Abstract
The purpose of this work was to determine whether cerebral endothelial cells have the capacity to synthesize histamine or to express mRNA of receptors that specifically respond to available free histamine. The histamine concentrations and the expression of L-histidine decarboxylase (HDC) and histamine H1 and H2 receptor mRNA, both in adult rat brain and in cultured immortalized RBE4 cerebral endothelial cells, were investigated. In this study endothelial cells were devoid of any kind of detectable histamine production, both in vivo and in the immortalized RBE4 cells in culture. Both the immunostainings for histamine and the in situ hybridizations for HDC were negative, as well as histamine determinations by HPLC, indicating that endothelial cells do not possess the capacity to produce histamine. Also, glucocorticoid (dexamethasone) treatment failed to induce histamine production in the cultured cells. Although the cerebral endothelial cells lack histamine production, a nonsaturable uptake in RBE4 cells is demonstrated. The internalized histamine is detected both in the cytoplasm and in the nucleus, which could indicate a role for histamine as an intracellular messenger. Histamine H1 and H2 receptor mRNA was expressed in RBE4 cells, and glucocorticoid treatment down-regulated the mRNA levels of both H1 and H2 receptors. This mechanism may be involved in glucocorticoid-mediated effects on cerebrovascular permeability and brain edema.
The presence of histamine in the central nervous system has been known since the beginning of this century, and several lines of evidence suggest that histamine can act as a neurotransmitter in the brain (see Schwartz et al., 1991). Intracerebral histamine is also a known regulator of cerebral blood flow (Edvinsson et al., 1993) and endothelial permeability (Dux and Joó, 1982). The effect of histamine on target cells and organs is conducted through three types of receptors, H1, H2 and H3, coupled to G proteins (Leurs et al., 1995).
Histamine has been detected in three different cellular compartments of the central nervous system: in hypothalamic tuberomammillary neurons and their processes (Panula et al., 1984; Watanabe et al. 1984), perivascular mast cells, and isolated cerebral microvessels (for review see Edvinsson et al., 1993). A relatively high content of histamine (Robinson-White and Beaven, 1982), but low activity of histamine-synthesizing (L-histidine decarboxylase; HDC) and -metabolizing (histamine-N-methyltransferase) enzymes, has been demonstrated (Karnushina et al., 1980) in microvessel-enriched fractions isolated from guinea pig brain. Histamine is also present in blood vessels isolated from the bovine brain (Jarrott et al., 1979). This could suggest that histamine is synthesized locally in the brain microvessels, as in endothelial cells of peripheral organs (El-Ackad and Brody, 1975; Schayer, 1966), but that it has a slow turnover. Despite the application of specific and sensitive immunocytochemical methods for histamine (Panula et al., 1984, 1988, 1990; Steinbusch et al., 1984) and HDC (Watanabe et al., 1984), histaminergic markers have not been documented in vivo in capillary endothelial cells in the rat brain. This study was conducted to reveal whether an active synthesis of histamine occurs within the cerebral endothelial cells and to elucidate the histamine receptor mRNA expression in these cells. To avoid contamination from other cell types associated with primary cultures, we used an immortalized rat brain endothelial cell line (RBE4), an accepted in vitro model of the blood-brain barrier (BBB). The RBE4 cell line is of rat cerebral origin (Durieu-Trautmann et al., 1993), and the cells have been characterized in detail (Roux et al., 1994) and found to retain the main features of cerebral endothelial cells kept in primary cultures.
Potent physiological effects of histamine on the brain vessels have been shown both in vivo and in vitro. In the periphery, histamine induces formation of interendothelial gaps (Majno and Palade, 1961), and ferritin-labeled histamine accumulates over the perijunctional filament web (Heltianu et al., 1982). Histamine also depolarizes endothelial cells (Northover, 1980), increases intracellular Ca2+ levels, and promotes trypan blue-albumin diffusion through a layer of polycarbonate filter and endothelial cell monolayer through an H1 receptor mechanism (Rotrosen and Gallin, 1986). Injected histamine has furthermore been reported to increase pinocytocis in rat brain, and histamine increases synthesis of prostaglandins in capillary-rich fractions of rat brain, a mechanism that clearly differs from that observed in peripheral endothelial cells (Joó et al., 1981). However, neither H1 nor H2 receptors have previously been demonstrated in endothelial cells derived from brain vessels.
Steroids have been used in treatment of cerebral ischemia and edema (De Reuck et al., 1988, Betz and Coester, 1990a), although the mechanism of action is not clearly defined. Dexamethasone also attenuates the development of ischemic brain edema in gerbils (Dux et al., 1990) and in rats (Betz and Coester, 1990b). Possible mechanisms include release of mediators that inhibit phospholipase A2 (Blackwell et al., 1980; Sztriha et al., 1986), but direct actions on endothelial cells have also been observed (De Kloet, 1984; Ziylan et al., 1989). Histamine H2 antagonists also have beneficial effects on ischemic brain edema (Tosaki et al., 1994) and traumatic edema (Mohanty et al., 1989) in rats. However, the expression and regulation of histamine receptors have not been studied at a cellular level in brain endothelium. Histamine induces pinocytotic activity in brain endothelial cells (Joó, 1986), an effect inhibited by application of the H2 antagonist metiamide (Dux and Joó 1982), which may indicate that the cellular mechanism of histamineinduced increase in BBB permeability involves H2 receptors. We hypothesized that endothelial cells may express H1 and/or H2 receptors that are regulated by steroids.
METHODS
Cell cultures
The cultured cells used in this study are immortalized rat brain microvessel endothelial cells. They originate from primary rat cerebral endothelial cells that are transfected with the plasmid pE1A-neo, containing the adenovirus E1A encoding sequence and a gene for neomycin resistance (Durieu-Trautmann et al., 1993). We used a clone, RBE4, characterized by Roux et al. (1994). Cells were grown on collagen-coated (Sigma, St. Louis, MO, U.S.A.; rat tail type I collagen) dishes or coverslips, in Dulbecco's modified Eagle's/Ham's F-10 (1:1) cell culture medium (Gibco, Grand island, NY, U.S.A.) supplemented with 10% fetal calf serum (Gibco), 2 mmol/L L-glutamine, and 1 ng/mL basic fibroblast growth factor (PeproTech, London, U.K.).
Prior to incubations with histamine, the cultured cells were washed with phosphate-buffered saline (PBS; pH 7.1) supplemented with 0.5 mmol/L ethylenediaminetetraacetate. The experiments were then done in the indicated culture medium with the fetal calf serum excluded. In experiments for histamine synthesis, the cells were maintained in normal culture medium supplemented with 1 mmol/L L-histidine and 10 μmol/L pyridoxal-5-phosphate. All dexamethasone incubations were carried out in freshly prepared normal culture medium supplemented with proper concentrations of dexamethasone (Helsinki Univ. Pharmacy, Helsinki, Finland).
Animal experiments
For immunostainings, male Sprague-Dawley rats (250 to 300 g) were anesthetized with sodium pentobarbital (Mebunat) and perfused through the left heart ventricle with freshly prepared 4% 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDAC; Sigma) in 0.1 mol/L sodium phosphate buffer (pH 7.4), as described by Panula et al. (1989), followed by 2% paraformaldehyde (PFA). For in situ hybridization, male Sprague-Dawley rats were fixed by a similar transcardial perfusion with 4% PFA in 0.1 mol/L phosphate buffer (pH 7.4). The PFA-fixed brains were removed and postfixed in the same fixative for 1 hour. For cryoprotection, all brains were, after fixation, kept in 0.1 mol/L phosphate buffer (pH 7.4) containing 20% sucrose until they sank.
Immunocytochemistry
Immunostaining for histamine in 20-μm-thick cryostat sections of the rat brain was carried out using the biotin-streptavidin method followed by visualization of peroxidase activity with diaminobenzidine and H2O2. Tissues were handled and all appropriate controls were done as previously described (Panula et al., 1984, 1988, 1990). The histamine antiserum (Panula et al., 1990) was diluted 1:250,000. The stained sections were examined with a Nikon Diaphot-TMD inverted microscope equipped with Nomarski optics. Cultured RBE4 endothelial cells were washed briefly in PBS (pH 7.4) and fixed in 4% EDAC and 0.5% PFA in 0.1 mol/L phosphate buffer (pH 7.4) at room temperature for 15 minutes. The coverslips were then washed in PBS containing 0.25% Triton X-100 (PBS-T), followed by incubation with a histamine anti-serum diluted (1:1,000) in PBS-T containing 1% normal swine serum for 1 hour at room temperature. After washing in PBS-T, the cells were incubated for 30 minutes at room temperature with fluorescein-conjugated swine anti-rabbit IgG (Dakopatts, Copenhagen, Denmark) diluted 1:40 in PBS-T. After a final wash with PBS, the coverslips were embedded in glycerol (80% in PBS) on glass slides and examined with a Leitz Aristoplan fluorescence microscope and a Leica TCS40 confocal laser scanning microscope using the program SCANware 4.2a. Twenty-five images were collected with 0.1- to 0.18-μm intervals through the thickness of the cells. Individual images were examined to reveal the subcellular distribution of histamine immunoreactivity, and maximal projection images were produced to visualize the total distribution in two-dimensional images. As controls, representative coverslips were incubated with histamine antiserum preabsorbed with a histamine-bovine serum albumin conjugate. Control slides with the primary antibody incubation omitted were also examined. The histamine antiserum has been characterized extensively, and it does not detect L-histidine or L-histidine-containing peptides (Panula et al., 1990).
HPLC measurements
For measurements of histamine uptake, the RBE4 cells were washed twice with PBS (pH 7.4) and then scraped loose in PBS with a cellscrape. The volume and weight of the cell pellet were measured, and the cells were resuspended and lysed in 10× original volume of 2% perchloric acid. Cells treated for studies on histamine synthesis were directly lysed on the culture dishes with 1 mol/L NaOH and collected with a glass rod. All samples were directly acidified by addition of perchloric acid to a final 2% concentration. Histamine concentrations were measured by HPLC according to the method of Yamatodani (1985; also Mochizuki et al., 1991) with a cation-exchange column followed by postcolumn derivatization and fluorescence detection.
Reverse transcriptase polymerase chain reaction amplification
Total RNA from all indicated tissues or cells was extracted according to Chomczynski and Sacci (1987). For HDC amplification, we also used mRNA isolated with the PolyATtract mRNA Isolation System 1000 (Promega Corp., Madison, WI, U.S.A.). cDNA was prepared with the First-Strand cDNA Synthesis Kit (Pharmacia Biotech, Sweden). All reactions were done with the pd(N)6 primer according to the protocols provided by the manufacturer. An aliquot of the first-strand cDNA was amplified by polymerase chain reaction (PCR) with specific primers. The primers used for rat HDC amplification were specific for nucleotides 1,119 to 1,148 and 1,733 to 1752 (nucleotides according to Joseph et al., 1990), for rat H1 receptor nucleotides 1,771 to 1,790 and 2,101 to 2,120 (nucleotides according to Fujimoto et al., 1993), and for rat H2 receptor nucleotides 1,119 to 1,146 and 1,630 to 1,647 (nucleotides according to Ruat et al., 1991). PCR was performed in a final volume of 50 μL containing 1 μmol/L of each primer, 200 μmol/L of each dNTP, 10 mmol/L Tris-HCl (pH 8.8) at 25°C, 1.5 mmol/L MgCl2, 50 mmol/L KCl, 0.1% Triton X-100, and 1 U DynaZyme DNA polymerase (Finnzymes Oy, Finland). After being overlaid with mineral oil, the samples were first heated for 5 minutes at 94°C, and then 40 cycles of amplification were done in a thermal cycler (Mini Cycler; MJ Research, Watertown, MA, U.S.A.). Each cycle consisted of 30 seconds at 94°C, 30 seconds at 69°C, and 90 seconds at 72°C (for HDC amplification); 30 seconds at 94°C, 1 minute at 63°C, and 1 minute at 72°C (H1 receptor); and 30 seconds at 94°C, 1 minute at 61°C, and 1 minute at 72°C (H2 receptor). All amplifications were ended with a 7-minute incubation at 72°C before the samples were cooled to 4°C. Aliquots of amplified cDNA were analyzed by electrophoresis on a 1.8% agarose gel in buffer containing 89 mmol/L Tris-HCl, 89 mmol/L boric acid, and 1 mmol/L ethylenediaminetetraacetate (pH 8.0). After electrophoresis, agarose gels were stained with ethidium bromide and documented on an ultraviolet transilluminator. The sizes of the amplified cDNA fragments were estimated from co-migrated size markers (λ-DNA HindIII digested and/or MX174-DNA HinfI digested).
In situ hybridization on tissue sections
L-Histidine decarboxylase in situ hybridization on rat brain sections was carried out with a nonradioactive digoxigenin (DIG)-labeled probe, and an in situ hybridization technique for free-floating sections was applied. As a template for probe synthesis, we used the 5′ end of a rat HDC cDNA clone, bases 1 to 1,132 (nucleotides according to Joseph et al., 1990), in the pGEM 7Zf(+) vector (Promega, U.S.A.). All probes were synthesized according to the instructions from the manufacturer of the DIG label (Boehringer-Mannheim, Mannheim, Germany). Twenty-micron-thick free-floating cryostat sections of PFA-fixed rat brains were prepared. The sections were washed in PBS and then pretreated in the following solutions: 0.02 mol/L HCl for 10 min, two rinses in PBS (5 minutes each), PBS-T for 3 minutes, two rinses in PBS (5 minutes each), 0.2 μg/mL proteinase K in 5 mmol/L ethylenediaminetetraacetate and 50 mmol/L Tris-HCl (pH 7.5) for 15 min, 2% PFA for 5 min, and two rinses in PBS for 5 minutes each. After these pretreatments, the sections were incubated for 2 hours at 50°C with a prehybridization solution containing 50% formamide, 750 mmol/L NaCl, 25 mmol/L ethylenediaminetetraacetate, 25 mmol/L piperazine-N,N'-bis(2-ethanesulfonic acid), 2.5× Denhardt's solution, 250 μg/mL tRNA, and 250 μg/mL ssDNA. Sections were then transferred to the hybridization solution for at least 16 hours at 50°C. The solution consisted of the prehybridization solution with 5% dextran sulfate and 200 ng/mL DIG-labeled RNA probe heated to 80°C and cooled on ice before use. Following the hybridization, the sections were washed briefly in 2× saline sodium citrate and treated for 30 minutes at 37°C with 50 μg/mL RNase A in 500 mmol/L NaCl, 1 mmol/L ethylenediaminetetraacetate, and 10 mmol/L Tris-HCl (pH 8.0). This was followed by rinsing in the same buffer without RNase A, for 30 minutes at room temperature, two washes in 2× saline sodium citrate at room temperature, two washes in 1× saline sodium citrate at room temperature, and one wash in 0.1× saline sodium citrate at 55°C (30 minutes each). The sections were washed twice for 10 minutes in 150 mmol/L NaCl and 100 mmol/L Tris-HCl (pH 7.5) (Buffer 1), incubated for 1 hour in 0.5% blocking reagent (Boehringer-Mannheim) in Buffer 1 with 0.03% Triton X-100, and then incubated overnight at 4°C with anti-DIG alkaline phosphatase-conjugated Fab fragments (Boehringer-Mannheim) diluted 1:1,000 in Buffer 1 containing 0.03% Triton X-100. The sections were washed twice for 30 minutes in Buffer 1 and for 10 minutes in 100 mmol/L NaCl, 50 mmol/L MgCl2, and Tris-HCl (pH 9.5) (Buffer 2). They were then incubated overnight in a chromogen solution containing 45 μL of nitro blue tetrazolium (toluidine salt, 75 mg/mL in 70% dimethylformamide), 35 μL of 5-bromo-4-chloro-3-indolyl phosphate (50 mg/mL in 100% dimethylformamide), and 2.4 mg of levamisole in 10 mL of Buffer 2. The reaction was stopped by washing the sections in 10 mmol/L Tris-HCl and 1 mmol/L ethylenediaminetetraacetate (pH 8.0). The sections were mounted on gelatin-coated slides, dehydrated, and coverslipped with Permount (Fisher Scientific, U.S.A.).
As specificity controls for HDC hybridizations, we used a sense probe complementary to the antisense probe. Also, pretreatment of the tissue sections with RNase A, to abolish specific hybridization, was routinely used. In all experiments, antisense and control hybridizations were done in parallel.
In situ hybridization on endothelial cell cultures
H1 and H2 receptor in situ hybridizations on cultured endothelial cells were done with [α-35S]UTP (56103H; ICN Pharmaceuticals, CA, U.S.A.) -labeled cRNA probes. The H1-specific antisense probe was made by cloning the SacI-KpnI restriction enzyme fragment from a rat genomic H1 receptor clone (Fujimoto et al., 1993) into the pGEM-7Zf(+) vector. After linearization of the template with BstEII, the synthesized cRNA probe was ∼500 bases long and covered the complete 5′ end of the coding sequence. To prepare a rat H2 receptor-specific probe, we applied reverse transcriptase PCR amplification. As template we used total RNA isolated from rat brain and 20-base-long primers covering the outermost ends of the coding region of the rat transcript (Ruat et al., 1991). The amplified coding region was cloned into the pBluescript KS vector and used as template for probe synthesis. The synthesized cRNA probe was ∼1,080 bases long, covering the complete coding sequence. Both for H1 and for H2 hybridizations, a corresponding sense probe was synthesized and used as a control. Before the endothelial cells were hybridized with respective probes, they were washed in PBS and fixed in 4% PFA (in PBS) at room temperature for 20 minutes. After two washes in PBS, the cells were treated with proteinase K (2 μg/mL in PBS) at 37°C for 30 minutes, followed by a 2-minute fixation in 2% PFA (in PBS). The pretreatment was terminated in two PBS washes and dehydration in ethanol (70, 96, and 100%). Coverslips were used directly or stored overnight at +4°C. The hybridization and posthybridization treatments were done as described by Simmons et al. (1989). In situ hybridized coverslips were dipped in Kodak NTB-2 liquid autoradiography emulsion. After 3 to 5 weeks of exposure, the slides were developed according to standard procedures. The cells were then counterstained with cresyl violet, dehydrated in ethanol, mounted with Permount (Fisher Scientific), and examined with a Leitz Aristoplan microscope under light- and dark-field illumination. Dexamethasone-treated and control samples were always included in the same in situ hybridizations and treated identically.
RESULTS
Histamine immunocytochemistry on rat brain sections
The histaminergic neuronal system in the rat and in other vertebrates has been mapped and characterized extensively. In this study, we focused on three areas in the rat brain that have been reported to contain histamine in different compartments: the hypothalamus containing the tuberomammillary histamine neurons and the highest amount of histamine in the brain, the thalamus containing perivascular histamine-rich mast cells, and the cerebral cortex, which frequently is used for preparation of primary endothelial cell cultures. In rat hypothalamus, histamine was localized exclusively in tuberomammillary neurons, varicose nerve fibers in different parts of the hypothalamus (Fig. 1A), and in a few mast cells in the median eminence. Blood vessels were frequently surrounded by networks of histamine-immunoreactive fibers, but no immunoreactivity was seen in the endothelium (Fig. 1A). In the thalamus, numerous histamine-immunoreactive mast cells were seen in association with blood vessels (Fig. 1B); in the cerebral cortex, varicose nerve fibers were the only histamine-immunoreactive structures (Fig. 1C). No immunoreactivity in blood vessel endothelium could be found in any part of the brain, either in capillaries or in larger blood vessels (data not shown).
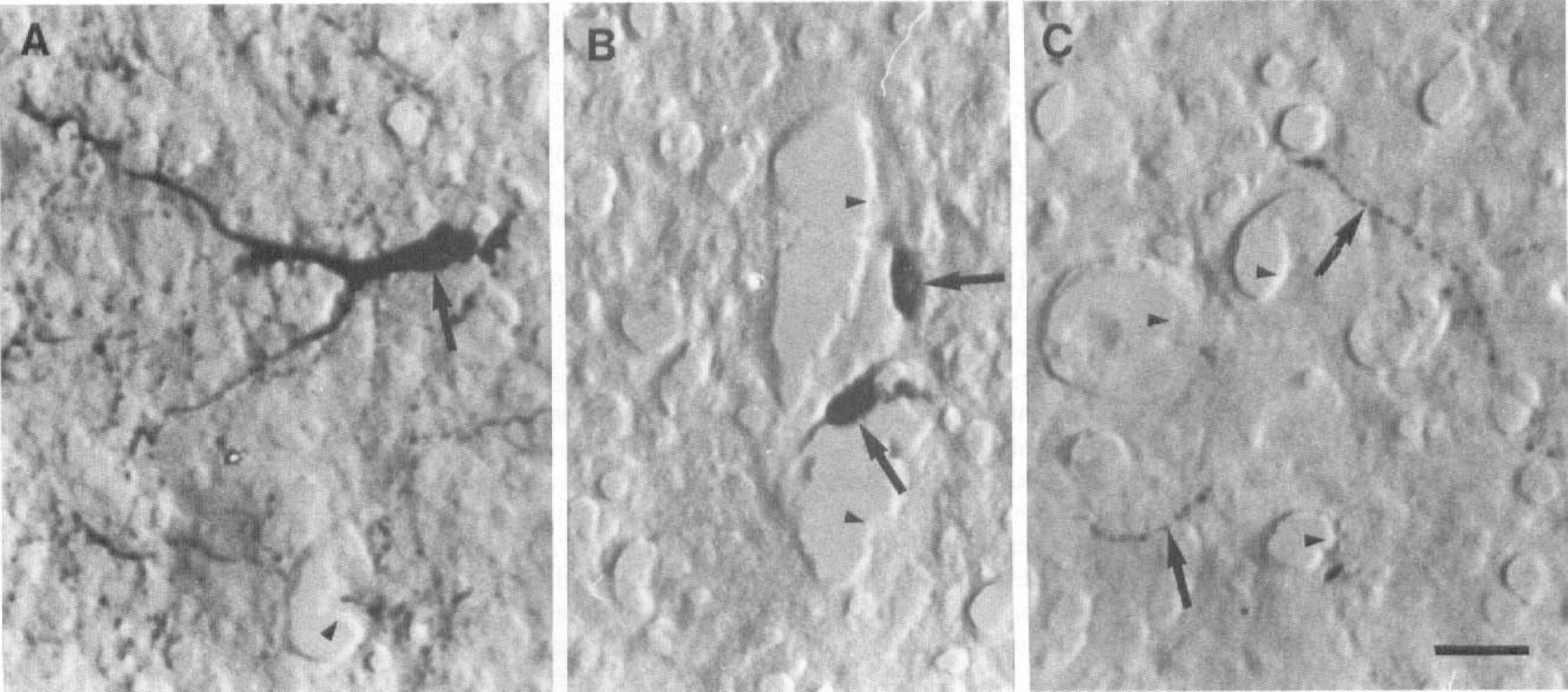
In these micrographs, the histamine-nonreactive cerebral blood vessel endothelium is indicated with arrowheads.
HDC in situ hybridization in rat brain sections
To be able to localize histamine-producing cells, we developed a sensitive in situ hybridization procedure for free-floating tissue sections using cRNA probes. The probes used for HDC detection were DIG labeled. The tuberomammillary neurons were intensely stained for HDC mRNA, whereas the identified capillaries and larger blood vessels within the same region (Fig. 2) and all capillaries in the cerebral cortex and the median eminence (data not shown) were devoid of detectable hybridization. No signal for HDC was detected in any cell type in cerebral cortex or thalamus. No signal was seen when the antisense probe was replaced by a sense probe or the sections were pretreated with RNase A before hybridization with the antisense cRNA probe. In situ hybridizations with 35S-labeled radioactive oligonucleotide probes were also performed, with identical results as with DIG-labeled probes (data not shown).
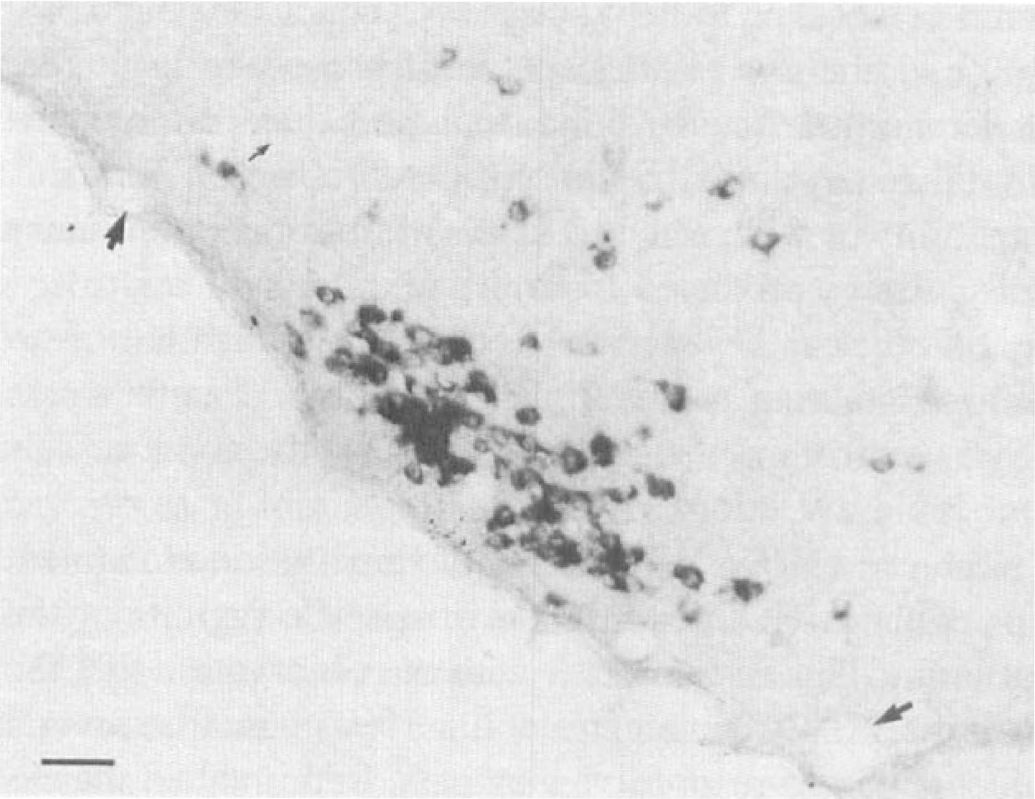
Expression of HDC in posterior basal hypothalamus of the rat. The hybridization is done with a DIG-labeled HDC cRNA probe. In the tuberomammillary nucleus, strongly stained cell bodies can be seen, whereas blood vessels (arrows) are nonreactive. Scale bar = 50 μm.
Endothelial cell cultures and histamine production
To further address the role of capillary endothelial cells in histamine metabolism, we used cultures of immortalized cerebral endothelial cells. Confluent cell cultures displayed only a weak unspecific background when stained for histamine immunoreactivity. We were further unable, with HPLC, to detect histamine in the cultured cells (detection limit was 5 fmol/sample). No detectable levels of histamine were found in the culture medium, so production and excretion of histamine are thus unlikely. The lack of HDC mRNA in these cells when tested by using in situ hybridization and reverse transcriptase PCR (Fig. 3) indicates that these cells, under normal cell culture conditions, lack the capability to synthesize histamine. Dexamethasone has been indicated as a upregulating agent for HDC expression. We tested this synthetic glucocorticoid on RBE4 cells from the nano- to micromolar range and from short (3-hour) to long (24-hour) time incubations, but no induction of HDC expression could be seen when tested with reverse transcriptase PCR (Fig. 3).
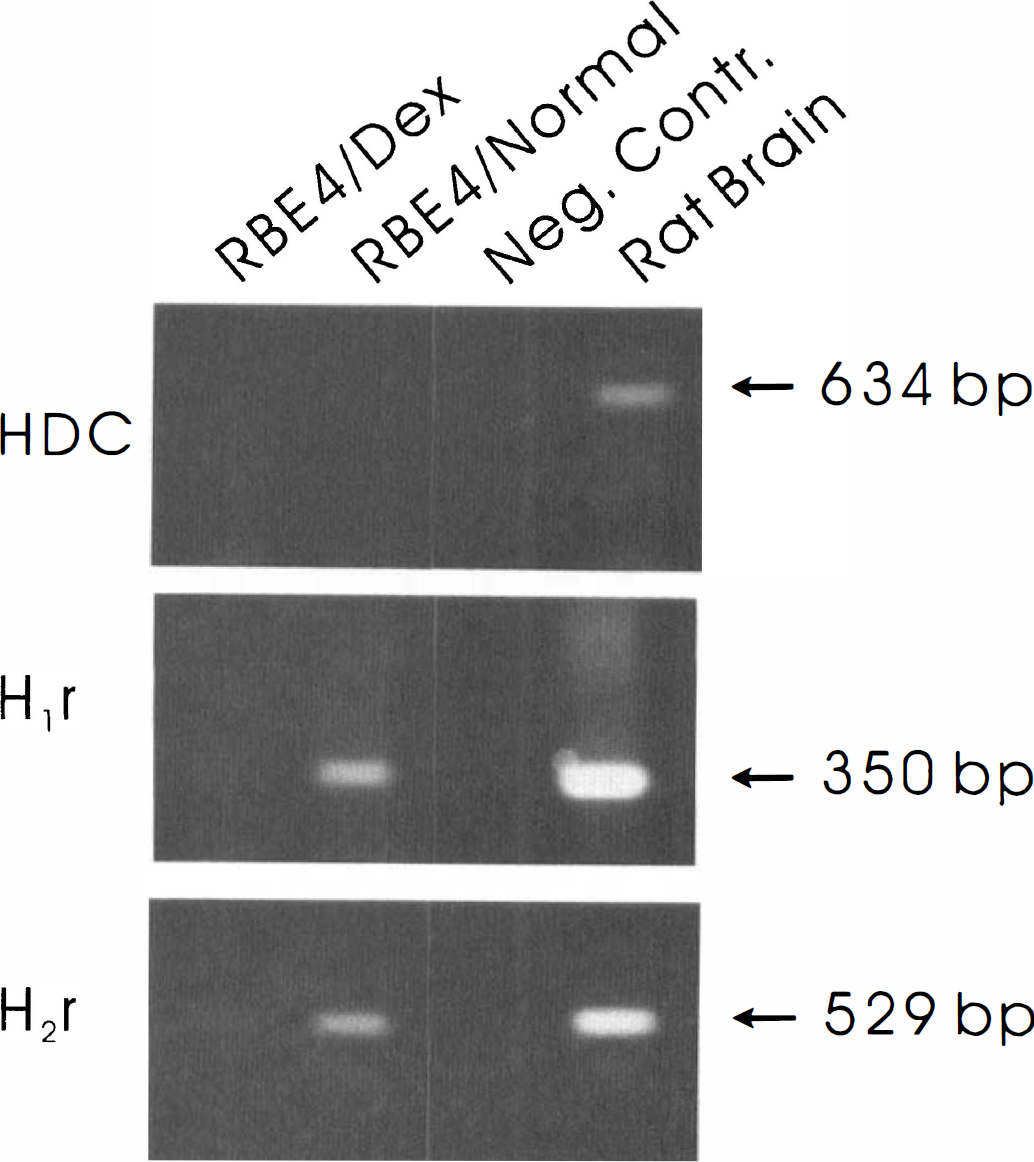
Reverse transcriptase PCR amplification of HDC, H1 receptor, and H2 receptor transcripts in rat brain tissue and cultured RBE4 cells. An equal amount of PCR reaction products was electrophoretically separated on a 1.8% agarose gel and stained with ethidium bromide. The PCR reactions were done on total RNA isolated from rat brain (positive control), nontreated RBE4 cells (RBE4/Normal), and RBE4 cells treated with 100 nmol/L dexamethasone for 24 hours (RBE4/Dex). In the negative controls, RNA is omitted from the reverse transcriptase reaction.
Uptake of histamine in RBE4 cells
The uptake mechanism of histamine was studied by culturing RBE4 cells in different concentrations of histamine followed by measurements of the internalized histamine at different time points. HPLC determinations indicate that the uptake is unspecific and concentration dependent (Fig. 4). The nonsaturable uptake suggests that there is no specific high-affinity histamine uptake mechanism in RBE4 cells, within the concentration range used. The uptake of histamine was confirmed by immunostainings and confocal laser scanning microscopy. Histamine was clearly detectable in the cytoplasm and partly also in the nucleus (Fig. 5A), in contrast to the control cells where only a weak background staining in the cytoplasm but nothing in the nucleus could be seen (Fig. 5B).
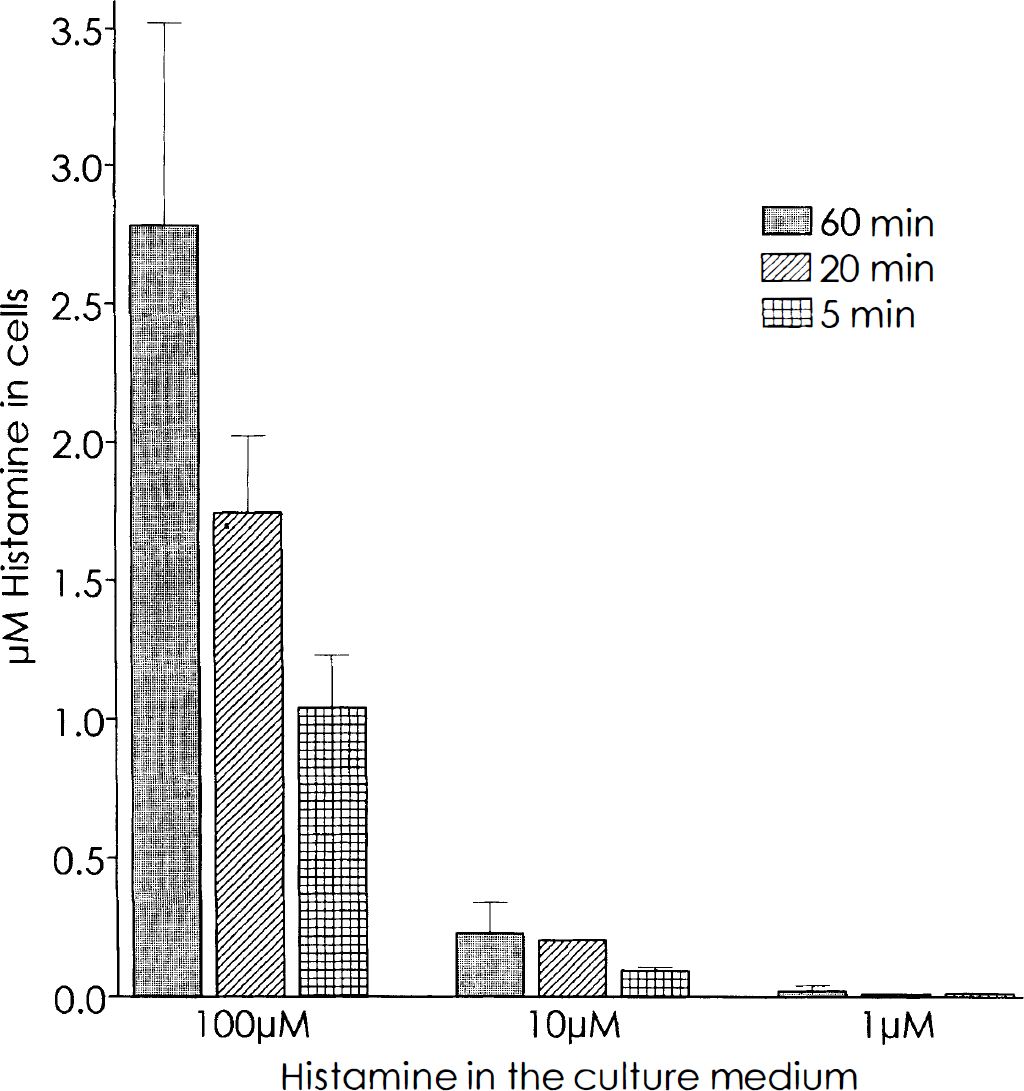
Analysis of histamine (HA) uptake in RBE4 cells after incubation in culture medium containing different concentrations of HA. The data are from one representative experiment with three samples measured per time and concentration point (error bars indicate SD).
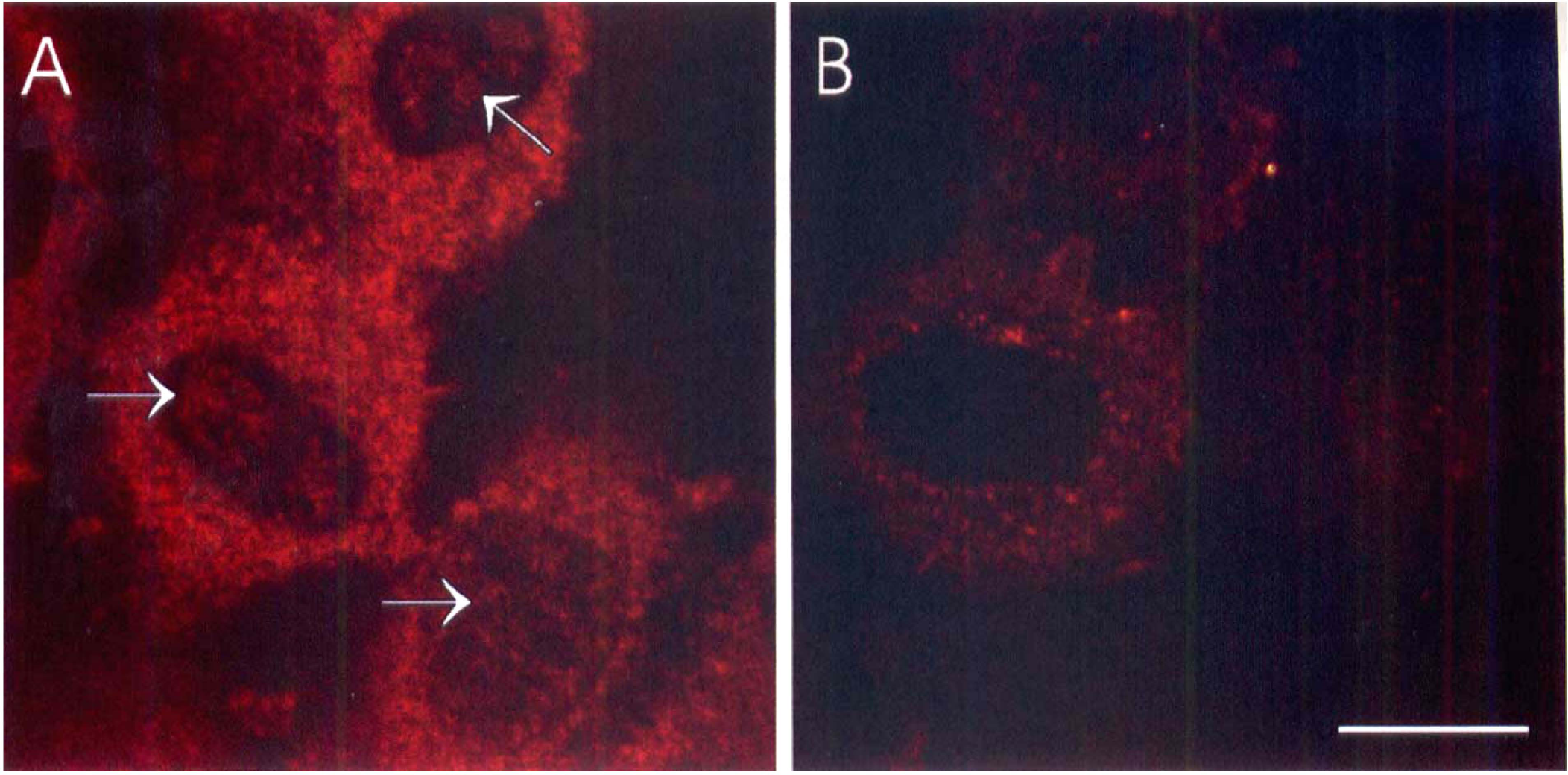
Confocal laser scanning micrographs with histamine-immunostained cultured RBE4 endothelial cells.
Histamine H1 and H2 receptor expression and regulation in RBE4 cells
The effects of histamine on endothelial cells can be mediated through H1 and H2 receptor mechanisms. As shown by reverse transcriptase PCR, both H1 and H2 receptor mRNA can be detected in RBE4 cells under normal cell culture conditions (Fig. 3). Dexamethasone treatment of RBE4 cells did not affect HDC expression, but both H1 and H2 expression were clearly downregulated (Fig. 3). In situ hybridization verified that both H1 and H2 receptors are expressed in RBE4 cells under normal cell culture conditions (Fig. 6A, B, E, and F). The down-regulation upon glucocorticoid treatment was also evident with in situ hybridization (Fig. 6C, D, G, and H).
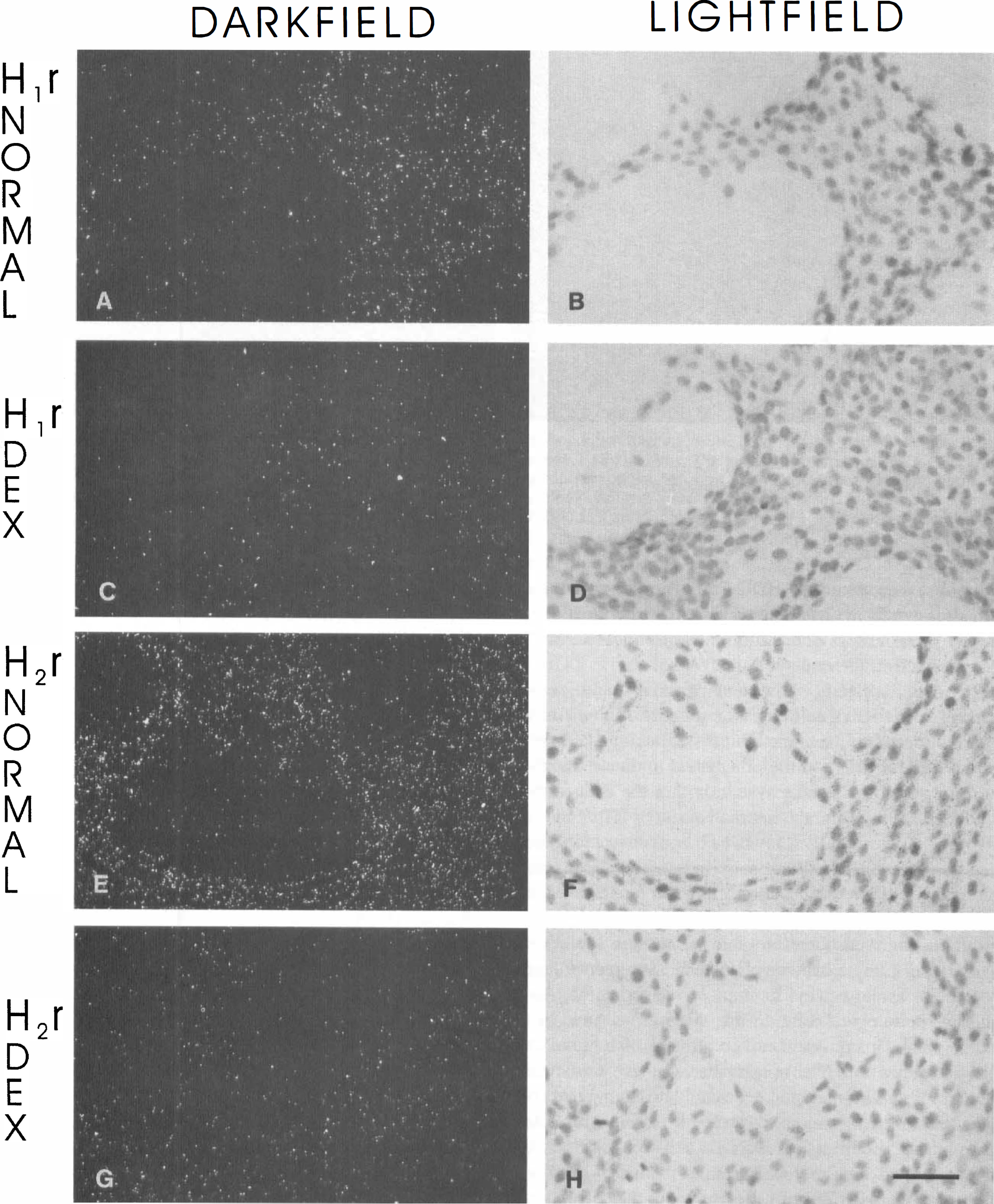
H1
DISCUSSION
Brain capillary endothelial cells have a crucial function not only as a barrier between the blood and the brain but also as a selective filter for substances circulating in the blood, of which only some are allowed to enter the brain tissue itself. Histamine concentrations in, e.g., human plasma may rise from 1 to 10 nmol/L to 4 μmol/L after intravenous injections of curare and to 10 μmol/L after morphine (Moss and Rosow, 1983), which suggests that variations in plasma histamine are very large and may activate mechanisms other than those associated with high-affinity receptors. As an in vitro model to study the BBB, we have used RBE4 endothelial cells. Although an immortalized cell line, these cells have in culture retained the endothelial “cobblestone” morphology. Several parameters, e.g., the expression of Factor VIII-related antigen, growth factor-dependent growth, and contact-inhibited proliferation, also indicate preserved endothelial characteristics (Roux et al., 1994). In a recent article, Quinonéro et al. (1997) showed that RBE4 cells can be useful as transplantation vehicles for gene products to adult rat brain. Grafted cells survived at least 1 year without any sign of tumor formation, and their growth location in close association with blood vessels further indicates a preserved endothelial-like phenotype. In this study, we show that endothelial cells can, under some physiological conditions, internalize high concentrations of histamine. This could indicate a possible role for histamine in intracellular signaling in endothelial cells.
There are no publications available with immunocytochemical evidence of histamine or HDC synthesis/expression in endothelial cells. Also, in this study we failed to detect any histamine immunoreactivity in the brain endothelium in vivo or in cultured RBE4 cerebral endothelial cells. Furthermore, HPLC measurements failed to detect any native histamine synthesis in RBE4 cells. There is, of course, a possibility that our methods do not detect histamine produced at very low levels. To overcome this problem, we developed a sensitive in situ hybridization protocol for detection of HDC expression. We used DIG-labeled probes, and in adult rat brain sections the only HDC-expressing structures detected were the neuronal cell bodies in the tuberomammillary nucleus in the hypothalamus. All other cell types in hypothalamus, thalamus, and cerebral cortex were devoid of HDC expression. We also performed conventional radioactive in situ hybridization to verify the negative results. The results with radioactive oligonucleotide probes were identical to the results from the DIG in situ hybridizations (data not shown). HDC in situ hybridization on RBE4 cells yielded similar negative results, indicating lack of HDC synthesis. Reverse transcriptase PCR amplification also showed lack of HDC expression in cultured cerebral endothelial cells. We concluded that cerebral endothelium is not capable of producing histamine under normal in vivo conditions and that cultured immortalized endothelial cells of cerebral origin lack this capability as well. The low levels of histamine in primary cell cultures produced from isolated cerebral microvessels (Joó et al., 1981) could be due to occasional histamine-containing mast cells and basophils (Panula et al., 1991) or from nerve endings attached to blood vessels (for review see Joó, 1985).
Zahnow (1995) showed that dexamethasone treatment up-regulates HDC expression in specific regions of the rat brain. This is consistent with the observation that the gene for HDC (Yatsunami et al., 1994) contains several glucocorticoid-responsive elements. In this study, the expression of HDC in RBE4 cells was unaffected by dexamethasone treatment. Reverse transcriptase PCR could not detect HDC up-regulation (or induction) after dexamethasone treatment, which suggests that the effect of dexamethasone on brain HDC is on neurons or mast cells. It is therefore histamine originating from the blood, nerve fibers, or mast cells within the brain that exerts its potent effect on the endothelium.
Huszti et al. (1995) showed that primary cultures of cerebral endothelial cells have a specific high-affinity uptake system for histamine. Our histamine uptake studies with RBE4 cells, on the other hand, indicate the absence of a specific uptake system within the concentration range studied, and the internalization of histamine in RBE4 cells was clearly shown to be concentration and time dependent. The discrepancy between these results can be due to the histamine concentration ranges used in the two studies. Huszti et al. (1995) used 3H-labeled histamine and were therefore able to measure low-level uptake of histamine, whereas HPLC measurements have a detection level at a few femtomoles of histamine. Another methodological difference is that we measured histamine uptake in confluent cell cultures on dishes, not on cells in suspension. The uptake that we have measured may represent luminal uptake, possibly reflecting the uptake mechanisms from the blood. To further elucidate this point, the polarization of RBE4 cells must be examined. The confocal microscopy data obtained on histamine uptake in RBE4 cells clearly show that the internalized histamine is distributed in the cytoplasm and partly also in the nucleus of the cells. Further experiments are needed to study if this nuclear transport occurs in living cells or represents a fixation artifact. Histamine has been proposed to be an intracellular messenger, mediating its effects through an intracellular histamine receptor associated with chromatin (see Brandes et al., 1990, 1992), but the significance of intracellular histamine remains to be elucidated.
To further characterize the significance of histamine in endothelial cells, we studied the expression and regulation of histamine receptors in the RBE4 cells. The receptor expression data presented are based on mRNA detection and thus represent regulation on the transcriptional level. Histamine is usually considered as a dilator of cerebral blood vessels, but there are clear differences in histamine responses depending on the size and location of the blood vessels (Hewett and Murray, 1993). Blood vessels of similar size in different brain areas do not always show identical responses to histamine, and differences between species have also been reported (for review see Edvinsson et al., 1993). Histamine H2 receptor binding (Karnushina et al., 1980) and H1 receptor binding (Peroutka et al., 1980) have both been reported in brain microvessel endothelium. However, the physiological relevance of the histamine receptors on endothelial cells has not been fully established. In peripheral endothelial cells, activation of H1 receptors can result in contraction and formation of junctional gaps large enough to allow leakage of plasma proteins (Hill, 1990). Such a mechanism has not been demonstrated in brain endothelial cells, in which histamine has been shown to increase pinocytotic activity instead of interendothelial leakage (Dux and Joó, 1982).
Our reverse transcriptase PCR and in situ hybridization experiments clearly indicated that RBE4 cells express both H1 and H2 receptors. It has previously been shown that H1 and H2 receptor antagonists can reduce BBB permeability and thereby inhibit formation of cerebral edema (Dux et al., 1987). We showed here that exposure of RBE4 cells to dexamethasone, a synthetic glucocorticoid, resulted in down-regulation of both H1 and H2 receptor expression. Glucocorticoids have been used to prevent edema formation, e.g., during surgery or acute ischemic stroke (De Reuck et al., 1988), although chronic dexamethasone pretreatment may also aggravate ischemic neuronal necrosis (Koide et al., 1986). Decreased permeability of the BBB as a result of down-regulated H1 and H2 receptors may contribute to decreased fluid accumulation in brain after both histamine receptor antagonist treatment and glucocorticoid administration. Dexamethasone down-regulates H1 receptor-induced inositol phosphate accumulation in human cultured airway smooth muscle cells (Hardy et al., 1996), which is in agreement with our findings on down-regulation of H1 receptor expression in RBE4 cells.
The data presented in this article have to be confirmed with receptor binding studies, and the physiological significance of these findings remains also to be verified in vivo. For this purpose, tools for detection of histamine receptor proteins will be needed to reveal the location of histamine receptors on the luminal and/or abluminal side of the endothelial cells in different classes of brain vessels.
