Abstract
Inactivation of the subthalamic nucleus (STN) has attracted interest as a therapeutic tool in Parkinson's disease. The functional consequences of the inactivation, however, are uncertain. In this study definition of the pattern of changes of cerebral functional activity associated with lesion of the STN and dopaminergic stimulation, by using the [14C]deoxyglucose method, was sought. Six or 7 days following unilateral lesion of the STN, the animals were divided into two groups: One group (n = 10) was administered apomorphine (1 mg/kg) subcutaneously; the second group (n = 10) received saline. The [14C]deoxyglucose procedure was initiated 10 minutes following the drug or saline injection. The results show that systemic administration of apomorphine to rats with unilateral lesion of the STN causes ipsiversive rotational behavior and asymmetries of glucose utilization of defined brain areas, including the substantia nigra reticulata, globus pallidus, and entopeduncular nucleus. These nuclei are the main targets of the subthalamic excitatory projections. Lesion of the nucleus per se (without challenge with apomorphine) has no significant consequences on glucose utilization. The findings indicate that the STN is involved in the activation of the basal ganglia output nuclei induced by systemic dopaminergic stimulation.
The subthalamic nucleus (STN) is a key component of the basal ganglia nuclei, a specialized neural circuit involved primarily in the control of voluntary movements. The circuit includes the corpus striatum, substantia nigra pars compacta, substantia nigra pars reticulata, medial globus pallidus, and lateral globus pallidus. According to a well known model (Albin et al., 1995; Gerfen, 1992), the striatum controls the basal ganglia outflow both directly and indirectly. A subset of striatal GABAergic neurons projects directly to the substantia nigra pars reticulata and medial globus pallidus, the output nuclei of the circuit. A different subset projects to the lateral globus pallidus, which sends GABAergic projections to STN. STN in turn sends excitatory, glutamatergic efferents to the substantia nigra pars reticulata, medial globus pallidus, and lateral globus pallidus (Kita and Kitai, 1987).
The model predicts that the dopaminergic denervation of the nigrostriatal pathway, the hallmark of Parkinson's disease, leads to increased activity of the output nuclei. This phenomenon is probably mediated by enhanced glutamatergic drive from STN. The prediction is based on a number of studies reporting increased neuronal firing rate (Bergman et al., 1994; Kreiss et al., 1997), increased glucose metabolism (Mitchell et al., 1989), and activity of mitochondrial enzymes (Porter et al., 1994), in STN or its projection nuclei, following denervation of the nigrostriatal pathway. Since hyperactivity of STN is thought to play a central role in the motor abnormalities of Parkinson's disease (DeLong, 1990), functional inactivation of STN has been proposed as a therapeutic tool for the disease (Limousin et al., 1995).
The model, however, has been the object of criticisms (Albin et al., 1995; Chesselet and Delfs, 1996). In particular, the role of STN in the functional organization of the basal ganglia nuclei is not defined. For example, the STN hyperactivity associated with the striatal dopamine denervation has been attributed to reduction of the inhibitory control exerted by the lateral globus pallidus (Albin et al., 1995). However, complete removal of the pallidal influence causes a slight increase in the firing rate of subthalamic neurons, far less pronounced than the increase observed in animal models of Parkinson's disease (Hassani et al., 1996). Thus, alternative explanations for STN hyperactivity may be taken into account. For example, the striatum might directly affect subthalamic activity (Kaatz and Albin, 1995). More importantly, STN receives dopaminergic afferents from the substantia nigra pars compacta (Canteras et al., 1990; Hassani et al., 1997). The roles of this pathway and of dopaminergic neurotransmission in general at the subthalamic level are uncertain. The mechanisms by which inactivation of the nucleus ameliorates parkinsonian symptoms are also unknown. In the rat, unilateral lesion of STN causes ipsilateral decreases in the activity of three different mitochondrial enzymes, complex I, II, and IV, in the substantia nigra pars reticulata, globus pallidus (the rodent homologues of the lateral globus pallidus), and entopeduncular nucleus (the rodent homologues of the medial globus pallidus) (Blandini and Greenamyre, 1995; Blandini et al., 1995). Furthermore, unilateral STN ablation reverses the effects on complex II and IV activity as well as the rotational response to apomorphine, a mixed D1/D2 dopamine agonist, induced by ipsilateral lesion of the nigrostriatal pathway (Blandini et al., 1997).
The definition of the mechanisms underlying the beneficial effects of STN inactivation on parkinsonian symptoms requires comprehension of the functional role of the nucleus under normal conditions. In this study we sought to evaluate the role of STN in mediating the effects of systemic dopaminergic stimulation on the functional activity of the basal ganglia output nuclei. To address this issue, we investigated the changes of local cerebral glucose utilization by means of the [14C]deoxyglucose method (Sokoloff et al., 1977) and motor behavior associated with inactivation of STN and subsequent dopaminergic stimulation. The measurements were carried out in freely moving rats, both in resting conditions and after pharmacological challenge with apomorphine.
MATERIALS AND METHODS
Male Sprague-Dawley rats (250-300 g) were used (Charles River, Calco, Italy). Animals were housed in group cages and maintained under standard temperature (21 ± 1°C) and light conditions (7 a.m. through 7 p.m.); food and water were available ad libitum, until the time of surgery.
Subthalamic lesions (day 1)
On day 1, animals were anesthetized with thiopental sodium (50 mg/kg body wt, intraperitoneally) and placed in a stereotaxic surgical apparatus; they received 0.2 to 0.3 µL of an N-methyl-
Apomorphine tests (days 3 and 5)
In a preliminary study, we had observed that the rotational response to apomorphine initiates ~24 hours after lesion and is most evident in the 2 to 4 following days (data not shown). Thus, 2 days after surgery (day 3), animals received a first subcutaneous injection of apomorphine, at a dose (1 mg/kg + 1% ascorbate) previously shown to elicit consistent rotational behavior in rats bearing unilateral lesion of STN (Murer et al., 1993). Three minutes after injection, animals were placed in fiberglass cylindrical boxes (40 × 40 cm) and attached to a transducer connected to an automated rotometer (Basile, Comerio, Italy). The total number of full rotations (360°) performed by the animals in the following 30 minutes were then registered. The test was repeated 2 days later (day 5), and the results from the two tests were averaged.
Animal groups and measurement of CMRglc (days 7 to 8)
Of all the animals that were submitted to the lesioning procedure, 26 rats showed consistent (>30 net rotations in 30 minutes) turning behavior in response to systemic apomorphine and were selected for the [14C]deoxyglucose study. The measurement was carried out on day 7 or 8. Animals were split into two groups: One group (n = 13) was administered apomorphine (1 mg/kg + 1% ascorbate) subcutaneously; the second group (n = 13) received saline. The [14C]deoxyglucose procedure was begun 10 minutes following the drug or saline injection. The procedure was carried out according to the original description (Sokoloff et al., 1977). Briefly, a pulse of [14C]deoxyglucose (100 µCi/kg, specific activity 50 to 55 mCi/mmol; Amersham, U.K.) was injected through the venous catheter. Timed arterial blood samples were then collected and centrifuged immediately. Plasma 14C concentrations were determined by liquid scintillation counting (Beckman, San Ramon, CA, U.S.A.), and plasma glucose concentrations were assayed by means of the Beckman II Glucose Analyzer. Approximately 45 minutes after administration of the tracer, animals were killed by an intravenous administration of a lethal dose of thiopental sodium. Brains were rapidly removed and frozen in isopentane at −40°C and then stored in a freezer at −70°C, until sectioning. Coronal brain sections (20 µm) were cut in a cryostat maintained at −21°C. Sections were picked up on glass coverslips, dried on a hot plate (+60°C), and then autoradiographed on Kodak Min-R X-ray films (Kodak, Rochester, NY, U.S.A.), along with a set of calibrated 14C plastic standards (Amersham, Milan, Italy). Films were exposed for 14 days and developed automatically. Autoradiograms were analyzed by quantitative densitometry using a computerized image-processing system (NIH Imaging, Bethesda, MD, U.S.A.). Optical density measurements for each brain area were made in at least five sections. Tissue 14C concentrations were determined from the optical densities and a calibration curve obtained from densitometric analysis of the autoradiograms of the calibrated standards. Rates of glucose utilization were then calculated from the local tissue 14C concentrations, the time course of the plasma glucose and 14C concentrations, and the appropriate constants according to the operational equation of the method (Sokoloff et al., 1977).
Histologic verification of lesion
Nissl staining was carried out in at least four STN-containing sections per animal of all the rats submitted to the procedure for lesioning STN. The extent of the lesion was assessed by comparing the number of subthalamic neurons of the two hemispheres.
Statistics
For analysis of the rates of glucose utilization, only data from animals with complete lesion of STN were considered. Statistical analysis was carried out using a data analysis software (Statview 4.01; Abacus Concepts, U.S.A.). Interhemispheric differences in glucose utilization, within each group of animals, were analyzed using a Student's t test for paired data. The t test was carried out separately for each brain area considered, for a total of 24 comparisons. A Student's t test for unpaired data was used to compare mean values of glucose utilization of the two groups of animals (apomorphine versus saline).
To control for the overall probability of false rejections among all pairs, we used a significance level of 0.05/24 = 0.002.
The existence of correlation between variables was explored by calculating the Pearson's coefficient of correlation (r).
RESULTS
Histologic verification of lesion
Twenty rats exhibited a virtually complete and circumscribed lesion of the right STN. The Nissl staining showed neuronal loss (>90%), accompanied by glial reaction replacing the large neurons (Fig. 1). In a few cases, a small lesion in the zona incerta ventralis was also found. In the remaining animals, partial lesions of the right STN (ranging from 10 to 60% of the nucleus) were found. Values of glucose utilization of the animals with incomplete lesion were not included in the final tabulation.
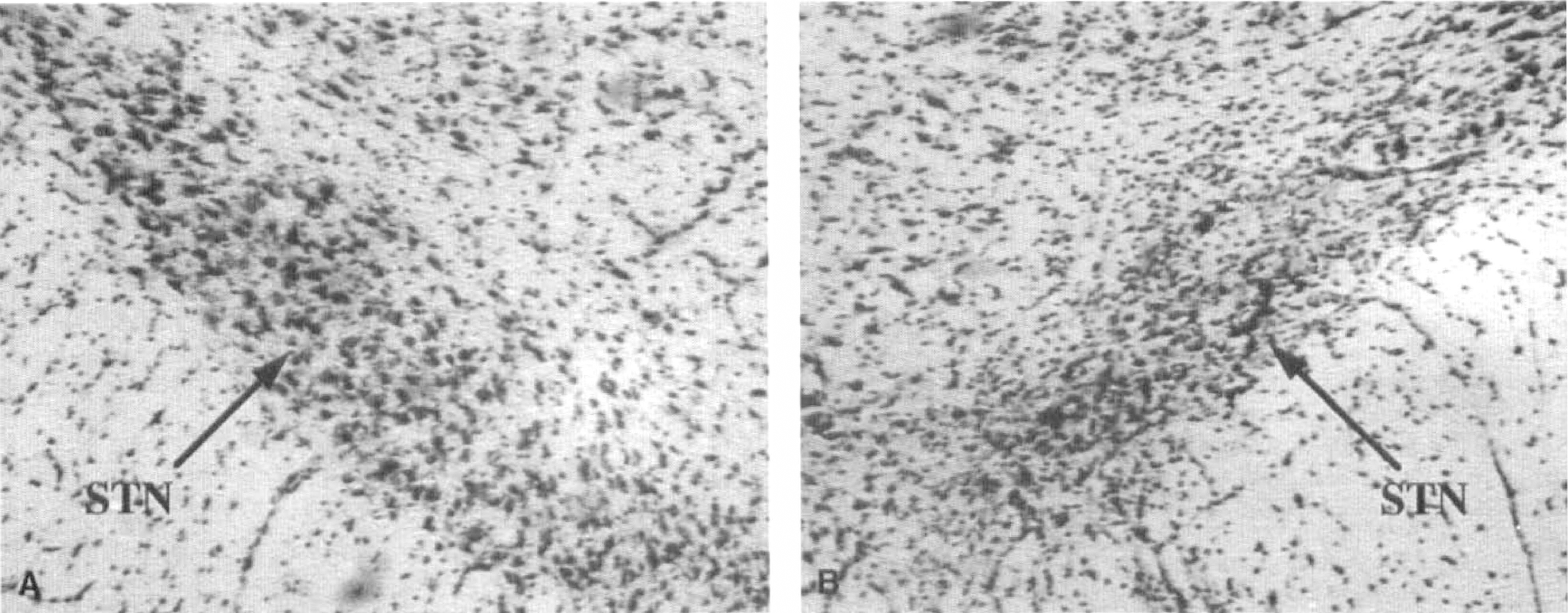
Histologic verification of subthalamic lesion. Nissl staining of a coronal section cut at the level of the subthalamic nucleus (STN). Note, on the lesioned side
Behavior
Upon awakening from anesthesia, rats with complete, unilateral lesion of STN exhibited “wet-dog shakes” and masticatory movements as well as a postural asymmetry of the head, which was turned toward the intact (left) side. As the animals recovered the ability to move, they displayed a weak spontaneous tendency to rotate contralaterally with respect to the lesioned side. This behavior gradually disappeared in 4 to 6 hours, but in most animals it could be elicited 24 hours later by gently pinching the tail.
Systemic administration of apomorphine to rats with complete lesion of the right STN caused rotational behavior ipsilateral to the side of the lesion. This response was enhanced following the second administration of the drug. Mean ± SD values of net ipsilateral turns per 30 minutes following the first (day 3) and the second (day 5) injection of the same dose of apomorphine (1 mg/kg subcutaneously) were 55 ± 39 and 118 ± 62, respectively (n = 20, P < 0.001, paired t test). Animals with partial lesions of STN exhibited a weaker rotational response to apomorphine. In fact, when all animals were considered, a positive correlation between the extent of the subthalamic lesion and the motor response to apomorphine was found (Fig. 2).
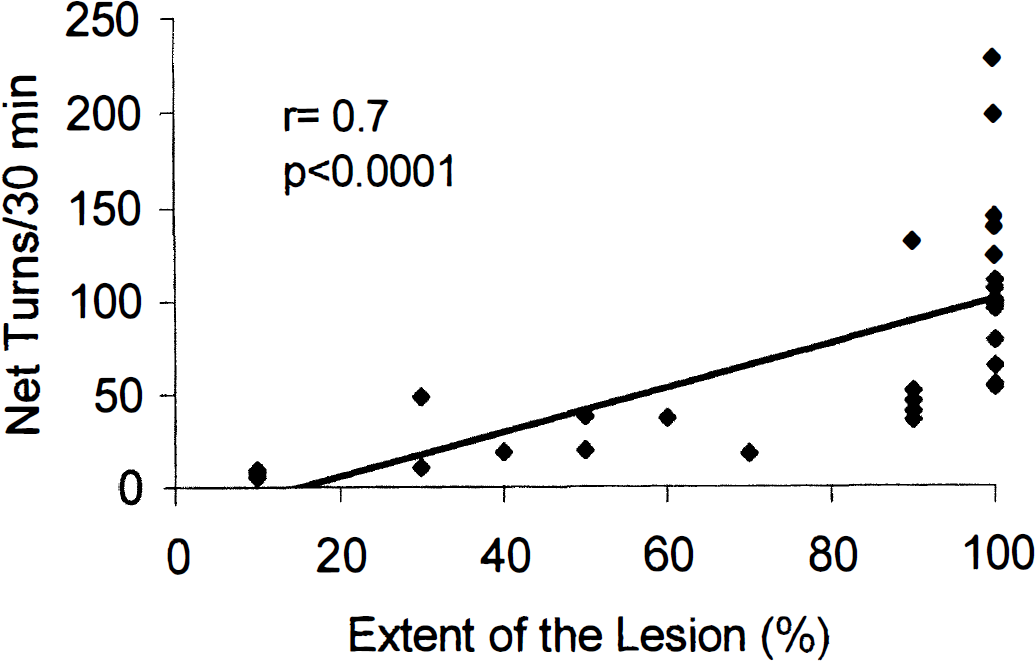
Correlation between the extent of the subthalamic lesion and the number of net ipsilateral rotations. Each dot represents an animal. Only animals with complete (>90%) lesion were considered for analysis of CMRglc data.
Glucose utilization
CMRglc values reported in this study refer to the 20 rats (10 per each treatment group) that showed a virtually complete lesion at the histological verification.
Vehicle Animals with complete lesions of the right STN that received saline did not show statistically significant asymmetries in glucose utilization between the two cerebral hemispheres (Table 1).
CMRglc of rats with unilateral lesion of the subthalamic nucleus after administration of apomorphine or saline
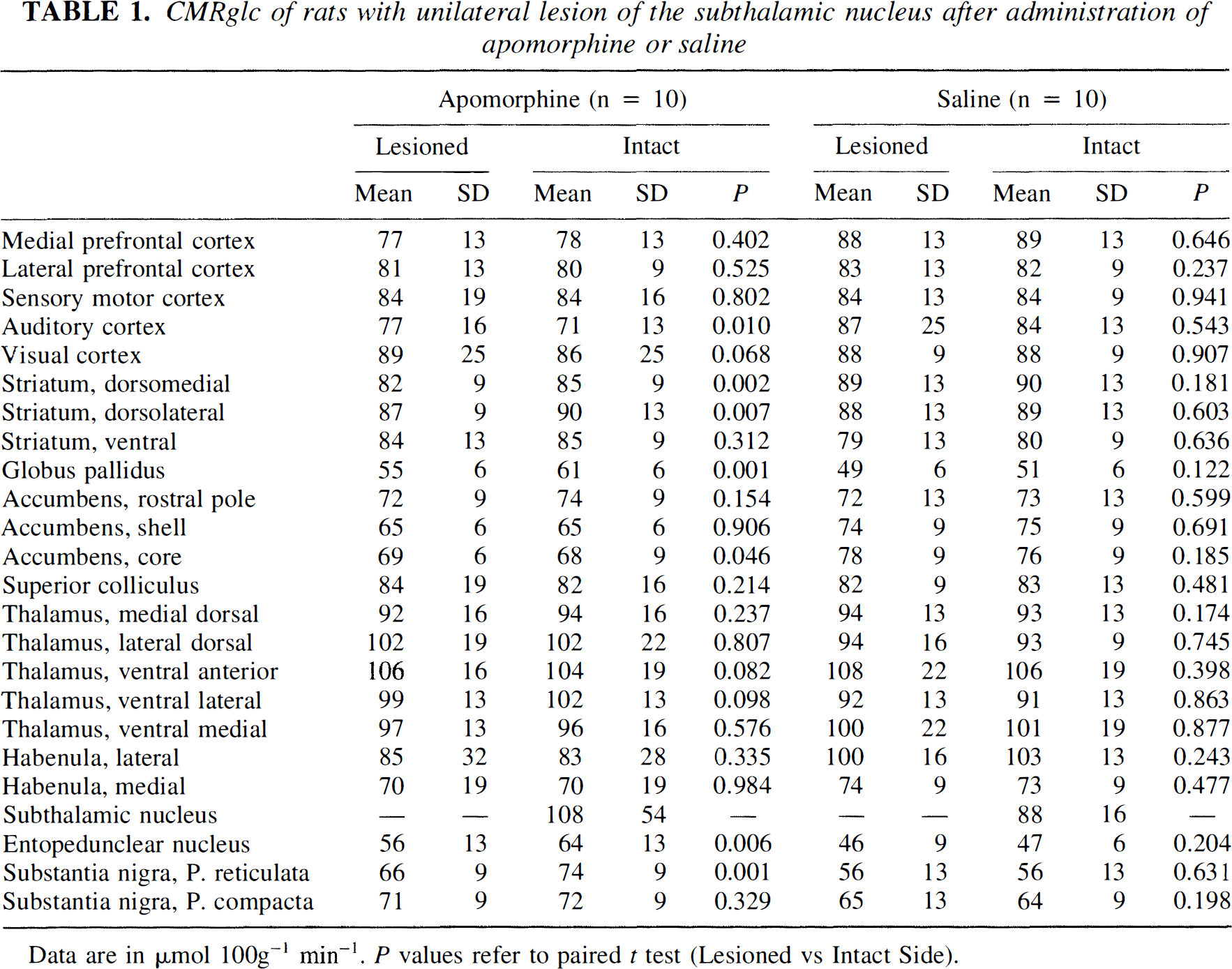
Data are in µmol 100g−1 min−1. P values refer to paired t test (Lesioned vs Intact Side).
Apomorphine Apomorphine administered to animals with lesion of STN caused interhemispheric asymmetries of CMRglc in defined brain areas (Table 1). Significant differences were found in the globus pallidus (Δ% intact side/lesioned side = 11%; P < 0.001) and substantia nigra pars reticulata (Δ% = 12%; P = 0.001). The entopeduncular nucleus showed a 14% side-to-side difference (P = 0.006). In these nuclei, asymmetries were due to increases of glucose utilization on the intact side rather than to decreases on the lesioned side (Table 1). In fact, apomorphine per se increased glucose utilization on the intact side by 36% in the entopeduncular nucleus (P = 0.003) and 32% in the substantia nigra pars reticulata (P = 0.001), with respect to corresponding areas of rats that received vehicle. We also found that mean CMRglc value of STN was 23% higher in the animal group administered apomorphine than in the animal group that received saline. The difference, however, was not significant (P = 0.221) because of the high variability encountered in the values of this brain area in particular. There was also an interhemispheric asymmetry of CMR-glc values of the dorsomedial striatum (P = 0.002).
All the animals that were administered apomorphine exhibited turning behavior ipsilateral to the lesioned side during the measurement of glucose utilization.
DISCUSSION
The results of this study show that systemic administration of apomorphine to rats with unilateral lesion of the STN causes ipsiversive rotational behavior coupled to significant asymmetries of glucose utilization of the substantia nigra pars reticulata and globus pallidus. An effect of apomorphine was also seen in the entopeduncular nucleus, although the side-to-side difference (14%; P = 0.006) just approached the critical level of statistical significance chosen in this study. Comparison of data obtained in animals that received vehicle with those that received apomorphine suggests that the drug-induced asymmetries reflect relative increases of glucose utilization in the intact hemisphere rather than decreases in the lesioned hemisphere.
The substantia nigra pars reticulata, entopeduncular nucleus, and globus pallidus represent the main targets of STN excitatory projections (Kita and Kitai, 1987; Tzagournissakis et al., 1994). Because changes in glucose utilization occur mainly at the neuronal terminal level, the effect of apomorphine on these nuclei may partly result from increased activity of subthalamic neurons. Indeed, various authors have shown that systemic administration of apomorphine or of combinations of D1 and D2 dopamine agonists causes a number of biochemical and functional changes in STN neurons of intact rats. The changes include enhanced glucose utilization (McCulloch et al., 1982; Pontieri et al., 1995), enhanced expression of Fos protein (Ruskin and Marshall, 1995), and increased firing rate (Kreiss et al., 1996). Dopamine receptors are present in the STN (Martes et al., 1985). It is therefore a possibility that apomorphine directly affects STN activity. Alternatively, apomorphine may affect STN by activating striatal neurons. Striatal activation may reach STN via the striato-pallidal-subthalamic pathway or via the striato-subthalamic pathway, whose existence has been recently hypothesized (Kaatz and Albin, 1995). The results of our study therefore indicate that the effects of apomorphine on the substantia nigra pars reticulata, entopeduncular nucleus, and globus pallidus are, at least in part, mediated by activation of the STN. The relevance of dopaminergic transmission at the subthalamic level is also supported by the observation that functional responses of subthalamic neurons to dopamine agonists change dramatically in rats with lesion of the dopaminergic, nigrostriatal pathway (Kreiss et al., 1997).
Apomorphine also caused a slight significant change of CMRglc of the dorsomedial striatum. The result is probably the functional expression of the small contingent of subthalamic projections that impinge into the nucleus (Kita and Kitai, 1987).
Animals with unilateral STN lesion show ipsiversive rotational behavior in response to systemic administration of apomorphine. This behavioral stereotypy, which is less characterized than the well known contralateral rotational response to apomorphine of rats with unilateral nigrostriatal lesion, had been previously described (Kafetzopoulos and Papadopoulos, 1983; Piallat et al., 1996). We observed that the rotational response is higher following the second than the first administration of the drug. This phenomenon, which is consistent with previous observations (Piallat et al., 1996), is reminiscent of the sensitization phenomenon described in animals with unilateral lesion of the nigrostriatal pathway. In animals bearing the nigrostriatal lesion, systemic apomorphine, acting at the level of the denervated and supersensitive striatum, induces an imbalance in the basal ganglia outflow of the two hemispheres, which causes animals to rotate contralaterally to the lesioned side. Repeated stimulation with apomorphine or D1 agonists potentiates the dopamine-mediated behavioral responses, probably as a consequence of progressive augmentation of dopamine receptor sensitivity at the level of the denervated striatum (Morelli et al., 1993). Given the presence of dopamine receptors in the STN and assuming that apomorphine acts directly on this nucleus, we may hypothesize that repeated challenges with dopamine agonists alter the response properties of the subthalamic neurons as well.
In this study we found that lesion of STN per se (without challenge with apomorphine) has no significant consequences on CMRglc. In previous studies we showed that unilateral lesion of STN causes ipsilateral reduction in the activity of mitochondrial enzymes of STN projection nuclei (Blandini and Greenamyre, 1995; Blandini et al., 1995). The apparent discrepancy between the results of the two studies is likely due to the different methods used. Persistent reduction of neuronal activity is necessary to down-regulate mitochondrial enzyme activity. The down-regulation probably reflects a long-lasting adaptive response (Iannuzzelli et al., 1994; Wong-Riley, 1989). Conversely, the [14C]deoxyglucose method visualizes changes of glucose utilization occurring during the measurement. Therefore, it provides an index of ongoing neuronal activity in response to a wide range of stimuli (Sokoloff et al., 1977). Lesion of STN per se does not alter functional activity of the basal ganglia circuitry under resting conditions, but it modifies the response of the same nuclei to dopaminergic stimulation.
In conclusion, the study shows that unilateral lesion of STN alters the metabolic response to apomorphine of defined areas of the basal ganglia, namely, the substantia nigra pars reticulata, the entopeduncular nucleus, and the globus pallidus. The metabolic asymmetries are associated with ipsiversive rotational behavior. These data indicate the involvement of the STN in the activation of the basal ganglia output nuclei induced by systemic dopaminergic stimulation.
