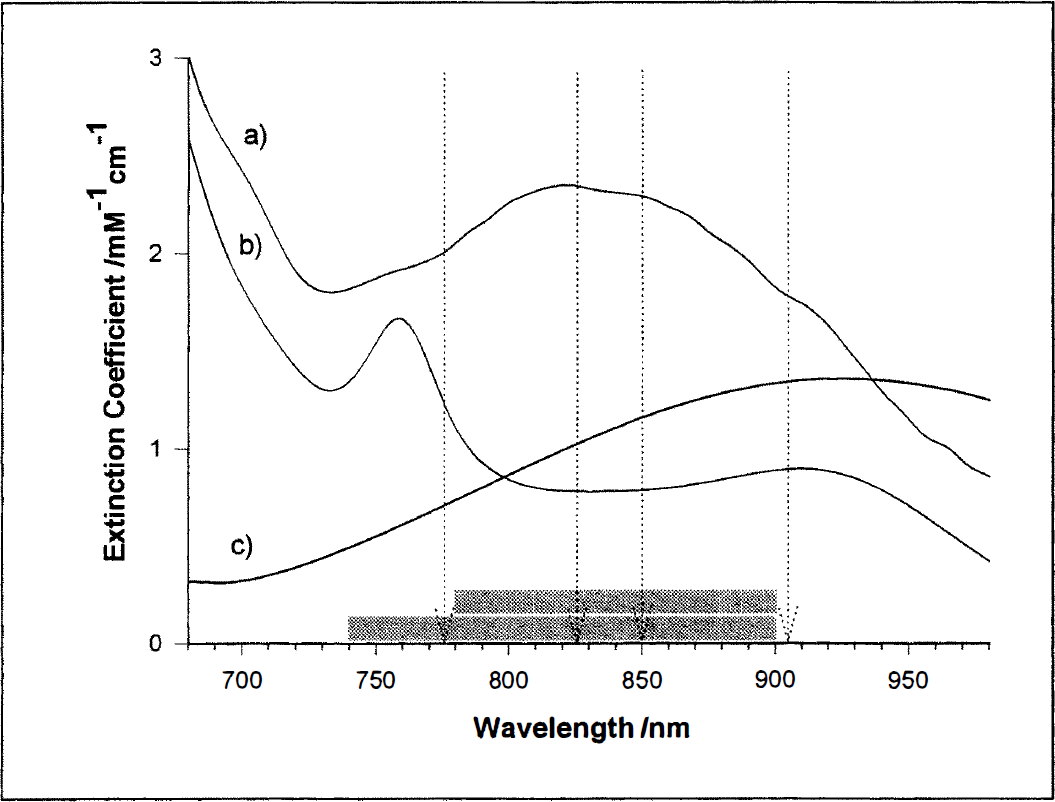
In vitro extinction coefficient (ε) spectra for Oxy-Hb
The different fitting procedures for the analysis of the spectra
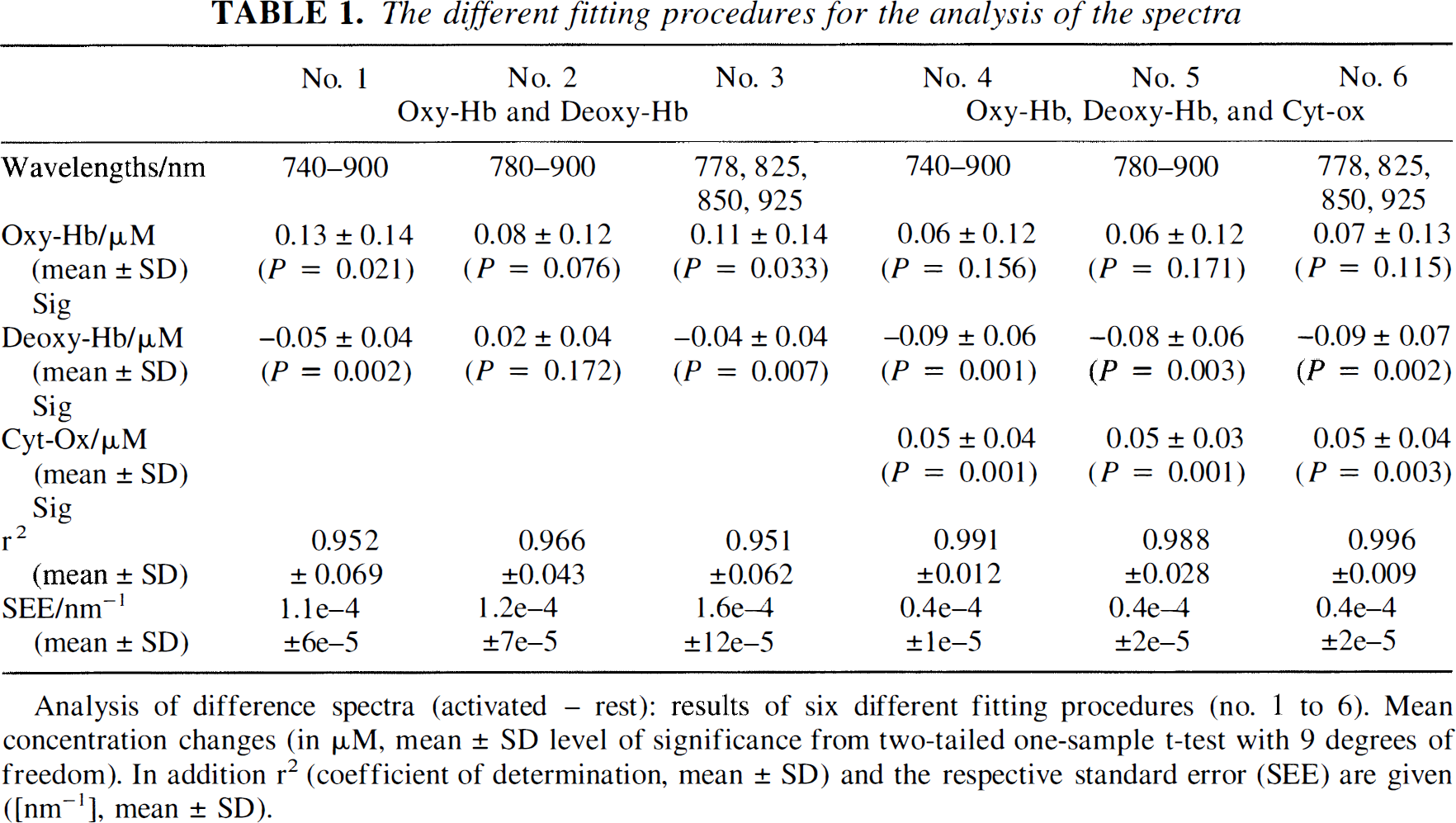
Analysis of difference spectra (activated – rest): results of six different fitting procedures (no. 1 to 6). Mean concentration changes (in µM. mean ± SD level of significance from two-tailed one-sample t-test with 9 degrees of freedom). In addition r2 (coefficient of determination, mean ± SD) and the respective standard error (SEE) are given ([nm−1], mean ± SD).
To further characterize the results of the described fitting procedures we calculated the coefficient of determination (r2, goodness of fit), as well as the standard error of the estimate (or standard deviation of the residuals). To allow for comparison between the different fitting procedures, the standard error of the estimate is given per nanometer.
As shown in the results section fit procedures 4 and 5 gave the best results with no significant difference between the two. We used fit procedure 4 for further analysis.
Statistical analysis. To test whether the CCD approach is sensitive enough to measure hemoglobin oxygenation and Cyt-Ox redox changes in the human occipital cortex during visual stimulation, we determined the time courses of these concentration changes. We used fit procedure 4, i.e., the attenuation changes over the range 740 to 900 nm were fitted to the known absorption spectra of oxygenated and deoxygenated hemoglobin (Matcher et al., 1995) (Fig. I), as well as the difference spectrum of oxidized minus reduced Cyt-Ox (Wray et al., 1988; Matcher et al., 1995).
To assess whether there was a statistically significant change in any of the three parameters, we determined the 2-second maximum amplitude for each parameter and calculated the mean concentration changes during this temporal window for each subject and parameter. From this value the mean concentration change under resting conditions (mean for the periods “1 to 5 seconds” and “38 to 40 seconds” of the stimulation cycle) was subtracted. With the resulting values we performed a one-sample t test against zero (two-tailed, n–1 degrees of freedom, with n = number of trials).
RESULTS
Individual DPF and [Deoxy-Hb]
The average DPF830 was 5.05 (± 0.77, standard deviation). The average Deoxy-Hb concentration was 15.32 (± 3.68) µmol/L.
Analysis of activated – rest difference spectra
Table 1 gives the results of the different fitting procedures to the stimulation minus rest difference spectra (ADiff). With all fit procedures there is an increase in [Oxy-Hb], but this is significant only with fit procedures 1 (0.13 ± 0.14 µmol/L; P = 0.021; one-sample t test; 9 degrees of freedom) and 3 (0.11 ± 0.14 µmol/L; P = 0.033). Using the two-component fits (procedures 1 through 3) the increase in [Oxy-Hb] is larger than with the three-component fits (4 through 6).
Using the two-component fits (excluding Cyt-Ox) there is a significant decrease in [Deoxy-Hb] with procedures 1 (−0.05 ± 0.04 µmol/L; P = 0.002) and 3 (−0.04 ± 0.04 µmol/L; P = 0.007). Using procedure 2 (780 to 900 nm, thus neglecting the Deoxy-Hb feature at 760 nm) there is a slight increase in [Deoxy-Hb] (0.02 ± 0.04 µmol/L; P = 0.172). With all procedures including three components (4 through 6), a statistically significant decrease in [Deoxy-Hb] is calculated (procedure 4, −0.09 ± 0.06 µmol/L; P = 0.001; procedure 5, −0.08 ± 0.06 µmol/L; P = 0.003; procedure 6, −0.09 ± 0.07 µmol/L; P = 0.002).
In all the fits that include the Cyt-Ox spectrum, a statistically significant increase in Cyt-Ox oxidation is obtained (procedure 4, 0.05 ± 0.04 µmol/L; P = 0.001; procedure 5, 0.05 ± 0.03 µmol/L; P = 0.001; procedure 6, 0.05 ± 0.04 µmol/L; P = 0.003). The coefficient of determination, r2, is larger for the three-component procedures, i.e., the part of the in vivo data that is explained by the estimate is larger when Cyt-Ox is included in the fit. The standard error of the estimate is given in units per nanometer to allow a comparison. It is smaller when Cyt-Ox is included in the fit. Fitting procedures 4 and 5 seem to explain the in vivo data quite well (r2 = 0.991 and 0.988, respectively) and produce the smallest error. Procedure 5 neglects the important Deoxy-Hb feature at 760 nm and might underestimate changes in Deoxy-Hb. We used procedure 4 for further analysis.
Figure 2 shows the average of the difference spectra of all subjects. It displays two models. First, the result of fitting procedure 4, i.e., the three-component fit (including Cyt-Ox), applied to 740 to 900 nm (Oxy-Hb, 0.05 µmol/L; Deoxy-Hb, −0.09 µmol/L; Cyt-Oxox/red, 0.05 µmol/L; r2 = 0.997) is shown. Furthermore the result of procedure 1, i.e., the two-component fit excluding Cyt-Ox, 740 to 900 nm (Oxy-Hb, 0.12 µmol/L; Deoxy-Hb, −0.05 µmol/L; r2 = 0.965) is presented. The shape of the model including Oxy-Hb, Deoxy-Hb, and Cyt-Ox is very similar to the averaged experimental Adiff spectrum, whereas the model including only Oxy-Hb and Deoxy-Hb is clearly different, especially around 820 nm.
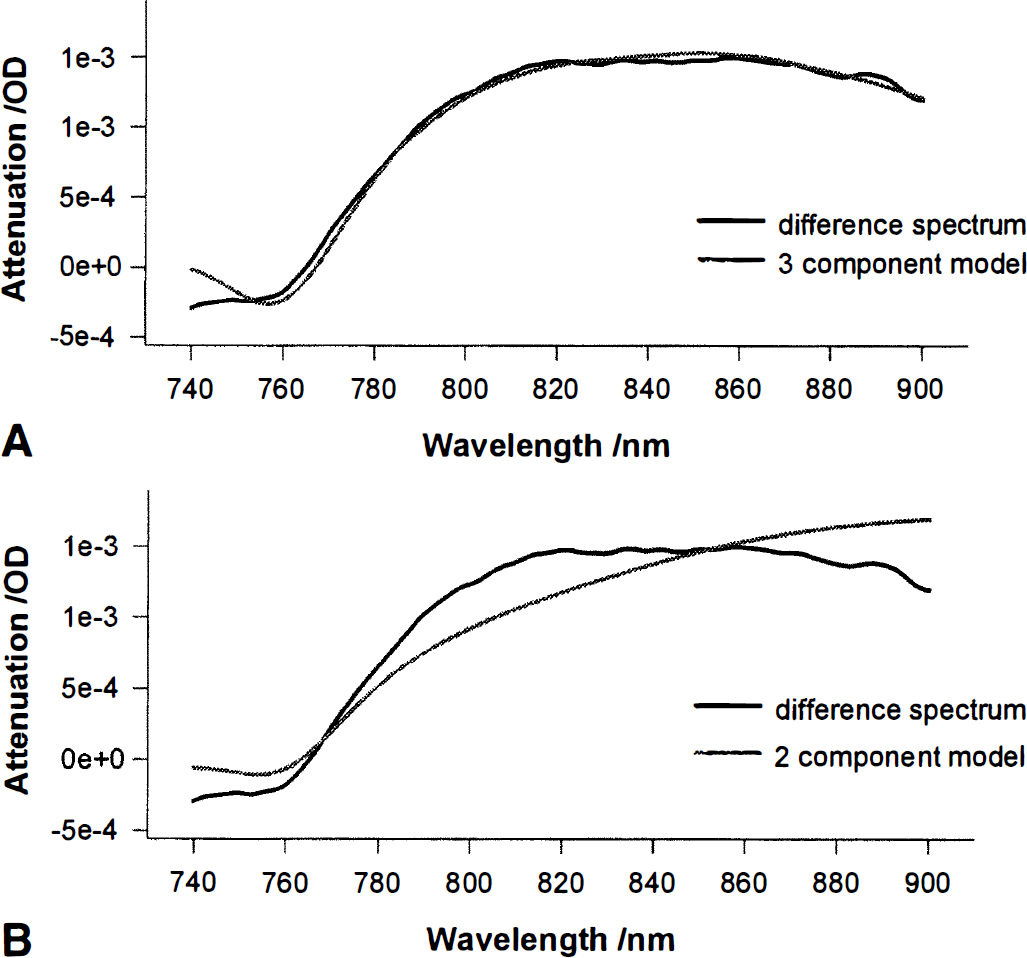
Averaged (n = 10) experimental (thick line in
Figure 3 gives the residuals of the models shown in Fig. 2 for procedures 4 and 1 (Fig. 3A), as well as the extinction coefficient spectrum of Cyt-Ox (Fig. 3B). For an ideal fitting procedure, the residuals are randomly distributed. Above 760 nm the residuals of procedure 4 seem to be distributed randomly. In contrast, when only Oxy-Hb and Deoxy-Hb are included in the fitting procedure, the residuals have essentially the same spectral dependence as the Cyt-Oxox/red absorption spectrum, which is given in Fig. 3B.
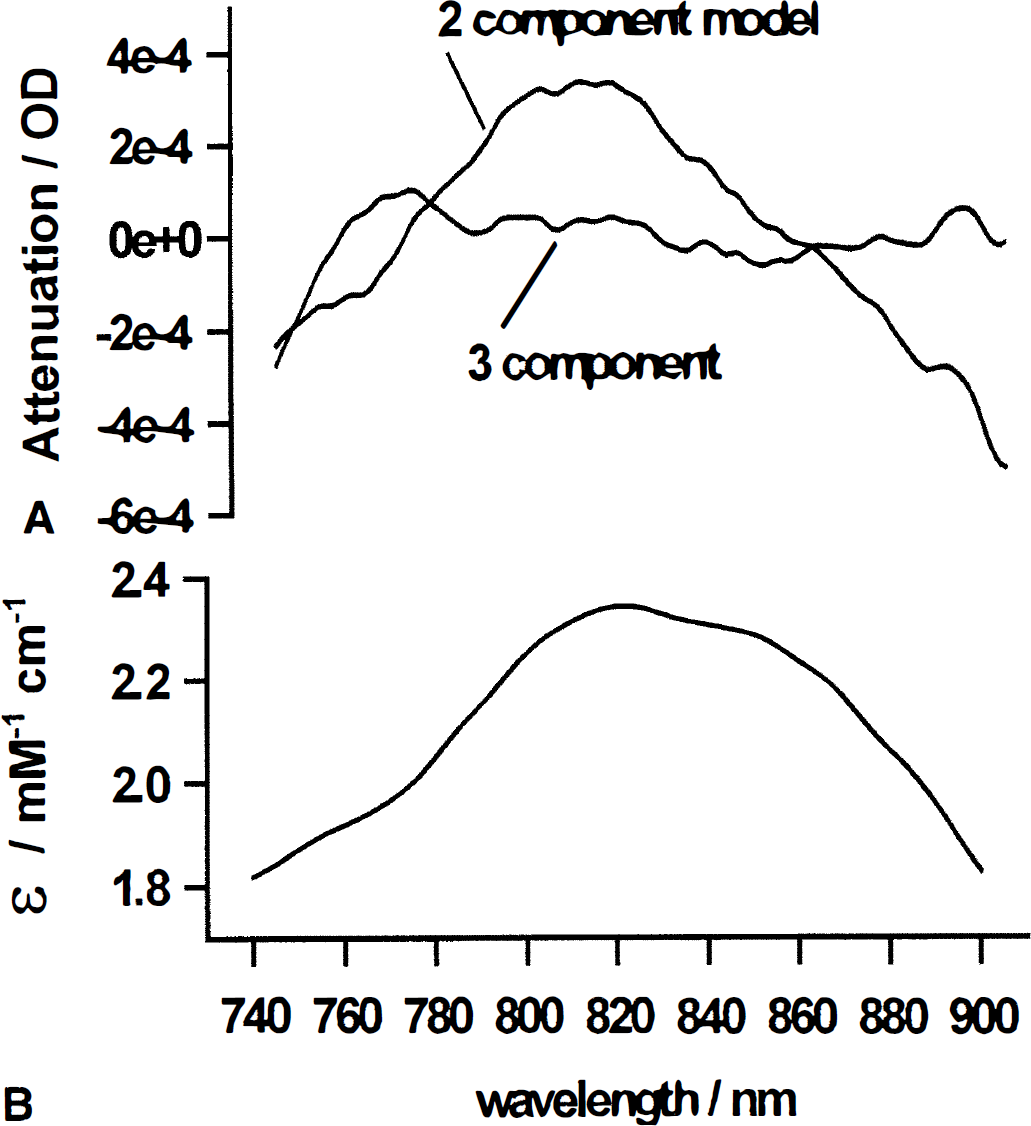
Statistical analysis
Figure 4 gives the grand average (n = 10) of the time courses of [Oxy-Hb], [Deoxy-Hb], and [Cyt-Ox] determined using fit procedure 4 during visual stimulation. As has been shown previously, there is a rise in [Oxy-Hb] and a drop in [Deoxy-Hb] with brain activation. In addition there is an increase in Cyt-Ox oxidation.
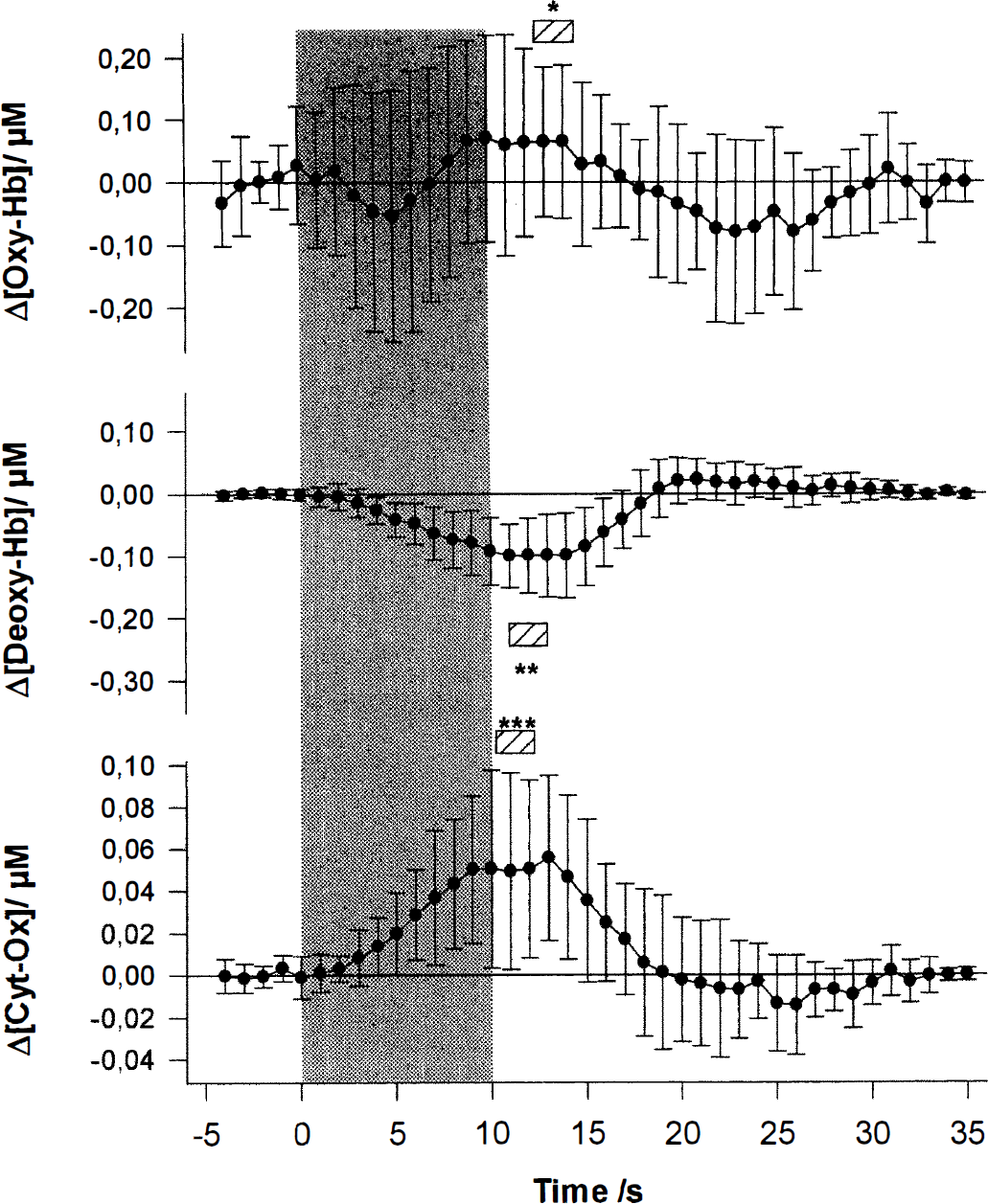
The grand average of the concentration changes for all 10 subjects as a function of time. The gray-shaded area depicts the 10-second visual stimulation period. The changes in [Oxy-Hb], [Deoxy-Hb], and redox state of Cyt-Ox are given in micromoles per liter. The data are averaged for 1 second. The error bars give the standard deviations of the mean. Note that the time course of [Cyt-Ox] differs from that of [Oxy-Hb] and [Deoxy-Hb]. The hatched bars indicate the average 2-second peak. The stars indicate the level of significance of the mean concentration changes during this period (one-sample t test, two-tailed, 9 degrees of freedom; *P = 0.002; **P < 0.001; ***P = 0.001). The large standard deviation indicates pronounced intersubject variability, which is caused by slight intersubject variations in optode position, sample volume, path length, and so forth. Note that the standard deviation is larger for [Oxy-Hb] than for [Deoxy-Hb]. The reason for the higher standard deviation in [Oxy-Hb] versus [Deoxy-Hb] is the higher intrinsic noise of the [Oxy-Hb] signal. One reason for this physiologic noise is pulse-induced concentration changes. We have analyzed the changes in [Oxy-Hb] and [Deoxy-Hb] during a single pulse and noted that the [Oxy-Hb] changes are approximately 10 times larger (data not shown). Because in this study we are undersampling with respect to the pulse, this leads to higher noise in the [Oxy-Hb] trace.
[Oxy-Hb], after an initial drop 5 seconds after the onset of the stimulation (not statistically significant and not in all subjects) rises to a plateau maximum approximately 5 seconds later, 10 seconds after the onset of stimulus presentation. After the end of the stimulation there is a poststimulus undershoot that peaks after 15 seconds. Finally [Oxy-Hb] increases toward prestimulation values.
The concentration of deoxygenated hemoglobin starts to decrease approximately 3 seconds after the onset of stimulation, reaching a plateau minimum after 11 seconds, and remains decreased for approximately 3 seconds. After the end of the stimulation, [Deoxy-Hb] increases and exceeds baseline values (poststimulus overshoot) reaching a maximum approximately 10 seconds later. The concentration of deoxygenated hemoglobin decreases to baseline values approximately 20 seconds after the end of the stimulation.
The [Cyt-Ox] time course is slightly different. The Cyt-Ox oxidation starts to increase 3 seconds after the beginning of the activation period and reaches a plateau after approximately 9 seconds. It remains elevated for approximately 4 seconds. Thereafter it decreases again, slightly exceeds baseline values approximately 15 seconds after the end of the stimulation, and finally reaches pre stimulation baseline values approximately 20 seconds after the end of the stimulation.
Table 2 gives the number of trials (n) for each subject, the time to peak (2-second interval, tmax), the mean concentration changes during this temporal window (± standard deviation), and the level of significance for these changes (one-sample t test against zero, two-tailed, n −1 degrees of freedom) for each subject and parameter. In 9 of the 10 subjects there is an increase in Oxy-Hb that is statistically significant in 5 subjects. The 2-second peak in Oxy-Hb is reached 12.7 ± 3.5 seconds after the onset of stimulation (compare Fig. 4). All 10 subjects show a significant decrease in Deoxy-Hb. The 2-second minimum in Deoxy-Hb is reached 11.0 ± 1.6 seconds after the beginning of the stimulation. In all of the subjects Cyt-Ox oxidation increases. This is statistically significant in 9 of 10. The 2-second peak in Cyt-Ox is reached 10.2 ± 1.6 seconds after the onset of stimulation.
Changes in [Oxy-Hb], [Deoxy-Hb], and [Cyt-Ox] in response to visual stimulation
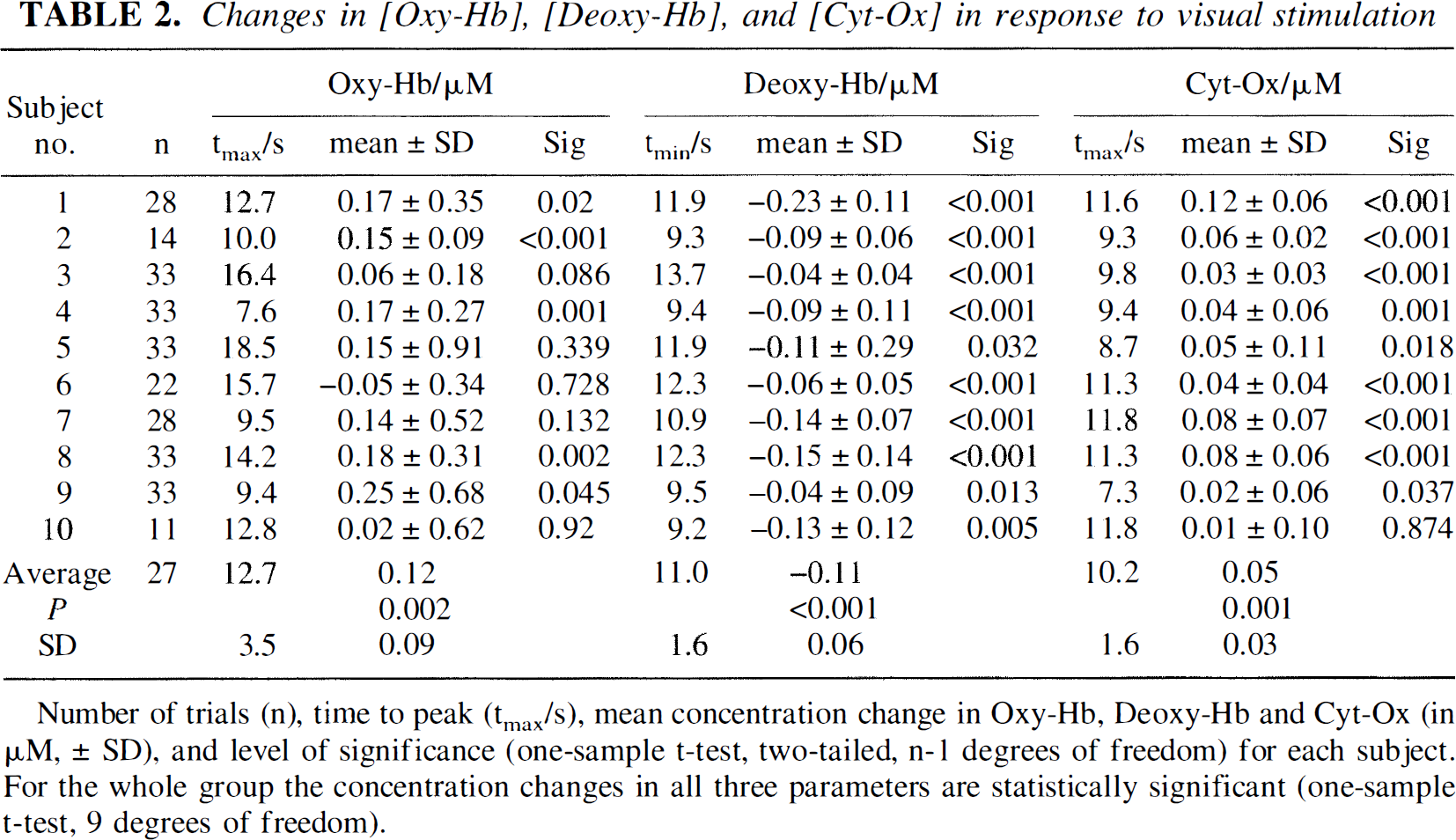
Number of trials (n), time to peak (tmax/s), mean concentration change in Oxy-Hb, Deoxy-Hb and Cyt-Ox (in µM, ± SD), and level of significance (one-sample t-test, two-tailed, n-1 degrees of freedom) for each subject. For the whole group the concentration changes in all three parameters are statistically significant (one-sample t-test, 9 degrees of freedom).
The average concentration changes (group average) during the respective peak response were 0.12 ± 0.03 µmol/L for Oxy-Hb, −0.10 ± 0.01 µmol/L for Deoxy-Hb, and 0.05 ± 0.01 µmol/L for the Cyt-Ox redox state. These changes were highly significant for all three parameters (Oxy-Hb, P = 0.002; Deoxy-Hb, P < 0.001; Cyt-Ox, P = 0.001; one-sample t test, two-tailed, 9 degrees of freedom).
DISCUSSION
Using NIRS, changes in cerebral hemoglobin oxygenation in response to increased brain activity can be assessed noninvasively in human subjects. This has been shown for motor activity, visual activation, auditory stimulation, and performance of cognitive tasks (for a recent review see Villringer and Chance, 1997).
In response to increased brain activity an increase in regional cerebral blood flow occurs that is larger than the increase in oxygen consumption (Fox et al., 1988). This leads to a transient focal hyperoxygenation. Therefore, a localized increase in [Oxy-Hb] and a decrease in [Deoxy-Hb] is seen when the NIRS probes are positioned over an activated brain area (Villringer and Chance, 1997).
The disadvantage of current NIRS systems for the assessment of brain activity is that they only approximate quantitation of the concentration changes. Furthermore, with measurements at only three to six individual wavelengths, the separation of hemoglobin signal from the Cyt-Ox signal has been regarded as questionable (Matcher et al., 1995; Cooper et al., 1994, 1998).
In this study we used an NIRS approach based on white light illumination and detection with a CCD camera (Cope et al., 1989), which allows for measurements over the entire wavelength range between 700 and 1,000 nm. Using this approach we demonstrate that (1) it is possible to assess changes in hemoglobin oxygenation of the human brain quantitatively and noninvasively during brain activation at high temporal resolution, confirming and improving results obtained with other NIRS instruments; (2) difference spectra cannot be explained by well-known changes of Oxy-Hb and Deoxy-Hb only; Cyt-Ox must be included; and (3) changes in Cyt-Ox redox state can be measured during functional activation. Our measurements indicate a transient increase in Cyt-Ox oxidation during increased brain activity.
Individual DPF and absolute [Deoxy-Hb] in the occipital cortex of human adult subjects
As suggested by Matcher et al. (1994), we used second differential spectroscopy to measure the mean optical path length over the occipital cortex in each adult human subject. The average DPFs30 was 5.05 ± 0.77 (mean ± SD). Using phase-resolved spectroscopy, Duncan et al. (1996) have reported values for the adult forehead and subsequently shown that the DPF increases as a function of age. Using their equation for the age-dependence from their study, a DPF830 of 5.46 would result for our subjects (the difference from our measurements was not statistically significant, data not shown). However, one has to be cautious when comparing values measured over different regions of the head as the proportion of the different absorbers and scatterers (bone, myelin, muscle, blood) in the illuminated tissue is likely to vary regionally. Using time-of-flight measurements a 15% variation of the DPF measured frontally and parietally has been found (Ferrari et al., 1993). The DPF830 reported here shows a large intersubject variability (SD approximately 15%). This is comparable to the variation of DPF found in studies using the same approach in infants (Cooper et al., 1996) and other approaches (phase-resolved spectroscopy, time-of-flight measurements, Duncan et al., 1996); for a detailed discussion of these methodological issues, see Kohl et al. (1998a).
It is important to point out that in this study we determined the individual DPF under resting conditions and assumed that the DPF does not change during the experiment. Using this individually determined DPF, we demonstrated a mean absolute increase of [Oxy-Hb] of 0.05 µmol/L and a mean absolute decrease in [Deoxy-Hb] of 0.09 µmol/L during visual stimulation. Although it is clearly an improvement that these measurements can now give absolute values, it must be emphasized that this concentration change refers to the entire sample volume including extracerebral tissue. To relate these changes to the baseline concentration in brain tissue, near-infrared imaging approaches, which are currently being developed, will be necessary.
In addition to the above mentioned quantitation of concentration changes, owing to its spectral feature in the second differential, the total absolute [Deoxy-Hb] can also be determined with this approach (Matcher and Cooper, 1994). We obtained a value of [Deoxy-Hb] of 15.3 ± 3.7 µmol/L. Again it must be emphasized that this value refers to the entire sample volume including extracerebral tissue. In a recent study Brun et al. (1997) have stated that NIRS detects hemoglobin in tissue in a venous to arterial ratio of 2: 1. Assuming an arterial blood hemoglobin oxygenation of 97% and a venous saturation of 70%, the mean absolute hemoglobin concentration can then be approximated to be 73 ± 18 µmol/L.
Changes in Cyt-Ox redox state with functional brain activation
The main finding of our study is an increase in Cyt-Ox oxidation during functional activation in human subjects. We subsequently review previous measurements and concepts about the Cyt-Ox redox state in animals and human subjects.
The systematic study of changes in the redox state of Cyt-Ox was initiated by Keilin's study of the thoracic muscles of the wax moth during wing motion (Keilin, 1925; for a detailed historical review see Chance et al., 1997).
Experiments using visible light
Changes in the cerebral Cyt-Ox redox state were first investigated by Rosenthal and coworkers. They reported Cyt-Ox in animal brain to be reduced approximately 30% in the control resting state. This was reported for rats (Sylvia and Rosenthal, 1979), cats, and rabbits (Rosenthal et al., 1976), in which Cyt-Ox oxidation was found to vary as a continuous function of inspired O2, Using this dual-wavelength approach (605 and 590 nm) the same group has reported small local increases in local blood volume and transient increases in Cyt-Ox in animals upon direct cortical stimulation (Rosenthal and Jobsis, 1971; LaManna et al., 1977; Lockwood et al., 1984).
This approach was further developed to rapid scanning photometry. This technique observes different wavelengths sequentially. Using this technique LaManna et al. (1987) underpinned previous findings of their group and found a fraction of Cyt-Ox in intact rat brain that was oxidized during direct cortical stimulation. However, these in vivo studies using visible light bear some problems.
The small penetration depth of the visible light restricts the studies to animal studies because skin, muscles, and skull have to be removed. The algorithm used for the dual-wavelength approaches to compensate for contamination of the Cyt-Ox signal by the much stronger hemoglobin signal might be unsatisfactory (Kariman and Burkhart, 1985). The scanning systems that observe different wavelengths sequentially produce spectral distortion (Cope et al., 1989).
Furthermore, these studies using visible light measured heme a and a3 and not CuA as in our study.
The near-infrared absorption features of Cyt-Ox
A near-infrared (NIR) absorption band centered around 830 nm in the oxidized minus reduced NIR difference spectrum of Cyt-Ox had been described earlier (Wharton and Tzagoloff, 1964). Cytochrome-c oxidase contains four redox-active metal centers that contribute to the signal. The two redox-active iron atoms are present in prosthetic heme a groups, which contain hydroxyethylfamesyl and propionate side chains and clearly perturb the visible spectrum compared with the more common heme b moiety usually found in heme enzymes.
It has been suggested that the NIR spectrum of Cyt-Ox is dominated by the CuA center (Wharton and Tzagoloff, 1964; Hartzell et al., 1973; Boelens et al., 1982). This has recently been confirmed by two findings: (1) Cyt-Ox from bacterial enzymes that contain CuA but do not contain heme a have NIR spectra that are very similar to those of the native enzyme (Ingledew et al., 1992; Kelly et al., 1993), and (2) bacterial enzymes that lack CuA but contain heme a have no strong NIR spectra (Henning et al., 1995).
However, the in vivo NIR spectrum is not only influenced by Cyt-Ox but also by cytochrome-c. Below 750 nm there might be significant interference (60% at 695 nm); above 780 nm this is unlikely (only approximately 10% of the absorption might be attributable to cytochrome-c, Cooper et al., 1994).
In vivo difference spectra of Cyt-Ox differences across species?
In blood—fluorocarbon exchange-transfused (i.e., blood-free) rats, Cyt-Ox is detectable in vivo (Wray et al., 1988). The detected difference spectrum of oxidized minus reduced Cyt-Ox is very similar to the spectrum of the isolated enzyme. Both have a characteristic feature centered at 820 to 840 nm.
A human Cyt-Ox spectrum in the NIR has not yet been reported, but a comparison of difference spectra (Cooper et al., 1997) of pig, rat, and cat brain (Ferrari et al., 1990; Miyake et al., 1991) shows similarity. Thus, it is unlikely that age and species differences affect the shape of the Cyt-Ox difference spectrum.
Detection of cytochrome-c oxidase redox changes in vivo?
It has been questioned whether this relatively weak Cyt-Ox signal can be detected in vivo in the presence of the much larger (10-fold) [Oxy-Hb] and [Deoxy-Hb]. Cross talk might occur, i.e., a change in hemoglobin concentration might yield an artifactual change in the Cyt-Ox signal unless the algorithms for deconvoluting the spectral contributions were perfect (Cooper et al., 1994; Matcher et al., 1995).
Matcher and colleagues (1995) challenged those algorithms for determining Cyt-Ox in the presence of hemoglobin concentration changes. They found that the algorithm using 112 wavelengths (770 to 900 nm; the algorithm was named UCLn) was most robust to extreme changes in Oxy-Hb, Deoxy-Hb, and total hemoglobin (sum of Oxy-Hb and Deoxy-Hb). The algorithm applies a spectral correction factor to account for the wavelength dependence of the DPF by scaling the extinction coefficients accordingly (Essenpreis et al., 1993). Proof that measurement of the Cyt-Ox signal in the brain using this multiwavelength algorithm is robust to large changes in hemoglobin concentration and oxygenation has recently been shown in animal models, both neonatal (Cooper et al., 1999) and adult (Cooper et al., 1998).
In this study it is important to point out that we assumed that the individual DPF remained constant during the experiment. In addition to changes in chromophore concentrations discussed here, there might be scattering changes associated with brain activation (Kohl et al., 1998b). These changes would directly influence the DPF values and interfere with Cyt-Ox measurements.
Cyt-Ox redox state during visual stimulation
Using algorithms that are similar to the UCLn algorithm (multiwavelength, 740 to 900 nm or 780 to 900 nm; Table 1) in this study we have shown that Cyt-Ox oxidation in the occipital cortex increases significantly in response to visual stimulation in 9 of 10 subjects (compare Table 2).
This effect is present irrespective of the involved fitting procedure (compare Table 1). In the temporal window of the maximum increase in Cyt-Ox oxidation the amplitude of the hemoglobin-related parameters are on the same order of magnitude as the changes in Cyt-Ox redox state. In a study analyzing potential cross talk only very large changes in Oxy-Hb or Deoxy-Hb (10 to 20 µmol/L compared with −0.09 to −0.13 µmol/L in our study) produced spurious changes in the Cyt-Ox redox state (Matcher et al., 1995). It is thus unlikely that the changes in Cyt-Ox oxidation are caused by cross talk.
The analysis of the difference spectra between the activated state and the resting state reveals that the measured spectra cannot be explained sufficiently by changes in [Oxy-Hb] and [Deoxy-Hb] alone as a significant part of the residuals of a two-component fit are nonrandom. However, this unexplained part of the spectrum becomes much smaller when Cyt-Ox is included into the fit (740 to 900 nm, 4.8% versus 0.9% if Cyt-Ox is included; 780 to 900 nm, 3.4% versus 1.2% when Cyt-Ox is included; compare Table 1 and Fig. 2). This point becomes even more evident when the part of the data that is not explained by the two-component (Oxy-Hb, Deoxy-Hb) model is analyzed: the residuals of the fitting procedures not including Cyt-Ox have similar features as the Cyt-Ox difference spectrum (Fig. 3). When Cyt-Ox is included as an additional component, the residuals are not only significantly smaller (Fig. 3 and Table 1) with only approximately 1% of the signal remaining unexplained (indicated by 1 – r2), but the residuals seem to be distributed randomly, i.e., the difference spectrum can sufficiently be described as a combination of Oxy-Hb, Deoxy-Hb, and Cyt-Ox.
The analysis of the mean concentration changes (Table 2) shows that a statistically significant increase in Cyt-Ox oxidation in response to visual stimulation is present in 9 of the 10 subjects. Thus we have shown that this CCD approach allows us to measure hemoglobin oxygenation changes and changes in the Cyt-Ox redox state in the human occipital cortex during visual stimulation quantitatively. It is evident from the data given in Fig. 4 and Table 2 that the time course of Cyt-Ox-oxidation is significantly different from [Oxy-Hb] and [Deoxy-Hb].
How does this relate to current concepts of brain energy metabolism during functional activation?
Increased neuronal activity leads to an accelerated activity of active ion pumps (Na+/K+-ATPase) to sustain and restore the membrane potentials, i.e., ATP is hydrolyzed to ADP + Pi and subsequently ATP has to be synthesized. This might be accomplished by oxidative phosphorylation or anaerobic glycolysis. There is an ongoing debate about how the brain meets this demand. In agreement with the traditional view that oxidative phosphorylation accounts for more than 95% of the ATP production of the brain (Siesjö, 1978), animal studies indicate that oxidative phosphorylation supplies the majority of energy during transiently increased brain activation (Hyder et al., 1996). In contrast to this position other authors hypothesized that glycolysis satisfies a major part of the additional energy demand (Fox et al., 1988).
The processes of glycolysis, β-oxidation, and the tricarboxylic acid cycle provide reducing equivalents to the respiratory chain in the form of NADH and FADH, which then fuel oxidative phosphorylation. The terminal enzyme of the respiratory chain is Cyt-Ox, and it catalyzes the transfer of electrons from its reduced substrate cytochrome-c to CuA and cytochromes-a and -a3 and then to molecular oxygen to form water (for a detailed review on regulation of oxidative phosphorylation, see Brand and Murphy, 1987; Brown, 1992; Balaban, 1990).
What is the physiologic relevance of the oxidation we observe in the Cyt-Ox CuA center? Our recent studies in the adult rat brain have suggested that CuA can be reduced as much as 15% in the normal brain (Cooper et al., 1998). Small oxidations from this basal redox level are therefore entirely possible. We have described previously the factors that affect the redox state of this center in vitro and in vivo (Cooper et al., 1994). The biggest changes are observed for large changes in the oxygen tension (Cooper et al., 1998); however, the relatively small changes seen in this study could also be caused by a number of other factors that affect the flow of electrons through this center.
Possible factors that might oxidize CuA in vitro include an increase in the oxygen tension, a decrease in the proton electrochemical potential across the inner mitochondrial membrane, a decrease in pH, or a decrease in the supply of reducing equivalents to the respiratory chain. The latter two appear unlikely to be true in vivo for functional activation as other studies show an increase in pH (Rango et al., 1997) and an increase (Hyder et al., 1996) in tricarboxylic acid flux (and hence the supply of electrons from NADH to the mitochondrial respiratory chain).
Although increases in the oxygen tension undoubtedly occur after functional activation, we do not believe that this is the most likely explanation of our data as this would imply that under physiologic conditions, the CuA in the adult brain is partly reduced owing to a low tissue Po2. This is inconsistent with animal studies from our own (Cooper et al., 1998) and other laboratories (Ferrari et al., 1995; Tamura, 1993). These studies demonstrate that lowering the supply of oxygen to the brain does not reduce CuA, suggesting that the normal brain Po2 is significantly greater than the region at which the CuA center shows oxygen-dependent changes in its steady-state redox level.
Thus we believe that the most likely current explanation for our data is that decreases in the proton electrochemical potential increase the rate of electron exit from the CuA, causing an increased steady-state oxidation. In the case of Cyt-Ox respiring on artificial electron donors in mitochondria, oxidation of CuA clearly occurs when the proton electrochemical potential is completely collapsed by uncouplers (Morgan and Wikström, 1991). However, there are few data relating to the changes in the redox state of CuA in mitochondria respiring on physiologic electron donors (e.g., NADH-linked substrates) when the potential is decreased by increasing the ADP concentration. This is an important point, as ADP stimulates oxidative phosphorylation by causing only a relatively small decrease in the mitochondrial proton electrochemical potential, whereas most studies using mitochondrial uncouplers result in complete loss of the potential.
In contrast to CuA, it is easier to look at the effects of varying the proton electrochemical potential on the mitochondrial cytochrome-c redox state. It is expected that these changes will be mirrored by those in CuA (Cooper et al., 1994), given the physical proximity (Tsukihara et al., 1995) and similar redox potentials (Rich et al., 1988) of cytochrome-c and CuA. Although there is some disagreement in the literature (Murphy and Brand, 1987), there are studies that clearly indicate that increasing ADP levels oxidize cytochrome-c, whether succinate or NADH is the source of electrons (Chance and Williams, 1955). We think that this is the most likely physiologic explanation for the oxidation we observed in this study, i.e., the increased demand for ATP to regenerate ion gradients in the brain leads to increased ATP hydrolysis. This in turn increases the ADP concentration, increasing the rate of oxidative phosphorylation by a decrease in the potential across the mitochondrial membrane. As a byproduct of this we see an oxidation in CuA through the mechanisms described above. Although by no means proven, such an explanation would mean that NIRS would provide an intriguing insight into the cellular, as well as the hemodynamic, events after functional activation.
Finally it has been suggested that the nitric oxide that is at least partly responsible for the increased blood flow after functional activation may inhibit Cyt-Ox activity (Brown, 1995). The present studies do not support this hypothesis as increases in nitric oxide would be expected to reduce Cyt-Ox CuA, not oxidize it, by binding to the ferrous heme a3 site, thus preventing electron exit from CuA.
It should be added that our data indicate an increase in oxygen consumption during functional activation in agreement with other recent studies (Hyder et al., 1997). Because the simultaneous blood flow increase exceeds the increase in oxygen consumption, and because this blood flow increase seems to be accomplished by a change in blood flow velocity (rather than an increase in blood volume) (Bereczki et al., 1993; Villringer et al., 1994) [Deoxy-Hb] drops (for a more detailed discussion see Villringer and Dirnagl, 1995). The rather small effects that we observed in [total-Hb] are consistent with this explanation. In summary, during functional activation, we see an increase in [Oxy-Hb], a decrease in [Deoxy-Hb], a rather small change in the concentration of total hemoglobin, and—the new finding in this study—an increase in Cyt-Ox oxidation.
We are now studying whether the increase in Cyt-Ox oxidation is present with all types of brain activation, such as motor and somatosensory, whether it is present in all cortical areas, and whether it is dependent on the local cortical Cyt-Ox concentration. From the knowledge of the normal interplay between intracellular Cyt-Oxox/red and the vascular oxygenation, it will be interesting to assess the effects of pathologic processes such as cerebral ischemia.
CONCLUSION
In this study we have shown the following. (1) The high spectral resolution CCD approach allows us to measure hemoglobin oxygenation changes and changes in the redox state of Cyt-Ox in the human occipital cortex during visual stimulation. Because the individual DPF is measured the concentration changes are quantitative. Additionally the absolute local [Deoxy-Hb] is determined. (2) The spectra obtained in a state of increased brain activity cannot be explained solely by the well-known changes in [Oxy-Hb] and [Deoxy-Hb]; Cyt-Ox has to be included in the analysis. Only if Cyt-Ox is considered can the spectral changes in response to increased brain activity be explained. (3) In 9 of 10 subjects Cyt-Ox oxidation transiently increases significantly during visual stimulation.
Whereas other functional neuroimaging techniques measure either parameters related to the neurovascular response (e.g., positron emission spectroscopy, functional magnetic resonance imaging, single photon emission computed tomography, see Villringer and Dirnagl, 1995, for a review) or intracellular events (e.g., magnetoencephalography, EEG), NIRS has thus far the unique feature of being able to measure intravascular and intracellular events simultaneously.
Footnotes
Acknowledgment
The authors thank Dr. Britton Chance, Philadelphia, PA, for his helpful comments and support.