Abstract
Changes in cerebral blood oxygenation due to functional activation of the primary sensorimotor cortex during a unilateral finger opposition task were simultaneously mapped by deoxyhemoglobin-sensitive magnetic resonance imaging (MRI) and monitored by near-infrared spectroscopy (NIRS). Activation foci along the contralateral central sulcus displayed task-associated increases in MRI signal intensity, indicating a concomitant decrease of the focal concentration of deoxyhemoglobin. This interpretation was confirmed by simultaneous reductions in deoxyhemoglobin measured optically. Since observation of the latter effect required exact spatial matching of the MRI-detected activation foci and position of the fiber optic bundles (“optodes”) used for transmitting and receiving light, it may be concluded that optical recordings of changes in deoxyhemoglobin during functional challenge probe only a restricted brain tissue region. While deoxyhemoglobin responses seen by NIRS were smaller for ipsi- than for contralateral finger movements, task-related increases in oxyhemoglobin were rather similar between both conditions and, thus, seem to be less specific. Furthermore, no consistent changes were obtained for total hemoglobin during task performance, possibly due to the short timing of the repetitive protocol. In general, results underline, in humans, the hitherto assumed signal physiology for functional brain mapping by oxygenation-sensitive MRI and allow assessment of both constraints and practicability of functional studies by NIRS.
Keywords
Functional challenge of the brain is accompanied by regional changes in cerebral blood flow (CBF), volume (CBV), and oxygenation (CBO). A variety of magnetic resonance imaging (MRI) techniques have been proposed to map sites of neuronal activation by monitoring changes in these parameters (for an overview see Society of Magnetic Resonance in Medicine, 1993). Work presented in this study benefits from recent modifications of gradient-echo MRI sequences that allow high-resolution functional neuroimaging of CBO changes, while reducing sensitivity to artifactual influences from altered flow velocity and motion (Frahm et al., 1994). Near-infrared spectroscopy (NIRS) is an optical method that assesses concentration changes of oxy-and deoxyhemoglobin (oxy-Hb and deoxy-Hb, respectively) by measuring changes of absorption at different wavelengths (Cope and Delpy, 1988; Villringer et al., 1993).
Although MRI and NIRS methods both detect CBO changes during functional brain activation, MRI is characterized by excellent spatial resolution while NIRS provides specificity of several oxygenation-related parameters. The purposes of this study were to combine respective strengths of each method and compare findings obtained by simultaneous recording of cortical activation during a sequential finger opposition task. Part of this work has been presented in abstract form (Obrig et al., 1994).
METHODS
NIRS recordings
A NIRO-500 monitor (Hamamatsu, Herrshing, Germany) measured changes in optical densities (ODs) at wavelengths of 775, 825, 850, and 904 nm with a sampling rate of 1.9 kHz. According to an algorithm described by Cope, absolute concentration changes of oxy-Hb, deoxy-Hb, and total-Hb (oxy-Hb + deoxy-Hb) were calculated (Cope et al., 1988) and data averaged over 1-s periods. Pertinent data are expressed in arbitrary units. If the mean optical pathlength is assumed to be 5.9 times the interoptode distance, 1 arbitrary unit equals 1 μM (compare with van der Zee et al., 1990). Two-fiber optic bundles (“optodes”), emitting and collecting near-infrared pulsed laser light, were fixed to a subject's head with an interoptode distance of 3.0–4.0 cm [1.5–2.0 cm medial and lateral position C3* in a modified 10–20 system (Steinmetz et al., 1989), i.e., always above the left hemisphere]. Thus, the interoptode axis was intended to superimpose onto the central sulcus. Initially, overall NIRS response quality and specific response patterns during contralateral task performance were assessed. If task-related deoxy-Hb decreases were observed, we proceeded to obtain simultaneous MRI and NIRS measurements. If no response could be elicited, we repositioned the optodes above the central sulcus and the presumptive primary sensorimotor hand area. This process was guided by anatomic MRI in which optode position was identified both by skin indentation and vitamin E capsules attached to the optodes' head. Even then, optimal precision was not achieved in all subjects since, for practical reasons, only up to three repositionings were allowed.
MRI recordings
Nine right-handed healthy subjects (32 ± 9 years of age, five men and four women) were studied at 2.0 T (Siemens Magnetom, Erlangen, Germany) using a standard circularly polarized headcoil. Informed written consent was obtained prior to the investigations. MRI of CBO was based on dynamic acquisition of spin density-weighted images with T*2 sensitivity (radio frequency-spoiled FLASH, repetition time/echo time/flip angle [TR/TE]/α = 62.5/30/10°). Sections of 4-mm thickness were recorded with an in-plane resolution of 0.78 × 1.56 mm2 (for further details see Frahm et al., 1994). As shown in Fig. 1, sections were oriented either parallel to the bicommisural plane or tangential to the hemispheric convexity and individually aligned to cover the sensorimotor hand area underlying the optodes.
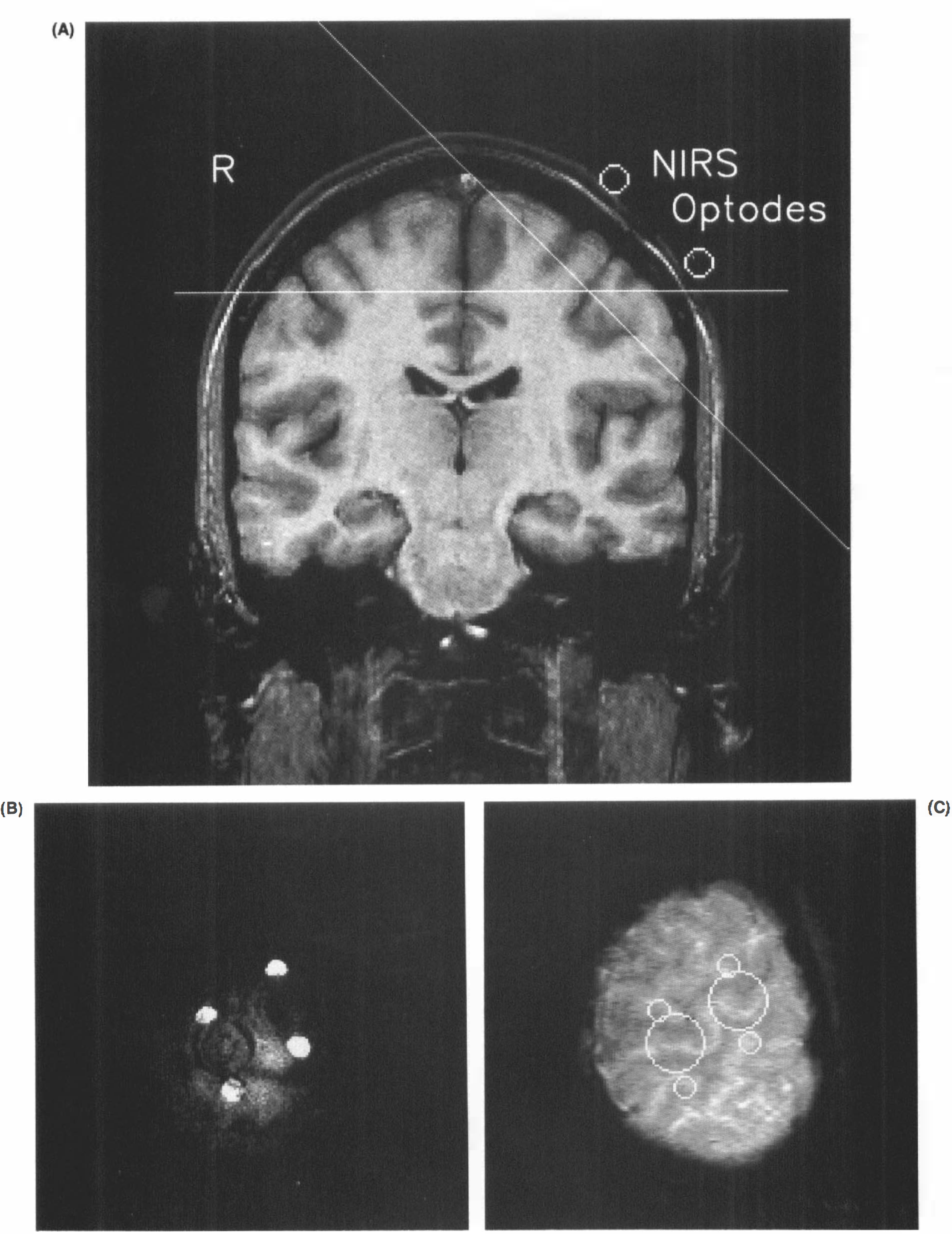
Design for simultaneous MRI and NIRS of brain activation.
MRI data analysis
The stimulation protocol comprised six cycles of unilateral sequential finger opposition (18 s) and rest (36 s). MRI-based activation maps were calculated on a pixel-by-pixel basis by correlating signal intensity time courses with a box-car reference waveform as an idealized “on/off” response function shifted by one datapoint (6 s) relative to the stimulation protocol to account for hemodynamic latency (Bandettini et al., 1993). To reduce single pixel noise, time course intensities were spatially filtered by averaging with the eight neighboring pixels. The resulting map of correlation coefficients was color-coded, thresholded, and overlaid on a corresponding flow-sensitized anatomic image (TR/TE/α = 70.3/7.5/60°) of identical spatial resolution. Thresholding of activation maps followed a recently introduced individual analysis procedure defining specificity of “activation” at an error probability level of 0.05% by accounting for global variations in the distribution of correlation coefficients and, subsequently, analyzing contingent spatial distribution (Kleinschmidt et al., 1995).
NIRS data analysis
For all MRI sections and activation cycles in each subject, NIRS data were time-locked to the onset of finger opposition. Three temporal windows were defined for statistical evaluation: T1, mean value of the 18-s period before motor activation (preactivation baseline); T2, mean value of 18-s period following onset of motor performance with a 6-s latency (maximal response); and T3, mean value of the remaining 12-s, period (postactivation baseline). Three hypotheses were submitted to statistical analysis: (i) a change of NIRS parameter values occurs in response to motor performance; (ii) this response is lateralized, i.e., different for contra- and ipsilateral task performance; and (iii) the NIRS response depends on the optode localization relative to the MRI activation focus. The first two hypotheses were tested in all subjects who performed the motor task contra- and ipsilateral to the optodes positioned over the left hemisphere (n = 8). The third hypothesis was tested in a single subject in whom contralateral motor performance was compared before and after optode repositioning (detailed in Results section).
A two-factorial multivariate analysis of variance (MANOVA) with polynomial contrasts (SPSS+ software) was used to test for the significance of influence of the condition factor, CI (contra-versus ipsilateral condition), and the response factor T (temporal window in either condition), as well as their cross-correlation LAT = CI × T. A significant influence of factor T indicates an NIRS signal change in response to motor performance (first hypothesis), whereas the cross-correlation factor, LAT, indicates response lateralization (second hypothesis). It is important to note that a significant influence of the condition factor, CI, does not adequately indicate lateralization, as only values in the temporal window T2 are expected to differ between conditions. The factor CI is not of interest in this analysis, but may, occasionally, become significant due to baseline drifts or lateralized responses. This, however, does not impair validity of the factor T and the cross-correlation factor LAT.
RESULTS
MRI
In all subjects, MRI recordings yielded activation maps showing task-related signal changes mainly in the primary sensorimotor hand area contralateral to the performing hand. An example is depicted in Figs. 2A,B, demonstrating that, although in principle equivalent, transverse MRI sections are not as favorable as tangential orientations for assessing the topographic relationship between optodes and activation sites. In the (left) primary sensorimotor hand area, weak responses to ipsilateral (left hand) task performance were detected in only a few runs. Activation was also seen close to the midline corresponding to the supplementary motor area and inconsistently in premotor and parietal cortices. A comprehensive comparison of response patterns with this and other hand motor tasks has appeared in abstract form (Boecker et al., 1994).
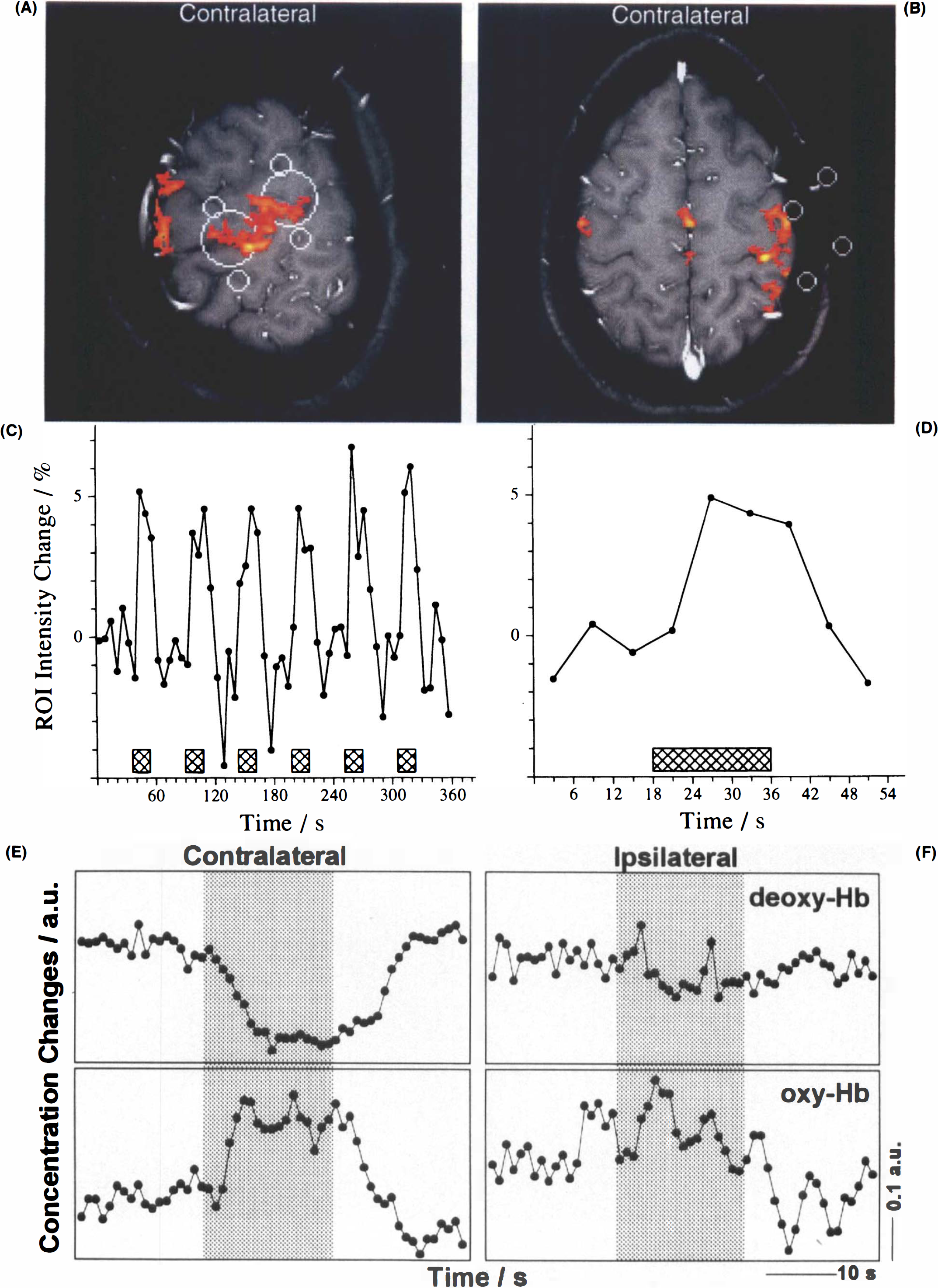
MRI of CBO and NIRS during right hand motor performance.
NIRS
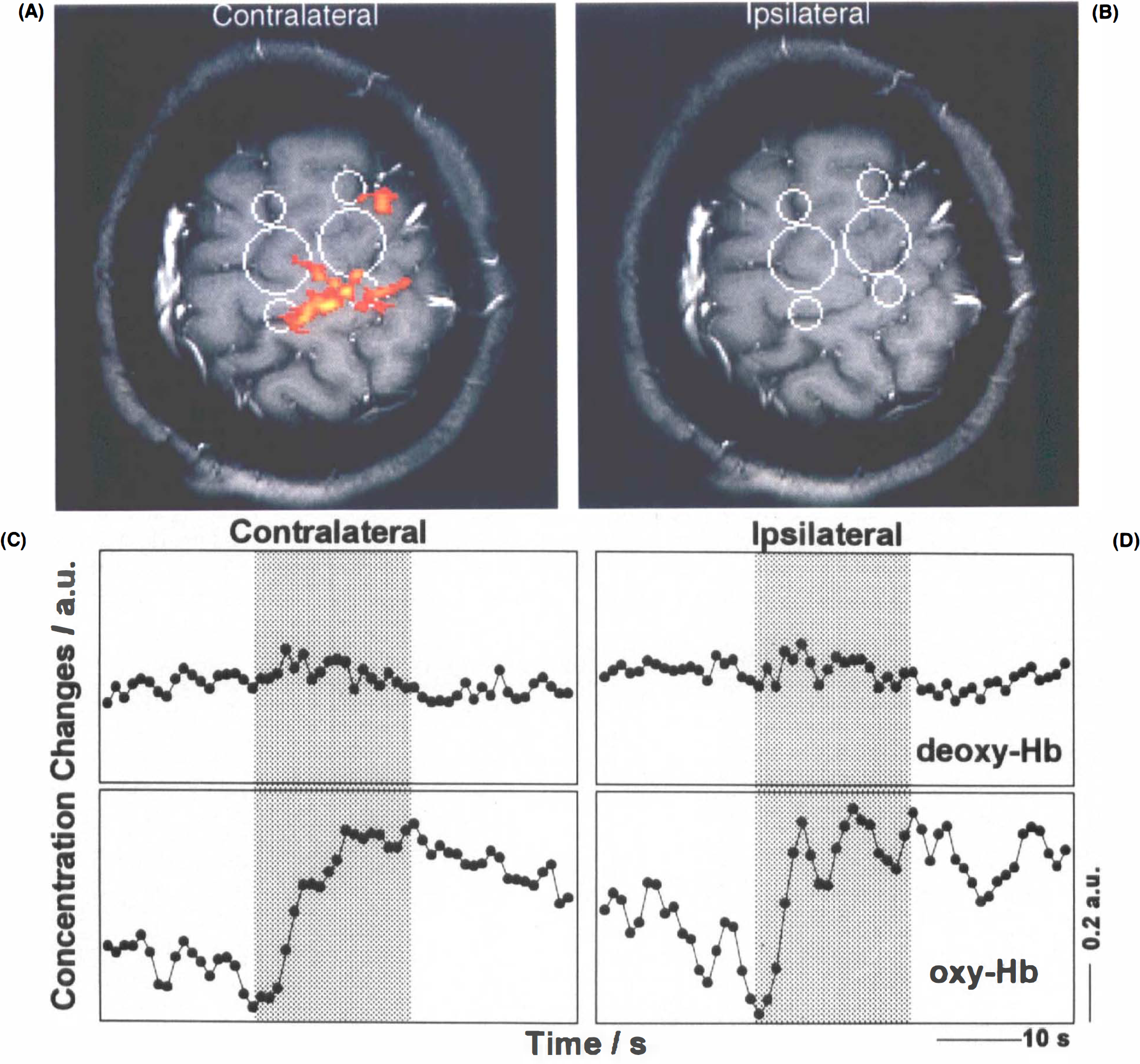
In all subjects but one, motor activity was accompanied by significant decreases in deoxy-Hb (p ≤ 0.01) (Table 1). The response followed the onset of task performance, with a latency of several seconds, and persisted for some seconds after task execution ceased. This is illustrated in Fig. 2 for contralateral hand performance in one of the subjects in whom optodes were in optimal proximity to the primary sensorimotor hand area. The figure displays one of several contiguous tangential (Fig. 2A) and transverse (Fig. 2B) activation maps as well as an original MRI signal intensity time course from an activated region (Fig. 2C), and a time-locked average of the same response (Fig. 2D). Corresponding NIRS findings averaged across all runs of contralateral task execution (Fig. 2E) are complemented by findings for ipsilateral hand activation (Fig. 2F). Although the temporal resolution of the MRI data is lower than that of NIRS, averaging the MRI responses from single runs, e.g., Fig. 2D, in an analogous manner as the NIRS data, yielded time courses that mirrored the deoxy-Hb signal detected by NIRS, e.g., Fig. 2E.
Statistical analysis of NIRS recordings
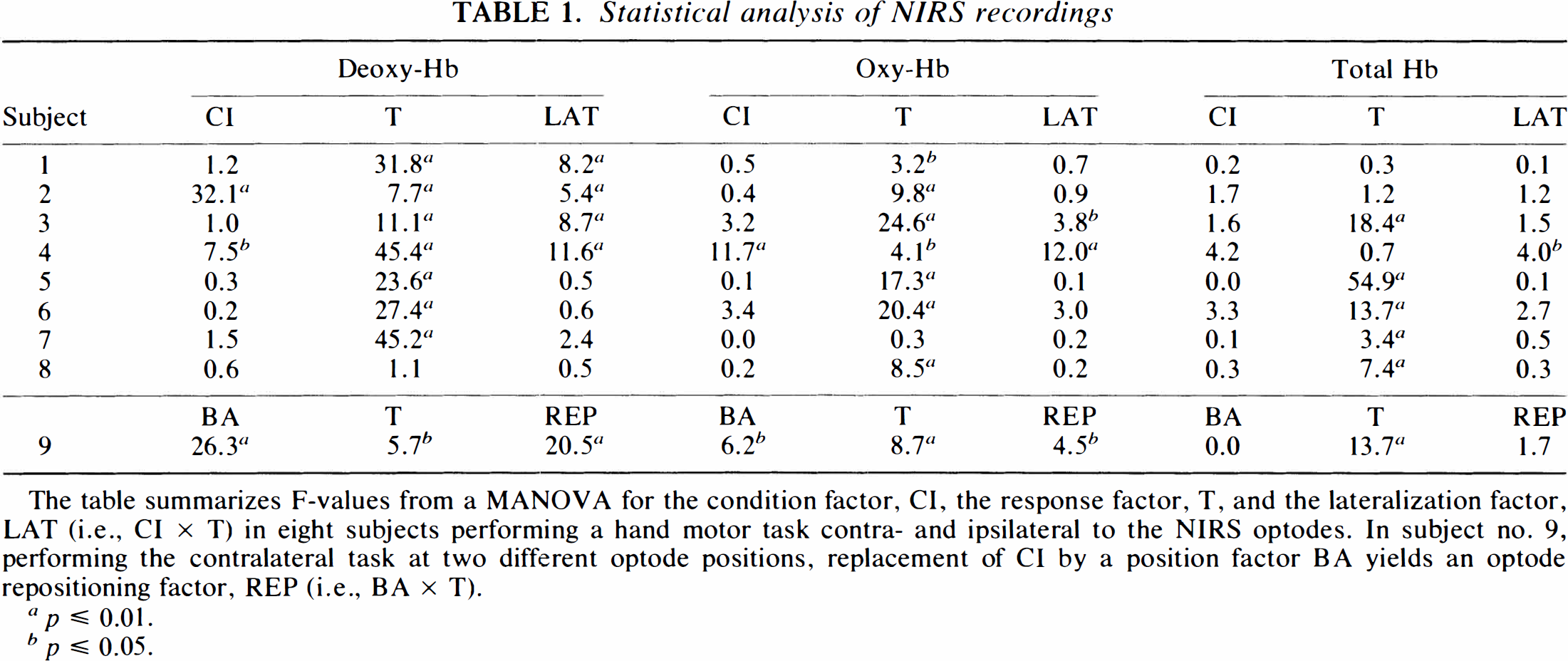
The table summarizes F-values from a MANOVA for the condition factor, CI, the response factor, T, and the lateralization factor, LAT (i.e., CI × T) in eight subjects performing a hand motor task contra- and ipsilateral to the NIRS optodes. In subject no. 9, performing the contralateral task at two different optode positions, replacement of CI by a position factor BA yields an optode repositioning factor, REP (i.e., BA × T).
p ≤ 0.01.
p ≤ 0.05
Recordings for both contra- and ipsilateral task performances at identical optode positions were obtained in eight subjects. As demonstrated in Fig. 3, deoxy-Hb concentration changes were strongly lateralized in favor of the contralateral side (p ≤ 0.01 when pooling all subjects, for single subjects see Table 1). Qualitatively, the closer the correspondence between interoptode path and hand area defined by MRI, the better the NIRS response strength during contralateral task execution than during ipsilateral. This did not hold true for ipsilateral task performance, in which no systematic dependence was detected.
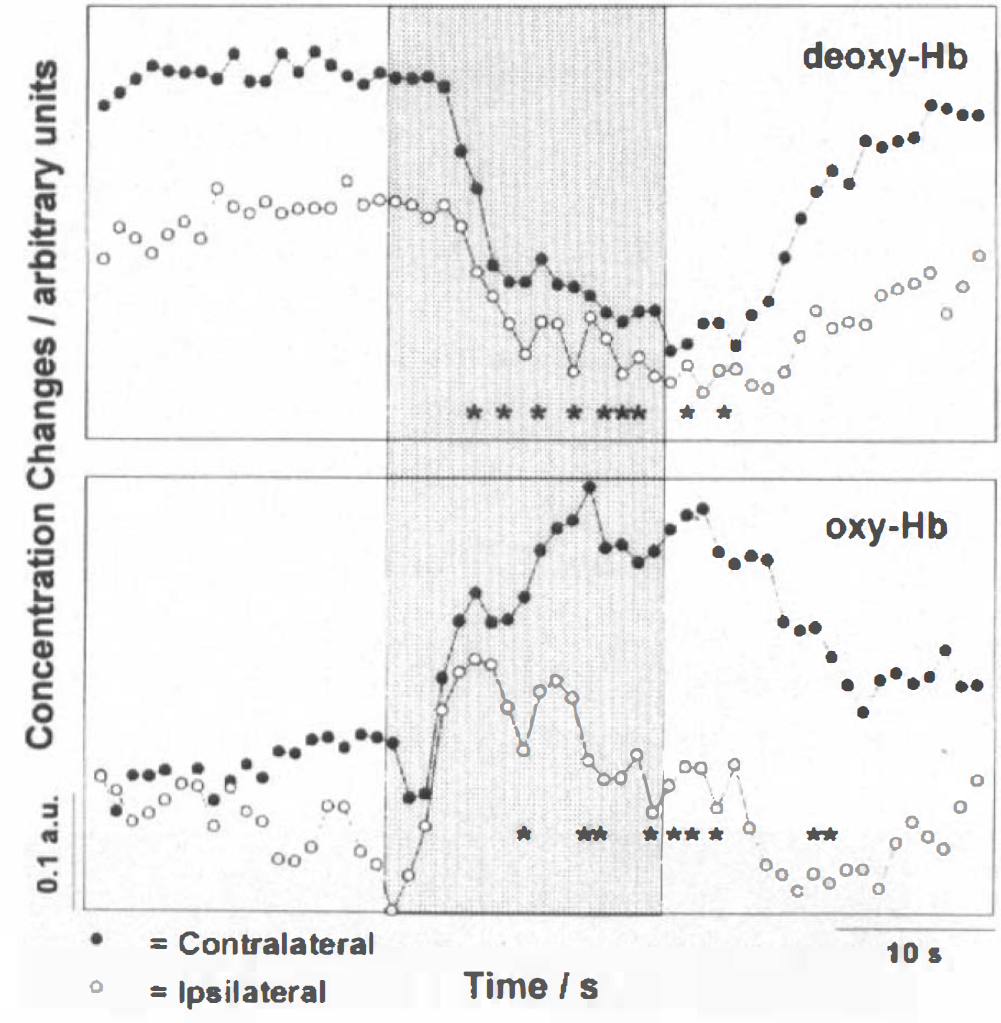
NIRS during contra- and ipsilateral hand motor performance. Time-locked grand average of NIRS recordings of deoxy-Hb and oxy-Hb for all sections and volunteers during contra- and ipsilateral task performances, respectively.
For oxy-Hb, increases (p ≤ 0.05 in seven subjects) (Table 1) better in the initial phase were parallel irrespective of contra- or ipsilateral task performance. Lateralization was not seen until later phases, as shown in Fig. 3, for the grand average of time-locked NIRS responses across all subjects. It achieved significance only in two subjects (p ≤ 0.05) (Table 1). One volunteer presented a task-related decrease in oxy-Hb despite a regular deoxy-Hb decrease. Interestingly, for contralateral task performance, oxy-Hb changes often mirrored, rather than exceeded, deoxy-Hb decreases (Fig. 2E). Consequently, total Hb remained virtually unchanged during stimulation in three volunteers and even decreased in one volunteer. When pooling total Hb data across subjects, no significant response or significant lateralization was detected. In several subjects, a more prominent feature of oxy-Hb and total Hb was a postactivation undershoot.
In one subject (no. 8 in Table 1), no deoxy-Hb response was detected by NIRS despite a pronounced and lateralized CBO-sensitive MRI response (Figs. 4A,B). In the absence of NIRS deoxy-Hb changes, however, this subject displayed significant, but not lateralized, increases in oxy-Hb and total Hb (Figs. 4C,D, Table 1). This may be explained by nonoptimal optode positioning and/or individual intervening variables, such as changes in skin perfusion (“red head” phenomenon).
MRI and NIRS of CBO in a subject without deoxy-Hb response.
For contralateral task execution, grand averages of NIRS parameters in all subjects time-locked to the performance of dynamic MRI are presented in Fig. 5. This group response comprises task-associated absolute deoxy-Hb decreases and oxy-Hb increases largely corresponding to those shown in Fig. 2 for an individual subject. While the deoxy-Hb responses and their lateralization were significant (p ≤ 0.01) when pooling all subjects, this was not the case for oxy-Hb changes, despite significant responses in most single subjects (Table 1). The lack of statistical significance in the pooled data is due to the greater intersubject variability of oxy-Hb, and, thus, also total-Hb responses.
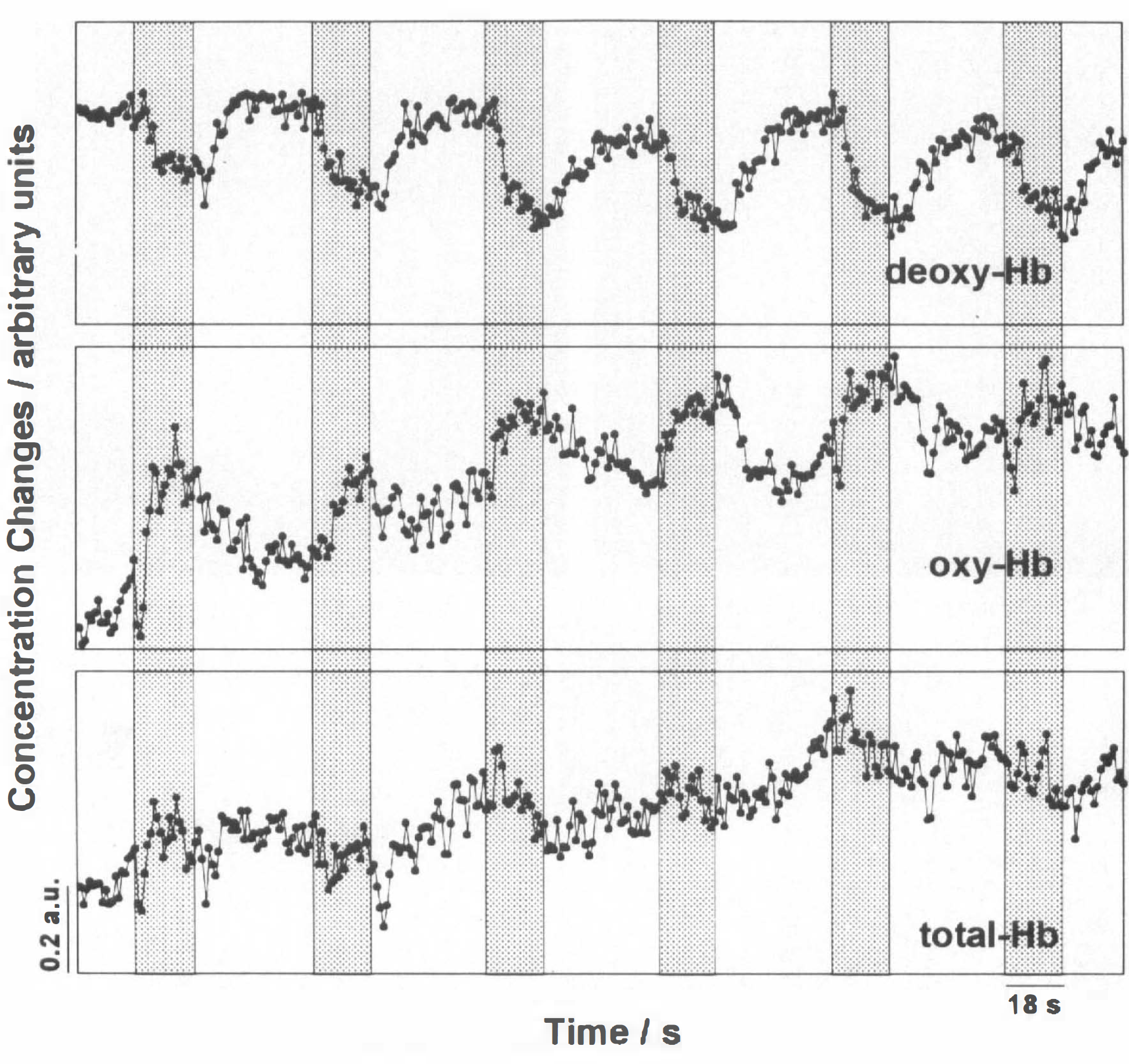
NIRS during contralateral motor performance across volunteers. Time-locked grand average of NIRS recordings of deoxy-Hb, oxy-Hb, and total-Hb for all MRI runs and volunteers.
A paramount factor accounting for intersubject variability even for the more robust deoxy-Hb responses is that topographic congruence of MRI and NIRS could only be achieved within certain limits. In many subjects, preliminary NIRS runs at the initial optode positions that followed external bony landmarks produced poor or no deoxy-Hb decreases. In these cases, the optode positions did not correspond to the actual position of the central sulcus. However, responses could be improved after realigning the optodes according to the individual anatomy even if the actual optode position was shifted by ≤1 cm. To address this issue formally, simultaneous MRI recordings before and after optode repositioning were performed in one subject. Corresponding activation maps and NIRS findings are shown in Fig. 6. In the MANOVA design for this subject, the response factor, T, was retained, but the condition factor, CI, replaced by the position factor, BA, and the repositioning factor, REP, calculated as the cross-correlation BA × T. The influence of this repositioning factor, REP, on NIRS values before and after correcting optode localization was significant for both deoxy-Hb (p ≤ 0.01) and oxy-Hb (p ≤ 0.05), but not total Hb (Table 1).
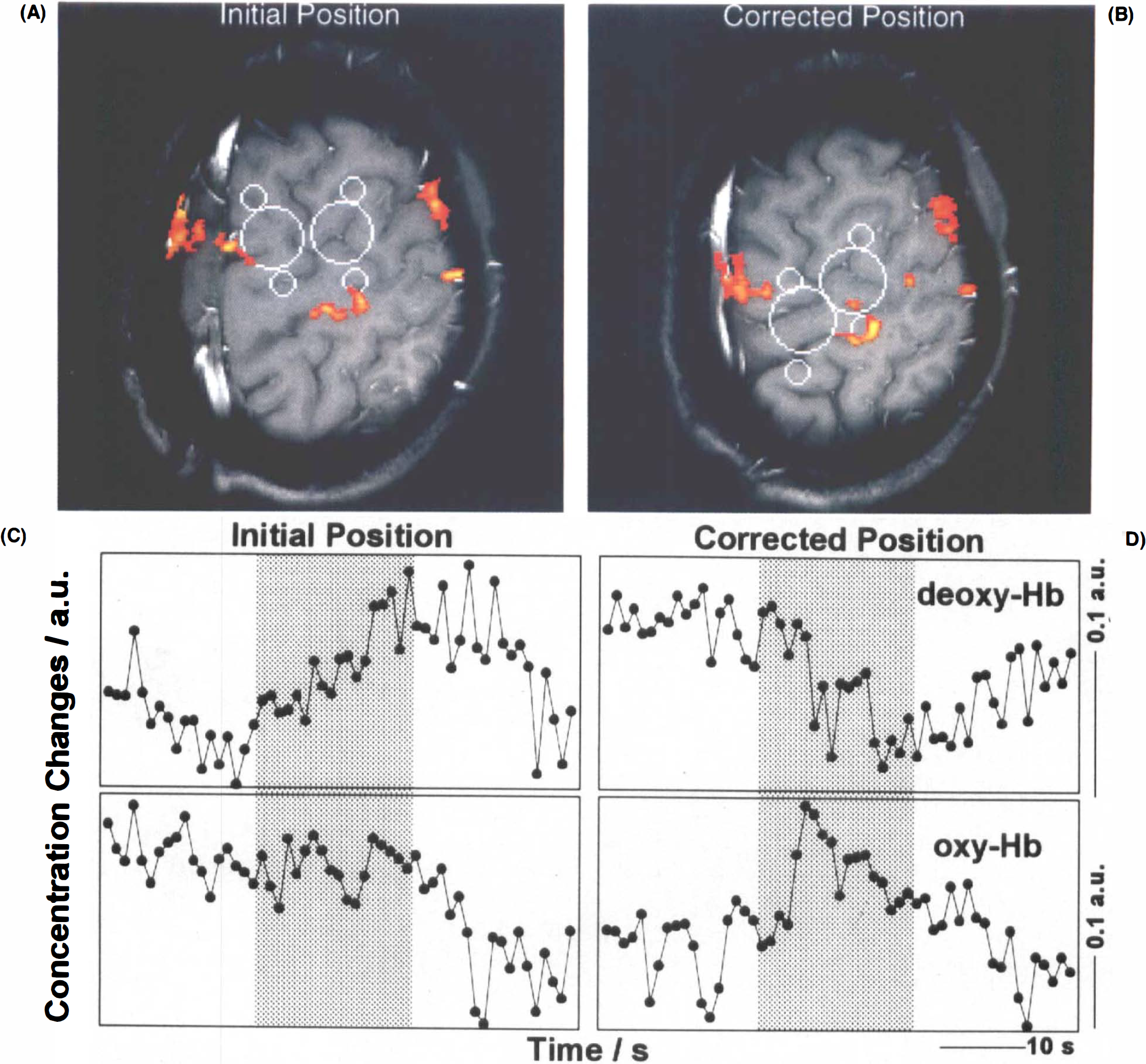
MRI and NIRS of CBO at initial and corrected optode positions.
DISCUSSION
Signal physiology in functional MRI
The two new methods introduced over the last few years for noninvasive monitoring of human brain function—CBO-sensitive MRI and NIRS—are at opposite ends of the existing span in spatial resolution provided by functional recording techniques, but share sensitivity for changes in blood oxygenation. While there are MRI techniques measuring changes in blood volume or flow, the most widely used approach for functional mapping—is assumed to reflect endogenous contrast related to the concentration of paramagnetic deoxy-Hb (Thulborn et al., 1981; Ogawa et al., 1990). In particular, absolute decreases in deoxy-Hb, which reduce microscopic susceptibility effects, yield MRI signal increases. Extending animal studies that combined MRI and optical recordings of CBO (Turner et al., 1993), the present study attempted to complement functional MRI in humans by noninvasive direct NIRS measurements of CBO. NIRS signals are believed to stem from a banana-shaped volume between the two optodes, with a penetration depth of ∼2–3 cm from the head surface (van der Zee et al., 1990).
Confirming expectations about the nature of focal and transient MRI signal changes, NIRS directly demonstrated simultaneous decreases in deoxy-Hb in response to task performance. Moreover, these effects were only observed if the presumptive optical sample volume covered the activation focus mapped by MRI. While this indicates that these foci actually display deoxy-Hb changes, it must be pointed out that MRI specificity may depend on the technique chosen for functional mapping. The use of low flip angle, gradient echo images implemented here (in conjunction with relatively long TRs) greatly reduces artifactual influences from flow (or motion) while retaining sensitivity to deoxy-Hb-dependent changes in magnetic susceptibility (Frahm et al., 1994). Under these conditions, task-related focal MRI signal changes temporally and spatially match changes in deoxy-Hb measured by NIRS, and, thus, underline the common physiologic mechanism monitored by both techniques.
Lack of deoxy-Hb responses in NIRS
There are two possible explanations for the absence of deoxy-Hb responses in optical recordings: mispositioned optodes with respect to activation foci or a failure to sample effectively from activated brain tissue due to individual differences in skull absorption or skin pigmentation. Therefore, interpretation of a lack of NIRS deoxy-Hb changes in relation to brain function would require control studies, which, for example, may lead to a positive response in a different paradigm.
As for optode positioning, this study has illustrated that the use of external bony landmarks is not precise enough to account for interindividual anatomic variability (Steinmetz et al., 1989) and the restricted NIRS sample volume. In fact, absence of task-related deoxy-Hb responses during initial recordings that were already close to a subsequently optimized position above the central sulcus clearly indicate that NIRS sensitivity is focused onto brain tissue beneath the interoptode axis. This finding holds true at least as far as deoxy-Hb changes are concerned. While qualitatively the relationship between optode position (relative to the MRI activation focus) and strength of corresponding NIRS signal changes was obvious, a quantitative modeling of optical responses would have to account for variability in such factors as skull thickness, skin absorption, interoptode distance, and angulation as well as scattering properties of underlying brain structures and potential wavelength dependencies. For functional NIRS studies, it may be concluded that careful optimization of optode positions, preferably in conjunction with anatomic MRI, is mandatory.
NIRS deoxy-Hb responses to ipsilateral performance
Given the topographic specificity of the deoxy-Hb response, it comes as some surprise that weaker, but still considerable, NIRS signal changes were seen during ipsilateral task execution (Fig. 3), whereas most MRI-based mapping revealed no such or extremely weak responses (e.g. Fig. 4B). Although a nonlateralized extracerebral response of the deoxy-Hb signal cannot be excluded with certainty, this possibility or that of a spatially unspecific “activation” effect do not seem probable, as no consistent deoxy-Hb changes were observed at optode positions only 1–2 cm away from the central sulcus. Furthermore, ipsilateral responses cannot be explained by very restricted areas of activity described in other functional MRI studies (Kim et al., 1993), since such foci would have been easily detected by the high-resolution MRI technique applied here (see Frahm et al., 1993). It remains possible that averaged NIRS responses are sensitive to weak NIRS alterations throughout the sample volume that escape detection when studied by a voxel matrix as in MRI. Sources for such CBO changes no only include primary sensorimotor cortex but also may extend to adjacent cortex activated during task performance.
Oxy-Hb responses in NIRS
Contrasting the findings for deoxy-Hb, changes in oxy-Hb did not display such a close relationship to topographically mapped activation sites, especially with respect to response lateralization. This underlines that oxy-Hb concentration changes provide information on an aspect of hemodynamic changes that differs in meaning from deoxy-Hb (or MRI) and seems less specific for functional recording of focal cerebral responses. Given the ratio of oxygenated and deoxygenated hemoglobin, the absolute oxy-Hb concentration is much higher and may, therefore, more readily reflect changes not so much of oxygenation but, rather, blood volume and flow. This may result in lower baseline stability due to systemic flow fluctuations, including conceivable “contamination” by extracerebral tissue. Interestingly, however, this could also account for the low degree of response lateralization seen during the first seconds of motor performance (compare Fig. 3), as global blood flow changes are known to occur during the transition from a “resting” to an “activated” state (Sitzer et al., 1994). Similar considerations may apply for an undershoot phenomenon that was observed in oxy-Hb rather than deoxy-Hb time courses.
Lack of total-Hb responses in NIRS
Another striking observation was the absence of a consistent task-related change in total-Hb. While previous studies often reported an increase in total-Hb or CBV, some of our volunteers even showed decreases under activation. Vasodilation is a consistent and localized feature of functional brain activation. A rise in overall CBV despite unchanged corpuscular CBV (equivalent to total-Hb) would reflect altered hematocrit. Changes of local cerebral hematocrit associated with flow velocity changes have been proposed based on animal experiments (for a review see Mchedlishvili, 1986). The difference between changes in corpuscular CBV and plasma volume may account for the discrepancy of the findings described above and those obtained by CBV measurements assessing plasma volume. Another possible explanation lies in the temporal protocol of task performance. The timespan chosen between subsequent intervals of task performance (pauses of 36 s) may be too short to allow for recovery of vasomotor tone. Grand averages of oxy-Hb and total-Hb across subjects (Fig. 5) tended to increase monotonously during the recording of six cycles of activation (18 s) and rest (36 s) for contra-but not ipsilateral motor activity. This suggests that the total-Hb increases might have been masked by the relatively rapid repetition of motor activation periods. Clearly, with protocols optimized for MRI of brain activation, deoxy-Hb becomes the main determinant of MRI signal changes, whereas the influence of volume (and flow) changes is further attenuated.
Footnotes
Acknowledgment:
We thank Dr. Ulrich Mansmann from the Institut für medizinische Statistik, Universitätsklinikum Benjamin-Franklin, Freie Universität, Berlin, for invaluable assistance in Statistical data evaluation. In part, data analysis and figure preparation used the facilities of the Gesellschaft für wissenschaftliche Datenverarbeitung Göttingen. A.V. and U.D. received support from the Deutsche Forschungsgmeinschaft (G. Hess Programm, VI 93/7/1).
